The Asymmetric A3(Aldehyde–Alkyne–Amine) Coupling: Highly Enantioselective Access to Propargylamines
Abstract
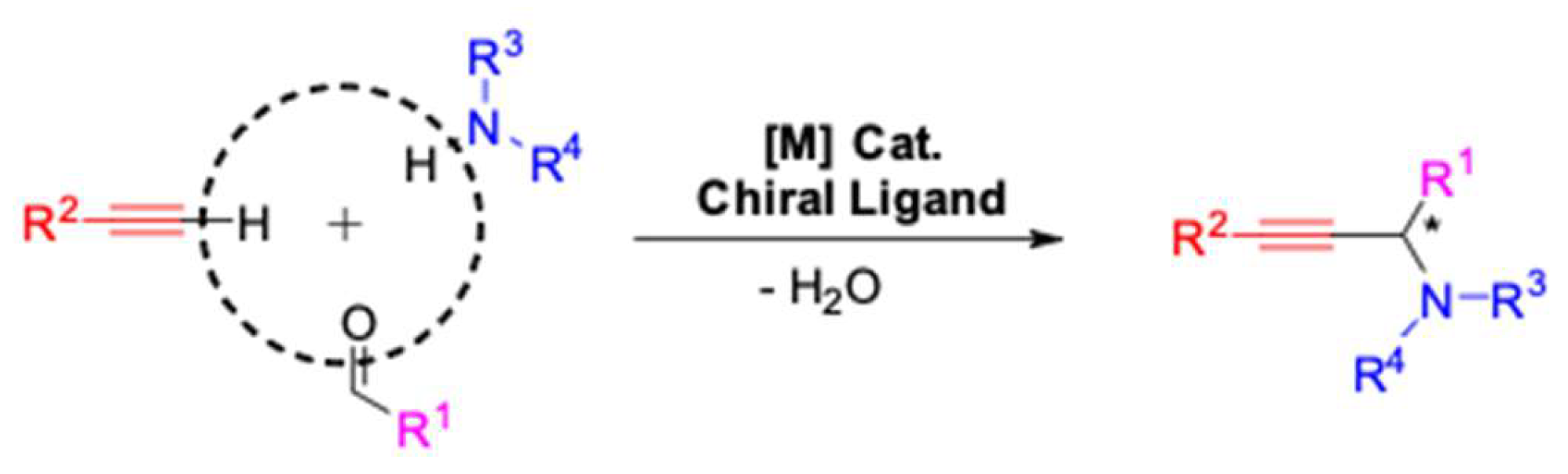
1. Introduction
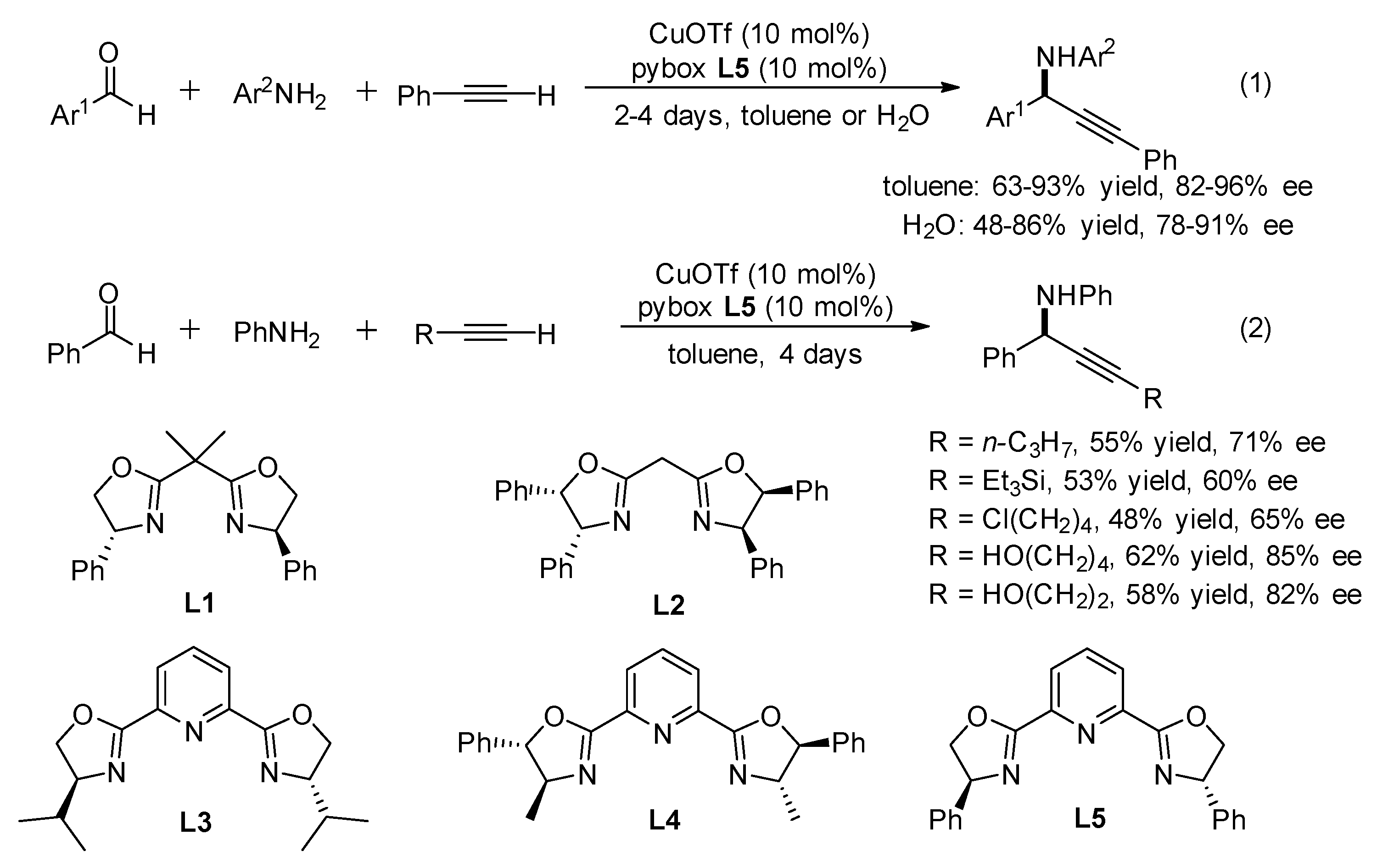
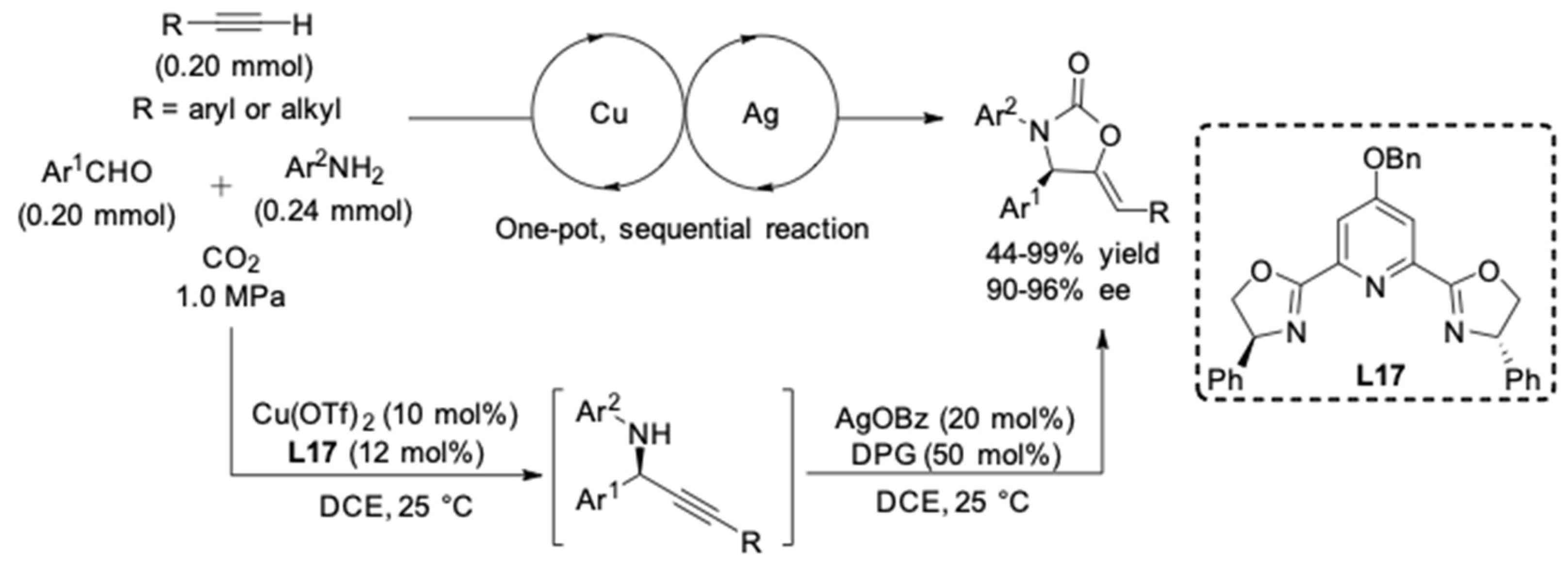
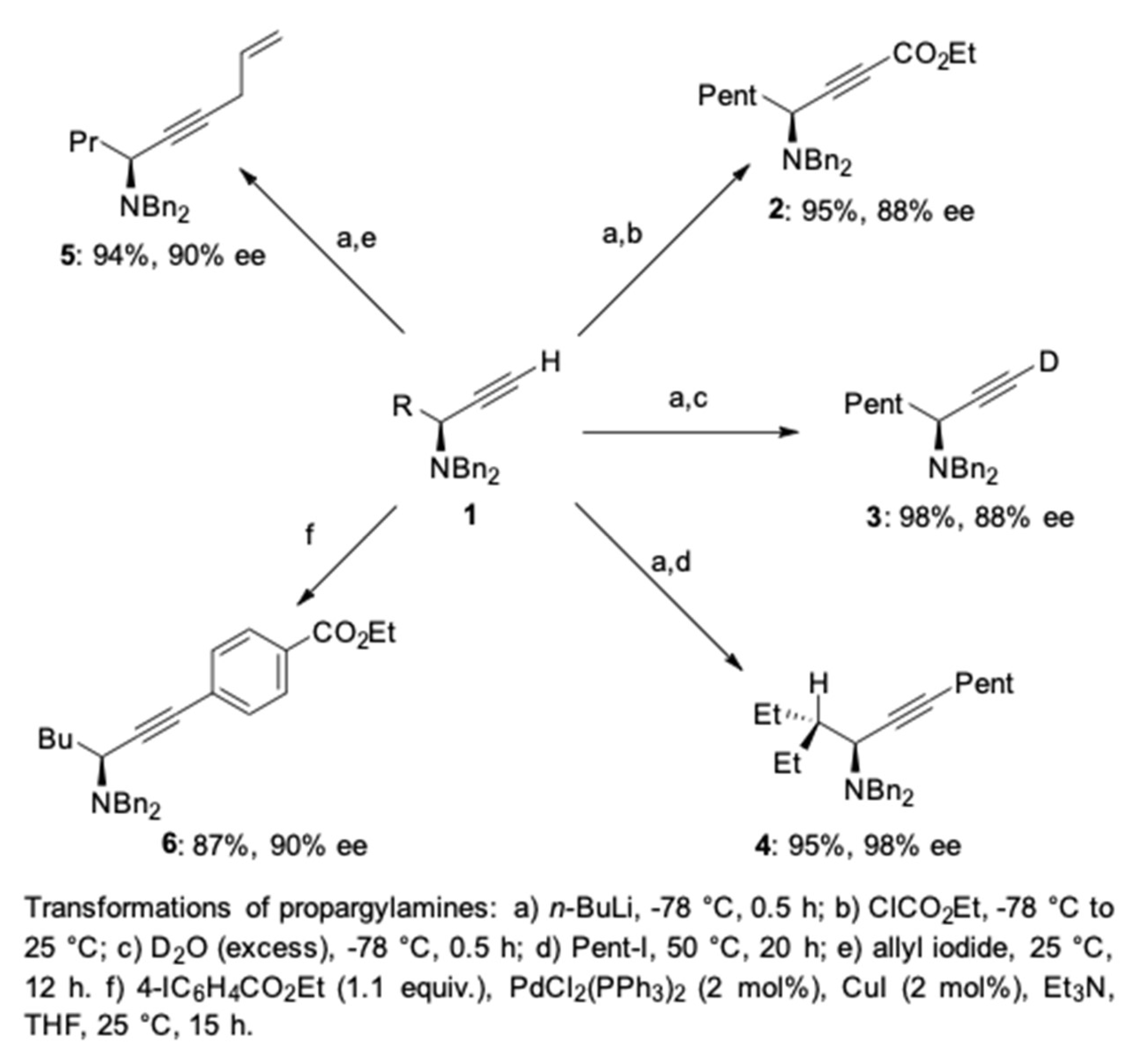
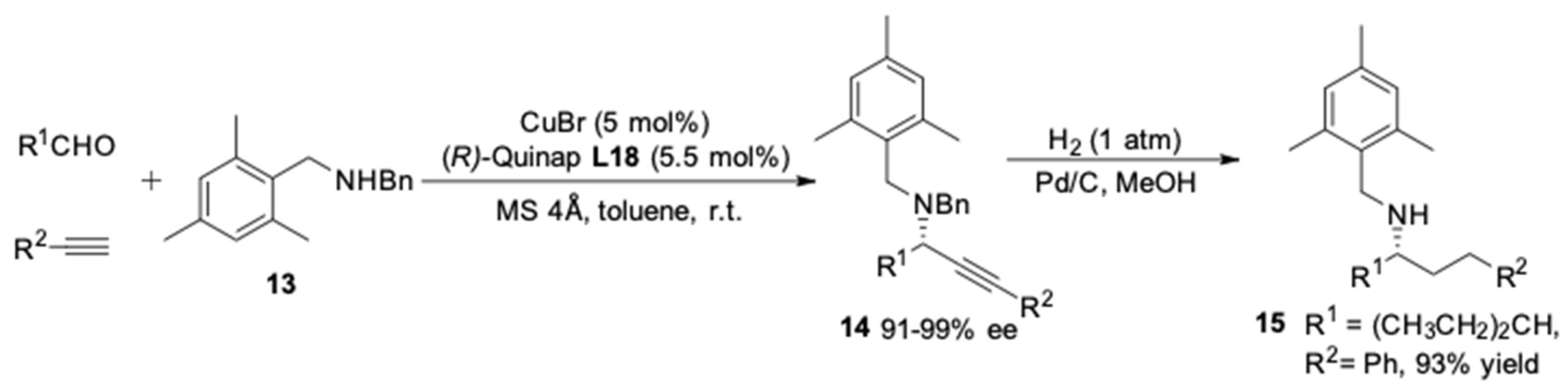
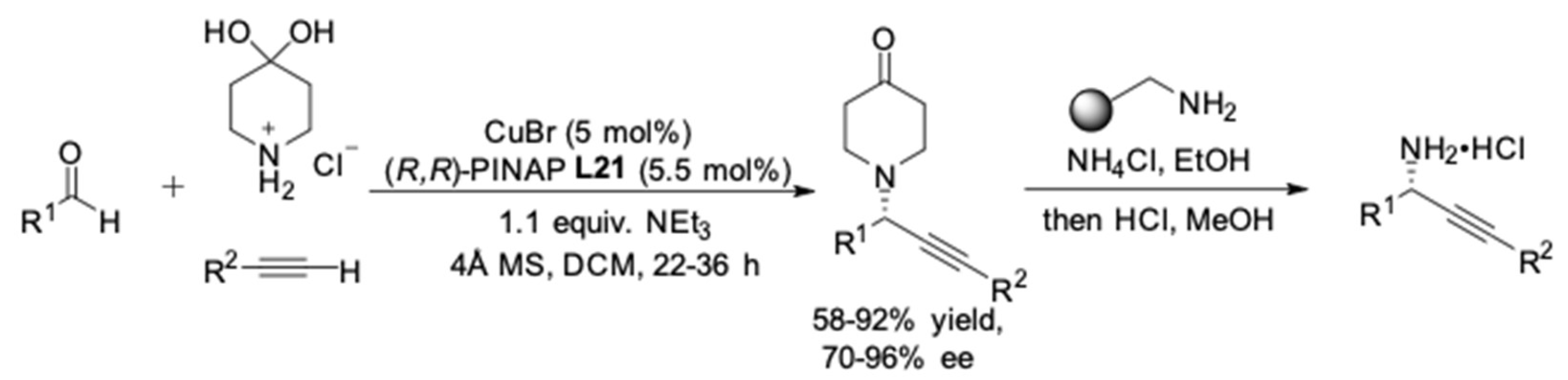
2. AA3 –Coupling Derived from Primary Amine
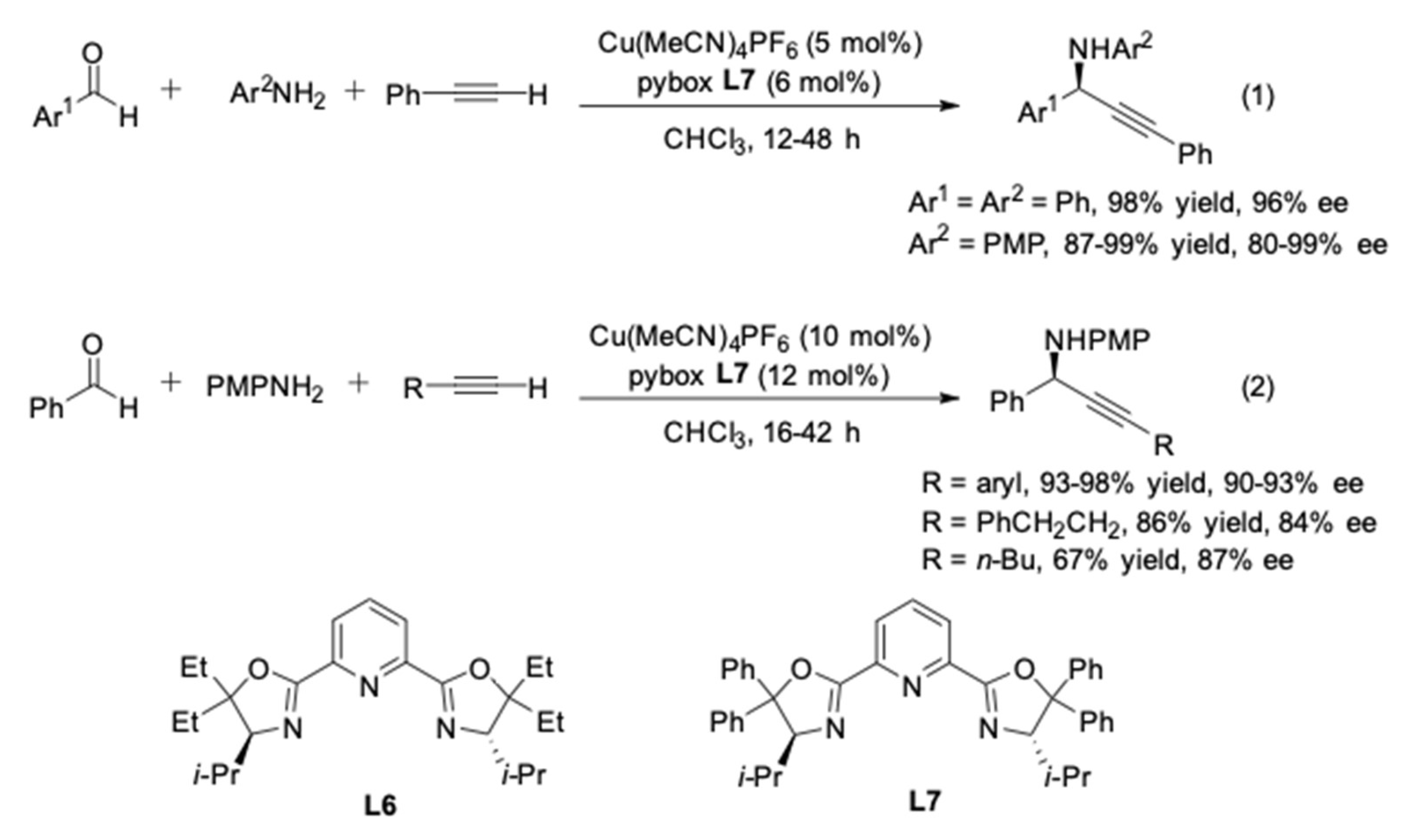
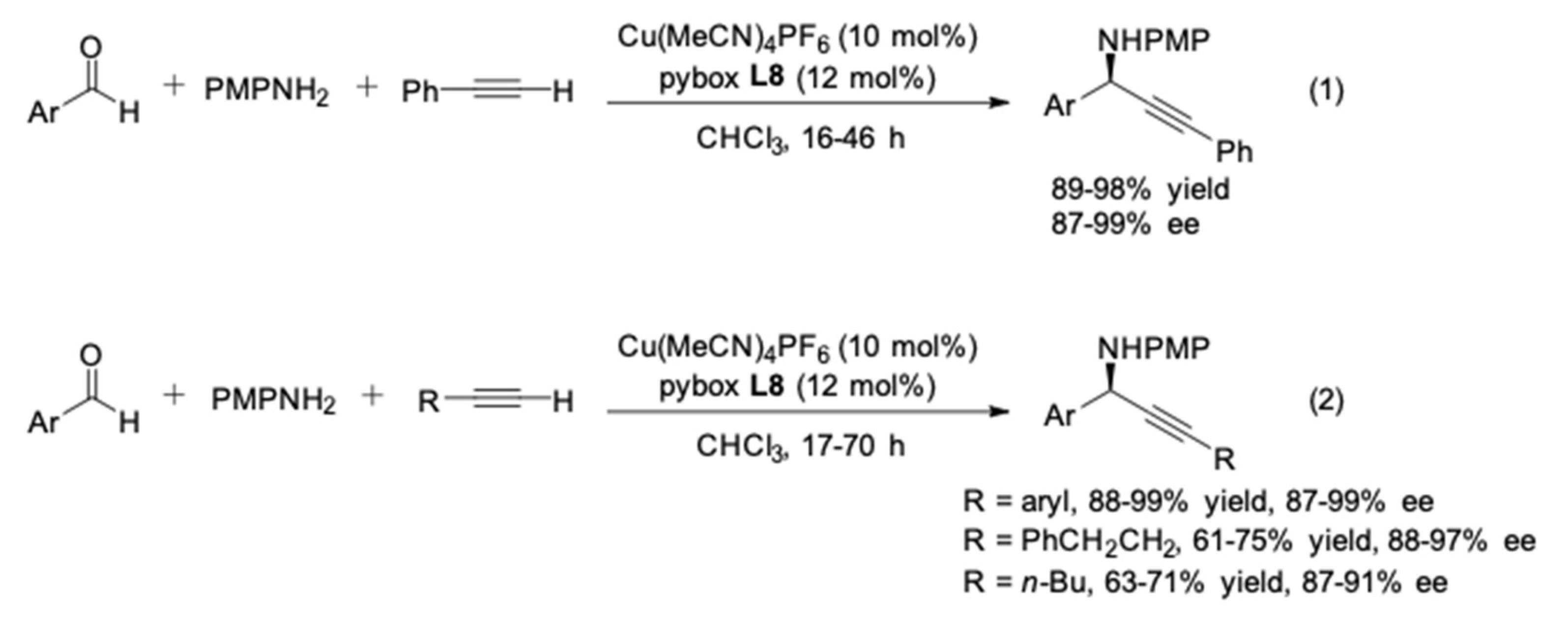
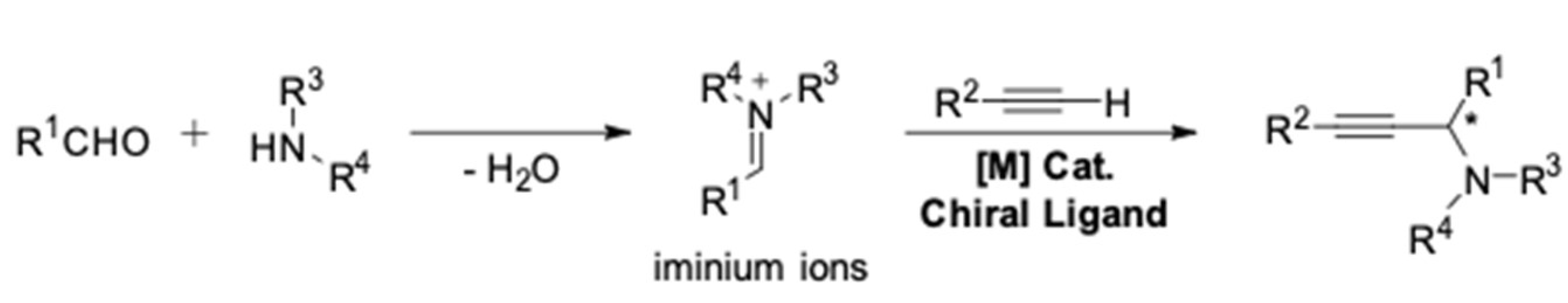
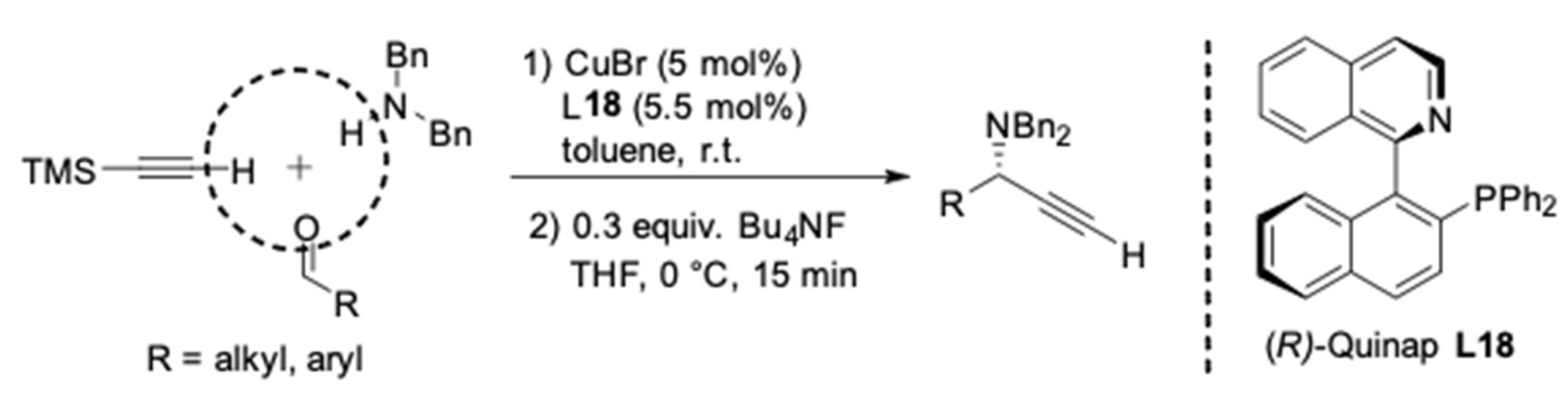
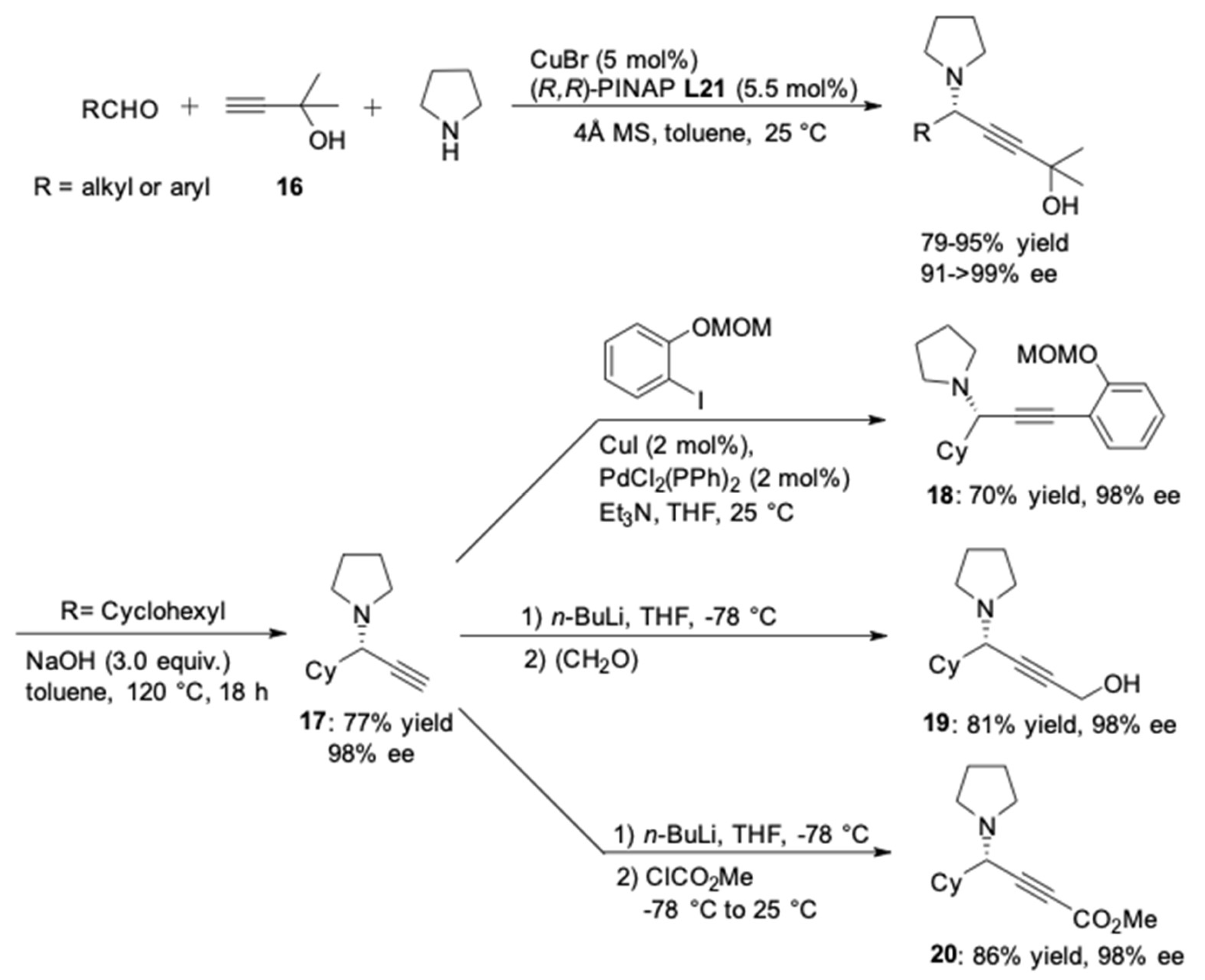
3. AA3–Coupling Derived from Secondary Amine
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ugi, I.; Dömling, A.; Hörl, W. Multicomponent Reactions in Organic Chemistry. Endeavour 1994, 18, 115–122. [Google Scholar] [CrossRef]
- Yu, J.; Shi, F.; Gong, L.-Z. Brønsted-Acid-Catalyzed Asymmetric Multicomponent Reactions for the Facile Synthesis of Highly Enantioenriched Structurally Diverse Nitrogenous Heterocycles. Acc. Chem. Res. 2011, 44, 1156–1171. [Google Scholar] [CrossRef] [PubMed]
- De Graaff, C.; Ruijter, E.; Orru, R.V.A. Recent Developments in Asymmetric Multicomponent Reactions. Chem. Soc. Rev. 2012, 41, 3969–4009. [Google Scholar] [CrossRef]
- Visbal, R.; Grau, S.; Herrera, R.P.; Gimeno, M.C. Gold Catalyzed Multicomponent Reactions beyond A3 Coupling. Molecules 2018, 23, 2255. [Google Scholar] [CrossRef]
- Touré, B.B.; Hall, D.G. Natural Product Synthesis Using Multicomponent Reaction Strategies. Chem. Rev. 2009, 109, 4439–4486. [Google Scholar] [CrossRef]
- Dömling, A.; Wang, W.; Wang, K. Chemistry and Biology of Multicomponent Reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed]
- Eckert, H. Diversity Oriented Syntheses of Conventional Heterocycles by Smart Multi Component Reactions (MCRs) of the Last Decade. Molecules 2012, 17, 1074–1102. [Google Scholar] [CrossRef]
- Trost, B.M.; Chung, C.K.; Pinkerton, A.B. Stereocontrolled Total Synthesis of (+)-Streptazolin by a Palladium-Catalyzed Reductive Diyne Cyclization. Angew. Chem. Int. Ed. 2004, 43, 4327–4329. [Google Scholar] [CrossRef]
- Fleming, J.J.; Bois, J.D. A Synthesis of (+)-Saxitoxin. J. Am. Chem. Soc. 2006, 128, 3926–3927. [Google Scholar] [CrossRef]
- Peshkov, V.A.; Pereshivko, O.P.; Van der Eycken, E.V. A Walk around the A3-Coupling. Chem. Soc. Rev. 2012, 41, 3790–3807. [Google Scholar] [CrossRef]
- Yoo, W.-J.; Zhao, L.; Li, C.-J. The A3-Coupling (Aldehyde–Alkyne–Amine) Reaction: A Versatile Method for the Preparation of Propargylamines. Aldrichimica Acta 2011, 44, 43–51. [Google Scholar]
- Gommermann, N.; Knochel, P. Practical Highly Enantioselective Synthesis of Propargylamines through a Copper-Catalyzed One-Pot Three-Component Condensation Reaction. Chem. Eur. J. 2006, 12, 4380–4392. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Li, C.-J. Enantioselective Direct-Addition of Terminal Alkynes to Imines Catalyzed by Copper(I)pybox Complex in Water and in Toluene. J. Am. Chem. Soc. 2002, 124, 5638–5639. [Google Scholar] [CrossRef]
- Wei, C.; Mague, J.T.; Li, C.-J. Cu(I)-Catalyzed Direct Addition and Asymmetric Addition of Terminal Alkynes to Imines. Proc. Natl. Acad. Sci. USA 2004, 101, 5749–5754. [Google Scholar] [CrossRef] [PubMed]
- Bisai, A.; Singh, V.K. Enantioselective One-Pot Three-Component Synthesis of Propargylamines. Org. Lett. 2006, 8, 2405–2408. [Google Scholar] [CrossRef]
- Córdova, A.; Notz, W.; Zhong, G.; Betancort, J.M.; Barbas, C.F. A Highly Enantioselective Amino Acid-Catalyzed Route to Functionalized α-Amino Acids. J. Am. Chem. Soc. 2002, 124, 1842–1843. [Google Scholar] [PubMed]
- Enders, D.; Grondal, C.; Vrettou, M.; Raabe, G. Asymmetric Synthesis of Selectively Protected Amino Sugars and Derivatives by a Direct Organocatalytic Mannich Reaction. Angew. Chem. Int. Ed. 2005, 44, 4079–4083. [Google Scholar]
- Ginotra, S.K.; Singh, V.K. Studies on Enantioselective Allylic Oxidation of Olefins Using Peresters Catalyzed by Cu(I)-Complexes of Chiral Pybox Ligands. Org. Biomol. Chem. 2006, 4, 4370–4374. [Google Scholar] [CrossRef]
- Ginotra, S.K.; Singh, V.K. Enantioselective Oxidation of Olefins Catalyzed by Chiral Copper Bis(oxazolinyl)pyridine Complexes: A Reassessment. Tetrahedron 2006, 62, 3573–3581. [Google Scholar] [CrossRef]
- Sekar, G.; DattaGupta, A.; Singh, V.K. Asymmetric Kharasch Reaction: Catalytic Enantioselective Allylic Oxidation of Olefins Using Chiral Pyridine Bis(diphenyloxazoline)−Copper Complexes and tert-Butyl Perbenzoate. J. Org. Chem. 1998, 63, 2961–2967. [Google Scholar] [CrossRef]
- DattaGupta, A.; Singh, V.K. Catalytic Enantioselective Allylic Oxidation of Olefins with Copper Complexes of Chiral Nonracemic Bis(oxazolinyl)pyridine Type Ligands. Tetrahedron Lett. 1996, 37, 2633–2636. [Google Scholar]
- Bisai, A.; Singh, V.K. Enantioselective One-Pot Three-Component Synthesis of Propargylamines Catalyzed by Copper(I)-Pyridine Bis-(oxazoline) Complexes. Tetrahedron. 2012, 68, 3480–3486. [Google Scholar]
- Irmak, M.; Groschner, A.; Boysen, M.M.K. glucoBox Ligand—A New Carbohydrate-Based Bis(oxazoline) Ligand.Synthesis and First Application. Chem. Commun. 2007, 2, 177–179. [Google Scholar]
- Irmak, M.; Lehnert, T.; Boysen, M.M.K. First Synthesis of a Carbohydrate-Derived Pyridyl Bis(thiazoline) Ligand. Tetrahedron Lett. 2007, 48, 7890–7893. [Google Scholar] [CrossRef]
- Irmak, M.; Boysen, M.M.K. A New Pyridyl Bis(oxazoline)Ligand Prepared from D-Glucosamine for Asymmetric Alkynylation of Imines. Adv. Synth. Catal. 2008, 350, 403–405. [Google Scholar] [CrossRef]
- Nakamura, S.; Hyodo, K.; Nakamura, Y.; Shibata, N.; Toru, T. Novel Enantiocomplementary C2-Symmetric Chiral Bis(imidazoline) Ligands: Highly Enantioselective Friedel–Crafts Alkylation of Indoles with Ethyl 3,3,3-Trifluoropyruvate. Adv. Synth. Catal. 2008, 350, 1443–1448. [Google Scholar] [CrossRef]
- Hara, N.; Nakamura, S.; Shibata, N.; Toru, T. First Enantioselective Synthesis of (R)-Convolutamydine B and E with N-(Heteroarenesulfonyl)prolinamides. Chem. Eur. J. 2009, 15, 6790–6793. [Google Scholar]
- Nakamura, S.; Hara, N.; Nakashima, H.; Kubo, K.; Shibata, N.; Toru, T. Enantioselective Synthesis of (R)-Convolutamydine A with New N-Heteroarylsulfonylprolinamides. Chem. Eur. J. 2008, 14, 8079–8081. [Google Scholar] [CrossRef]
- Nakamura, S.; Nakashima, H.; Sugimoto, H.; Sano, H.; Hattori, M.; Shibata, N.; Toru, T. Enantioselective C–C Bond Formation to Sulfonylimines through Use of the 2-Pyridinesulfonyl Group as a Novel Stereocontroller. Chem. Eur. J. 2008, 14, 2145–2152. [Google Scholar] [CrossRef]
- Nakamura, S.; Nakashima, H.; Yamamura, A.; Shibata, N.; Toru, T. Organocatalytic Enantioselective Hydrophosphonylation of Sulfonylimines having a Heteroarenesulfonyl Group as a Novel Stereocontroller. Adv. Synth. Catal. 2008, 350, 1209–1212. [Google Scholar]
- Nakamura, S.; Sakurai, Y.; Nakashima, H.; Shibata, N.; Toru, T. Organocatalytic Enantioselective Aza-Friedel-Crafts Alkylation of Pyrroles with N-(Heteroarenesulfonyl)imines. Synlett 2009, 10, 1639–1642. [Google Scholar] [CrossRef]
- Nakamura, S.; Ohara, M.; Nakamura, Y.; Shibata, N.; Toru, T. Copper-Catalyzed Enantioselective Three-Component Synthesis of Optically Active Propargylamines from Aldehydes, Amines, and Aliphatic Alkynes. Chem. Eur. J. 2010, 16, 2360–2362. [Google Scholar] [CrossRef] [PubMed]
- Shao, Z.; Pu, X.; Li, X.; Fan, B.; Chan, A.S.C. Enantioselective, Copper(I)-Catalyzed Three-Component Reaction for the Synthesis of β, γ-Alkynyl α-Amino Acid Derivatives. Tetrahedron: Asymmetry 2009, 20, 225–229. [Google Scholar] [CrossRef]
- Abdulganeeva, S.A.; Erzhanov, K.B. Acetylenic Amino Acids. Russ. Chem. Rev. 1991, 60, 676–687. [Google Scholar] [CrossRef]
- Angst, C. Stereoselective Synthesis of β, γ-Unsaturated Amino Acids. Pure Appl. Chem. 1987, 59, 373–380. [Google Scholar] [CrossRef]
- Gao, X.-T.; Gan, C.-C.; Liu, S.-Y.; Zhou, F.; Wu, H.-H.; Zhou, J. Utilization of CO2 as a C1 Building Block in a Tandem Asymmetric A3 Coupling-Carboxylative Cyclization Sequence to 2-Oxazolidinones. ACS Catal. 2017, 7, 8588–8593. [Google Scholar] [CrossRef]
- Alcock, N.W.; Brown, J.M.; Hulmes, D.I. Synthesis and Resolution of 1-(2-Diphenylphosphino-1-naphthyl)isoquinoline; a P–N Chelating Ligand for Asymmetric Catalysis. Tetrahedron: Asymmetry 1993, 4, 743–756. [Google Scholar] [CrossRef]
- Gommermann, N.; Koradin, C.; Polborn, K.; Knochel, P. Enantioselective, Copper(I)-Catalyzed Three-Component Reaction for the Preparation of Propargylamines. Angew. Chem. Int. Ed. 2003, 42, 5763–5766. [Google Scholar] [CrossRef]
- Koradin, C.; Polborn, K.; Knochel, P. Enantioselective Synthesis of Propargylamines by Copper-Catalyzed Addition of Alkynes to Enamines. Angew. Chem. Int. Ed. 2002, 41, 2535–2538. [Google Scholar] [CrossRef]
- Koradin, C.; Gommermann, N.; Polborn, K.; Knochel, P. Synthesis of Enantiomerically Enriched Propargylamines by Copper-Catalyzed Addition of Alkynes to Enamines. Chem. Eur. J. 2003, 9, 2797–2811. [Google Scholar] [CrossRef]
- Gommermann, N.; Knochel, P. Practical Highly Enantioselective Synthesis of Terminal Propargylamines. An Expeditious Synthesis of (S)-(+)-Coniine. Chem. Commun. 2004, 2324–2325. [Google Scholar] [CrossRef] [PubMed]
- Gommermann, N.; Knochel, P. Preparation of Functionalized Primary Chiral Amines and Amides via an Enantioselective Three-Component Synthesis of Propargylamines. Tetrahedron 2005, 61, 11418–11426. [Google Scholar] [CrossRef]
- Dube, H.; Gommermann, N.; Knochel, P. Synthesis of Chiral α-Aminoalkylpyrimidines Using an Enantioselective Three-Component Reaction. Synthesis 2004, 12, 2015–2025. [Google Scholar] [CrossRef]
- Gommermann, N.; Gehrig, A.; Knochel, P. Enantioselective Synthesis of Chiral a-Aminoalkyl-1,2,3-triazoles Using α-Three-Component Reaction. Synlett 2005, 18, 2796–2798. [Google Scholar] [CrossRef]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Kolb, H.C.; Sharpless, K.B. The Growing Impact of Click Chemistry on Drug Discovery. Drug. Discov. Today 2003, 8, 1128–1137. [Google Scholar] [CrossRef]
- Fürstner, A.; Szillat, H.; Gabor, B.; Mynott, R. Platinum- and Acid-Catalyzed Enyne Metathesis Reactions: Mechanistic Studies and Applications to the Syntheses of Streptorubin B and Metacycloprodigiosin. J. Am. Chem. Soc. 1998, 120, 8305–8314. [Google Scholar] [CrossRef]
- Karpov, A.S.; Müller, T.J.J. New Entry to a Three-Component Pyrimidine Synthesis by TMS−Ynones via Sonogashira Coupling. Org. Lett. 2003, 5, 3451–3454. [Google Scholar] [CrossRef]
- Adlington, R.M.; Baldwin, J.E.; Catterick, D.; Pritchard, G.J. A Versatile Approach to Pyrimidin-4-yl Substituted α-Aminoacids from Alkynyl Ketones; the Total Synthesis of L-Lathyrine. Chem. Commun. 1997, 1757–1758. [Google Scholar] [CrossRef]
- Adlington, R.M.; Baldwin, J.E.; Catterick, D.; Pritchard, G.J. The Synthesis of Pyrimidin-4-yl Pubstituted α-Amino Acids. Aversatile Approach from Alkynyl Ketones. J. Chem. Soc., Perkin Trans. 1 1999, 855–866. [Google Scholar] [CrossRef]
- Adlington, R.M.; Baldwin, J.E.; Pritchard, G.J.; Spencer, K.C. Synthesis of Novel C-Nucleosides with Potential Applications in Combinatorial and Parallel Synthesis. Tetrahedron Lett. 2000, 41, 575–578. [Google Scholar] [CrossRef]
- Bois, F.; Gardette, D.; Gramain, J.-C. A New Asymmetric Synthesis of (S)-(+)-Pipecoline and (S)-(+)- and (R)-(−)-Coniine by Reductive Photocyclization of Dienamides. Tetrahedron Lett. 2000, 41, 8769–8772. [Google Scholar]
- Chippindale, A.M.; Davies, S.G.; Iwamoto, K.; Parkin, R.M.; Smethurst, C.A.P.; Smith, A.D.; Rodriguez-Solla, H. Asymmetric Synthesis of Cyclic β-Amino Acids and Cyclic Amines via Sequential Diastereoselective Conjugate Addition and Ring Closing Metathesis. Tetrahedron 2003, 59, 3253–3265. [Google Scholar] [CrossRef]
- Hayes, J.F.; Shipman, M.; Twin, H. Asymmetric Synthesis of 2-Substituted Piperidines Using a Multi-Component Coupling Reaction: Rapid Assembly of (S)-Coniine from (S)-1-(1-phenylethyl)-2-Methyleneaziridine. Chem. Commun. 2001, 1784–1785. [Google Scholar] [CrossRef]
- Gommermann, N.; Knochel, P. 2-Phenallyl as a Versatile Protecting Group for the Asymmetric One-Pot Three-Component Synthesis of Propargylamines. Chem. Commun. 2005, 4175–4177. [Google Scholar] [CrossRef]
- Gommermann, N.; Knochel, P. Highly Enantioselective Synthesis of Propargylamines Using (Mesitylmethyl)benzylamine. Synlett 2005, 18, 2799–2801. [Google Scholar] [CrossRef]
- Knöpfel, T.F.; Aschwanden, P.; Ichikawa, T.; Watanabe, T.; Carreira, E.M. Readily Available Biaryl P,N Ligands for Asymmetric Catalysis. Angew. Chem. Int. Ed. 2004, 43, 5971–5973. [Google Scholar] [CrossRef]
- Aschwanden, P.; Stephenson, C.R.J.; Carreira, E.M. Highly Enantioselective Access to Primary Propargylamines: 4-Piperidinone as a Convenient Protecting Group. Org. Lett. 2006, 8, 2437–2440. [Google Scholar] [CrossRef]
- Fan, W.; Ma, S. An Easily Removable Stereo-dictating Group for Enantioselective Synthesis of Propargylic Amines. Chem. Commun. 2013, 49, 10175–10177. [Google Scholar] [CrossRef]
- Neenan, T.X.; Whitesides, G.M. Synthesis of High Carbon Materials from Acetylenic Precursors. Preparation of Aromatic Monomers Bearing Multiple Ethynyl Groups. J. Org. Chem. 1988, 53, 2489–2496. [Google Scholar]
- Schmittel, M.; Ammon, H. Preparation of a Rigid Macrocycle with Two Exotopic Phenanthroline Binding Sites. Synlett 1999, 6, 750–752. [Google Scholar] [CrossRef]
- Tykwinski, R.R. Evolution in the Palladium-Catalyzed Cross-Coupling of sp- and sp2-Hybridized Carbon Atoms. Angew. Chem. Int. Ed. 2004, 42, 1566–1568. [Google Scholar] [CrossRef]
- Heuft, M.A.; Collins, S.K.; Yep, G.P.A.; Fallis, A.G. Synthesis of Diynes and Tetraynes from in Situ Desilylation/Dimerization of Acetylenes. Org. Lett. 2001, 3, 2883–2886. [Google Scholar] [CrossRef]
- Fan, W.; Yuan, W.; Ma, S. Unexpected E-stereoselective Reductive A3-Coupling Reaction of Terminal Alkynes with Aldehydes and Amines. Nat. Commun. 2014, 5, 3884–3892. [Google Scholar] [CrossRef]
- Cardoso, F.S.P.; Abboud, K.A.; Aponick, A. Design, Preparation, and Implementation of an Imidazole-Based Chiral Biaryl P,N-Ligand for Asymmetric Catalysis. J. Am. Chem. Soc. 2013, 135, 14548–14551. [Google Scholar] [CrossRef]
- Paioti, P.H.S.; Abboud, K.A.; Aponick, A. Incorporation of Axial Chirality into Phosphino-Imidazoline Ligands for Enantioselective Catalysis. ACS Catal. 2017, 7, 2133–2138. [Google Scholar] [CrossRef]
- Aponick, A.; Li, C.-Y.; Malinge, J.; Marques, E.F. An Extremely Facile Synthesis of Furans, Pyrroles, and Thiophenes by the Dehydrative Cyclization of Propargyl Alcohols. Org. Lett. 2009, 11, 4624–4627. [Google Scholar] [CrossRef]
- Naeimi, H.; Moradian, M. Thioether-based Copper(I) Schiff Base Complex as a Catalyst for a Direct and Asymmetric A3-coupling Reaction. Tetrahedron: Asymmetry 2014, 25, 429–434. [Google Scholar] [CrossRef]
- Zhao, C.; Seidel, D. Enantioselective A3 Reactions of Secondary Amines with a Cu(I)/Acid−Thiourea Catalyst Combination. J. Am. Chem. Soc. 2015, 137, 4650–4653. [Google Scholar] [CrossRef]
- Periasamy, M.; Sanjeevakumar, N.; Dalai, M.; Gurubrahamam, R.; Reddy, P.O. Highly Enantioselective Synthesis of Chiral Allenes by Sequential Creation of Stereogenic Center and Chirality Transfer in a Single Pot Operation. Org. Lett. 2012, 14, 2932–2935. [Google Scholar] [CrossRef]

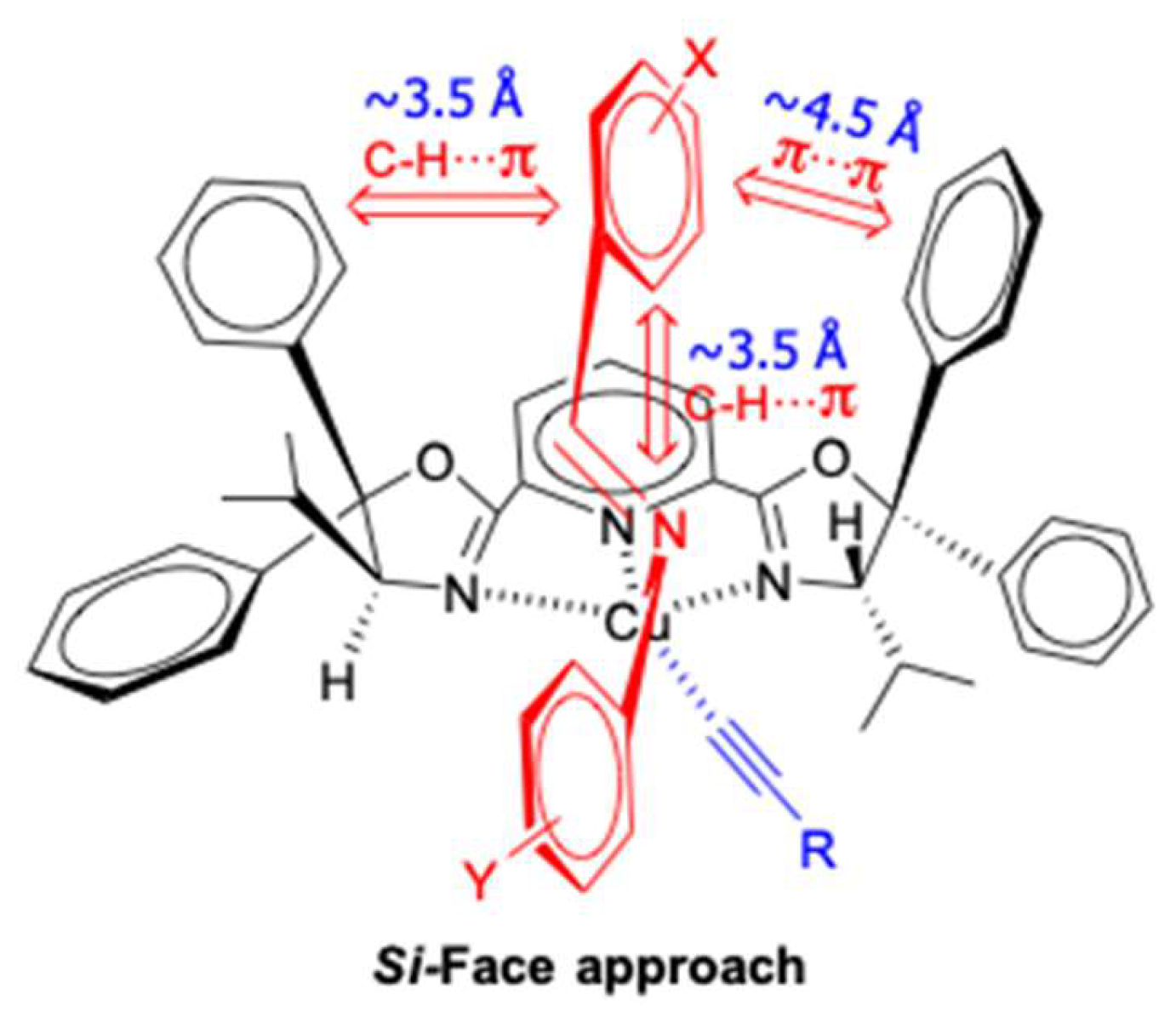
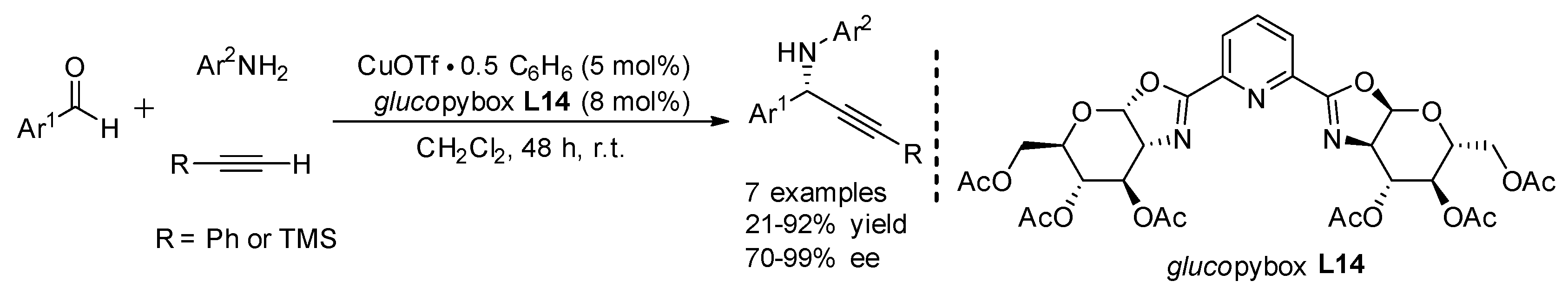
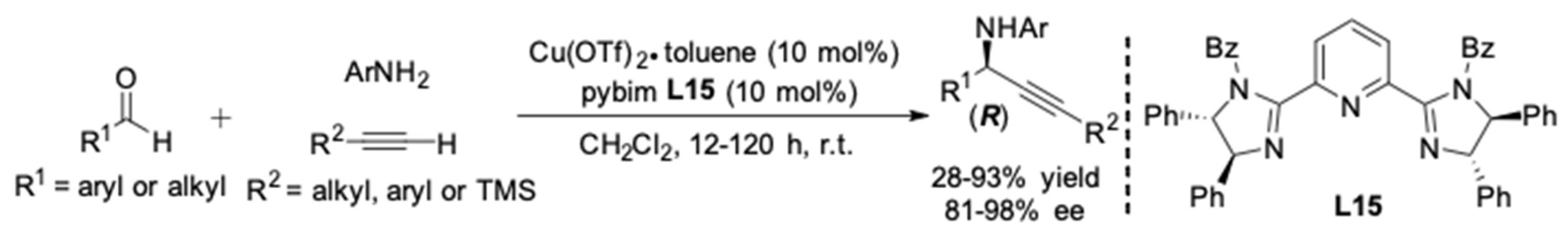
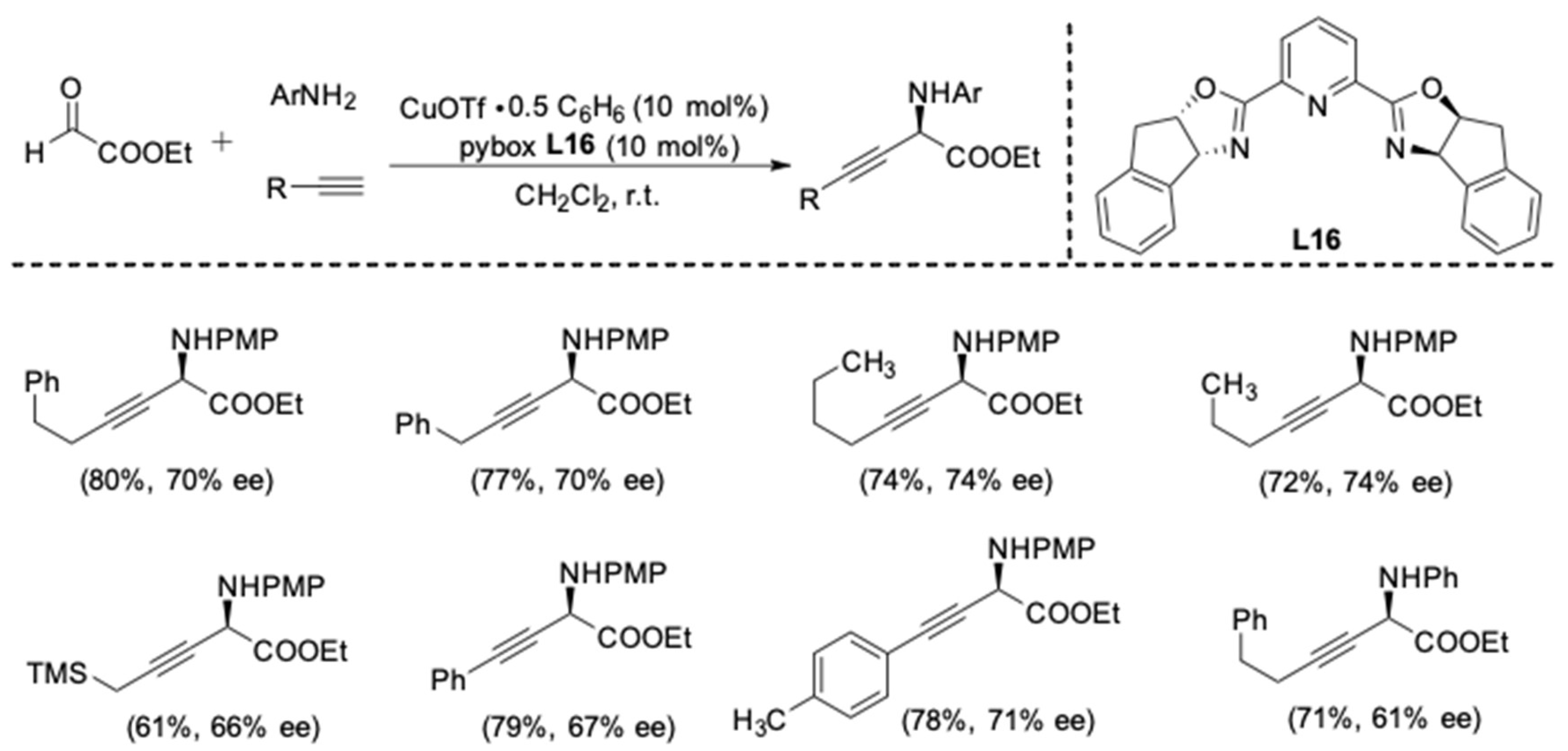
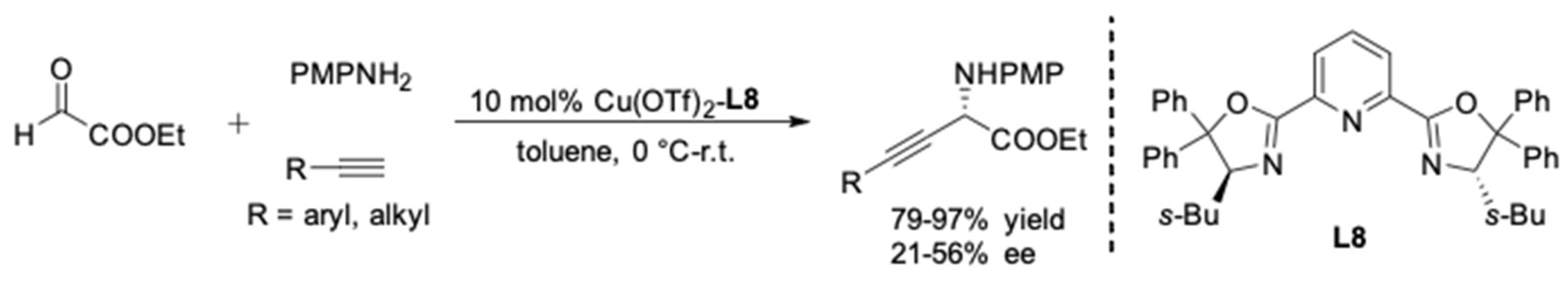



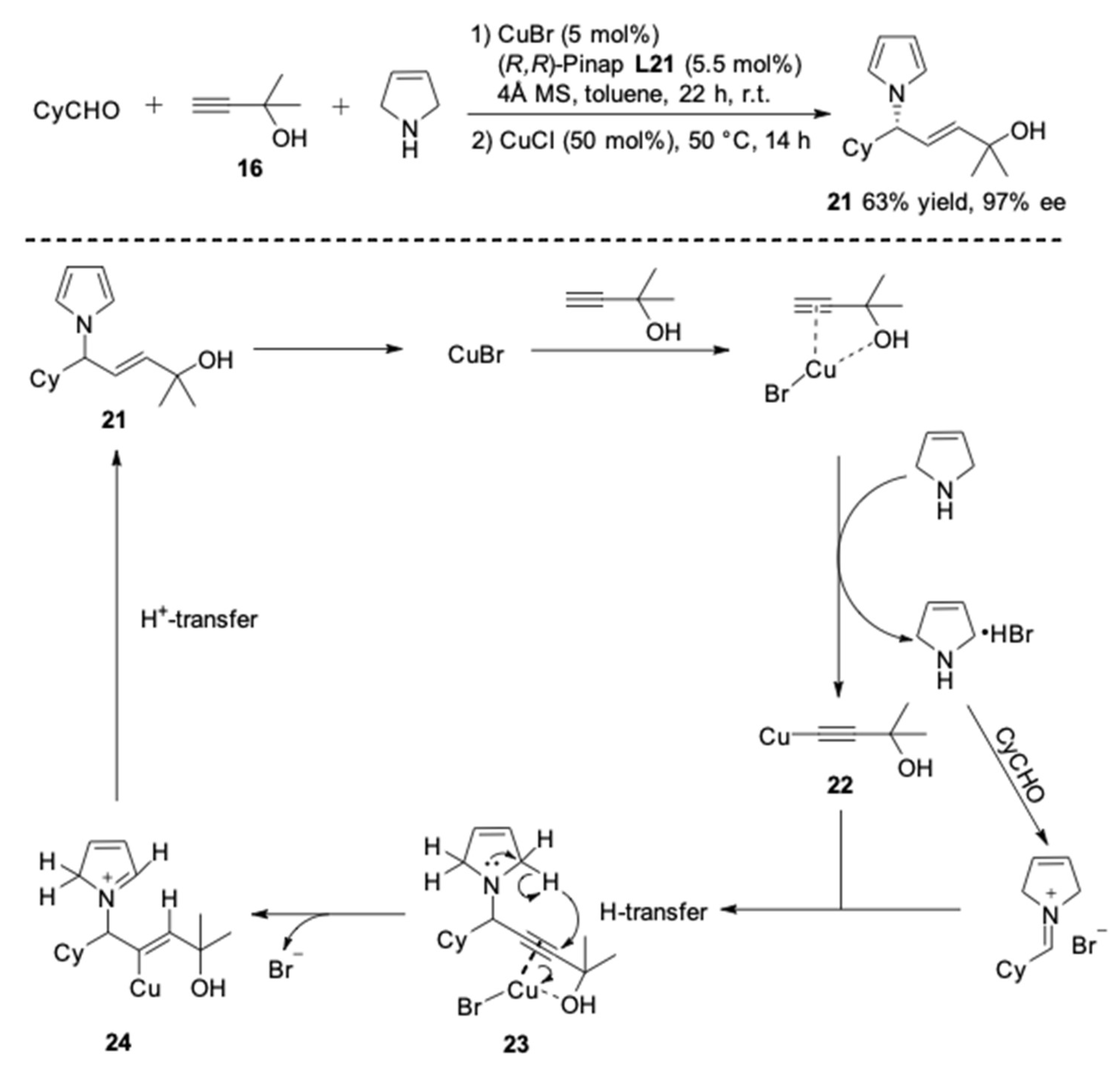
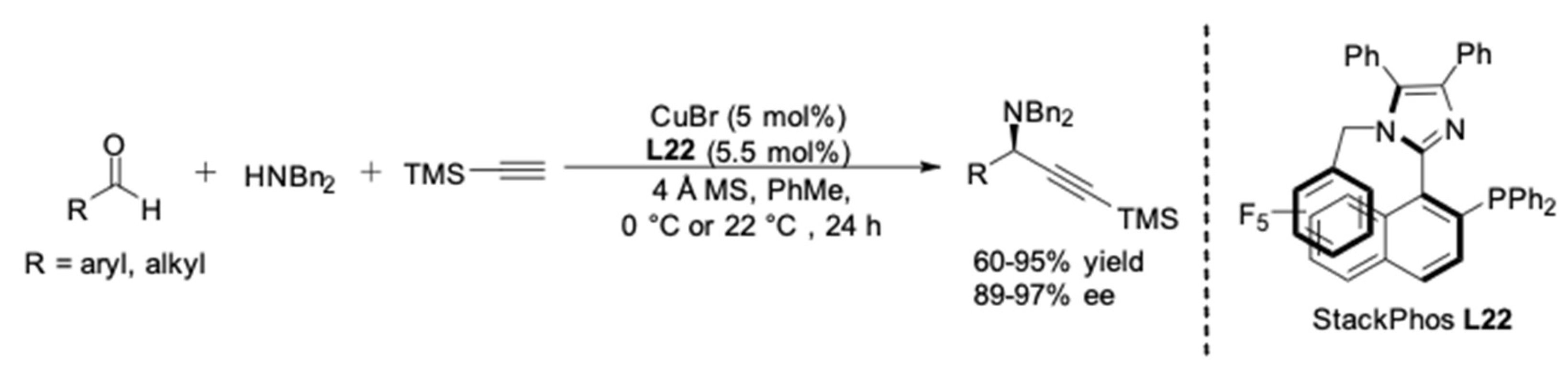
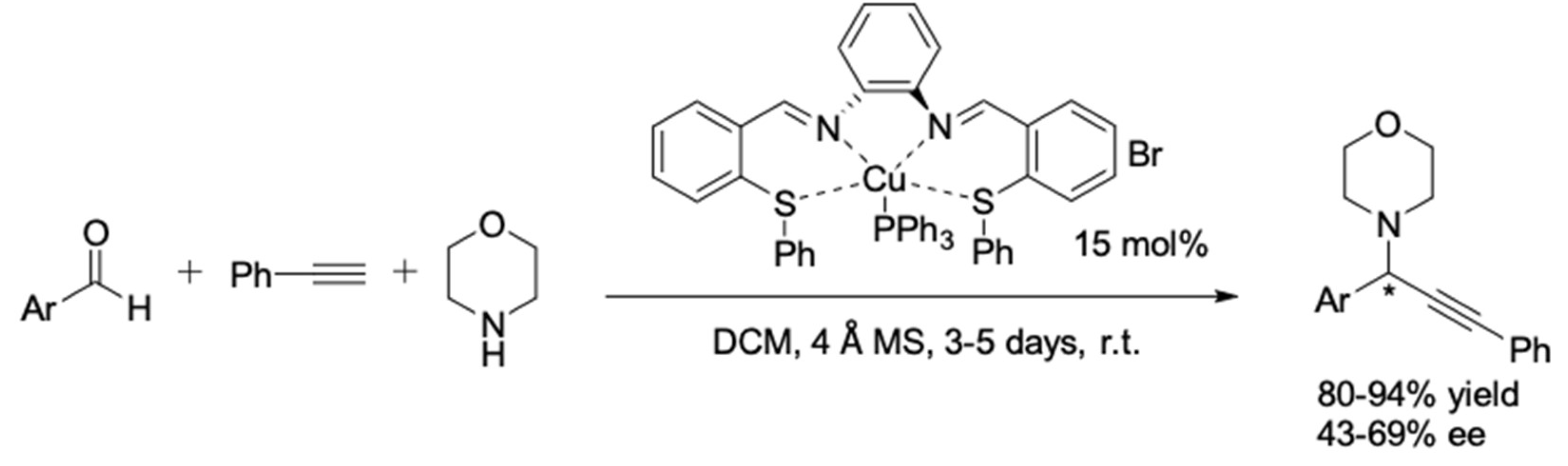
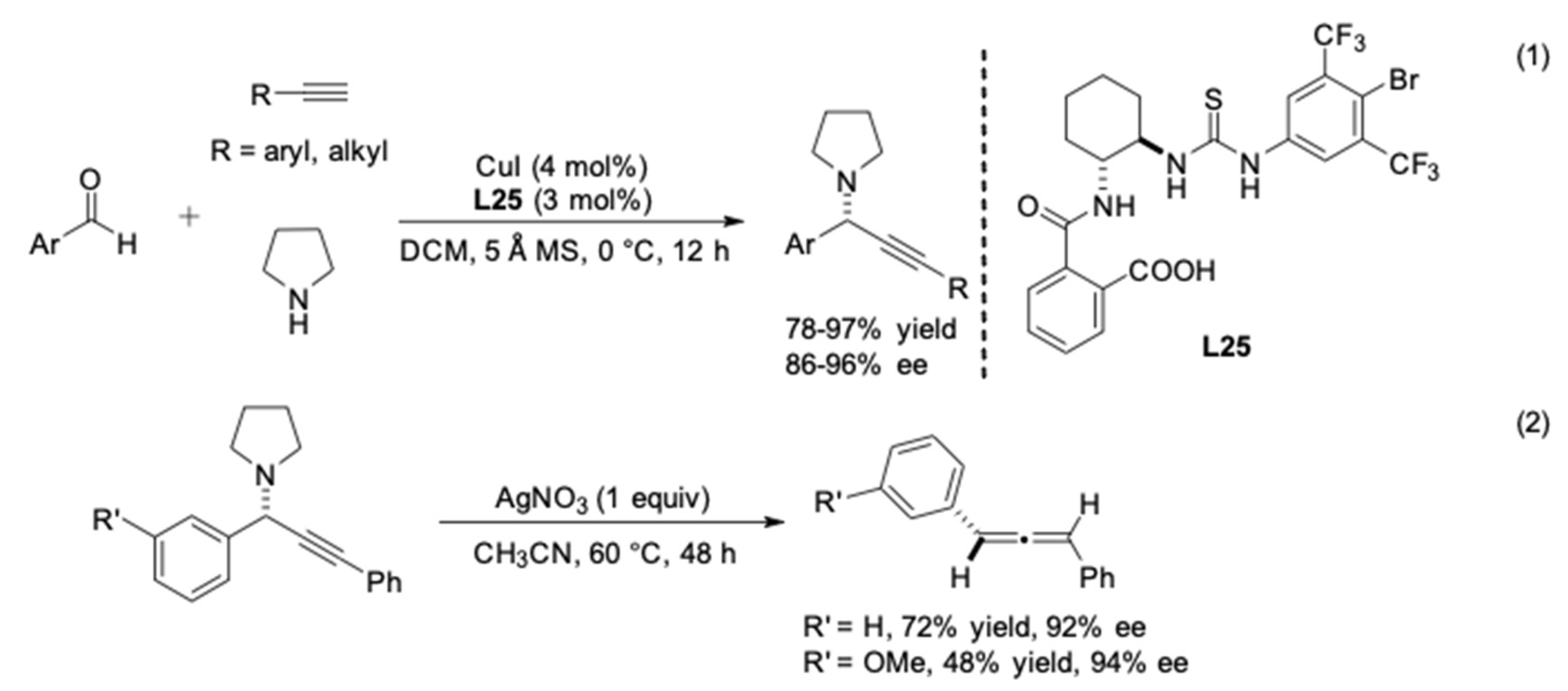
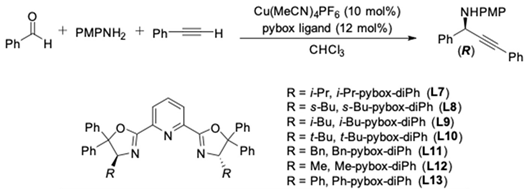
| Entry | Pybox | Time | Yield a (%) | ee (%) |
|---|---|---|---|---|
| 1 | i-Pr-pybox-diPh (L7) | 16 h | 98 | 90 |
| 2 | s-Bu-pybox-diPh (L8) | 18 h | 97 | 93 |
| 3 | i-Bu-pybox-diPh (L9) | 4 days | 56 | 63 |
| 4 | t-Bu-pybox-diPh (L10) | 22 h | 90 | 68 |
| 5 | Bn-pybox-diPh (L11) | 5 days | 45 | 64 |
| 6 | Me-pybox-diPh (L12) | 4 days | 51 | 53 |
| 7 | Ph-pybox-diPh (L13) | 28 h | 96 | 75(S) b |
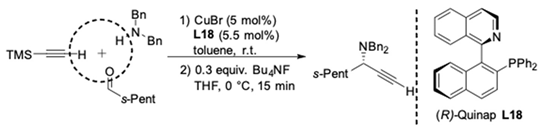
| Run | Yield (%) a | ee (%) b |
|---|---|---|
| 1 | 99 | 98 |
| 2 | 87 | 97 |
| 3 | 89 | 98 |
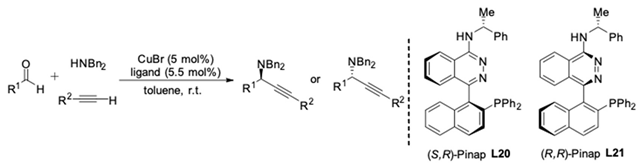
| R1 | R2 | Ligand | Yield [%] | Ee [%] | Quinap [% ee] |
|---|---|---|---|---|---|
| i-Pr | Me3Si | L20 | 84 | 98 (R) | 92 |
| L21 | 82 | 99 (S) | |||
| i-Pr | Ph | L20 | 88 | 90 (R) | 84 |
| L21 | 82 | 95 (S) | |||
| i-Bu | i-Bu | L20 | 74 | 91 (R) | 82 |
| L21 | 72 | 94 (S) |
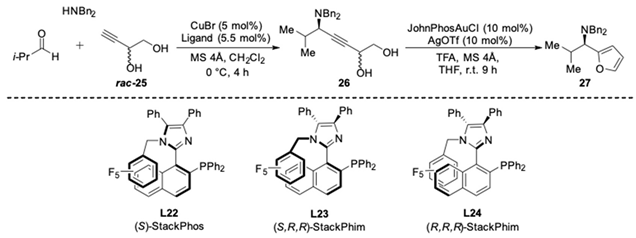
| Entry | Ligand | Product | Yield 26/26′ | Yield 27/ent-27 | ee |
|---|---|---|---|---|---|
| 1 | L22 |  | 86% | 80% | 66% |
| 2 | L23 | 64% | 75% | 82% | |
| 3 | L24 |  | 74% | 81% | −94% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mo, J.-N.; Su, J.; Zhao, J. The Asymmetric A3(Aldehyde–Alkyne–Amine) Coupling: Highly Enantioselective Access to Propargylamines. Molecules 2019, 24, 1216. https://doi.org/10.3390/molecules24071216
Mo J-N, Su J, Zhao J. The Asymmetric A3(Aldehyde–Alkyne–Amine) Coupling: Highly Enantioselective Access to Propargylamines. Molecules. 2019; 24(7):1216. https://doi.org/10.3390/molecules24071216
Chicago/Turabian StyleMo, Jia-Nan, Junqi Su, and Jiannan Zhao. 2019. "The Asymmetric A3(Aldehyde–Alkyne–Amine) Coupling: Highly Enantioselective Access to Propargylamines" Molecules 24, no. 7: 1216. https://doi.org/10.3390/molecules24071216
APA StyleMo, J.-N., Su, J., & Zhao, J. (2019). The Asymmetric A3(Aldehyde–Alkyne–Amine) Coupling: Highly Enantioselective Access to Propargylamines. Molecules, 24(7), 1216. https://doi.org/10.3390/molecules24071216






