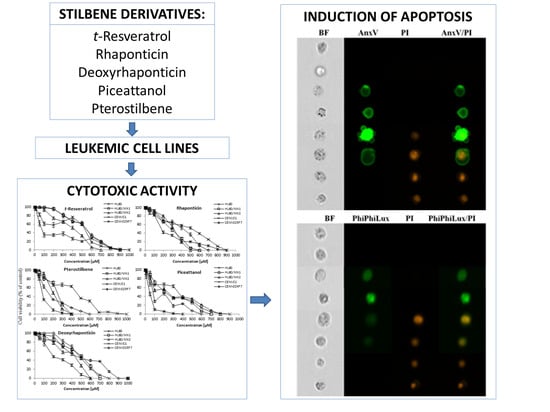
Imaging Flow Cytometric Analysis of Stilbene-Dependent Apoptosis in Drug Resistant Human Leukemic Cell Lines
Abstract
1. Introduction
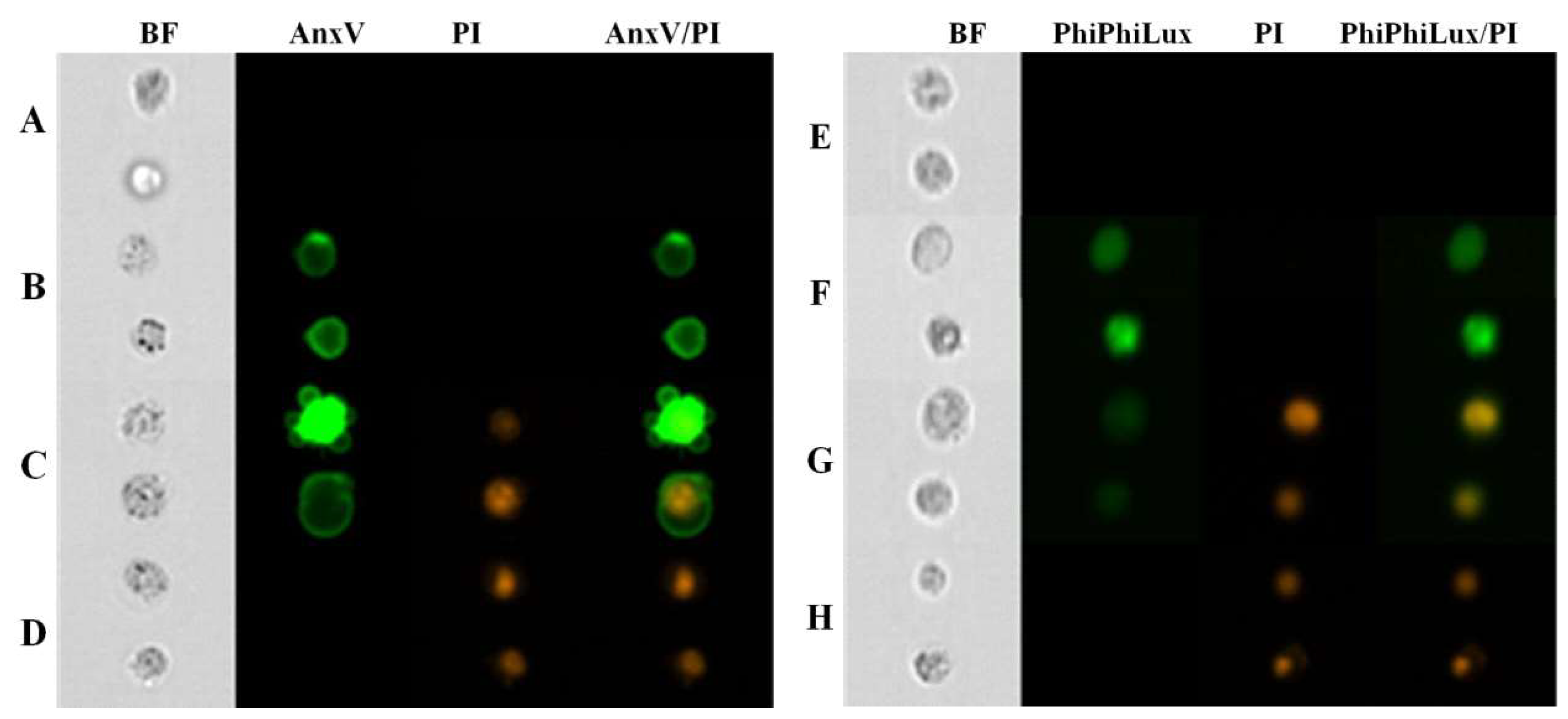
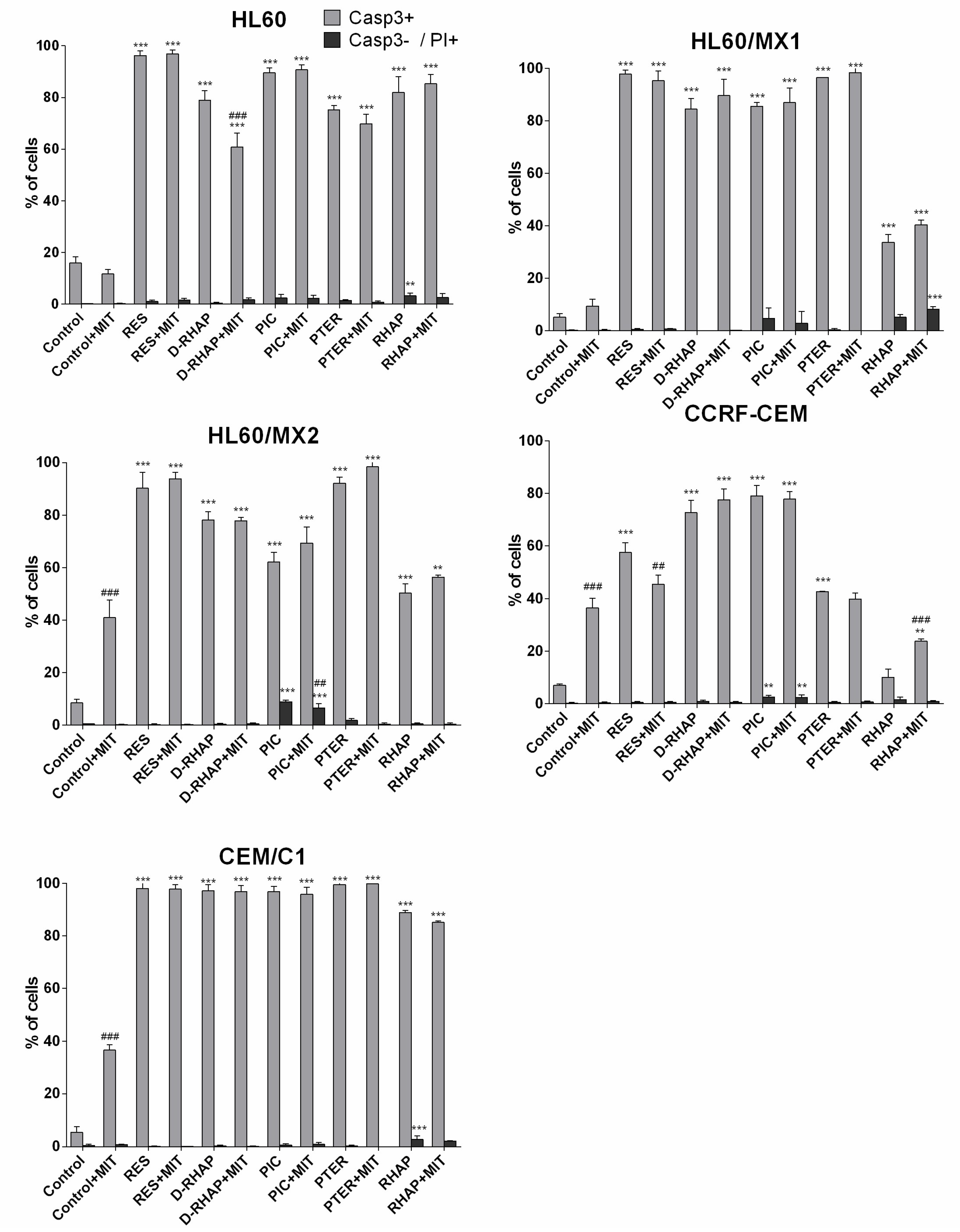
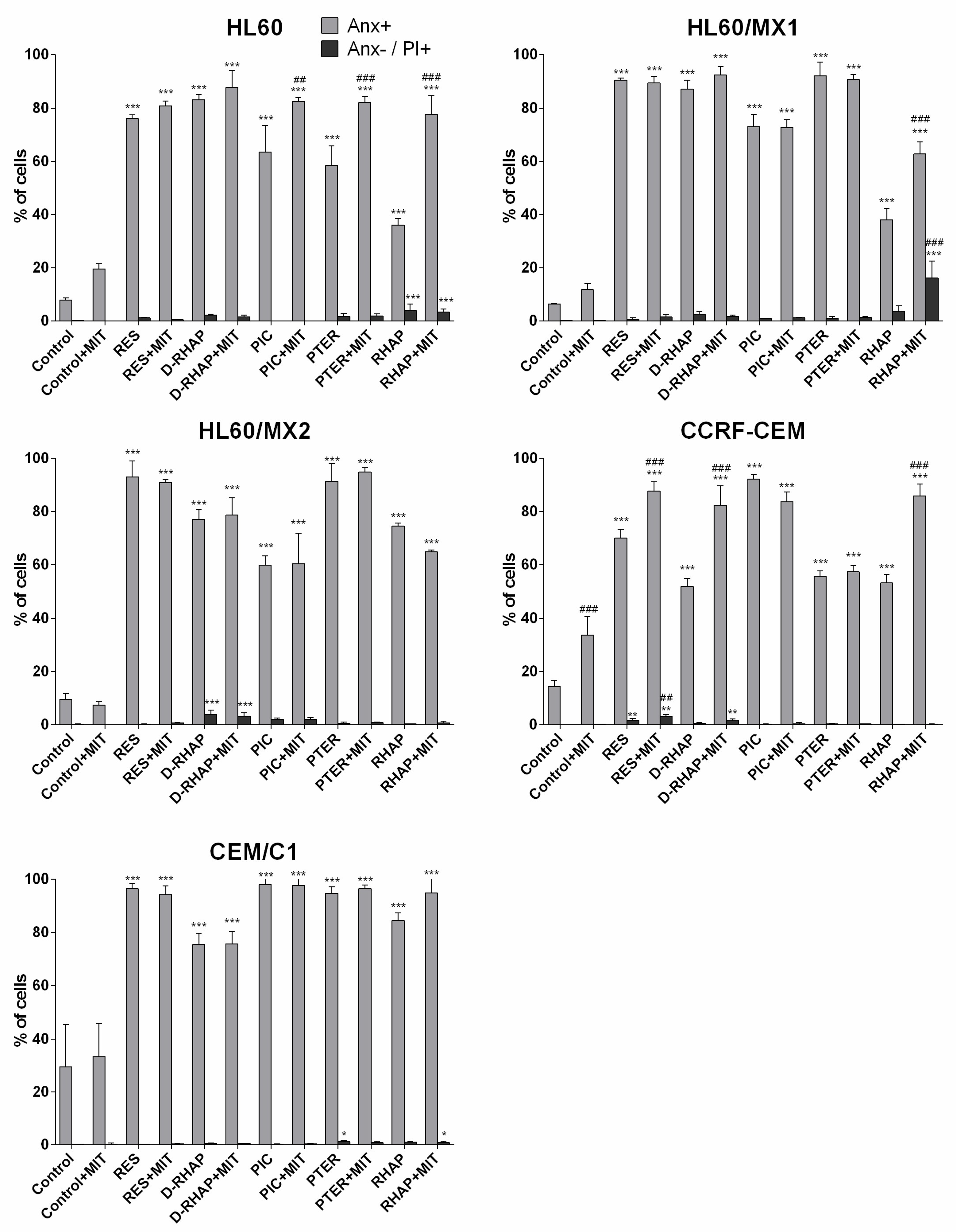
2. Results
3. Discussion
4. Materials and Methods
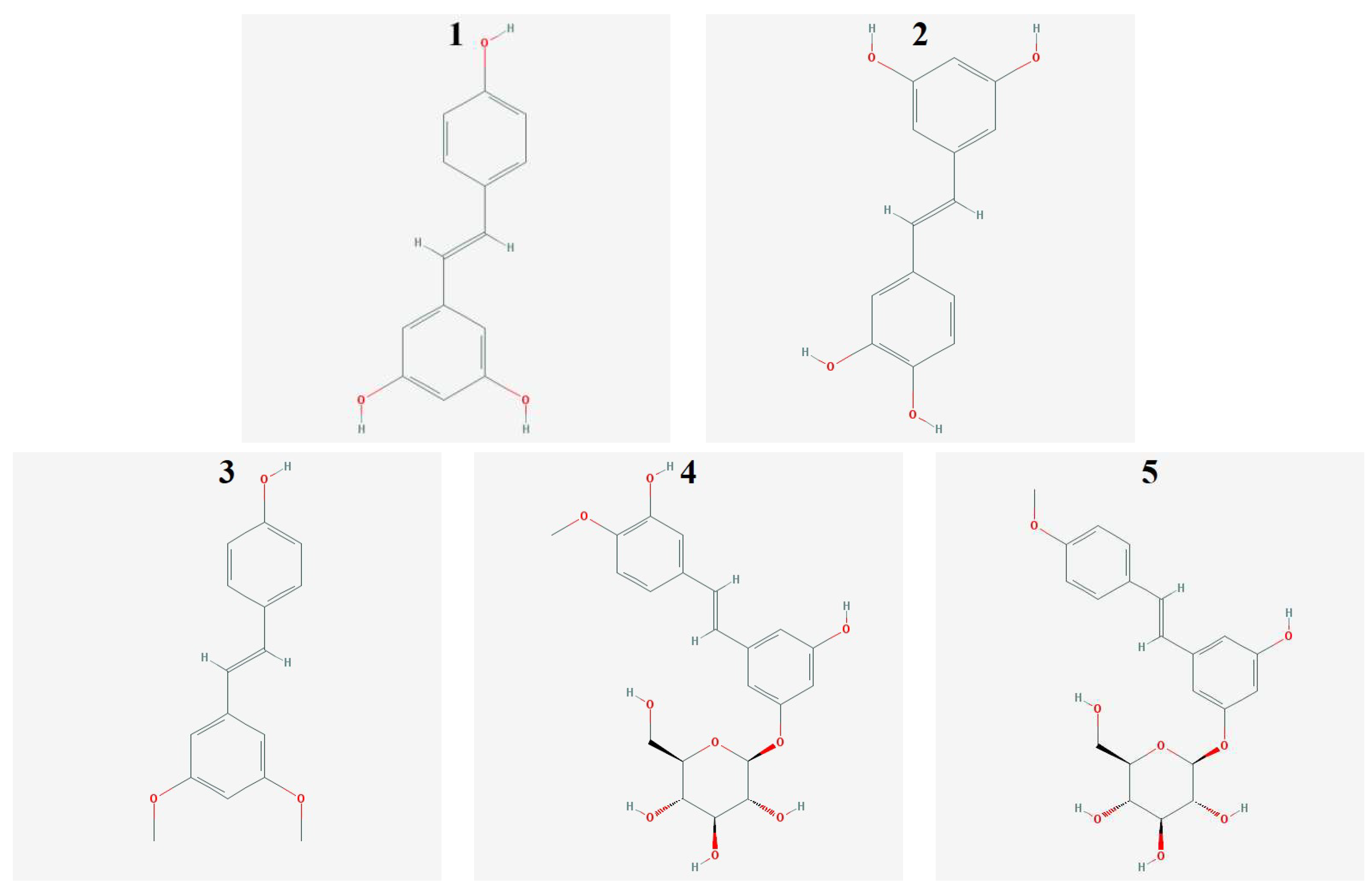
4.1. Stilbene Derivatives
4.2. Cell Lines and Cell Culture
4.3. Analysis of Cell Viability
4.4. Leukemic Cells Preparation
4.5. Annexin V-FITC/PI Assay
4.6. PhiPhiLux-G1D2/PI Assay
4.7. FlowSight Cytometric Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Efferth, T.; Volm, M. Multiple resistance to carcinogens and xenobiotics: P-glycoproteins as universal detoxifiers. Arch. Toxicol. 2017, 91, 2515–2538. [Google Scholar] [CrossRef]
- Chen, Z.; Shi, T.; Zhang, L.; Zhu, P.; Deng, M.; Huang, C.; Hu, T.; Jiang, L.; Li, J. Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: A review of the past decade. Cancer Lett. 2016, 370, 153–164. [Google Scholar] [CrossRef] [PubMed]
- López-Marqués, R.L.; Poulsen, L.R.; Bailly, A.; Geisler, M.; Pomorski, T.G.; Palmgren, M.G. Structure and mechanism of ATP-dependent phospholipid transporters. Biochim. Biophys. ActaGen. Subj. 2015, 1850, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, J.I.; Williams, R.T.; Henderson, M.J.; Norris, M.D.; Haber, M. ABC transporters as mediators of drug resistance and contributors to cancer cell biology. Drug Resist. Updat. 2016, 26, 1–9. [Google Scholar] [CrossRef]
- Senthilkumar, R.; CHEN, B.-A.; CAI, X.-H.; FU, R. Anticancer and multidrug-resistance reversing potential of traditional medicinal plants and their bioactive compounds in leukemia cell lines. Chin. J. Nat. Med. 2014, 12, 881–894. [Google Scholar] [CrossRef]
- Wu, C.-P.; Calcagno, A.M.; Ambudkar, S. V Reversal of ABC drug transporter-mediated multidrug resistance in cancer cells: evaluation of current strategies. Curr. Mol. Pharmacol. 2008, 1, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Gillet, J.-P.; Gottesman, M.M. Mechanisms of Multidrug Resistance in Cancer. In Methods in Molecular Biology (Clifton, N.J.); Zhou, J., Ed.; Humana Press: New York, NY, USA, 2010; Volume 596, pp. 47–76. [Google Scholar]
- Pui, C.-H.; Robison, L.L.; Look, A.T. Acute lymphoblastic leukaemia. Lancet 2008, 371, 1030–1043. [Google Scholar] [CrossRef]
- Inaba, H.; Greaves, M.; Mullighan, C.G. Acute lymphoblastic leukaemia. Lancet 2013, 381, 1943–1955. [Google Scholar] [CrossRef]
- Wang, Z.-Y.; Chen, Z. Acute promyelocytic leukemia: from highly fatal to highly curable. Blood 2008, 111, 2505–2515. [Google Scholar] [CrossRef]
- Park, J.H.; Qiao, B.; Panageas, K.S.; Schymura, M.J.; Jurcic, J.G.; Rosenblat, T.L.; Altman, J.K.; Douer, D.; Rowe, J.M.; Tallman, M.S. Early death rate in acute promyelocytic leukemia remains high despite all-trans retinoic acid. Blood 2011, 118, 1248–1254. [Google Scholar] [CrossRef] [PubMed]
- Roupe, K.A.; Remsberg, C.M.; Yáñez, J.A.; Davies, N.M. Pharmacometrics of stilbenes: seguing towards the clinic. Curr. Clin. Pharmacol. 2006, 1, 81–101. [Google Scholar] [CrossRef]
- Chong, J.; Poutaraud, A.; Hugueney, P. Metabolism and roles of stilbenes in plants. Plant Sci. 2009, 177, 143–155. [Google Scholar] [CrossRef]
- Signorelli, P.; Ghidoni, R. Resveratrol as an anticancer nutrient: Molecular basis, open questions and promises. J. Nutr. Biochem. 2005, 16, 449–466. [Google Scholar] [CrossRef]
- Ferreira, M.-J.U.; Duarte, N.; Gyémánt, N.; Radics, R.; Cherepnev, G.; Varga, A.; Molnár, J. Interaction between doxorubicin and the resistance modifier stilbene on multidrug resistant mouse lymphoma and human breast cancer cells. Anticancer Res. 2006, 26, 3541–3546. [Google Scholar]
- Ruiz, B.R.; Hernández, S.P. Cancer chemoprevention by dietary phytochemicals: Epidemiological evidence. Maturitas 2016, 94, 13–19. [Google Scholar] [CrossRef]
- Tsai, H.-Y.; Ho, C.-T.; Chen, Y.-K. Biological actions and molecular effects of resveratrol, pterostilbene, and 3′-hydroxypterostilbene. J. food drug Anal. 2017, 25, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Varoni, E.M.; Lo Faro, A.F.; Sharifi-Rad, J.; Iriti, M. Anticancer Molecular Mechanisms of Resveratrol. Front. Nutr. 2016, 3, 8. [Google Scholar] [CrossRef]
- Estrov, Z.; Shishodia, S.; Faderl, S.; Harris, D.; Van, Q.; Kantarjian, H.M.; Talpaz, M.; Aggarwal, B.B. Resveratrol blocks interleukin-1beta-induced activation of the nuclear transcription factor NF-kappaB, inhibits proliferation, causes S-phase arrest, and induces apoptosis of acute myeloid leukemia cells. Blood 2003, 102, 987–995. [Google Scholar] [CrossRef]
- Singh, C.K.; Ndiaye, M.A.; Ahmad, N. Resveratrol and cancer: Challenges for clinical translation. Biochim. Biophys. Acta - Mol. Basis Dis. 2015, 1852, 1178–1185. [Google Scholar] [CrossRef]
- Piver, B.; Fer, M.; Vitrac, X.; Merillon, J.-M.; Dreano, Y.; Berthou, F.; Lucas, D. Involvement of cytochrome P450 1A2 in the biotransformation of trans-resveratrol in human liver microsomes. Biochem. Pharmacol. 2004, 68, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Bavaresco, L.; Vezzulli, S.; Battilani, P.; Giorni, P.; Pietri, A.; Bertuzzi, T. Effect of Ochratoxin A-Producing Aspergilli on Stilbenic Phytoalexin Synthesis in Grapes. J. Agric. Food Chem. 2003, 51, 6151–6157. [Google Scholar] [CrossRef]
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, J.; Ballington, J.R. Resveratrol, Pterostilbene, and Piceatannol in Vaccinium Berries. J. Agric. Food Chem. 2004, 52, 4713–4719. [Google Scholar] [CrossRef]
- Brinker, A.M.; Seigler, D.S. Isolation and identification of piceatannol as a phytoalexin from sugarcane. Phytochemistry 1991, 30, 3229–3232. [Google Scholar] [CrossRef]
- Cantos, E.; Espín, J.C.; Fernández, M.J.; Oliva, J.; Tomás-Barberán, F.A. Postharvest UV-C-Irradiated Grapes as a Potential Source for Producing Stilbene-Enriched Red Wines. J. Agric. Food Chem. 2003, 51, 1208–1214. [Google Scholar] [CrossRef] [PubMed]
- Wieder, T.; Prokop, A.; Bagci, B.; Essmann, F.; Bernicke, D.; Schulze-Osthoff, K.; Dörken, B.; Schmalz, H.G.; Daniel, P.T.; Henze, G. Piceatannol, a hydroxylated analog of the chemopreventive agent resveratrol, is a potent inducer of apoptosis in the lymphoma cell line BJAB and in primary, leukemic lymphoblasts. Leukemia 2001, 15, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Remsberg, C.M.; Yáñez, J.A.; Ohgami, Y.; Vega-Villa, K.R.; Rimando, A.M.; Davies, N.M. Pharmacometrics of pterostilbene: preclinical pharmacokinetics and metabolism, anticancer, antiinflammatory, antioxidant and analgesic activity. Phyther. Res. 2008, 22, 169–179. [Google Scholar] [CrossRef]
- Liang, X.; Sun, Y.; Zeng, W.; Liu, L.; Ma, X.; Zhao, Y.; Fan, J. Synthesis and biological evaluation of a folate-targeted rhaponticin conjugate. Bioorg. Med. Chem. 2013, 21, 178–185. [Google Scholar] [CrossRef]
- Li, P.; Tian, W.; Wang, X.; Ma, X. Inhibitory effect of desoxyrhaponticin and rhaponticin, two natural stilbene glycosides from the Tibetan nutritional food Rheum tanguticum Maxim. ex Balf., on fatty acid synthase and human breast cancer cells. Food Funct. 2014, 5, 251–256. [Google Scholar] [CrossRef]
- Portt, L.; Norman, G.; Clapp, C.; Greenwood, M.; Greenwood, M.T. Anti-apoptosis and cell survival: A review. Biochim. Biophys. Acta - Mol. Cell Res. 2011, 1813, 238–259. [Google Scholar] [CrossRef] [PubMed]
- Van Engeland, M.; Nieland, L.J.W.; Ramaekers, F.C.S.; Schutte, B.; Reutelingsperger, C.P.M. Annexin V-affinity assay: A review on an apoptosis detection system based on phosphatidylserine exposure. Cytometry 1998, 31, 1–9. [Google Scholar] [CrossRef]
- Hsiao, P.-C.; Chou, Y.-E.; Tan, P.; Lee, W.-J.; Yang, S.-F.; Chow, J.-M.; Chen, H.-Y.; Lin, C.-H.; Lee, L.-M.; Chien, M.-H. Pterostilbene Simultaneously Induced G0/G1-Phase Arrest and MAPK-Mediated Mitochondrial-Derived Apoptosis in Human Acute Myeloid Leukemia Cell Lines. PLoS One 2014, 9, e105342. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Park, Y.H.; Choi, S.W.; Yang, E.K.; Lee, W.J. Resveratrol derivatives potently induce apoptosis in human promyelocytic leukemia cells. Exp. Mol. Med. 2003, 35, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.-H.; Moon, D.-O.; Choi, Y.H.; Choi, I.-W.; Moon, S.-K.; Kim, W.-J.; Kim, G.-Y. Piceatannol enhances TRAIL-induced apoptosis in human leukemia THP-1 cells through Sp1- and ERK-dependent DR5 up-regulation. Toxicol. Vitr. 2011, 25, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Keum, Y.-S.; Jeong, W.-S.; Tony Kong, A.N. Chemoprevention by isothiocyanates and their underlying molecular signaling mechanisms. Mutat. Res. Mol. Mech. Mutagen. 2004, 555, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Filomeni, G.; Graziani, I.; Rotilio, G.; Ciriolo, M.R. trans-Resveratrol induces apoptosis in human breast cancer cells MCF-7 by the activation of MAP kinases pathways. Genes Nutr. 2007, 2, 295–305. [Google Scholar] [CrossRef]
- Cecchinato, V.; Chiaramonte, R.; Nizzardo, M.; Cristofaro, B.; Basile, A.; Sherbet, G.V.; Comi, P. Resveratrol-induced apoptosis in human T-cell acute lymphoblastic leukaemia MOLT-4 cells. Biochem. Pharmacol. 2007, 74, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Tsan, M.F.; White, J.E.; Maheshwari, J.G.; Bremner, T.A.; Sacco, J. Resveratrol induces Fas signalling-independent apoptosis in THP-1 human monocytic leukaemia cells. Br. J. Haematol. 2000, 109, 405–412. [Google Scholar] [CrossRef]
- Roman, V.; Billard, C.; Kern, C.; Ferry-Dumazet, H.; Izard, J.-C.; Mohammad, R.; Mossalayi, D.M.; Kolb, J.-P. Analysis of resveratrol-induced apoptosis in human B-cell chronic leukaemia. Br. J. Haematol. 2002, 117, 842–851. [Google Scholar] [CrossRef]
- Dörrie, J.; Gerauer, H.; Wachter, Y.; Zunino, S.J. Resveratrol induces extensive apoptosis by depolarizing mitochondrial membranes and activating caspase-9 in acute lymphoblastic leukemia cells. Cancer Res. 2001, 61, 4731–4739. [Google Scholar]
- Opydo-Chanek, M.; Rak, A.; Cierniak, A.; Mazur, L. Combination of ABT-737 and resveratrol enhances DNA damage and apoptosis in human T-cell acute lymphoblastic leukemia MOLT-4 cells. Toxicol. Vitr. 2017, 42, 38–46. [Google Scholar] [CrossRef]
- Tolomeo, M.; Grimaudo, S.; Di Cristina, A.; Roberti, M.; Pizzirani, D.; Meli, M.; Dusonchet, L.; Gebbia, N.; Abbadessa, V.; Crosta, L.; et al. Pterostilbene and 3′-hydroxypterostilbene are effective apoptosis-inducing agents in MDR and BCR-ABL-expressing leukemia cells. Int. J. Biochem. Cell Biol. 2005, 37, 1709–1726. [Google Scholar] [CrossRef]
- Siedlecka-Kroplewska, K.; Jozwik, A.; Kaszubowska, L.; Kowalczyk, A.; Boguslawski, W. Pterostilbene induces cell cycle arrest and apoptosis in MOLT4 human leukemia cells. Folia Histochem. Cytobiol. 2012, 50, 574–580. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, B.; Ammazzalorso, A.; Fantacuzzi, M.; Giampietro, L.; Maccallini, C.; Amoroso, R. Anticancer Activity of Stilbene-Based Derivatives. ChemMedChem 2017, 12, 558–570. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.A.; Kishino, K.; Satoh, R.; Hashimoto, K.; Kikuchi, H.; Nishikawa, H.; Shirataki, Y.; Sakagami, H. Tumor-specificity and apoptosis-inducing activity of stilbenes and flavonoids. Anticancer Res. 2005, 25, 2055–2063. [Google Scholar] [PubMed]
- Wang, B.H.; Lu, Z.X.; Polya, G.M. Inhibition of eukaryote serine/threonine-specific protein kinases by piceatannol. Planta Med. 1998, 64, 195–199. [Google Scholar] [CrossRef]
- Ren, W.; Qiao, Z.; Wang, H.; Zhu, L.; Zhang, L. Flavonoids: Promising anticancer agents. Med. Res. Rev. 2003, 23, 519–534. [Google Scholar] [CrossRef]
- Kang, T.; Liang, N. Studies on the inhibitory effects of quercetin on the growth of HL-60 leukemia cells. Biochem. Pharmacol. 1997, 54, 1013–1018. [Google Scholar] [CrossRef]
- Dajas, F. Life or death: Neuroprotective and anticancer effects of quercetin. J. Ethnopharmacol. 2012, 143, 383–396. [Google Scholar] [CrossRef]
- Kubrak, T.; Podgórski, R.; Stompor, M. Natural and Synthetic Coumarins and their Pharmacological Activity. Eur. J. Clin. Exp. Med. 2017, 15, 169–175. [Google Scholar] [CrossRef]
- Kubrak, T.; Bogucka-Kocka, A.; Komsta, Ł.; Załuski, D.; Bogucki, J.; Galkowski, D.; Kaczmarczyk, R.; Feldo, M.; Cioch, M.; Kocki, J. Modulation of Multidrug Resistance Gene Expression by Coumarin Derivatives in Human Leukemic Cells. Oxid. Med. Cell. Longev. 2017, 2017, 5647281. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, L.; Gonzalez, V. Selected isothiocyanates rapidly induce growth inhibition of cancer cells. Mol. Cancer Ther. 2003, 2, 1045–1052. [Google Scholar] [PubMed]
- Jakubikova, J.; Bao, Y.; Sedlak, J. Isothiocyanates induce cell cycle arrest, apoptosis and mitochondrial potential depolarization in HL-60 and multidrug-resistant cell lines. Anticancer Res. 2005, 25, 3375–3386. [Google Scholar] [PubMed]
- Roupe, K.A.; Yáñez, J.A.; Teng, X.W.; Davies, N.M. Pharmacokinetics of selected stilbenes: rhapontigenin, piceatannol and pinosylvin in rats. J. Pharm. Pharmacol. 2006, 58, 1443–1450. [Google Scholar] [CrossRef]
- Wang, P.; Sang, S. Metabolism and pharmacokinetics of resveratrol and pterostilbene. BioFactors 2018, 44, 16–25. [Google Scholar] [CrossRef]
- Vervandier-Fasseur, D.; Vang, O.; Latruffe, N. Special Issue: Improvements for Resveratrol Efficacy. Molecules 2017, 22, 1737. [Google Scholar] [CrossRef]
- He, H.; Ge, Y.; Dai, H.; Cui, S.; Ye, F.; Jin, J.; Shi, Y.; He, H.; Ge, Y.; Dai, H.; et al. Design, Synthesis and Biological Evaluation of Stilbene Derivatives as Novel Inhibitors of Protein Tyrosine Phosphatase 1B. Molecules 2016, 21, 1722. [Google Scholar] [CrossRef] [PubMed]
- Chalal, M.; Delmas, D.; Meunier, P.; Latruffe, N.; Vervandier-Fasseur, D.; Chalal, M.; Delmas, D.; Meunier, P.; Latruffe, N.; Vervandier-Fasseur, D. Inhibition of Cancer Derived Cell Lines Proliferation by Synthesized Hydroxylated Stilbenes and New Ferrocenyl-Stilbene Analogs. Comparison with Resveratrol. Molecules 2014, 19, 7850–7868. [Google Scholar] [CrossRef]
- Pentek, T.; Newenhouse, E.; O’Brien, B.; Chauhan, A.S. Development of a Topical Resveratrol Formulation for Commercial Applications Using Dendrimer Nanotechnology. Molecules 2017, 22, 137. [Google Scholar] [CrossRef]
- Collins, S.J. The HL-60 promyelocytic leukemia cell line: proliferation, differentiation, and cellular oncogene expression. Blood 1987, 70, 1233–1244. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
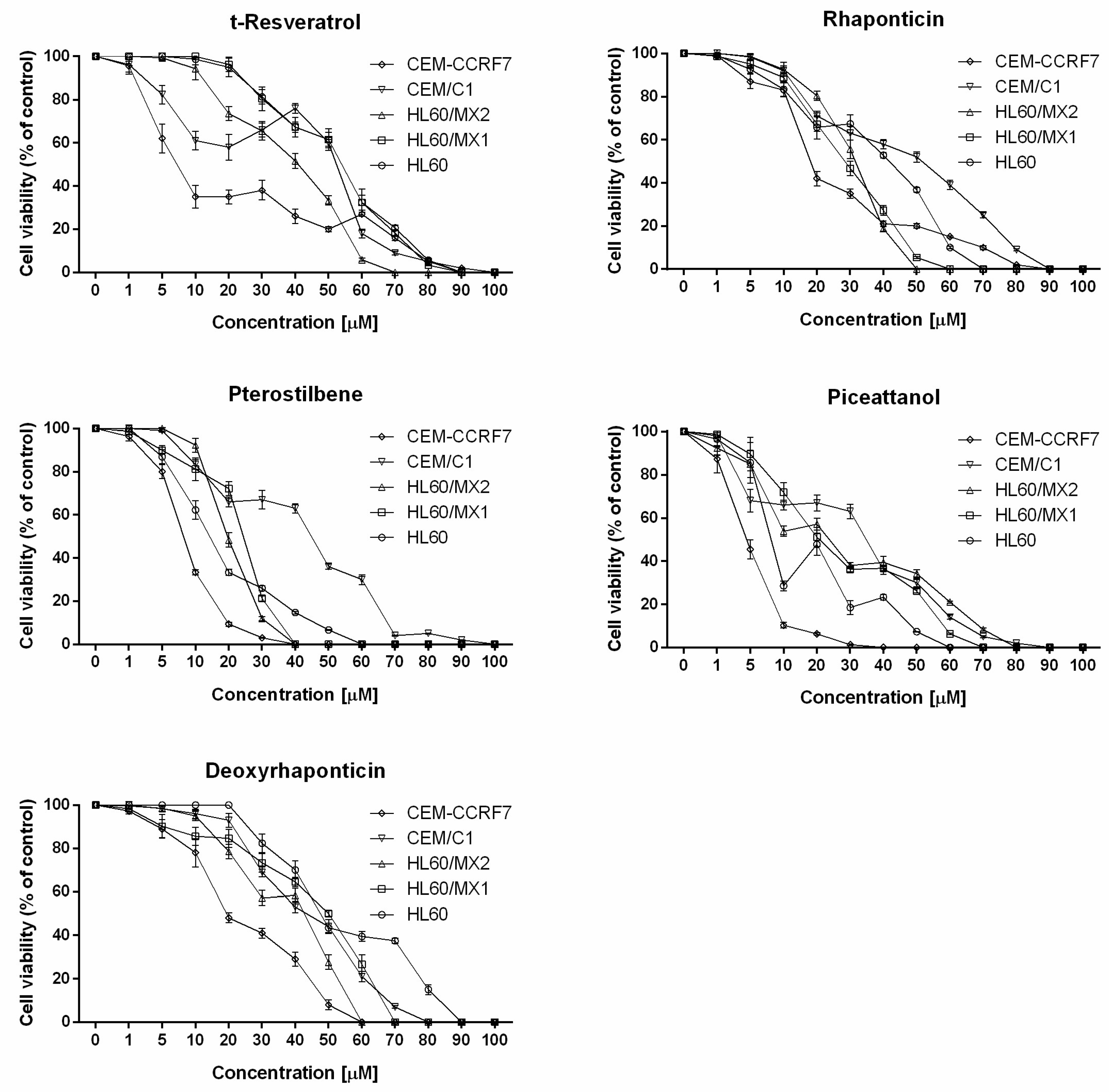
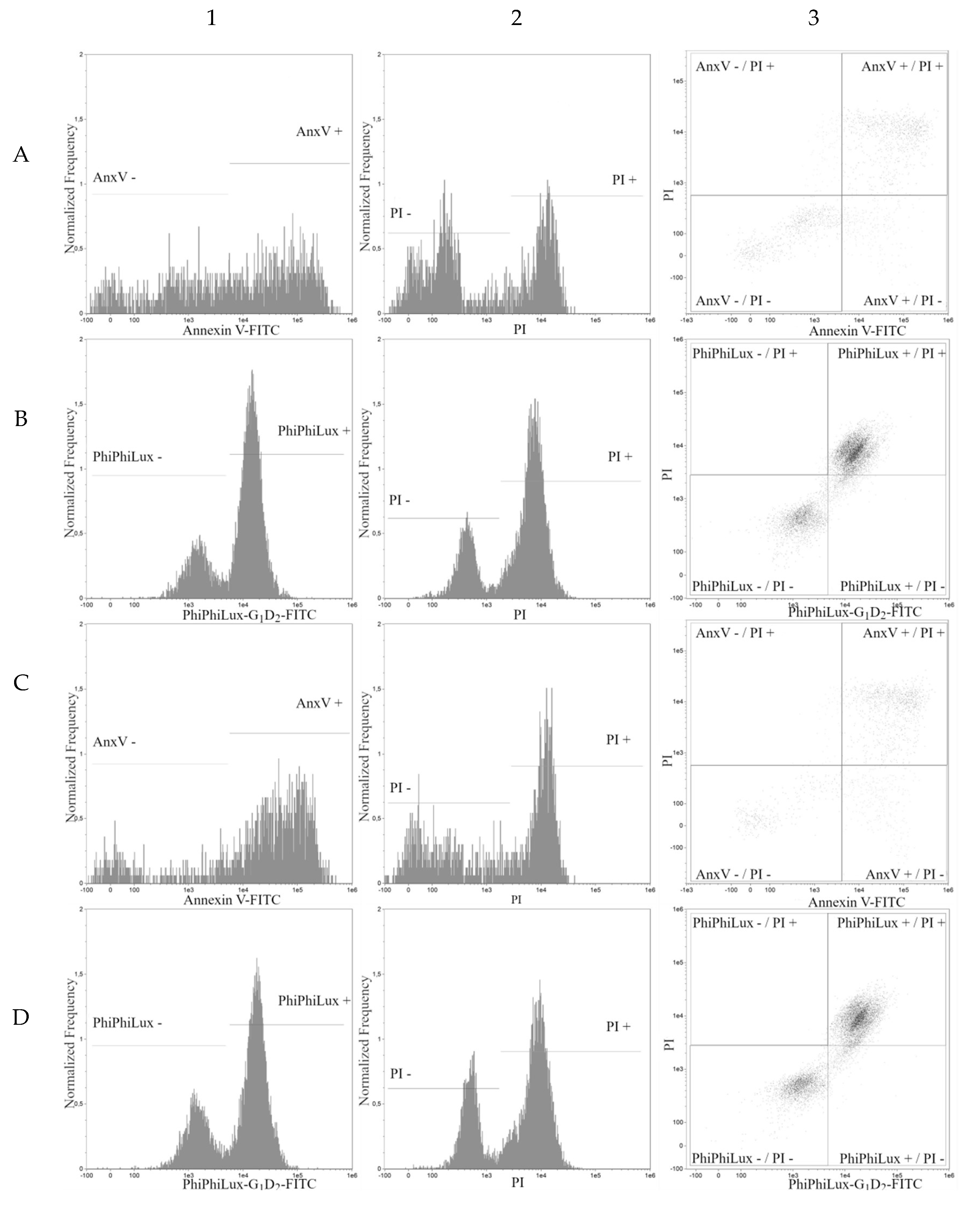
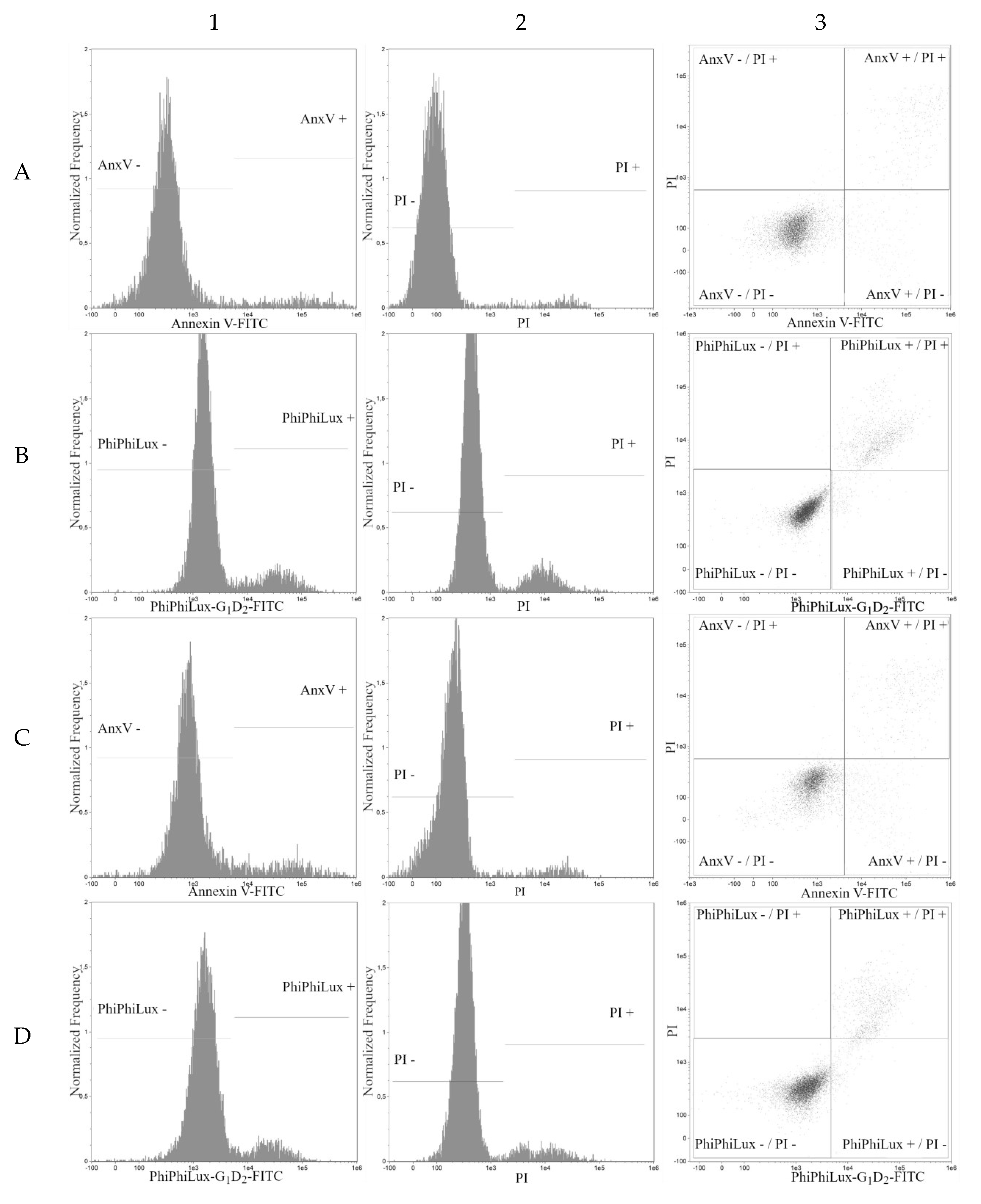
| Cell Line | t-Resveratrol (Mean ± SD) | Deoxyrhaponticin (Mean ± SD) | Pterostilbene (Mean ± SD) | Rhapontin (Mean ± SD) | Piceatannol (Mean ± SD) |
|---|---|---|---|---|---|
| HL60 | 54.09 ± 5.69 | 47.54 ± 3.90 | 14.25 ± 1.48 | 41.75 ± 2.33 | 8.12 ± 2.17 |
| HL60/MX1 | 53.95 ± 1.18 | 50.52 ± 2.46 | 24.35 ± 2.57 | 28.38 ± 1.26 | 20.76 ± 2.15 |
| HL60/MX2 | 40.88 ± 1.85 | 42.71 ± 2.60 | 19.62 ± 1.95 | 31.51 ± 2.51 | 23.73 ± 1.63 |
| CEM/C1 | 52.38 ± 3.28 | 43.33 ± 3.59 | 44.82 ± 2.75 | 51.54 ± 2.36 | 34.82 ± 3.87 |
| CCRF-CEM | 7.22 ± 1.78 | 19.33 ± 1.51 | 8.21 ± 0.72 | 18.05 ± 2.22 | 4.57 ± 0.12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Czop, M.; Bogucka-Kocka, A.; Kubrak, T.; Knap-Czop, K.; Makuch-Kocka, A.; Galkowski, D.; Wawer, J.; Kocki, T.; Kocki, J. Imaging Flow Cytometric Analysis of Stilbene-Dependent Apoptosis in Drug Resistant Human Leukemic Cell Lines. Molecules 2019, 24, 1896. https://doi.org/10.3390/molecules24101896
Czop M, Bogucka-Kocka A, Kubrak T, Knap-Czop K, Makuch-Kocka A, Galkowski D, Wawer J, Kocki T, Kocki J. Imaging Flow Cytometric Analysis of Stilbene-Dependent Apoptosis in Drug Resistant Human Leukemic Cell Lines. Molecules. 2019; 24(10):1896. https://doi.org/10.3390/molecules24101896
Chicago/Turabian StyleCzop, Marcin, Anna Bogucka-Kocka, Tomasz Kubrak, Karolina Knap-Czop, Anna Makuch-Kocka, Dariusz Galkowski, Joanna Wawer, Tomasz Kocki, and Janusz Kocki. 2019. "Imaging Flow Cytometric Analysis of Stilbene-Dependent Apoptosis in Drug Resistant Human Leukemic Cell Lines" Molecules 24, no. 10: 1896. https://doi.org/10.3390/molecules24101896
APA StyleCzop, M., Bogucka-Kocka, A., Kubrak, T., Knap-Czop, K., Makuch-Kocka, A., Galkowski, D., Wawer, J., Kocki, T., & Kocki, J. (2019). Imaging Flow Cytometric Analysis of Stilbene-Dependent Apoptosis in Drug Resistant Human Leukemic Cell Lines. Molecules, 24(10), 1896. https://doi.org/10.3390/molecules24101896