Role of Some Food-Grade Synthesized Flavonoids on the Control of Ochratoxin A in Aspergillus carbonarius
Abstract
1. Introduction
2. Results
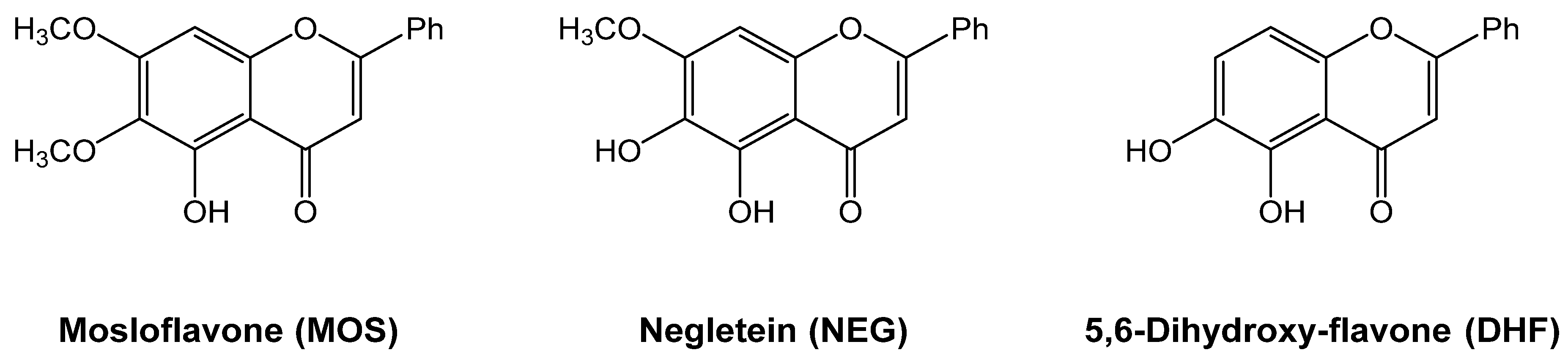
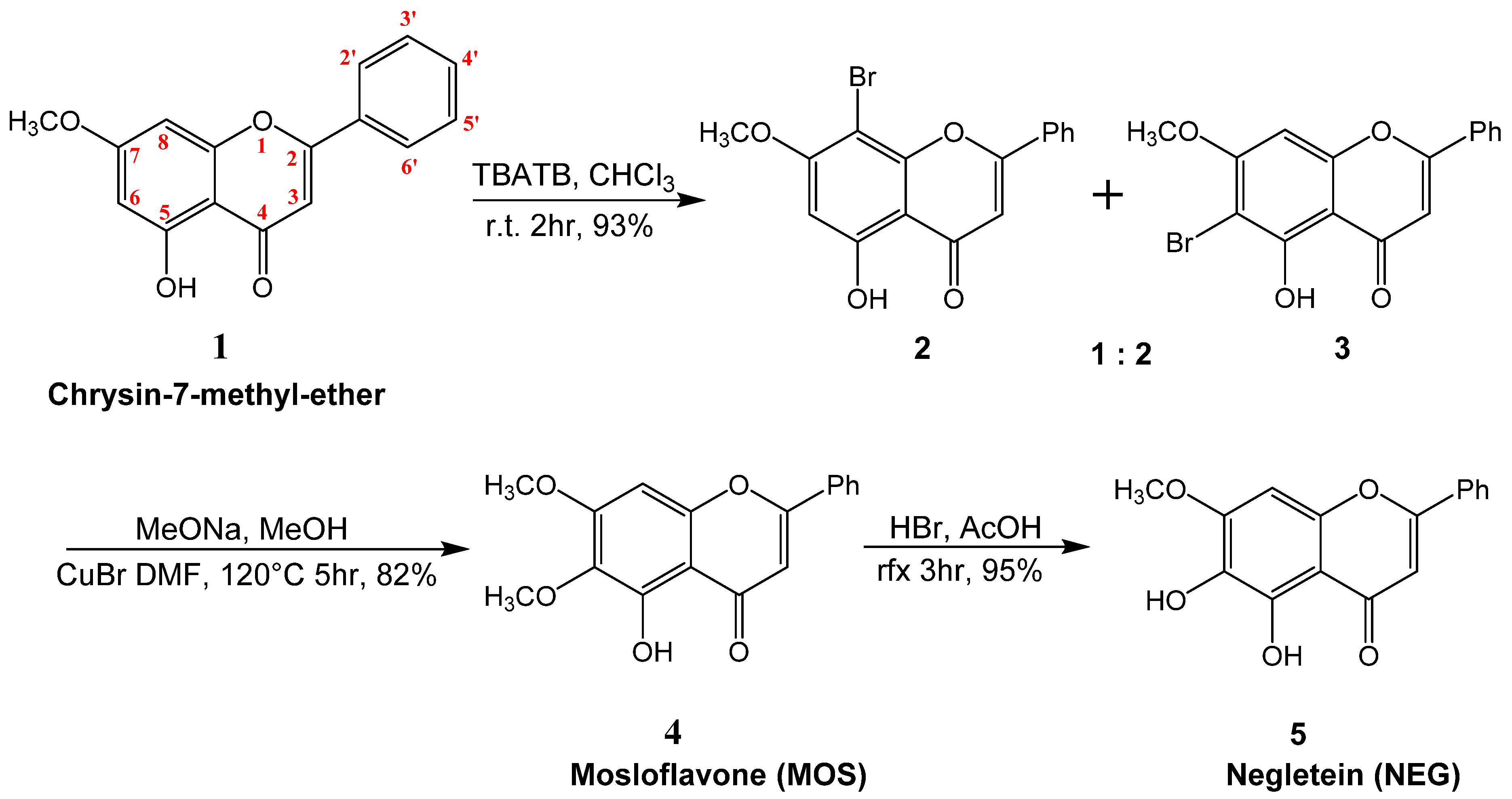
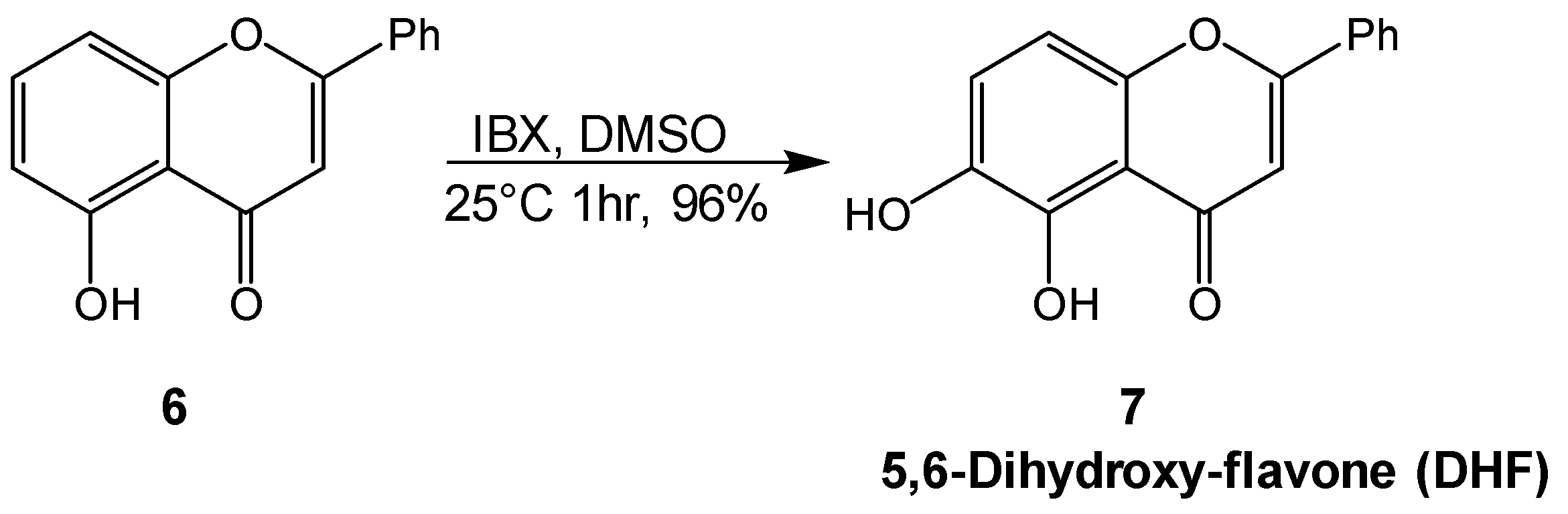
2.1. Synthesis of Mosloflavone 4, Negletein 5, and 5,6-Dihydroxy-flavone 6
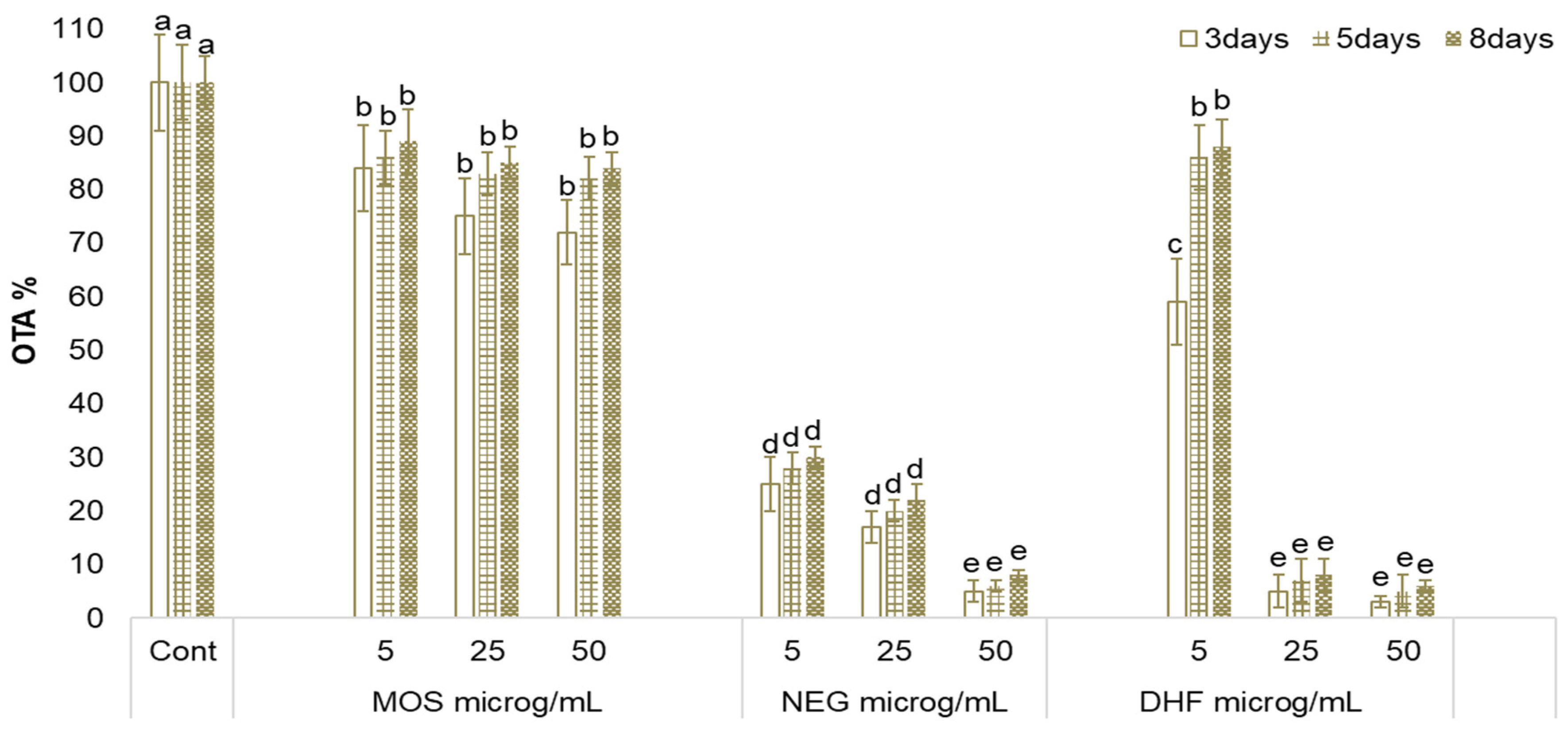
2.2. Effect of MOS, NEG, and DHF on A. carbonarius Growth and OTA Biosynthesis
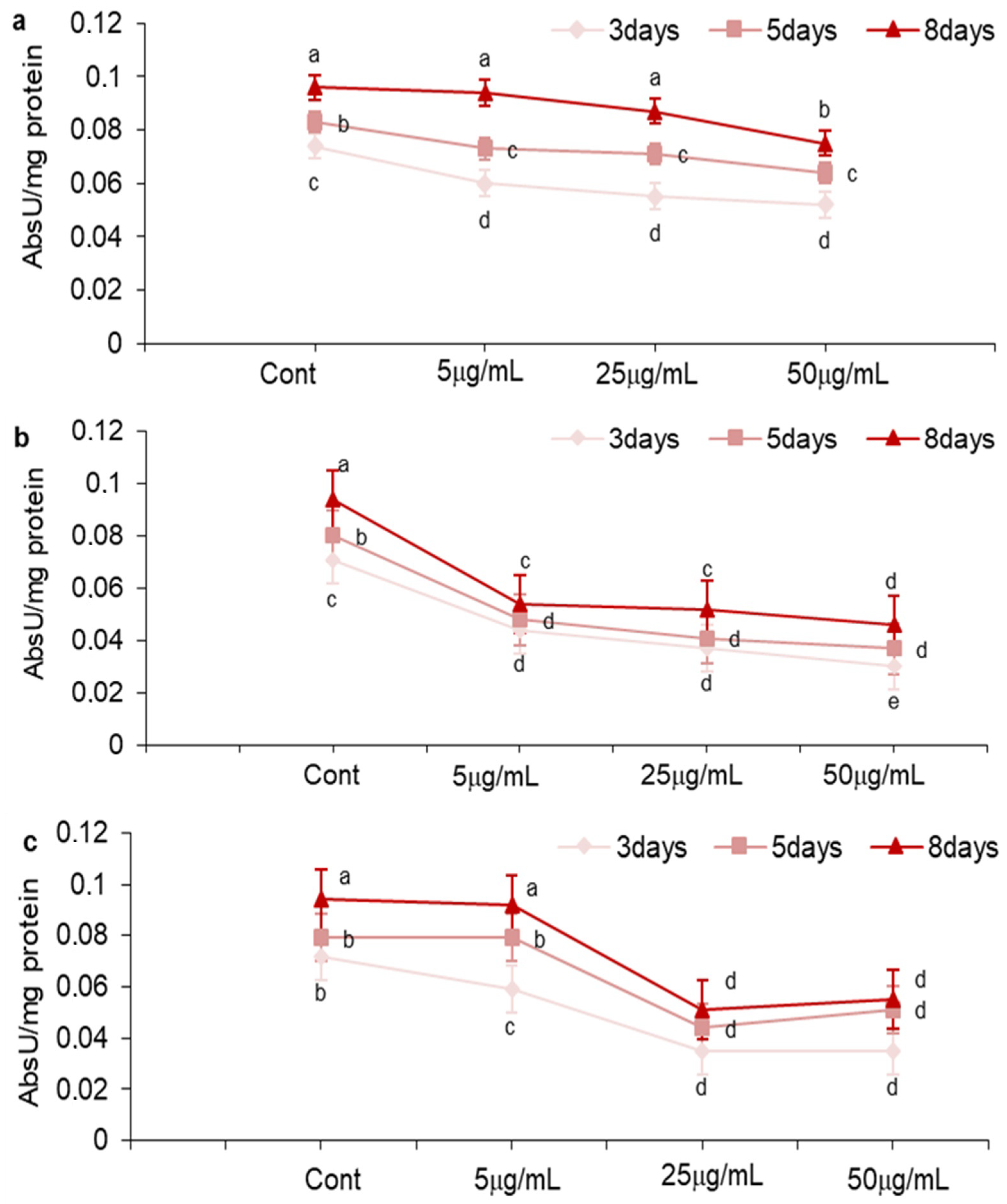
2.3. Effect of MOS, NEG, and DHF on LOX Activity in A. carbonarius
3. Discussion
4. Materials and Methods
4.1. Synthesis of 5-hydroxy-7-methoxy-8-bromo-flavone 2, and 5-hydroxy-6-bromo-7-methoxy-flavone 3
4.2. Synthesis of 5-hydroxy-6,7-dimethoxy-flavone 4 (MOS)
4.3. Synthesis of 5,6-dihydroxy-7-methoxy-flavone 5 (NEG)
4.4. Synthesis of 5,6-diydroxy-flavone 7 (DHF)
4.5. Fungal Cultures
4.6. OTA Extraction and Analysis, LOX Activity Determination
4.7. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Wang, Y.; Wang, L.; Liu, F.; Wang, Q.; Nimal Selvaraj, J.; Xing, F.; Zhao, Y.; Liu, Y. Ochratoxin A producing fungi biosynthetic pathway and regulatory mechanisms. Toxins 2016, 8, 83. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, A.; Atoui, A. Ochratoxin A: General overview and actual molecular status. Toxins 2010, 2, 461–493. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). Some naturally occurring substances: Food items and constituents, heterocyclic aromatic amines and mycotoxins. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 1993; p. 56. [Google Scholar]
- Commission, E. Commission regulation (EC) No. 1881/2006 of 19/12/2006 Setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union L 2006, I364, 4–5. [Google Scholar]
- Heussner, A.H.; Bingle, L.E.H. Comparative ochratoxin toxicity: A review of the available data. Toxins 2015, 7, 4253–4282. [Google Scholar] [CrossRef] [PubMed]
- Channaiah, L.H.; Maier, D.E. Best Stored Maize Management Practices for the Prevention of Mycotoxin Contamination. In Mycotoxin Reduction in Grain Chains, 1st ed.; Leslie, J.F., Logrieco, A.F., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2014; pp. 78–87. [Google Scholar]
- Pittet, A. Natural occurrence of mycotoxins in foods and feeds, an updated review. Rev. Med. Vet. 1998, 149, 479–492. [Google Scholar]
- Neme, K.; Mohammed, A. Mycotoxin occurrence in grains and the role of postharvest management as mitigation strategies. A review. Food Control 2017, 78, 412–425. [Google Scholar] [CrossRef]
- Atanasova-Penichon, V.; Barreau, C.; Richard-Forget, F. Antioxidant secondary metabolites in cereals: Potential involvement in resistance to Fusarium and mycotoxin accumulation. Front. Microbiol. 2016, 7, 566. [Google Scholar] [CrossRef] [PubMed]
- Reverberi, M.; Punelli, F.; Scarpari, M.; Camera, E.; Zjalic, S.; Ricelli, A.; Fanelli, C.; Fabbri, A.A. Lipoperoxidation affects ochratoxin A biosynthesis in Aspergillus ochraceus and its interaction with wheat seeds. Appl. Microbiol. Biotechnol. 2010, 85, 1935–1946. [Google Scholar] [CrossRef] [PubMed]
- Morcia, C.; Tumino, G.; Ghizzoni, R.; Bara, A.; Salhi, N.; Terzi, V. In Vitro evaluation of sub-lethal concentrations of plant-derived antifungal compounds on Fusaria growth and mycotoxin production. Molecules 2017, 22, 1271. [Google Scholar] [CrossRef]
- Montenegro, C.D.; Goncalves, G.F.; De Oliveira, A.A.; Lira, A.B.; Cassiano, T.T.M.; De Lima, N.T.R.; Barbosa, J.M.; Diniz, M.D.F.M.; Pessoa, H.L.F. In silico study and bioprospection of the antibacterial and antioxidant effects of flavone and its hydroxylated derivatives. Molecules 2017, 22, 869. [Google Scholar] [CrossRef]
- Wang, J.; Yue, Y.D.; Tang, F.; Sun, J. TLC Screening for antioxidant activity of extracts from fifteen bamboo species and identification of antioxidant flavone glycosides from leaves of Bambusa textilis McClure. Molecules 2012, 17, 12297–12311. [Google Scholar] [CrossRef] [PubMed]
- Robens, J.; Cardwell, K. The costs of mycotoxin management to the USA.: Management of aflatoxins in the United States. J. Toxicol. Toxin Rev. 2003, 22, 139–152. [Google Scholar] [CrossRef]
- Sorrenti, V.; Di Giacomo, C.; Acquaviva, R.; Barbagallo, I.; Bognanno, M.; Galvano, F. Toxicity of ochratoxin A and its modulation by antioxidants: A review. Toxins 2013, 5, 1742–1766. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, H.; Sumarah, M.W.; Gao, Q.; Wang, D.; Zhang, Y. veA gene acts as a positive regulator of conidia production, ochratoxin A biosynthesis, and oxidative stress tolerance in Aspergillus niger. J. Agric. Food Chem. 2018, 66, 13199–13208. [Google Scholar] [CrossRef]
- Vaya, J.; Belinky, P.A.; Aviram, M. Antioxidant constituents from licorice roots: Isolation, structure elucidation and antioxidative capacity toward LDL oxydation. Free Radic. Biol. Med. 1997, 23, 302–313. [Google Scholar] [CrossRef]
- Barreca, D.; Bellocco, E.; Leuzzi, U.; Gattuso, G. First evidence of C- and O-glycosyl flavone in blood orange (Citrus sinensis (L.) Osbeck) juice and their influence on antioxidant properties. Food Chem. 2014, 149, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Yun, K.J.; Lim, D.H.; Kim, J.; Jang, Y.P. Anti-inflammatory properties of flavone di-C-glycosides as active principles of Camellia mistletoe, Korthalsella japonica. Biomol. Ther. 2016, 24, 630–637. [Google Scholar] [CrossRef]
- Choi, H.J.; Song, H.H.; Lee, J.S.; Ko, H.J.; Song, J.-H. Inhibitory effects of norwogonin, oroxylin A, and mosloflavone on enterovirus 71. Biomol. Ther. 2016, 24, 552–558. [Google Scholar] [CrossRef]
- Gonzalez Collado, I.; Macias, F.A.; Massanet, G.M.; Rodriguez, L. Flavonoids from Centaurea clementei. J. Nat. Prod. 1985, 48, 819–822. [Google Scholar] [CrossRef]
- Van Kiem, P.; Van Minh, C.; Hoang, T.H.; Jung, J.L.; Im, S.L.; Young, H.K. Phenolic constituents with inhibitory activity against NFAT transcription from Desmos chinensis. Arch. Pharm. Res. 2005, 28, 1345–1349. [Google Scholar] [CrossRef]
- Phan, C.W.; Sabaratnam, V.; Bovicelli, P.; Righi, G.; Saso, L. Negletein as a neuroprotectant enhances the action of nerve growth factor and induces neurite outgrowth in PC12 cells. Biofactors 2016, 42, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Righi, G.; Antonioletti, R.; Silvestri-Proietti, I.; D’Antona, N.; Lambusta, D.; Bovicelli, P. Convergent synthesis of mosloflavone, negletein and baicalein from crysin. Tetrahedron 2010, 66, 1294–1298. [Google Scholar] [CrossRef]
- Barontini, M.; Bernini, R.; Crisante, F.; Fabrizi, G. Selective and efficient oxidative modifications of flavonoids with 2-iodoxybenzoic acid. Tetrahedron 2010, 66, 6047–6053. [Google Scholar] [CrossRef]
- Gao, H.; Nishioka, T.; Kawabata, J.; Kasai, T. Structure-activity relationship for alpha-glucosidase inhibition of baicalein, 5,6,7-trihydroxyflavone: The effect of A-ring substitution. Biosci. Biotechnol. Biochem. 2004, 68, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, E.; Sabellico, C.; Hajek, J.; Stankova, V.; Filipsky, T.; Balducci, V.; De Vito, P.; Leone, S.; Bavavea, E.I.; Proietti-Silvestri, I.; et al. Protection of cells against oxidative stress by nanomolar levels of hydroxyflavones indicates a new type of intracellular antioxidant mechanism. PloS ONE 2013, 8, e60796. [Google Scholar] [CrossRef] [PubMed]
- Antonioletti, R.; Viglianti, A.; Cristofoli, S.; Ricelli, A. Role of some neosynthesized styryl heterocycles in ochratoxin A control in Aspergillus carbonarius. Curr. Org. Chem. 2016, 20, 2029–2035. [Google Scholar] [CrossRef][Green Version]
- Cheynier, V.; Comte, G.; Davies, K.M.; Lattanzio, V.; Martens, S. Plant phenolics: Recent advances on their biosynthesis, genetics and ecophysiology. Plant Physiol. Biochem. 2013, 72, 1–20. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic compounds in Brassica vegetables. Molecules 2011, 16, 251–280. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 162750. [Google Scholar] [CrossRef]
- Havermann, S.; Chovolou, Y.; Humpf, H.U.; Watjen, W. Modulation of the Nrf2 signalling pathway in Hct116 colon carcinoma cells by baicalein and its methylated derivative negletein. Pharm. Biol. 2016, 54, 1491–1502. [Google Scholar] [CrossRef]
- Frigerio, M.; Santagostino, M.; Sputore, S. A user-friendly entry to 2-iodoxybenzoic acid (IBX). J. Org. Chem. 1999, 64, 4537–4538. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds mosloflavone, negletein are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ricelli, A.; De Angelis, M.; Primitivo, L.; Righi, G.; Sappino, C.; Antonioletti, R. Role of Some Food-Grade Synthesized Flavonoids on the Control of Ochratoxin A in Aspergillus carbonarius. Molecules 2019, 24, 2553. https://doi.org/10.3390/molecules24142553
Ricelli A, De Angelis M, Primitivo L, Righi G, Sappino C, Antonioletti R. Role of Some Food-Grade Synthesized Flavonoids on the Control of Ochratoxin A in Aspergillus carbonarius. Molecules. 2019; 24(14):2553. https://doi.org/10.3390/molecules24142553
Chicago/Turabian StyleRicelli, Alessandra, Martina De Angelis, Ludovica Primitivo, Giuliana Righi, Carla Sappino, and Roberto Antonioletti. 2019. "Role of Some Food-Grade Synthesized Flavonoids on the Control of Ochratoxin A in Aspergillus carbonarius" Molecules 24, no. 14: 2553. https://doi.org/10.3390/molecules24142553
APA StyleRicelli, A., De Angelis, M., Primitivo, L., Righi, G., Sappino, C., & Antonioletti, R. (2019). Role of Some Food-Grade Synthesized Flavonoids on the Control of Ochratoxin A in Aspergillus carbonarius. Molecules, 24(14), 2553. https://doi.org/10.3390/molecules24142553




