Plants are Capable of Synthesizing Animal Steroid Hormones
Abstract
:1. Introduction
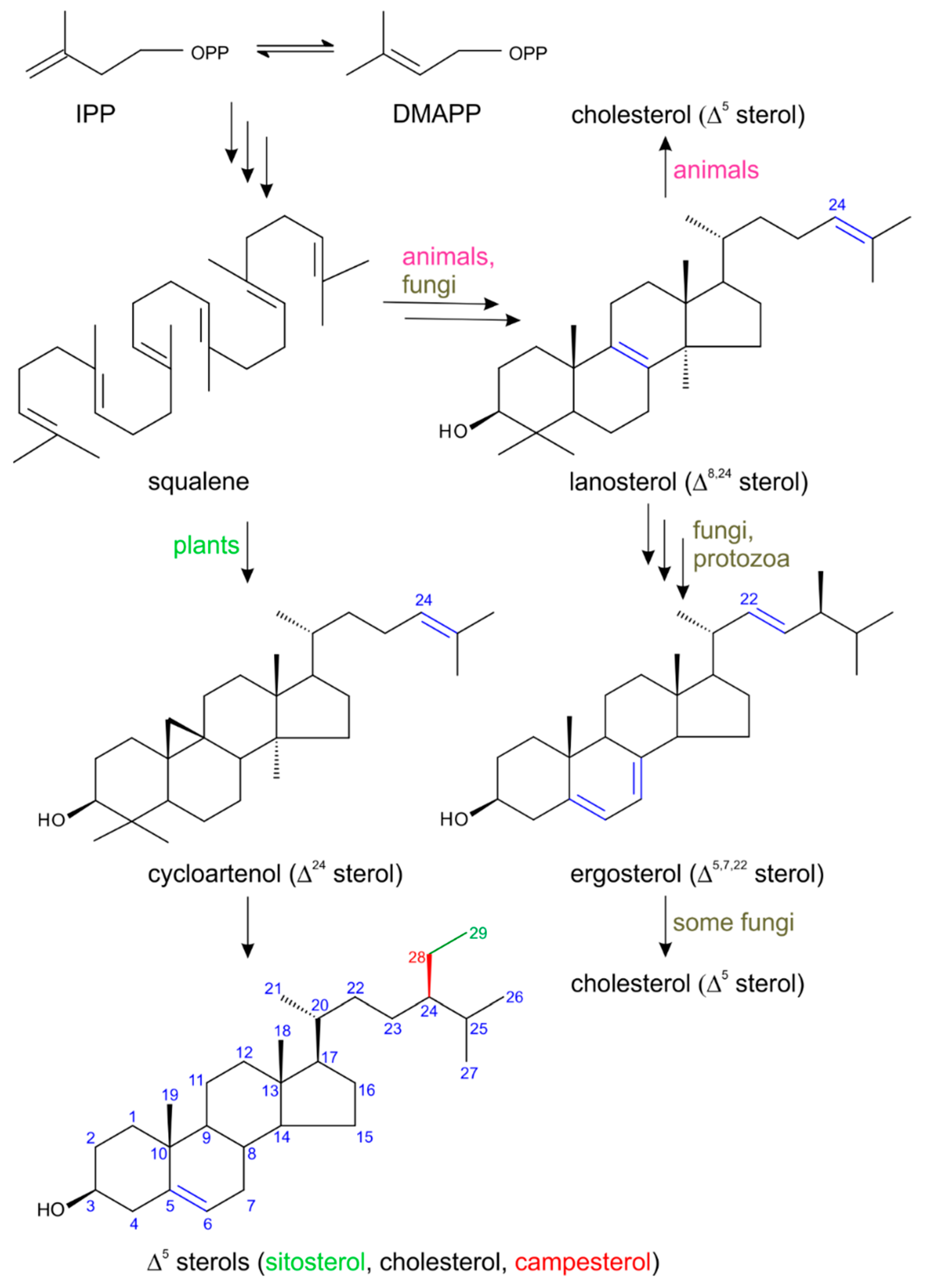
2. Biosynthesis of Plant Sterols with Respect to Steroid Hormone Formation
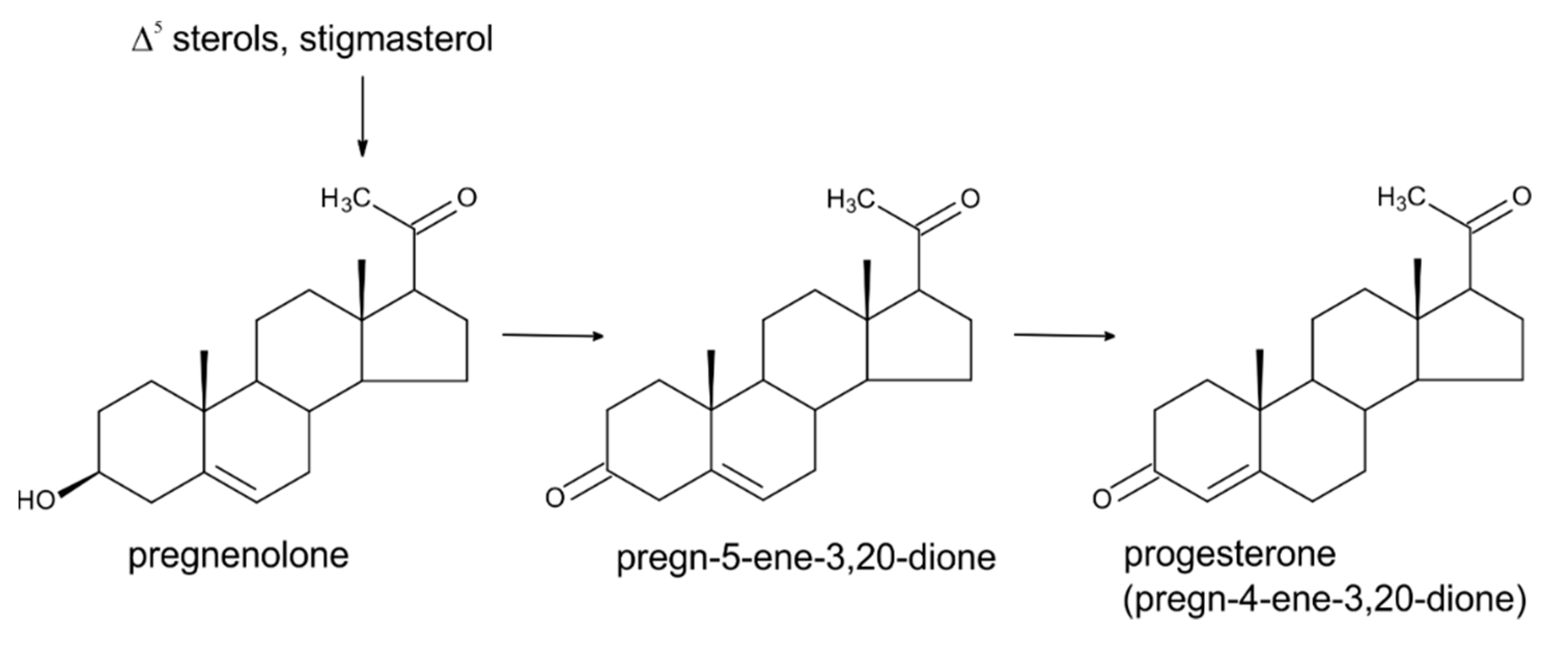
3. Progesterone
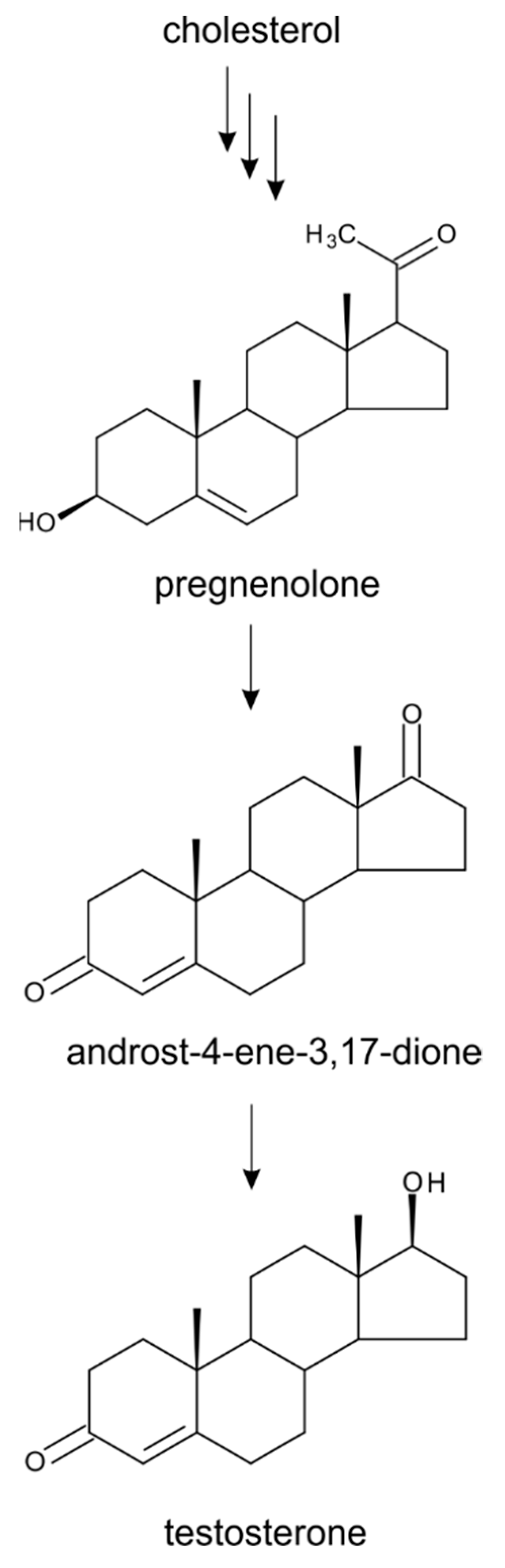
4. Testosterone
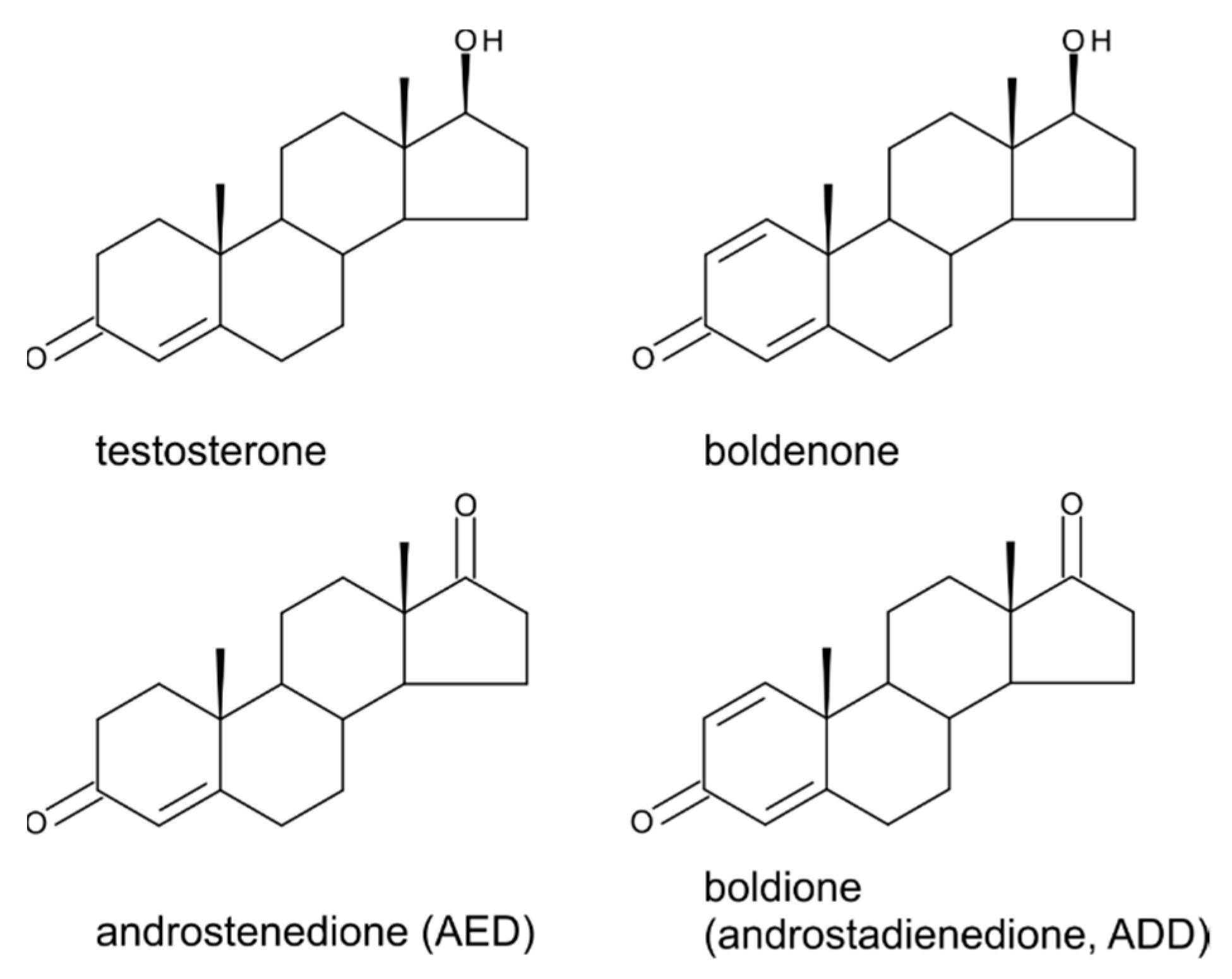
5. Boldenone, Boldione, and Other Testosterone Derivatives
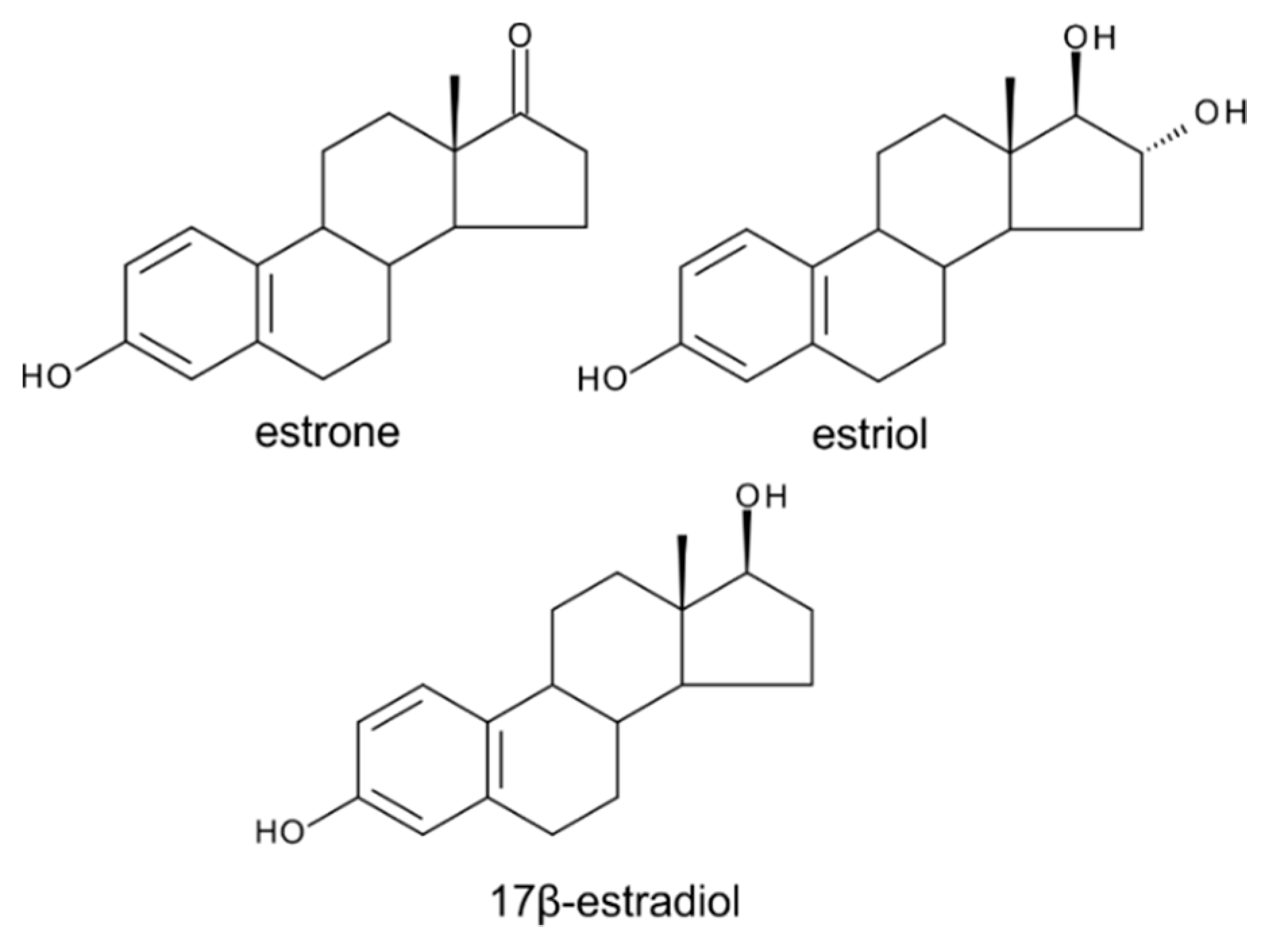
6. Estrogens
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tarkowská, D.; Strnad, M. Isoprenoid-derived plant signaling molecules: Biosynthesis and biological importance. Planta 2018, 247, 1051–1066. [Google Scholar] [CrossRef] [PubMed]
- Behmer, S.T.; Nes, W.D. Insect sterol nutrition and physiology: A global overview. Adv. Insect Physiol. 2003, 31, 1–72. [Google Scholar] [CrossRef]
- Lindsey, K.; Pullen, M.L.; Topping, J.F. Importance of plant sterols in pattern formation and hormone signalling. Trends Plant Sci. 2003, 8, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Espenshade, P.J.; Hughes, A.L. Regulation of sterol synthesis in eukaryotes. Annu. Rev. Genet. 2007, 41, 401–427. [Google Scholar] [CrossRef]
- Silvestro, D.; Andersen, T.G.; Schaller, H.; Jensen, P.E. Plant Sterol Metabolism. Δ7-Sterol-C5-Desaturase (STE1/DWARF7), Δ5,7-Sterol-Δ7-Reductase (DWARF5) and Δ24-Sterol-Δ24-Reductase (DIMINUTO/DWARF1) Show Multiple Subcellular Localizations in Arabidopsis thaliana (Heynh) L. PLoS ONE 2013, 8, e56429. [Google Scholar] [CrossRef] [PubMed]
- Nes, W.D. Biosynthesis of Cholesterol and Other Sterols. Chem. Rev. 2011, 111, 6423–6451. [Google Scholar] [CrossRef]
- Hartmann, M.-A. Plant sterols and the membrane environment. Trends Plant Sci. 1998, 3, 170–175. [Google Scholar] [CrossRef]
- Goldstein, J.L.; Brown, M.S. Regulation of the mevalonate pathway. Nature 1990, 343, 425–430. [Google Scholar] [CrossRef]
- Payne, A.H.; Hales, D.B. Overview of steroidogenic enzymes in the pathway from cholesterol to active steroid hormones. Endocrin. Rev. 2004, 25, 947–970. [Google Scholar] [CrossRef]
- Fellner, O.O. Zuckestoffwechsel, Sexualorgane, und Insulin. Med. Klin. 1926, 22, 1886–1888. [Google Scholar]
- Dohrn, M.; Faure, W.; Poll, H.; Blotevogel, W. Tokokine, Stoff mit sexualhormonartiger Wirkung aus Pfanzenzellen. Med. Klin. 1926, 22, 1417–1419. [Google Scholar]
- Butenandt, A. Über progynon ein krytallisiertes weibliches sexualhormon. Naturwissenschaften 1929, 17, 879. [Google Scholar] [CrossRef]
- Doisy, E.A.; Thayer, S.; Veler, C.D. The crystals of the follicular ovarian hormone. Proc. Soc. Exp. Biol. Med. 1929, 27, 417–419. [Google Scholar] [CrossRef]
- Sláma, K. Development and metamorphosis in invertebrates–hormonal control. Gen. Comp. Endocrinol. 1980, 40, 313–314. [Google Scholar]
- World Anti-Doping Agency. Available online: https://www.wada-ama.org/en/content/what-is-prohibited (accessed on 14 July 2019).
- Heftman, E. Functions of steroids in plants. Phytochemistry 1975, 14, 891–901. [Google Scholar] [CrossRef]
- Geuns, J.M.C. Steroid hormones and plant growth and development. Phytochemistry 1978, 17, 1–14. [Google Scholar] [CrossRef]
- Janeczko, A.; Skoczowski, A. Mammalian sex hormones in plants. Folia Histochem. Cytobiol. 2005, 43, 71–79. [Google Scholar] [PubMed]
- Janeczko, A. The presence and activity of progesterone in the plant kingdom. Steroids 2012, 77, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Pollak, O.J. Reduction of blood cholesterol in man. Circulation 1953, 7, 702–706. [Google Scholar] [CrossRef]
- Moghadasian, M.H.; Frohlich, J.J. Effects of dietary phytosterols on cholesterol metabolism and atherosclerosis: Clinical and experimental evidence. Am. J. Med. 1999, 107, 588–594. [Google Scholar] [CrossRef]
- Piironen, V.; Lindsay, D.G.; Miettinen, T.A.; Toivo, J.; Lampi, A.-M. Plant sterols: Biosynthesis, biological function and their importance to human nutrition. J. Sci. Food Agric. 2000, 80, 939–966. [Google Scholar] [CrossRef]
- Connolly, J.D.; Hill, R.A. Dictionary of Terpenoids, 1st ed.; Chapman and Hall/CRC: New York, NY, USA, 1992; ISBN 978-0412257704. [Google Scholar]
- Lichtenthaler, H.K. The plants’ 1-deoxy-d-xylulose-5-phosphate pathway for biosynthesis of isoprenoids. Fett-Lipid 1998, 100, 128–138. [Google Scholar] [CrossRef]
- McCaskill, D.; Croteau, R. Some caveats for bioengineering terpenoids metabolism in plants. Trends Biotechnol. 1998, 16, 349–355. [Google Scholar] [CrossRef]
- Benveniste, P. Sterol biosynthesis. Ann. Rev. Plant Physiol. 1986, 37, 275–308. [Google Scholar] [CrossRef]
- Nes, W.D. The biochemistry of plant sterols. Adv. Lipid Res. 1977, 15, 233–324. [Google Scholar] [CrossRef]
- Leboeuf, M.; Cave, A.; Goutarel, R. Steroid alkaloids. Presence of progesterone in the leaves of Holarrhena floribunda (G. Don) Duer and Schinz. C. R. Acad. Sci. Paris 1964, 259, 3401–3403. [Google Scholar]
- Iino, M.; Nomura, T.; Tamaki, Y.; Yamada, Y.; Yoneyama, K.; Takeuchi, Y.; Mori, M.; Asami, T.; Nakano, T.; Yokota, T. Progesterone: Its occurrence in plants and involvement in plant growth. Phytochemistry 2007, 68, 1664–1673. [Google Scholar] [CrossRef]
- Gawienowski, A.M.; Gibbs, C.C. Identification of cholesterol and progesterone in apples seeds. Steroids 1968, 12, 545–550. [Google Scholar] [CrossRef]
- Simons, R.G.; Grinwich, D.L. Immunoreactive detection of four mammalian steroids in plants. Can. J. Bot. 1989, 67, 288–296. [Google Scholar] [CrossRef]
- Carson, J.D.; Jenkins, R.L.; Wilson, M.M.; Howell, W.M.; Moore, R. Naturally occurring progesterone in loblolly pine (Pinus taeda L.): A major steroid precursor of environmental androgens. Environ. Toxicol. Chem. 2008, 27, 1273–1278. [Google Scholar] [CrossRef]
- Simerský, R.; Novák, O.; Morris, D.A.; Pouzar, V.; Strnad, M. Identification and quantification of several mammalian steroid hormones in plants by UPLC-MS/MS. J. Plant Growth Regul. 2009, 28, 125–136. [Google Scholar] [CrossRef]
- Lindemann, P. Steroidogenesis in plants–Biosynthesis and conversions of progesterone and other pregnane derivatives. Steroids 2015, 103, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Caspi, E.; Lewis, D.O.; Piatak, D.M.; Thimann, K.V.; Winter, A. Biosynthesis of plant sterols. Conversion of cholesterol to pregnenolone in Digitalis purpurea. Experientia 1966, 22, 506–507. [Google Scholar] [CrossRef]
- Bennett, R.D.; Heftmann, E.; Winter, B.J. Conversion of sitosterol to progesterone by Digitalis lanata. Naturwissenschaften 1969, 56, 463. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.D.; Heftmann, E.; Winter, B.J. A function of sitosterol. Phytochemistry 1969, 8, 2325–2328. [Google Scholar] [CrossRef]
- Lindemann, P.; Luckner, M. Biosynthesis of pregnane derivatives in somatic embryos of Digitalis lanata. Phytochemistry 1997, 46, 507–513. [Google Scholar] [CrossRef]
- Bhattacharya, B.; Gupta, K. Steroid hormone effects on growth and apical dominance of sunflower. Phytochemistry 1981, 20, 989–991. [Google Scholar] [CrossRef]
- Janeczko, A.; Filek, W. Stimulation of generative development in partly vernalized winter wheat by animal sex hormones. Acta Physiol. Plant. 2002, 24, 291–295. [Google Scholar] [CrossRef]
- Janeczko, A.; Filek, W.; Biesaga-Kosćielniak, J.; Marcińska, I.; Janeczko, Z. The influence of animal sex hormones on the induction of flowering in Arabidopsis thaliana: Comparison with the effect of 24-epibrassinolide. Plant Cell Tissue Org. Cult. 2003, 72, 147–151. [Google Scholar] [CrossRef]
- Ylstra, B.; Touraev, A.; Brinkmann, A.O.; Heberle-Bors, E.; Tunen, A. Steroid hormones stimulate germination and tube growth of in Vitro matured tobacco pollen. Plant Physiol. 1995, 107, 639–643. [Google Scholar] [CrossRef]
- Speranza, A.; Crosti, P.; Malerba, M.; Stocchi, O.; Scoccianti, V. The environmental endocrine disruptor, bisphenol A, affects germination, elicits stress response and alters steroid hormone production in kiwifruit pollen. Plant Biol. 2011, 13, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.J. Plant Hormones. Biosynthesis, Signal Transduction, Action! 3th ed.; Springer: Dordrecht, The Netherland, 2010; ISBN 9781402026850. [Google Scholar]
- Yang, X.H.; Xu, Z.H.; Xue, H.W. Arabidopsis membrane steroid binding protein 1 is involved in inhibition of cell elongation. Plant Cell 2005, 17, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Song, L.; Xue, H.W. Membrane steroid binding protein 1 (MSBP1) stimulates tropism by regulating vesicle trafficking and auxin redistribution. Mol. Plant 2008, 1, 1077–1087. [Google Scholar] [CrossRef] [PubMed]
- Janeczko, A.; Budziszewska, B.; Skoczowski, A.; Dybała, M. Specific binding sites for progesterone and 17β-estradiol in cells of Triticum aestivum L. Acta Biochim. Pol. 2008, 55, 701–711. [Google Scholar]
- Šaden-Krehula, M.; Tajić, M.; Kolbah, D. Sex hormones and corticosteroids in pollen of Pinus nigra. Phytochemistry 1979, 18, 345–346. [Google Scholar] [CrossRef]
- Šaden-Krehula, M.; Tajić, M.; Kolbah, D. Testosterone, epitestosterone and androstenedione in the pollen of Scotch pine P. siivestris L. Experimentia 1971, 27, 108–109. [Google Scholar] [CrossRef]
- Hartmann, S.; Lacorn, M.; Steinhart, H. Natural occurrence of steroid hormones in food. Food Chem. 1998, 62, 7–20. [Google Scholar] [CrossRef]
- Mohareb, A.S.O.; Kherallah, I.E.A.; Badawy, M.E.I.; Salem, M.Z.M.; Yousef, H.A. Chemical composition and activity of bark and leaf extracts of Pinus halepensis and Olea europaea grown in AL-Jabel AL-Akhdar region, Libya against some plant phytopathogens. J. Appl. Biotechnol. Bioeng. 2017, 3, 331–342. [Google Scholar] [CrossRef]
- Šaden-Krehula, M.; Kolbah, D.; Tajić, M.; Blažević, N. 17-ketosteroids in Pinus nigra Ar. Steroid hormones in the pollen of pine species. IV. Naturwissenschaften 1983, 70, 520–522. [Google Scholar] [CrossRef]
- Dean, P.D.G.; Exley, D.; Goodwin, T.W. Steroid oestrogens in plants: Re-estimation of oestrone in pomegranate seeds. Phytochemistry 1971, 10, 2215–2216. [Google Scholar] [CrossRef]
- Amin, S.; Paleologou, A.M. Estrone in Hyphaene thebaica kernel and pollen grains. Phytochemistry 1973, 12, 899–901. [Google Scholar] [CrossRef]
- Amin, S.; Bassiouny, A.R. Estrone in Olea europaea kernel. Phytochemistry 1979, 18, 344. [Google Scholar] [CrossRef]
- Young, I.J.; Knights, B.A.; Hillman, J.R. Oestradiol and its biosynthesis in Phaseolus vulgaris L. Nature 1977, 267, 429. [Google Scholar] [CrossRef] [PubMed]
- ZhongHan, Y.; Yin, T.; ZongXun, C. Changes of the contents of testosterone-steroidal sex hormone in reproductive organs of Lilium davidii Duch. Acta Bot. Sin. 1994, 36, 215–220. [Google Scholar]
- Farquhar, J.W.; Sokolow, M. Response of serum lipids and lipoproteins of man to beta-sitosterol and safflower oil. Circulation 1958, 17, 890–899. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.V.K.; Santos, R.F.; Siqueira, J.A.C.; Bariccatti, R.A.; Lenz, N.B.G.; Cruz, G.S.; Tokura, L.K.; Klajn, F.F. Chemical characterization of oil and biodiesel from four safflower genotypes. Ind. Crop Prod. 2018, 123, 192–196. [Google Scholar] [CrossRef]
- Hall III, C. Overview of the oilseed safflower (Carthamus tinctorius L.). Ref. Module Food Sci. 2016. [Google Scholar] [CrossRef]
- Heftmann, E. Biochemistry of plant steroids. Annu. Rev. Plant Physiol. 1963, 14, 225–248. [Google Scholar] [CrossRef]
- Heftmann, E. Function of sterols in plants. Lipids 1971, 6, 128–133. [Google Scholar] [CrossRef]
- Lin, J.; Proebsting, W.M.; Heftmann, E. Conversion of 4-androstene-3,17-dione to testosterone by Pisum sativum. Phytochemistry 1979, 18, 1667–1669. [Google Scholar] [CrossRef]
- Lin, J.; Palevitch, D.; Heftmann, E. Reduction of 4-androstene-3,17-dione by growing cucumber plants. Phytochemistry 1983, 22, 1149–1154. [Google Scholar] [CrossRef]
- Hirotani, M.; Furuya, T. Biotransformation of testosterone and other androgens by suspension cultures of Nicotiana tabacum “bright yellow”. Phytochemistry 1974, 13, 2135–2142. [Google Scholar] [CrossRef]
- De Brabander, H.F.; Poelmans, S.; Schilt, R.; Stephany, R.W.; Le Bizec, B.; Draisci, R.; Sterk, S.S.; van Ginkel, L.A.; Courtheyn, D.; Van Hoff, N.; et al. Presence and metabolism of the anabolic steroid boldenone in various animal species: a review. Food Addit. Contam. 2004, 21, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Poelmans, S.; DeWasch, K.; Martele, Y.; Schilt, R.; VanHoof, N.; Noppe, H.; Verslycke, T.; Janssen, C.; Courtheyn, D.; De Brabander, H.F. The possible transformation of phytosterols to boldenone. In Proceedings Euro Food Chem XII: Strategies for Safe Food; D-Facto Public: Bruges, Belgium, 2003; pp. 74–78. [Google Scholar]
- Kostova, I.; Dinchev, D. Saponins in Tribulus terrestris–chemistry and bioactivity. Phytochem. Rev. 2005, 4, 111–137. [Google Scholar] [CrossRef]
- Janeczko, A. Influence of Selected Steroids on Plant Physiological Processes-Especially Flowering Induction. Ph.D. Thesis, Agricultural University, Krakow, Poland, 2000. [Google Scholar]
- Janeczko, A.; Filek, W.; Skoczowski, A. Influence of human sex hormones on the growth response of winter wheat immature embryos and callus (in Polish). Zesz. Probl. Post Nauk. Roln. 2002, 488, 667–673. [Google Scholar]
- Loewe, S.; Spohr, E. Über weibliche Sexualhormone (Thelytropine). XII. Brunsterzeugende Stoffe (Thelykinine) als Erzeugnisse des Pflanzenreiches. Biochem. Z. 1927, 180, 1–26. [Google Scholar]
- Butenandt, A.; Jakobi, H. Über die Darstellung eines kristallisierten pflanzlichen Tokokinins (Thelykinins) und seine Identifizierung mit dem ex-Follikelhormon. Untersuchungen über das weibliche Sexualhormon, 10. Mitteilung. Z. Physiol. Chem. 1933, 218, 104–112. [Google Scholar] [CrossRef]
- Skarzynski, B. An oestrogenic substance from plant material. Nature 1933, 131, 766. [Google Scholar]
- Hassan, A.; Wafa, M.A. An estrogenic substance in pollen-grains of date palm tree Phaenix dactylifera L., Palmae. Nature 1947, 159, 409–410. [Google Scholar] [CrossRef]
- El Ridi, M.S.; Wafa, M.A. Estrogenic substance in palm-pollen grains of the date palm. J. Roy. Egypt Med. Assoc. 1948, 30, 124–127. [Google Scholar]
- Bennett, R.D.; Ko, S.T.; Heftmann, E. Isolation of estrone and cholesterol from the date palm, Phoenix dactylifera L. Phytochemistry 1966, 5, 231–235. [Google Scholar] [CrossRef]
- Awad, O. Steroidal estrogens of Prunus armeniaca seeds. Phytochemistry 1974, 13, 678–679. [Google Scholar] [CrossRef]
- Kopcewicz, J. Estrogens in developing bean (Phaseolus vulgaris) plants. Phytochemistry 1971, 10, 1423–1427. [Google Scholar] [CrossRef]
- Jacobsohn, G.M.; Frey, M.J.; Hochberg, R.B. The absence of steroid estrogens in plants. Steroids 1965, 6, 93–99. [Google Scholar] [CrossRef]
- Van Rompuy, L.L.L.; Zeevaart, J.A.D. Ann. Report MSU/ERDA Plant Research Laboratory; Michigan State University: East Lansing, MI, USA, 1975; Volume 47. [Google Scholar]
- Milanesi, L.; Monje, P.; Boland, R. Presence of estrogen and estrogen receptor-like proteins in Solanum glaucophyllum. Biochem. Biophys. Res. Commun. 2001, 289, 1175–1179. [Google Scholar] [CrossRef]
- Helmkamp, G.; Bonner, J. Some relationships of sterols to plant growth. Plant Physiol. 1953, 28, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Kopcewicz, J. Influence of estrone on growth and endogenous gibberellins content in dwarf pea. Bull. Sci. Cl. 1969, 17, 727–731. [Google Scholar]
- Guan, M.; Roddick, J.G. Epibrassinolide - inhibition of development of excised, adventitious and intact root of tomato (Lycopersicon esculentum): Comparison with the effects of steroidal estrogens. Physiol. Plant. 1988, 74, 720–726. [Google Scholar] [CrossRef]
- Chouard, P. Action combinee de la folliculine et la durée d´éclairement sur la floraison des Reines-Marguerites. Compt. Rend. Soc. Biol. 1937, 126, 509–512. [Google Scholar]
- Kopcewicz, J. Influence of estrogens on the flower formation in Cichorium intybus L. Naturwissenschaften 1970, 57, 136. [Google Scholar] [CrossRef]
- Kopcewicz, J.; Porazinski, Z. Effect of growth regulators, steroids and estrogen fraction from sage plants on flowering of a long-day plant, Salvia splendens, grown under non-inductive light conditions. Biol. Plant. 1974, 16, 132–135. [Google Scholar] [CrossRef]
- Kopcewicz, J. Influence of steroidal hormones on flower sex expression in Ecballium elaterium (L.). Z. Pflanzenphysiol. 1971, 65, 92–94. [Google Scholar]
- Gawienowski, M.; Cheney, R.W.; Marsh, H.V. Alteration of sex expression in the cucumber by testosterone and estradiol. Phytochemistry 1971, 10, 2033–2034. [Google Scholar] [CrossRef]
- Young, I.J.; Knights, B.A.; Hillman, J.R. The metabolism of estrogens in vivo and in vitro by Phaseolus vulgaris. Z. Pflanz. 1979, 94, 307–316. [Google Scholar] [CrossRef]
| Steroid | CAS Number | Mol. Formula | Mol. Weight (g·mol−1) | Amount | Origin | Ref. |
|---|---|---|---|---|---|---|
| progesterone | 57-83-0 | C21H30O2 | 314.46 | 0.08 µg·g−1 | Pinus nigra | [48] |
| 3–1600 ng·g−1 | 31 plant species | [31] | ||||
| 6–1540 ng·kg−1 | 8 plant species | [29] | ||||
| 1.19–15.5 µg·g−1 | Pinus taeda | [32] | ||||
| 0.66 ng·g−1 | Inula helenium | [33] | ||||
| 17.4 ng·g−1 | Nicotiana tabacum | [33] | ||||
| 18.5 ng·g−1 | Digitalis purpurea | [33] | ||||
| 0.02–15.39 ng·g−1 | Tribulus terrestris | Tarkowská, unpublished | ||||
| testosterone | 58-22-0 | C19H28O2 | 288.42 | 0.08 µg·g−1 | Pinus silvestris | [49] |
| 0.09 µg·kg−1 | wheat | [50] | ||||
| 0.05 µg·kg−1 | corn oil | [50] | ||||
| 0.21 µg·kg−1 | safflower oil | [50] | ||||
| 0.01–0.02 ng·g−1 | Tribulus terrestris | Tarkowská, unpublished | ||||
| androsta-1,4-diene-3,17-dione (boldione) | 897-06-3 | C19H24O2 | 284.40 | not quantified | Pinus halepensis | [51] |
| 0.1–2.7 pg·g−1 | Tribulus terrestris | Tarkowská, unpublished | ||||
| androst-4-ene-3,17-dione | 63-05-8 | C19H26O2 | 286.41 | 0.59 µg·g−1 | Pinus silvestris | [49] |
| 0.09 µg·g−1 | Pinus nigra | [48] | ||||
| 0.08 µg·g−1 | Pinus nigra | [52] | ||||
| 0.05 ng·g−1 | potato | [50] | ||||
| 0.48 ng·g−1 | wheat | [50] | ||||
| 2.20 ng·g−1 | Nicotiana tabacum | [33] | ||||
| 3.20 ng·g−1 | Inula helenium | [33] | ||||
| 0.01–0.05 ng·g−1 | Tribulus terrestris | Tarkowská, unpublished | ||||
| estrone | 53-16-7 | C18H22O2 | 270.37 | 2.5–4.5 µg·kg−1 | pomegranate | [53] |
| 5.13/5.25 µg·g−1 | Haphaene thebaica | [54] | ||||
| 0.04 µg·ml−1 | corn oil | [54] | ||||
| 33.75 µg·g−1 | Olea europea | [55] | ||||
| 28–420 ng·g−1 | 20 plant species | [31] | ||||
| 17β-estradiol | 50-28-2 | C18H24O2 | 272.38 | 2–10 µg·kg−1 | Phaseolus vulgaris | [56] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tarkowská, D. Plants are Capable of Synthesizing Animal Steroid Hormones. Molecules 2019, 24, 2585. https://doi.org/10.3390/molecules24142585
Tarkowská D. Plants are Capable of Synthesizing Animal Steroid Hormones. Molecules. 2019; 24(14):2585. https://doi.org/10.3390/molecules24142585
Chicago/Turabian StyleTarkowská, Danuše. 2019. "Plants are Capable of Synthesizing Animal Steroid Hormones" Molecules 24, no. 14: 2585. https://doi.org/10.3390/molecules24142585
APA StyleTarkowská, D. (2019). Plants are Capable of Synthesizing Animal Steroid Hormones. Molecules, 24(14), 2585. https://doi.org/10.3390/molecules24142585