Evaluation of Anti-Tumorigenic Effects of Diosmetin against Human Colon Cancer Xenografts in Athymic Nude Mice
Abstract
1. Introduction
2. Results
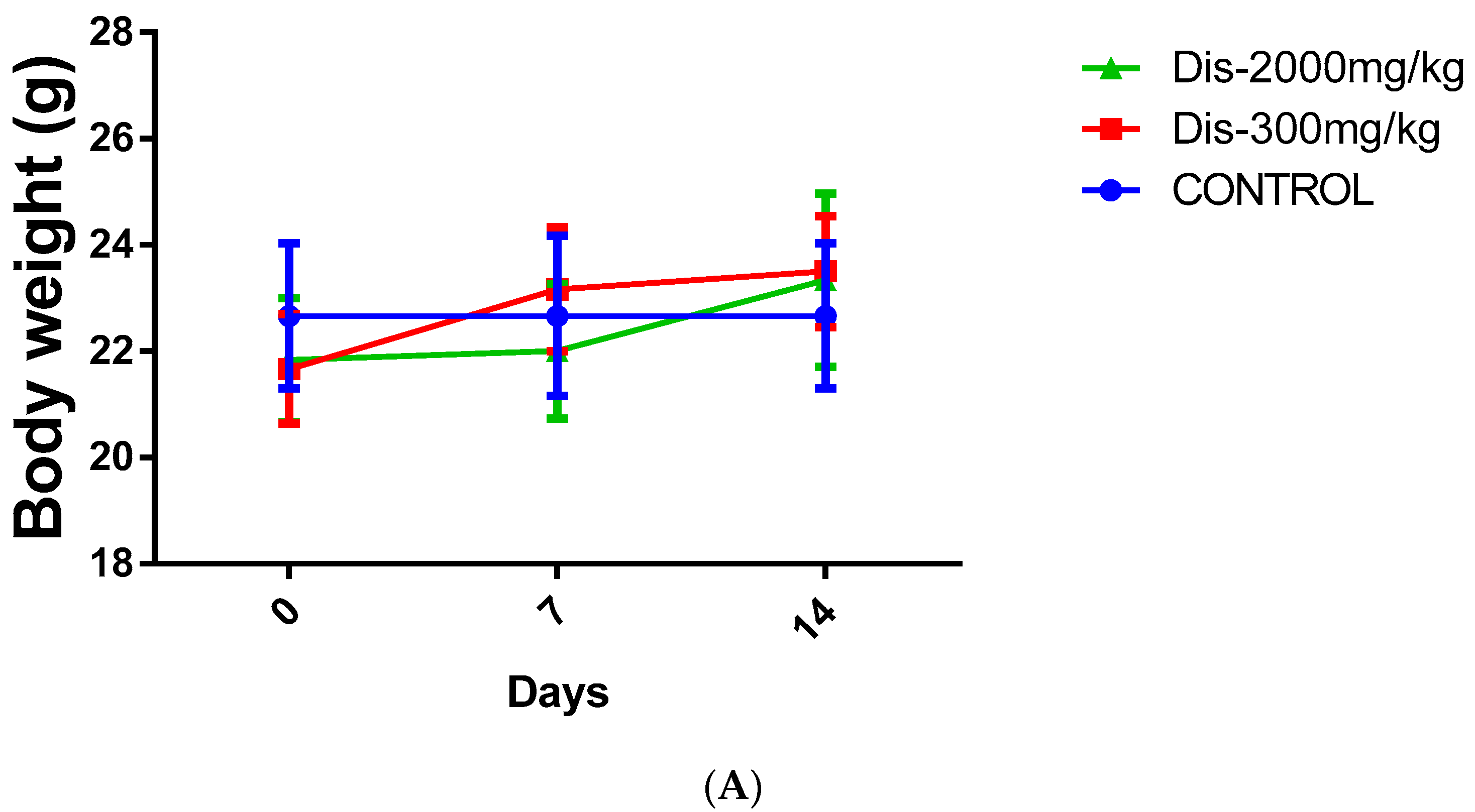
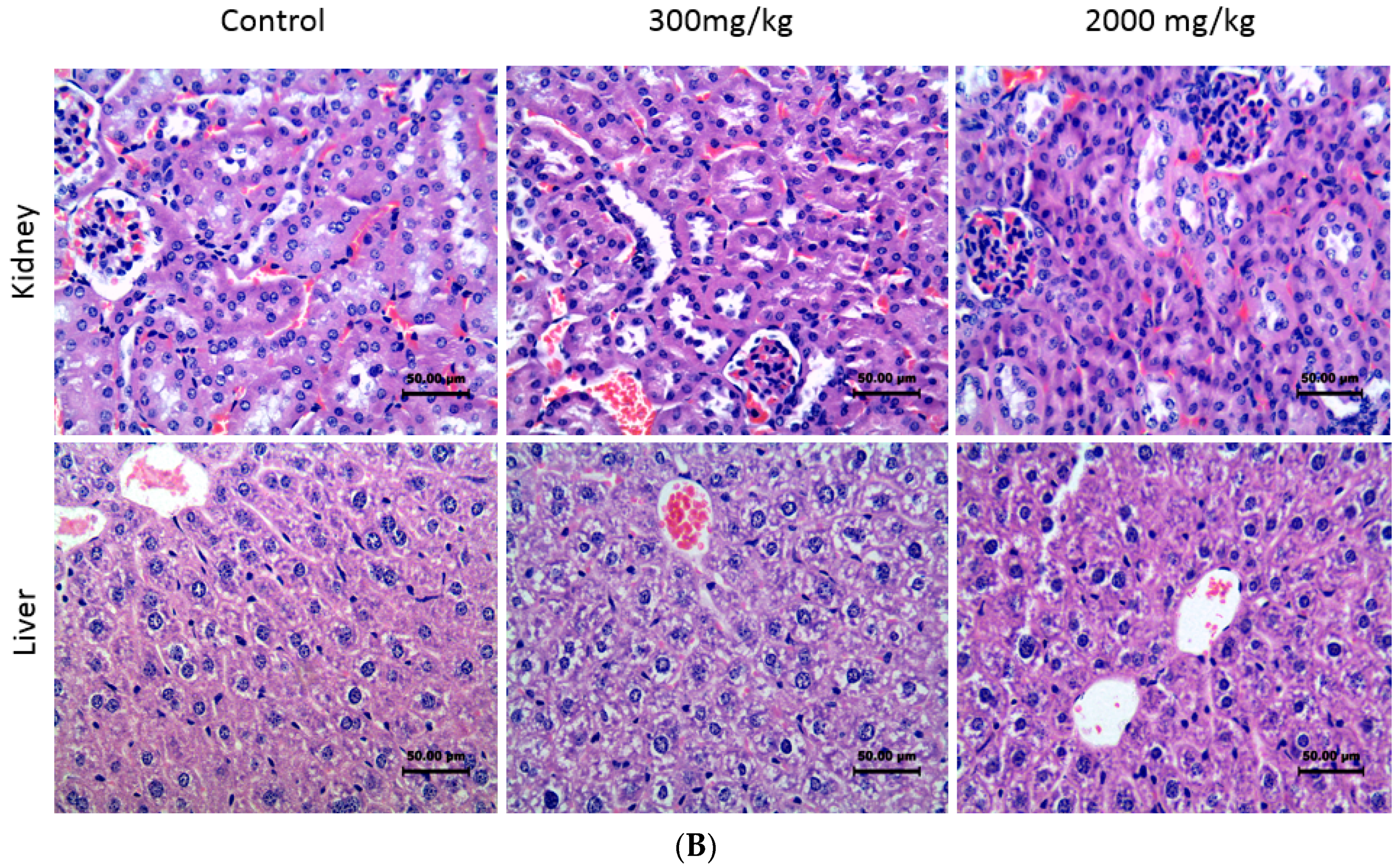
2.1. Acute Toxicity of Diosmetin
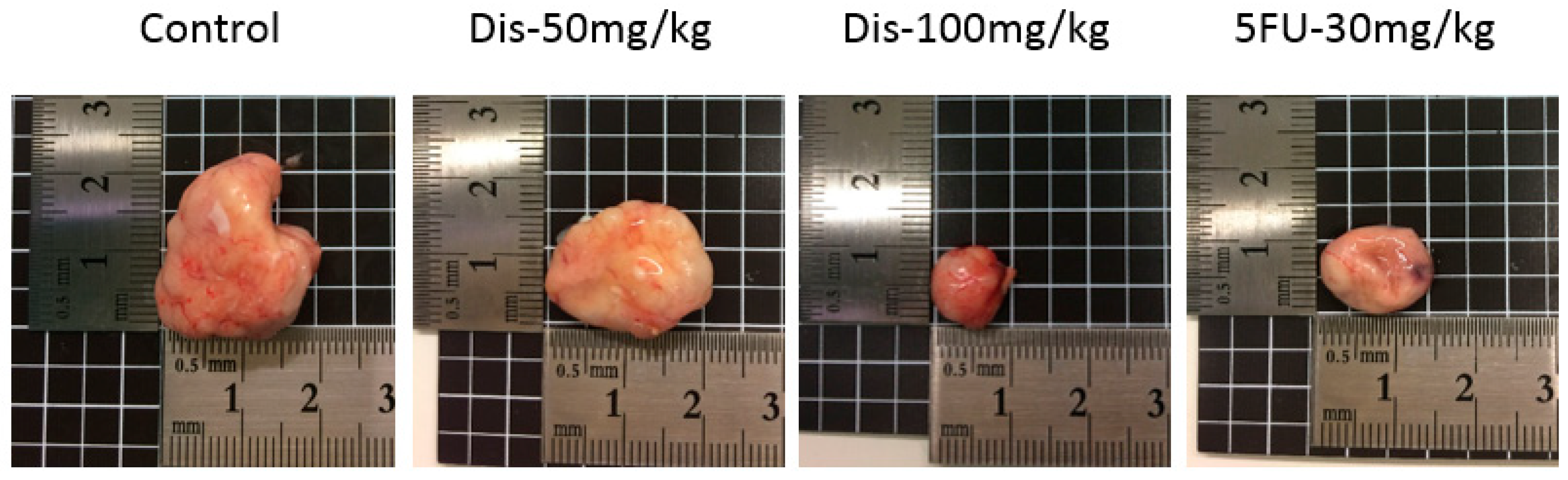
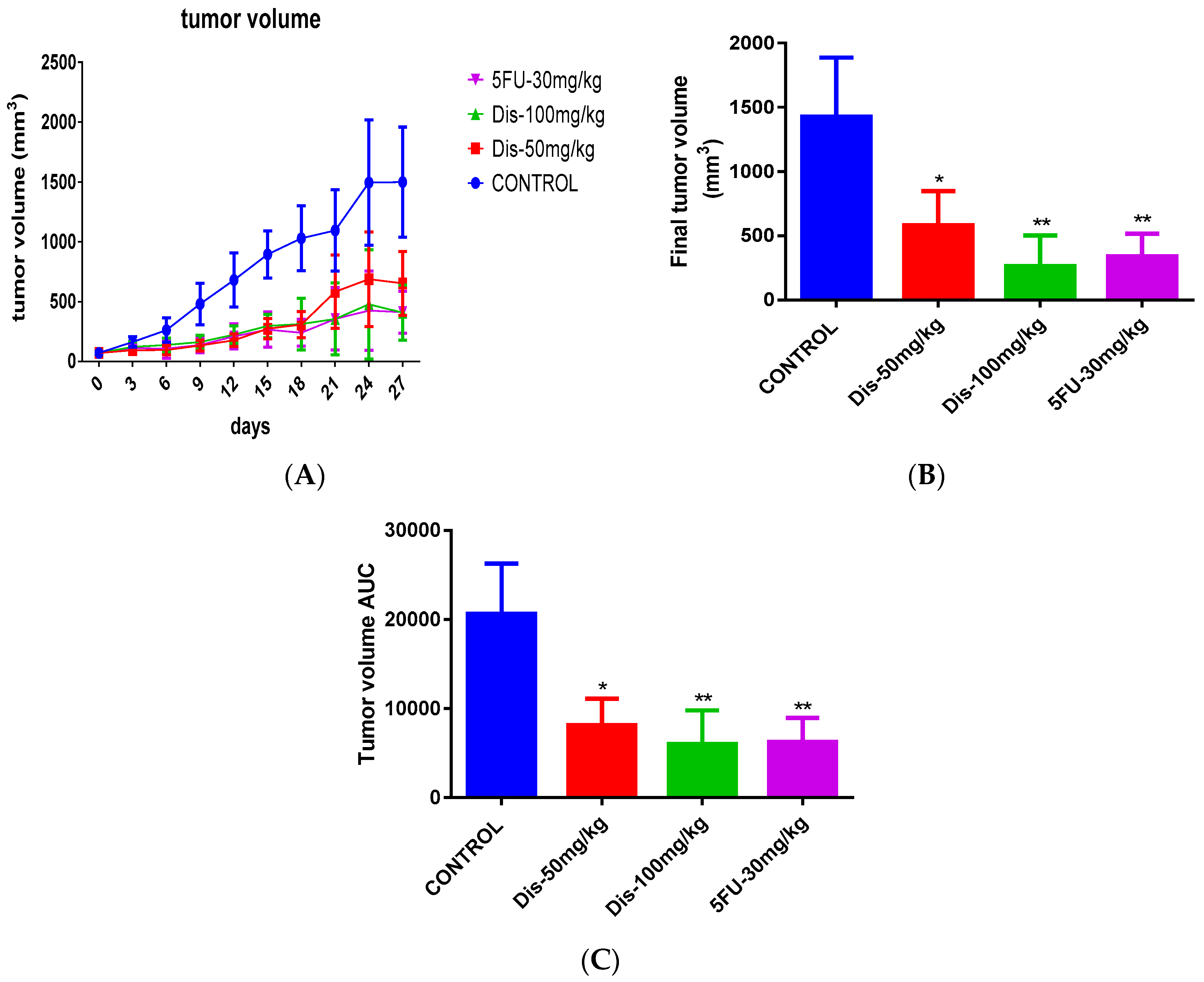
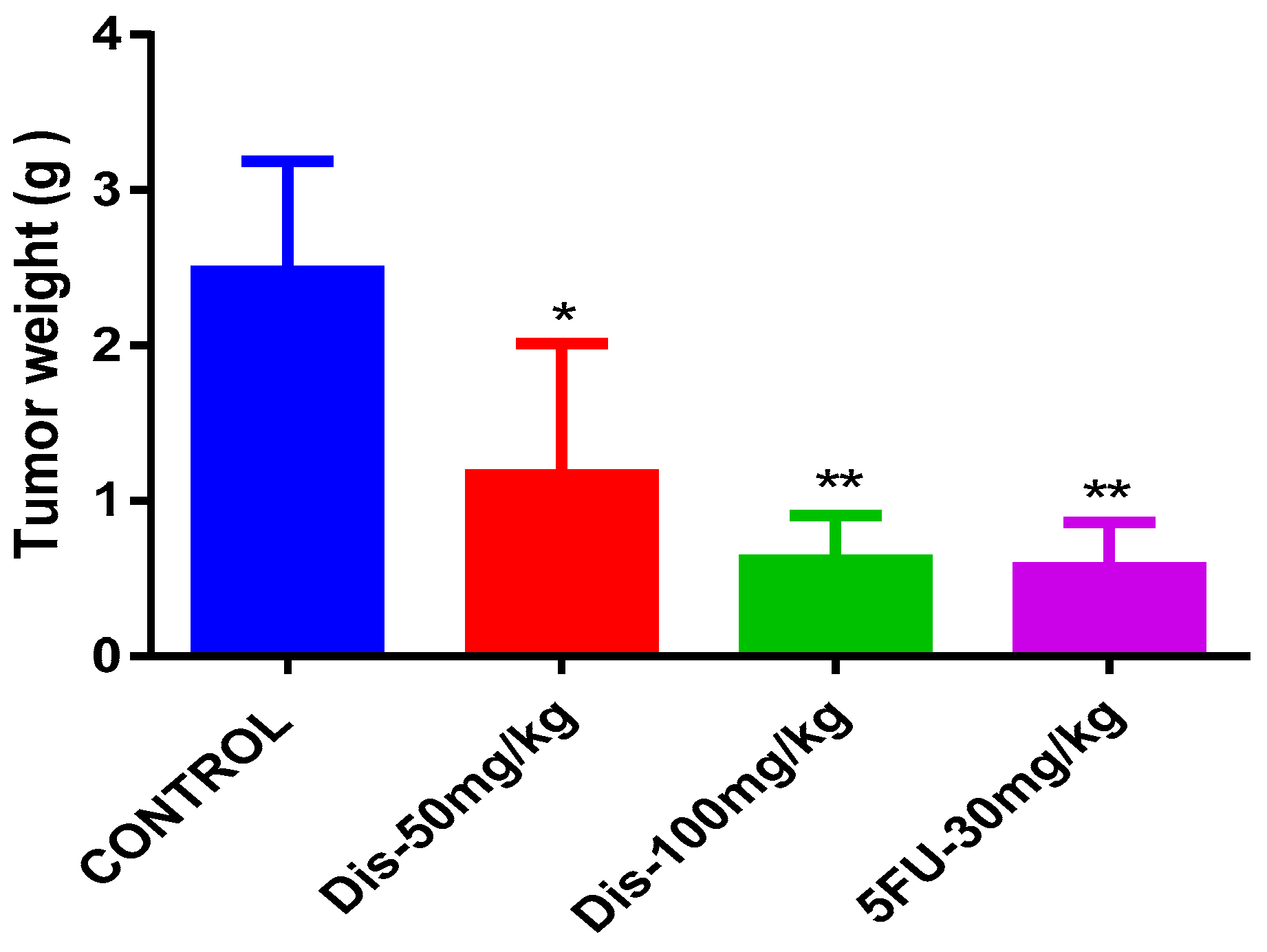
2.2. Dis Reduces Tumor Size in HCT-116 Colon Cancer Xenograft Nude Mice
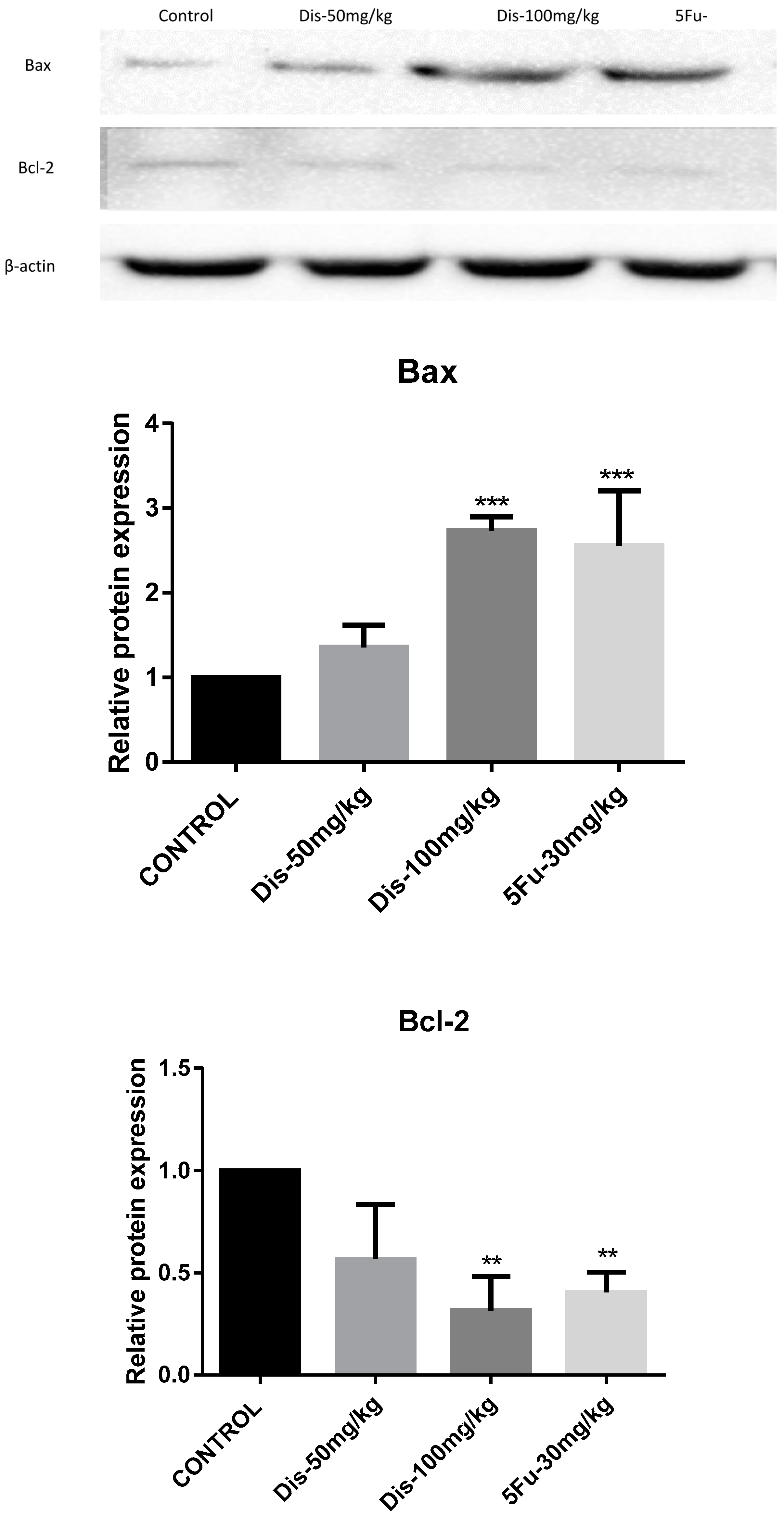
2.3. Apoptotic Effects of Dis in HCT-116 Xenograft Nude Mice
3. Discussion
4. Materials and Methods
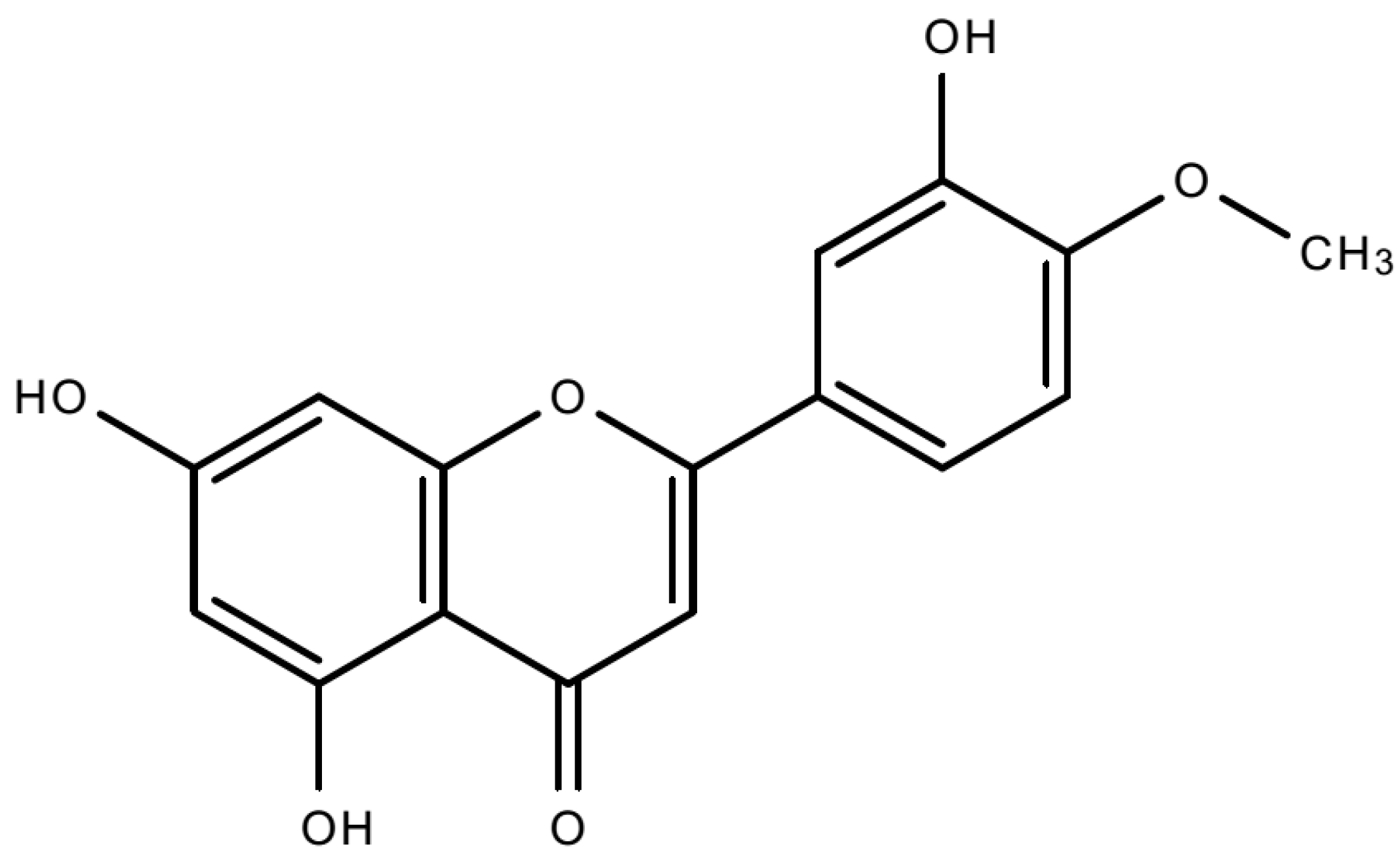
4.1. Compound and Cell Lines
4.2. Acute Toxicity
4.3. Human Colorectal Cancer Xenografts in Athymic Nude Mice
4.4. Western Blot
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2016, 66, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Focaccetti, C.; Bruno, A.; Magnani, E.; Bartolini, D.; Principi, E.; Dallaglio, K.; Bucci, E.O.; Finzi, G.; Sessa, F.; Noonan, D.M. Effects of 5-fluorouracil on morphology, cell cycle, proliferation, apoptosis, autophagy and ROS production in endothelial cells and cardiomyocytes. PLoS ONE 2015, 10, e0115686. [Google Scholar] [CrossRef] [PubMed]
- Koosha, S.; Alshawsh, M.A.; Looi, C.Y.; Seyedan, A.; Mohamed, Z. An association map on the effect of flavonoids on the signaling pathways in colorectal cancer. Int. J. Med. Sci. 2016, 13, 374. [Google Scholar] [CrossRef] [PubMed]
- AlAjmi, M.F.; Rehman, M.T.; Hussain, A.; Rather, G.M. Pharmacoinformatics approach for the identification of Polo-like kinase-1 inhibitors from natural sources as anti-cancer agents. Int. J. Biol. Macromol. 2018, 116, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Androutsopoulos, V.P.; Mahale, S.; Arroo, R.R.; Potter, G. Anticancer effects of the flavonoid diosmetin on cell cycle progression and proliferation of MDA-MB 468 breast cancer cells due to CYP1 activation. Oncol. Rep. 2009, 21, 1525–1528. [Google Scholar] [PubMed]
- Oak, C.; Khalifa, A.; Isali, I.; Bhaskaran, N.; Walker, E.; Shukla, S. Diosmetin suppresses human prostate cancer cell proliferation through the induction of apoptosis and cell cycle arrest. Int. J. Oncol. 2018, 53, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Yan, Y.; Xiao, L.; Dai, S.; Zeng, S.; Qian, L.; Wang, L.; Yang, X.; Xiao, Y.; Gong, Z. Radiosensitizing effect of diosmetin on radioresistant lung cancer cells via Akt signaling pathway. PLoS ONE 2017, 12, e0175977. [Google Scholar] [CrossRef] [PubMed]
- Roma, A.; Rota, S.G.; Spagnuolo, P.A. Diosmetin Induces Apoptosis of Acute Myeloid Leukemia Cells. Mol. Pharm. 2018, 15, 1353–1360. [Google Scholar] [CrossRef]
- Liu, J.; Wen, X.; Liu, B.; Zhang, Q.; Zhang, J.; Miao, H.; Zhu, R. Diosmetin inhibits the metastasis of hepatocellular carcinoma cells by downregulating the expression levels of MMP-2 and MMP-9. Mol. Med. Rep. 2016, 13, 2401–2408. [Google Scholar] [CrossRef]
- Liu, B.; Shi, Y.; Peng, W.; Zhang, Q.; Liu, J.; Chen, N.; Zhu, R. Diosmetin induces apoptosis by upregulating p53 via the TGF-β signal pathway in HepG2 hepatoma cells. Mol. Med. Rep. 2016, 14, 159–164. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, J.; Jia, K.; Li, N.; Liu, B.; Zhang, Q.; Zhu, R. Diosmetin triggers cell apoptosis by activation of the p53/Bcl-2 pathway and inactivation of the Notch3/NF-κB pathway in HepG2 cells. Oncol. Lett. 2016, 12, 5122–5128. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.Y.; Yuan, D.; Yang, J.Y.; Wang, L.H.; Wu, C.F. Cytotoxic activity of flavonoids from the flowers of Chrysanthemum morifolium on human colon cancer Colon205 cells. J. Asian Nat. Prod. Res. 2009, 11, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.S.; Lin, C.R.; Du, Y.C.; Lübken, T.; Chiang, M.Y.; Chen, I.H.; Wu, C.C.; Hwang, T.L.; Chen, S.L.; Yen, M.H. Acasiane A and B and farnesirane A and B, diterpene derivatives from the roots of Acacia farnesiana. Planta Med. 2009, 75, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, S.; Wenzel, U.; Daniel, H. Comparative analysis of the effects of flavonoids on proliferation, cytotoxicity, and apoptosis in human colon cancer cell lines. Eur. J. Nutrition 1999, 38, 133–142. [Google Scholar] [CrossRef]
- Watson, A. Apoptosis and colorectal cancer. Gut 2004, 53, 1701–1709. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Koosha, S.; Mohamed, Z.; Sinniah, A.; Alshawsh, M.A. Investigation into the Molecular Mechanisms underlying the Anti-proliferative and Anti-tumorigenesis activities of Diosmetin against HCT-116 Human Colorectal Cancer. Sci. Rep. 2019, 9, 5148. [Google Scholar] [CrossRef] [PubMed]
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food sources, bioavailability, metabolism, and bioactivity. Adv. Nut. 2017, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780. [Google Scholar] [CrossRef]
- Juarez-Reyes, K.; Brindis, F.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; Bye, R.; Linares, E.; Mata, R. Hypoglycemic, antihyperglycemic, and antioxidant effects of the edible plant Anoda cristata. J. Ethnopharmacol. 2015, 161, 36–45. [Google Scholar] [CrossRef]
- Liu, X.Y.; Fan, M.L.; Wang, H.Y.; Yu, B.Y.; Liu, J.H. Metabolic profile and underlying improved bio-activity of Fructus aurantii immaturus by human intestinal bacteria. Food Funct. 2017, 8, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Malmir, M.; Gohari, A.R.; Saeidnia, S.; Silva, O. A new bioactive monoterpene-flavonoid from Satureja khuzistanica. Fitoterapia 2015, 105, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Schinella, G.R.; Giner, R.M.; Recio, M.C.; Mordujovich de Buschiazzo, P.; Rios, J.L.; Manez, S. Anti-inflammatory effects of South American Tanacetum vulgare. J. Pharm. Pharmacol. 1998, 50, 1069–1074. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gong, X.B.; Huang, L.G.; Wang, Z.X.; Wan, R.Z.; Zhang, P.; Zhang, Q.Y.; Chen, Z.; Zhang, B.S. Diosmetin exerts anti-oxidative, anti-inflammatory and anti-apoptotic effects to protect against endotoxin-induced acute hepatic failure in mice. Oncotarget 2017, 8, 30723–30733. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, Y.; Li, Q.; Shi, X.; Guo, Y. Acute and Subacute Oral Toxicity Evaluation of Eriobotrya japonica Leaf Triterpene Acids in ICR Mice. Evid.-Based Complement. Alternat. Med. 2017, 2017, 4837839. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, M.; Jestoi, M.; Nathanail, A.V.; Kokkonen, U.M.; Anttila, M.; Koivisto, P.; Karhunen, P.; Peltonen, K. Application of OECD Guideline 423 in assessing the acute oral toxicity of moniliformin. Food Chem. Toxicol. 2013, 53, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Shentu, S.; Aggarwal, B.B.; Krishnan, S. Curcumin Sensitizes Human Colorectal Cancer Xenografts in Nude Mice to;-Radiation byTargeting Nuclear Factor-KB^ Regulated Gene Products. Clin. Cancer Res. 2008, 14, 7. [Google Scholar]
- Pyo, J.H.; Byeon, S.J.; Lee, H.; Min, Y.W.; Min, B.H.; Lee, J.H.; Kim, K.M.; Ahn, H.S.; Kim, K.; Choi, Y.H. Measurement of tumor volume is not superior to diameter for prediction of lymph node metastasis in early gastric cancer with minute submucosal invasion. Oncotarget 2017, 8, 113758. [Google Scholar] [CrossRef][Green Version]
- Corwin, W.L.; Ebrahimi-Nik, H.; Floyd, S.M.; Tavousi, P.; Mandoiu, I.I.; Srivastava, P.K. Tumor Control Index as a new tool to assess tumor growth in experimental animals. J. Immunol. Methods 2017, 445, 71–76. [Google Scholar] [CrossRef]
- Aipire, A.; Li, J.; Yuan, P.; He, J.; Hu, Y.; Liu, L.; Feng, X.; Li, Y.; Zhang, F.; Yang, J. Glycyrrhiza uralensis water extract enhances dendritic cell maturation and antitumor efficacy of HPV dendritic cell-based vaccine. Sci. Rep. 2017, 7, 43796. [Google Scholar] [CrossRef]
- Hather, G.; Liu, R.; Bandi, S.; Mettetal, J.; Manfredi, M.; Shyu, W.C.; Donelan, J.; Chakravarty, A. Growth Rate Analysis and Efficient Experimental Design for Tumor Xenograft Studies: Supplementary Issue: Array Platform Modeling and Analysis (A). Cancer Inf. 2014, 13, CIN. S13974. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, A.B.; Guha, S.; Krishnan, S.; Diagaradjane, P.; Gelovani, J.; Aggarwal, B.B. Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-κB–regulated gene products. Cancer Res. 2007, 67, 3853–3861. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koosha, S.; Mohamed, Z.; Sinniah, A.; Alshawsh, M.A. Evaluation of Anti-Tumorigenic Effects of Diosmetin against Human Colon Cancer Xenografts in Athymic Nude Mice. Molecules 2019, 24, 2522. https://doi.org/10.3390/molecules24142522
Koosha S, Mohamed Z, Sinniah A, Alshawsh MA. Evaluation of Anti-Tumorigenic Effects of Diosmetin against Human Colon Cancer Xenografts in Athymic Nude Mice. Molecules. 2019; 24(14):2522. https://doi.org/10.3390/molecules24142522
Chicago/Turabian StyleKoosha, Sanaz, Zahurin Mohamed, Ajantha Sinniah, and Mohammed A. Alshawsh. 2019. "Evaluation of Anti-Tumorigenic Effects of Diosmetin against Human Colon Cancer Xenografts in Athymic Nude Mice" Molecules 24, no. 14: 2522. https://doi.org/10.3390/molecules24142522
APA StyleKoosha, S., Mohamed, Z., Sinniah, A., & Alshawsh, M. A. (2019). Evaluation of Anti-Tumorigenic Effects of Diosmetin against Human Colon Cancer Xenografts in Athymic Nude Mice. Molecules, 24(14), 2522. https://doi.org/10.3390/molecules24142522





