Anticancer Ruthenium Complexes with HDAC Isoform Selectivity
Abstract
1. Introduction
2. Results and Discussion
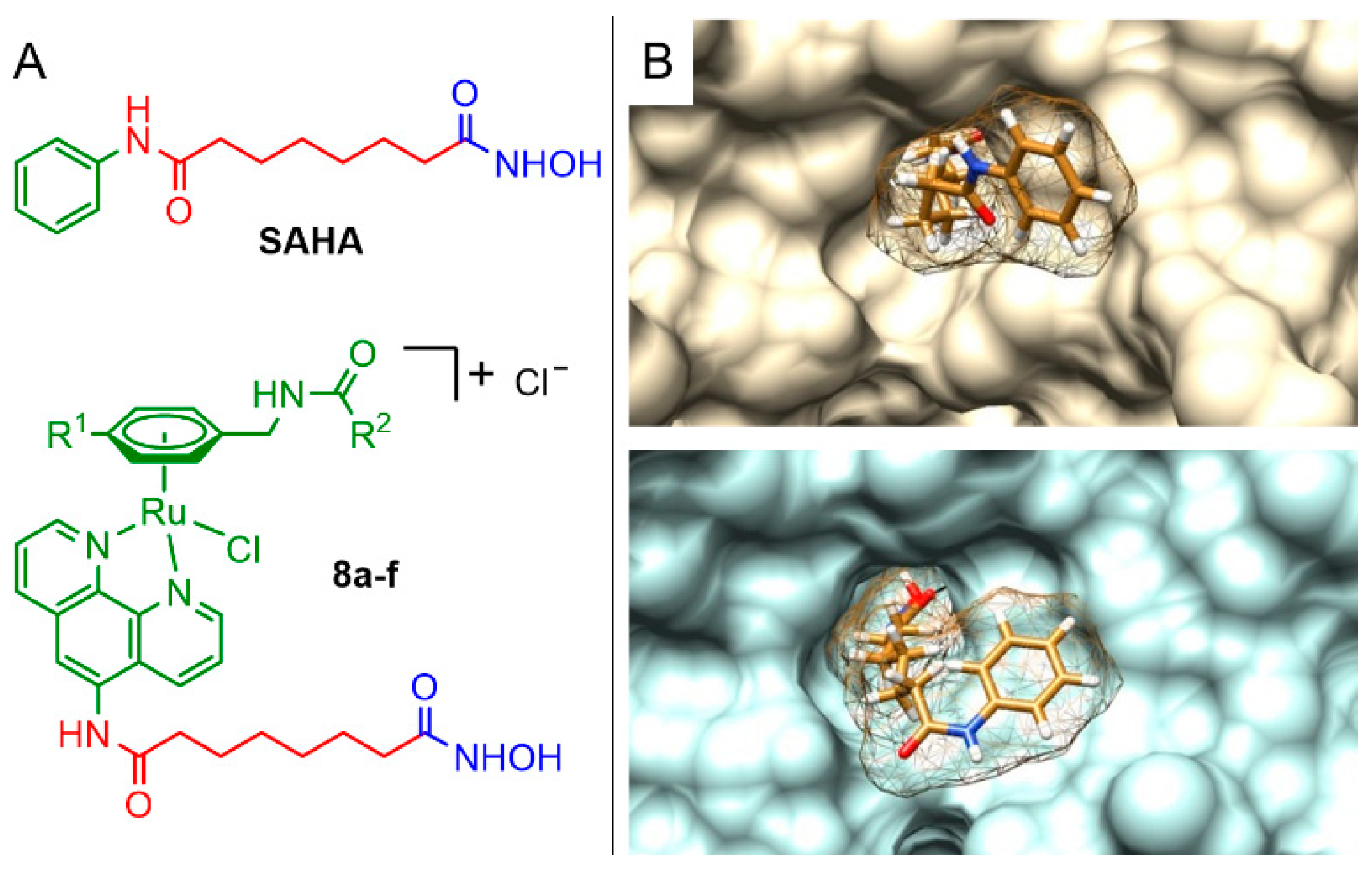
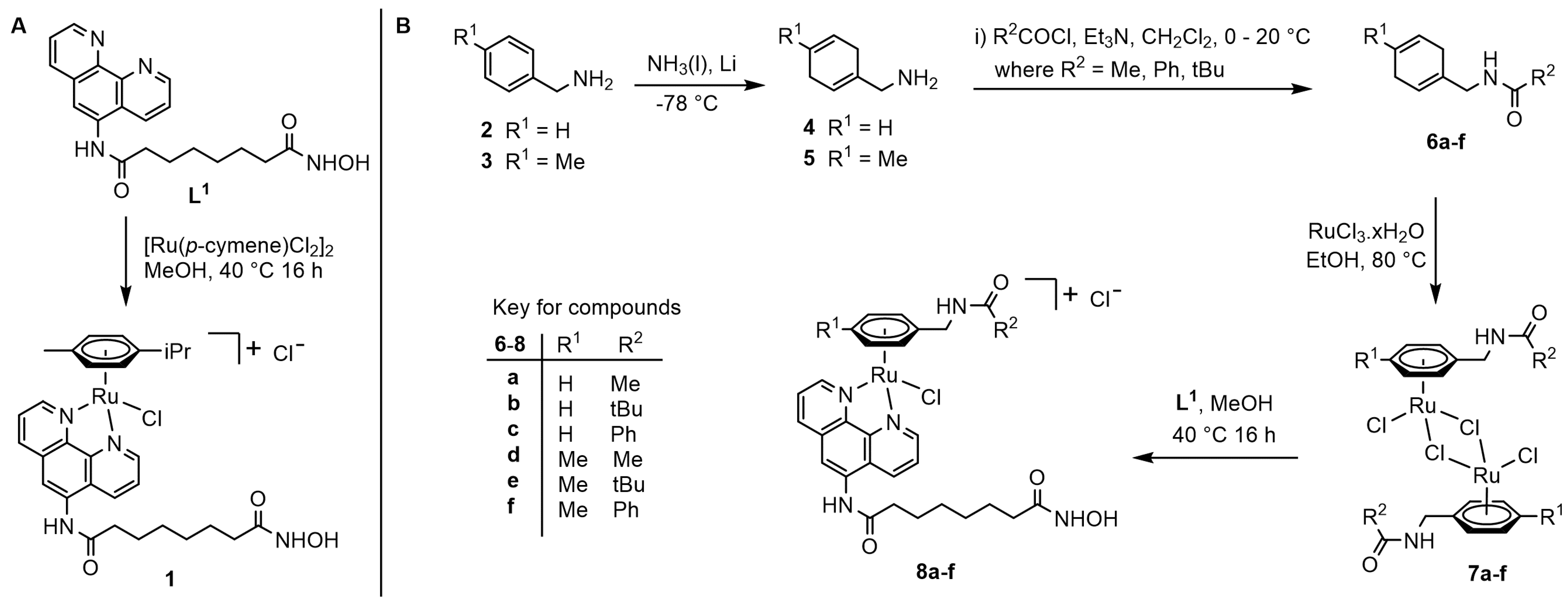
2.1. Synthesis and Characterisation
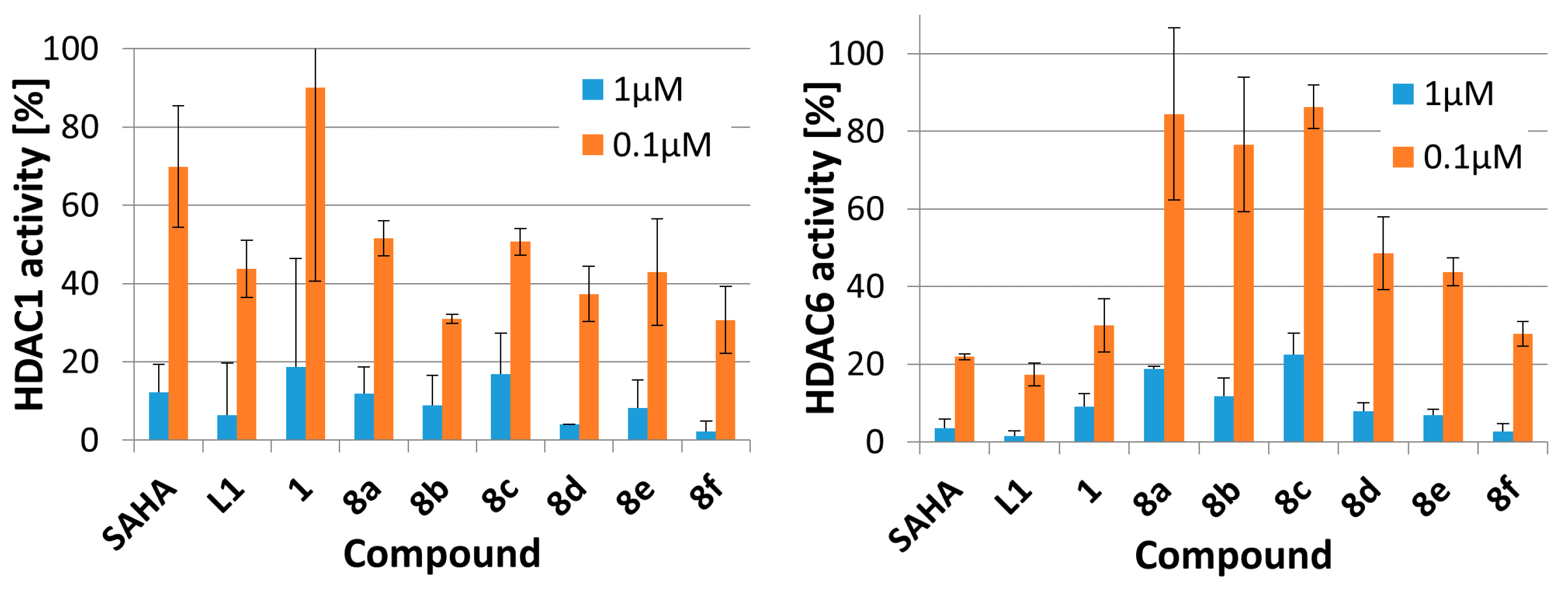
2.2. Enzyme Inhibition Assays
2.3. In Vitro Assays
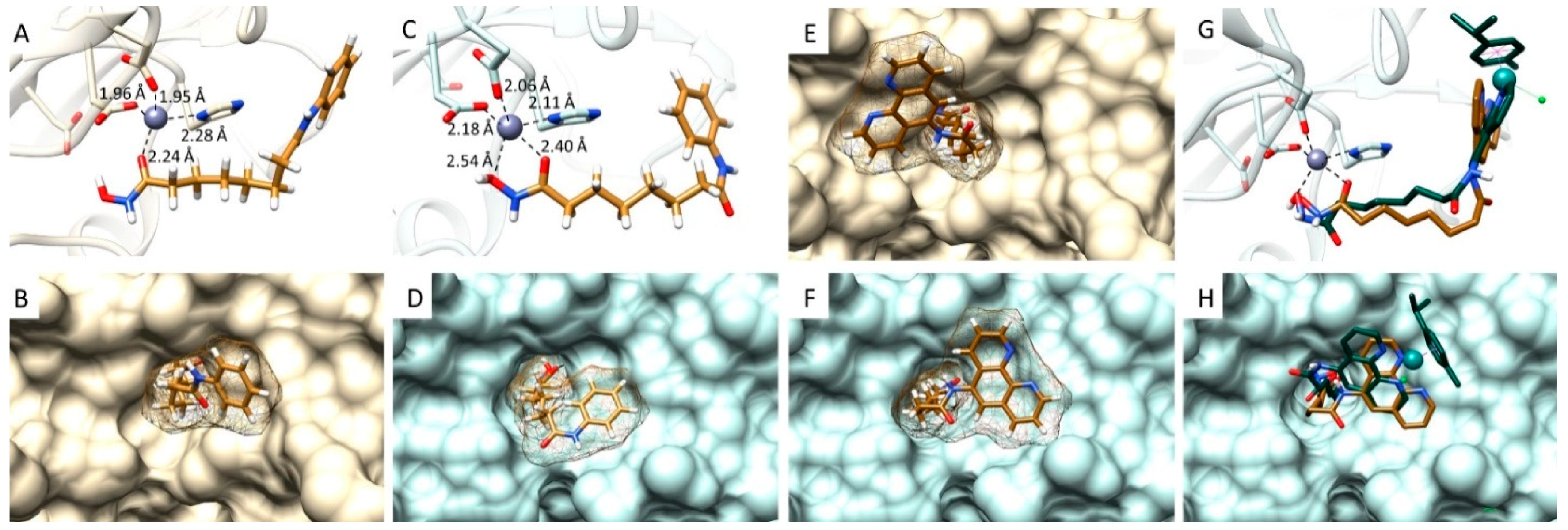
2.4. Computational Docking Study
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Grunstein, M. Histone acetylation in chromatin structure and transcription. Nature 1997, 389, 349–352. [Google Scholar] [CrossRef]
- Kuo, M.H.; Allis, C.D. Roles of histone acetyltransferases and deacetylases in gene regulation. BioEssays 1998, 20, 615–626. [Google Scholar] [CrossRef]
- Davie, J.R.; Hendzel, M.J. Multiple functions of dynamic histone acetylation. J. Cell. Biochem. 1994, 55, 98–105. [Google Scholar] [CrossRef]
- Luo, J.; Su, F.; Chen, D.; Shiloh, A.; Gu, W. Deacetylation of p53 modulates its effect on cell growth and apoptosis. Nature 2000, 408, 377–381. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, W.G. Targeting histone deacetylases for cancer therapy: From molecular mechanisms to clinical implications. Int. J. Biol. Sci. 2014, 10, 757–770. [Google Scholar] [CrossRef]
- Eckschlager, T.; Plch, J.; Stiborova, M.; Hrabeta, J. Histone deacetylase inhibitors as anticancer drugs. Int. J. Mol. Sci. 2017, 18, 1414. [Google Scholar] [CrossRef]
- Butler, L.M.; Zhou, X.; Xu, W.S.; Scher, H.I.; Rifkind, R.A.; Marks, P.A.; Richon, V.M. The histone deacetylase inhibitor SAHA arrests cancer cell growth, up-regulates thioredoxin-binding protein-2, and down-regulates thioredoxin. Proc. Natl. Acad. Sci. USA 2002, 99, 11700–11705. [Google Scholar] [CrossRef]
- Richon, V.M.; Webb, Y.; Merger, R.; Sheppard, T.; Jursic, B.; Ngo, L.; Civoli, F.; Breslow, R.; Rifkind, R.A.; Marks, P.A. Second generation hybrid polar compounds are potent inducers of transformed cell differentiation. Proc. Natl. Acad. Sci. USA 1996, 93, 5705–5708. [Google Scholar] [CrossRef]
- Marks, P.A.; Breslow, R. Dimethyl sulfoxide to vorinostat: Development of this histone deacetylase inhibitor as an anticancer drug. Nat. Biotechnol. 2007, 25, 84–90. [Google Scholar] [CrossRef]
- Olaharski, A.J.; Ji, Z.; Woo, J.Y.; Lim, S.; Hubbard, A.E.; Zhang, L.; Smith, M.T. The histone deacetylase inhibitor trichostatin a has genotoxic effects in human lymphoblasts in vitro. Toxicol. Sci. 2006, 93, 341–347. [Google Scholar] [CrossRef]
- Shen, S.; Kozikowski, A.P. Why Hydroxamates May Not Be the Best Histone Deacetylase Inhibitors - What Some May Have Forgotten or Would Rather Forget? ChemMedChem 2016, 11, 15–21. [Google Scholar] [CrossRef]
- Bieliauskas, A.V.; Pflum, M.K.H. Isoform-selective histone deacetylase inhibitors. Chem. Soc. Rev. 2008, 37, 1402–1413. [Google Scholar] [CrossRef]
- Thaler, F.; Mercurio, C. Towards selective inhibition of histone deacetylase isoforms: What has been achieved, where we are and what will be next. ChemMedChem 2014, 9, 523–536. [Google Scholar] [CrossRef]
- Ganesan, A. Targeting the Zinc-Dependent Histone Deacetylases (HDACs) for Drug Discovery; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–27. [Google Scholar]
- Kilpin, K.J.; Dyson, P.J. Enzyme inhibition by metal complexes: Concepts, strategies and applications. Chem. Sci. 2013, 4, 1410–1419. [Google Scholar] [CrossRef]
- Dörr, M.; Meggers, E. Metal complexes as structural templates for targeting proteins. Curr. Opin. Chem. Biol. 2014, 19, 76–81. [Google Scholar] [CrossRef]
- Feng, L.; Geisselbrecht, Y.; Blanck, S.; Wilbuer, A.; Atilla-Gokcumen, G.E.; Filippakopoulos, P.; Kräling, K.; Celik, M.A.; Harms, K.; Maksimoska, J.; et al. Structurally sophisticated octahedral metal complexes as highly selective protein kinase inhibitors. J. Am. Chem. Soc. 2011, 133, 5976–5986. [Google Scholar] [CrossRef]
- Salmon, A.J.; Williams, M.L.; Innocenti, A.; Vullo, D.; Supuran, C.T.; Poulsen, S.-A. Inhibition of carbonic anhydrase isozymes I, II and IX with benzenesulfonamides containing an organometallic moiety. Bioorg. Med. Chem. Lett. 2007, 17, 5032–5035. [Google Scholar] [CrossRef]
- Can, D.; Spingler, B.; Schmutz, P.; Mendes, F.; Raposinho, P.; Fernandes, C.; Carta, F.; Innocenti, A.; Santos, I.; Supuran, C.T.; et al. [(Cp-R)M(CO)3] (M=Re or 99mTc) Arylsulfonamide, Arylsulfamide, and Arylsulfamate Conjugates for Selective Targeting of Human Carbonic Anhydrase IX. Angew. Chem. Int. Ed. 2012, 51, 3354–3357. [Google Scholar] [CrossRef]
- Parker, L.J.; Italiano, L.C.; Morton, C.J.; Hancock, N.C.; Ascher, D.B.; Aitken, J.B.; Harris, H.H.; Campomanes, P.; Rothlisberger, U.; De Luca, A.; et al. Studies of Glutathione Transferase P1-1 Bound to a Platinum(IV)-Based Anticancer Compound Reveal the Molecular Basis of Its Activation. Chem. Eur. J. 2011, 17, 7806–7816. [Google Scholar] [CrossRef]
- McGivern, T.J.P.; Slator, C.; Kellett, A.; Marmion, C.J. Innovative DNA-Targeted Metallo-prodrug Strategy Combining Histone Deacetylase Inhibition with Oxidative Stress. Mol. Pharm. 2018, 15, 5058–5071. [Google Scholar] [CrossRef]
- Spencer, J.; Amin, J.; Wang, M.; Packham, G.; Alwi, S.S.S.; Tizzard, G.J.; Coles, S.J.; Paranal, R.M.; Bradner, J.E.; Heightman, T.D. Synthesis and biological evaluation of JAHAs: Ferrocene-based histone deacetylase inhibitors. ACS Med. Chem. Lett. 2011, 2, 358–362. [Google Scholar] [CrossRef]
- Librizzi, M.; Longo, A.; Chiarelli, R.; Amin, J.; Spencer, J.; Luparello, C. Cytotoxic Effects of Jay Amin Hydroxamic Acid (JAHA), a Ferrocene-Based Class I Histone Deacetylase Inhibitor, on Triple-Negative MDA-MB231 Breast Cancer Cells. Chem. Res. Toxicol. 2012, 25, 2608–2616. [Google Scholar] [CrossRef]
- Spencer, J.; Amin, J.; Boddiboyena, R.; Packham, G.; Cavell, B.E.; Syed Alwi, S.S.; Paranal, R.M.; Heightman, T.D.; Wang, M.; Marsden, B.; et al. Click JAHAs: Conformationally restricted ferrocene-based histone deacetylase inhibitors. Med. Chem. Comm. 2012, 3, 61–64. [Google Scholar] [CrossRef]
- Griffith, D.; Morgan, M.P.; Marmion, C.J. A novel anti-cancer bifunctional platinum drug candidate with dual DNA binding and histone deacetylase inhibitory activity. Chem. Commun. 2009, 44, 6735–6737. [Google Scholar] [CrossRef]
- Parker, J.P.; Nimir, H.; Griffith, D.M.; Duff, B.; Chubb, A.J.; Brennan, M.P.; Morgan, M.P.; Egan, D.A.; Marmion, C.J. A novel platinum complex of the histone deacetylase inhibitor belinostat: Rational design, development and in vitro cytotoxicity. J. Inorg. Biochem. 2013, 124, 70–77. [Google Scholar] [CrossRef]
- Ye, R.R.; Ke, Z.F.; Tan, C.P.; He, L.; Ji, L.N.; Mao, Z.W. Histone-deacetylase-targeted fluorescent ruthenium(II) polypyridyl complexes as potent anticancer agents. Chem. Eur. J. 2013, 19, 10160–10169. [Google Scholar] [CrossRef]
- Ye, R.R.; Tan, C.P.; Lin, Y.N.; Ji, L.N.; Mao, Z.W. A phosphorescent rhenium(I) histone deacetylase inhibitor: Mitochondrial targeting and paraptosis induction. Chem. Commun. 2015, 51, 8353–8356. [Google Scholar] [CrossRef]
- Ye, R.R.; Tan, C.P.; He, L.; Chen, M.H.; Ji, L.N.; Mao, Z.W. Cyclometalated Ir(III) complexes as targeted theranostic anticancer therapeutics: Combining HDAC inhibition with photodynamic therapy. Chem. Commun. 2014, 50, 10945–10948. [Google Scholar] [CrossRef]
- Ocasio, C.A.; Sansook, S.; Jones, R.; Roberts, J.M.; Scott, T.G.; Tsoureas, N.; Coxhead, P.; Guille, M.; Tizzard, G.J.; Coles, S.J.; et al. Pojamide: An HDAC3-Selective Ferrocene Analogue with Remarkably Enhanced Redox-Triggered Ferrocenium Activity in Cells. Organometallics 2017, 36, 3276–3283. [Google Scholar] [CrossRef]
- Göbel, P.; Ritterbusch, F.; Helms, M.; Bischof, M.; Harms, K.; Jung, M.; Meggers, E. Probing Chiral Recognition of Enzyme Active Sites with Octahedral Iridium(III) Propeller Complexes. Eur. J. Inorg. Chem. 2015, 2015, 1654–1659. [Google Scholar] [CrossRef]
- Cross, J.M.; Blower, T.R.; Gallagher, N.; Gill, J.H.; Rockley, K.L.; Walton, J.W. Anticancer RuII and RhIII Piano-Stool Complexes that are Histone Deacetylase Inhibitors. ChemPlusChem 2016, 81, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Hai, Y.; Christianson, D.W. Histone deacetylase 6 structure and molecular basis of catalysis and inhibition. Nat. Chem. Biol. 2016, 12, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Butler, K.V.; Kalin, J.; Brochier, C.; Vistoli, G.; Langley, B.; Kozikowski, A.P. Rational design and simple chemistry yield a superior, neuroprotective HDAC6 inhibitor, tubastatin A. J. Am. Chem. Soc. 2010, 132, 10842–10846. [Google Scholar] [CrossRef]
- Gasser, G.; Metzler-Nolte, N. The potential of organometallic complexes in medicinal chemistry. Curr. Opin. Chem. Biol. 2012, 16, 84–91. [Google Scholar] [CrossRef]
- Zeng, L.; Gupta, P.; Chen, Y.; Wang, E.; Ji, L.; Chao, H.; Chen, Z.S. The development of anticancer ruthenium(II) complexes: From single molecule compounds to nanomaterials. Chem. Soc. Rev. 2017, 46, 5771–5804. [Google Scholar] [CrossRef]
- Golbaghi, G.; Castonguay, A. Rationally designed ruthenium complexes for breast cancer therapy. Molecules 2020, 25, 265. [Google Scholar] [CrossRef]
- Richard, J.P.; Melikov, K.; Vives, E.; Ramos, C.; Verbeure, B.; Gait, M.J.; Chernomordik, L.V.; Lebleu, B. Cell-penetrating peptides: A reevaluation of the mechanism of cellular uptake. J. Biol. Chem. 2003, 278, 585–590. [Google Scholar] [CrossRef]
- New, E.J.; Congreve, A.; Parker, D. Definition of the uptake mechanism and sub-cellular localisation profile of emissive lanthanide complexes as cellular optical probes. Chem. Sci. 2010, 1, 111–118. [Google Scholar] [CrossRef]
- Miyake, Y.; Keusch, J.J.; Wang, L.; Saito, M.; Hess, D.; Wang, X.; Melancon, B.J.; Helquist, P.; Gut, H.; Matthias, P. Structural insights into HDAC6 tubulin deacetylation and its selective inhibition. Nat. Chem. Biol. 2016, 12, 748–754. [Google Scholar] [CrossRef]
- Porter, N.J.; Mahendran, A.; Breslow, R.; Christianson, D.W. Unusual zinc-binding mode of HDAC6-selective hydroxamate inhibitors. Proc. Natl. Acad. Sci. USA 2017, 114, 13459–13464. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| Compound | HDAC1 IC50 (nM) | HDAC6 IC50 (nM) | Selectivity Factor |
|---|---|---|---|
| 1 | 240 ± 30 | 32 ± 1 | 7.5 |
| 8f | 80 ± 10 | 35 ± 6 | 2.3 |
| L1 | 105 ± 15 | 41 ± 2 | 2.6 |
| SAHA | 30 ± 4 | 12 ± 1 | 2.5 |
| Compound | EC50 (μM) | Cellular Uptake (%) | Compound | EC50 (μM) | Cellular Uptake (%) |
|---|---|---|---|---|---|
| SAHA | 1.5 ± 0.2 | - | 8d | 1.2 ± 0.3 | 13.1 |
| 1 | 1.5 ± 0.4 | 8.0 | 8e | 5.1 ± 1.5 | 6.2 |
| 8a | 32 ± 2 | 0.5 | 8f | 2.1 ± 0.2 | 11.2 |
| 8b | 1.4 ± 0.2 | 18.9 | L1 | 3.0 ± 0.6 | - |
| 8c | 1.7 ± 0.3 | 13.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cross, J.M.; Blower, T.R.; Kingdon, A.D.H.; Pal, R.; Picton, D.M.; Walton, J.W. Anticancer Ruthenium Complexes with HDAC Isoform Selectivity. Molecules 2020, 25, 2383. https://doi.org/10.3390/molecules25102383
Cross JM, Blower TR, Kingdon ADH, Pal R, Picton DM, Walton JW. Anticancer Ruthenium Complexes with HDAC Isoform Selectivity. Molecules. 2020; 25(10):2383. https://doi.org/10.3390/molecules25102383
Chicago/Turabian StyleCross, Jasmine M., Tim R. Blower, Alexander D. H. Kingdon, Robert Pal, David M. Picton, and James W. Walton. 2020. "Anticancer Ruthenium Complexes with HDAC Isoform Selectivity" Molecules 25, no. 10: 2383. https://doi.org/10.3390/molecules25102383
APA StyleCross, J. M., Blower, T. R., Kingdon, A. D. H., Pal, R., Picton, D. M., & Walton, J. W. (2020). Anticancer Ruthenium Complexes with HDAC Isoform Selectivity. Molecules, 25(10), 2383. https://doi.org/10.3390/molecules25102383







