Biological Activities and Chemical Constituents of Essential Oils from Piper cubeba Bojer and Piper nigrum L.
Abstract
:1. Introduction
2. Results
2.1. Antioxidant and Antihyperuricemic Activities
2.2. Herbicidal Activity of P. cubeba and P. nigrum EOs against B. pilosa and E. crus-galli
2.3. Physiological and Biochemical Responses of B. pilosa and E. crus-galli to P. cubeba and P. nigrum EOs
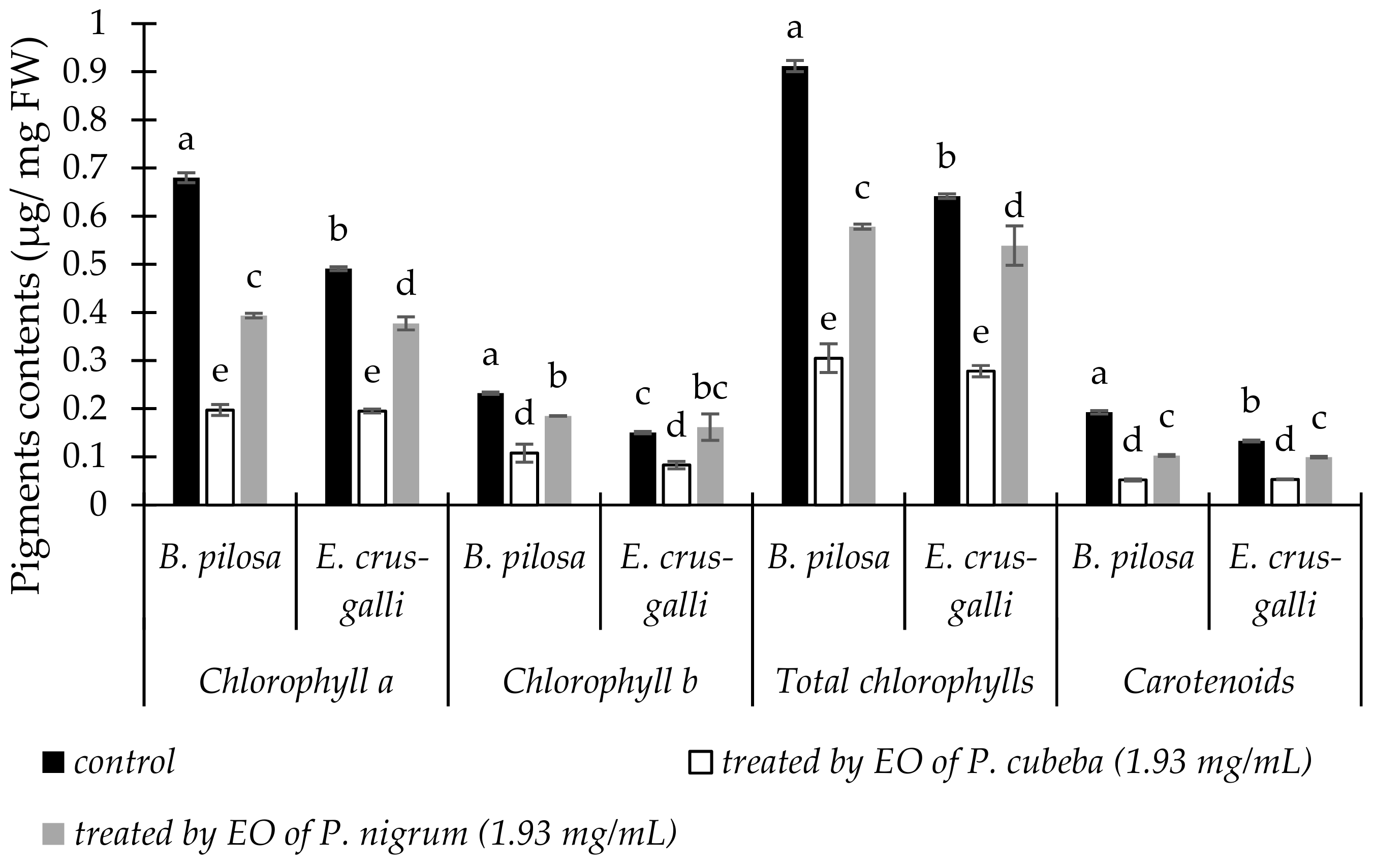
2.3.1. Pigments Contents
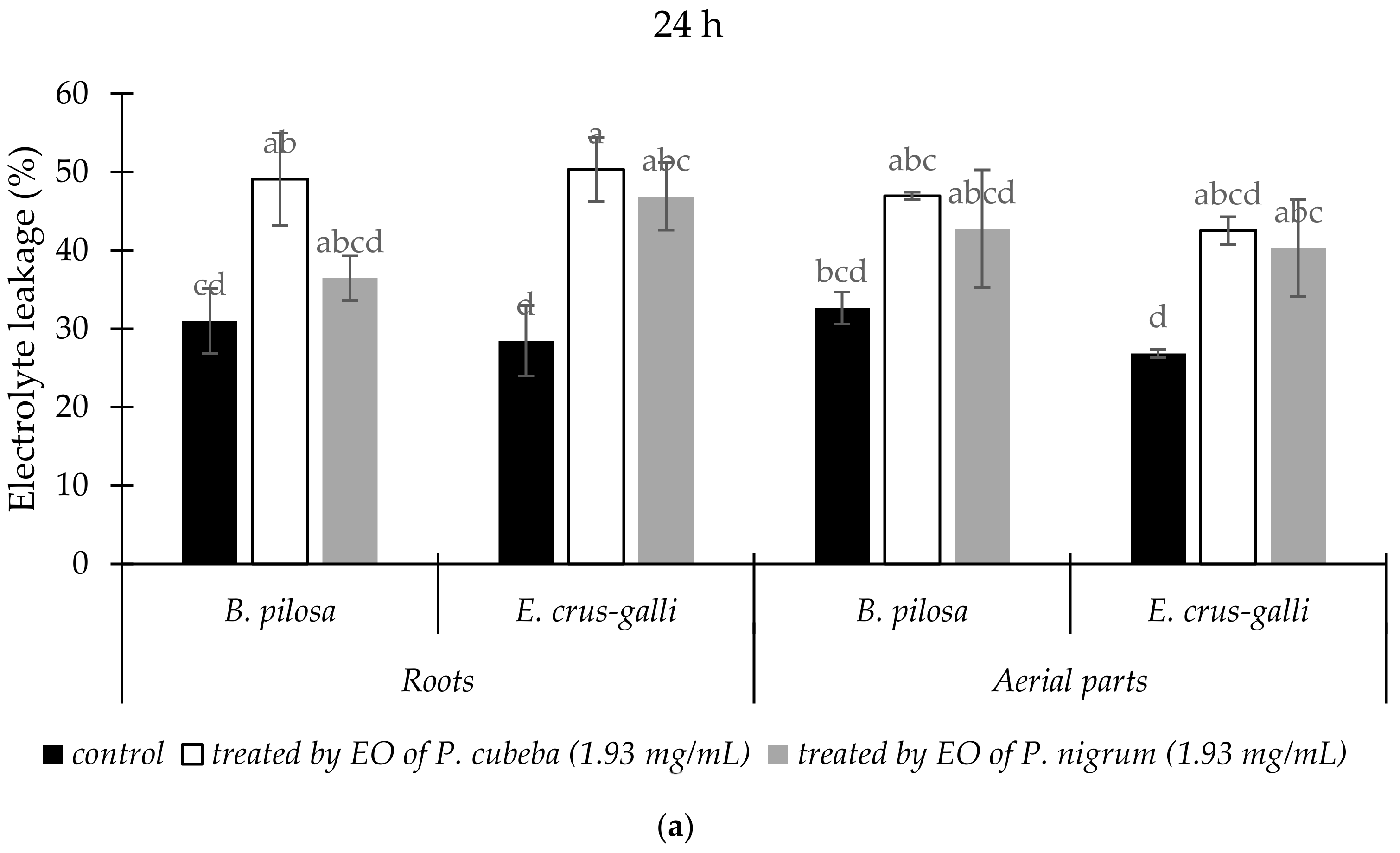
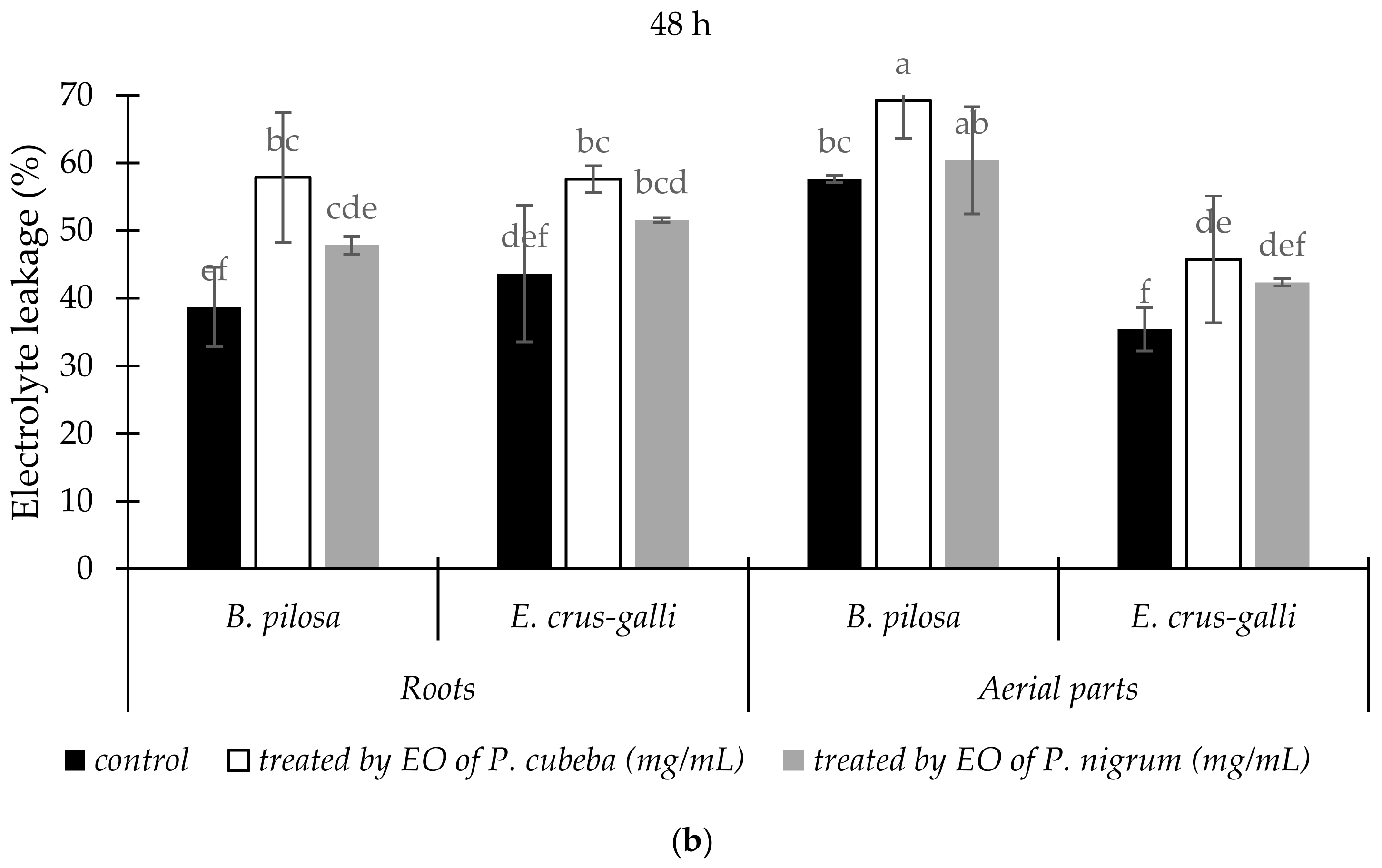
2.3.2. Electrolyte Leakage
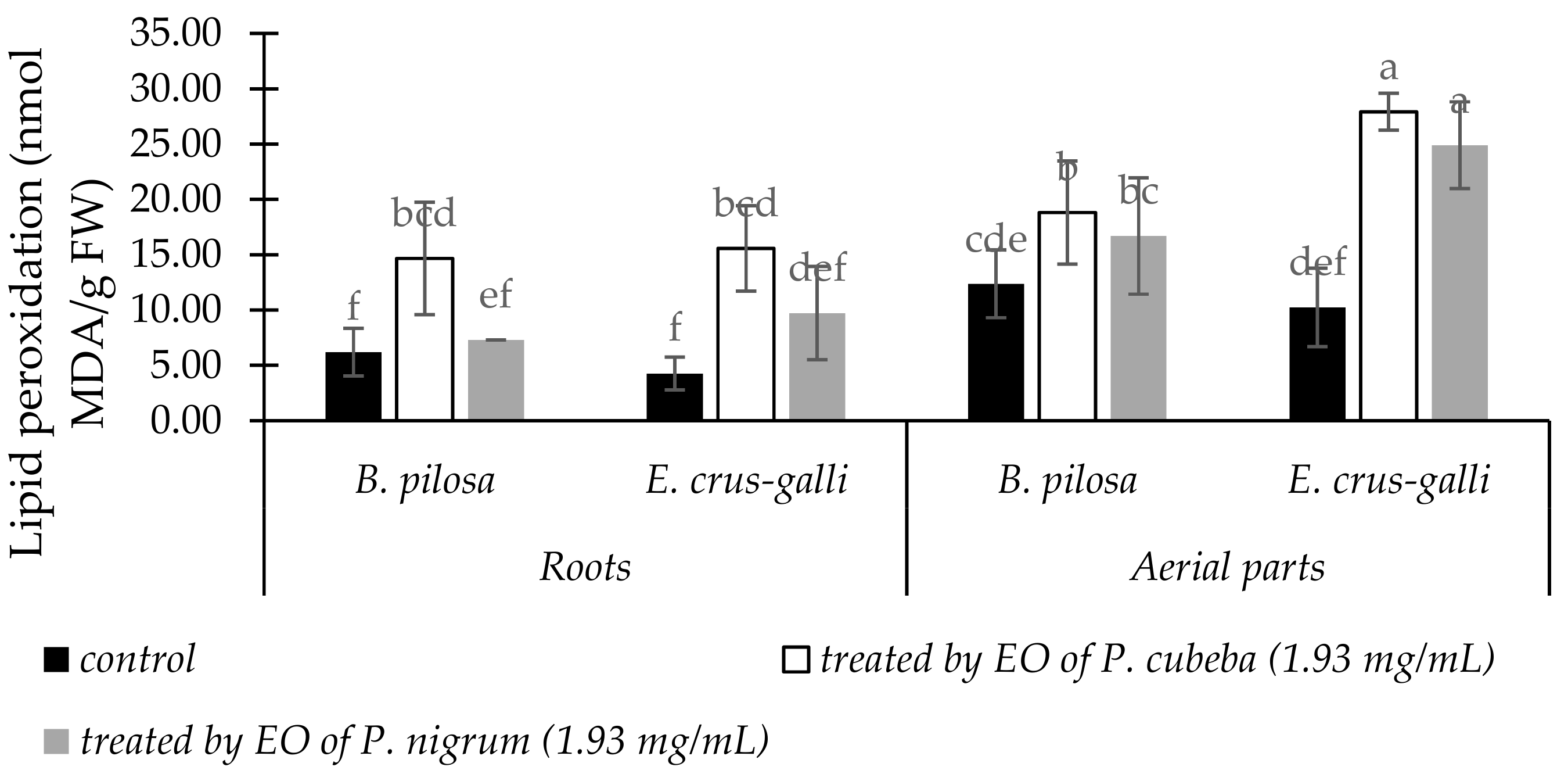
2.3.3. Lipid Peroxidation
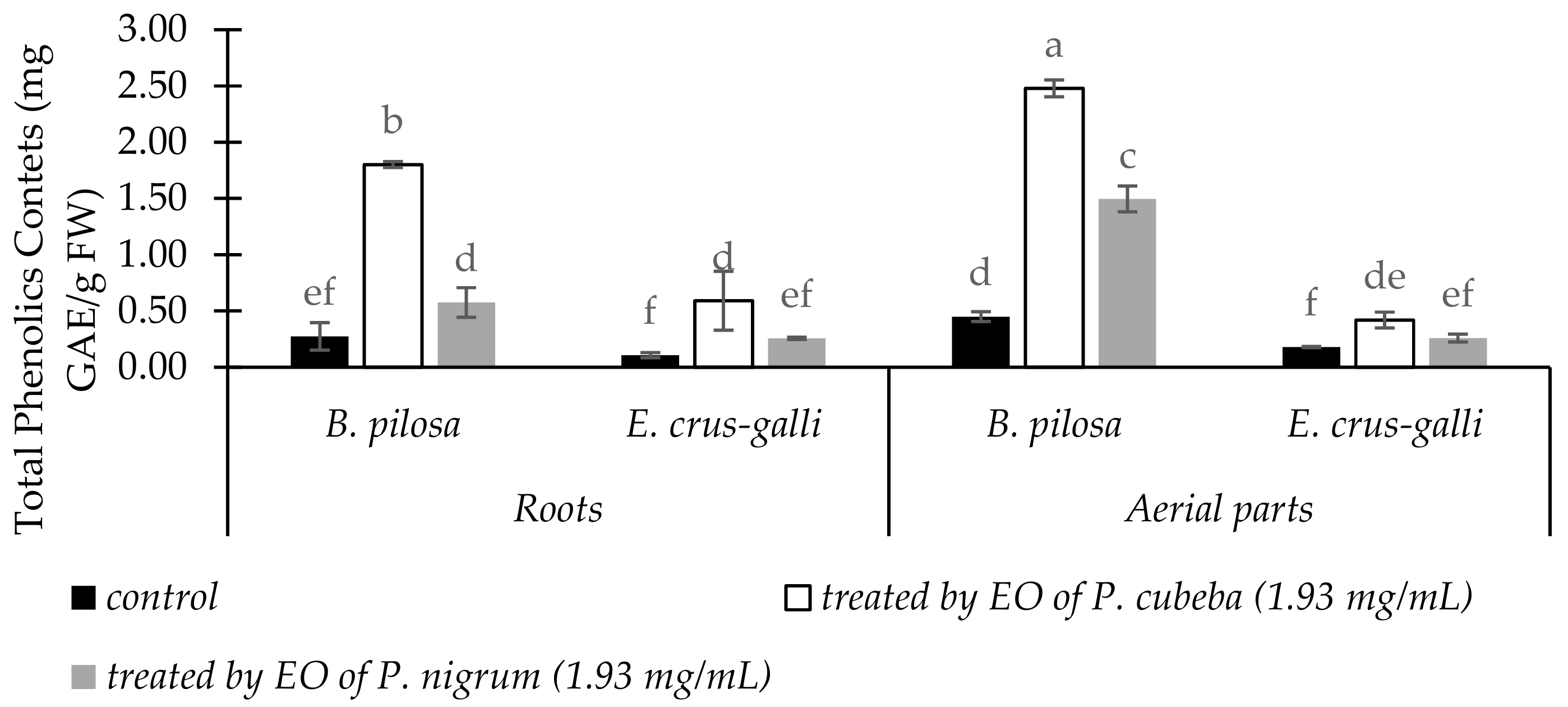
2.3.4. Total Phenolic Contents
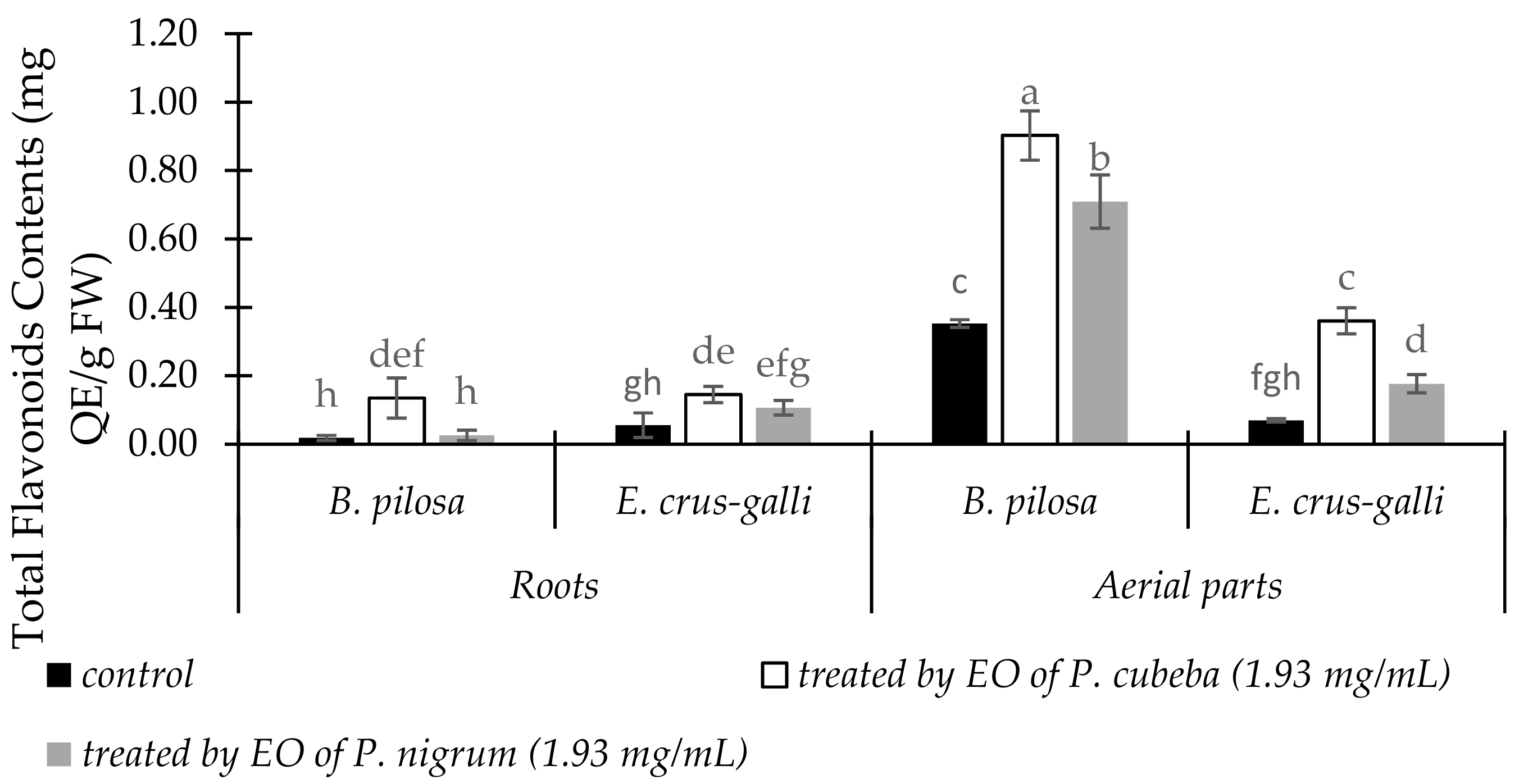
2.3.5. Total Flavonoid Contents
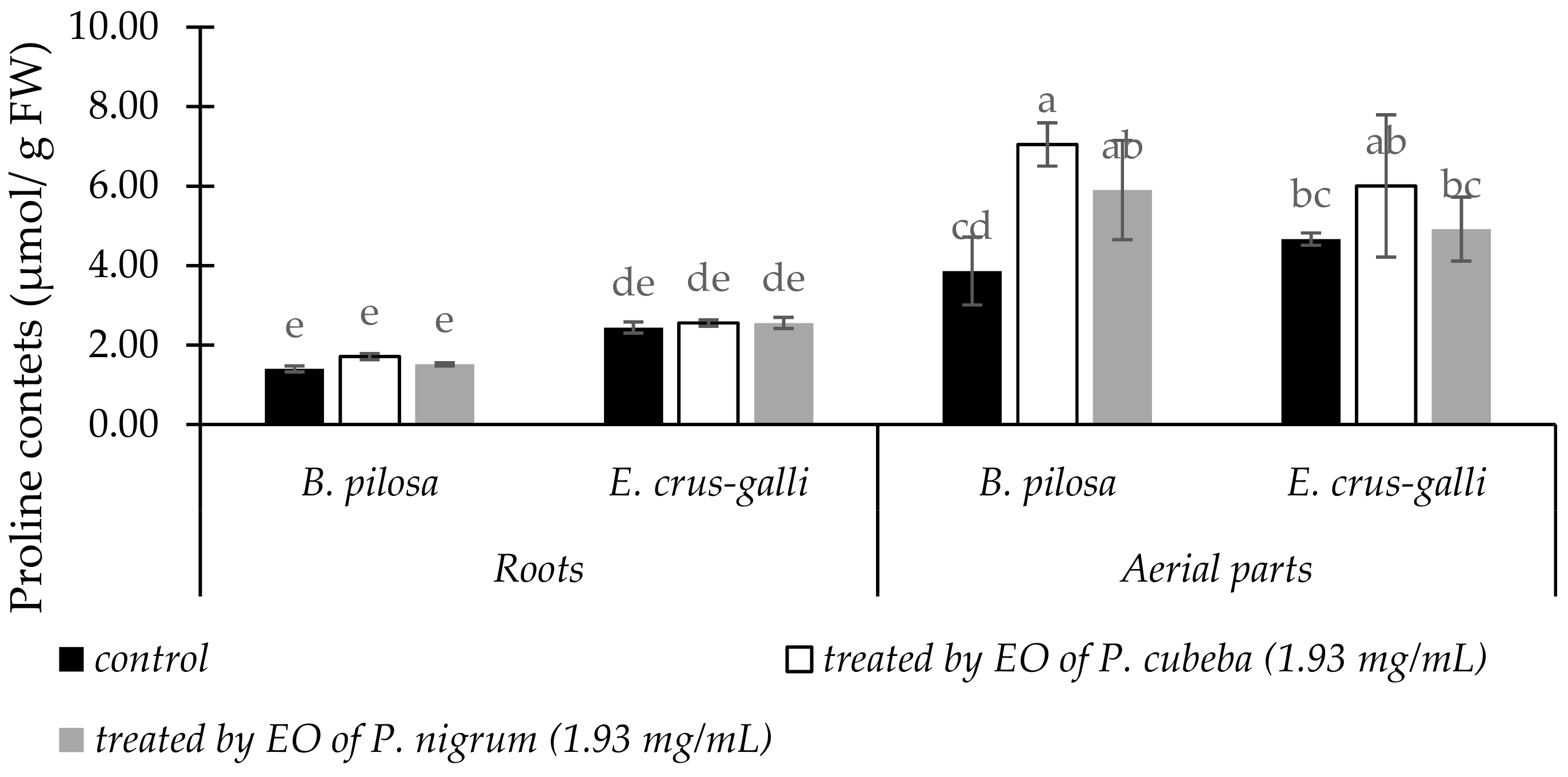
2.3.6. Proline Contents
2.3.7. Essential Oil Yields
2.3.8. Chemicals Composition of P. cubeba and P. nigrum Essential Oils
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Plant Materials and Seeds
4.3. Essential Oils Extraction
4.4. Antioxidant Assays
4.4.1. DPPH Radical Scavenging Assay
4.4.2. ABTS Radical Scavenging Assay
4.5. Xanthine Oxidase Inhibitory Assay
4.6. Herbicidal Assays
4.7. Physiological and Biochemical Responses
4.7.1. Chlorophyll and Carotenoid Contents
4.7.2. Electrolyte Leakage
4.7.3. Lipid Peroxidation
4.7.4. Proline Contents
4.7.5. Total Phenolic Contents
4.7.6. Total Flavonoid Contents
4.7.7. Identification of Chemical Constituents by Gas Chromatography-Mass Spectrometry (GC-MS)
4.7.8. Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (LC-ESI-MS) Analysis
4.7.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Laosinwattana, C.; Wichittrakarn, P.; Teerarak, M. Chemical composition and herbicidal action of essential oil from Tagetes erecta L. leaves. Ind. Crop. Prod. 2018, 126, 129–134. [Google Scholar] [CrossRef]
- Prakash, B.; Mishra, P.K.; Kedia, A.; Dubey, N.K. Antifungal, antiaflatoxin and antioxidant potential of chemically characterized Boswellia carterii Birdw essential oil and its in vivo practical applicability in preservation of Piper nigrum L. fruits. LWT Food Sci. Technol. 2014, 56, 240–247. [Google Scholar] [CrossRef]
- Abd El-Gawad, A.M. Chemical constituents, antioxidant and potential allelopathic effect of the essential oil from the aerial parts of Cullen plicata. Ind. Crops Prod. 2016, 80, 36–41. [Google Scholar] [CrossRef]
- Tu, P.T.B.; Tawata, S. Anti-oxidant, anti-aging, and anti-melanogenic properties of the essential oils from two varieties of Alpinia zerumbet. Molecules 2015, 20, 16723–16740. [Google Scholar] [CrossRef]
- Hazrati, H.; Saharkhiz, M.J.; Niakousari, M.; Moein, M. Natural herbicide activity of Satureja hortensis L. essential oil nanoemulsion on the seed germination and morphophysiological features of two important weed species. Ecotoxicol. Environ. Saf. 2017, 142, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Fagodia, S.K.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Phytotoxicity and cytotoxicity of Citrus aurantiifolia essential oil and its major constituents: Limonene and citral. Ind. Crops Prod. 2017, 108, 708–715. [Google Scholar] [CrossRef]
- Ben Ghnaya, A.; Amri, I.; Hanana, M.; Gargouri, S.; Jamoussi, B.; Romane, A.; Hamrouni, L. Tetraclinis articulata (Vahl.) Masters essential oil from Tunisia: Chemical characterization and herbicidal and antifungal activities assessment. Ind. Crops Prod. 2016, 83, 113–117. [Google Scholar] [CrossRef]
- Mesquita, J.M.O.; Oliveira, A.B.; Braga, F.C.; Lombardi, J.A.; da Cunha, A.P.; Salgueiro, L.; Cavaleiro, C. Essential oil constituents of Piper vicosanum Yunker from the Brazilia Atlantic Forest. J. Essent. Oil Res. 2006, 18, 392–395. [Google Scholar] [CrossRef]
- Hoff Brait, D.R.; Mattos Vaz, M.S.; da Silva Arrigo, J.; Borges de Carvalho, L.N.; Souza de Araújo, F.H.; Vani, J.M.; da Silva Mota, J.; Cardoso, C.A.L.; Oliveira, R.J.; Negrão, F.J.; et al. Toxicological analysis and anti-inflammatory effects of essential oil from Piper vicosanum leaves. Regul. Toxicol. Pharmacol. 2015, 73, 699–705. [Google Scholar] [CrossRef]
- De Morais, S.M.; Facundo, V.A.; Bertini, L.M.; Cavalcanti, E.S.B.; dos Anjos, J.F., Jr.; Ferreira, S.A.; de Brito, E.S.; de Souza Neto, M.A. Chemical composition and larvicidal activity of essential oils from Piper species. Biochem. Syst. Ecol. 2007, 35, 670–675. [Google Scholar] [CrossRef]
- Bosquiroli, L.S.S.; Demarque, D.P.; Rizk, Y.S.; Cunha, M.C.; Marques, M.C.S.; De Matos, M.F.C.; Kadri, M.C.T.; Carollo, C.A.; Arruda, C.C.P. In vitro anti-leishmania infantum activity of essential oil from Piper angustifolium. Braz. J. Pharmacogn. 2015, 25, 124–128. [Google Scholar] [CrossRef]
- Nahak, G.; Sahu, R.K. Phytochemical evaluation and antioxidant activity of Piper cubeba and Piper nigrum. J. Appl. Pharm. Sci. 2011, 01, 153–157. [Google Scholar]
- Elfahmi, K.R.; Batterman, S.; Bos, R.; Kayser, O.; Woerdenbag, H.J.; Quax, W.J. Lignan profile of Piper cubeba, an Indonesian medicinal plant. Biochem. Syst. Ecol. 2007, 35, 397–402. [Google Scholar] [CrossRef]
- Magalhães, L.G.; de Souza, J.M.; Wakabayashi, K.A.L. In vitro efficacy of the essential oil of Piper cubeba L. (Piperaceae) against Schistosoma mansoni. Parasitol. Res. 2012, 110, 1747–1754. [Google Scholar] [CrossRef]
- Mothana, R.; Alsaid, M.; Khaled, J.M.; Alharbi, N.S.; Alatar, A.; Raish, M.; Al-Yahya, M.; Rafatullah, S.; Parvez, M.K.; Ahamad, S.R. Assessment of antinociceptive, antipyretic and antimicrobial activity of Piper cubeba L. essential oil in animal models. Pak. J. Pharm. Sci. 2016, 29, 671–677. [Google Scholar] [PubMed]
- Chaubey, M.K. Insecticidal properties of Zingiber officinale and Piper cubeba essential oils against Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). J. Biol. Act. Prod. Nat. 2011, 1, 306–313. [Google Scholar]
- Bos, R.; Woerdenbag, H.J.; Kayser, O.; Quax, W.J.; Ruslan, K.; Elfami. Essential oil constituents of Piper cubeba L. fils. from Indonesia. J. Essent. Oil Res. 2007, 19, 14–17. [Google Scholar] [CrossRef]
- Vinturelle, R.; Mattos, C.; Meloni, J.; Nogueira, J.; Nunes, M.J.; Jr, I.S.V.; Rocha, L.; Lione, V.; Castro, H.C.; das Chagas, E.F. In vitro evaluation of essential oils derived from Piper nigrum (Piperaceae) and Citrus limonum (Rutaceae) against the tick Rhipicephalus (Boophilus) microplus (Acari: Ixodidae). Biochem. Res. Int. 2017, 1–9. [Google Scholar] [CrossRef]
- Upadhyay, R.K.; Jaiswal, G. Evaluation of biological activities of Piper nigrum oil against Tribolium castaneum. Bull. Insectol. 2007, 60, 57–61. [Google Scholar]
- Amer, A.; Mehlhorn, H. Larvicidal effects of various essential oils against Aedes, Anopheles, and Culex larvae (Diptera, Culicidae). Parasitol. Res. 2006, 99, 466–472. [Google Scholar] [CrossRef]
- Morsy, N.F.S.; Abd El-Salam, E.A. Antimicrobial and antiproliferative activities of black pepper (Piper nigrum L.) essential oil and oleoresin. J. Essent. Oil Bear. Plants 2017, 20, 779–790. [Google Scholar] [CrossRef]
- Morshed, S.; Hossain, M.D.; Ahmad, M.; Junayed, M. Physicochemical characteristics of essential oil of black pepper (Piper nigrum) cultivated in Chittagong, Bangladesh. J. Food Qual. Hazards Control 2017, 4, 66–69. [Google Scholar]
- Hari, R.; Vasuki, R.; Prasoon, G.P.; Singh, H.P.; Mishra, N. Evaluation of in-vitro antioxidant and xanthine oxidase inhibitory activity of selected Indian plants. Int. J. Biotech. Trend Tech. 2012, 2, 1–9. [Google Scholar]
- Gawlik-Dziki, U. Dietary spices as a natural effectors of lipoxygenase, xanthine oxidase, peroxidase and antioxidant agents. LWT Food Sci. Technol. 2012, 47, 138–146. [Google Scholar] [CrossRef]
- Sabina, E.P.; Nagar, S.; Rasool, M. A role of piperine on monosodium urate crystal-induced inflammation—an experimental model of gouty arthritis. Inflammation 2011, 34, 184–192. [Google Scholar] [CrossRef] [PubMed]
- William, E.A., Jr.; James, S.C. Phase transition enthalpy measurements of organic and organometallic compounds. In NIST Chemistry WebBook, NIST Standard Reference Database Number 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2018. [Google Scholar]
- Bagheri, H.; Abdul Manap, M.Y.; Abdul Manap, M.Y.B.A.; Solati, Z. Antioxidant activity of Piper nigrum L. essential oil extracted by supercritical CO2 extraction and hydro-distillation. Talanta 2014, 121, 220–228. [Google Scholar]
- Zaka, M.S.; Iqbal, N.; Saeed, Q.; Akrem, A.; Batool, M.; Khan, A.A.; Anwar, A.; Bibi, M.; Azeem, S.; Rizvi, D.; et al. Toxic effects of some insecticides, herbicides, and plant essential oils against Tribolium confusum Jacquelin du val (Insecta: Coleoptera: Tenebrionidae). Saudi J. Biol. Sci 2018, in press. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Xuan, T.D.; Koyama, H.; Tawata, S. Antioxidant activity and contents of essential oil and phenolic compounds in flowers and seeds of Alpinia zerumbet (Pers.) B.L. Burtt. & R.M. Sm. Food Chem. 2007, 104, 1648–1653. [Google Scholar]
- Minh, T.N.; Xuan, T.D.; Van, T.M.; Andriana, Y.; Viet, T.D.; Khanh, T.D.; Tran, H.-D. Phytochemical analysis and potential biological activities of essential oil from rice leaf. Molecules 2019, 24, 889. [Google Scholar] [CrossRef] [PubMed]
- García, P.; Ramallo, I.A.; Salazar, M.O.; Furlan, R.L.E. Chemical diversification of essential oils, evaluation of complex mixtures and identification of a xanthine oxidase inhibitor. RSC Adv. 2016, 6, 57245–57252. [Google Scholar] [CrossRef]
- Yan, G.; Zhu, C.; Luo, Y.; Yang, Y.; Wei, J. Potential allelopathic effects of Piper nigrum, Mangifera indica and Clausena lansium. J. App. Ecol. 2006, 17, 1633–1636. (in Chinese). [Google Scholar]
- Sheng-li, Z.H.A.N. Preliminary study on the allelopathy of Piper nigrum L. Available online: http://en.cnki.com.cn/Article_en/CJFDTotal-AHNY200903016.htm (accessed on 12 May 2019).
- Siddiqui, Z.S. Allelopathic effects of black pepper leachings on Vigna mungo (L.) Hepper. Acta Physiol. Plant. 2007, 29, 303–308. [Google Scholar] [CrossRef]
- Jalaei, Z.; Fattahi, M.; Aramideh, S. Allelopathic and insecticidal activities of essential oil of Dracocephalum kotschyi Boiss. from Iran: A new chemotype with highest limonene-10-al and limonene. Ind. Crops Prod. 2015, 73, 109–117. [Google Scholar] [CrossRef]
- López, M.L.; Bonzani, N.E.; Zygadlo, J.A. Allelopathic potential of Tagetes minuta terpenes by a chemical, anatomical and phytotoxic approach. Biochem. Syst. Ecol. 2008, 36, 882–890. [Google Scholar] [CrossRef]
- Dias, J.F.G.; Miguel, O.G.; Miguel, M.D. Composition of essential oil and allelopathic activity of aromatic water of Aster lanceolatus Willd: (Asteraceae). Braz. J. Pharm. Sci. 2009, 45, 469–474. [Google Scholar] [CrossRef]
- Almarie, A.A.; Mamat, A.S.; Wahab, Z.; Rukunudin, I.H. Chemical composition and phytotoxicity of essential oils isolated from Malaysian plants. Allelopath. J. 2016, 37, 55–70. [Google Scholar]
- Andriana, Y.; Xuan, T.D.; Quan, N.V.; Quy, T.N. Allelopathic potential of Tridax procumbens L. on radish and identification of allelochemicals. Allelopath. J. 2018, 43, 223–238. [Google Scholar] [CrossRef]
- Jaballah, S.B.; Zribi, I.; Haouala, R. Physiological and biochemical responses of two lentil varieties to chickpea (Cicer arietinum L.) aqueous extracts. Sci. Hortic. 2017, 225, 74–80. [Google Scholar] [CrossRef]
- Kaur, S.; Singh, H.P.; Mittal, S.; Batish, D.R.; Kohli, R.K. Phytotoxic effects of volatile oil from Artemisia scoparia against weeds and its possible use as a bioherbicide. Ind. Crops Prod. 2010, 32, 54–61. [Google Scholar] [CrossRef]
- Mutlu, S.; Atici, Ö.; Esim, N.; Mete, E. Essential oils of catmint (Nepeta meyeri Benth.) induce oxidative stress in early seedlings of various weed species. Acta Physiol. Plant. 2011, 33, 943–951. [Google Scholar] [CrossRef]
- Ladhari, A.; Omezzine, F.; Haouala, R. The impact of Tunisian Capparidaceae species on cytological, physiological and biochemical mechanisms in lettuce. South Afr. J. Bot. 2014, 93, 222–230. [Google Scholar] [CrossRef]
- Zhang, D.-Y.; Yao, X.-H.; Duan, M.-H.; Wei, F.-Y.; Wu, G.-H.; Li, L. Variation of essential oil content and antioxidant activity of Lonicera species in different sites of China. Ind. Crops Prod. 2015, 77, 772–779. [Google Scholar] [CrossRef]
- Quan, N.V.; Dang, X.T.; Tran, H.-D.; Thuy, N.T.D.; Trang, L.T.; Huong, C.T.; Andriana, Y.; Tuyen, P.T. Antioxidant, α-amylase and α-glucosidase inhibitory activities and potential constituents of Canarium tramdenum Bark. Molecules 2019, 24, 482. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.T.T.; Awale, S.; Tezuka, Y.; Le Tran, Q.; Watanabe, H.; Kadota, S. Xanthine oxidase inhibitory activity of Vietnamese medicinal plants. Biol. Pharm. Bull. 2004, 27, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Andriana, Y.; Xuan, T.D.; Quy, T.N.; Ngoc, M.T.; Van, T.M.; Viet, T.D. Antihyperuricemia, antioxidant, and antibacterial activities of Tridax procumbens L. Foods 2019, 8, 21. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.; Wellburn, A. Determinations of total carotenoids and chlorophylls b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Physiological and biochemical mechanisms of allelochemicals in aqueous extracts of diploid and mixoploid Trigonella foenum-graecum L. South African J. Bot. 2014, 93, 167–178. [CrossRef]
- Lutts, S.; Kinet, J.M.; Bouharmont, J. NaCl-induced senescence in leaves of rice (Oryza sativa L.) cultivars differing in salinity resistance. Ann. Bot. 1996, 78, 389–398. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Gopi, R.; Sankar, B.; Manivannan, P.; Kishorekumar, A.; Sridharan, R.; Panneerselvam, R. Studies on germination, seedling vigour, lipid peroxidation and proline metabolism in Catharanthus roseus seedlings under salt stress. South Afr. J. Bot. 2007, 73, 190–195. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Tawata, S. Antioxidant capacity and phenolic content of Rumex dentatus L. grown in Egypt. J. Crop Sci. Biotechnol. 2012, 15, 59–64. [Google Scholar] [CrossRef]
- Banerjee, S.; Mazumdar, S. Electrospray ionization mass spectrometry: A technique to access the information beyond the molecular weight of the analyte. Int. J. Anal. Chem. 2012, 2012, 1–40. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of P. cubeba and P. nigrum EOs are available from the authors. |
| Samples | Antioxidant Activity (IC50 (mg/mL)) | XOI Activity (IC50 (µg/mL)) | |
|---|---|---|---|
| DPPH | ABTS | ||
| P. cubeba EO | 0.82 ± 0.06 b | 1.32 ± 0.04 b | 54.87 ± 1.69 a b |
| P. nigrum EO | 1.15 ± 0.08 a | 1.74 ± 0.03 a | 77.11 ± 2.11 a |
| BHT * | 0.009 ± 0.02 c | 0.071 ± 0.001 c | - |
| Allopurinol ** | - | - | 20.45 ± 0.3 b |
| Treatments | Indicator Plants | IC50 (mg/mL) | ||
|---|---|---|---|---|
| Germination | Roots | Shoots | ||
| P. cubeba EOs | B. pilosa | 5.19 ± 0.22 a | 2.63 ± 0.58 a | 1.93 ± 0.68 b |
| E. crus-galli | 5.26 ± 0.34 a | 3.30 ± 0.97 a | 5.86 ± 0.94 a b | |
| P. nigrum EOs | B. pilosa | 5.44 ± 0.00 a | 2.67 ± 0.64 a | 3.1 ± 0.56 a b |
| E. crus-galli | > 8.00 | 2.71 ± 0.11 a | 7.21 ± 2.02 a | |
| Samples | Dry Weight (g) | EOs (g) | Yields (% w/w) (% w/w DW) |
|---|---|---|---|
| P. cubeba | 300.00 | 3.69 ± 0.05 a | 1.23 ± 0.01 a |
| P. nigrum | 300.00 | 3.35 ± 0.01 b | 1.11 ± 0.01 b |
| No. | Compounds | Rt (min) | Chemical Formula | MW (g/mol) | Chemical Class | Area (%) | RI * | |
|---|---|---|---|---|---|---|---|---|
| P. cubeba EO | P. nigrum EO | |||||||
| 1 | β-Thujene | 5.32 | C10H16 | 136.238 | Monoterpenes | - | 20.58 | 929 |
| 2 | Terpinen-4-ol | 7.81 | C10H18O | 154.253 | Monoterpenes | 42.41 | 1.85 | 1179 |
| 3 | trans-Chrysanthenyl acetate | 8.38 | C12H18O2 | 194.274 | Monoterpenes | 0.1 | - | 1228 |
| 4 | δ-EIemene | 9.96 | C15H24 | 204.357 | Monoterpenes | 0.58 | 5.03 | 1338 |
| 5 | α-Cubebene | 10.13 | C15H24 | 204.357 | Sesquiterpenes | 6.54 | - | 1349 |
| 6 | α-Copaene | 10.55 | C15H24 | 204.357 | Sesquiterpenes | 20.04 | 4.79 | 1377 |
| 7 | D-Germacrene | 10.69 | C15H24 | 204.357 | Sesquiterpenes | 2.50 | - | 1451 |
| 8 | γ-Elemene | 11.21 | C15H24 | 204.357 | Sesquiterpenes | 17.68 | - | 1455 |
| 9 | β-Caryophyllene | 11.23 | C15H24 | 204.357 | Sesquiterpenes | - | 51.12 | 1467 |
| 10 | Humulene | 11.60 | C15H24 | 204.357 | Sesquiterpenes | - | 3.81 | 1474 |
| 11 | β-Selinene | 12.03 | C15H24 | 204.357 | Sesquiterpenes | - | 5.59 | 1490 |
| 12 | δ-Cadinene | 12.33 | C15H24 | 204.357 | Sesquiterpenes | 2.7 | 2.04 | 1525 |
| 13 | α-Elemol | 12.68 | C15H26O | 222.372 | Sesquiterpenes | 1.78 | - | 1551 |
| 14 | Spathulenol | 13.07 | C15H24O | 220.356 | Sesquiterpenes | 0.18 | - | 1581 |
| 15 | Caryophyllene oxide | 13.16 | C15H24O | 220.356 | Sesquiterpenes | - | 1.51 | 1595 |
| 16 | Cubenol | 13.65 | C15H26O | 222.372 | Sesquiterpenes | 0.44 | 0.97 | 1642 |
| 17 | β-Eudesmol | 14.00 | C15H26O | 222.372 | Sesquiterpenes | 0.64 | - | 1655 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andriana, Y.; Xuan, T.D.; Quy, T.N.; Tran, H.-D.; Le, Q.-T. Biological Activities and Chemical Constituents of Essential Oils from Piper cubeba Bojer and Piper nigrum L. Molecules 2019, 24, 1876. https://doi.org/10.3390/molecules24101876
Andriana Y, Xuan TD, Quy TN, Tran H-D, Le Q-T. Biological Activities and Chemical Constituents of Essential Oils from Piper cubeba Bojer and Piper nigrum L. Molecules. 2019; 24(10):1876. https://doi.org/10.3390/molecules24101876
Chicago/Turabian StyleAndriana, Yusuf, Tran Dang Xuan, Tran Ngoc Quy, Hoang-Dung Tran, and Quang-Tri Le. 2019. "Biological Activities and Chemical Constituents of Essential Oils from Piper cubeba Bojer and Piper nigrum L." Molecules 24, no. 10: 1876. https://doi.org/10.3390/molecules24101876
APA StyleAndriana, Y., Xuan, T. D., Quy, T. N., Tran, H.-D., & Le, Q.-T. (2019). Biological Activities and Chemical Constituents of Essential Oils from Piper cubeba Bojer and Piper nigrum L. Molecules, 24(10), 1876. https://doi.org/10.3390/molecules24101876








