Level of Biogenic Amines in Red and White Wines, Dietary Exposure, and Histamine-Mediated Symptoms upon Wine Ingestion
Abstract
1. Introduction
2. Results and Discussion
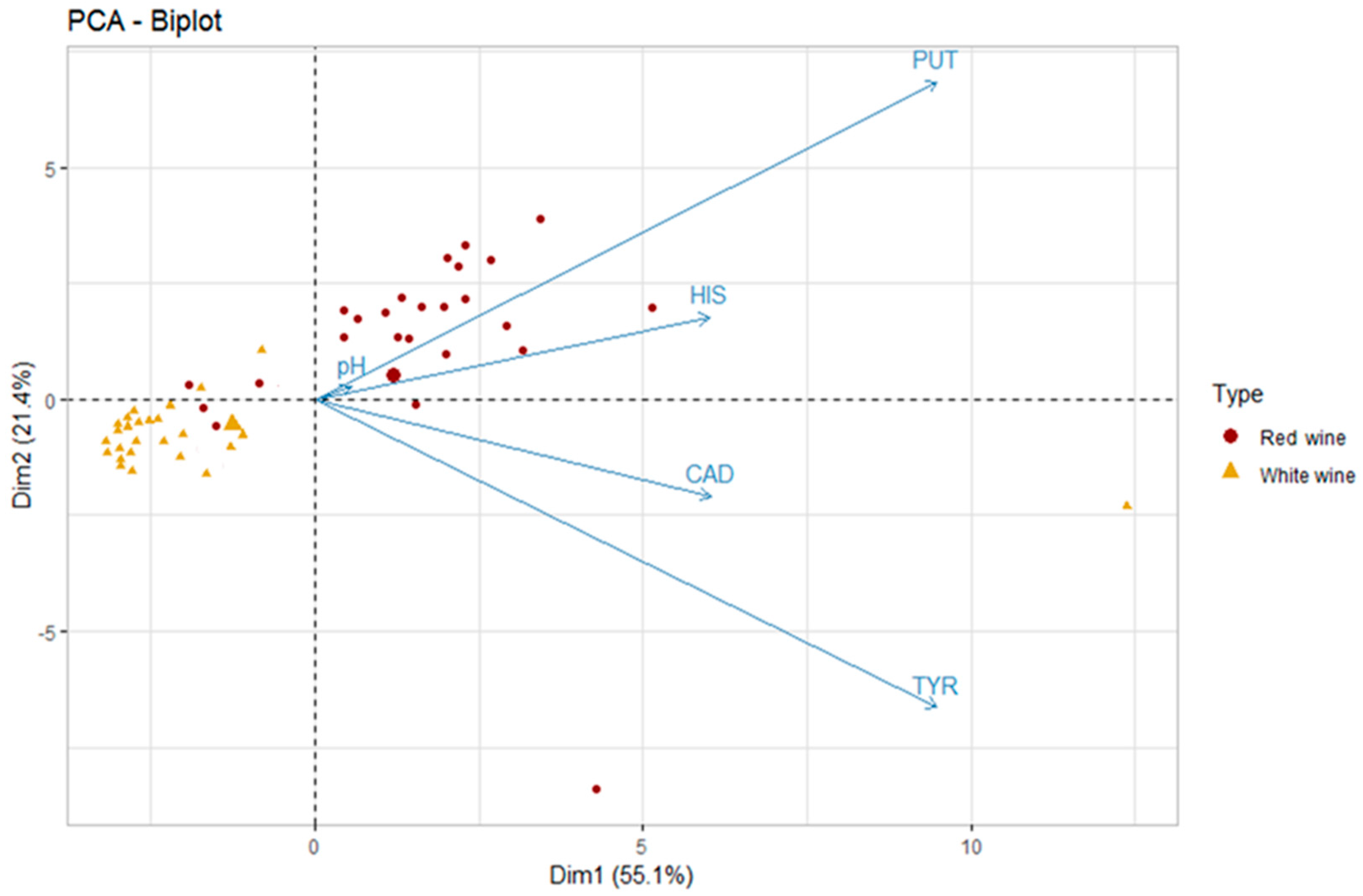
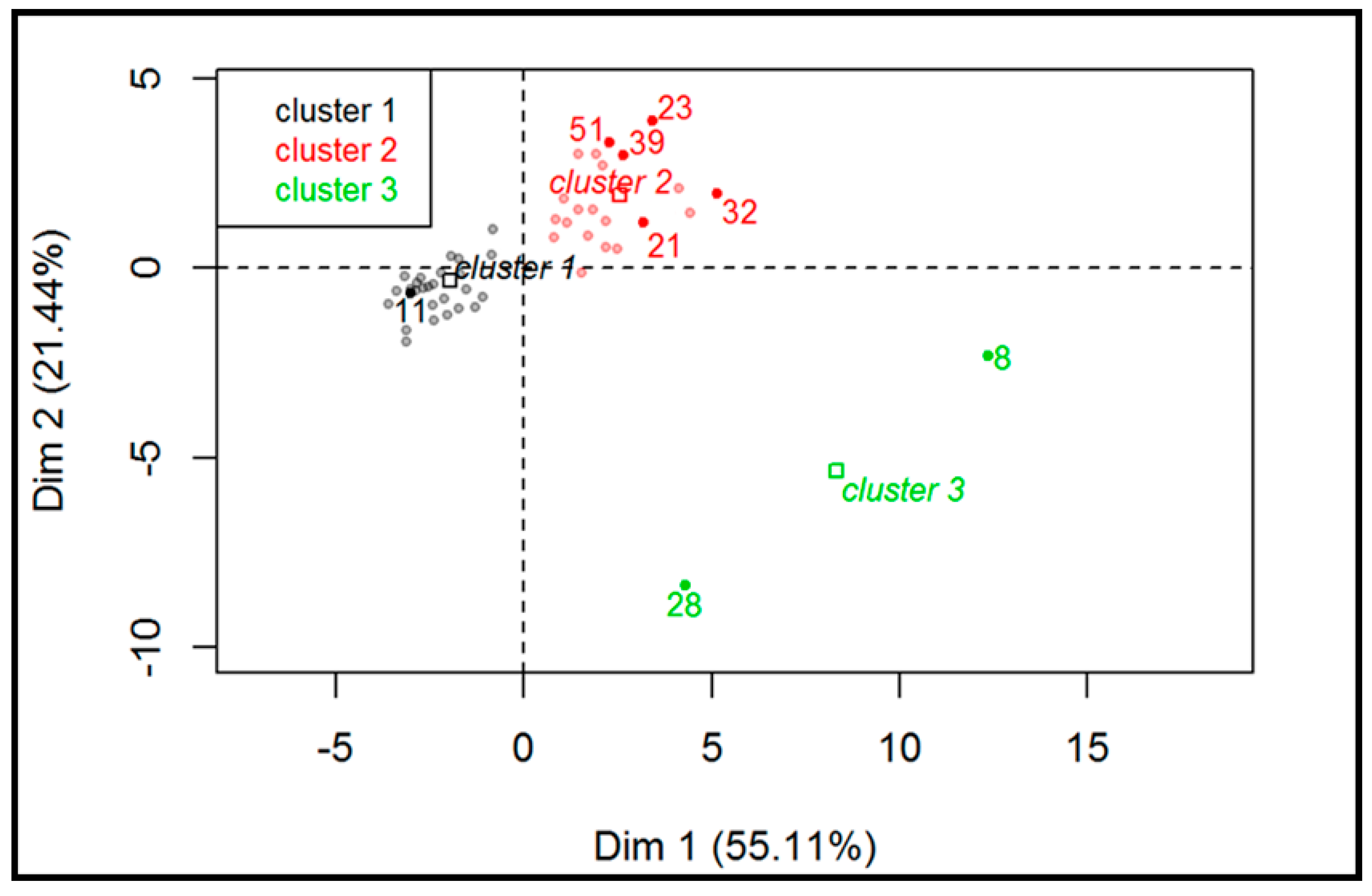
2.1. Principal Components Analysis
- Value of the jth sample in the ith column of the dataset
- Mean value of the ith column of the dataset
- Standard deviation of the ith column of the dataset
2.2. Exposure to BAs through Consumption of Wine
- DI = Daily intake of BAs (mg/day)
- C = The 50th and 95th percentile concentration of BAs detected in the samples (mg/L).
- Q = Individual wine daily consumption of population within different age groups and for median and 95th percentile consumers (L/day).
2.3. Case Report
3. Materials and Methods
3.1. Sampling and Extraction
3.2. Chemicals and Reagents
3.3. Derivatization
3.4. Chromatographic Conditions
3.5. Recoveries, Determination and Quantification Limits
3.6. Statistical Analysis
3.7. Case Study
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Greifová, G.; Májeková, H.; Greif, G.; Body, P.; Greifová, M.; Dubničková, M. Analysis of antimicrobial and immunomodulatory substances produced by heterofermentative Lactobacillus reuteri. Folia Microbiol. 2017, 62, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Peña-Gallego, A.; Hernández-Orte, P.; Cacho, J.; Ferreira, V. High-Performance Liquid Chromatography Analysis of Amines in Must and Wine: A Review. Food Rev. Int. 2012, 28, 71–96. [Google Scholar] [CrossRef]
- Chong, C.Y.; Bakar, F.A.; Russly, A.R.; Jamilah, B.; Mahyudin, N.A. The effects of food processing on biogenic amines formation. Int. Food Res. J. 2011, 18, 867–876. [Google Scholar]
- Park, J.S.; Lee, C.H.; Kwon, E.Y.; Lee, H.J.; Kim, J.Y.; Kim, S.H. Monitoring the contents of biogenic amines in fish and fish products consumed in Korea. Food Control 2010, 21, 1219–1226. [Google Scholar] [CrossRef]
- McCabe-Sellers, B.; Frankel, E.H.; Wolfe, J.J. Handbook of Food-Drug Interactions; CRC press: Boca Raton, FL, USA, 2003. [Google Scholar]
- González-Fernández, C.; Santos, E.M.; Jaime, I.; Rovira, J. Influence of starter cultures and sugar concentrations on biogenic amine contents in chorizo dry sausage. Food Microbiol. 2003, 20, 275–284. [Google Scholar] [CrossRef]
- Russo, P.; Spano, G.; Arena, M.P.; Capozzi, V.; Fiocco, D.; Grieco, F.; Beneduce, L. Are consumers aware of the risks related to biogenic amines in food. Curr Res. Technol. Edu. Top. Appl. Microbiol. Microb. Biotechnol. 2010, 1087–1095. [Google Scholar]
- Alvarez, M.A.; Moreno-Arribas, M.V. The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends Food Sci. Technol. 2014, 39, 146–155. [Google Scholar] [CrossRef]
- Wei, F.; Xu, X.; Zhou, G.; Zhao, G.; Li, C.; Zhang, Y.; Chen, L.; Qi, J. Irradiated Chinese Rugao ham: Changes in volatile N-nitrosamine, biogenic amine and residual nitrite during ripening and post-ripening. Meat Sci. 2009, 81, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Linares, D.M.; Martín, M.; Ladero, V.; Alvarez, M.A.; Fernández, M. Biogenic Amines in Dairy Products. Crit. Rev. Food. Sci. Nutr. 2011, 51, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Prester, L. Biogenic Amines in Ready-to-Eat Foods; Academic Press: San Diego, CA, USA, 2016; ISBN 9780128020081. [Google Scholar]
- Prester, L. Biogenic amines in fish, fish products and shellfish: A review. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2011, 28, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- EFSA Scientific Opinion on risk based control of biogenic amine formation in fermented foods. EFSA J. 2011, 9, 1–93.
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Esselen, M.; Schrenk, D. Chapter 11—Other Process-Generated Toxicants in Foods. In Chemical Contaminants and Residues in Food, 2nd ed.; Schrenk, D., Cartus, A., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 223–249. ISBN 978-0-08-100674-0. [Google Scholar]
- Toro-Funes, N.; Bosch-Fuste, J.; Latorre-Moratalla, M.L.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Biologically active amines in fermented and non-fermented commercial soybean products from the Spanish market. Food Chem. 2015, 173, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Silla Santos, M.H. Biogenic amines: Their importance in foods. Int. J. Food Microbiol. 1996, 29, 213–231. [Google Scholar] [CrossRef]
- Schirone, M.; Visciano, P.; Tofalo, R.; Suzzi, G. Histamine Food Poisoning. In Histamine and Histamine Receptors in Health and Disease; Hattori, Y., Seifert, R., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 217–235. ISBN 978-3-319-58194-1. [Google Scholar]
- Konakovsky, V.; Focke, M.; Hoffmann-Sommergruber, K.; Schmid, R.; Scheiner, O.; Moser, P.; Jarisch, R.; Hemmer, W. Levels of histamine and other biogenic amines in high-quality red wines. Food Addit. Contam. Part A 2011, 28, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Bieber, T.; Novak, N. Histamine intolerance in clinical practice. Dtsch Arztebl 2006, 103, 3477–3483. [Google Scholar]
- Sattler, J.; Häfner, D.; Klotter, H.-J.; Lorenz, W.; Wagner, P.K. Food-induced histaminosis as an epidemiological problem: Plasma histamine elevation and haemodynamic alterations after oral histamine administration and blockade of diamine oxidase (DAO). Agents Actions 1988, 23, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Barone, C.; Barbera, M.; Barone, M.; Parisi, S.; Zaccheo, A. Biogenic Amines in Cheeses: Types and Typical Amounts. In Chemical Evolution of Nitrogen-Based Compounds in Mozzarella Cheeses; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–18. ISBN 978-3-319-65739-4. [Google Scholar]
- Menne, A.; Bodmer, S.; Amon, U. Der Sektprovokationstest in der Diagnostik einer enteralen Histaminose TT - null. Akt Dermatol 2001, 27, 58–62. [Google Scholar] [CrossRef]
- Önal, A.; Tekkeli, S.E.K.; Önal, C. A review of the liquid chromatographic methods for the determination of biogenic amines in foods. Food Chem. 2013, 138, 509–515. [Google Scholar] [CrossRef]
- Benkerroum, N. Biogenic Amines in Dairy Products: Origin, Incidence, and Control Means. Compr. Rev. Food Sci. Food Saf. 2016, 15, 801–826. [Google Scholar] [CrossRef]
- Bulushi, I.A.; Poole, S.; Deeth, H.C.; Dykes, G.A. Biogenic Amines in Fish: Roles in Intoxication, Spoilage, and Nitrosamine Formation—A Review. Crit. Rev. Food Sci. Nutr. 2009, 49, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Önal, A. A review: Current analytical methods for the determination of biogenic amines in foods. Food Chem. 2007, 103, 1475–1486. [Google Scholar] [CrossRef]
- De Borba, B.M.; Rohrer, J.S. Determination of biogenic amines in alcoholic beverages by ion chromatography with suppressed conductivity detection and integrated pulsed amperometric detection. J. Chromatogr. A 2007, 1155, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Park, H.K.; Kim, J.K.; Kim, M. Determination of biogenic amines in Korean traditional fermented soybean paste (Doenjang). Food Chem. Toxicol. 2010, 48, 1191–1195. [Google Scholar] [CrossRef] [PubMed]
- Spano, G.; Russo, P.; Lonvaud-Funel, A.; Lucas, P.; Alexandre, H.; Grandvalet, C.; Coton, E.; Coton, M.; Barnavon, L.; Bach, B.; et al. Biogenic amines in fermented foods. Eur. J. Clin. Nutr. 2010, 64, S95–S100. [Google Scholar] [CrossRef] [PubMed]
- Martuscelli, M.; Mastrocola, D. Biogenic Amines: A Claim for Wines. In Biogenic Amines; Proestos, C., Ed.; IntechOpen: Rijeka, Croatia, 2019. [Google Scholar]
- Tuberoso, C.I.G.; Congiu, F.; Serreli, G.; Mameli, S. Determination of dansylated amino acids and biogenic amines in Cannonau and Vermentino wines by HPLC-FLD. Food Chem. 2015, 175, 29–35. [Google Scholar] [CrossRef]
- Martuscelli, M.; Arfelli, G.; Manetta, A.C.; Suzzi, G. Biogenic amines content as a measure of the quality of wines of Abruzzo (Italy). Food Chem. 2013, 140, 590–597. [Google Scholar] [CrossRef]
- Ancín-Azpilicueta, C.; González-Marco, A.; Jiménez-Moreno, N. Current Knowledge about the Presence of Amines in Wine. Crit. Rev. Food Sci. Nutr. 2008, 48, 257–275. [Google Scholar] [CrossRef]
- Polo, L.; Ferrer, S.; Peña-Gallego, A.; Hernández-Orte, P.; Pardo, I. Biogenic amine synthesis in high quality Tempranillo wines. Relationship with lactic acid bacteria and vinification conditions. Ann. Microbiol. 2011, 61, 191–198. [Google Scholar] [CrossRef]
- Henríquez-Aedo, K.; Durán, D.; Garcia, A.; Hengst, M.B.; Aranda, M. Identification of biogenic amines-producing lactic acid bacteria isolated from spontaneous malolactic fermentation of chilean red wines. LWT-Food Sci. Technol. 2016, 68, 183–189. [Google Scholar] [CrossRef]
- Ancín-Azpilicueta, C.; Nieto-Rojo, R.; Gómez-Cordón, J. Influence of fertilisation with foliar urea on the content of amines in wine. Food Addit. Contam. Part A 2011, 28, 877–884. [Google Scholar] [CrossRef] [PubMed]
- EC Commission regulation (EC) No 2073/2005 of 15 November 2005 on microbial criteria for foodstuffs. Off. J. Eur. Union 2005, L338, 1–26. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32005R2073&from=EN (accessed on 8 August 2019).
- EC Commission regulation (EC) n°1019/2013 of 23 October 2013 amending Annex I to Regulation (EC) n°2073/2005 as regards histamine in fishery products. Off. J. Eur. Union 2013, 9, 46–47. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32013R1019&from=EN (accessed on 8 August 2019).
- Bover-Cid, S.; Iquierdo-Pulido, M.; Mariné-Font, A.; Vidal-Carou, M.C. Biogenic mono-, di- and polyamine contents in Spanish wines and influence of a limited irrigation. Food Chem. 2006, 96, 43–47. [Google Scholar] [CrossRef]
- Costantini, A.; Pietroniro, R.; Doria, F.; Pessione, E.; Garcia-Moruno, E. Putrescine production from different amino acid precursors by lactic acid bacteria from wine and cider. Int. J. Food Microbiol. 2013, 165, 11–17. [Google Scholar] [CrossRef] [PubMed]
- La Torre, G.L.; Rando, R.; Saitta, M.; Alfa, M.; Maisano, R.; Dugo, G. Determination of biogenic amine and heavy metal contents in sicilian wine samples. Ital. J. Food Sci. 2010, 22, 28–40. [Google Scholar]
- Leclercq, C.; Arcella, D.; Piccinelli, R.; Sette, S.; Le Donne, C.; Turrini, A. The Italian National Food Consumption Survey INRAN-SCAI 2005-06: Main results in terms of food consumption. Public Health Nutr. 2009, 12, 2504–2532. [Google Scholar] [CrossRef]
- Preti, R.; Antonelli, M.L.; Bernacchia, R.; Vinci, G. Fast determination of biogenic amines in beverages by a core-shell particle column. Food Chem. 2015, 187, 555–562. [Google Scholar] [CrossRef]
- Chiacchierini, E.; Restuccia, D.; Vinci, G. Evaluation of two different extraction methods for chromatographic determination of bioactive amines in tomato products. Talanta 2006, 69, 548–555. [Google Scholar] [CrossRef]
- R Core Team. R: A language and environment for statistical computing. 2019. Available online: https://www.r-project.org/ (accessed on 8 August 2019).
- Lê, S.; Josse, J.; Husson, F. FactoMineR: A Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Kassambara, A.; Mundt, F. factoextra: Extract and Visualize the Results of Multivariate Data Analyses 2017. Available online: https://CRAN.R-project.org/package=factoextra (accessed on 8 August 2019).
- Thuleau, S.; Husson, F. FactoInvestigate: Automatic Description of Factorial Analysis 2018. Available online: https://CRAN.R-project.org/package=FactoInvestigate (accessed on 8 August 2019).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer-Verlag: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Kassambara, A. ggpubr: “ggplot2” Based Publication Ready Plots 2018. Available online: https://CRAN.R-project.org/package=ggpubr (accessed on 8 August 2019).
Sample Availability: Samples of the biogenic amines are available from the authors. |
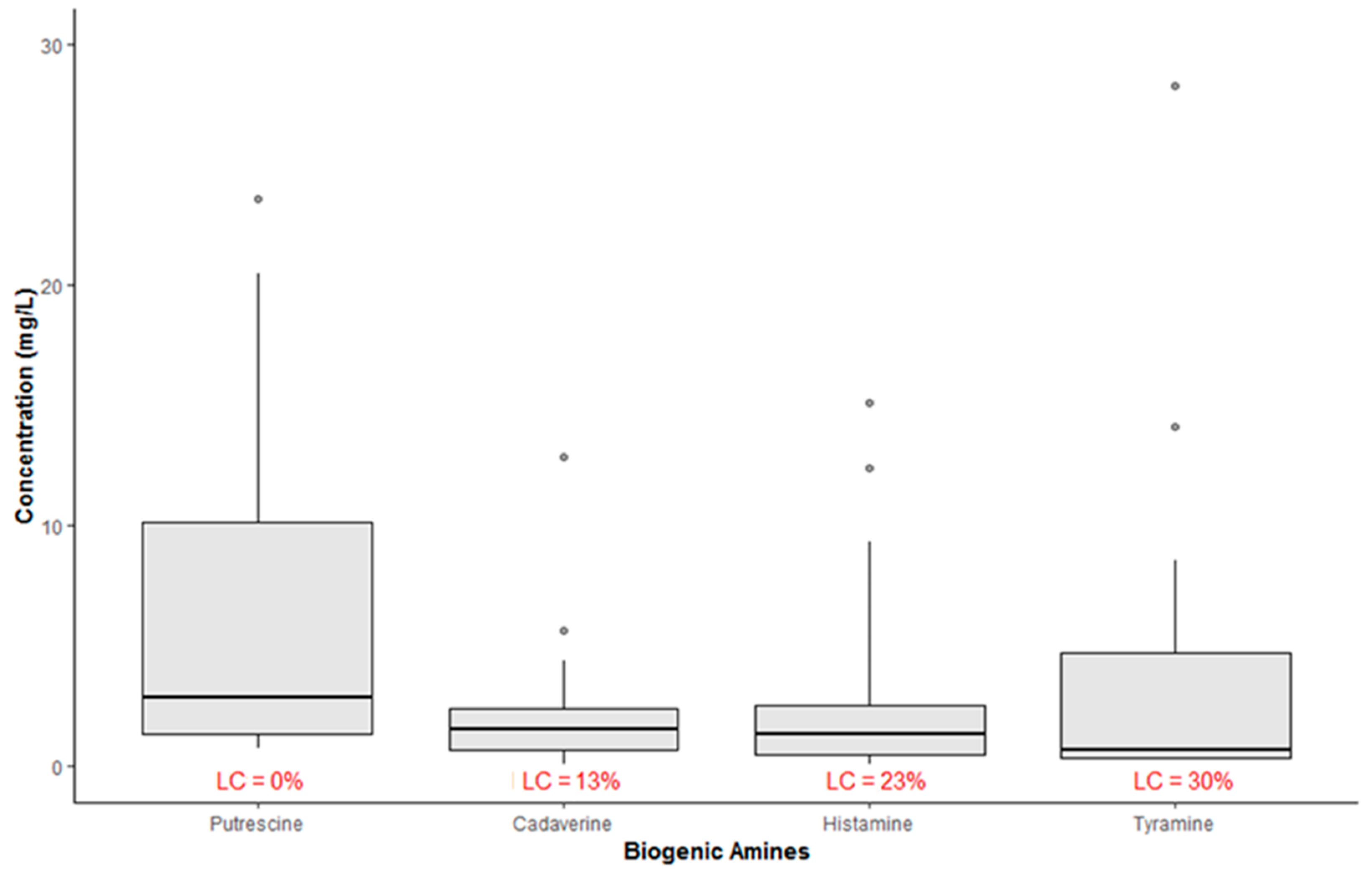
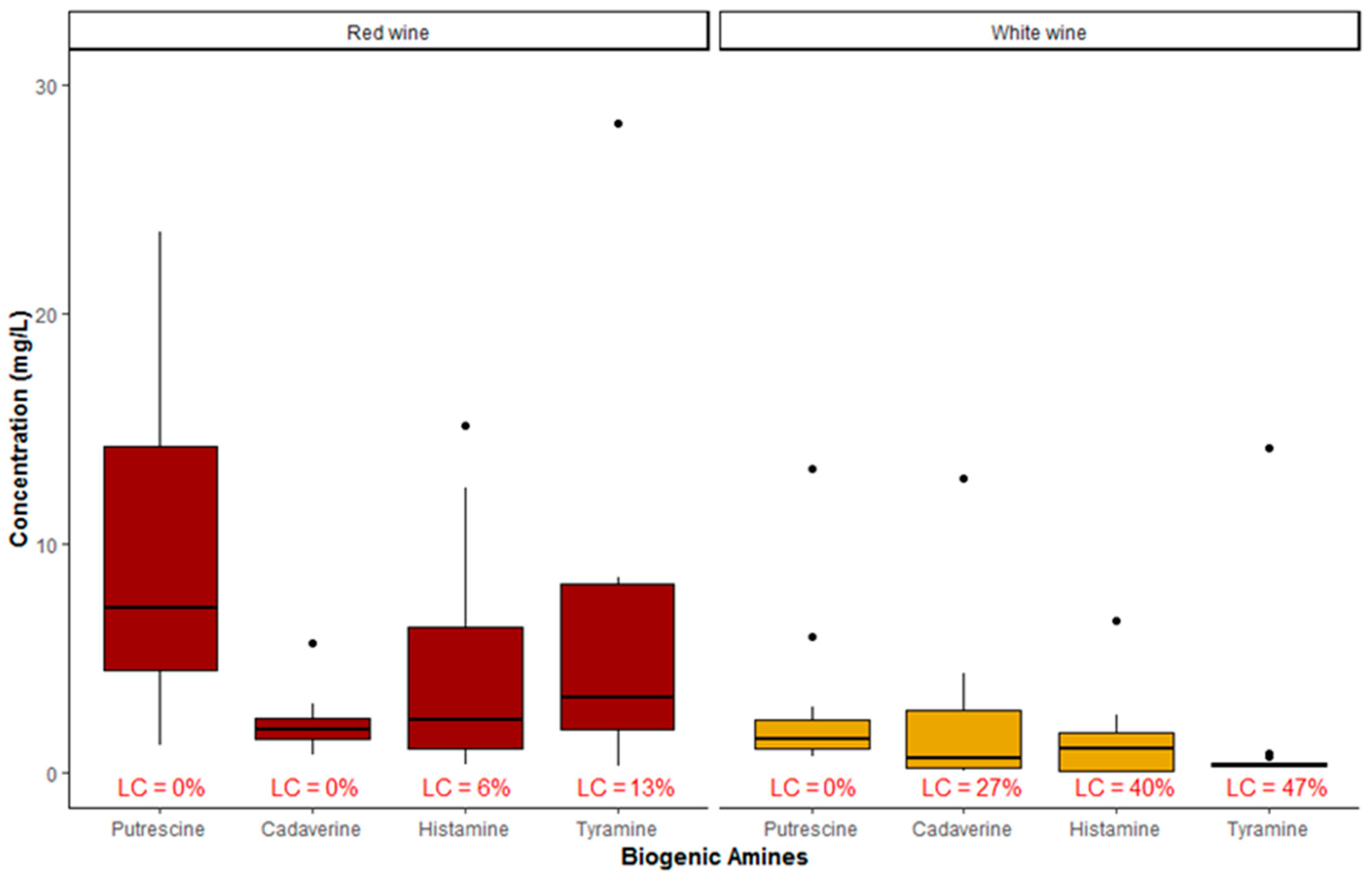
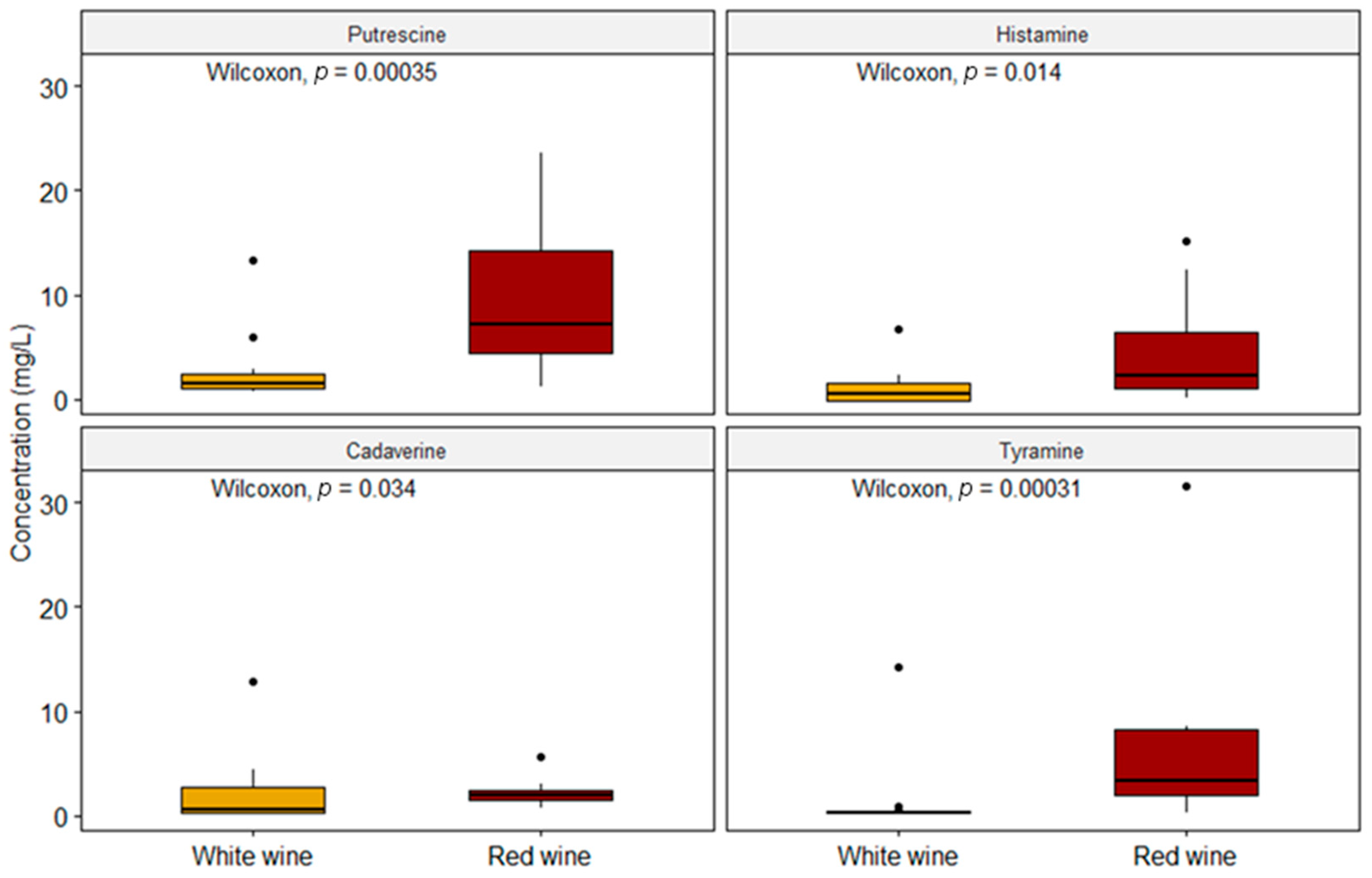
| BA | This Study | EFSA, 2011 [13] | Tuberoso et al. (2014) [32] | Martuscelli et al. (2013) [33] | Bover-Cid et al. (2006) [40] | |||
|---|---|---|---|---|---|---|---|---|
| Mean | Median | 95th pctl | Mean | 95th pctl | Mean | Mean | Mean | |
| Putrescine | 1.85 | 1.55 | 3.96 | 1.4–1.5 | 3.9–4.4 | 5.96 | 2.24 | 4.00 |
| Cadaverine | 1.49 | 0.92 | 4.36 | 0.1–0.2 | 0.3–0.4 | 2.06 | 0.79 | 0.10 |
| Histamine | 0.76 | 0.28 | 2.41 | 0.8–0.9 | 2.6 | ND | 0.18 | 0.20 |
| Tyramine | 0.38 | 0.31 | 0.78 | 1.1–1.2 | 4.3–4.5 | NQ | 0.41 | 0.20 |
| BA | This Study | EFSA, 2011 [13] | Tuberoso et al. (2014) [32] | Martuscelli et al. (2013) [33] | Konakovsy et al. (2011) [19] | Bover-Cid et al. (2006) [40] | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Median | 95th pctl | Mean | 95th pctl | Mean | Mean | Median | Mean | |
| Putrescine | 9.98 | 7.30 | 20.31 | 4.2–4.8 | 9.5–11.5 | 20.50 | 7.88 | 19.4 | 27.90 |
| Cadaverine | 1.71 | 1.93 | 4.25 | 0.2–0.5 | 0.6–1.6 | 2.13 | 0.11 | 0.58 | 0.20 |
| Histamine | 2.36 | 2.45 | 9.32 | 3.6–3.7 | 12.3–12.4 | 6.61 | 2.91 | 7.20 | 3.90 |
| Tyramine | 3.43 | 3.20 | 8.24 | 2.7–2.9 | 7.8–8.5 | 9.06 | 5.22 | 3.52 | 3.30 |
| Gender | BA | Adolescent (10–18 Aged) | Adult (18–65 Aged) | Elderly (>65 Aged) | |||
|---|---|---|---|---|---|---|---|
| Best Case | Worst Case | Best Case | Worst Case | Best Case | Worst Case | ||
| Males | PUT | ≤0.001 | 0.002 | 0.279 | 1.808 | 0.463 | 3.002 |
| CAD | ≤0.001 | ≤0.001 | 0.155 | 0.437 | 0.257 | 0.725 | |
| HIS | ≤0.001 | ≤0.001 | 0.128 | 0.932 | 0.212 | 1.547 | |
| TYR | ≤0.001 | ≤0.001 | 0.062 | 0.842 | 0.103 | 1.397 | |
| Females | PUT | ≤0.001 | 0.002 | 0.056 | 0.363 | 0.167 | 1.085 |
| CAD | ≤0.001 | ≤0.001 | 0.031 | 0.088 | 0.093 | 0.262 | |
| HIS | ≤0.001 | ≤0.001 | 0.026 | 0.187 | 0.077 | 0.559 | |
| TYR | ≤0.001 | ≤0.001 | 0.012 | 0.169 | 0.037 | 0.505 | |
| Gender | BA | Adolescent (10–18 Aged) | Adult (18–65 Aged) | Elderly (>65 Aged) | |||
|---|---|---|---|---|---|---|---|
| Best Case | Worst Case | Best Case | Worst Case | Best Case | Worst Case | ||
| Males | PUT | 0.112 | 0.723 | 1.116 | 7.237 | 1.339 | 8.680 |
| CAD | 0.062 | 0.175 | 0.619 | 1.747 | 0.743 | 2.096 | |
| HIS | 0.051 | 0.373 | 0.512 | 3.729 | 0.614 | 4.473 | |
| TYR | 0.025 | 0.337 | 0.248 | 3.368 | 0.297 | 4.040 | |
| Females | PUT | ≤0.001 | 0.004 | 0.613 | 3.978 | 0.781 | 5.063 |
| CAD | ≤0.001 | ≤0.001 | 0.340 | 0.960 | 0.433 | 1.222 | |
| HIS | ≤0.001 | 0.002 | 0.282 | 2.050 | 0.358 | 2.609 | |
| TYR | ≤0.001 | 0.002 | 0.136 | 1.852 | 0.173 | 2.357 | |
| Compound | Concentration (mg/L) |
|---|---|
| Putrescine | 13.01 ± 0.30 |
| Cadaverine | 1.51 ± 0.17 |
| Histamine | 9.97 ± 0.21 |
| Tyramine | 8.23 ± 0.22 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, F.; Montuori, P.; Schettino, M.; Velotto, S.; Stasi, T.; Romano, R.; Cirillo, T. Level of Biogenic Amines in Red and White Wines, Dietary Exposure, and Histamine-Mediated Symptoms upon Wine Ingestion. Molecules 2019, 24, 3629. https://doi.org/10.3390/molecules24193629
Esposito F, Montuori P, Schettino M, Velotto S, Stasi T, Romano R, Cirillo T. Level of Biogenic Amines in Red and White Wines, Dietary Exposure, and Histamine-Mediated Symptoms upon Wine Ingestion. Molecules. 2019; 24(19):3629. https://doi.org/10.3390/molecules24193629
Chicago/Turabian StyleEsposito, Francesco, Paolo Montuori, Mario Schettino, Salvatore Velotto, Tommaso Stasi, Raffaele Romano, and Teresa Cirillo. 2019. "Level of Biogenic Amines in Red and White Wines, Dietary Exposure, and Histamine-Mediated Symptoms upon Wine Ingestion" Molecules 24, no. 19: 3629. https://doi.org/10.3390/molecules24193629
APA StyleEsposito, F., Montuori, P., Schettino, M., Velotto, S., Stasi, T., Romano, R., & Cirillo, T. (2019). Level of Biogenic Amines in Red and White Wines, Dietary Exposure, and Histamine-Mediated Symptoms upon Wine Ingestion. Molecules, 24(19), 3629. https://doi.org/10.3390/molecules24193629









