Development of a Validated UPLC-MS/MS Method for Analyzing Major Ginseng Saponins from Various Ginseng Species
Abstract
1. Introduction
2. Results and Discussion
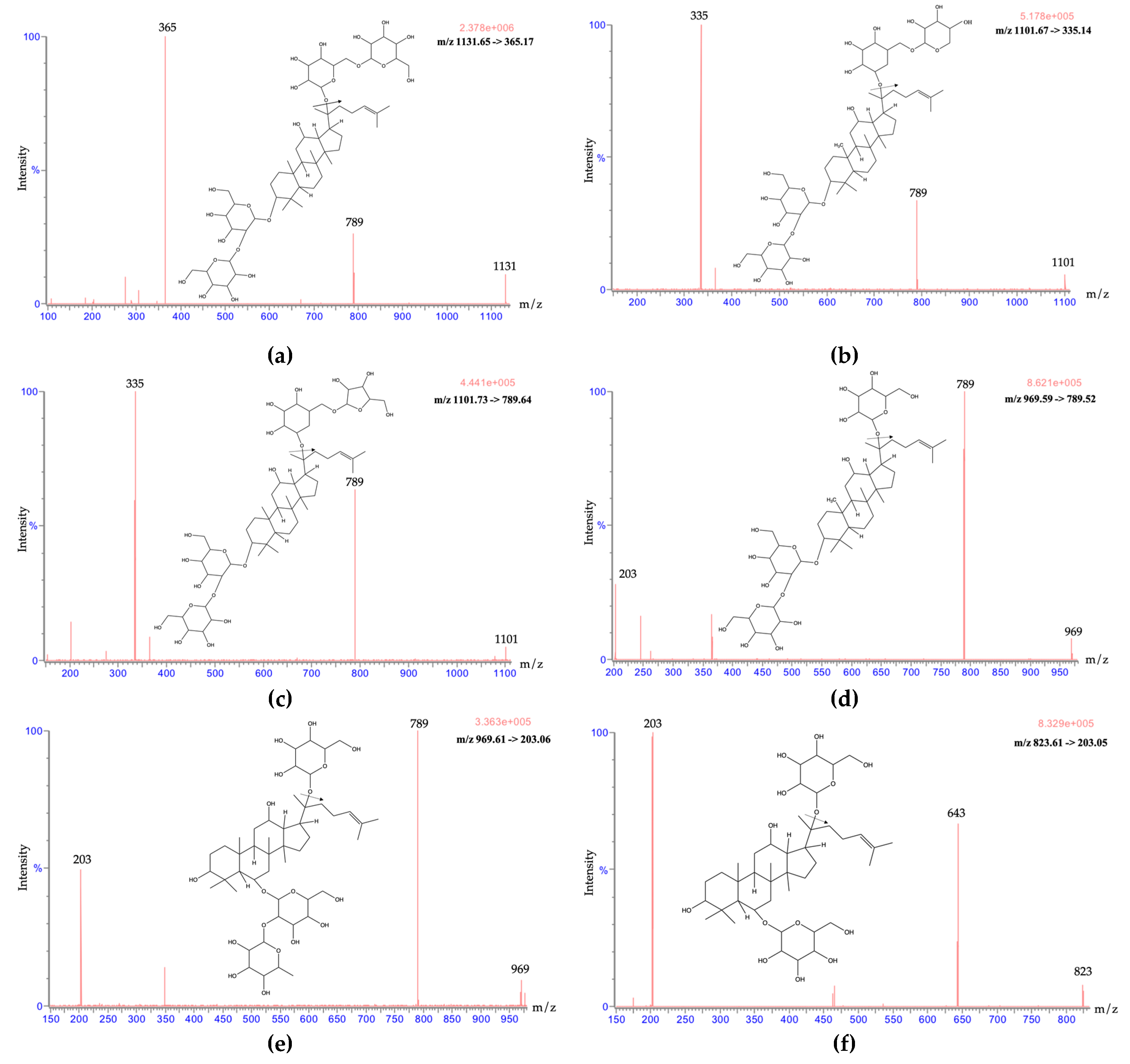
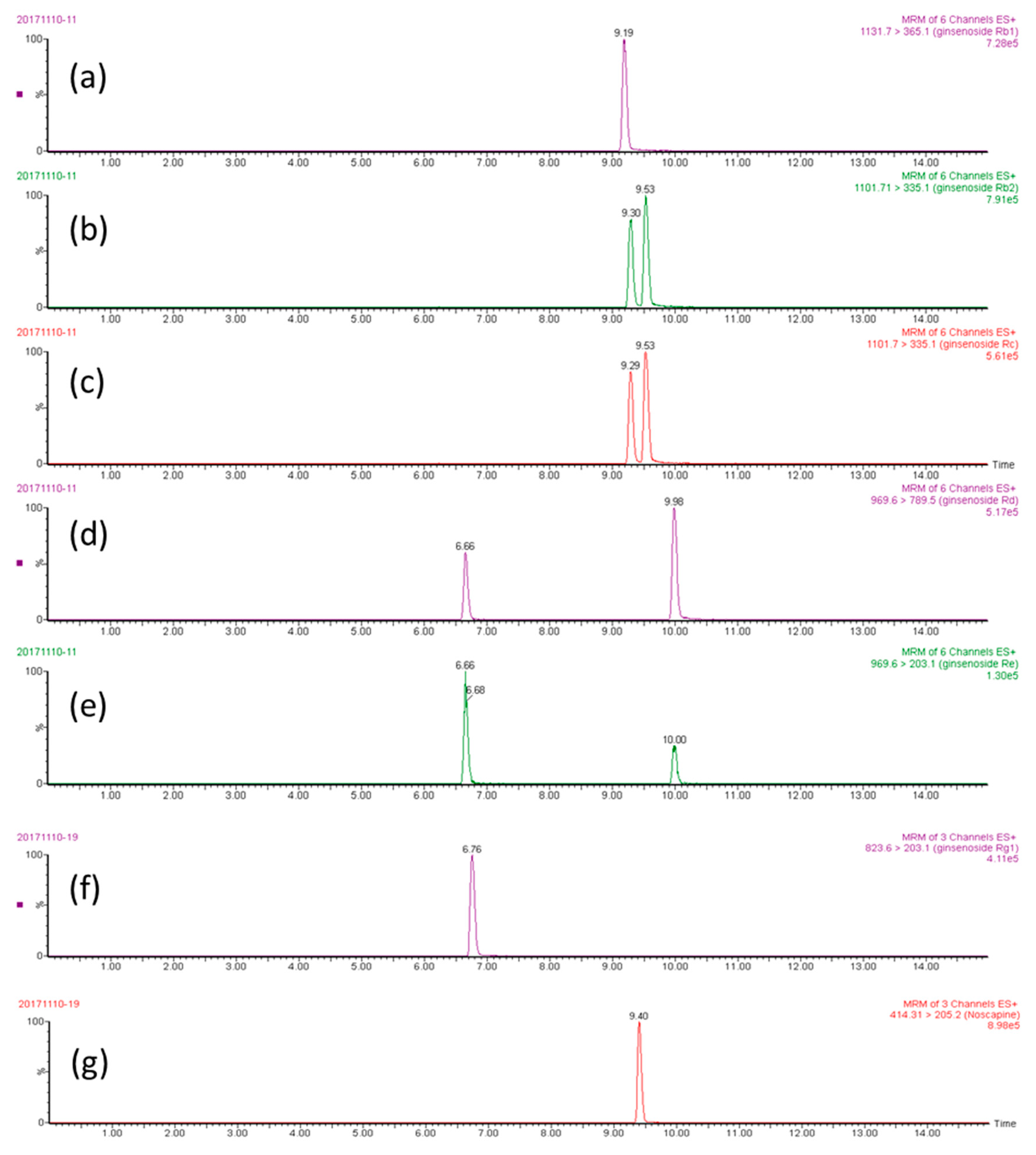
2.1. Optimization of UPLC-MS/MS Conditions
2.2. Method Validation
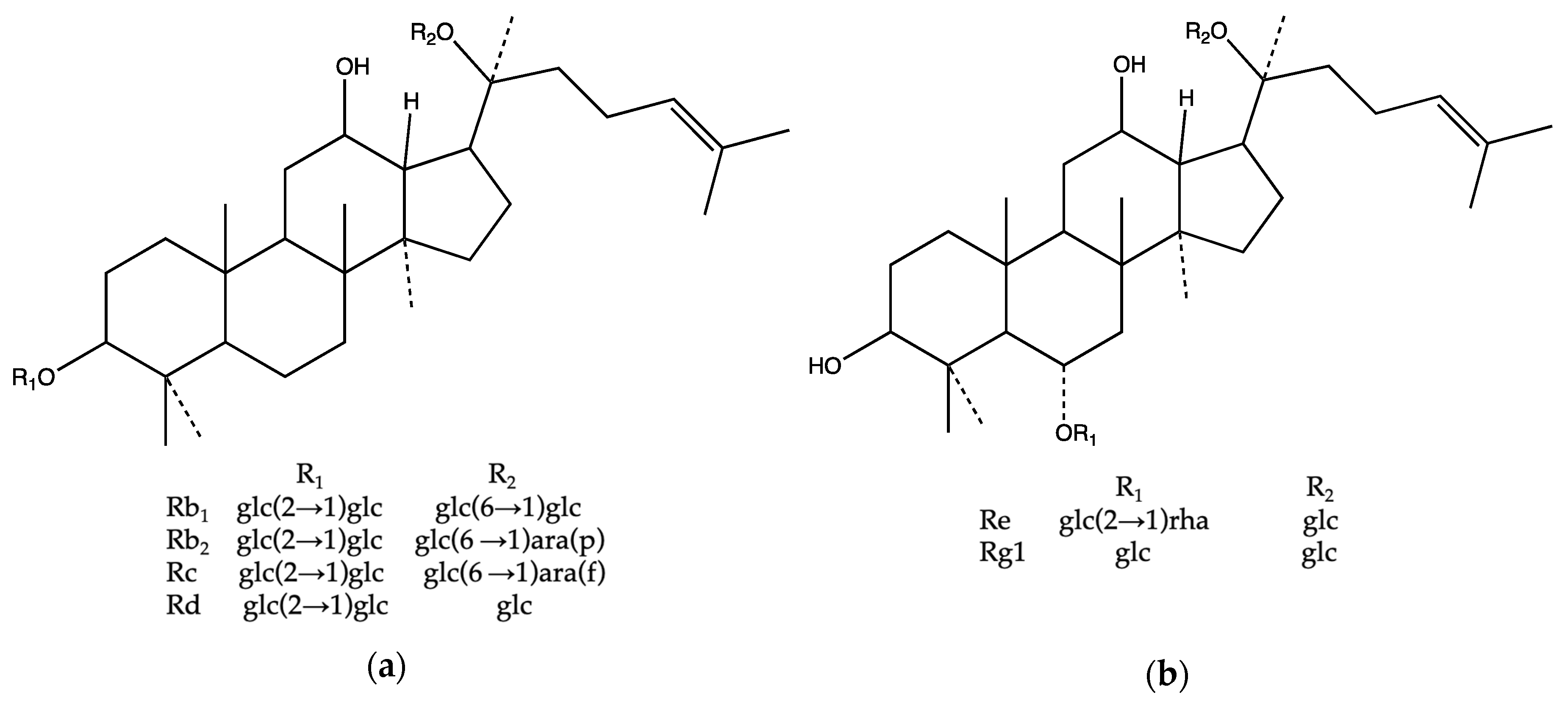
2.3. The Correlation between the Chemical Structure and Retention Time
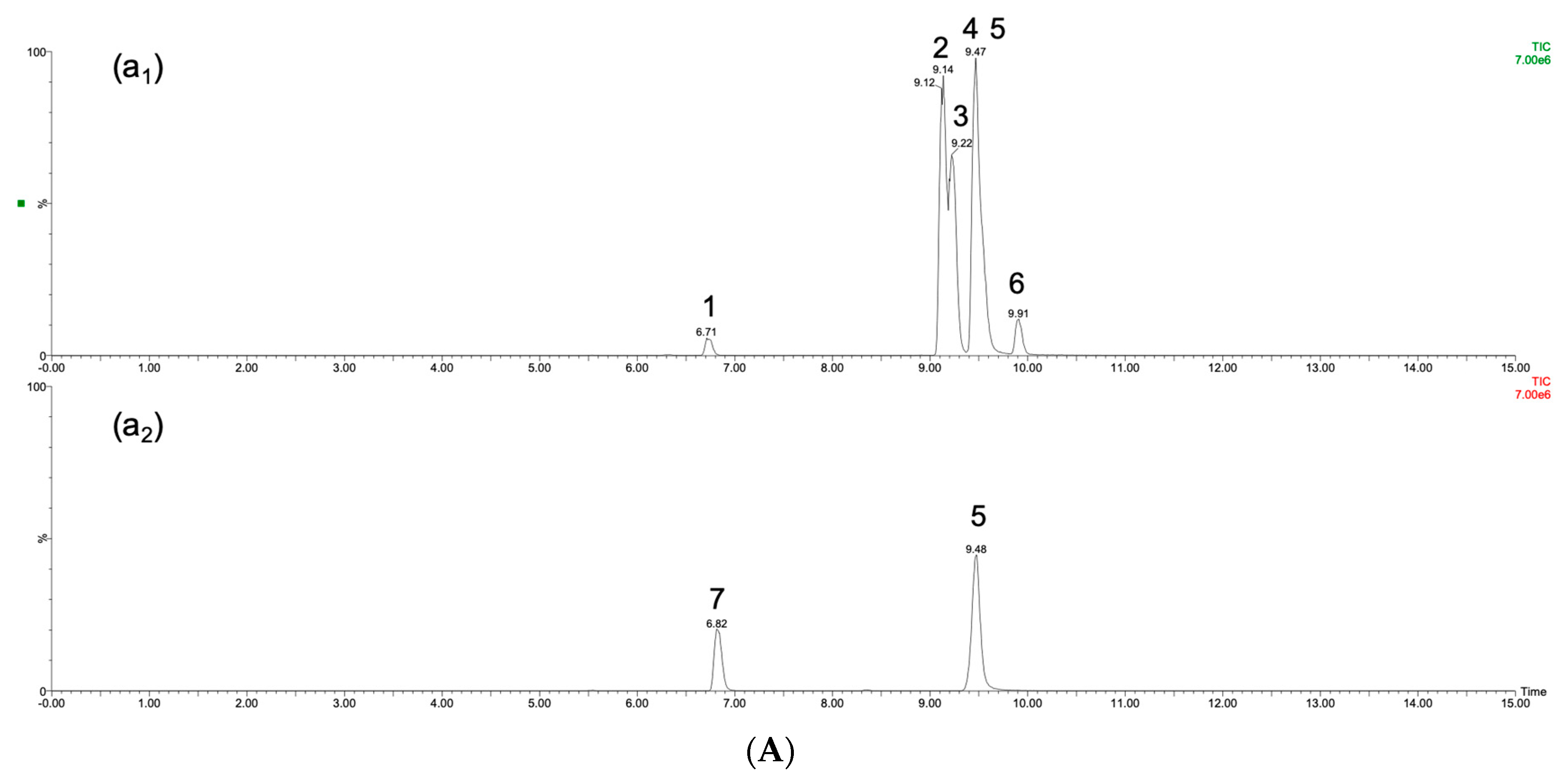
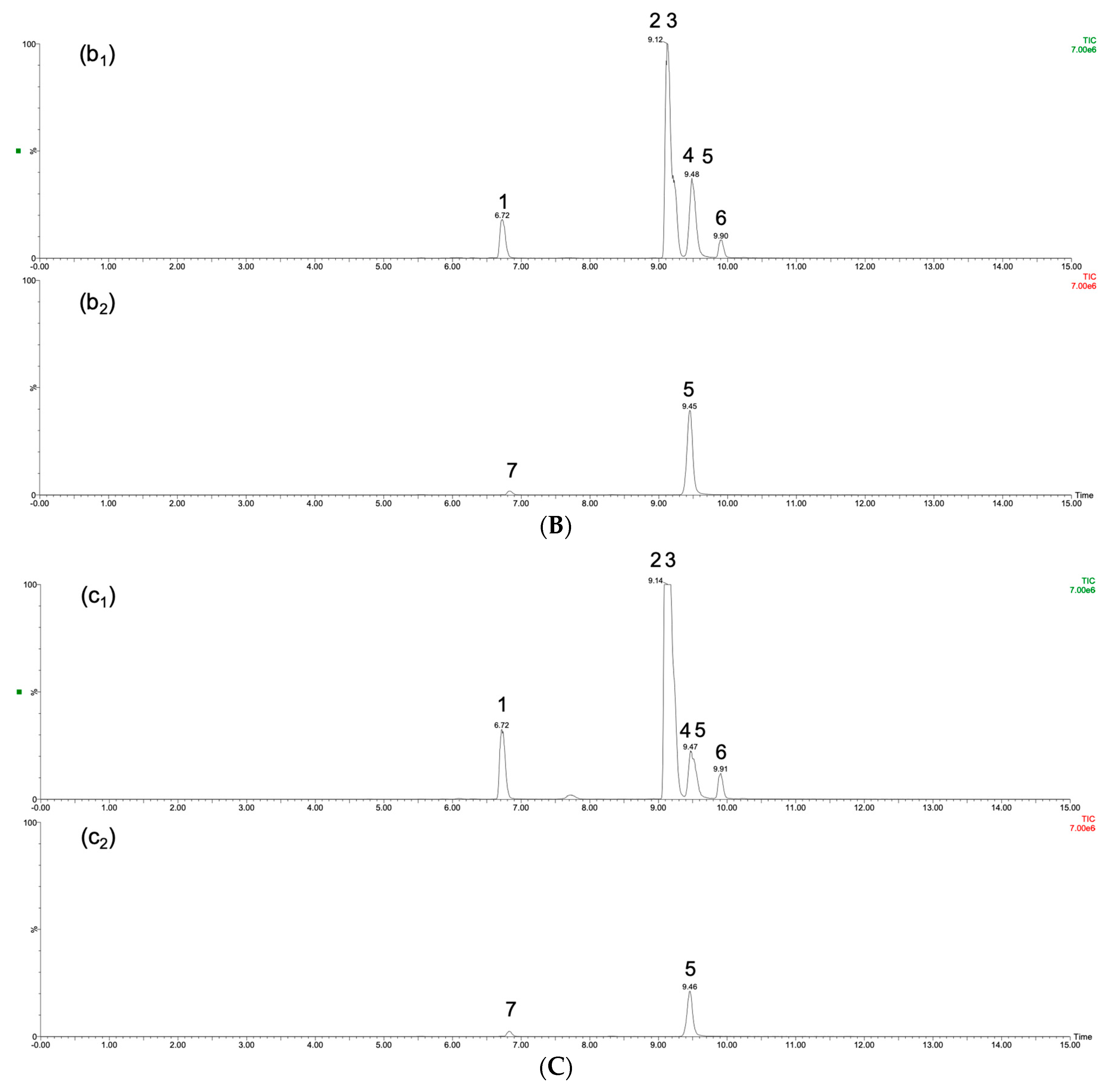
2.4. Ginseng Sample Quality
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Origin of Ginseng Samples
3.3. Preparation of Standard Solutions
3.4. Preparation of Ginseng Samples
3.5. Instruments and Conditions
3.6. Validation of the Analytical Method
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baeg, I.H.; So, S.H. The world ginseng market and the ginseng (Korea). J. Ginseng Res. 2013, 37, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.L.; He, Z.M.; Zhu, H.Y.; Gao, Y.G.; Zhao, Y.; Yang, H.; Zhang, L.X. Involvement of serotonergic, noradrenergic and dopaminergic systems in the antidepressant-like effect of ginsenoside Rb1, a major active ingredient of Panax ginseng C.A. Meyer. J. Ethnopharmacol. 2017, 204, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Qi, B.; Liu, L.; Zhang, H.; Zhou, G.X.; Wang, S.; Duan, X.Z.; Bai, X.Y.; Wang, S.M.; Zhao, D.Q. Anti-fatigue effects of proteins isolated from Panax quinquefolium. J. Ethnopharmacol. 2014, 153, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Pérez, Z.E.; Singh, P.; Kim, Y.J.; Mathiyalagan, R.; Kim, D.H.; Lee, M.H.; Yang, D.C. Applications of Panax ginseng leaves-mediated gold nanoparticles in cosmetics relation to antioxidant, moisture retention, and whitening effect on B16BL6 cells. J. Ginseng Res. 2017, 42, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Mucalo, I.; Jovanovski, E.; Rahelic, D.; Bozikov, V.; Romic, Z.; Vuksan, V. Effect of American ginseng (Panax quinquefolius L.) on arterial stiffness in subjects with type-2 diabetes and concomitant hypertension. J. Ethnopharmacol. 2013, 150, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Li, W.; Sha, Y.; Tezuka, Y.; Kadota, S.; Li, X. Triterpenoid saponins from leaves and stems of Panax quinquefolium L. J. Asian Nat. Prod. Res. 2001, 3, 123–130. [Google Scholar] [CrossRef] [PubMed]
- González-Burgos, E.; Fernandez-Moriano, C.; Gómez-Serranillos, M.P. Potential neuroprotective activity of Ginseng in Parkinson’s disease: A review. J. Neuroimmune Pharmacol. 2015, 10, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Choi, P.; Kim, T.; Ko, H.; Kim, H.K.; Kang, K.S.; Ham, J. Protective Effects of Processed Ginseng and Its Active Ginsenosides on Cisplatin-Induced Nephrotoxicity: In Vitro and in Vivo Studies. J. Agric. Food Chem. 2015, 63, 5964–5969. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yi, Y.S.; Kim, M.Y.; Cho, J.Y. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.S.; Lee, Y.; Lee, H.; Kim, J.; Kwon, B.R.; Yu, K.Y.; Cha, J.D.; Hwang, S.M.; Choi, K.M.; Jeong, Y.S. Fermented red ginseng extract inhibits cancer cell proliferation and viability. J. Med. Food 2015, 18, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Park, S.Y.; Yin, C.S.; Kim, H.T.; Kim, Y.M.; Yi, T.H. Antiaging effects of the mixture of Panax ginseng and Crataegus pinnatifida in human dermal fibroblasts and healthy human skin. J. Ginseng Res. 2017, 41, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Kim, J.H. A review on the medicinal potentials of ginseng and ginsenosides on cardiovascular diseases. J. Ginseng Res. 2014, 38, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Min, H. Ginseng, the ‘Immunity Boost’: The Effects of Panax ginseng on Immune System. J. Ginseng Res. 2012, 36, 354–368. [Google Scholar] [CrossRef] [PubMed]
- Nag, S.A.; Qin, J.J.; Wang, W.; Wang, M.H.; Wang, H.; Zhang, R. Ginsenosides as Anticancer Agents: In vitro and in vivo Activities, Structure-Activity Relationships, and Molecular Mechanisms of Action. Front. Pharmacol. 2012, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Sun, L.; Zhang, Z.; Guo, Y.; Liu, S. Profiling and multivariate statistical analysis of Panax ginseng based on ultra-high-performance liquid chromatography coupled with quadrupole-time-of-flight mass spectrometry. J. Pharm. Biomed. Anal. 2015, 107, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Ru, W.; Wang, D.; Xu, Y.; He, X.; Sun, Y.E.; Qian, L.; Zhou, X.; Qin, Y. Chemical constituents and bioactivities of Panax ginseng (C. A. Mey.). Drug Discov. Ther. 2015, 9, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.H.; Bae, O.N.; Park, J.H. Recent methodology in ginseng analysis. J. Ginseng Res. 2012, 36, 119–134. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.M.; Lee, S.D.; Kang, H.S.; Cho, M.G.; Hong, S.S.; Park, C.K.; Lee, J.T.; Jeon, B.S.; Ko, S.R.; Shon, H.J.; et al. Rapid and Simultaneous Determination of Ginsenosides Rb1, Rb2, Rc and Re in Korean Red Ginseng Extract by HPLC using Mass:Mass Spectrometry and UV Detection. J. Ginseng Res. 2008, 32, 390–396. [Google Scholar]
- Wang, H.P.; Zhang, Y.B.; Yang, X.W.; Zhao, D.Q.; Wang, Y.P. Rapid characterization of ginsenosides in the roots and rhizomes of Panax ginseng by UPLC-DAD-QTOF-MS/MS and simultaneous determination of 19 ginsenosides by HPLC-ESI-MS. J. Ginseng Res. 2016, 40, 382–394. [Google Scholar] [CrossRef] [PubMed]
- Pace, R.; Martinelli, E.M.; Sardone, N. Metabolomic evaluation of ginsenosides distribution in Panax genus (Panax ginseng and Panax quinquefolius) using multivariate statistical analysis. Fitoterapia 2015, 101, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Li, Y.; Li, X.; Ruan, C.C.; Wang, L.J.; Sun, G.Z. The effects of dynamic changes of malonyl ginsenosides on evaluation and quality control of Panax ginseng C.A. Meyer. J. Pharm. Biomed. Anal. 2012, 64–65, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Abozeid, A.; Zu, Y.G.; Tang, Z.H. The integration of GC-MS and LC-MS to assay the metabolomics profiling in Panax ginseng and Panax quinquefolius reveals a tissue- and species-specific connectivity of primary metabolites and ginsenosides accumulation. J. Pharm. Biomed. Anal. 2017, 135, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Song, F.; Guo, D.J.; Mi, J.; Qin, Q.J.; Yu, Q.; Liu, S.Y. Mass Spectrometry-Based Approach in Ginseng Research- A Promising Way to Metabolomic. Curr. Anal. Chem. 2012, 8, 43–66. [Google Scholar] [CrossRef]
- Wu, W.; Jiao, C.; Li, H.; Ma, Y.; Jiao, L.; Liu, S. LC-MS based metabolic and metabonomic studies of Panax ginseng. Phytochem. Anal. 2018, 29, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.Y.; Fan, Y.; Yu, Q.T.; Ge, Y.Z.; Yan, C.P.; Alolga, R.N.; Li, P.; Ma, Z.H.; Qi, L.W. Integrated evaluation of malonyl ginsenosides, amino acids and polysaccharides in fresh and processed ginseng. J. Pharm. Biomed. Anal. 2015, 107, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.L.; Pan, H.Q.; Wang, H.; Yao, S.; Yang, W.Z.; Hou, J.J.; Jin, Q.H.; Wu, W.Y.; Guo, D.A. Global profiling combined with predicted metabolites screening for discovery of natural compounds: Characterization of ginsenosides in the leaves of Panax notoginseng as a case study. J. Chromatogr. A 2018, 1538, 34–44. [Google Scholar] [CrossRef] [PubMed]
- He, Y.F.; Cai, H.Q.; Li, W.Y.; Xiu, Y.; Liu, W.L.; Chi, H.Y.; Shen, H.; Yang, M.G.; Pei, J.; Liu, S.Y. A discrimination study of Asia ginseng and America ginseng by a comparison of ginsenosides, oligosaccharides and amino acids using a UPLC-MS method. J. Liq. Chromatogr. Relat. Technol. 2019, 41, 825–830. [Google Scholar] [CrossRef]
- Yuan, Y.X.; Cheng, Y.J.; Ma, J.F.; Wang, Y.M.; Liang, Q.L.; Luo, G.A. Steaming-Induced Chemical Transformations and Holistic Quality Assessment of Red Ginseng Derived from Panax ginseng by Means of HPLC-ESI-MS/MSn-Based Multicomponent Quantification Fingerprint. J. Agric. Food Chem. 2012, 60, 8213–8224. [Google Scholar]
- Chen, Y.; Zhao, Z.; Chen, H.; Brand, E.; Yi, T.; Qin, M.; Liang, Z. Determination of ginsenosides in Asian and American ginsengs by liquid chromatography-quadrupole/time-of-flight MS: Assessing variations based on morphological characteristics. J. Ginseng Res. 2017, 41, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Kadian, N.; Raju, K.S.; Rashid, M.; Malik, M.Y.; Taneja, I.; Wahajuddin, M. Comparative assessment of bioanalytical method validation guidelines for pharmaceutical industry. J. Pharm. Biomed. Anal. 2016, 126, 83–97. [Google Scholar] [CrossRef] [PubMed]
- McEachran, A.D.; Newton, S.R.; Beverly, B.E.; Sobus, J.R.; Williams, A.J. A comparison of three liquid chromatography (LC) retention time prediction models. Talanta 2018, 182, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhao, Z.; Chen, H.; Yi, T.; Qin, M.; Liang, Z. Chemical Differentiation and Quality Evaluation of Commercial Asian and American Ginsengs based on a UHPLC-QTOF/MS/MS Metabolomics Approach. Phytochem. Anal. 2015, 26, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.K.; Ahn, M.S.; Park, J.S.; Liu, J.R.; In, D.S.; Min, B.W.; Kim, S.W. Discrimination of cultivation ages and cultivars of ginseng leaves using Fourier transform infrared spectroscopy combined with multivariate analysis. J. Ginseng Res. 2014, 38, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Choo, G.C.; Cho, H.S.; Lim, J.T. Soil properties of cultivation sites for mountain-cultivated ginseng at local level. J. Ginseng Res. 2015, 39, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Balan, P.; Popovich, D.G. Analysis of Ginsenoside Content (Panax ginseng) from Different Regions. Molecules 2019, 24, 3491. [Google Scholar] [CrossRef] [PubMed]
- Zhen, G.; Zhang, L.; Du, Y.; Yu, R.; Liu, X.; Cao, F.; Chang, Q.; Deng, X.; Xia, M.; He, H. De novo assembly and comparative analysis of root transcriptomes from different varieties of Panax ginseng C. A. Meyer grown in different environments. Sci. China Life Sci. 2015, 58, 1099–1110. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Wang, C.Z.; Zhou, C.J.; Wang, B.; Han, L.; Zhang, C.F.; Wu, X.H.; Yuan, C.S. Adulteration and cultivation region identification of American ginseng using HPLC coupled with multivariate analysis. J. Pharm. Biomed. Anal. 2014, 99, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Bae, B.S.; Park, H.W.; Ahn, N.G.; Cho, B.G.; Cho, Y.L.; Kwak, Y.S. Characterization of Korean Red Ginseng (Panax ginseng Meyer): History, preparation method, and chemical composition. J. Ginseng Res. 2015, 39, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Z.; Anderson, S.; Du, W.; He, T.C.; Yuan, C.S. Red ginseng and cancer treatment. Chin. J. Nat. Med. 2016, 14, 7–16. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds ginsenoside Rb1, ginsenoside Rb2, ginsenoside Rc, ginsenoside Rd, ginsenoside Re, and ginsenoside Rg1 are available from the authors. |
| Compound | Nominal Concentration (ng/mL) | Intraday | ||
|---|---|---|---|---|
| Observed Concentration (ng/mL) | Precision RSD (%) | Accuracy Bias (%) | ||
| ginsenoside Rb1 | 25 | 25.93 0.94 | 3.61 | 3.70 |
| 250 | 275.2 3.80 | 1.38 | 10.06 | |
| 500 | 500.7 31.42 | 6.27 | 0.13 | |
| ginsenoside Rb2 | 25 | 23.79 0.82 | 3.43 | −4.86 |
| 250 | 278.8 6.38 | 2.29 | 11.54 | |
| 500 | 458.8 29.00 | 6.32 | −8.52 | |
| ginsenoside Rc | 25 | 26.37 0.61 | 2.33 | 5.49 |
| 250 | 250.5 5.6 | 2.23 | 0.19 | |
| 500 | 464.5 31.02 | 6.68 | −7.11 | |
| ginsenoside Rd | 25 | 28.21 0.82 | 2.90 | 12.5 |
| 250 | 266.9 9.38 | 3.52 | 6.76 | |
| 500 | 480.3 23.44 | 4.88 | −3.94 | |
| ginsenoside Re | 25 | 28.22 0.70 | 2.50 | 12.88 |
| 250 | 277.6 9.19 | 3.31 | 11.06 | |
| 500 | 533.2 11.59 | 2.17 | 6.64 | |
| ginsenoside Rg1 | 25 | 22.78 0.59 | 2.58 | −8.86 |
| 250 | 259.1 3.87 | 1.49 | 3.65 | |
| 500 | 489.3 15.77 | 3.22 | −2.14 | |
| Ginsenoside | Structure Type | Molecular Formula | cLog P Value | No. of Sugars | Position of Sugar | Position of OH | Retention Time | |||
|---|---|---|---|---|---|---|---|---|---|---|
| C-3 | C-6 | C-20 | C-3 | C-12 | ||||||
| Re | panaxatriol | C48H82O18 | 2.18 | 3 | 0 | 2 | 1 | 1 | 1 | 6.75 |
| Rg1 | panaxatriol | C42H72O14 | 2.27 | 2 | 0 | 1 | 1 | 1 | 1 | 6.85 |
| Rb1 | panaxadiol | C54H92O23 | 1.64 | 4 | 2 | 0 | 2 | 0 | 1 | 9.21 |
| Rb2 | panaxadiol | C53H90O22 | 1.67 | 4 | 2 | 0 | 2 | 0 | 1 | 9.32 |
| Rc | panaxadiol | C53H90O22 | 2.29 | 4 | 2 | 0 | 2 | 0 | 1 | 9.55 |
| Rd | panaxadiol | C48H82O18 | 1.63 | 3 | 2 | 0 | 1 | 0 | 1 | 10.01 |
| Scientific Name (Common Name) | Cultivation Region | Cultivation Method | Sample No. | Rb1 (mg/g) | Rb2 (mg/g) | Rc (mg/g) | Rd (mg/g) | Re (mg/g) | Rg1 (mg/g) |
|---|---|---|---|---|---|---|---|---|---|
| P. ginseng C.A. Meyer (Korean ginseng) | Korea | Wood- cultivated | A | 10 ± 1 | 5 ± 1 | 5 ± 1 | 3 ± 1 | 1 ± 0.3 | 7 ± 1 |
| B | 6 ± 1 | 2 ± 0.4 | 2 ± 0.3 | 2 ± 0.2 | ND | 5 ± 1 | |||
| C | 16 ± 0.4 | 4 ± 0.1 | 3 ± 0.1 | 5 ± 0.1 | 1 ± 0.0 | 9 ± 1 | |||
| D | 9 ± 1 | 4 ± 0.4 | 4 ± 1 | 3 ± 1 | 0.1 ± 0.1 | 5 ± 0.4 | |||
| Mean | 10 ± 4 * | 4 ± 1 * | 3 ± 1 | 3 ± 1 * | 0.5 ± 0.4 * | 6 ± 2 | |||
| P. japonicus C.A. Meyer (Japanese ginseng) | Japan | Wood- cultivated | E | 7 ± 0.4 | 4 ± 0.2 | 3 ± 0.2 | 3 ± 0.3 | ND | 5 ± 1 |
| F | 3 ± 1 | 4 ± 1 | ND | 2 ± 0.4 | ND | ND | |||
| Mean | 5 ± 2 * | 4 ± 0.1 * | 2 ± 0.0 | 2 ± 1 * | ND | 3 ± 0.4 | |||
| P. quinquefolius L. (American ginseng) | Canada | Truly wild | G | 42 ± 1 | 1 ± 0.1 | 5 ± 0.1 | 43 ± 1 | 3 ± 0.2 | 7 ± 0.1 |
| United States | H | 41 ± 3 | 1 ± 0.1 | 5 ± 0.3 | 22 ± 1 | 9 ± 1 | 1 ± 0.1 | ||
| Wisconsin | I | 39 ± 2 | 0.1 ± 0.0 | 2 ± 0.2 | 31 ± 2 | 6 ± 1 | 3 ± 0.2 | ||
| Mean | 41 ± 1 * | 1 ± 0.3 * | 4 ± 2 | 32 ± 10 * | 6 ± 3 * | 3 ± 0.1 | |||
| Wood- cultivated | J | 51 ± 4 | 1 ± 0.1 | 5 ± 1 | 26 ± 2 | 3 ± 1 | 1 ± 0.1 | ||
| K | 46 ± 4 | 0.4 ± 0.1 | 3 ± 1 | 15 ± 2 | 3 ± 1 | 12 ± 4 | |||
| Mean | 48 ± 3 * | 1 ± 0.1 * | 4 ± 2 | 20 ± 7 * | 3 ± 0.2 * | 6 ± 8 | |||
| Wild- simulated | L | 4 ± 0.4 | 0.7 ± 0.1 | 1 ± 0.1 | 1 ± 0.2 | ND | 6 ± 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.; Li, C.-L.; Cheng, Y.-Y.; Tsai, T.-H. Development of a Validated UPLC-MS/MS Method for Analyzing Major Ginseng Saponins from Various Ginseng Species. Molecules 2019, 24, 4065. https://doi.org/10.3390/molecules24224065
Yang L, Li C-L, Cheng Y-Y, Tsai T-H. Development of a Validated UPLC-MS/MS Method for Analyzing Major Ginseng Saponins from Various Ginseng Species. Molecules. 2019; 24(22):4065. https://doi.org/10.3390/molecules24224065
Chicago/Turabian StyleYang, Ling, Chi-Lin Li, Yung-Yi Cheng, and Tung-Hu Tsai. 2019. "Development of a Validated UPLC-MS/MS Method for Analyzing Major Ginseng Saponins from Various Ginseng Species" Molecules 24, no. 22: 4065. https://doi.org/10.3390/molecules24224065
APA StyleYang, L., Li, C.-L., Cheng, Y.-Y., & Tsai, T.-H. (2019). Development of a Validated UPLC-MS/MS Method for Analyzing Major Ginseng Saponins from Various Ginseng Species. Molecules, 24(22), 4065. https://doi.org/10.3390/molecules24224065







