Anthocyanin Profile of Elderberry Juice: A Natural-Based Bioactive Colouring Ingredient with Potential Food Application
Abstract
1. Introduction
2. Results and Discussion
2.1. Bioactivity and Anthocyanin Profile of S. nigra Juice
2.2. Nutritional and Chemical Characterisation of Croissants
2.3. Colour Parameters
2.4. Antioxidant Activity and Anthocyanin Content of Prepared Croissants
3. Experimental Section
3.1. Standards and Reagents
3.2. Sample Collection and Preparation
3.3. Bioactive Properties
3.3.1. Antioxidant Activity
3.3.2. Cytotoxicity
3.4. Anthocyanin Analysis
3.5. Croissant Preparation
3.6. Nutritional Value
3.7. Evaluation of Colour Changes
3.8. Antioxidant Activity of the Prepared Croissants
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments

Conflicts of Interest
References
- Martins, N.; Roriz, C.L.; Morales, P.; Barros, L.; Ferreira, I.C.F.R. Food colorants: Challenges, opportunities and current desires of agro-industries to ensure consumer expectations and regulatory practices. Trends Food Sci. Technol. 2016, 52, 1–15. [Google Scholar] [CrossRef]
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Adding molecules to food, pros and cons: A review on synthetic and natural food additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Amchova, P.; Kotolova, H.; Ruda-Kucerova, J. Health safety issues of synthetic food colorants. Regul. Toxicol. Pharmacol. 2015, 73, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Gostner, J.M.; Becker, K.; Ueberall, F.; Fuchs, D. The good and bad of antioxidant foods: An immunological perspective. Food Chem. Toxicol. 2015, 80, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Masone, D.; Chanforan, C. Study on the interaction of artificial and natural food colorants with human serum albumin: A computational point of view. Comput. Biol. Chem. 2015, 56, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Munawar, N.; bt Hj Jamil, H.M.T. The Islamic Perspective Approach on Plant Pigments as Natural Food Colourants. Procedia Soc. Behav. Sci. 2014, 121, 193–203. [Google Scholar] [CrossRef]
- Veberic, R.; Jakopic, J.; Stampar, F.; Schmitzer, V. European elderberry (Sambucus nigra L.) rich in sugars, organic acids, anthocyanins and selected polyphenols. Food Chem. 2009, 114, 511–515. [Google Scholar] [CrossRef]
- Sidor, A.; Gramza-Michałowska, A. Advanced research on the antioxidant and health benefit of elderberry (Sambucus nigra) in food—A review. J. Funct. Foods 2015, 18, 941–958. [Google Scholar] [CrossRef]
- Młynarczyk, K.; Walkowiak-Tomczak, D.; Łysiak, G.P. Bioactive properties of Sambucus nigra L. As a functional ingredient for food and pharmaceutical industry. J. Funct. Foods 2018, 40, 377–390. [Google Scholar] [CrossRef]
- Swer, T.L.; Chauhan, K. Stability studies of enzyme aided anthocyanin extracts from Prunus nepalensis L. LWT 2019, 102, 181–189. [Google Scholar] [CrossRef]
- Sun-Waterhouse, D.; Jin, D.; Waterhouse, G.I.N. Effect of adding elderberry juice concentrate on the quality attributes, polyphenol contents and antioxidant activity of three fibre-enriched pastas. Food Res. Int. 2013, 54, 781–789. [Google Scholar] [CrossRef]
- Massoud, R.; Khosravi-Darani, K.; Nakhsaz, F.; Varga, L. Evaluation of Physicochemical, Microbiological and Sensory Properties of Croissants Fortified with Arthrospira platensis (Spirulina). Czech J. Food Sci. 2016, 4, 350–355. [Google Scholar] [CrossRef]
- Fallaize, R.; Wilson, L.; Gray, J.; Morgan, L.M.; Griffin, B.A. Variation in the effects of three different breakfast meals on subjective satiety and subsequent intake of energy at lunch and evening meal. Eur. J. Nutr. 2013, 52, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Raposo, A.; Carrascosa, C.; Pérez, E.; Tavares, A.; Sanjuán, E.; Saavedra, P.; Millán, R. Vending machine foods: Evaluation of nutritional composition. Ital. J. Food Sci. 2016, 28, 448–463. [Google Scholar]
- Olivas-Aguirre, F.J.; Rodrigo-García, J.; Martínez-Ruiz, N.D.R.; Cárdenas-Robles, A.I.; Mendoza-Díaz, S.O.; Álvarez-Parrilla, E.; González-Aguilar, G.A.; De La Rosa, L.A.; Ramos-Jiménez, A.; Wall-Medrano, A. Cyanidin-3-O-glucoside: Physical-chemistry, foodomics and health effects. Molecules 2016, 21, 1264. [Google Scholar] [CrossRef] [PubMed]
- Duymuş, H.G.; Göger, F.; Başer, K.H.C. In vitro antioxidant properties and anthocyanin compositions of elderberry extracts. Food Chem. 2014, 155, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.; Ferreira, S.; Nunes, F.M. Elderberry (Sambucus nigra L.) by-products a source of anthocyanins and antioxidant polyphenols. Ind. Crops Prod. 2017, 95, 227–234. [Google Scholar] [CrossRef]
- Wallace, T.; Giusti, M. Anthocyanins. Adv. Nutr. 2015, 6, 620–622. [Google Scholar] [CrossRef]
- Van Wymelbeke, V.; Brondel, L.; Bon, F.; Martin-Pfitzenmeyer, I.; Manckoundia, P. An innovative brioche enriched in protein and energy improves the nutritional status of malnourished nursing home residents compared to oral nutritional supplement and usual breakfast: FARINE+ project. Clin. Nutr. ESPEN 2016, 15, 93–100. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Chawla, S.P.; Chander, R.; Sharma, A. Antioxidant properties of Maillard reaction products obtained by gamma-irradiation of whey proteins. Food Chem. 2009, 116, 122–128. [Google Scholar] [CrossRef]
- Bessada, S.M.F.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb.f.: An underexploited and highly disseminated species. Ind. Crops Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef]
- Barros, L.; Pereira, E.; Calhelha, R.C.; Dueñas, M.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Bioactivity and chemical characterization in hydrophilic and lipophilic compounds of Chenopodium ambrosioides L. J. Funct. Foods 2013, 5, 1732–1740. [Google Scholar] [CrossRef]
- Guimarães, R.; Barros, L.; Dueñas, M.; Carvalho, A.M.; Queiroz, M.J.R.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterisation of phenolic compounds in wild fruits from Northeastern Portugal. Food Chem. 2013, 141, 3721–3730. [Google Scholar] [CrossRef] [PubMed]
- Lockowandt, L.; Pinela, J.; Roriz, C.L.; Pereira, C.; Abreu, R.M.V.; Calhelha, R.C.; Alves, M.J.; Barros, L.; Bredol, M.; Ferreira, I.C.F.R. Chemical features and bioactivities of cornflower (Centaurea cyanus L.) capitula: The blue flowers and the unexplored non-edible part. Ind. Crops Prod. 2019, 128, 496–503. [Google Scholar] [CrossRef]
- AOAC Official Methods of Analysis. Assoc. Off. Anal. Chem. Int. 2016, 38, 431.
- Caleja, C.; Barros, L.; Barreira, J.C.M.; Ciric, A.; Sokovic, M.; Calhelha, R.C.; Beatriz, M.; Oliveira, P.P.; Ferreira, I.C.F.R. Suitability of lemon balm (Melissa officinalis L.) extract rich in rosmarinic acid as a potential enhancer of functional properties in cupcakes. Food Chem. 2018, 250, 67–74. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples are available from the authors. |

| Antioxidant Activity (EC50, mg/mL) | |||||
| DPPH Radical-Scavenging | Reducing Power | β-Carotene Bleaching Inhibition | |||
| 3.1 ± 0.1 | 3.7 ± 0.2 | 9.4 ± 0.3 | |||
| Antitumor Activity (GI50, μg/mL) | |||||
| MCF-7 | HeLa | NCI-H460 | HepG2 | PLP2 | |
| 58 ± 1 | 16 ± 1 | 16 ± 1 | 98 ± 4 | >400 | |
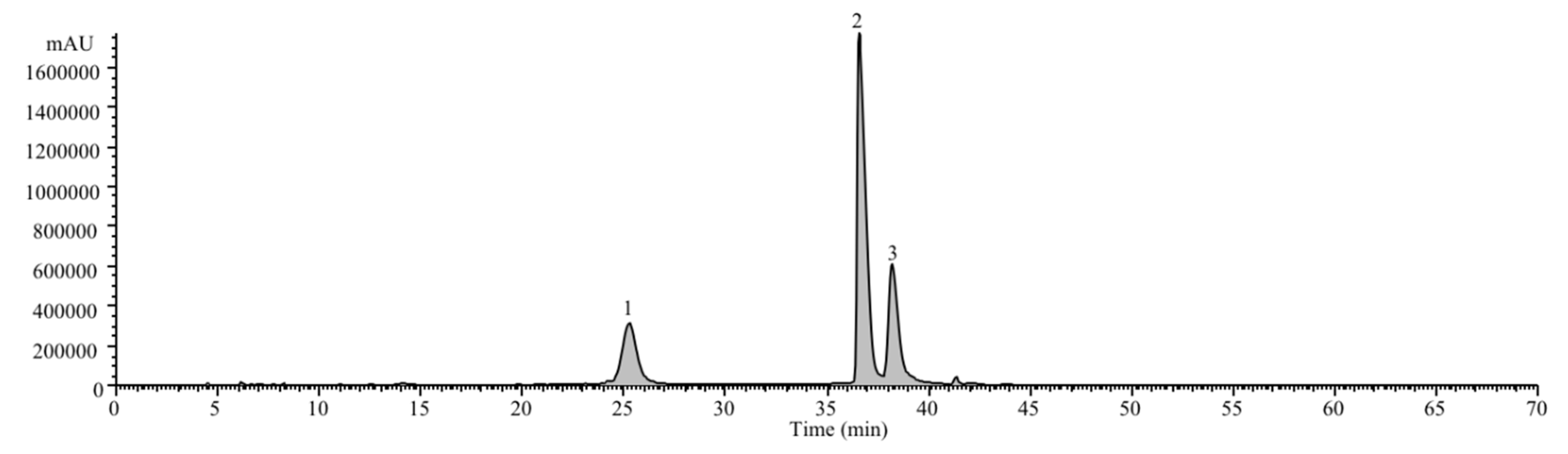
| Anthocyanins Profile of S. nigra | |||||
| Peak | RT (min) | λmax (nm) | Pseudomolecular ion [M + H]+ (m/z) | Identification | Quantification (μg/mL) |
| 1 | 25.31 | 515 | 581(79), 449(100), 287(77) | Cyanidin-3-O-sambubioside-5-O-glucoside | 140 ± 21 |
| 2 | 36.63 | 517 | 287(100) | Cyanidin-3-O-sambubioside | 560 ± 45 |
| 3 | 38.26 | 517 | 287(100) | Cyanidin-3-O-glucoside | 390 ± 30 |
| Total anthocyanins | 1090± 35 | ||||
| Sample | Nutritional Value (g/100 g) | Energy (Kcal/100 g) | Free Sugars (g/100 g) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Moisture | Fat | Proteins | Carbohydrates | Ash | Fructose | Glucose | Maltose | Trehalose | Sugars | ||
| BC | 0.23 ± 0.02 | 17.0 ± 0.4 a | 10.3 ± 0.2 b | 70 ± 1 | 2.0 ± 0.1 | 474 ± 2 ab | 4.1 ± 0.1 b | 2.4 ± 0.1 b | 3.9 ± 0.2 a | 1.8 ± 0.1 a | 12.2 ± 0.2 b |
| EJ2 | 0.23 ± 0.03 | 16.7 ± 0.3 b | 10.4 ± 0.3 b | 71 ± 1 | 2.1 ± 0.1 | 476 ± 4 a | 5.1 ± 0.3 a | 2.4 ± 0.3 b | 3.7 ± 0.1 b | 1.6 ± 0.1 b | 12.8 ± 0.5 ab |
| EJ4 | 0.24 ± 0.02 | 16.4 ± 0.4 bc | 11.0 ± 0.1 a | 70 ± 1 | 2.1 ± 0.2 | 472 ± 4 b | 5.2 ± 0.5 a | 2.7 ± 0.5 b | 3.6 ± 0.1 b | 1.4 ± 0.1 c | 12.9 ± 0.5 ab |
| EJ8 | 0.24 ± 0.03 | 16.1 ± 0.2 c | 11.1 ± 0.1 a | 71 ± 1 | 2.0 ± 0.1 | 473 ± 2 b | 5.6 ± 0.5 a | 3.4 ± 0.5 a | 4.0 ± 0.1 a | 1.3 ± 0.2 cd | 14,3 ± 0.5 a |
| CCD | 0.24 ± 0.02 | 16.5 ± 0.2 b | 10.9 ± 0.1 a | 70 ± 1 | 2.0 ± 0.1 | 472 ± 2 b | 4.2 ± 0.2 b | 1.6 ± 0.1 c | 3.6 ± 0.1 b | 1.2 ± 0.2 d | 10.6 ± 0.2 c |
| Homoscedasticity 1 (p value) (n = 45) | <0.001 | 0.006 | <0.001 | 0.012 | <0.001 | 0.001 | <0.001 | <0.001 | <0.001 | 0.003 | <0.001 |
| ANOVA 2 (p value) (n = 45) | 0.191 | 0.001 | <0.001 | 0.071 | 0.084 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| C6:0 | C10:0 | C12:0 | C14:0 | C16:0 | C16:1 | C18:0 | C18:1 | C18:2 | C18:3n3 | SFA | MUFA | PUFA | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BC | 0.9 ± 0.1 c | 1.4 ± 0.1 c | 1.9 ± 0.1 c | 5.9 ± 0.1 c | 21 ± 1 b | 1.0 ± 0.1 d | 7.0 ± 0.1 b | 28 ± 1 cd | 28 ± 1 a | 0.7 ± 0.1 | 41 ± 1 c | 30 ± 1 c | 29 ± 1 b |
| EJ2 | 1.0 ± 0.2 c | 1.3 ± 0.1 c | 1.7 ± 0.3 c | 4.7 ± 0.5 d | 17 ± 1 c | 1.1 ± 0.2 cd | 6.8 ± 0.4 b | 28 ± 1 d | 29 ± 2 a | 1.3 ± 0.1 | 37 ± 2 d | 30 ± 1 bc | 33 ± 3 a |
| EJ4 | 1.3 ± 0.1 b | 1.7 ± 0.2 b | 2.2 ± 0.2 b | 6.3 ± 0.4 bc | 19 ± 1 c | 1.4 ± 0.1 ab | 8.0 ± 0.1 a | 30 ± 1 a | 22 ± 1 b | 1.3 ± 0.1 | 44 ± 1 b | 32 ± 1 a | 24 ± 1 c |
| EJ8 | 1.6 ± 0.1 a | 1.9 ± 0.1 a | 2.5 ± 0.1 a | 7.6 ± 0.1 a | 24 ± 3 a | 1.3 ± 0.1 bc | 8.3 ± 0.5 a | 29 ± 1 bc | 20 ± 2 b | 1.0 ± 0.1 | 47 ± 2 a | 31 ± 1 b | 22 ± 2 c |
| CCD | 1.0 ± 0.1 c | 1.8 ± 0.2 ab | 2.2 ± 0.2 b | 6.8 ± 0.5 ab | 21 ± 1 b | 1.6 ± 0.2 a | 8.3 ± 0.4 a | 29 ± 1 ab | 22 ± 2 b | 0.9 ± 0.1 | 44 ± 1 b | 32 ± 1 a | 24 ± 2 c |
| Homoscedasticity 1 (p value) (n = 45) | <0.001 | 0.003 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.001 |
| ANOVA2 (p value) (n = 45) | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.017 | <0.001 | <0.001 | 0.042 | 0.072 | 0.041 | 0.006 | <0.001 |
| L* | a* | b* | |
|---|---|---|---|
| BC | 79 ± 1 a | 6.4 ± 0.1 c | 31 ± 1 a |
| EJ2 | 62 ± 1 c | 8.5 ± 0.4 b | 19 ± 1 b |
| EJ4 | 57 ± 1 d | 13.9 ± 0.1a | 12 ± 1c |
| EJ8 | 54 ± 1 e | 14.0 ± 0.1a | 9 ± 1d |
| CCD | 69 ± 3 b | 8.6 ± 0.1 b | 19 ± 1 b |
| Homoscedasticity 1 (p-value) (n = 45) | <0.001 | <0.001 | <0.001 |
| ANOVA 2 (p-value) (n = 45) | <0.001 | <0.001 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, R.F.R.; Barreira, J.C.M.; Heleno, S.A.; Barros, L.; Calhelha, R.C.; Ferreira, I.C.F.R. Anthocyanin Profile of Elderberry Juice: A Natural-Based Bioactive Colouring Ingredient with Potential Food Application. Molecules 2019, 24, 2359. https://doi.org/10.3390/molecules24132359
da Silva RFR, Barreira JCM, Heleno SA, Barros L, Calhelha RC, Ferreira ICFR. Anthocyanin Profile of Elderberry Juice: A Natural-Based Bioactive Colouring Ingredient with Potential Food Application. Molecules. 2019; 24(13):2359. https://doi.org/10.3390/molecules24132359
Chicago/Turabian Styleda Silva, Ricardo F. R., João C. M. Barreira, Sandrina A. Heleno, Lillian Barros, Ricardo C. Calhelha, and Isabel C. F. R. Ferreira. 2019. "Anthocyanin Profile of Elderberry Juice: A Natural-Based Bioactive Colouring Ingredient with Potential Food Application" Molecules 24, no. 13: 2359. https://doi.org/10.3390/molecules24132359
APA Styleda Silva, R. F. R., Barreira, J. C. M., Heleno, S. A., Barros, L., Calhelha, R. C., & Ferreira, I. C. F. R. (2019). Anthocyanin Profile of Elderberry Juice: A Natural-Based Bioactive Colouring Ingredient with Potential Food Application. Molecules, 24(13), 2359. https://doi.org/10.3390/molecules24132359










