Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral
Abstract
1. Introduction
2. Where Do Zeolites Come From?
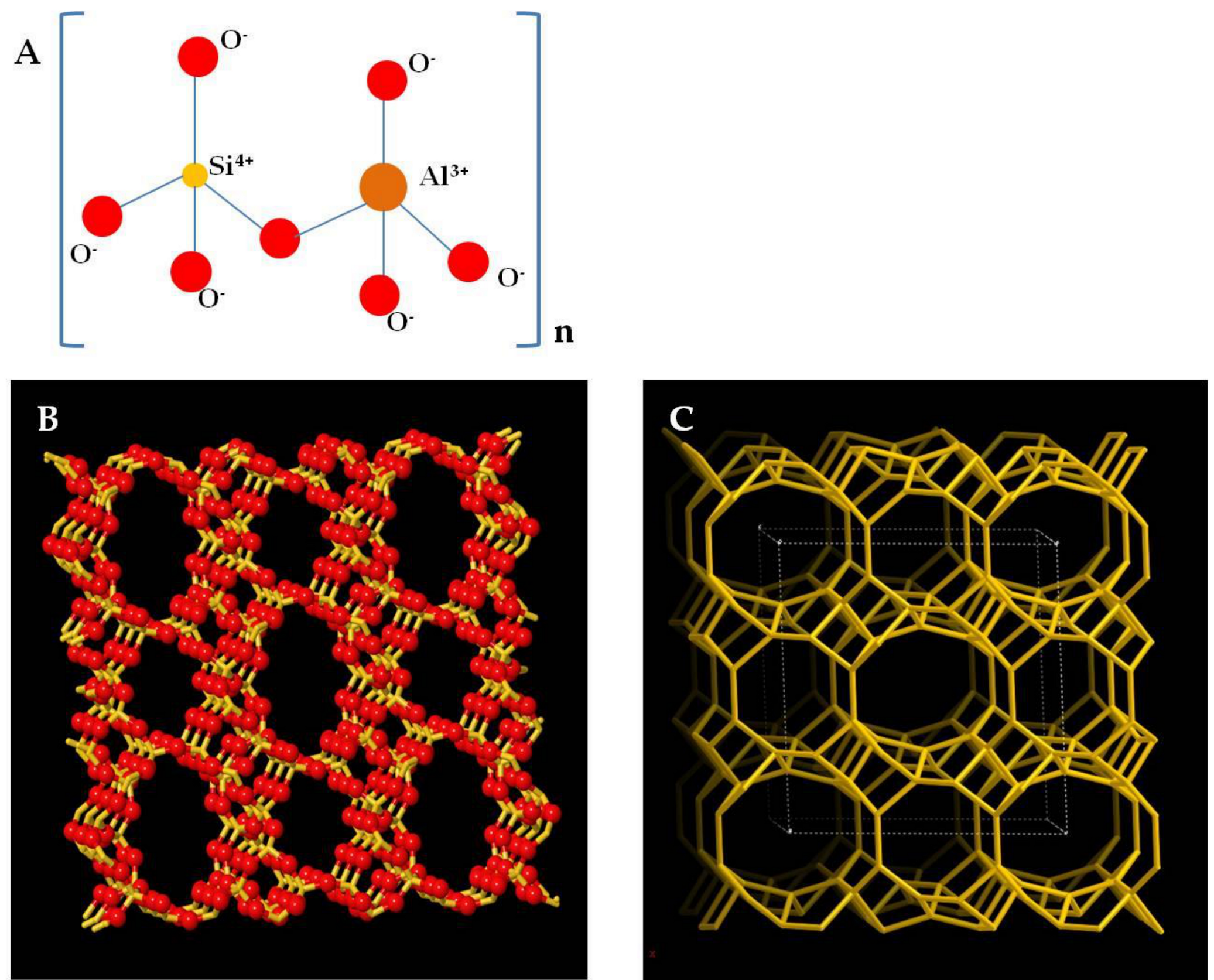
3. Zeolite Clinoptilolite: A Multifaceted Rock
3.1. Micronized Zeolite Clinoptilolite: A Special Treatment
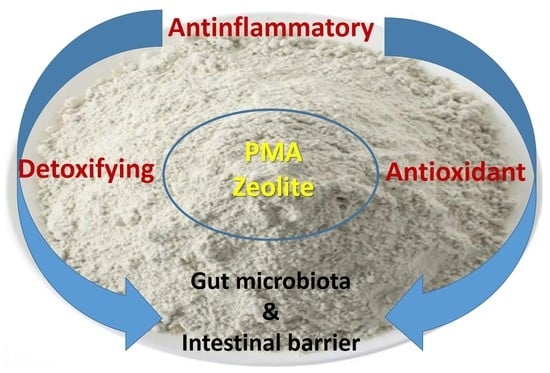
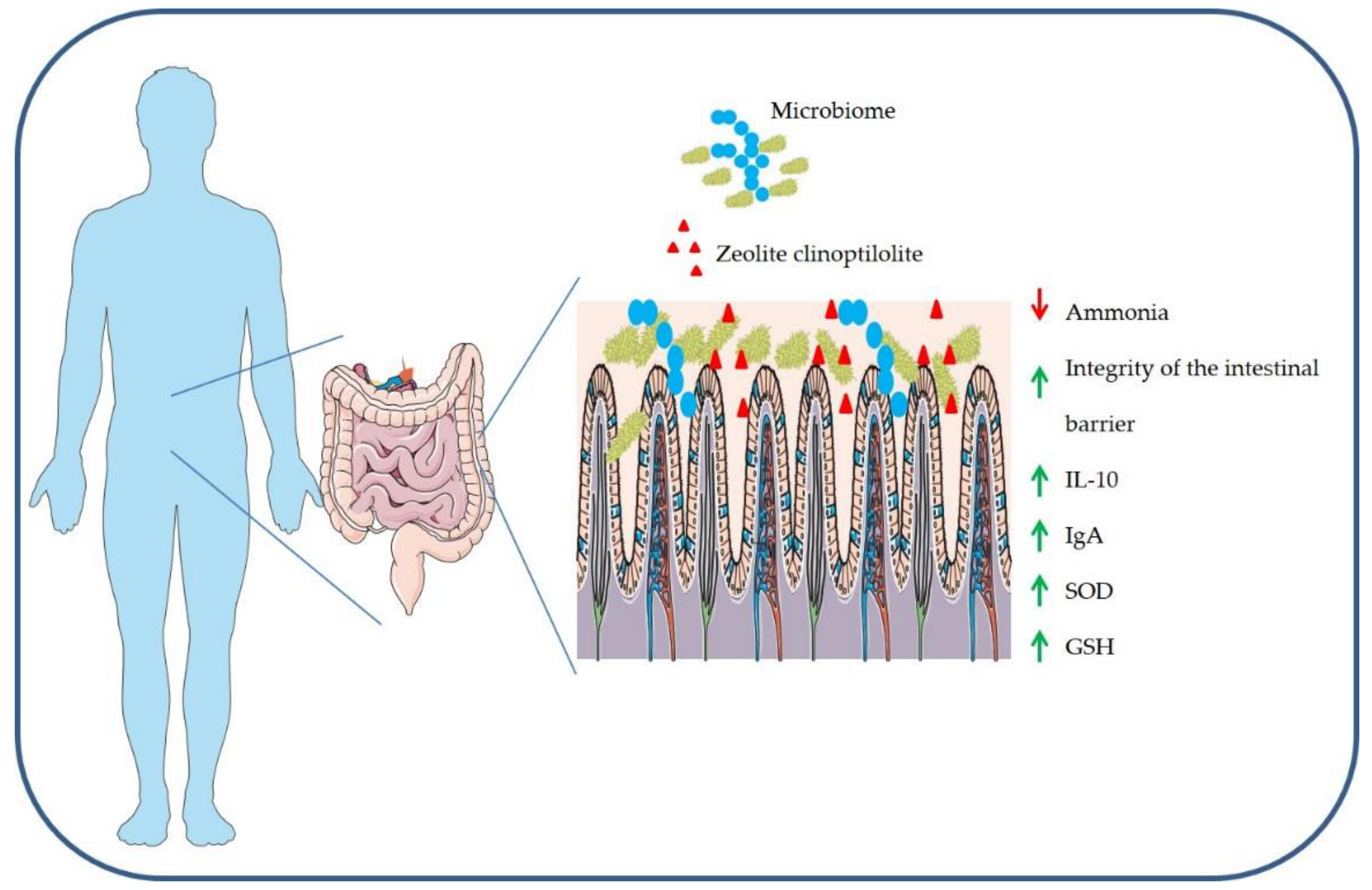
3.2. Detoxifying Effects
3.3. Anti-Inflammatory Effects
3.4. Antioxidant Effects
4. Brain Effects of Zeolite Clinoptilolite
4.1. What is the Action of Zeolites in the Brain?
4.2. Zeolites, Gut-Brain Axis and Alzheimer Disease
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pekov, I.V.; Grigorieva, A.A.; Turchkova, A.G.; Lovskaya, E.V. Natural ion exchange in microporous minerals: Different aspects and implications. Miner. Adv. Mater. I 2008, 7–15. [Google Scholar] [CrossRef]
- Jha, B.; Singh, D.N. Basics of Zeolites. In Fly Ash Zeolites; Springer: Singapore, 2016; Volume 78, pp. 5–31. [Google Scholar]
- Margeta, K.; Zabukovec, N.; Siljeg, M.; Farkas, A. Natural Zeolites in Water Treatment—How Effective is Their Use. In Water Treatment; Elshorbagy, W., Chowdhury, R., Eds.; IntechOpen: London, UK, 2013; Volume 5, pp. 81–112. [Google Scholar]
- Kraljević Pavelić, S.; Micek, V.; Filošević, A.; Gumbarević, D.; Žurga, P.; Bulog, A.; Orct, T.; Yamamoto, Y.; Preočanin, T.; Plavec, J.; et al. Novel, oxygenated clinoptilolite material efficiently removes aluminium from aluminium chloride-intoxicated rats in vivo. Microporous Mesoporous Mater. 2017, 249, 146–156. [Google Scholar] [CrossRef]
- Kraljević Pavelić, S.; Simović Medica, J.; Gumbarević, D.; Filošević, A.; Pržulj, N.; Pavelić, K. Critical Review on Zeolite Clinoptilolite Safety and Medical Applications in vivo. Front. Pharmacol. 2018, 9, 1350. [Google Scholar] [CrossRef]
- Lamprecht, M.; Bogner, S.; Steinbauer, K.; Schuetz, B.; Greilberger, J.F.; Leber, B.; Wagner, B.; Zinser, E.; Petek, T.; Wallner-Liebmann, S.; et al. Effects of zeolite supplementation on parameters of intestinal barrier integrity, inflammation, redoxbiology and performance in aerobically trained subjects. J. Int. Soc. Sports Nutr. 2015, 12, 40. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.J.; Wang, L.C.; Zhou, Y.M.; Zhang, J.F.; Wang, T. Effects of clinoptilolite and modified clinoptilolite on the growth performance, intestinal microflora, and gut parameters of broilers. Poult. Sci. 2013, 92, 684–692. [Google Scholar] [CrossRef]
- Kaelberer, M.M.; Buchanan, K.L.; Klein, M.E.; Barth, B.B.; Montoya, M.M.; Shen, X.; Bohórquez, D.V. A gut-brain neural circuit for nutrient sensory transduction. Science 2018, 361, eaat5236. [Google Scholar] [CrossRef]
- Iijima, A. Geology of Natural Zeolites and Zeolitic Rocks. Pure Appl. Chem. 1980, 52, 2115–2130. [Google Scholar] [CrossRef]
- Mumpton, F.A.; Ormsby, W.C. Morphology of Zeolites in Sedimentary-Rocks by Scanning Electron-Microscopy. Clays Clay Miner. 1976, 24, 1–23. [Google Scholar] [CrossRef]
- De’gennaro, M.; Cappelletti, P.; Langella, A.; Perrotta, A.; Scarpati, C. Genesis of zeolites in the Neapolitan Yellow Tuff: Geological, volcanological and mineralogical evidence. Contrib. Miner. Petrol. 2000, 139, 17–35. [Google Scholar] [CrossRef]
- Gottardi, G. The Genesis of Zeolites. Eur. J. Miner. 1989, 1, 479–487. [Google Scholar] [CrossRef]
- Li, Y.; Pera-Titus, M.; Xiong, G.; Yang, W.; Landrivon, E.; Miachon, S.; Dalmon, J.A. Nanocomposite MFI-alumina membranes via pore-plugging synthesis: Genesis of the zeolite material. J. Membr. Sci 2008, 325, 973–981. [Google Scholar] [CrossRef]
- Machiels, L.; Garces, D.; Snellings, R.; Vilema, W.; Morante, F.; Paredes, C.; Elsen, J. Zeolite occurrence and genesis in the Late-Cretaceous Cayo arc of Coastal Ecuador: Evidence for zeolite formation in cooling marine pyroclastic flow deposits. Appl. Clay Sci. 2014, 87, 108–119. [Google Scholar] [CrossRef]
- Woodall, L.J.; Anderson, P.A.; Armstrong, A.R.; Edwards, P.P. Dissolving alkali metals in zeolites: Genesis of the perfect cluster crystal. J. Chem. Soc. Dalton Trans. 1996, 719–727. [Google Scholar] [CrossRef]
- Marantos, I.; Christidis, G.E.; Ulmanu, M. Zeolite Formation and Deposits. In Natural Zeolites Handbook; Inglezakis, V.J., Zorpas, A.A., Eds.; Bentham Science Publishers: Dubai, United Arab Emirates, 2012; Chapter 2.1; pp. 28–51. [Google Scholar]
- Weisenberger, T.; Bucher, K. Zeolites in fissures of granites and gneisses of the Central Alps. J. Metamorph. Geol. 2010, 28, 825–847. [Google Scholar] [CrossRef]
- Kovič, P.; Krošl-Kuščer, N. Hydrothermal Zeolite Occurrence from the Smrekovec Mt. Area, Slovenia, Yugoslavia. Stud. Surf. Sci. Catal. 1986, 28, 87–92. [Google Scholar]
- Monzón, J.D.; Pereyra, A.M.; Conconi, M.S.; Basaldella, E.I. Phase transformations during the zeolitization of fly ashes. J. Environ. Chem. Eng. 2017, 5, 1548–1553. [Google Scholar] [CrossRef]
- Hall, A. Zeolitization of volcaniclastic sediments; the role of temperature and pH. J. Sediment. Res. 1998, 68, 739–745. [Google Scholar] [CrossRef]
- Ghiara, M.R. Occurrence of Clinoptilolite and Mordenite in Tertiary Calc-Alkaline Pyroclastites from Sardinia (Italy). Clays Clay Miner. 1999, 47, 319–328. [Google Scholar] [CrossRef]
- Laurino, C.; Palmieri, B. Zeolite: “the Magic Stone”; Main Nutritional, Environmental, Experimental and Clinical Fields of Application. Nutr. Hosp. 2015, 32, 573–581. [Google Scholar]
- Ivanova, I.I.; Knyazeva, E.E. Micro-mesoporous materials obtained by zeolite recrystallization: Synthesis, characterization and catalytic applications. Chem. Soc. Rev. 2013, 42, 3671–3688. [Google Scholar] [CrossRef] [PubMed]
- Ambrozova, P.; Kynicky, J.; Urubek, T.; Nguyen, V. Synthesis and Modification of Clinoptilolite. Molecules 2017, 22, 1107. [Google Scholar] [CrossRef] [PubMed]
- Benning, L.G.; Wilkin, R.T.; Barnes, H.L. Solubility and stability of zeolites in aqueous solution: II. Calcic clinoptilolite and mordenite. Am. Miner. 2000, 85, 495–508. [Google Scholar] [CrossRef]
- Ham, K.; Kim, B.S.; Choi, K.-Y. Enhanced ammonium removal efficiency by ion exchange process of synthetic zeolite after Na+ and heat pretreatment. Water Sci. Technol. 2018, 788, 1417–1425. [Google Scholar] [CrossRef] [PubMed]
- Gorimbo, J.; Taenzana, B.; Muleja, A.A.; Kuvarega, A.T.; Jewell, L.L. Adsorption of cadmium, nickel and lead ions: Equilibrium, kinetic and selectivity studies on modified clinoptilolites from the USA and RSA. Environ. Sci. Pollut. Res. 2018, 25, 30962–30978. [Google Scholar] [CrossRef] [PubMed]
- Giordani, M.; Mattioli, M.; Ballirano, P.; Pacella, A.; Cenni, M.; Boscardin, M.; Valentini, L. Geological occurrence, mineralogical characterization, and risk assessment of potentially carcinogenic erionite in Italy. J. Toxicol. Environ. Health Part B 2017, 20, 81–103. [Google Scholar] [CrossRef]
- Beaucham, C.; King, B.; Feldmann, K.; Harper, M.; Dozier, A. Assessing occupational erionite and respirable crystalline silica exposure among outdoor workers in Wyoming, South Dakota, and Montana. J. Occup. Environ. Hyg. 2018, 15, 455–465. [Google Scholar] [CrossRef]
- Demirer, E.; Ghattas, C.F.; Radwan, M.O.; Elamin, E.M. Clinical and Prognostic Features of Erionite-Induced Malignant Mesothelioma. Yonsei Med. J. 2015, 56, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Metintas, S.; Ak, G.; Metintas, M. A review of the cohorts with environmental and occupational mineral fiber exposure. Arch. Environ. Occup. Health 2018, 74, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Cangiotti, M.; Salucci, S.; Battistelli, M.; Falcieri, E.; Mattioli, M.; Giordani, M.; Ottaviani, M.F. EPR, TEM and cell viability study of asbestiform zeolite fibers in cell media. Colloids Surf. B Biointerfaces 2018, 161, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Bacakova, L.; Vandrovcova, M.; Kopova, I.; Jirka, I. Applications of zeolites in biotechnology and medicine—A review. Biomater. Sci. 2018, 6, 974–989. [Google Scholar] [CrossRef] [PubMed]
- Katic, M.; Bosnjak, B.; Gall-Troselj, K.; Dikic, I.; Pavelic, K. A clinoptilolite effect on cell media and the consequent effects on tumor cells in vitro. Front. Biosci. J. Virtual Libr. 2006, 11, 1722–1732. [Google Scholar] [CrossRef]
- Dogliotti, G.; Malavazos, A.E.; Giacometti, S.; Solimene, U.; Fanelli, M.; Corsi, M.M.; Dozio, E. Natural zeolites chabazite/phillipsite/analcime increase blood levels of antioxidant enzymes. J. Clin. Biochem. Nutr. 2012, 50, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Superchi, P.; Saleri, R.; Ossiprandi, M.C.; Riccardi, E.; Passaglia, E.; Cavalli, V.; Beretti, V.; Sabbioni, A. Natural zeolite (chabazite/phillipsite) dietary supplementation influences faecal microbiota and oxidant status of working dogs. Ital. J. Anim. Sci. 2016, 16, 115–121. [Google Scholar] [CrossRef]
- Ferreira, L.; Fonseca, A.M.; Botelho, G.; Aguiar, C.A.; Neves, I.C. Antimicrobial activity of faujasite zeolites doped with silver. Microporous Mesoporous Mater. 2012, 160, 126–132. [Google Scholar] [CrossRef]
- Attanoos, R.L.; Churg, A.; Galateau-Salle, F.; Gibbs, A.R.; Roggli, V.L. Malignant Mesothelioma and Its Non-Asbestos Causes. Arch. Pathol. Lab. Med. 2018, 142, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, K.O.; Nur Aainaa, H.; Haruna Ahmed, O.; Ab Majid, N.M. Effects of clinoptilolite zeolite on phosphorus dynamics and yield of Zea Mays L. cultivated on an acid soil. PLoS ONE 2018, 13, e0204401. [Google Scholar]
- Sun, J.; Wu, Y.; Zhou, Y.; Lu, C.; Ahmad, H.; Zhang, H.; He, J.; Zhang, L.; Wang, T. Influence of Butyrate Loaded Clinoptilolite Dietary Supplementation on Growth Performance, Development of Intestine and Antioxidant Capacity in Broiler Chickens. PLoS ONE 2016, 11, e0154410. [Google Scholar]
- Nizet, S.; Muñoz, E.; Fiebich, B.L.; Abuja, P.M.; Kashofer, K.; Zatloukal, K.; Tangermann, S.; Kenner, L.; Tschegg, C.; Nagl, D.; et al. Clinoptilolite in Dextran Sulphate Sodium-Induced Murine Colitis: Efficacy and Safety of a Microparticulate Preparation. Inflamm. Bowel Dis. 2018, 24, 54–66. [Google Scholar] [CrossRef]
- Hossein Nia, B.; Khorram, S.; Rezazadeh, H.; Safaiyan, A.; Tarighat-Esfanjani, A. The Effects of Natural Clinoptilolite and Nano-Sized Clinoptilolite Supplementation on Glucose Levels and Oxidative Stress in Rats with Type 1 Diabetes. Can. J. Diabetes 2018, 42, 31–35. [Google Scholar] [CrossRef]
- Potgieter, W.; Samuels, S.; Snyman, J.R. Potentiated clinoptilolite: Artificially enhanced aluminosilicate reduces symptoms associated with endoscopically negative gastroesophageal reflux disease and nonsteroidal anti-inflammatory drug induced gastritis. Clin. Exp. Gastroenterol. 2014, 7, 215–220. [Google Scholar]
- Zarkovic, N.; Zarkovic, K.; Kralj, M.; Borovic, S.; Sabolovic, S.; Blazi, M.P.; Cipak, A.; Pavelic, K. Anticancer and antioxidative effects of micronized zeolite clinoptilolite. Anticancer. Res. 2003, 23, 1589–1595. [Google Scholar] [PubMed]
- Montinaro, M.; Uberti, D.; Maccarinelli, G.; Bonini, S.A.; Ferrari-Toninelli, G.; Memo, M. Dietary zeolite supplementation reduces oxidative damage and plaque generation in the brain of an Alzheimer’s disease mouse model. Life Sci. 2013, 92, 903–910. [Google Scholar] [CrossRef]
- Andrić, L.; Trumić, M.; Trumić, M.; Nikolić, V. Micronization of zeolite in vibration mill. Reciklaza I Odrziv. Razvoj 2018, 11, 63–71. [Google Scholar] [CrossRef]
- Beltcheva, M.; Metcheva, R.; Popov, N.; Teodorova, S.E.; Heredia-Rojas, J.A.; Rodríguez-de la Fuente, A.O.; Rodríguez-Flores, L.E.; Topashka-Ancheva, M. Modified Natural Clinoptilolite Detoxifies Small Mammal’s Organism Loaded with Lead I. Lead Disposition and Kinetic Model for Lead Bioaccumulation. Biol. Trace Elem. Res. 2011, 147, 180–188. [Google Scholar] [CrossRef]
- Nikpey, A.; Kazemian, H.; Safari-Varyani, A.; Rezaie, M.; Sirati-Sabet, M. Protective Effect of Microporous Natural Clinoptilolite on Lead-Induced Learning and Memory Impairment in Rats. Health Scope 2013, 2, 52–57. [Google Scholar] [CrossRef]
- Papaioannou, D.; Katsoulos, P.D.; Panousis, N.; Karatzias, H. The role of natural and synthetic zeolites as feed additives on the prevention and/or the treatment of certain farm animal diseases: A review. Microporous Mesoporous Mater. 2005, 84, 161–170. [Google Scholar] [CrossRef]
- Kanyılmaz, M.; Tekelioğlu, N.; Sevgili, H.; Uysal, R.; Aksoy, A. Effects of dietary zeolite (clinoptilolite) levels on growth performance, feed utilization and waste excretions by gilthead sea bream juveniles (Sparus aurata). Anim. Feed Sci. Technol. 2015, 200, 66–75. [Google Scholar] [CrossRef]
- Burmańczuk, A.; Markiewicz, W.; Burmańczuk, A.; Kowalski, C.; Roliński, Z.; Burmańczuk, N. Possibile use of natural zeolites in animal production and environment protection. J. Elem. 2015, 4, 803–811. [Google Scholar] [CrossRef]
- Liu, R.; Lal, R. Nanoenhanced Materials for Reclamation of Mine Lands and Other Degraded Soils: A Review. J. Nanotechnol. 2012, 2012, 1–18. [Google Scholar] [CrossRef]
- Katsoulos, P.D.; Karatzia, M.A.; Boscos, C.; Wolf, P.; Karatzias, H. In-field evaluation of clinoptilolite feeding efficacy on the reduction of milk aflatoxin M1 concentration in dairy cattle. J. Anim. Sci. Technol. 2016, 58, 24. [Google Scholar] [CrossRef][Green Version]
- Katsoulos, P.D.; Karatzia, M.A.; Polizopoulou, Z.; Florou-Paneri, P.; Karatzias, H. Effects of prolonged consumption of water with elevated nitrate levels on certain metabolic parameters of dairy cattle and use of clinoptilolite for their amelioration. Environ. Sci. Pollut. Res. 2015, 22, 9119–9126. [Google Scholar] [CrossRef] [PubMed]
- Goff, J.P. Invited review: Mineral absorption mechanisms, mineral interactions that affect acid–base and antioxidant status, and diet considerations to improve mineral status. J. Dairy Sci. 2018, 101, 2763–2813. [Google Scholar] [CrossRef]
- Schneider, A.F.; Zimmermann, O.F.; Gewehr, C.E. Zeolites in poultry and swine production. Cienc. Rural 2017, 47, e20160344. [Google Scholar] [CrossRef]
- Nahm, K.H. Efficient feed nutrient utilization to reduce pollutants in poultry and swine manure. Crit. Rev. Environ. Sci. Technol. 2002, 32, 1–16. [Google Scholar] [CrossRef]
- Fotidis, I.A.; Kougias, P.G.; Zaganas, I.D.; Kotsopoulos, T.A.; Martzopoulos, G.G. Inoculum and zeolite synergistic effect on anaerobic digestion of poultry manure. Environ. Technol. 2014, 35, 1219–1225. [Google Scholar] [CrossRef]
- Walker, V. Ammonia Metabolism and Hyperammonemic Disorders. Adv. Clin. Chem. 2014, 67, 73–150. [Google Scholar] [PubMed]
- Genuis, S.J.; Birkholz, D.; Ralitsch, M.; Thibault, N. Human detoxification of perfluorinated compounds. Public Health 2010, 124, 367–375. [Google Scholar] [CrossRef]
- Bischoff, S.C.; Barbara, G.; Buurman, W.; Ockhuizen, T.; Schulzke, J.-D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability—A new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef]
- Colella, C. A critical reconsideration of biomedical and veterinary applications ofnatural zeolites. Clay Miner. 2018, 46, 295–309. [Google Scholar] [CrossRef]
- McGhee, J.R.; Fujihashi, K. Inside the Mucosal Immune System. PLoS Biol. 2012, 10, e1001397. [Google Scholar] [CrossRef]
- Ivkovic, S.; Deutsch, U.; Silberbach, A.; Walraph, E.; Mannel, M. Dietary supplementation with the tribomechanically activated zeolite clinoptilolite in immunodeficiency: Effects on the immune system. Adv. Ther. 2004, 21, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Bermudez-Brito, M.; Plaza-Díaz, J.; Muñoz-Quezada, S.; Gómez-Llorente, C.; Gil, A. Probiotic Mechanisms of Action. Ann. Nutr. Metab. 2012, 61, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Hemarajata, P.; Versalovic, J. Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Ther. Adv. Gastroenterol. 2012, 6, 39–51. [Google Scholar] [CrossRef]
- Sabbioni, A.; Ferrario, C.; Milani, C.; Mancabelli, L.; Riccardi, E.; di Ianni, F.; Beretti, V.; Superchi, P.; Ossiprandi, M.C. Modulation of the Bifidobacterial Communities of the Dog Microbiota by Zeolite. Front. Microbiol. 2016, 7, 1491. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Grandhi, R.; Patterson, T.; Nicholson, S. A Review of Traumatic Brain Injury and the Gut Microbiome: Insights into Novel Mechanisms of Secondary Brain Injury and Promising Targets for Neuroprotection. Brain Sci. 2018, 8, 113. [Google Scholar] [CrossRef] [PubMed]
- Belkaid, Y.; Hand, T.W. Role of the Microbiota in Immunity and Inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Rotfogel, Z.; Hillman, D.; Popugailo, A.; Arad, G.; Supper, E.; Osman, F.; Kaempfer, R. Superantigens hyperinduce inflammatory cytokines by enhancing the B7-2/CD28 costimulatory receptor interaction. Proc. Natl. Acad. Sci. USA 2016, 113, E6437–E6446. [Google Scholar] [CrossRef]
- Actor, J.K. Role of Major Histocompatibility Complex in the Immune Response. In Elsevier’s Integrated Review Immunology and Microbiology, 2nd ed.; Actor, J.K., Ed.; Elsevier: Philadelphia, PA, USA, 2012; Volume 5, pp. 33–41. [Google Scholar]
- He, L.; He, T.; Farrar, S.; Ji, L.; Liu, T.; Ma, X. Antioxidants Maintain Cellular Redox Homeostasis by Elimination of Reactive Oxygen Species. Cell. Physiol. Biochem. 2017, 44, 532–553. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell. Longev. 2017, 2017, 1–13. [Google Scholar] [CrossRef]
- Araújo, R.F.F.D.; Martins, D.B.G.; Borba, M.A.C.S.M. Oxidative Stress and Disease. In The Transcription Factor Nrf2; Morales-Gonzales, J.A., Ed.; IntechOpen: London, UK, 2016; Volume 10, pp. 185–199. [Google Scholar]
- Saribeyoglu, K.; Aytac, E.; Pekmezci, S.; Saygili, S.; Uzun, H.; Ozbay, G.; Aydin, S.; Seymen, H.O. Effects of clinoptilolite treatment on oxidative stress after partial hepatectomy in rats. Asian J. Surg. 2011, 34, 153–157. [Google Scholar] [CrossRef]
- Cai, Q.; Turner, B.D.; Sheng, D.; Sloan, S. The kinetics of fluoride sorption by zeolite: Effects of cadmium, barium and manganese. J. Contam. Hydrol. 2015, 177, 136–147. [Google Scholar] [CrossRef] [PubMed]
- Bintaş, E.; Bozkurt, M.; Küçükyılmaz, K.; Konak, R.; Çınar, M.; Akşit, H.; Seyrek, K.; Çatlı, A.U. Efficacy of Supplemental Natural Zeolite in Broiler Chickens Subjected to Dietary Calcium Deficiency. Ital. J. Anim. Sci. 2014, 13, 275–283. [Google Scholar] [CrossRef]
- Rana, C.; Mediha, H.A.; Naceur, M.H. Effects of the Incorporation of Zeolite on Zoo technical Parameters of Chicken. J. Anim. Sci. Livest. Prod. 2018, 2, 011. [Google Scholar] [CrossRef]
- Shariatmadari, F. The application of zeolite in poultry production. World’s Poult. Sci. J. 2008, 64, 76–84. [Google Scholar] [CrossRef]
- Abraha, I.; Rimland, J.M.; Trotta, F.M.; Dell’Aquila, G.; Cruz-Jentoft, A.; Petrovic, M.; Gudmundsson, A.; Soiza, R.; O’Mahony, D.; Guaita, A.; et al. Systematic review of systematic reviews of non-pharmacological interventions to treat behavioural disturbances in older patients with dementia. The SENATOR-OnTop series. BMJ Open 2017, 7, e012759. [Google Scholar] [CrossRef] [PubMed]
- Hecht, K. Biological rhythms and sleep rhythms. Aerosp. Environ. Med. 2007, 41, 59–64. [Google Scholar]
- Mück-Šeler, D.; Pivac, N. The effect of natural clinoptilolite on the serotonergic receptors in the brain of mice with mammary carcinoma. Life Sci. 2003, 73, 2059–2069. [Google Scholar] [CrossRef]
- Golokhvast, K.; Sergievich, A.; Grigoriev, N. Geophagy (rock eating), experimental stress and cognitive idiosyncrasy. Asian Pac. J. Trop. Biomed. 2014, 4, 362–366. [Google Scholar] [CrossRef]
- Delavarian, M.; Hassanvand, A.; Gharibzadeh, S. Increasing performance in children with ADHD by trapping lead with a nano-zeolite. J. Neuropsychiatry Clin. Neurosci. 2013, 25, E23. [Google Scholar] [CrossRef]
- Delavarian, M.; Hassanvand, A.; Gharibzadeh, S. Using natural zeolite as a transporter of dopamine. J. Neuropsychiatry Clin. Neurosci. 2013, 25, E21. [Google Scholar] [CrossRef]
- Hassanvand, A.; Gharibzadeh, S. Is it possible to treat some brain diseases by drug-substituted zeolites? J. Neuropsychiatry Clin. Neurosci. 2013, 25, E04. [Google Scholar] [CrossRef]
- Hassanvand, A.; Hajihassani, M.; Abdi, M.; Gharibzadeh, S. Drug delivery using nano-pore zeolites and ultrasound. J. Neuropsychiatry Clin. Neurosci. 2013, 25, E20. [Google Scholar] [CrossRef] [PubMed]
- Apostolova, L.G. Alzheimer Disease. Contin. Lifelong Learn. Neurol. 2016, 22, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Kumar, A.; Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Memo, M.; Mastinu, A. Cannabimimetic plants: Are they new cannabinoidergic modulators? Planta 2019. [Google Scholar] [CrossRef] [PubMed]
- Mastinu, A.; Premoli, M.; Ferrari-Toninelli, G.; Tambaro, S.; Maccarinelli, G.; Memo, M.; Bonini, S.A. Cannabinoids in health and disease: Pharmacological potential in metabolic syndrome and neuroinflammation. Horm. Mol. Biol. Clin. Investig. 2018, 36, 20180013. [Google Scholar] [CrossRef]
- Premoli, M.; Aria, F.; Bonini, S.A.; Maccarinelli, G.; Gianoncelli, A.; Pina, S.D.; Tambaro, S.; Memo, M.; Mastinu, A. Cannabidiol: Recent advances and new insights for neuropsychiatric disorders treatment. Life Sci. 2019, 224, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Moos, W.H.; Faller, D.V.; Harpp, D.N.; Kanara, I.; Pernokas, J.; Powers, W.R.; Steliou, K. Microbiota and Neurological Disorders: A Gut Feeling. Biores. Open Access 2016, 5, 137–145. [Google Scholar] [CrossRef]
- Giau, V.; Wu, S.; Jamerlan, A.; An, S.; Kim, S.; Hulme, J. Gut Microbiota and Their Neuroinflammatory Implications in Alzheimer’s Disease. Nutrients 2018, 10, 1765. [Google Scholar] [CrossRef]
- Griffiths, J.A.; Mazmanian, S.K. Emerging evidence linking the gut microbiome to neurologic disorders. Genome Med. 2018, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Z.; Wang, Y.; Li, F.; Jia, J.; Song, X.; Qin, S.; Wang, R.; Jin, F.; Kitazato, K.; et al. The Gut-Microglia Connection: Implications for Central Nervous System Diseases. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Alkasir, R.; Li, J.; Li, X.; Jin, M.; Zhu, B. Human gut microbiota: The links with dementia development. Protein Cell 2016, 8, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wang, T.; Jin, F. Alzheimer’s disease and gut microbiota. Sci. China Life Sci. 2016, 59, 1006–1023. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, F.; Clerici, M.; Ratto, D.; Occhinegro, A.; Licito, A.; Romeo, M.; Iorio, C.; Rossi, P. The Gut-Brain Axis in Alzheimer’s Disease and Omega-3. A Critical Overview of Clinical Trials. Nutrients 2018, 10, 1267. [Google Scholar] [CrossRef] [PubMed]
- Berti, V.; Walters, M.; Sterling, J.; Quinn, C.G.; Logue, M.; Andrews, R.; Matthews, D.C.; Osorio, R.S.; Pupi, A.; Vallabhajosula, S.; et al. Mediterranean diet and 3-year Alzheimer brain biomarker changes in middle-aged adults. Neurology 2018, 90, e1789–e1798. [Google Scholar] [CrossRef] [PubMed]

| Zeolite Mineral Species | Formula | Effects | Ref. |
|---|---|---|---|
| Analcime | NaAlSi2O6·H2O | Antioxidant | [35] |
| Phillipsite | (Ca,Na2,K2)3Al6Si10O32·12H2O | Antioxidant and gut microbiota | [35,36] |
| Faujasite | (Na2,Ca,Mg)3.5[Al7Si17O48]·32(H2O) | Antimicrobial | [37] |
| Erionite | (Na2,K2,Ca)2[Al4Si14O36]·15H2O | Carcinogenic | [28,29,30,38] |
| Clinoptilolite | (Na,K,Ca)2-3Al3(Al,Si)2Si13O36·12H2O | Environmental purification, intestinal detoxifier, antioxidant, anti-inflammatory, antitumor | [39,40,41,42,43] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mastinu, A.; Kumar, A.; Maccarinelli, G.; Bonini, S.A.; Premoli, M.; Aria, F.; Gianoncelli, A.; Memo, M. Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral. Molecules 2019, 24, 1517. https://doi.org/10.3390/molecules24081517
Mastinu A, Kumar A, Maccarinelli G, Bonini SA, Premoli M, Aria F, Gianoncelli A, Memo M. Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral. Molecules. 2019; 24(8):1517. https://doi.org/10.3390/molecules24081517
Chicago/Turabian StyleMastinu, Andrea, Amit Kumar, Giuseppina Maccarinelli, Sara Anna Bonini, Marika Premoli, Francesca Aria, Alessandra Gianoncelli, and Maurizio Memo. 2019. "Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral" Molecules 24, no. 8: 1517. https://doi.org/10.3390/molecules24081517
APA StyleMastinu, A., Kumar, A., Maccarinelli, G., Bonini, S. A., Premoli, M., Aria, F., Gianoncelli, A., & Memo, M. (2019). Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral. Molecules, 24(8), 1517. https://doi.org/10.3390/molecules24081517