Composition of Sugars in Wild and Cultivated Lingonberries (Vaccinium vitis-idaea L.)
Abstract
1. Introduction
2. Results
2.1. Method Validation
2.2. Qualitative Analysis of Sugars in Wild and Cultivated Lingonberries
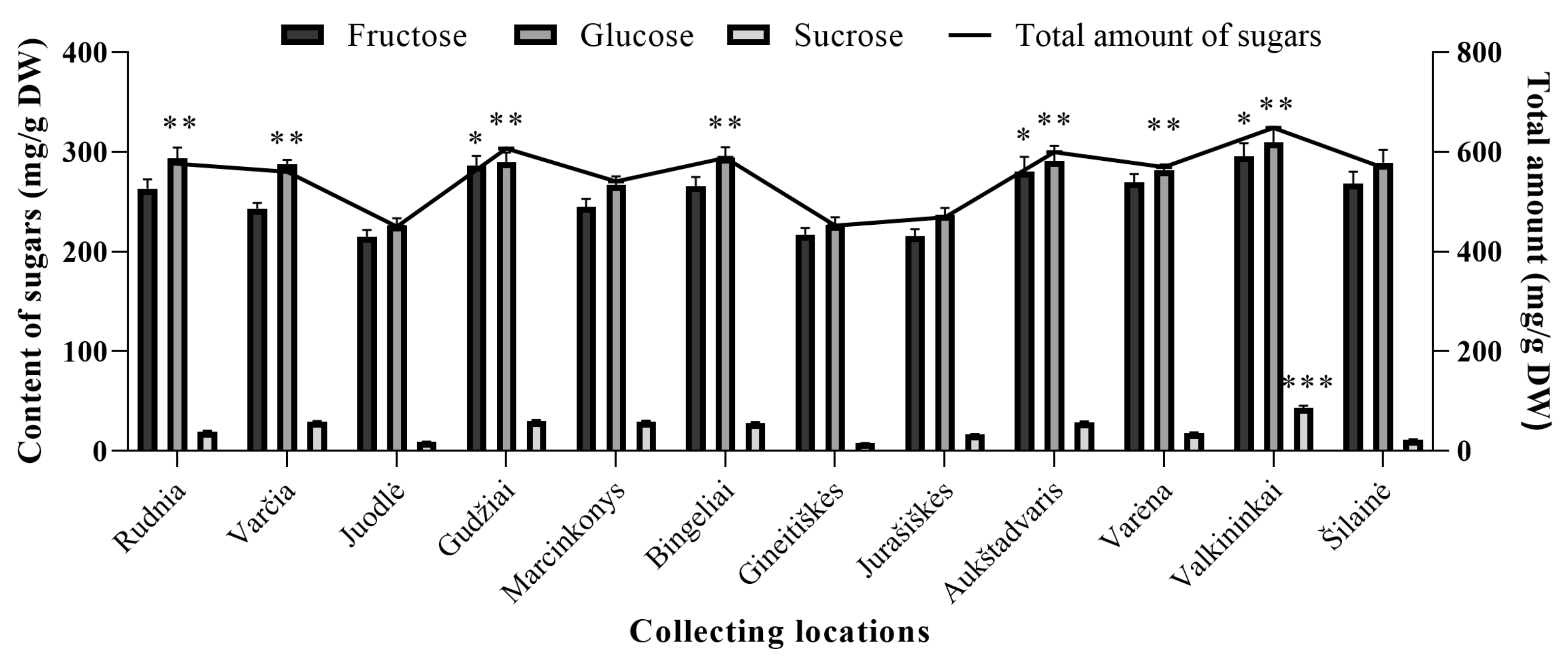
2.3. Quantitative Analysis of Sugars in Wild Lingonberries
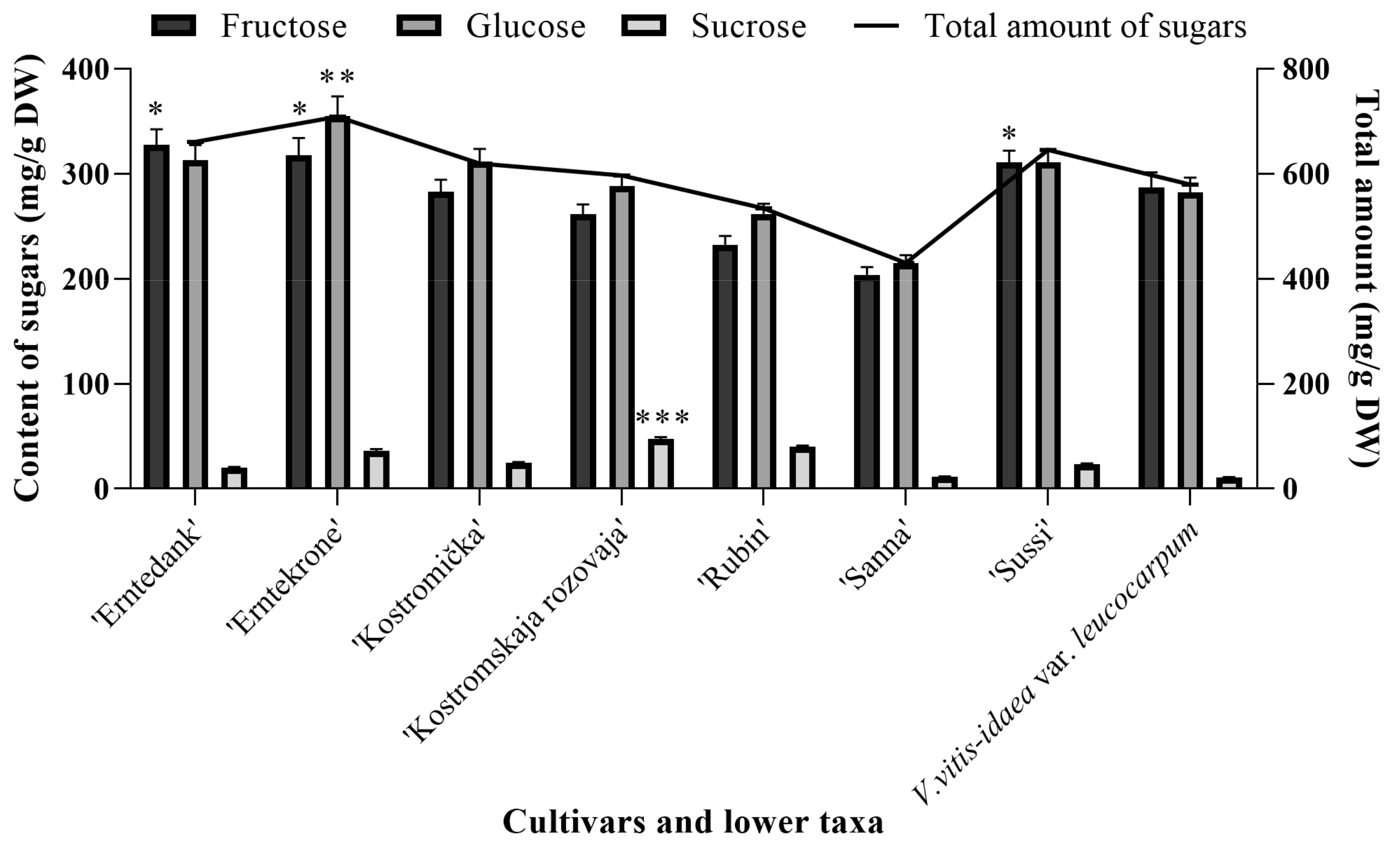
2.4. Quantitative Analysis of Sugars in Cultivated Lingonberries
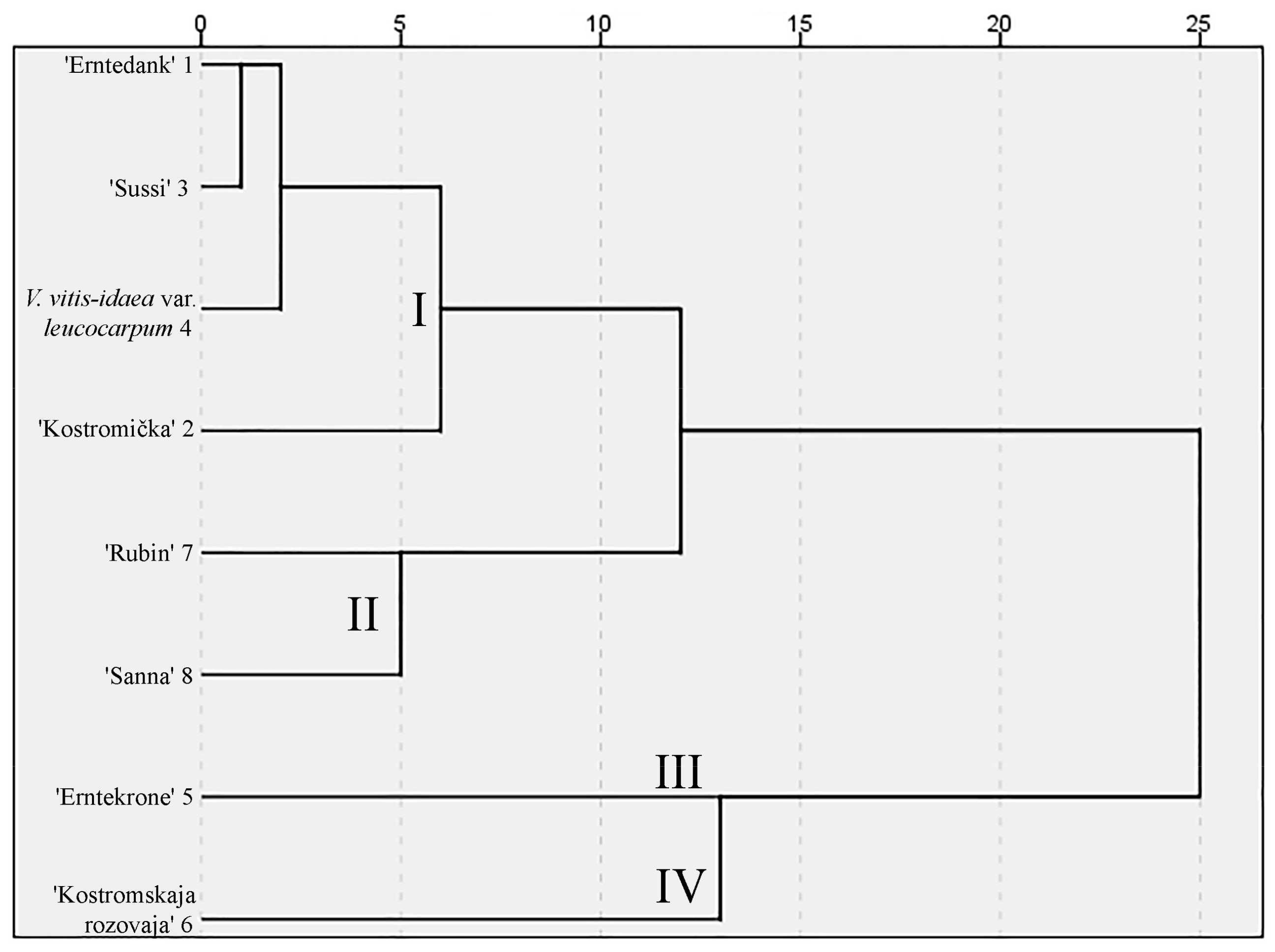
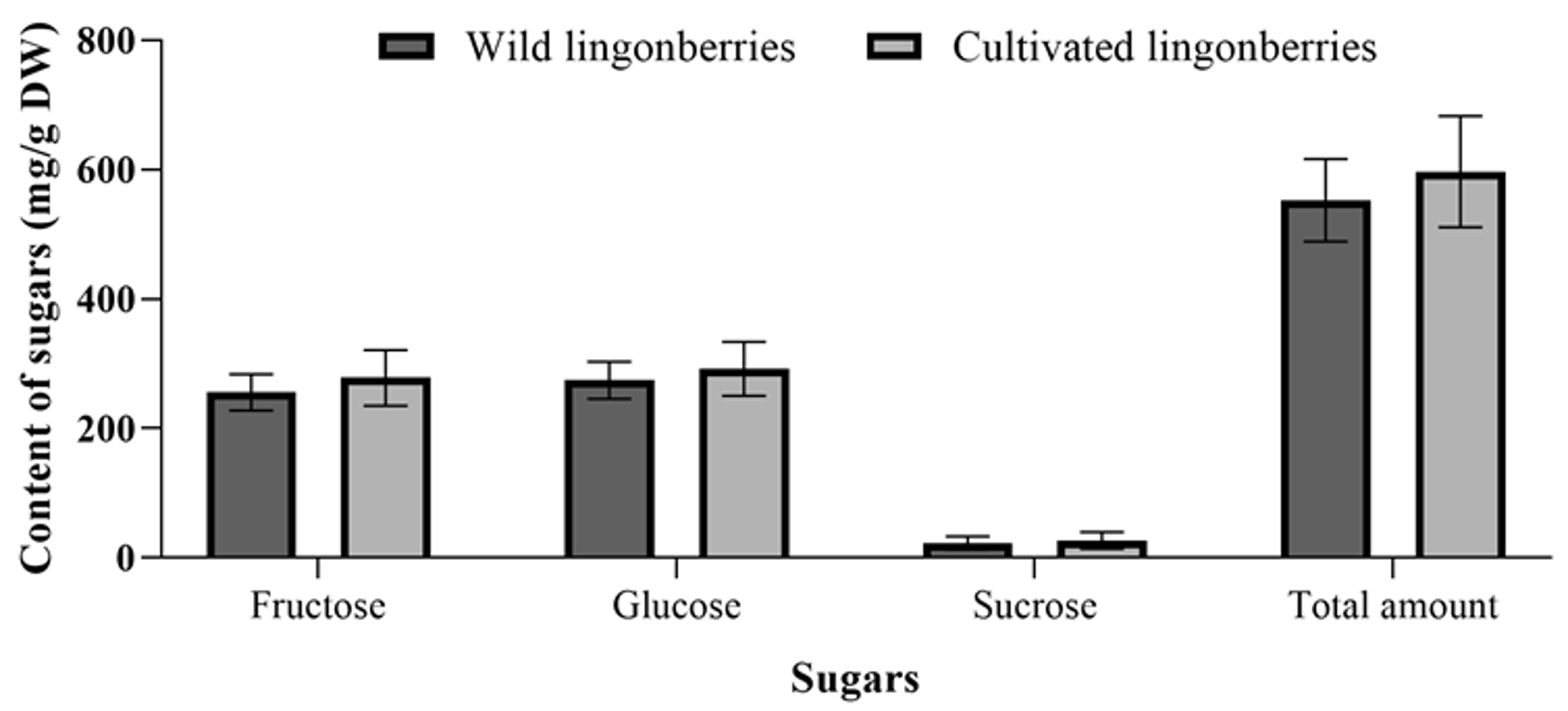
2.5. Comparison of Sugars Between Wild and Cultivated Lingonberries
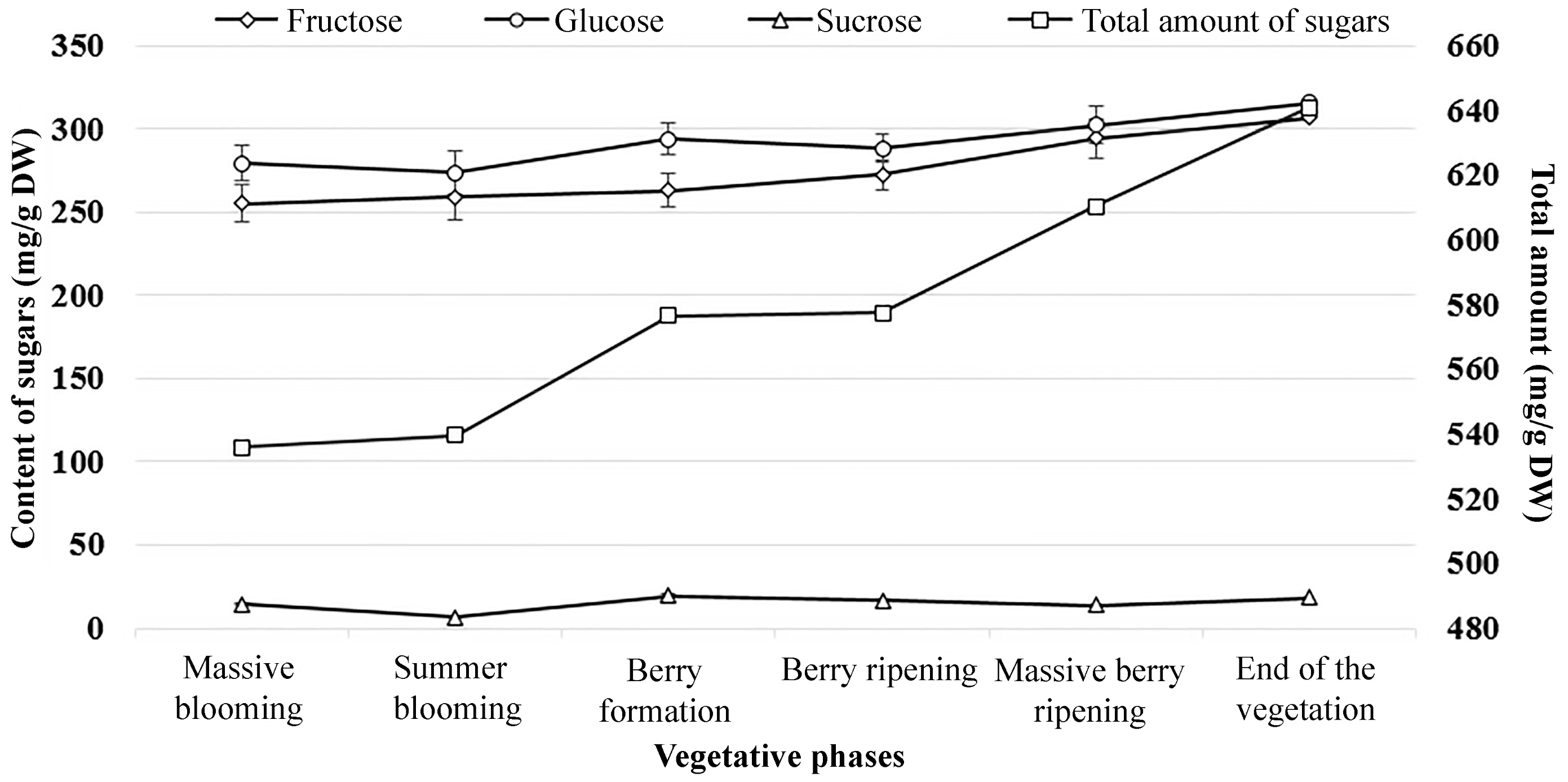
2.6. Sugars of Lingonberries During the Growing Season
3. Discussion
4. Materials and Methods
4.1. Chemicals and Solvents
4.2. Plant Material
4.2.1. Wild Lingonberries
4.2.2. Cultivated Lingonberries
4.3. Sample Preparation and Extraction of Sugars
4.4. Qualitative and Quantitative Analysis of Sugars by HPLC–ELSD Method
4.5. Method Validation
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Guideline: Sugars Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2015; pp. 1–5. [Google Scholar]
- Van Dam, R.M.; Seidell, J.C. Carbohydrate Intake and Obesity. Eur. J. Clin. Nutr. 2007, 61, 75–99. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.K.; Appel, L.J.; Brands, M.; Howard, B.V.; Lefevre, M.; Lustig, R.H.; Sacks, F.; Steffen, L.M.; Wylie-Rosett, J. Dietary Sugars Intake and Cardiovascular Health a Scientific Statement from the American Heart Association. Circulation 2009, 120, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Stephen, A.M. Carbohydrate Terminology and Classification. Eur. J. Clin. Nut. 2007, 61, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Muir, J.G.; Barrett, J.S.; Gibson, P.R. Fermentable Oligosaccharides, Disaccharides, Monosaccharides and Polyols: Role in Irritable Bowel Syndrome. Expert. Rev. Gastroent. 2014, 8, 819–834. [Google Scholar] [CrossRef] [PubMed]
- Rizkalla, S.W. Health Implications of Fructose Consumption: A Review of Recent Data. Nutr. Metab. (Lond) 2010, 7, 82. [Google Scholar] [CrossRef]
- Rosa, M.; Prado, C.; Podazza, G.; Interdonato, R.; González, J.A.; Hilal, M.; Prado, F.E. Soluble Sugars-Metabolism, Sensing and Abiotic Stress a Complex Network in the Life of Plants. Plant. Signal. Behav. 2009, 4, 388–393. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Schmitzer, V.; Slatnar, A.; Stampar, F.; Veberic, R. Composition of Sugars, Organic Acids, and Total Phenolics in 25 Wild or Cultivated Berry Species. J. Food. Sci. 2012, 77, 1064–1070. [Google Scholar] [CrossRef]
- Hisano, M.; Bruschini, H.; Nicodemo, A.C.; Srougi, M. Cranberries and Lower Urinary Tract Infection Prevention. Clinics (Sao Paulo) 2012, 67, 661–667. [Google Scholar] [CrossRef]
- Ofek, I.; Hasty, D.L.; Sharon, N. Anti-Adhesion Therapy of Bacterial Diseases: Prospects and Problems. FEMS. Immunol. Med. Microbiol. 2003, 38, 181–191. [Google Scholar] [CrossRef]
- Foxman, B. The Epidemiology of Urinary Tract Infection. Nat. Rev. Urol. 2010, 7, 653–660. [Google Scholar] [CrossRef]
- Drózdz, P.; Seziene, V.; Wójcik, J.; Pyrzyńska, K. Evaluation of Bioactive Compounds, Minerals and Antioxidant Activity of Lingonberry (Vaccinium vitis-idaea L.) Fruits. Molecules 2018, 23, 53. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Finn, C.E. Lingonberry (Vaccinium vitis-idaea L.) Grown in the Pacific Northwest of North America: Anthocyanin and Free Amino Acid Composition. J. Funct. Foods. 2012, 4, 213–218. [Google Scholar] [CrossRef]
- Suomela, J.P.; Vaarno, J.; Sandell, M.; Lehtonen, H.M.; Tahvonen, R.; Viikari, J.; Kallio, H. Children’s Hedonic Response to Berry Products: Effect of Chemical Composition of Berries and HTAS2R38 Genotype on Liking. Food. Chem. 2012, 135, 1210–1219. [Google Scholar] [CrossRef] [PubMed]
- Kylli, P.; Nohynek, L.; Puupponen-Pimiä, R.; Westerlund-Wikström, B.; Leppänen, T.; Welling, J.; Moilanen, E.; Heinonen, M. Lingonberry (Vaccinium vitis-idaea) and European Cranberry (Vaccinium microcarpon) Proanthocyanidins: Isolation, Identification, and Bioactivities. J. Agric. Food. Chem. 2011, 59, 3373–3384. [Google Scholar] [CrossRef] [PubMed]
- Klavins, L.; Klavina, L.; Huna, A.; Klavins, M. Polyphenols, Carbohydrates and Lipids in Berries of Vaccinium Species. Environ. Exp. Biol. 2015, 13, 147–158, 2255-958. [Google Scholar]
- Fan, Z.L.; Wang, Z.Y.; Liu, J.R. Cold-Field Fruit Extracts Exert Different Antioxidant and Antiproliferative Activities in Vitro. Food. Chem. 2011, 129, 402–407. [Google Scholar] [CrossRef]
- Fan, Z.L.; Wang, Z.Y.; Zuo, L.L.; Tian, S.Q. Protective Effect of Anthocyanins from Lingonberry on Radiation-Induced Damages. Inter. J. Env. Res. Pub. Heal. 2012, 9, 4732–4743. [Google Scholar] [CrossRef]
- Alam, Z.; Roncal, J.; Peña-Castillo, L. Genetic Variation Associated with Healthy Traits and Environmental Conditions in Vaccinium Vitis-Idaea. BMC. Genomics. 2018, 19. [Google Scholar] [CrossRef]
- Gustavsson, B.A. Genetic Variation in Horticulturally Important Traits of Fifteen Wild Lingonberry Vaccinium Vitis-Idaea L. Populations. Euphytica 2001, 120, 173–182. [Google Scholar] [CrossRef]
- Viljanen, K.; Heiniö, R.L.; Juvonen, R.; Kössö, T.; Puupponen-Pimiä, R. Relation of Sensory Perception with Chemical Composition of Bioprocessed Lingonberry. Food. Chem. 2014, 157, 148–156. [Google Scholar] [CrossRef]
- Varo, P.; Laine, R.; Veijalainen, K.; Espo, A.; Wetterhoff, A.; Koivistoinen, P. Dietary Fibre and Available Carbohydrates in Finnish Vegetables and Fruits. Agric. Food. Sci. 1984, 56, 49–59. [Google Scholar] [CrossRef]
- Viljakainen, S.; Visti, A.; Laakso, S. Concentrations of Organic Acids and Soluble Sugars in Juices from Nordic Berries. Acta. Agric. Scand. Sect. B. Soil. Plant. Sci. 2002, 52, 101–109. [Google Scholar] [CrossRef]
- Antolak, H.; Czyzowska, A.; Sakač, M.; Mišan, A.; Đuragić, O.; Kregiel, D. Phenolic Compounds Contained in Little-Known Wild Fruits as Antiadhesive Agents Against the Beverage-Spoiling Bacteria Asaia Spp. Molecules 2017, 22, 1256. [Google Scholar] [CrossRef] [PubMed]
- Ross, K.A.; Godfrey, D.; Fukumoto, L. The Chemical Composition, Antioxidant Activity and α-Glucosidase Inhibitory Activity of Water-Extractable Polysaccharide Conjugates from Northern Manitoba Lingonberry. Cogent. Food. Agric. 2015, 1. [Google Scholar] [CrossRef]
- Bandzaitiene, Z.; Daubaras, R.; Labokas, J. Brukne: Vaccinium vitis-idaea L.; Botanikos instituto leidykla: Vilnius, Lithuania, 2007; pp. 25–133. [Google Scholar]
- Paal, T. Lingonberry (Vaccinium vitis-idaea L.) Research in Estonia: An overview. Acta. Hortic. 2006, 715, 203–218. [Google Scholar] [CrossRef]
- Haffner, K.E. Ecology of Vaccinium growing. Acta. Hortic. 1993, 346, 214–220. [Google Scholar] [CrossRef]
- Penhallegon, R. Lingonberry Production Guide for the Pacific Northwest. Available online: https://catalog.extension.oregonstate.edu/pnw583 (accessed on 29 September 2019).
- Raudone, L.; Vilkickyte, G.; Pitkauskaite, L.; Raudonis, R.; Vainoriene, R.; Motiekaityte, V. Antioxidant Activities of Vaccinium vitis-idaea L. Leaves within Cultivars and Their Phenolic Compounds. Molecules 2019, 24, 844. [Google Scholar] [CrossRef]
- Ma, B.; Chen, J.; Zheng, H.; Fang, T.; Ogutu, C.; Li, S.; Han, Y.; Wu, B. Comparative Assessment of Sugar and Malic Acid Composition in Cultivated and Wild Apples. Food. Chem. 2015, 172, 86–91. [Google Scholar] [CrossRef]
- Guerrero, C.J.; Ciampi, P.L.; Castilla, C.A.; Medel, S.F.; Schalchli, S.H.; Hormazabal, U.E.; Bensch, T.E.; Alberdi, L.M. Antioxidant Capacity, Anthocyanins, and Total Phenols of Wild and Cultivated Berries in Chile. Chil. J. Agric. Res. 2010, 70, 537–544. [Google Scholar] [CrossRef]
- Yang, L.; Wen, K.S.; Ruan, X.; Zhao, Y.X.; Wei, F.; Wang, Q. Response of Plant Secondary Metabolites to Environmental Factors. Molecules 2018, 23, 762. [Google Scholar] [CrossRef]
- Jaakola, L.; Hohtola, A. Effect of Latitude on Flavonoid Biosynthesis in Plants. Plant. Cell. Environ. 2010, 33, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Vyas, P.; Curran, N.H.; Igamberdiev, A.U.; Debnath, S.C. Antioxidant Properties of Lingonberry (Vaccinium vitis-idaea L.) Leaves within a Set of Wild Clones and Cultivars. Can. J. Plant. Sci. 2015, 95, 663–669. [Google Scholar] [CrossRef]
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive Compounds and Antioxidant Activity in Different Types of Berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef] [PubMed]
- Bujor, O.-C.; Le Bourvellec, C.; Volf, I.; Popa, V.I.; Dufour, C. Seasonal Variations of the Phenolic Constituents in Bilberry (Vaccinium myrtillus L.) Leaves, Stems and Fruits, and Their Antioxidant Activity. Food. Chem. 2016, 213, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Caffin, N.; D’Arcy, B.; Jiang, Y.; Shi, J.; Singanusong, R.; Liu, X.; Datta, N.; Kakuda, Y.; Xu, Y. Seasonal Variations of Phenolic Compounds in Australia-Grown Tea (Camellia sinensis). J. Agric. Food. Chem. 2005, 53, 6477–6483. [Google Scholar] [CrossRef]
- Drózdz, P.; Seziene, V.; Pyrzynska, K. Phytochemical Properties and Antioxidant Activities of Extracts from Wild Blueberries and Lingonberries. Plant. Foods. Hum. Nutr. 2017, 72, 360–364. [Google Scholar] [CrossRef]
- Eid, H.M.; Ouchfoun, M.; Brault, A.; Vallerand, D.; Musallam, L.; Arnason, J.T.; Haddad, P.S. Lingonberry (Vaccinium Vitis-Idaea L.) Exhibits Antidiabetic Activities in a Mouse Model of Diet-Induced Obesity. Evid-Based. Compl. Alt. 2014, 645812, 1–10. [Google Scholar] [CrossRef]
- Ho, G.T.T.; Nguyen, T.K.Y.; Kase, E.T.; Tadesse, M.; Barsett, H.; Wangensteen, H. Enhanced Glucose Uptake in Human Liver Cells and Inhibition of Carbohydrate Hydrolyzing Enzymes by Nordic Berry Extracts. Molecules 2017, 22, 1806. [Google Scholar] [CrossRef]
- Kivimäki, A.S.; Siltari, A.; Ehlers, P.I.; Korpela, R.; Vapaatalo, H. Lingonberry Juice Lowers Blood Pressure of Spontaneously Hypertensive Rats (SHR). J. Funct. Foods. 2013, 5, 1432–1440. [Google Scholar] [CrossRef]
- Addor, F.A.S. Antioxidants in Dermatology. An. Bras. Dermatol. 2017, 92, 356–362. [Google Scholar] [CrossRef]
- Pimple, B.P.; Badole, S.L. Polyphenols: A Remedy for Skin Wrinkles. In Polyphenols in Human Health and Disease; Academic Press: Cambridge, MA, USA, 2013; Volume 1, pp. 861–869. [Google Scholar]
- Malciute, A.; Naujalis, J.R.; Vilkonis, K.K. Cowberry (Vaccinium vitis-idaea) Collection in the Botanical Garden of Siauliai University: Composition and General Condition. Jaunųjų. Mokslininkų. darbai. 2008, 5, 81–84, 1648-8776. [Google Scholar]
- Zymone, K.; Raudone, L.; Raudonis, R.; Marksa, M.; Ivanauskas, L.; Janulis, V. Phytochemical Profiling of Fruit Powders of Twenty Sorbus L. Cultivars. Molecules 2018, 23, 2593. [Google Scholar] [CrossRef] [PubMed]
- ICH Q2(R1). Validation of Analytical Procedures: Text and Methodology. Current Step 4 Version. 2005. Available online: https://pacificbiolabs.com/wp-content/uploads/2017/12/Q2_R1__Guideline-4.pdf (accessed on 27 October 1994).
Sample Availability: Samples of the compounds are available from the authors. |
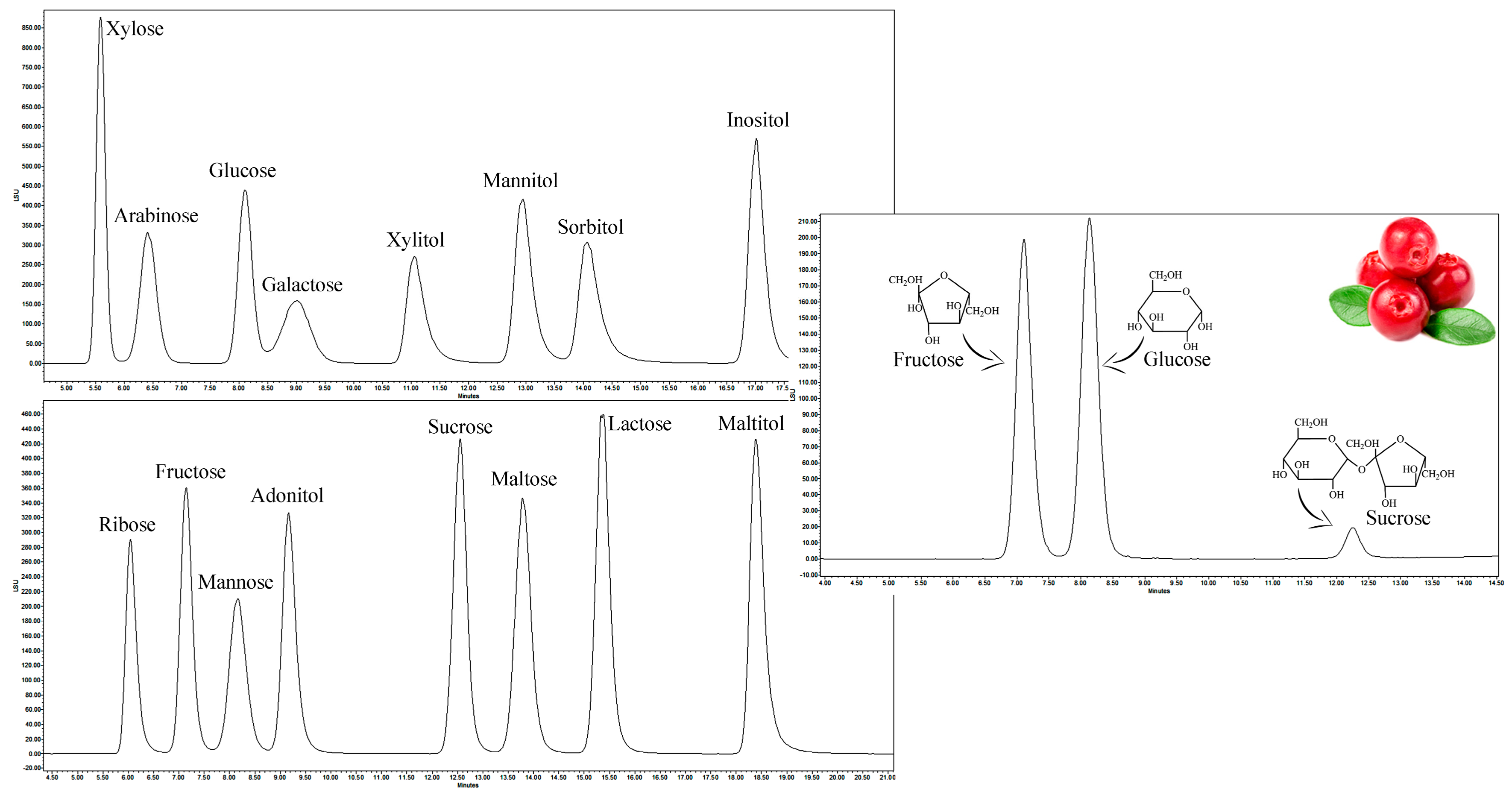

| Component | Calibration Equation | Coefficient of Determination R2 | Coefficient of Correlation R |
|---|---|---|---|
| Fructose | Y = 1.80 X + 44.9 | 0.9998 | 0.9999 |
| Glucose | Y = 2.00 X − 26.3 | 0.9876 | 0.9938 |
| Sucrose | Y = 1.77 X + 82.2 | 0.9999 | 0.9999 |
| Forest | Latitude (°) | Longitude (°) | Altitude (m) |
|---|---|---|---|
| Rudnia | 54.40 | 24.49 | 137 |
| Gineitiškės | 54.49 | 24.39 | 155 |
| Marcinkonys | 54.07 | 24.43 | 123 |
| Varėna | 54.29 | 24.44 | 136 |
| Valkininkai | 54.36 | 24.85 | 116 |
| Gudžiai | 54.36 | 24.43 | 136 |
| Bingeliai | 54.15 | 24.25 | 112 |
| Šilainė | 54.08 | 23.71 | 135 |
| Varčia | 54.32 | 24.21 | 148 |
| Aukštadvaris | 54.57 | 24.61 | 167 |
| Juodlė | 55.83 | 22.94 | 141 |
| Jurašiškės | 54.10 | 23.89 | 135 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilkickyte, G.; Raudonis, R.; Motiekaityte, V.; Vainoriene, R.; Burdulis, D.; Viskelis, J.; Raudone, L. Composition of Sugars in Wild and Cultivated Lingonberries (Vaccinium vitis-idaea L.). Molecules 2019, 24, 4225. https://doi.org/10.3390/molecules24234225
Vilkickyte G, Raudonis R, Motiekaityte V, Vainoriene R, Burdulis D, Viskelis J, Raudone L. Composition of Sugars in Wild and Cultivated Lingonberries (Vaccinium vitis-idaea L.). Molecules. 2019; 24(23):4225. https://doi.org/10.3390/molecules24234225
Chicago/Turabian StyleVilkickyte, Gabriele, Raimondas Raudonis, Vida Motiekaityte, Rimanta Vainoriene, Deividas Burdulis, Jonas Viskelis, and Lina Raudone. 2019. "Composition of Sugars in Wild and Cultivated Lingonberries (Vaccinium vitis-idaea L.)" Molecules 24, no. 23: 4225. https://doi.org/10.3390/molecules24234225
APA StyleVilkickyte, G., Raudonis, R., Motiekaityte, V., Vainoriene, R., Burdulis, D., Viskelis, J., & Raudone, L. (2019). Composition of Sugars in Wild and Cultivated Lingonberries (Vaccinium vitis-idaea L.). Molecules, 24(23), 4225. https://doi.org/10.3390/molecules24234225









