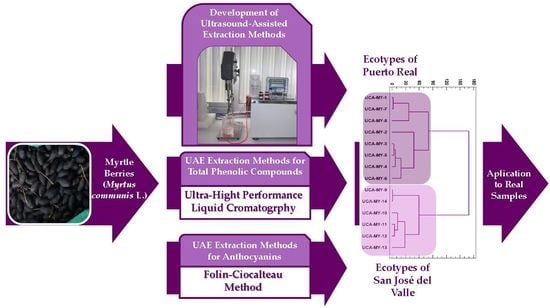
Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Development of the UAE Method
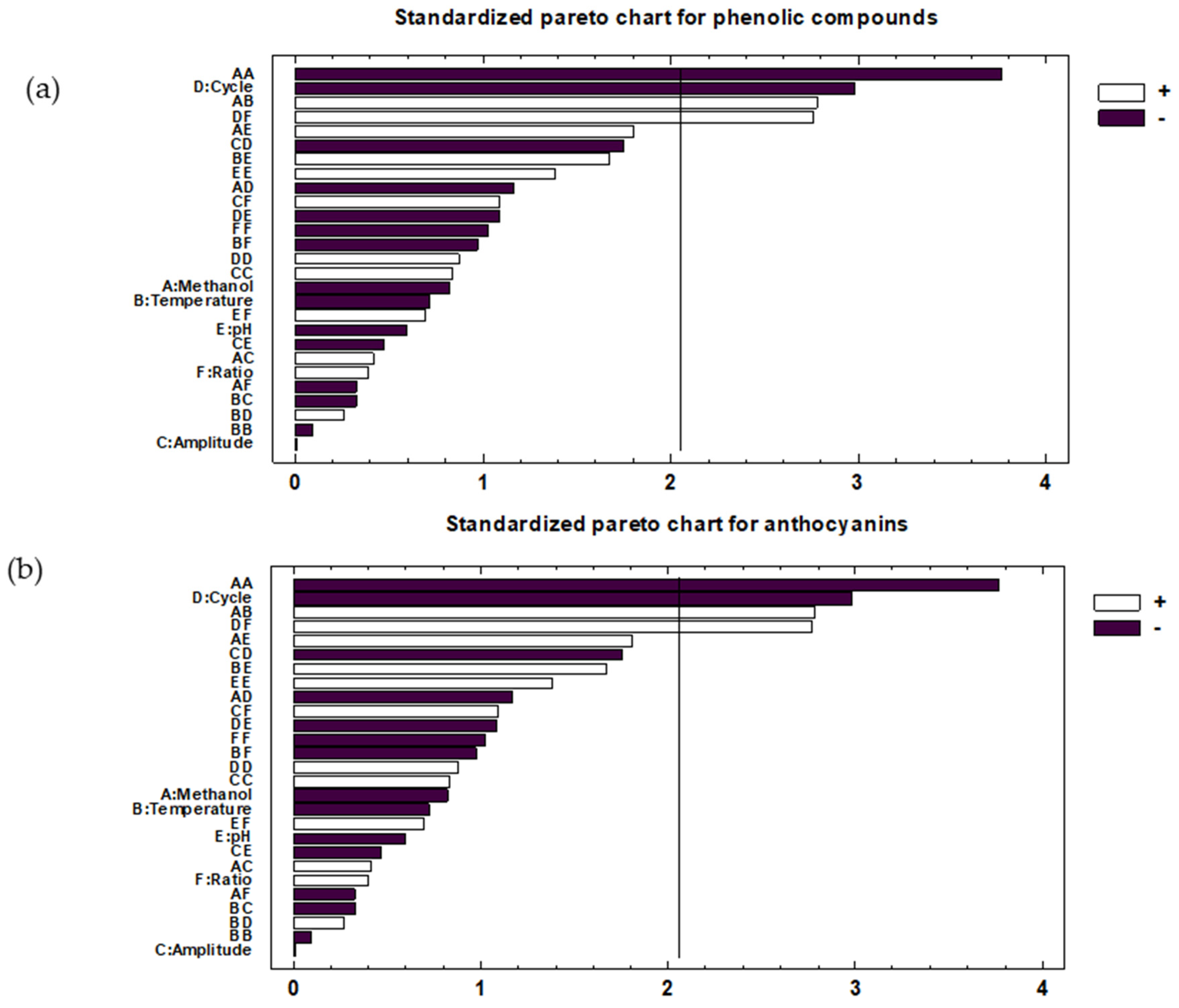
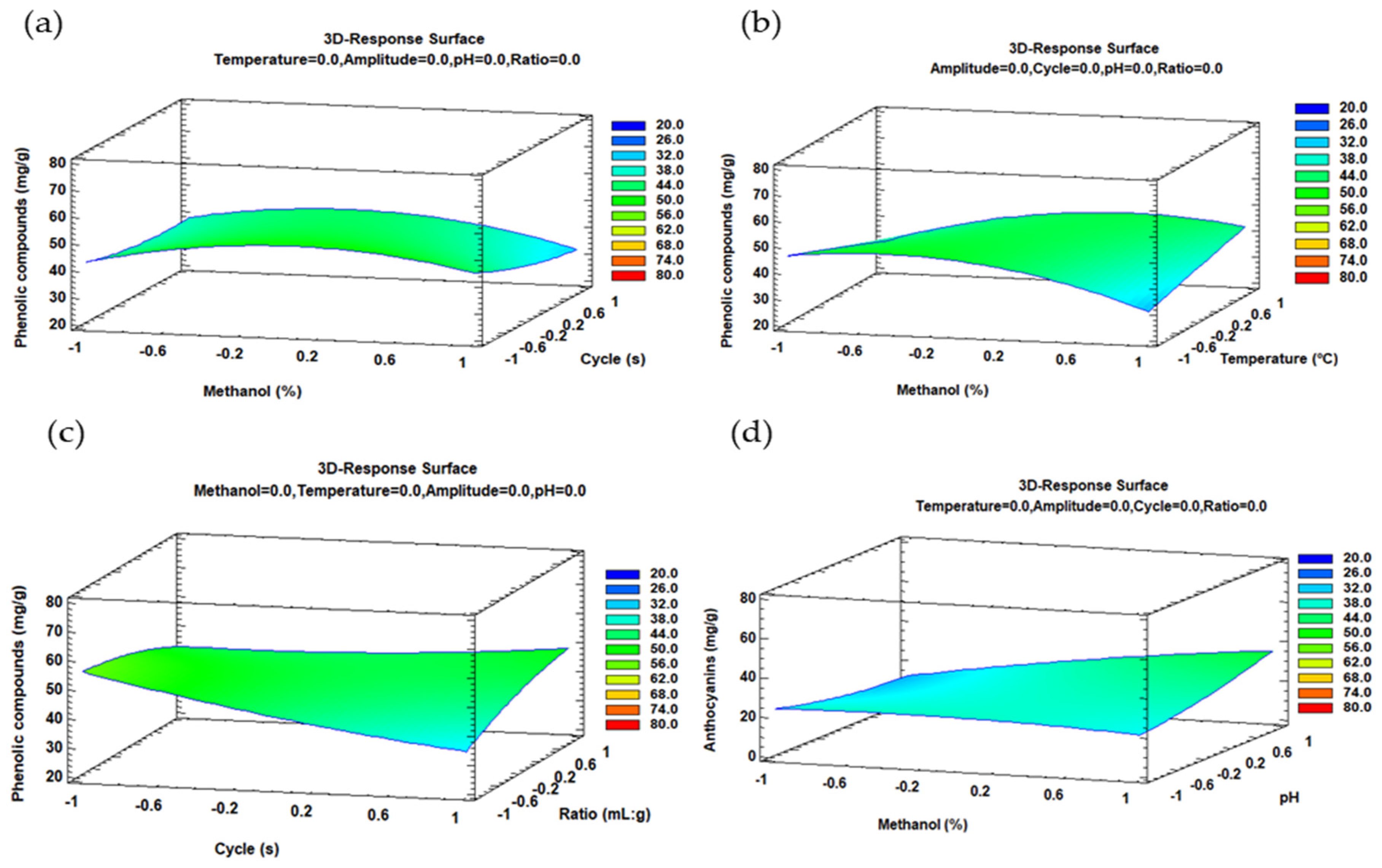
2.2. Optimal Conditions
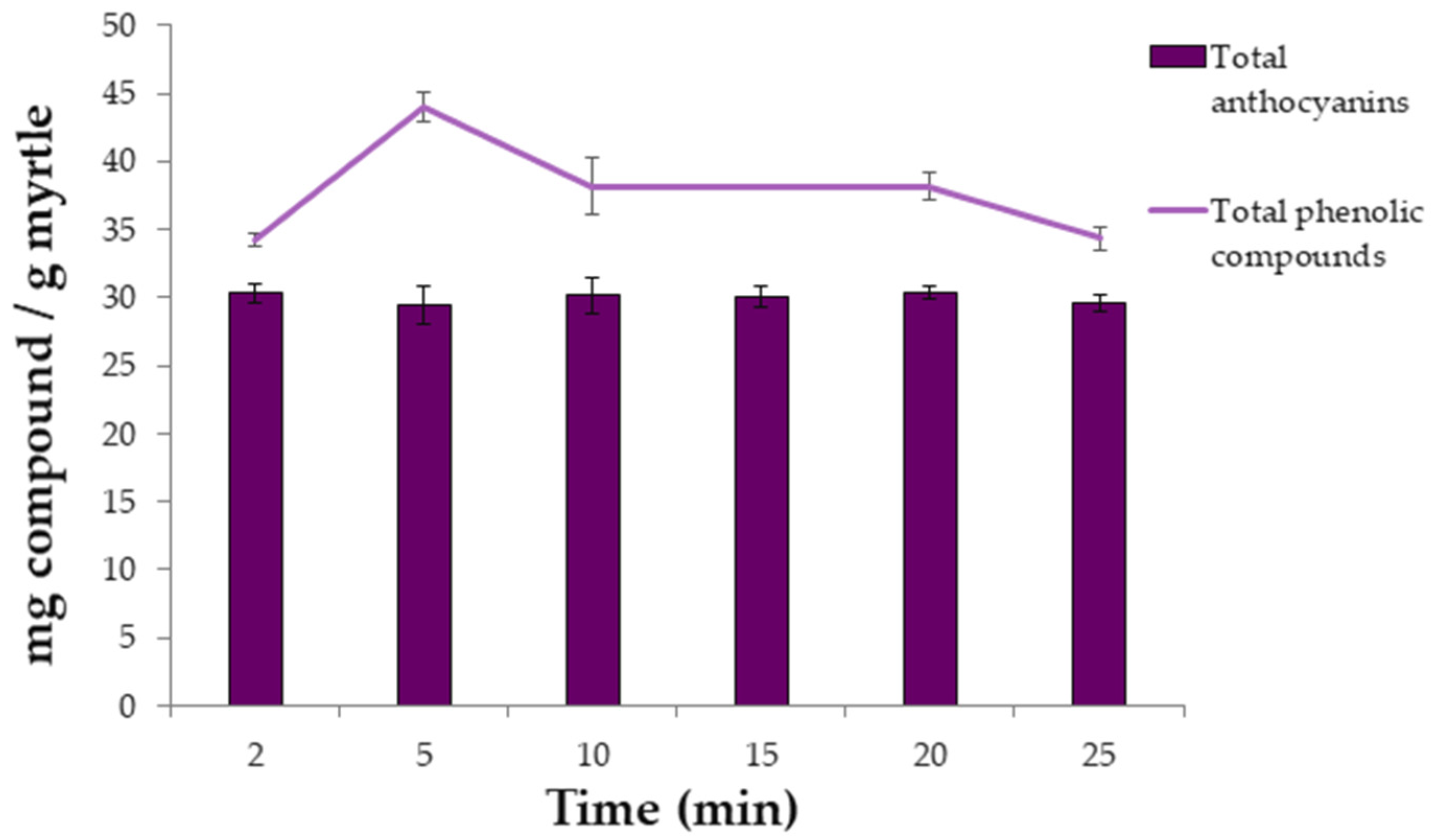
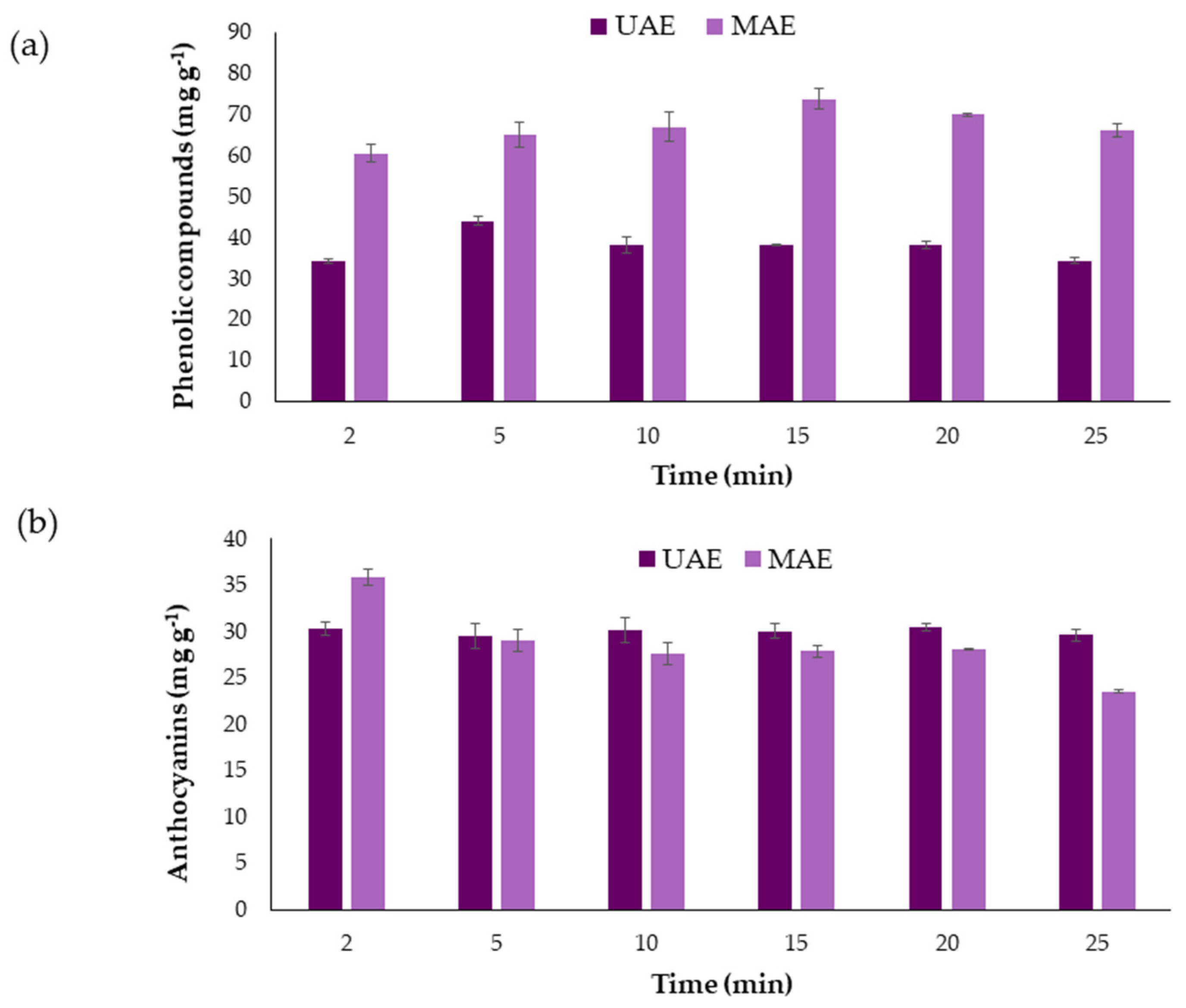
2.3. Extraction Time
2.4. Repeatability and Intermediate Precision of UAE Methods
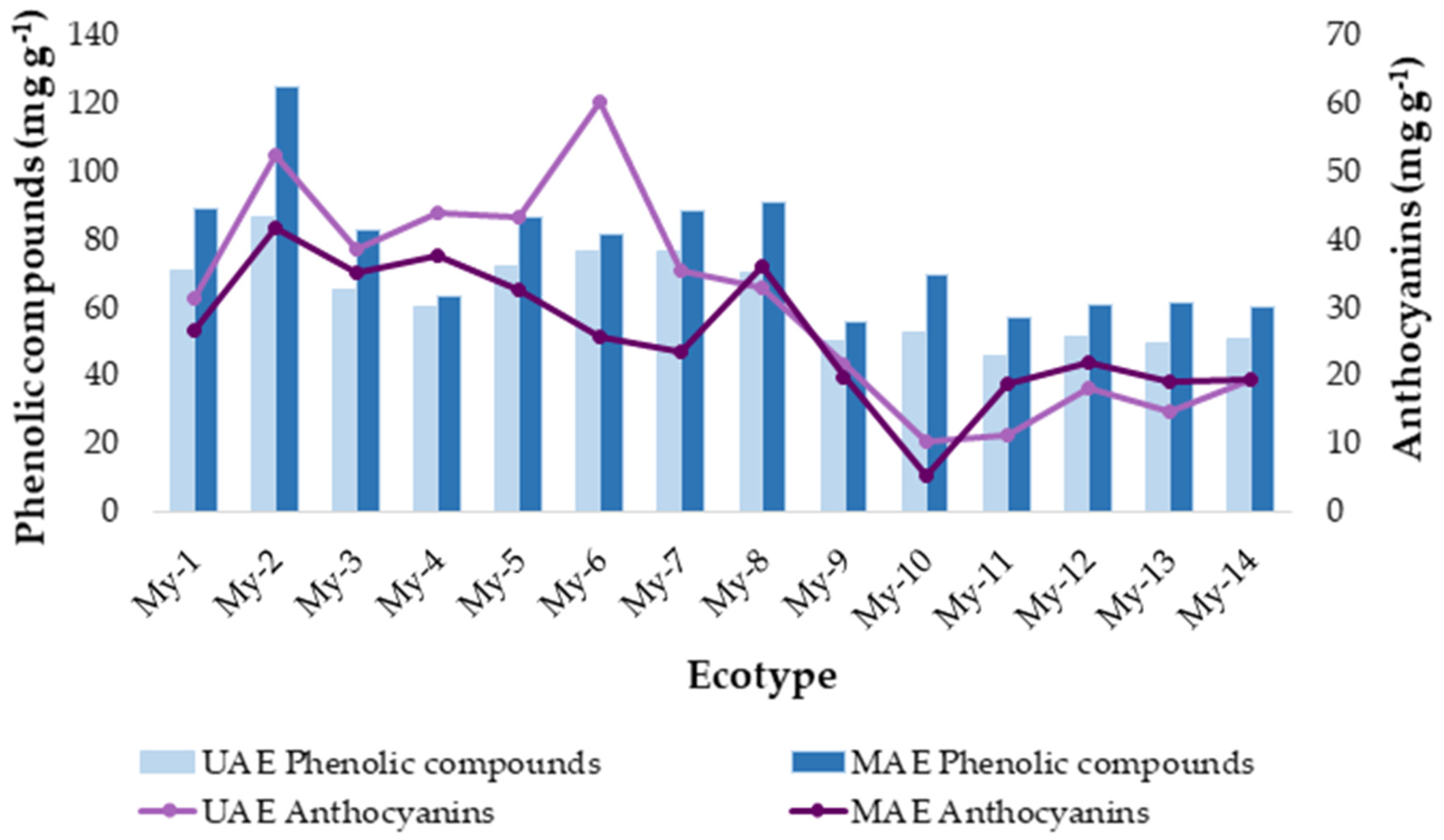
2.5. Application of the Developed Methods to Ecotypes from Two Locations
2.6. Analysis by Conglomerate
2.7. Comparison Study: UAE vs. MAE
3. Materials and Methods
3.1. Plant Materials
3.2. Chemicals and Solvents
3.3. Ultrasound-Assisted Extraction Procedure
3.4. Determining the Content of Total Phenolic Compounds by Folin-Ciocalteau Essay
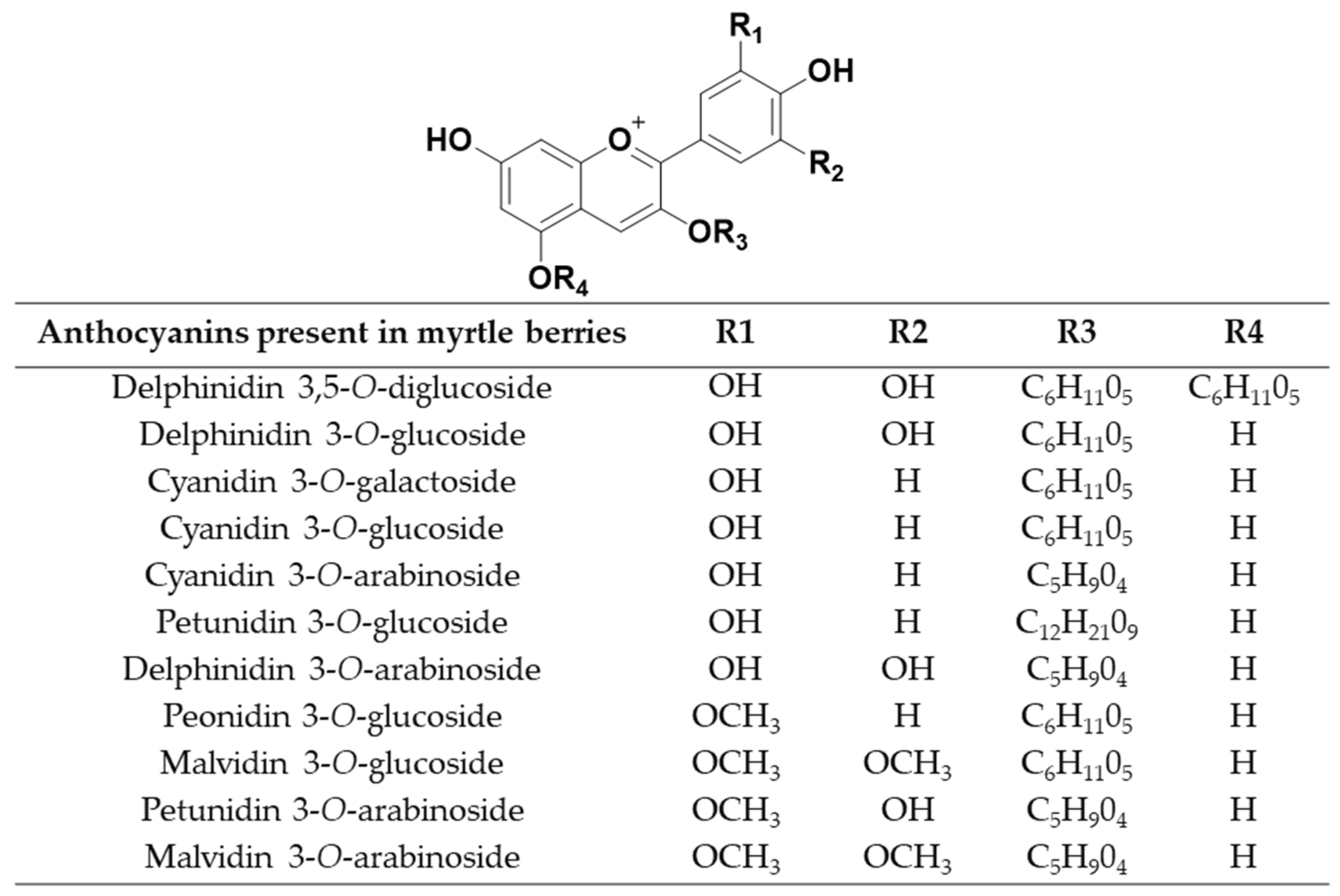
3.5. Identification of Anthocyanins by UHPLC-QToF-MS
3.6. Determination of Anthocyanins by UHPLC-UV-Vis System
3.7. Application of Box-Behnken Design (BBD) to the Optimization of the Extraction Methods
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tian, Y.; Puganen, A.; Alakomi, H.L.; Uusitupa, A.; Saarela, M.; Yang, B. Antioxidative and antibacterial activities of aqueous ethanol extracts of berries, leaves, and branches of berry plants. Food Res. Int. 2018, 106, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Ye, Q.; Jiang, L.; Luo, Z. Effects of nano-TiO2 packaging on postharvest quality and antioxidant activity of strawberry (Fragaria × ananassa Duch.) stored at low temperature. J. Sci. Food Agric. 2017, 97, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Łata, B.; Trampczynska, A.; Paczesna, J. Cultivar variation in apple peel and whole fruit phenolic composition. Sci. Hortic. 2009, 121, 176–181. [Google Scholar] [CrossRef]
- Ahmad, N.; Zuo, Y.; Lu, X.; Anwar, F.; Hammed, S. Anwar. Characterization of free and conjugated phenolic compounds in fruits of selected wild plants. Food Chem. 2016, 190, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Bamba, B.S.B.; Shi, J.; Tranchant, C.C.; Xue, S.J.; Forney, C.F.; Lim, L.-T. Influence of Extraction Conditions on Ultrasound-Assisted Recovery of Bioactive Phenolics from Blueberry Pomace and Their Antioxidand Activity. Molecules 2018, 23, 1685. [Google Scholar] [CrossRef] [PubMed]
- Wannes, A.; Mhamdi, B.; Sriti, J.; Ben Jemia, M.; Ouchikh, O.; Hamdaoui, G.; Kchouk, M.E.; Marzouk, B. Antioxidant activities of the essential oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf, stem and flower. Food Chem. Toxicol. 2010, 48, 1362–1370. [Google Scholar] [CrossRef] [PubMed]
- Limwachiranon, J.; Jiang, L.; Huang, H.; Sun, J.; Luo, Z. Improvement of phenolic compounds extraction from high-starch lotus (Nelumbo nucifera G.) seed kernels using glycerol: New insights to amylose/amylopectin—Phenolic relationships. Food Chem. 2019, 274, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.C.; Cebola, M.-J.; Bernardo-Gil, M.G. Evolution of the Yields and Composition of Essential Oil from Portuguese Myrtle (Myrtus comunis L.) through the Vegetative Cycle. Molecules 2009, 14, 3094–3105. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, F.; San, B.; Yildirim, A.N.; Polat, M.; Ercişli, S. Mineral Composition of Leaves and Fruit in Some Myrtle (Myrtus communis L.). Genotypes. Erwerbs Obstbau 2015, 57, 149–152. [Google Scholar] [CrossRef]
- Kuskoski, E.M.; Asuero, A.G.; Troncoso, A.M.; Mancini-Filho, J.; Fett, R. Aplicación de diversos métodos químicos para determinar actividad antioxidante en pulpa de frutos. Ciênc. Tecnol. Aliment. 2005, 25, 726–732. [Google Scholar] [CrossRef] [Green Version]
- Maldini, M.; Chessa, M.; Petretto, G.L.; Montoro, P.; Rourke, J.P.; Foddai, M.; Nicoletti, M.; Pintore, G. Profiling and Simultaneous Quantitative Determination of Anthocyanins in Wild Myrtus communis L. Berries from Different Geographical Areas in Sardinia and their Comparative Evaluation. Phytochem. Anal. 2016, 27, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Babou, L.; Hadidi, L.; Grosso, C.; Zaidi, F.; Valentão, P.; Andrade, P.B. Study of phenolic composition and antioxidant activity of myrtle leaves and fruits as a function of maturation. Eur. Food Res. Technol. 2016, 242, 1447–1457. [Google Scholar] [CrossRef]
- Tuberos, C.I.G.; Rosa, A.; Bifulco, E.; Melis, M.P.; Atseri, A.; Pirisi, F.M.; Dessi, M.A. Chemical composition and antioxidant activities of Myrtus communis L. berries extracts. Food Chem. 2010, 123, 1242–1251. [Google Scholar] [CrossRef]
- Scorrano, S.; Lazzoi, M.R.; Mergola, L.; Di Bello, M.P.D.; Del Sole, R.; Vasapollo, G. Anthocyanins Profile by Q-TOF LC/MS in Myrtus communis Berries from Salento Area. Food Anal. Methods 2017, 10, 2404–2411. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Melis, M.; Angioni, A.; Pala, M.; Cabras, P. Myrtle hydroalcoholic extracts obtained from different selections of Myrtus communis L. Food Chem. 2007, 101, 806–811. [Google Scholar] [CrossRef]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Messaoud, C.; Boussaid, M. Myrtus communis berry color morphs: A comparative analysis of essential oils, fatty acids, phenolic compounds, and antioxidant activities. Chem. Biodiv. 2011, 8, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Serreli, G.; Jerković, I.; Gil, K.A.; Marijanović, Z.; Pacini, V.; Tuberoso, C.I.G. Phenolic Compounds, Volatiles and Antioxidant Capacity of White Myrtle Berry Liqueurs. Plant Foods Hum. Nutr. 2017, 72, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Aidi Wannes, W.; Marzouk, B. Differences between myrtle fruit parts (Myrtus communis var. italica) in phenolics and antioxidant contents. J. Food Biochem. 2013, 37, 585–594. [Google Scholar] [CrossRef]
- Pereira, P.; Cebola, M.J.; Oliveira, M.C.; Bernardo-Gil, M.G. Supercritical fluid extraction vs conventional extraction of myrtle leaves and berries: Comparison of antioxidant activity and identification of bioactive compounds. J. Supercrit. Fluids. 2016, 113, 1–9. [Google Scholar] [CrossRef]
- Picó, Y. Ultrasound-assisted extraction for food and environmental samples. TrAC Trends Anal. Chem. 2013, 43, 84–99. [Google Scholar] [CrossRef]
- González de Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Jiménez-Cantizano, A.; Ferreiro-González, M.; Amores-Arrocha, A.; Barbero, G.F. Development of new analytical microwave-assisted extraction methods for bioactive compounds from myrtle (myrtus communis L.). Molecules 2018, 23, 2992. [Google Scholar] [CrossRef] [PubMed]
- Mason, T.J.; Paniwnyk, L.; Lorimer, K.P. The uses of ultrasound in food technology. Ultrason. Sonochem. 1996, 3, S253–S260. [Google Scholar] [CrossRef]
- Briones-Labarca, V.; Plaza-Morales, M.; Giovagnoli-Vicuña, C.; Jamett, F. High hydrostatic pressure and ultrasound extractions of antioxidant compounds, sulforaphane and fatty acids from Chilean papaya (Vasconcellea pubescens) seeds: Effects of extraction conditions and methods. LWT Food Sci. Technol. 2015, 60, 525–534. [Google Scholar] [CrossRef]
- Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of the ultrasound-assisted extraction of anthocyanins and total phenolic compounds in mulberry (Morus nigra) pulp. Food Chem. 2017, 219, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.K.; Abert-Vian, M.; Fabiano-Tixier, A.S.; Dangles, O.; Chemat, F. Ultrasound-assisted extraction of polyphenols (flavanone glycosides) from orange (Citrus sinensis L.) peel. Food Chem. 2010, 119, 851–858. [Google Scholar] [CrossRef]
- Feng, S.; Luo, Z.; Tao, B.; Chen, C. Ultrasonic-assisted extraction and purification of phenolic compounds from sugarcane (Saccharum officinarum L.) rind. LWT Food Sci. Technol. 2015, 60, 970–976. [Google Scholar] [CrossRef]
- Sarais, G.; D’Urso, G.; Lai, C.; Pirisi, F.M.; Pizza, C.; Montoro, P. Targeted and untargeted mass spectrometric approaches in discrimination between Myrtus communis cultivars from Sardinia region. J. Mass Spectrom. 2016, 51, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.; Cebola, M.J.; Oliveira, M.C.; Bernardo Gil, M.G. Antioxidant capacity and identification of bioactive compounds of Myrtus communis L. extract obtained by ultrasound-assisted extraction. J. Food Sci. Technol. 2017, 54, 4362–4369. [Google Scholar] [CrossRef] [PubMed]
- Bridgers, E.N.; Chinn, M.S.; Truong, V.D. Extraction of anthocyanins from industrial purple-fleshed sweetpotatoes and enzymatic hydrolysis of residues for fermentable sugars. Ind. Crops Prod. 2010, 32, 613–620. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Michiels, J.; Joris, A.Y.; Hosseini, M. Kolkhoung (Pistacia khinjuk) kernel oil quality is affected by different parameters in pulsed ultrasound-assisted solvent extraction. Ind. Crops Prod. 2015, 70, 28–33. [Google Scholar] [CrossRef]
- Kazemi, M.; Karim, R.; Mirhosseini, H.; Abdul Hamid, A. Optimization of pulsed ultrasound-assisted technique for extraction of phenolics from pomegranate peel of Malas variety: Punicalagin and hydroxybenzoic acids. Food Chem. 2016, 206, 156–166. [Google Scholar] [CrossRef] [PubMed]
- You, Q.; Yin, X.; Ji, C. Pulsed counter-current ultrasound-assisted extraction and characterization of polysaccharides from Boletus edulis. Carbohydr. Polym. 2014, 101, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.P.D.F.; Pereira, A.L.D.; Barbero, G.F.; Martínez, J. Recovery of anthocyanins from residues of Rubus fruticosus, Vaccinium myrtillus and Eugenia brasiliensis by ultrasound assisted extraction, pressurized liquid extraction and their combination. Food Chem. 2017, 231, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kidak, R.; Ince, N.H. Ultrasonic destruction of phenol and substituted phenols: A review of current research. Ultrason. Sonochem. 2006, 13, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.P.D.F.; Pasquel-Reátegui, J.L.; Barbero, G.F.; Martínez, J. Pressurized liquid extraction of bioactive compounds from blackberry (Rubus fruticosus L.) residues: A comparison with conventional methods. Food Res. Int. 2015, 77, 675–683. [Google Scholar] [CrossRef]
- Mustafa, A.; Turner, C. Pressurized liquid extraction as a green approach in food and herbal plants extraction: A review. Anal. Chim. Acta 2011, 703, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, R.N.; Santos, D.T.; Meireles, M.A.A. Non-thermal stabilization mechanisms of anthocyanins in model and food systems-An overview. Food Res. Int. 2011, 44, 499–509. [Google Scholar] [CrossRef]
- Carrera, C.; Ruiz-Rodriguez, A.; Palma, M.; Barroso, C.G. Ultrasound assisted extraction of phenolic compounds from grapes. Anal. Chim. Acta 2012, 732, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.T.V.; Tarone, A.G.; Cazarin, C.B.B.; Barbero, G.F.; Martínez, J. Pressurized liquid extraction of bioactive compounds from grape marc. J. Food Eng. 2019, 240, 105–113. [Google Scholar] [CrossRef]
- Tiwari, B.K.; O’Donnell, C.P.; Cullen, P.J. Effect of non thermal processing technologies on the anthocyanin content of fruit juices. Trends Food Sci. Technol. 2009, 20, 137–145. [Google Scholar] [CrossRef]
- Tiwari, B.K.; Patras, A.; Brunton, N.; Cullen, P.J.; O’Donnell, C.P. Effect of ultrasound processing on anthocyanins and color of red grape juice. Ultrason. Sonochem. 2009, 17, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Fleschhut, J.; Kratzer, F.; Rechkemmer, G.; Kulling, S.E. Stability and biotransformation of various dietary anthocyanins in vitro. Eur. J. Nutr. 2006, 45, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Stipcovich, T.; Barbero, G.F.; Ferreiro-González, M.; Palma, M.; Barroso, C.G. Fast analysis of capsaicinoids in Naga Jolokia extracts (Capsicum chinense) by high-performance liquid chromatography using fused core columns. Food Chem. 2018, 239, 217–224. [Google Scholar] [CrossRef] [PubMed]
- AOAC International (Ed.) AOAC Peer Verified Methods Program. In Manual on Policies and Procedures; AOAC International: Arlingt, MD, USA, 1998. [Google Scholar]
- González-Varo, J.P.; Albaladejo, R.G.; Aparicio, A. Mating patterns and spatial distribution of conspecific neighbours in the Mediterranean shrub Myrtus communis (Myrtaceae). Plant Ecol. 2009, 203, 207–215. [Google Scholar] [CrossRef]
- Mulas, M. Germplasm Evaluation of Spontaneous Myrtle (Myrtus communis L.) for Cultivar Selection and Crop Development. J. Herbs Spices Med. Plants 1999, 6, 31–49. [Google Scholar] [CrossRef]
- Ghafoor, K.; Choi, Y.H.; Jeon, J.Y.; Jo, I.H. Optimization of ultrasound- assisted extraction of phenolic compounds, antioxidants, and anthocyanins from grape (Vitis vinifera) seeds. J. Agric. Food Chem. 2009, 57, 4988–4994. [Google Scholar] [CrossRef] [PubMed]
- Casazza, A.; Aliakbarian, B.; Mantegna, S.; Cravotto, G.; Perego, P. Extraction of phenolics from Vitis vinifera wastes using non-conventional techniques. J. Food Eng. 2010, 100, 50–55. [Google Scholar] [CrossRef]
- Wijngaard, H.; Hossain, M.B.; Rai, D.K.; Brunton, N. Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 2012, 46, 505–513. [Google Scholar] [CrossRef]
- Tušek, A.J.; Benković, M.; Cvitanović, A.B.; Valinger, D.; Jurina, T.; Kljusurić, J.G. Kinetics and thermodynamics of the solid-liquid extraction process of total polyphenols, antioxidants and extraction yield from Asteraceae plants. Ind. Crops Prod. 2016, 91, 205–214. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Vallverdú-Queralt, A.; Medina-Remón, A.; Andres-Lacueva, C.; Lamuela-Raventos, R.M. Changes in phenolic profile and antioxidant activity during production of diced tomatoes. Food Chem. 2011, 126, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Skouroumounis, G.K.; Elsey, G.M.; Taylor, D.K. Microwave-assistance provides very rapid and efficient extraction of grape seed polyphenols. Food Chem. 2011, 129, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Batista, Â.G.; Ferrari, A.S.; Da Cunha, D.C.; da Silva, J.K.; Cazarin, C.B.B.; Correa, L.C.M.; Prado, M.A.; de Carvalho-Silva, L.B.; Esteves, E.A.; Junior, M.R.M. Polyphenols, antioxidants, and antimutagenic effects of Copaifera langsdorffii fruit. Food Chem. 2016, 197, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Gan, R.-Y.; Zhou, T.; Xu, D.-P.; Li, H.-B. Optimization of Ultrasound-Assisted Extraction of Antioxidants from the Mung Bean Coat. Molecules 2017, 22, 638. [Google Scholar] [CrossRef] [PubMed]
- Maran, J.; Manikandan, S.; Thirugnanasambandham, K.; Vigna Nivetha, C.; Dinesh, R. Box-Behnken design based statistical modeling for ultrasound-assisted extraction of corn silk polysaccharide. Carbohydr. Polym. 2013, 92, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.L.C.; Bruns, R.E.; Ferreira, H.S.; Matos, G.D.; David, J.M.; Brandao, G.C.; da Silva, E.G.P.; Portugal, L.A.; dos Reis, P.S.; Souza, A.S.; et al. Box-Behnken design: An alternative for the optimization of analytical methods. Anal. Chim. Acta 2007, 597, 179–186. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples with the compounds delphinidin 3,5-O-diglucoside, delphinidin 3-O-glucoside, cyanidin 3-O-galactoside, cyanidin 3-O-glucoside, cyanidin 3-O-arabinoside, petunidin 3-O-glucoside, delphinidin 3-O-arabinoside, peonidin 3-O-glucoside, malvidin 3-O-glucoside, petunidin 3-O-arabinoside and malvidin 3-O-arabinoside are available from the authors. |


| Variables | Studied Ranges | |
|---|---|---|
| Phenolic Compounds | Anthocyanins | |
| Temperature (°C) | 10, 35, 60 | 10, 35, 60 |
| Amplitude (%) | 30, 50, 70 | 30, 50, 70 |
| Cycle (s) | 0.2, 0.45, 0.7 | 0.2, 0.45, 0.7 |
| pH | 2, 4.5, 7 | 2, 4.5, 7 |
| Solvent-sample ratio (mL/0.5 g) | 10, 15, 20 | 10, 15, 20 |
| Solvent composition (% methanol in water) | 50, 75, 100 | 25, 50, 75 |
| Source | Coefficient | Sum of Squares | Degrees of Freedom | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|---|
| Model | 2940.04 | 27 | 108.89 | 2.31 | 0.0179 | ||
| Methanol | X1 | −1.15 | 31.97 | 1 | 31.97 | 0.6787 | 0.4175 |
| Temperature | X2 | −1.01 | 24.50 | 1 | 24.50 | 0.5202 | 0.4772 |
| Amplitude | X3 | 0.0166 | 0.0066 | 1 | 0.0066 | 0.0001 | 0.9906 |
| Cycle | X4 | −4.17 | 418.01 | 1 | 418.01 | 8.88 | 0.0062 |
| pH | X5 | −0.8371 | 16.82 | 1 | 16.82 | 0.3571 | 0.5553 |
| Ratio | X6 | 0.5498 | 7.26 | 1 | 7.26 | 0.1541 | 0.6979 |
| Methanol × Temperature | X1X2 | 6.75 | 364.53 | 1 | 364.53 | 7.74 | 0.0099 |
| Methanol × Amplitude | X1X3 | 1.01 | 8.14 | 1 | 8.14 | 0.1729 | 0.6809 |
| Methanol × Cycle | X1X4 | −2.00 | 63.93 | 1 | 63.93 | 1.36 | 0.2545 |
| Methanol × pH | X1X5 | 4.38 | 153.56 | 1 | 153.56 | 3.26 | 0.0826 |
| Methanol × Ratio | X1X6 | −0.7961 | 5.07 | 1 | 5.07 | 0.1077 | 0.7454 |
| Temperature × Amplitude | X2X3 | −0.7882 | 4.97 | 1 | 4.97 | 0.1055 | 0.7479 |
| Temperature × Cycle | X2X4 | 0.6373 | 3.25 | 1 | 3.25 | 0.0690 | 0.7949 |
| Temperature × pH | X2X5 | 2.87 | 132.23 | 1 | 132.23 | 2.81 | 0.1058 |
| Temperature × Ratio | X2X6 | −2.36 | 44.72 | 1 | 44.72 | 0.9495 | 0.3388 |
| Amplitude × Cycle | X3X4 | −4.26 | 144.96 | 1 | 144.96 | 3.08 | 0.0911 |
| Amplitude × pH | X3X5 | −1.14 | 10.47 | 1 | 10.47 | 0.2222 | 0.6413 |
| Amplitude × Ratio | X3X6 | 1.87 | 56.01 | 1 | 56.01 | 1.19 | 0.2855 |
| Cycle × pH | X4X5 | −2.64 | 55.80 | 1 | 55.80 | 1.18 | 0.2863 |
| Cycle × Ratio | X4X6 | 6.71 | 359.66 | 1 | 359.66 | 7.64 | 0.0104 |
| pH × Ratio | X5X6 | 1.68 | 22.66 | 1 | 22.66 | 0.4812 | 0.4940 |
| Methanol × Methanol | X12 | −8.06 | 668.48 | 1 | 668.48 | 14.19 | 0.0009 |
| Temperature × Temperature | X22 | −0.2020 | 0.4197 | 1 | 0.4197 | 0.0089 | 0.9255 |
| Amplitude × Amplitude | X32 | 1.79 | 32.81 | 1 | 32.81 | 0.6967 | 0.4115 |
| Cycle × Cycle | X42 | 1.87 | 36.12 | 1 | 36.12 | 0.7669 | 0.3892 |
| pH × pH | X52 | 2.96 | 90.01 | 1 | 90.01 | 1.91 | 0.1786 |
| Ratio × Ratio | X62 | −2.20 | 49.57 | 1 | 49.57 | 1.05 | 0.3144 |
| Residual | 45.85 | 1224.48 | 26 | 47.10 | |||
| Lack of Fit | 1075.70 | 21 | 51.22 | 1.72 | 0.2858 | ||
| Pure Error | 148.78 | 5 | 29.76 | ||||
| Total | 4164.52 | 53 |
| Source | Coefficient | Sum of Squares | Degrees of Squares | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|---|
| Model | 2514.45 | 27 | 93.13 | 1.47 | 0.1631 | ||
| Methanol | X1 | 4.80 | 553.02 | 1 | 553.02 | 8.75 | 0.0065 |
| Temperature | X2 | −2.05 | 100.49 | 1 | 100.49 | 1.59 | 0.2185 |
| Amplitude | X3 | −1.88 | 85.15 | 1 | 85.15 | 1.35 | 0.2563 |
| Cycle | X4 | −2.47 | 146.43 | 1 | 146.43 | 2.32 | 0.1400 |
| pH | X5 | 1.13 | 30.92 | 1 | 30.92 | 0.4892 | 0.4905 |
| Ratio | X6 | −0.1120 | 0.3008 | 1 | 0.3008 | 0.0048 | 0.9455 |
| Methanol × Temperature | X1X2 | −5.59 | 250.28 | 1 | 250.28 | 3.96 | 0.0572 |
| Methanol × Amplitude | X1X3 | −4.68 | 174.94 | 1 | 174.94 | 2.77 | 0.1082 |
| Methanol × Cycle | X1X4 | −2.18 | 76.24 | 1 | 76.24 | 1.21 | 0.2821 |
| Methanol × pH | X1X5 | 6.51 | 338.73 | 1 | 338.73 | 5.36 | 0.0288 |
| Methanol × Ratio | X1X6 | 0.0864 | 0.0598 | 1 | 0.0598 | 0.0009 | 0.9757 |
| Temperature × Amplitude | X2X3 | −0.1454 | 0.1691 | 1 | 0.1691 | 0.0027 | 0.9591 |
| Temperature × Cycle | X2X4 | −1.16 | 10.77 | 1 | 10.77 | 0.1704 | 0.6831 |
| Temperature × pH | X2X5 | −3.14 | 157.76 | 1 | 157.76 | 2.50 | 0.1262 |
| Temperature × Ratio | X2X6 | 0.8116 | 5.27 | 1 | 5.27 | 0.0834 | 0.7751 |
| Amplitude × Cycle | X3X4 | 3.73 | 111.16 | 1 | 111.16 | 1.76 | 0.1963 |
| Amplitude × pH | X3X5 | −1.63 | 21.28 | 1 | 21.28 | 0.3368 | 0.5667 |
| Amplitude × Ratio | X3X6 | −1.04 | 17.31 | 1 | 17.31 | 0.2739 | 0.6052 |
| Cycle × pH | X4X5 | −0.7232 | 4.18 | 1 | 4.18 | 0.0662 | 0.7990 |
| Cycle × Ratio | X4X6 | 0.2305 | 0.4249 | 1 | 0.4249 | 0.0067 | 0.9353 |
| pH × Ratio | X5X6 | −0.9561 | 7.31 | 1 | 7.31 | 0.1157 | 0.7365 |
| Methanol × Methanol | X12 | −1.95 | 39.12 | 1 | 39.12 | 0.6191 | 0.4385 |
| Temperature × Temperature | X22 | 2.23 | 50.98 | 1 | 50.98 | 0.8068 | 0.3773 |
| Amplitude × Amplitude | X32 | 2.89 | 86.14 | 1 | 86.14 | 1.36 | 0.2536 |
| Cycle × Cycle | X42 | −1.17 | 14.04 | 1 | 14.04 | 0.2221 | 0.6414 |
| pH × pH | X52 | 2.03 | 42.24 | 1 | 42.24 | 0.6683 | 0.4211 |
| Ratio × Ratio | X62 | −1.64 | 27.72 | 1 | 27.72 | 0.4386 | 0.5136 |
| Residual | 23.34 | 1643.02 | 26 | 63.19 | |||
| Lack of Fit | 1469.28 | 21 | 69.97 | 2.01 | 0.2248 | ||
| Pure Error | 173.75 | 5 | 34.75 | ||||
| Total | 4157.47 | 53 |
| Compounds 1 | Myrtle ecotypes from Puerto Real | Myrtle ecotypes from San José del Valle | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MY-1 | MY-2 | MY-3 | MY-4 | MY-5 | MY-6 | MY-7 | MY-8 | MY-9 | MY-10 | MY-11 | MY-12 | MY-13 | MY-14 | |
| D-3,5-diGl | 0.166 ± 0.006 | 0.415 ± 0.016 | 0.404 ± 0.015 | 0.504 ± 0.020 | 0.377 ± 0.014 | 0.375 ± 0.016 | 0.183 ± 0.008 | 0.103 ± 0.0034 | 0.127 ± 0.004 | 0.065 ± 0.002 | 0.079 ± 0.0023 | 0.176 ± 0.0067 | 0.113 ± 0.003 | 0.134 ± 0.004 |
| Del-3-Glu | 10.552 ± 0.161 | 16.309 ± 0.534 | 10.305 ± 0.358 | 11.114 ± 0.384 | 12.557 ± 0.481 | 12.258 ± 0.432 | 8.315 ± 0.326 | 5.929 ± 0.165 | 3.667 ± 0.152 | 1.566 ± 0.008 | 2.145 ± 0.084 | 3.823 ± 0.124 | 2.987 ± 0.121 | 3.346 ± 0.135 |
| Cy-3-Ga | 0.150 ± 0.006 | 0.382 ± 0.008 | 0.216 ± 0.009 | 0.359 ± 0.007 | 0.319 ± 0.012 | 0.371 ± 0.0135 | 0.176 ± 0.007 | 0.392 ± 0.012 | 0.050 ± 0.001 | 0.052 ± 0.002 | 0.068 ± 0.0018 | 0.1757 ± 0.005 | 0.077 ± 0.002 | 0.036 ± 0.001 |
| Cy-3-Gl | 1.794 ± 0.084 | 3.004 ± 0.15 | 1.126 ± 0.023 | 1.241 ± 0.036 | 1.613 ± 0.03 | 1.313 ± 0.0578 | 1.946 ± 0.034 | 0.791 ± 0.043 | 0.852 ± 0.024 | 0.293 ± 0.01 | 0.301 ± 0.016 | 0.512 ± 0.025 | 0.452 ± 0.013 | 0.786 ± 0.032 |
| Cy-3-Ar | 0.092 ± 0.003 | 0.126 ± 0.002 | 0.076 ± 0.002 | 0.130 ± 0.002 | 0.112 ± 0.004 | 0.170 ± 0.006 | 0.061 ± 0.14 | 0.081 ± 0.0025 | 0.170 ± 0.003 | 0.040 ± 0.001 | 0.410 ± 0.021 | 0.657 ± 0.027 | 0.549 ± 0.23 | 0.188 ± 0.006 |
| Pet-3-Gl | 6.217 ± 0.110 | 11.007 ± 0.38 | 7.173 ± 0.314 | 8.691 ± 0.301 | 8.329 ± 0.297 | 10.242 ± 0.392 | 5.538 ± 0.0423 | 5.082 ± 0.176 | 6.810 ± 0.235 | 1.580 ± 0.018 | 1.676 ± 0.065 | 2.123 ± 0.085 | 1.823 ± 0.067 | 6.643 ± 0.114 |
| Del-3-Ara | 0.840 ± 0.005 | 2.378 ± 0.102 | 1.635 ± 0.062 | 1.775 ± 0.067 | 1.917 ± 0.064 | 2.009 ± 0.081 | 1.335 ± 0.021 | 1.562 ± 0.035 | 1.833 ± 0.076 | 0.367 ± 0.006 | 0.324 ± 0.009 | 0.679 ± 0.021 | 0.454 ± 0.014 | 1.234 ± 0.033 |
| Peo-3-Gl | 1.030 ± 0.017 | 1.179 ± 0.043 | 0.659 ± 0.019 | 0.689 ± 0.021 | 0.610 ± 0.023 | 0.816 ± 0.029 | 0.755 ± 0.34 | 0.481 ± 0.019 | 0.473 ± 0.015 | 0.305 ± 0.017 | 0.357 ± 0.012 | 0.921 ± 0.032 | 0.564 ± 0.013 | 0.446 ± 0.012 |
| Mal-3-Gl | 9.834 ± 0.314 | 16.691 ± 0.641 | 16.050 ± 0.65 | 18.301 ± 0.614 | 12.748 ± 0.768 | 23.817 ± 1.032 | 8.244 ± 0.273 | 9.072 ± 0.348 | 6.987 ± 0.246 | 5.195 ± 0.068 | 5.453 ± 0.185 | 7.679 ± 0.23 | 6.456 ± 0.241 | 5.979 ± 0.263 |
| Pet-3-Ar | 0.269 ± 0.009 | 0.551 ± 0.034 | 0.448 ± 0.014 | 0.587 ± 0.013 | 0.482 ± 0.028 | 0.751 ± 0.0013 | 0.332 ± 0.012 | 0.583 ± 0.015 | 0.438 ± 0.017 | 0.197 ± 0.002 | 0.206 ± 0.008 | 0.657 ± 0.023 | 0.454 ± 0.012 | 0.446 ± 0.012 |
| Mal-3-Ar | 0.226 ± 0.004 | 0.302 ± 0.009 | 0.402 ± 0.012 | 0.436 ± 0.121 | 0.296 ± 0.015 | 0.598 ± 0.019 | 0.240 ± 0.009 | 0.395 ± 0.014 | 0.185 ± 0.005 | 0.221 ± 0.001 | 0.244 ± 0.012 | 0.846 ± 0.032 | 0.679 ± 0.031 | 0.165 ± 0.006 |
| Total anthocyanins | 31.170 ± 1.023 | 52.346 ± 1.124 | 38.493 ± 3.516 | 43.827 ± 2.557 | 43.274 ± 1.904 | 60.252 ± 0.002 | 35.207 ± 1.175 | 32.903 ± 1.162 | 21.591 ± 0.92 | 10.381 ± 0.037 | 11.263 ± 0.431 | 18.249 ± 2.30 | 14.608 ± 1.964 | 19.403 ± 1.97 |
| Total Phenolic compounds | 70.747 ± 1.433 | 86.439 ± 3.125 | 64.7586 ± 0.914 | 60.034 ± 0.44 | 72.155 ± 3.174 | 76.124 ± 1.170 | 76.070 ± 3.417 | 69.815 ± 2.947 | 49.824 ± 0.362 | 52.66 ± 0.038 | 45.789 ± 1.824 | 51.278 ± 2.051 | 49.103 ± 0.192 | 50.897 ± 1.564 |
| Run | Factors | Responses | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X4 | X5 | X6 | YTP (mg·g−1) | YTA (mg·g−1) | |||
| Experimental | Predicted | Experimental | Predicted | |||||||
| 1 | 0 | 0 | −1 | 0 | −1 | −1 | 55.4367 | 51.0831 | 20.7595 | 23.8541 |
| 2 | 0 | 0 | 1 | 0 | −1 | −1 | 48.6499 | 49.6621 | 29.8821 | 25.4294 |
| 3 | 0 | 0 | −1 | 0 | 1 | −1 | 51.4343 | 48.3304 | 29.0606 | 31.2985 |
| 4 | 0 | 0 | 1 | 0 | 1 | −1 | 41.6667 | 42.334 | 27.9193 | 26.3493 |
| 5 | 0 | 0 | −1 | 0 | −1 | 1 | 46.1649 | 45.0747 | 27.6777 | 27.6226 |
| 6 | 0 | 0 | 1 | 0 | −1 | 1 | 47.6107 | 51.1375 | 25.6501 | 25.0375 |
| 7 | 0 | 0 | −1 | 0 | 1 | 1 | 50.4893 | 49.0542 | 28.4150 | 31.2426 |
| 8 | 0 | 0 | 1 | 0 | 1 | 1 | 45.7652 | 50.5417 | 23.6023 | 22.1329 |
| 9 | 0 | −1 | 0 | −1 | −1 | 0 | 57.1168 | 57.3752 | 28.5256 | 24.7846 |
| 10 | 0 | 1 | 0 | −1 | −1 | 0 | 44.7735 | 48.3304 | 26.8602 | 29.2928 |
| 11 | 0 | −1 | 0 | 1 | −1 | 0 | 41.9739 | 53.0361 | 27.755 | 23.6113 |
| 12 | 0 | 1 | 0 | 1 | −1 | 0 | 41.3703 | 46.5404 | 26.6112 | 23.4784 |
| 13 | 0 | −1 | 0 | −1 | 1 | 0 | 57.8351 | 55.2335 | 27.0548 | 34.781 |
| 14 | 0 | 1 | 0 | −1 | 1 | 0 | 71.3186 | 57.688 | 27.1787 | 26.729 |
| 15 | 0 | −1 | 0 | 1 | 1 | 0 | 41.3184 | 40.33 | 28.5542 | 30.715 |
| 16 | 0 | 1 | 0 | 1 | 1 | 0 | 48.1604 | 45.3335 | 18.8744 | 18.0219 |
| 17 | −1 | 0 | −1 | −1 | 0 | 0 | 37.8811 | 41.5155 | 14.3354 | 19.5394 |
| 18 | 1 | 0 | −1 | −1 | 0 | 0 | 39.2436 | 41.1874 | 19.2532 | 42.8583 |
| 19 | −1 | 0 | 1 | −1 | 0 | 0 | 48.7908 | 48.0444 | 18.0553 | 17.6695 |
| 20 | 1 | 0 | 1 | −1 | 0 | 0 | 43.5972 | 51.7521 | 21.1111 | 22.2834 |
| 21 | −1 | 0 | −1 | 1 | 0 | 0 | 54.6703 | 45.6802 | 17.6946 | 11.5099 |
| 22 | 1 | 0 | −1 | 1 | 0 | 0 | 35.7746 | 37.3562 | 20.6987 | 26.097 |
| 23 | −1 | 0 | 1 | 1 | 0 | 0 | 37.9611 | 35.1821 | 10.1680 | 24.5504 |
| 24 | 1 | 0 | 1 | 1 | 0 | 0 | 33.6931 | 30.8939 | 20.6241 | 20.4326 |
| 25 | 0 | −1 | −1 | 0 | 0 | −1 | 39.6751 | 44.4043 | 25.5771 | 30.4887 |
| 26 | 0 | 1 | −1 | 0 | 0 | −1 | 42.5789 | 48.6886 | 26.0578 | 25.0639 |
| 27 | 0 | −1 | 1 | 0 | 0 | −1 | 43.7849 | 42.272 | 27.3046 | 29.0925 |
| 28 | 0 | 1 | 1 | 0 | 0 | −1 | 42.0191 | 43.4037 | 24.5787 | 23.0862 |
| 29 | 0 | −1 | −1 | 0 | 0 | 1 | 47.4523 | 46.4907 | 27.6042 | 30.7219 |
| 30 | 0 | 1 | −1 | 0 | 0 | 1 | 39.3819 | 41.3177 | 28.7060 | 28.5433 |
| 31 | 0 | −1 | 1 | 0 | 0 | 1 | 58.3748 | 51.8422 | 25.7965 | 25.1653 |
| 32 | 0 | 1 | 1 | 0 | 0 | 1 | 48.6688 | 43.5167 | 28.9420 | 22.4052 |
| 33 | −1 | −1 | 0 | 0 | −1 | 0 | 58.0268 | 57.5556 | 22.9897 | 19.5292 |
| 34 | 1 | −1 | 0 | 0 | −1 | 0 | 41.2425 | 32.9846 | 19.5373 | 27.3024 |
| 35 | −1 | 1 | 0 | 0 | −1 | 0 | 38.5507 | 36.2848 | 24.6490 | 32.9035 |
| 36 | 1 | 1 | 0 | 0 | −1 | 0 | 46.8626 | 38.7149 | 20.2517 | 18.3034 |
| 37 | −1 | −1 | 0 | 0 | 1 | 0 | 35.7902 | 41.3694 | 17.7105 | 15.0653 |
| 38 | 1 | −1 | 0 | 0 | 1 | 0 | 34.6257 | 34.3231 | 17.7143 | 48.8664 |
| 39 | −1 | 1 | 0 | 0 | 1 | 0 | 20.7714 | 31.5978 | 19.0511 | 15.8794 |
| 40 | 1 | 1 | 0 | 0 | 1 | 0 | 48.5130 | 51.5526 | 19.2534 | 27.3073 |
| 41 | −1 | 0 | 0 | −1 | 0 | −1 | 49.3362 | 46.1577 | 18.9945 | 14.4979 |
| 42 | 1 | 0 | 0 | −1 | 0 | −1 | 49.0123 | 49.4397 | 21.8380 | 28.2914 |
| 43 | −1 | 0 | 0 | 1 | 0 | −1 | 30.0802 | 28.3987 | 16.72389 | 13.4627 |
| 44 | 1 | 0 | 0 | 1 | 0 | −1 | 24.1848 | 23.6849 | 20.7428 | 18.5244 |
| 45 | −1 | 0 | 0 | −1 | 0 | 1 | 35.7748 | 35.4395 | 16.4343 | 13.6402 |
| 46 | 1 | 0 | 0 | −1 | 0 | 1 | 33.0203 | 35.537 | 19.5057 | 27.7794 |
| 47 | −1 | 0 | 0 | 1 | 0 | 1 | 44.0929 | 44.5007 | 14.9678 | 13.5268 |
| 48 | 1 | 0 | 0 | 1 | 0 | 1 | 34.2591 | 36.6024 | 19.4501 | 18.9343 |
| 49 | 0 | 0 | 0 | 0 | 0 | 0 | 37.2240 | 45.8533 | 27.1667 | 23.3422 |
| 50 | 0 | 0 | 0 | 0 | 0 | 0 | 52.1910 | 45.8533 | 27.2028 | 23.3422 |
| 51 | 0 | 0 | 0 | 0 | 0 | 0 | 47.5538 | 45.8533 | 17.7051 | 23.3422 |
| 52 | 0 | 0 | 0 | 0 | 0 | 0 | 50.5994 | 45.8533 | 14.0658 | 23.3422 |
| 53 | 0 | 0 | 0 | 0 | 0 | 0 | 43.5576 | 45.8533 | 26.5943 | 23.3422 |
| 54 | 0 | 0 | 0 | 0 | 0 | 0 | 43.9939 | 45.8533 | 27.3184 | 23.3422 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
V. González de Peredo, A.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Ferreiro-González, M.; Amores-Arrocha, A.; Palma, M.; F. Barbero, G.; Jiménez-Cantizano, A. Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.). Molecules 2019, 24, 882. https://doi.org/10.3390/molecules24050882
V. González de Peredo A, Vázquez-Espinosa M, Espada-Bellido E, Ferreiro-González M, Amores-Arrocha A, Palma M, F. Barbero G, Jiménez-Cantizano A. Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.). Molecules. 2019; 24(5):882. https://doi.org/10.3390/molecules24050882
Chicago/Turabian StyleV. González de Peredo, Ana, Mercedes Vázquez-Espinosa, Estrella Espada-Bellido, Marta Ferreiro-González, Antonio Amores-Arrocha, Miguel Palma, Gerardo F. Barbero, and Ana Jiménez-Cantizano. 2019. "Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.)" Molecules 24, no. 5: 882. https://doi.org/10.3390/molecules24050882
APA StyleV. González de Peredo, A., Vázquez-Espinosa, M., Espada-Bellido, E., Ferreiro-González, M., Amores-Arrocha, A., Palma, M., F. Barbero, G., & Jiménez-Cantizano, A. (2019). Alternative Ultrasound-Assisted Method for the Extraction of the Bioactive Compounds Present in Myrtle (Myrtus communis L.). Molecules, 24(5), 882. https://doi.org/10.3390/molecules24050882