Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease
Abstract
1. Introduction
2. Alzheimer’s Disease
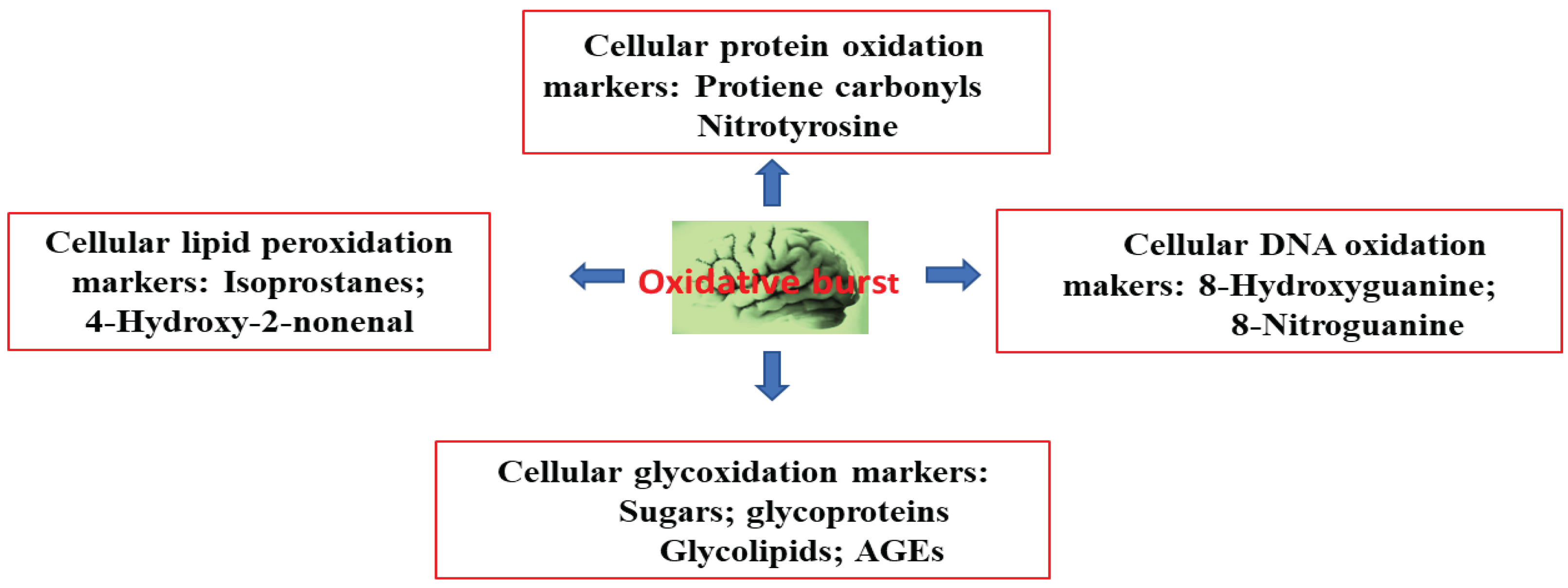
3. Alzheimer’s Disease and Oxidative Stress
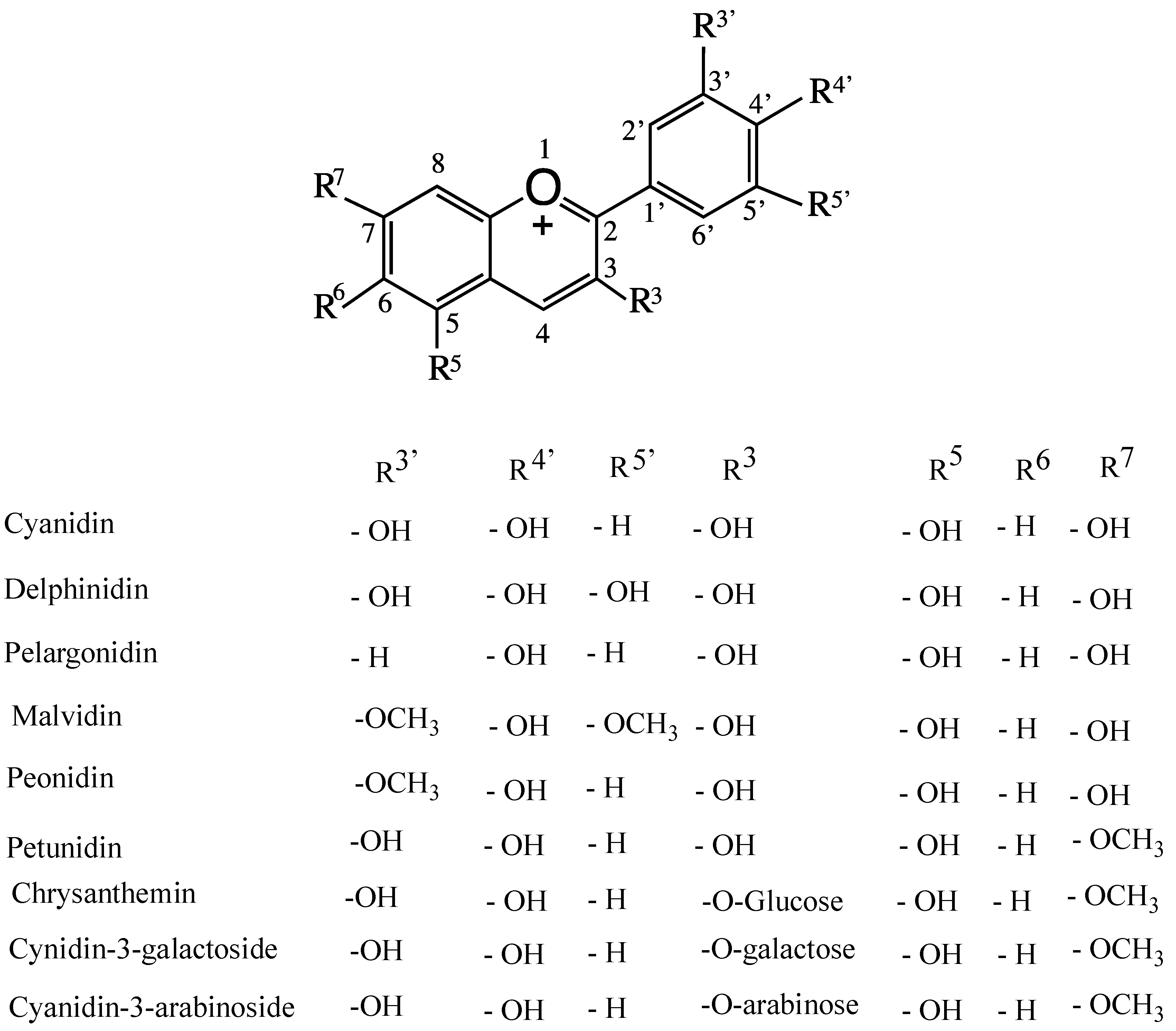
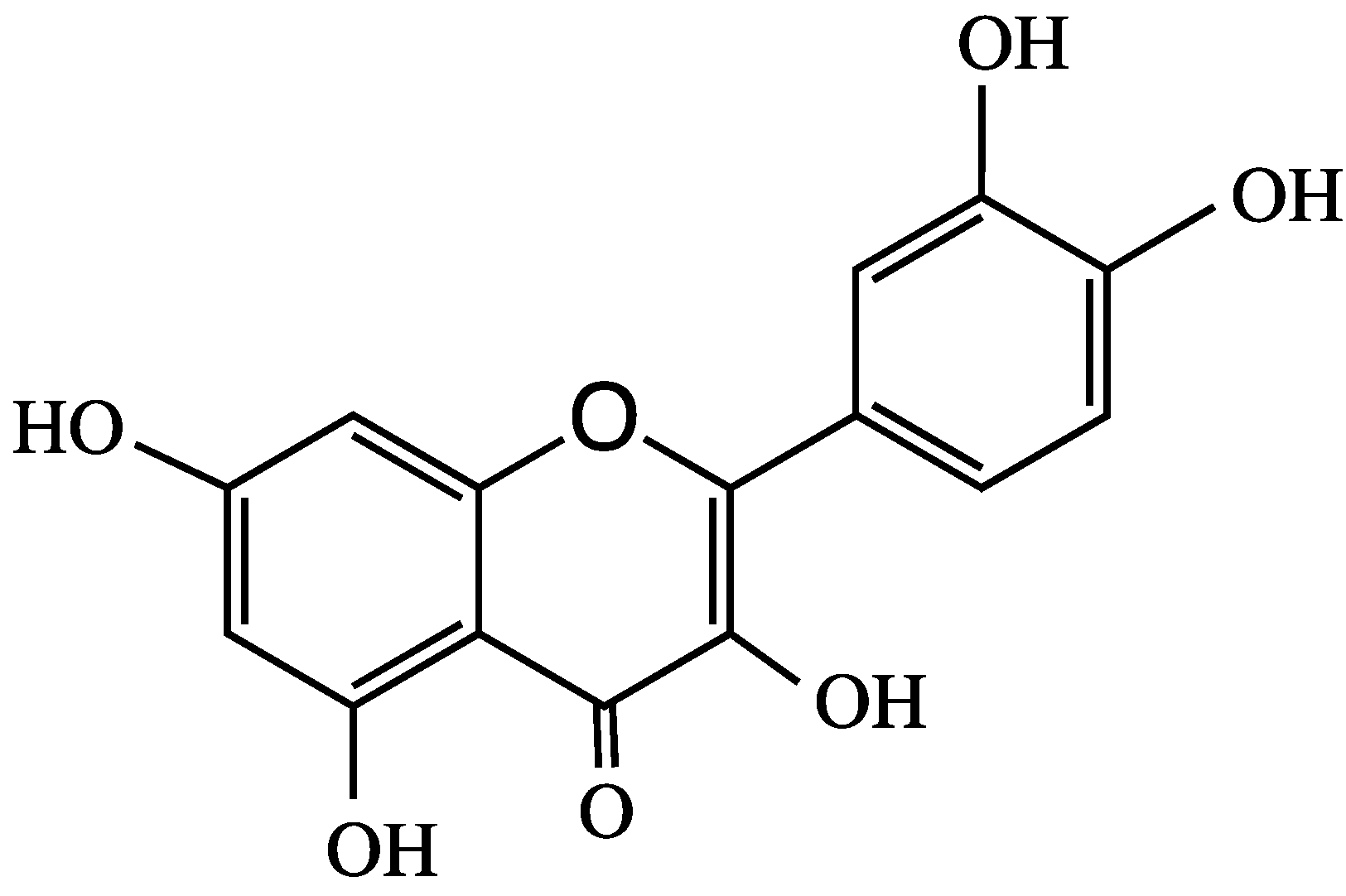
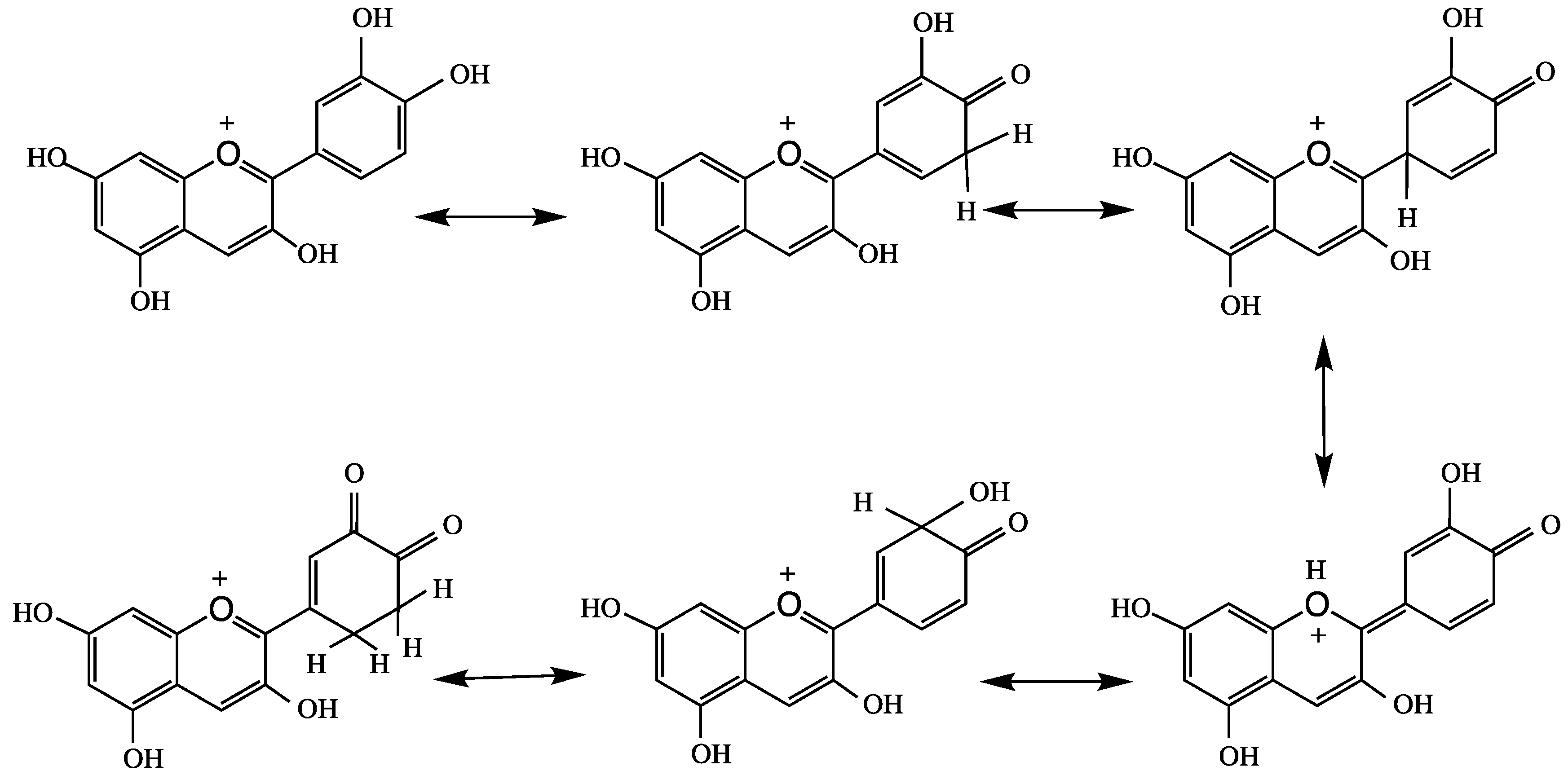
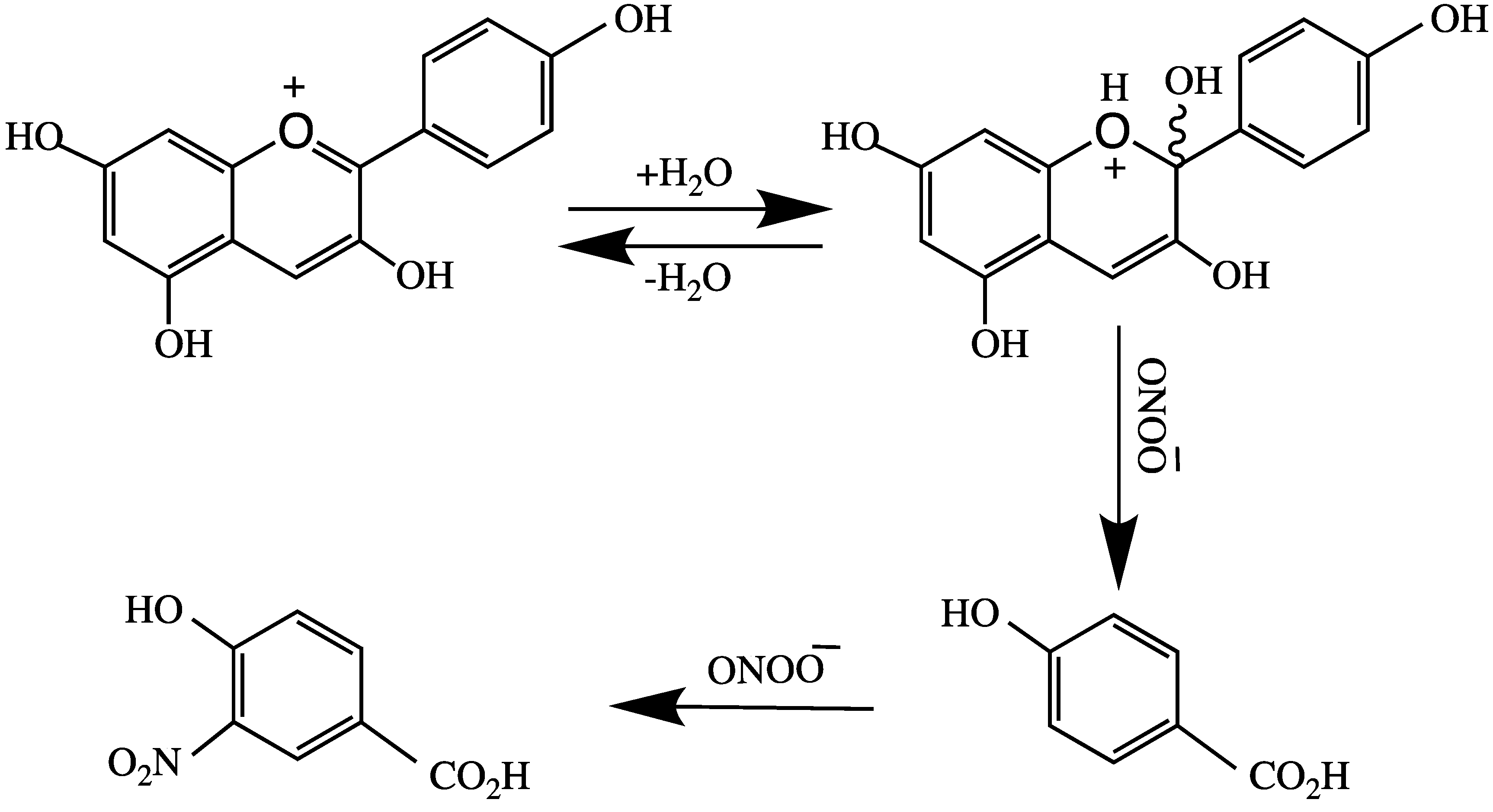
4. Anthocyanins and their Antioxidant Activity
5. Anthocyanin Absorption, Bioavailability, and Metabolism
6. Alzheimer’s Disease and Anthocyanins
7. Anthocyanin Nanoparticles
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, X.; Prior, R.L. Identification and characterization of anthocyanins by HPLC-ESI-MS/MS in common foods in the United States: Vegetables, nuts and grains. J. Agric. Food Chem. 2005, 53, 3101–3113. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.G. Anthocyanins: Antioxidants and/or anti-inflammatory activities. J. Appl. Pharm. Sci. 2011, 1, 7–15. [Google Scholar]
- Castañeda-Ovando, A.; Pacheco-Hernández, M.L.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Wallace, T.C. Anthocyanins in cardiovascular disease. Adv. Nutr. 2011, 2, 1–7. [Google Scholar] [CrossRef]
- Ho, L.; Ferruzzi, M.G.; Janle, E.M.; Wang, J.; Gong, B.; Chen, T.Y.; Lobo, J.; Cooper, B.; Wu, Q.L.; Talcott, S.T.; et al. Identification of brain-targeted bioactive dietary quercetin-3-O-glucuronide as a novel intervention for Alzheimer’s disease. FASEB J. 2013, 27, 769–781. [Google Scholar] [CrossRef]
- Shih, P.H.; Wu, C.H.; Yeh, C.T.; Yen, G.C. Protective effects of anthocyanins against amyloid beta-peptide-induced damage in neuro-2A cells. J. Agric. Food Chem. 2011, 59, 1683–1689. [Google Scholar] [CrossRef]
- Roy, H.J.; Lundy, S.; Eriksen, C.; Kalicki, B. Anthocyanins. Pennington Nutrition Series. 2009. Available online: http://www.pbrc.edu/training-and-education/pdf/pns/pns_anthocyanins.pdf (accessed on 22 November 2019).
- Lee, Y.M.; Yoon, Y.; Yoon, H.; Park, H.M.; Song, S.; Yeum, K.J. Dietary anthocyanins against obesity and inflammation. Nutrients 2017, 9, 1089. [Google Scholar] [CrossRef]
- Nanashima, N.; Horie, K.; Chiba, M.; Nakano, M.; Maeda, H.; Nakamura, T. Anthocyanin rich blackcurrant extract inhibits proliferation of the MCF10A healthy human breast epithelial cell line through induction of G0/G1 arrest and apoptosis. Mol. Med. Rep. 2017, 16, 6134–6141. [Google Scholar] [CrossRef]
- Nanashima, N.; Horei, K. Blackcurrent extract with phytoestrogen activity alleviates hair loss in ovariectomized rats. Molecules 2019, 24, 1272. [Google Scholar] [CrossRef]
- Shaw, O.M.; Nyanhanda, T.; McGhie, T.K.; Harper, J.L.; Hurst, R.D. Blackcurrant anthocyanins modulate CCL11 secretion and suppress allergic airway inflammation. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Serrano, A.; Ros, G.; Nieto, G. Bioactive compounds and extracts from traditional herbs and their potential anti-inflammatory health effects. Medicines 2018, 5, 76. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Strikoudi, P.; Spel, M.E.; Parpinel, M.; Serraino, D.; Montella, M.; Libra, M.; La Vecchia, C.; Rosato, V. Flavonoids and bladder cancer risk. Cancer Causes Control 2019, 30, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Gowd, V.; Bao, T.; Wang, L.; Huang, Y.; Chen, S.; Zheng, X.; Cui, S.; Chen, W. Antioxidant and antidiabetic activity of blackberry after gastrointestinal digestion and human gut microbiota fermentation. Food Chem. 2018, 269, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Baby, B.; Antony, P.; Vijayan, R. Antioxidant and anticancer properties of berries. Crit. Rev. Food Sci. Nutr. 2018, 58, 2491–2507. [Google Scholar] [CrossRef]
- Kaume, L.; Howard, L.R.; Devareddy, L. The Blackberry fruit: A review on its composition and chemistry, metabolism and bioavailability, and health benefits. J. Agric. Food Chem. 2012, 60, 5716–5727. [Google Scholar] [CrossRef] [PubMed]
- Kosmala, M.; Jurgoński, A.; Juśkiewicz, J.; Karlińska, E.; Macierzyński, J.; Rój, E.; Zduńczyk, Z. Chemical composition of Blackberry press cake, polyphenolic extract, and defatted seeds, and their effects on cecal fermentation, bacterial metabolites, and blood lipid profile in rats. J. Agric. Food Chem. 2017, 65, 5470–5479. [Google Scholar] [CrossRef] [PubMed]
- Kolniak-Ostek, J.; Alicja, Z.K.; Anna, S.L.; Izabela, F. Characterization of Phenolic Compounds of Thorny and Thorn less Blackberries. J. Agric. Food Chem. 2015, 63, 3012–3021. [Google Scholar] [CrossRef]
- de Gomes, M.G.; Del Fabbro, L.; Rossito, G.A.T.; Souza, L.C.; Donato, F.; Boeira, S.P.; Prigol, M.; Jesse, C.R. Blackberry juice anthocyanidins limit cisplatin-induced renal pathophysiology in mice. Pathophysiology 2019. [Google Scholar] [CrossRef]
- Gutierrez-Albanchez, E.; Kirakosyan, A.; Bolling, S.F.; Garcia-Villaraco, A.; Gutierrez-Manero, J.; Ramos-Solano, B. Biotic elicitation as a tool to improve strawberry and raspberry extract potential on metabolic syndrome-related enzymes in vitro. J. Sci. Food Agric. 2019, 99, 2939–2946. [Google Scholar] [CrossRef]
- Fernández-Demeneghi, R.; Rodríguez-Landa, J.F.; Guzmán-Gerónimo, R.I.; Acosta-Mesa, H.G.; Meza-Alvarado, E.; Vargas-Moreno, I.; Herrera-Meza, S. Effect of blackberry juice (Rubus fruticosus L.) on anxiety-like behavior in Wistar rats. Int. J. Food Sci. Nutr. 2019, 195, 1–12. [Google Scholar]
- Day, A.J.; Rothwell, J.A.; Morgan, R. Characterization of polyphenol metabolites. In Phytochemicals in Health and Disease; Bao, Y., Fenwick, R., Eds.; Dekker: New York, NY, USA, 2004; pp. 50–67. ISBN 0-8247-4023-8. [Google Scholar]
- Prossnitz, E.R.; Barton, M. Estrogen biology: New insights into GPER function and clinical opportunities. (Review). Mol. Cell. Endocrinol. 2014, 389, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Sohanaki, H.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Pelargonidin improves memory deficit in amyloid beta25-35 rat model of Alzheimer’s disease by inhibition of glial activation, cholinesterase, and oxidative stress. Biomed. Pharmacother. 2016, 83, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Sohanaki, H.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Pelargonidin Improves Passive Avoidance Task Performance in a Rat Amyloid Beta25-35 Model of Alzheimer’s Disease Via Estrogen Receptor Independent Pathways. Acta Med. Iran 2016, 54, 245–250. [Google Scholar] [PubMed]
- Maggiolini, M.; Vivacqua, A.; Fasanella, G.; Recchia, A.G.; Sisci, D.; Pezzi, V.; Montanaro, D.; Musti, A.M.; Picard, D.; Andò, S. The G protein-coupled receptor GPR30 mediates c-fos up-regulation by 17beta-estradiol and phytoestrogens in breast cancer cells. J. Biol. Chem. 2004, 279, 27008–27016. [Google Scholar] [CrossRef]
- Castellani, R.J.; Rolston, R.K.; Smith, M.A. Alzheimer disease. Disease-a-Month 2010, 56, 484–546. [Google Scholar] [CrossRef]
- Burns, A.; Iliffe, S. Alzheimer’s disease. Brit. Med. J. 2009, 338, b158. [Google Scholar] [CrossRef]
- Brookmeyer, R.; Johnson, E.; Zeigler-Graham, K.; Arrighi, H.M. Forecasting the global burden of Alzheimer’s disease. Alzheimer’s Demntia 2007, 3, 186–191. [Google Scholar] [CrossRef]
- Braak, H.; Del Tredici, K. Where, when, and in what form does sporadic Alzheimer’s disease begin? Curr. Opin. Neurol. 2012, 25, 708–714. [Google Scholar] [CrossRef]
- Thummayot, S.; Tocharus, C.; Suksamrarn, A.; Tocharus, J. Neuroprotective effects of cyanidin against Abeta-induced oxidative and ER stress in SK-N-SH cells. Neurochem. Int. 2016, 101, 15–21. [Google Scholar] [CrossRef]
- Kotzbauer, P.T.; Trojanowski, J.Q.; Lee, V.M. Lewy body pathology in Alzheimer’s disease. J. Mol. Neurosci. 2001, 17, 225–232. [Google Scholar] [CrossRef]
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Todd, S.; Barr, S.; Roberts, M.; Passmore, A.P. Survival in dementia and predictors of mortality: A review. Int. J. Geriatr. Psych. 2013, 28, 1109–1124. [Google Scholar] [CrossRef] [PubMed]
- Hansson, O.; Seibyl, J.; Stomrud, E.; Zetterberg, H.; Trojanowski, J.Q.; Bittner, T.; Lifke, V.; Corradini, V.; Eichenlaub, U.; Batria, R.; et al. CSF biomarkers of Alzheimer’s disease concord with amyloid-β PET and predict clinical progression: A study of fully automated immunoassays in BioFINDER and ADNI cohorts. Alzheimer’s Dement. 2018, 14, 1470–1481. [Google Scholar] [CrossRef]
- Smith, D.G.; Cappal, R.; Barnham, K.J. The redox chemistry of the Alzheimer’s disease amyloid beta peptide. Biochim. Biophys. Acta 2007, 1768, 1976–1990. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Rockenstein, E.; Crews, L.; Masliah, E. Role of protein aggregation in mitochondrial dysfunction and neurodegeneration in Alzheimer’s and Parkinson’s disease. Neuromol. Med. 2003, 4, 21–36. [Google Scholar] [CrossRef]
- Wang, X.; Wang, W.; Li, L.; Perry, G.; Lee, H.; Zhu, Z. Oxidative stress and mitochondrial dysfunction in Alzheimer’s disease. Biochim. Biophys. Acta 2014, 1842, 1240–1247. [Google Scholar] [CrossRef]
- Turner, P.R.; O’Connor, K.; Tate, W.P.; Abraham, W.C. Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory. Prog. Neurobiol. 2003, 70, 1–32. [Google Scholar] [CrossRef]
- Su, B.; Wang, X.; Nunomura, A.; Moreira, P.I.; Lee, H.G.; Perry, G.; Smith, M.A.; Zhu, X. Oxidative stress signaling in Alzheimer’s disease. Curr. Alzheimer Res. 2008, 5, 525–532. [Google Scholar] [CrossRef]
- Francis, P.T.; Palmer, A.M.; Snape, M.; Wilcock, G.K. The cholinergic hypothesis of Azheimer’s disease: A review of progress. J. Neurol. Neurosurg. Psych. 1999, 66, 137–147. [Google Scholar] [CrossRef]
- Hardy, J.; Allsop, D. Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol. Sci. 1991, 12, 383–388. [Google Scholar] [CrossRef]
- González-Barrio, R.; Borges, G.; Mullen, W.; Crozier, A. Bioavailability of anthocyanins and ellagitannins following consumption of raspberries by healthy humans and subjects with an ileostomy. J. Agric. Food Chem. 2010, 58, 3933–3939. [Google Scholar] [CrossRef]
- Marcus, D.I.; Thomas, C.; Rodriguez, C.; Simberkoff, K.; Tasi, J.S.; Strafaci, J.A.; Freedman, M.I. Increased peroxidation and reduced antioxidant enzyme activity in Alzheimer’s disease. Exp. Neurol. 1998, 150, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Foy, C.J.; Passmore, A.P.; Vahidassr, M.D.; Young, I.S.; Lawson, J.T. Plasma chain-breaking antioxidants in Alzheimer’s disease, vascular dementiaand Parkin’s disease. J. Assoc. Phys. QJM 1999, 92, 39–45. [Google Scholar]
- Kim, T.S.; Pae, C.U.; Yoon, S.J.; Jang, W.Y.; Lee, N.J.; Kim, J.J.; Lee, S.J.; Lee, C.; Paik, I.H.; Lee, C.U. Decreased plasma antioxidants in patients with Alzheimer’s disease. Int. J. Geriat. Psych. 2006, 21, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Dore, S. Decreased activity of the antioxidant heme oxygenase enzyme: Implications in ischemia and in Alzheimer’s disease. Free Radic. Biol. Med. 2002, 32, 1276–1282. [Google Scholar]
- Omar, R.A.; Chyan, Y.J.; Andorn, A.C.; Poeggeler, B.; Robakis, N.K.; Pappolla, M.A. Increased expressionbut reduced activity of antioxidant enzymes in Alzheimer’s disease. J. Alzheimer’s Dis. 1999, 1, 139–145. [Google Scholar] [CrossRef]
- Nunomura, A.; Perry, G.; Aliev, G.; Hirai, K.; Takeda, A.; Balraj, E.K.; Jones, P.K.; Ghanbari, H.; Wataya, T.; Shimohama, S.; et al. Oxidative damage is the earliest event in Alzheimer’s disease. J. Neuropathol. Exp. Neurol. 2001, 60, 759–767. [Google Scholar] [CrossRef]
- Swerdlow, R.H. Barin aging, Alzheimer’s disease, and mitochondria. Biochem. Biophys. Acta 2011, 1812, 1630–1639. [Google Scholar]
- Xu, H.; Finkelstein, D.I.; Adlard, P.A. Interaction of metals and apolipoprotien E in Alzheimer’s disease. Front. Aging Neurosci. 2014, 6, 121. [Google Scholar] [CrossRef]
- Kastenholz, B.; Garfin, D.E.; Horst, J.; Nagel, K.A. Plant metal chaperones: A novel perspective in dementia therapy. Amyloid 2009, 16, 81–83. [Google Scholar] [CrossRef]
- Hattori, M.; Sugino, E.; Minoura, K.; In, Y.; Sumida, M.; Taniguchi, T.; Tomoo, K.; Hiltunen, M. Anthocyanin-enriched bilberry and blackcurrant extracts modulate amyloid precursor protein processing and alleviate behavioral abnormalities in the APP/PSI mouse of Alzheimer’s disease. J. Nutr. Biochem. 2013, 24, 360–370. [Google Scholar]
- Ali, F.E.; Leung, A.; Cherny, R.A.; Mavros, C.; Barnham, K.J.; Separovic, F.; Barrow, C.J. Dimerisation of N-acetyl-L-tyrosine ethyl ester and Abeta peptides via formation of dityrosine. Free Radical Res. 2006, 40, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Atwood, C.S.; Perry, G.; Zeng, H.; Kato, Y.; Jones, W.D.; Ling, K.Q.; Huang, X.; Moir, R.D.; Wand, D.; Syre, L.M.; et al. Copper mediates dityrosine cross-linking of Alzheimer’s amyloid-beta. Biochemistry 2004, 43, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Al-Hilaly, Y.K.; Williams, T.L.; Stewart-Parker, M.; Ford, L.; Skaria, E.; Cole, M.; Bucher, W.G.; Morris, K.L.; Sadia, A.A.; Thrope, J.P.; et al. A central role for dityrosine crosslinking of Amuloid-β in Alzheimer’s disease. Acta Neuropathol. Commun. 2013, 1, 83. [Google Scholar] [CrossRef] [PubMed]
- Kandimalla, R.; Vallamkondu, J.; Corgiat, E.B.; Gill, K.D. Understanding aspects of aluminum exposure in Alzheimer’s disease development. Brain Pathol. 2016, 26, 139–154. [Google Scholar] [CrossRef]
- Gyori, J.; Platoshyn, O.; Carpenter, D.O.; Salanki, J. Effect of inorganic and organic tin compounds on ACh-and voltage-activated Na currents. Cell Mol. Neurobiol. 2000, 20, 591–604. [Google Scholar] [CrossRef]
- Maeda-Yamamoto, M.; Saito, T.; Nesumi, A.; Tokuda, Y.; Ema, K.; Honma, D.; Ogino, A.; Monobe, M.; Murakami, A.; Murakami, A.; et al. Chemical analysis and acetylcholinesterase inhibitory effect of anthocyanin-rich red leaf tea (cv. Sunrouge). J. Sci. Food Agric. 2012, 92, 2379–2386. [Google Scholar] [CrossRef]
- Parrado-Fernandez, C.; Sandebring-Matton, A.; Rodriguez-Rodriguez, P.; Aarsland, D.; Cedazo-Minguez, A. Anthocyanins protect from complex I inhibition and APPswe mutation through modulation of the mitochondrial fission/fusion pathways. Biochim. Biophys. Acta 2016, 1862, 2110–2118. [Google Scholar] [CrossRef]
- Qin, L.; Zhang, J.; Qin, M. Protective effect of cyanidin 3-O-glucoside on beta-amyloid peptide-induced cognitive impairment in rats. Neurosci. Lett. 2013, 534, 285–288. [Google Scholar] [CrossRef]
- Song, N.; Zhang, L.; Chen, W.; Zhu, H.; Deng, W.; Han, Y.; Guo, J.; Qin, C. Cyanidin 3-O-beta-glucopyranoside activates peroxisome proliferator-activated receptor-gamma and alleviates cognitive impairment in the APP(swe)/PS1(DeltaE9) mouse model. Biochem. Biophys. Acta 2016, 1862, 1786–1800. [Google Scholar]
- Dröge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Jing, P. Purple Corn Anthocyanins: Chemical Structure, Chemopreventive Activity and Structure/Function Relationships. Ph.D. Thesis, The Ohio State University, Columbus, OH, USA, 2006; pp. 5–90. [Google Scholar]
- Zerovnik, E. Amyloid fibril formation by human stefins: Structure, mechanism and putative functions. Biochimie 2010, 92, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Jomova, K.; Vondrakova, D.; Lawsom, M.; Valko, M. Metals oxidative stress and neurodegenerative disorders. Mol. Cell. Biochem. 2010, 345, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Guo, J.; Jiang, X.; Sun, J.; Tian, L.; Jiao, R.; Tang, Y.; Bai, W. Cyanidin-3-O-glucoside protects against cadmium-induced dysfunction of sex hormone secretion via the regulation of hypothalamus-pituitary-gonadal axis in male pubertal mice. Food Chem. Toxicol. 2019, 129, 13–21. [Google Scholar] [CrossRef]
- Buchweitz, M.; Carle, R.; Kammerer, D.R. Bathochromic and stabilizing effects of sugar beet pectin and an isolated pectic fraction on anthocyanins exhibiting pyrogallol and catechol moieties. Food Chem. 2012, 135, 3010–3019. [Google Scholar] [CrossRef] [PubMed]
- Shiono, M.; Matsugaki, N.; Takeda, K. Structure of commelinin, a blue complex pigment from the blue flowers of Commelina communis. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2008, 84, 452–456. [Google Scholar] [CrossRef]
- Mori, M.; Kondo, T.; Yoshida, K. Cyanosalvianin, a supramolecular blue metalloanthocyanin from petals of Salvia uliginosa. Phytochemistry 2008, 69, 3151–3158. [Google Scholar] [CrossRef]
- Kay, C. Analysis of the Bioactivity, Metabolism, and Pharmacokinetics of Anthocyanins in Humans. Ph.D. Thesis, University of Guelph, Guelph, ON, Canada, 2004; pp. 1–9. [Google Scholar]
- Antal, D.S.; Gârban, G.; Gârban, Z. The anthocyans: Biologically-active substances of food and pharmaceutic interest. Ann. Univ. Dunarea Jos Galati Food Technol. 2003, 6, 106–115. [Google Scholar]
- Wang, D.; Ho, L.; Faith, J.; Ono, K.; Janle, E.M.; Lachcik, P.J.; Cooper, B.R.; Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- Hahkonen, M.P.; Heinonen, M. Antioxidant activity of anthocyanins, and their aglycones. J. Agric. Food Chem. 2003, 51, 628–633. [Google Scholar] [CrossRef]
- Muselík, J.; García-Alonso, M.; Martín-López, M.P.; Žemlička, M.; Rivas-Gonzalo, J.C. Measurement of antioxidant activity of wine catechins, procyanidins, anthocyanins and pyranoanthocyanins. Int. J. Mol. Sci. 2007, 8, 797–809. [Google Scholar] [CrossRef]
- Tsuda, T.; Shiga, K.; Ohshima, K.; Kawakishi, S.; Osawa, T. Inibhition of lipid peroxidation and the active oxygen radical scavenging effect of anthocyanin pigments isolated from Phaseolus vulgaris L. Biochem. Pharmacol. 1996, 52, 1033–1039. [Google Scholar] [CrossRef]
- Vitaglione, P.; Donnarumma, G.; Napolitano, A.; Glavano, F.; Gallo, A.; Scalfi, L.; Fogliano, V. Protocatechuic acid is the major human metabolite of cyanidin-glucosides. J. Nutr. 2007, 137, 2043–2048. [Google Scholar] [CrossRef]
- Forester, S.C.; Waterhouse, A.L. Identification of Carbernet Sauvignon anthocyanin gut microflora metabolites. J. Agric. Food Chem. 2008, 56, 9299–9304. [Google Scholar] [CrossRef]
- Hou, D.X.; Yanagita, T.; Uto, T.; Masuzaki, S.; Fujii, M. Anthocyanidins inhibit cyclooxygenase-2 expression in LPS-evoked macrophages: Structure-activity relationship and molecular mechanism involved. Biochem. Pharmacol. 2005, 70, 417–425. [Google Scholar] [CrossRef]
- Hämäläinen, M.; Nieminen, R.; Vuorela, P.; Heinonen, M.; Moilanen, E. Anti-Inflammatory effects of flavonoids: Genistein, kaempferol, quercetin, and daidzein inhibit STA T-1 and NF-κB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-κB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat. Inflamm. 2007. [Google Scholar] [CrossRef]
- Seeram, N.P.; Momin, R.A.; Nair, M.G.; Bourquin, L.D. Cyclooxygenase inhibitory and antioxidant cyaniding glycosides in cherries and berries. Phytomedicine 2001, 8, 362–369. [Google Scholar] [CrossRef]
- Shih, P.H.; Chan, Y.C.; Liao, J.W.; Wang, M.F.; Yen, G.C. Antioxidantand cognitive promotion effects of anthocyanin-rich mulberry (Morus atropurpurea L.) on senescence-accelerated mice and prevention of Alzheimer’s disease. J. Nutr. Biochem. 2010, 21, 598–605. [Google Scholar] [CrossRef]
- Kalt, W.; McDonald, J.E.; Vinqvist-Tymchuk, M.R.; Liu, Y.; Fillmore, S.A.E. Human anthocyanin bioavailability: Effect of intake duration and dosing. Food Funct. 2017, 8, 4563–4569. [Google Scholar] [CrossRef]
- Felgines, C.; Texier, O.; Besson, C.; Lyan, B.; Lamaison, J.L.; Scalbert, A. Strawberry pelargonidin glycosides are excreted in urine as intact glycosides and glucuronidated pelargonidin derivatives in rats. Br. J. Nutr. 2007, 98, 1126–1131. [Google Scholar] [CrossRef]
- Mihailovic, N.R.; Mihailovic, V.B.; Ciric, A.R.; Sreckovic, N.Z.; Cvijovic, M.R.; Joksovic, L.G. Analysis of Wild Raspberries (Rubus idaeus L.): Optimization of the Ultrasonic-Assisted Extraction of Phenolics and a New Insight in Phenolics Bioaccessibility. Plant Foods Hum. Nutr. 2019. [Google Scholar] [CrossRef]
- Aura, A.M. In Vitro Digestion Models for Dietary Phenolic Compounds. Ph.D. Thesis, Technical Research Centre of Finland, Espoo, Finland, 2005; pp. 63–67. [Google Scholar]
- Borges, G.; Roowi, S.; Rouanet, J.M.; Duthie, G.G.; Lean, M.E.J.; Crozier, A. The bioavailability of raspberry anthocyanins and ellagitannins in rats. Mol. Nutr. Food Res. 2007, 51, 714–725. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of intestinal microbiota in the bioavailablity and physiological functions of dietary polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef]
- Gu, J.; Thomas-Ahner, J.M.; Riedl, K.M.; Bailey, M.T.; Vodovotz, Y.; Schwartz, S.J.; Clinton, S.K. Dietary Black Raspberries Impact the Colonic Microbiome and Phytochemical Metabolites in Mice. Mol. Nutr. Food Res. 2019, 63, e1800636. [Google Scholar] [CrossRef]
- Gutierres-Albanchez, J.M.; Carvalho, F.B.; Schetinger, M.R.; Marisco, P.; Agostinho, P.; Rodrigues, M.; Rubin, M.A.; Schmatz, R.; da Silva, C.R.; Cognato, G.; et al. Anthocyanins restore behavioral and biochemical changes caused by streptozotocin-induced sporadic dementia of Alzheimer’s type. Life Sci. 2014, 96, 7–17. [Google Scholar] [CrossRef]
- Van de Velde, F.; Esposito, D.; Grace, M.H.; Pirovani, M.E.; Lila, M.A. Anti-inflammatory and wound healing properties of polyphenolic extracts from strawberry and blackberry fruits. Food Res. Int. 2019, 121, 453–462. [Google Scholar] [CrossRef]
- Galvano, F.; La Fauci, L.; Lazzarino, G.; Fogliano, V.; Ritieni, A.; Ciappellano, S.; Battistini, N.C.; Tavazzi, B.; Galvano, G. Cyanidins: Metabolism and biological properties. J. Nutr. Biochem 2004, 15, 2–11. [Google Scholar] [CrossRef]
- Amin, F.U.; Shah, S.A.; Badshah, H.; Khan, M.; Kim, M.O. Anthocyanins encapsulated b PLGA@PEG nanoparticles potentially improved its free radical-scavenging capabilities via p38/JNK pathway against Aβ1–42-induced oxidative stress. J. Nanobiotechnol. 2017, 15, 12–28. [Google Scholar] [CrossRef]
- Sun, Q.; Jia, N.; Li, X.; Yang, J.; Chen, G. Grape seed proanthocyanins ameliorate neuronal oxidative damage by inhibiting GSK-3β-dependent mitochondrial permeability transition pore opening in an experimental model of sporadic Alzheimer’s disease. Aging Albany NY 2019, 11, 4107–4124. [Google Scholar]
- Yamakawa, M.Y.; Uchino, K.; Watanabe, Y.; Adachi, T.; Nakanishi, M.; Ichino, H.; Hongo, K.; Mizobata, T.; Kobayashi, S.; Nakashima, K.; et al. Anthocyanin suppresses the toxicity of Abeta deposits through diversion of molecular forms in in vitro and in vivo models of Alzheimer’s disease. Nutr. Neurosci. 2016, 19, 32–42. [Google Scholar] [CrossRef]
- Subash, S.; Essa, M.M.; Braidy, N.; Awlad-Thani, K.; Vaishnav, R.; Al-Adawi, S.; Al-Asmi, A.; Guillemin, G.J. Diet rich in date palm fruits improves memory, learning and reduces beta amyloid in transgenic mouse model of Alzheimer’s disease. J Ayuveda Integr. Med. 2015, 6, 111–120. [Google Scholar]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Nat. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef]
- Nikkhah, E.; Khayami, M.; Heidari, R. In vitro screening for antioxidant activity and cancer suppressive effect on blackberry (Morus nigra). Iranian J. Cancer Prev. 2008, 1, 167–172. [Google Scholar]
- Burton-Freeman, B.M.; Sandhu, A.K.; Edirisinghe, I. Red raspberries and their bioactive polyphenols:cardiometabolic and neuronal health links. Adv. Nutr. 2016, 7, 44–65. [Google Scholar] [CrossRef]
- Fernando, W.; Somartne, G.; Goozee, K.G.; Willaims, S.; Singh, H.; Matins, R.N. Diabetes and Alzheimeir’s disease: Can tea phytochemials play a role in prevention? J. Alzheimer’s Dis. 2017, 59, 481–501. [Google Scholar] [CrossRef]
- Belkacemi, A.; Ramassamy, C. Innovative anthocyanin/anthocyanidin formulation protects SK-N-SH cells against the amyloid-beta peptide-induced toxicity:Relevance to Alzheimer’s disease. Cent. Nerv. Syst. Agents Med. Chem. 2015, 16, 37–49. [Google Scholar] [CrossRef]
- Isaak, C.K.; Petkau, J.C.; Blewett, H.O.K.; Siow, Y.L. Lingonberry anthocyanins protect cardiac cells from oxidative-stress-induced apoptosis. Canad. J. Physiol. Pharmacol. 2017, 95, 904–910. [Google Scholar] [CrossRef]
- Badshah, H.; Kim, T.H.; Kim, M.O. Protective effects of anthocyanins against amyloid beta-induced neurotoxicity in vivo and in vitro. Nurochem. Int. 2015, 80, 51–59. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, M.; Jiang, J. Mitochondrial dysfucntion in merodegeneration diseases and drug targets via appoptic signalling. Mitochondrion 2019, 49, 35–45. [Google Scholar] [CrossRef]
- Pacheco, S.M.; Soares, M.S.P.; Gutierres, J.M.; Gerzson, M.F.B.; Carvalho, F.B.; Azambuja, J.H.; Schetinger, M.R.C.; Stefanello, F.M.; Spanevello, R.M. Anthocyanins as a potential pharmacological agent to manage memory deficit, oxidative stress and alterations in ion pump activity induced by experimental sporadic dementia of Alzheimer’s type. J. Nutr. Biochem. 2018, 56, 193–204. [Google Scholar] [CrossRef]
- Meng, L.; Xin, G.; Li, B.; Li, D.; Sun, X.; Yan, T.; Li, L.; Shi, L.; Cao, S.; Meng, X. Anthocyanins Extracted from Aronia melanocarpa Protect SH-SY5Y Cells against Amyloid-beta (1-42)-Induced Apoptosis by Regulating Ca(2+) Homeostasis and Inhibiting Mitochondrial Dysfunction. J. Agric. Food Chem. 2018, 66, 12967–12977. [Google Scholar] [CrossRef]
- Fraige, K.; Pereira-Filho, E.R.; Carrilho, E. Fingerprinting of anthocyanins from grapes produced in Brazil using HPLC-DAD-MS and exploratory analysis of principal component anaylysis. Food Chem. 2014, 145, 395–403. [Google Scholar] [CrossRef]
- Celik, E.; Sanlier, N. Effects of neutrients and bioactive food components on Alzheimer’s disease and epigentic. Crit. Rev. Food Sci. Nutr. 2017, 59, 102–113. [Google Scholar] [CrossRef]
- Afzal, M.; Safer, A.M.; Menon, M. Green tea polyphenols and their potential in health and disease. Inflammopharmacology 2015, 23, 151–161. [Google Scholar] [CrossRef]
- Safer, A.M.; Hanafi, N.; Bharali, D.J.; Cui, H.; Mousa, S.S. Effect of green tea extract encapsulated into chitosan nanoparticles in hepatice fibrosis collagen fibers assessed atomic force microscopy in rat hepatic fibrosis model. J. Nanosci. Nanotechnol. 2015, 15, 6452–6459. [Google Scholar] [CrossRef]
- Engin, A.B.; Engin, A. Nanoparticles and neurotoxicity dual response of glutamatergic receptors. Prog. Brain Res. 2019, 245, 281–303. [Google Scholar]
- Kim, M.J.; Rehman, S.U.; Amin, F.U.; Kim, M.O. Enhanced neuroprotection of anthocyanin-loaded PEG-gold nanoparticles against Abeta1-42-induced neuroinflammation and neurodegeneration via the NF-KB/JNK/GSK3beta signaling pathway. Nanomedicine 2017, 13, 2533–2544. [Google Scholar] [CrossRef]
- Ali, T.; Kim, T.; Rehman, S.U.; Khan, M.S.; Amin, F.U.; Khan, M.; Ikram, M. Aluminium and Alzheimer’s Disease. Facts about Dementia; Alzheimer’s Society: London, UK, 2005. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afzal, M.; Redha, A.; AlHasan, R. Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease. Molecules 2019, 24, 4255. https://doi.org/10.3390/molecules24234255
Afzal M, Redha A, AlHasan R. Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease. Molecules. 2019; 24(23):4255. https://doi.org/10.3390/molecules24234255
Chicago/Turabian StyleAfzal, Mohammad, Amina Redha, and Redha AlHasan. 2019. "Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease" Molecules 24, no. 23: 4255. https://doi.org/10.3390/molecules24234255
APA StyleAfzal, M., Redha, A., & AlHasan, R. (2019). Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease. Molecules, 24(23), 4255. https://doi.org/10.3390/molecules24234255




