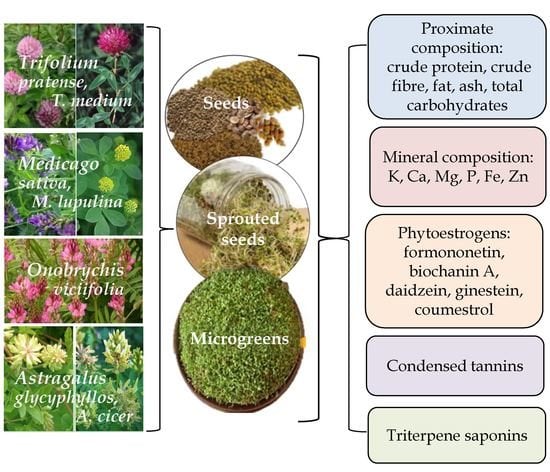
Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens
Abstract
1. Introduction
2. Results and Discussion
2.1. Proximate Composition
2.2. Mineral Profile
2.3. Bioactive Compounds
3. Materials and Methods
3.1. Materials
3.2. Seed Sprouting
3.3. Growing of Microgreens
3.4. Sample Preparation
3.5. Proximate Analysis
3.6. Determination of Minerals
3.7. Secondary Metabolite Analyses
3.7.1. Reagents
3.7.2. Reference Standard Solutions
3.7.3. Hydrolysis and Extraction of Phytoestrogens
3.7.4. Extraction of Condensed Tannins
3.7.5. Hydrolysis and Extraction of Triterpene Saponins
3.7.6. Quantification of Phytoestrogens
3.7.7. Quantification of Condensed Tannins
3.7.8. Quantification of Total Triterpene Saponins
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Muzquiz, M.; Varela, A.; Burbano, C.; Cuadrado, C.; Guillamón, E.; Pedrosa, M.M. Bioactive compounds in legumes: Pronutritive and antinutritive actions. Implications for nutrition and health. Phytochem. Rev. 2012, 11, 227–244. [Google Scholar] [CrossRef]
- Shahidi, F. Functional foods: Their role in health promotion and disease prevention. J. Food Sci. 2004, 69, R146–R149. [Google Scholar] [CrossRef]
- International Legume Database & Information Service World Database of Legumes. University of Reading, Reading. Available online: http://www.ildis.org/ (accessed on 26 September 2017).
- Bouchenak, M.; Lamri-Senhadji, M. Nutritional quality of legumes, and their role in cardiometabolic risk prevention: A review. J. Med. Food. 2013, 16, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Weide, A.; Riehl, S.; Zeidi, M.; Conard, N.J. Reconstructing subsistence practices: Taphonomic constraints and the interpretation of wild plant remains at aceramic Neolithic Chogha Golan, Iran. Veg. Hist. Archaeobot. 2017, 26, 487–504. [Google Scholar] [CrossRef]
- Butler, A. The small-seeded legumes: An enigmatic prehistoric resource. Acta Palaeobot. 1995, 1, 105–115. [Google Scholar]
- Dini, C.; García, M.A.; Vina, S.Z. Non-traditional flours: Frontiers between ancestral heritage and innovation. Food Funct. 2012, 3, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Bora, K.S.; Sharma, A. Phytochemical and pharmacological potential of Medicago sativa: A review. Pharm. Biol. 2011, 49, 211–220. [Google Scholar] [CrossRef]
- Łuczaj, Ł. Ethnobotanical review of wild edible plants of Slovakia. Acta Soc. Bot. Pol. 2012, 81, 245–255. [Google Scholar] [CrossRef]
- Ionkova, I. Anticancer compounds from in vitro cultures of rare medicinal plants. Pharmacogn. Rev. 2008, 2, 206–218. [Google Scholar]
- Adams, F. The Seven Books of Paulus Aegineta. Translated from Greek. With a Commentary Embracing a Complete View of the Knowledge Possessed by the Greeks, Romans, and Arabians on all Subjects Connected with Medicine and Surgery; book VII; Syndenham Society: London, UK, 1847; Volume 3, p. 278. Available online: https://archive.org/details/sevenbooksofpaul03pauluoft. (accessed on 30 October 2018).
- Malisch, C.S.; Luscher, A.; Baert, N.; Engstrom, M.T.; Studer, B.; Fryganas, C.; Suter, D.; Mueller-Harvey, I.; Salminen, J.P. Large variability of proanthocyanidin content and composition in sainfoin (Onobrychis viciifolia). J. Agric. Food Chem. 2015, 63, 10234–10242. [Google Scholar] [CrossRef]
- Saviranta, N.M.M.; Anttonen, M.J.; von Wright, A.; Karjalainen, R.O. Red clover (Trifolium pratense L.) isoflavones: Determination of concentrations by plant stage, flower colour, plant part and cultivar. J. Sci. Food Agric. 2008, 88, 125–132. [Google Scholar] [CrossRef]
- Butkutė, B.; Dagilytė, A.; Benetis, R.; Padarauskas, A.; Cesevičienė, J.; Olšauskaitė, V.; Lemežienė, N. Mineral and phytochemical profiles and antioxidant activity of herbal material from two temperate Astragalus species. BioMed Res. Int. 2018, 6318630. [Google Scholar] [CrossRef] [PubMed]
- Butkutė, B.; Lemežienė, N.; Padarauskas, A.; Norkevičienė, E.; Taujenis, L. Chemical composition of zigzag clover (Trifolium medium L.). In Breeding Grasses and Protein Crops in the Era of Genomics; Brazauskas, G., Statkevičiūtė, G., Jonavičienė, K., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 83–87. [Google Scholar]
- Butkutė, B.; Padarauskas, A.; Cesevičienė, J.; Taujenis, L.; Norkevičienė, E. Phytochemical composition of temperate perennial legumes. Crop Pasture Sci. 2018, 69, 1020–1030. [Google Scholar] [CrossRef]
- Butkutė, B.; Benetis, R.; Padarauskas, A.; Cesevičienė, J.; Dagilytė, A.; Taujenis, L.; Rodovičius, H.; Lemežienė, N. Young herbaceous legumes—A natural reserve of bioactive compounds and antioxidants for healthy food and supplements. J. Appl. Bot. Food Qual. 2017, 90, 346–353. [Google Scholar] [CrossRef]
- Butkutė, B.; Padarauskas, A.; Cesevičienė, J.; Pavilonis, A.; Taujenis, L.; Lemežienė, N. Perennial legumes as a source of ingredients for healthy food: Proximate, mineral and phytoestrogen composition and antibacterial activity. J. Food Sci. Technol. 2017, 54, 2661–2669. [Google Scholar] [CrossRef] [PubMed]
- Prati, S.; Baravelli, V.; Fabbri, D.; Schwarzinger, C.; Brandolini, V.; Maietti, A.; Tedeschi, P.; Benvenuti, S.; Macchia, M.; Marotti, I.; et al. Composition and content of seed flavonoids in forage and grain legume crops. J. Sep. Sci. 2007, 30, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; De Bruyne, T.; Hermans, N.; Apers, S.; Vanden Berghe, D.; Vlietinck, A.J. Proanthocyanidins in health care: Current and new trends. Curr. Med. Chem. 2004, 11, 1345–1359. [Google Scholar] [CrossRef]
- Seguin, P.; Zheng, W. Phytoestrogen content of alfalfa cultivars grown in eastern Canada. J. Sci. Food Agric. 2006, 86, 765–771. [Google Scholar] [CrossRef]
- Boe, A.; Bortnem, R.; Johnson, P.J. Changes in weight and germinability of black medic seed over a growing season, with a new seed predator. Proc. S. D. Acad. Sci. 2016, 95, 105–117. [Google Scholar]
- Agelet, A.; Valles, J. Studies on pharmaceutical ethnobotany in the region of Pallars (Pyrenees, Catalonia, Iberian Peninsula). Part II. New or very rare uses of previously known medicinal plants. J. Ethnopharmacol. 2003, 84, 211–227. [Google Scholar] [CrossRef]
- Aguilera, Y.; Díaz, M.F.; Jiménez, T.; Benítez, V.; Herrera, T.; Cuadrado, C.; Martín-Pedrosa, M.; Martín-Cabrejas, M.A. Changes in nonnutritional factors and antioxidant activity during germination of nonconventional legumes. J. Agric. Food Chem. 2013, 61, 8120–8125. [Google Scholar] [CrossRef] [PubMed]
- Cevallos-Casals, B.A.; Cisneros-Zevallos, L. Impact of germination on phenolic content and antioxidant activity of 13 edible seed species. Food Chem. 2010, 119, 1485–1490. [Google Scholar] [CrossRef]
- Plaza, L.; de Ancos, B.; Cano, M.P. Nutritional and health-related compounds in sprouts and seeds of soybean (Glycine max), wheat (Triticum aestivum L.) and alfalfa (Medicago sativa) treated by a new drying method. Eur. Food Res. Technol. 2003, 216, 138–144. [Google Scholar] [CrossRef]
- Danilčenko, H.; Dabkevičius, Z.; Jarienė, E.; Tarasevičienė, Ž.; Televičiūtė, D.; Tamošiūnas, A.; Jeznach, M. The effect of stinging nettle and field horsetail extracts on the synthesis of biologically active compounds in germinated leguminous and quinoa seed. Zemdirbyste 2017, 104, 337–344. [Google Scholar] [CrossRef]
- Ebert, A.W.; Wu, T.H.; Yang, R.Y. Amaranth sprouts and microgreens–a homestead vegetable production option to enhance food and nutrition security in the rural-urban continuum. In Proceedings of the Regional Symposium on Sustaining Small-Scale Vegetable Production and Marketing Systems for Food and Nutrition Security (SEAVEG2014), Bangkog, Tailand, 25–27 February 2014; Hughes, J.d., Kasemsap, P., Dasgupta, S., Dutta, O.P., Ketsa, S., Chaikiattiyos, S., Linwattana, G., Kosiyachinda, S., Chantrasmi, V., Eds.; The World Vegetable Center: Shanhua, Taiwan, 2014; pp. 233–244. [Google Scholar]
- Márton, M.; Mándoki, Z.; Csapo, J. Evaluation of biological value of sprouts I. Fat content, fatty acid composition. Acta Univ. Sapientiae Aliment. 2010, 3, 53–65. [Google Scholar]
- Oleszek, W.A. Composition and quantitation of saponins in alfalfa (Medicago sativa L.) seedlings. J. Agric. Food Chem. 1998, 46, 960–962. [Google Scholar] [CrossRef]
- Budryn, G.; Gałązka-Czarnecka, I.; Brzozowska, E.; Grzelczyk, J.; Mostowski, R.; Żyżelewicz, D.; Cerón-Carrasco, J.P.; Pérez-Sánchez, H. Evaluation of estrogenic activity of red clover (Trifolium pratense L.) sprouts cultivated under different conditions by content of isoflavones, calorimetric study and molecular modelling. Food Chem. 2018, 245, 324–336. [Google Scholar] [CrossRef]
- Baldinger, L.; Hagmüller, W.; Minihuber, U.; Matzner, M.; Zollitsch, W. Sainfoin seeds in organic diets for weaned piglets—Utilizing the protein-rich grains of a long-known forage legume. Renew. Agric. Food 2016, 31, 12–21. [Google Scholar] [CrossRef]
- Ebert, A.W.; Chang, C.H.; Yan, M.R.; Yang, R.Y. Nutritional composition of mungbean and soybean sprouts compared to their adult growth stage. Food Chem. 2017, 237, 15–22. [Google Scholar] [CrossRef]
- Dueñas, M.; Sarmento, T.; Aguilera, Y.; Benitez, V.; Mollá, E.; Esteban, R.M.; Martín-Cabrejas, M.A. Impact of cooking and germination on phenolic composition and dietary fibre fractions in dark beans (Phaseolus vulgaris L.) and lentils (Lens culinaris L.). LWT-Food Sci. Technol. 2016, 66, 72–78. [Google Scholar] [CrossRef]
- Hedley, C.L. Grain legume carbohydrates. In Carbohydrates in Grain Legume Seeds: Improving Nutritional Quality and Agronomic Characteristics; Hedley, C.L., Ed.; CABI Publishing: Norwich, UK, 2000; pp. 11–14. [Google Scholar]
- Masood, T.; Shah, H.U.; Zeb, A. Effect of sprouting time on proximate composition and ascorbic acid level of mung bean (Vigna radiate L.) and chickpea (Cicer Arietinum L.) seeds. J. Anim. Plant Sci. 2014, 24, 850–859. [Google Scholar]
- Maneemegalai, S.; Nandakumar, S. Biochemical studies on the germinated seeds of Vigna radiata (L.) R. Wilczek, Vigna mungo (L.) Hepper and Pennisetum typhoides (Burm f.) Stapf and CE Hubb. Int. J. Agric. Res. 2011, 6, 601–606. [Google Scholar] [CrossRef]
- Machado, A.L.D.L.; Barcelos, M.D.F.P.; Teixeira, A.H.R.; Nogueira, D.A. Evaluation of chemical compounds in Fabaceae sprouts for the human consumption. Cienc. Agrotec. 2009, 33, 1071–1078. [Google Scholar] [CrossRef]
- Khalil, A.W.; Zeb, A.; Mahmood, F.; Tariq, S.; Khattak, A.B.; Shah, H. Comparison of sprout quality characteristics of desi and kabuli type chickpea cultivars (Cicer arietinum L.). LWT-Food Sci. Technol. 2007, 40, 937–945. [Google Scholar] [CrossRef]
- Bau, H.M.; Villaume, C.; Nicolas, J.P.; Méjean, L. Effect of germination on chemical composition, biochemical constituents and antinutritional factors of soya bean (Glycine max) seeds. J. Sci. Food Agric. 1997, 73, 1–9. [Google Scholar] [CrossRef]
- Devi, C.B.; Kushwaha, A.; Kumar, A. Sprouting characteristics and associated changes in nutritional composition of cowpea (Vigna unguiculata). J. Food Sci. Technol. 2015, 52, 6821–6827. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking in human diets–iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Picciano, M.F.; Betz, J.M.; Fisher, K.D.; Saldanha, L.G.; Yetley, E.A.; Coates, P.M.; Radimer, K.; Bindewald, B.; Sharpless, K.E.; et al. Progress in development of an integrated dietary supplement ingredient database at the NIH Office of Dietary Supplements. J. Food Compost. Anal. 2006, 19, S108–S114. [Google Scholar] [CrossRef]
- Iqbal, A.; Khalil, I.A.; Ateeq, N.; Khan, M.S. Nutritional quality of important food legumes. Food Chem. 2006, 97, 331–335. [Google Scholar] [CrossRef]
- Sangronis, E.; Machado, C.J. Influence of germination on the nutritional quality of Phaseolus vulgaris and Cajanus cajan. LWT-Food Sci. Technol. 2007, 40, 116–120. [Google Scholar] [CrossRef]
- Özcan, M.M.; Al Juhaimi, F. Effect of sprouting and roasting processes on some physico-chemical properties and mineral contents of soybean seed and oils. Food Chem. 2014, 154, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Adatorwovor, R.; Roggenkamp, K.; Anderson, J. Intakes of calcium and phosphorus and calculated calcium-to-phosphorus ratios of older adults: NHANES 2005–2006 data. Nutrients 2015, 7, 9633–9639. [Google Scholar] [CrossRef] [PubMed]
- Kemi, V.E.; Kärkkäinen, M.U.; Rita, H.J.; Laaksonen, M.M.; Outila, T.A.; Lamberg-Allardt, C.J. Low calcium: Phosphorus ratio in habitual diets affects serum parathyroid hormone concentration and calcium metabolism in healthy women with adequate calcium intake. Br. J. Nutr. 2010, 103, 561–568. [Google Scholar] [CrossRef] [PubMed]
- McClure, S.T.; Chang, A.R.; Selvin, E.; Rebholz, C.M.; Appel, L.J. Dietary sources of phosphorus among adults in the United States: Results from NHANES 2001–2014. Nutrients 2017, 9, 95. [Google Scholar] [CrossRef] [PubMed]
- Dave, S.; Yadav, B.K.; Tarafdar, J.C. Phytate phosphorus and mineral changes during soaking, boiling and germination of legumes and pearl millet. J. Food Sci. Technol. 2008, 45, 344. [Google Scholar]
- Bains, K.; Uppal, V.; Kaur, H. Optimization of germination time and heat treatments for enhanced availability of minerals from leguminous sprouts. J. Food Sci. Technol. 2014, 51, 1016–1020. [Google Scholar] [CrossRef]
- Karadima, V.; Kraniotou, C.; Bellos, G.; Tsangaris, G.T. Drug-micronutrient interactions: Food for thought and thought for action. EPMA J. 2016, 7, 10. [Google Scholar] [CrossRef]
- Gregory, P.J.; Wahbi, A.; Adu-Gyamfi, J.; Heiling, M.; Gruber, R.; Joy, E.J.; Broadley, M.R. Approaches to reduce zinc and iron deficits in food systems. Glob. Food Secur. 2017, 15, 1–10. [Google Scholar] [CrossRef]
- La Frano, M.R.; De Moura, F.F.; Boy, E.; Lönnerdal, B.; Burri, B.J. Bioavailability of iron, zinc, and provitamin A carotenoids in biofortified staple crops. Nutr. Rev. 2014, 72, 289–307. [Google Scholar] [CrossRef]
- Hunt, J.R. Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. Am. J. Clin. Nutr. 2003, 78, 633S–639S. [Google Scholar] [CrossRef]
- Luo, Y.W.; Xie, W.H.; Jin, X.X.; Wang, Q.; He, Y.J. Effects of germination on iron, zinc, calcium, manganese, and copper availability from cereals and legumes. CYTA-J. Food. 2014, 12, 22–26. [Google Scholar] [CrossRef]
- Urbano, G.; López-Jurado, M.; Aranda, C.; Vilchez, A.; Cabrera, L.; Porres, J.M.; Aranda, P. Evaluation of zinc and magnesium bioavailability from pea (Pisum sativum, L.) sprouts. Effect of illumination and different germination periods. Int. J. Food Sci. Technol. 2006, 41, 618–626. [Google Scholar] [CrossRef]
- Forslund, L.C.; Andersson, H.C. Phytoestrogens in Foods on the Nordic Market: A Literature Review on Occurrence and Levels. Nordic Council of Ministers, 2017. Available online: https://www.diva-portal.org/smash/get/diva2:1137191/fulltext01.pdf (accessed on 5 November 2018).
- Mattioli, S.; Dal Bosco, A.; Martino, M.; Ruggeri, S.; Marconi, O.; Sileoni, V.; Falcinelli, B.; Castellini, C.; Benincasa, P. Alfalfa and flax sprouts supplementation enriches the content of bioactive compounds and lowers the cholesterol in hen egg. J. Funct. Foods 2016, 22, 454–462. [Google Scholar] [CrossRef]
- Wu, Z.; Song, L.; Feng, S.; Liu, Y.; He, G.; Yioe, Y.; Liu, S.Q.; Huang, D. Germination dramatically increases isoflavonoid content and diversity in chickpea (Cicer arietinum L.) seeds. J. Agric. Food Chem. 2012, 60, 8606–8615. [Google Scholar] [CrossRef]
- López, A.; El-Naggar, T.; Dueñas, M.; Ortega, T.; Estrella, I.; Hernández, T.; Gómez-Serranillos, M.P.; Palomino, O.M.; Carretero, M.E. Effect of cooking and germination on phenolic composition and biological properties of dark beans (Phaseolus vulgaris L.). Food Chem. 2013, 138, 547–555. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Ahn, J.K.; Khanh, T.D.; Chun, S.C.; Kim, S.L.; Ro, H.M.; Song, H.K.; Chung, I.M. Comparison of isoflavone concentrations in soybean (Glycine max (L.) Merrill) sprouts grown under two different light conditions. J. Agric. Food Chem. 2007, 55, 9415–9421. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.Y.; Lai, H.M. Bioactive compounds in legumes and their germinated products. J. Agric. Food Chem. 2006, 54, 3807–3814. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.Y.; Seo, D.B.; Shin, H.J.; Lee, S.J. Effect of Aspergillus oryzae-challenged germination on soybean isoflavone content and antioxidant activity. J. Agric. Food Chem. 2012, 60, 2807–2814. [Google Scholar] [CrossRef]
- Shimoyamada, M.; Okubo, K. Variation in saponin contents in germinating soybean seeds and effect of light irradiation. Agric. Biol. Chem. 1991, 55, 577–579. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Singh, N.; Kaur, A. Saponins in pulses and their health promoting activities: A review. Food Chem. 2017, 233, 540–549. [Google Scholar] [CrossRef]
- Pecetti, L.; Tava, A.; Romani, M.; De Benedetto, M.G.; Corsi, P. Variety and environment effects on the dynamics of saponins in lucerne (Medicago sativa L.). Eur. J. Agron. 2006, 25, 187–192. [Google Scholar] [CrossRef]
- Troszyńska, A.; Wołejszo, A.; Narolewska, O. Effect of germination time on the content of phenolic compounds and sensory quality of mung bean (Vigna radiata L.) sprouts. Pol. J. Food Nutr. Sci. 2006, 15/56, 453–459. [Google Scholar]
- Kalpanadevi, V.; Mohan, V.R. Effect of processing on antinutrients and in vitro protein digestibility of the underutilized legume, Vigna unguiculata (L.) Walp subsp. unguiculata. LWT-Food Sci. Technol. 2013, 51, 455–461. [Google Scholar] [CrossRef]
- Świeca, M.; Gawlik-Dziki, U.; Kowalczyk, D.; Złotek, U. Impact of germination time and type of illumination on the antioxidant compounds and antioxidant capacity of Lens culinaris sprouts. Sci. Hortic. 2012, 140, 87–95. [Google Scholar] [CrossRef]
- Klopsch, R.; Baldermann, S.; Voss, A.; Rohn, S.; Schreiner, M.; Neugart, S. Bread enriched with legume microgreens and leaves–ontogenetic and baking-driven changes in the profile of secondary plant metabolites. Front. Chem. 2018, 6, 322. [Google Scholar] [CrossRef] [PubMed]
- Cornara, L.; Xiao, J.; Burlando, B. Therapeutic potential of temperate forage legumes: A review. Crit. Rev. Food Sci. Nutr. 2016, 56, S149–S161. [Google Scholar] [CrossRef] [PubMed]
- Owusu-Apenten, R. Introduction to Food Chemistry; CRC press: Boca Raton, FL, USA, 2004; p. 272. [Google Scholar]
- Leśniewicz, A.; Jaworska, K.; Żyrnicki, W. Macro-and micro-nutrients and their bioavailability in polish herbal medicaments. Food Chem. 2006, 99, 670–679. [Google Scholar] [CrossRef]
- Pecetti, L.; Biazzi, E.; Tava, A. Variation in saponin content during the growing season of spotted medic (Medicago arabica (L.) Huds.). J. Agric. Food Chem. 2010, 90, 2405–2410. [Google Scholar] [CrossRef]
- Taujenis, L.; Padarauskas, A.; Cesevičienė, J.; Lemežienė, N.; Butkutė, B. Determination of coumestrol in lucerne by ultra-high pressure liquid chromatography-mass spectrometry. Chemija 2016, 27, 60–64. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Sample | Ash | Crude Protein | Crude Fat | Crude Fibre | Total Carbohydrates |
|---|---|---|---|---|---|
| Raw seeds | |||||
| Tpr | 4.71 ± 0.34 cdB | 34.5 ± 0.19 abC | 8.98 ± 0.01 bA | 13.8 ± 0.71 abC | 38.0 |
| Tme | 7.32 ± 0.24 bA | 34.8 ± 0.22 abB | n.a. | n.a. | n.a. |
| Msa | 4.32 ± 0.02 dB | 34.7 ± 0.41 abB | 11.6 ± 0.07 aA | 13.3 ± 0.23 bB | 36.1 |
| Mlu | 3.97 ± 0.03 eC | 34.1 ± 0.66 abC | 3.10 ± 0.35 dC | 12.8 ± 0.77 bAB | 46.0 |
| Ovi | 4.46 ± 0.08 dC | 35.8 ± 0.43 aC | 6.62 ± 0.21 cA | 12.2 ± 0.60 bcA | 40.9 |
| Agl | 8.19 ± 0.10 aA | 32.0 ± 0.37 bB | 2.85 ± 0.14 dB | 16.2 ± 1.00 aA | 40.8 |
| Aci | 5.24 ± 0.23 cA | 36.1 ± 0.13 aB | 3.78 ± 0.14 dA | 14.9 ± 0.69 aA | 40.0 |
| AVG* | 5.46 | 34.6 | 6.15 | 13.9 | 40.3 |
| AVG** | 4.37 | 34.8 | 7.56 | 13.0 | 40.3 |
| Sprouted seeds | |||||
| Tpr | 4.42 ± 0.37 bB | 38.9 ± 0.09 dB | 9.28 ± 0.01 aA | 15.6 ± 0.88 bB | 31.8 |
| Tme | 4.99 ± 0.01 abB | 39.8 ± 0.30 cdA | 6.62 ± 0.21 b | 15.3 ± 0.61 b | 33.3 |
| Msa | 4.65 ± 0.25 bB | 42.8 ± 0.38 bA | 8.88 ± 0.33 aB | 14.0 ± 0.45 bA | 29.7 |
| Mlu | 4.38 ± 0.31 bB | 42.6 ± 0.13 bB | 5.14 ± 0.24 cA | 13.9 ± 0.67 bA | 34.0 |
| Ovi | 5.35 ± 0.08 aB | 45.2 ± 0.41 aB | 5.58 ± 0.03 cB | 11.4 ± 0.54 cA | 32.5 |
| Agl | 4.24 ± 0.21 bB | 37.0 ± 0.26 eA | 3.95 ± 0.11 dA | 17.2 ± 0.43 aA | 37.6 |
| Aci | 5.29 ± 0.44 aA | 40.3 ± 0.03 cA | 4.08 ± 0.08 dA | 15.0 ± 0.58 bA | 35.3 |
| AVG* | 4.76 | 40.9 | 6.22 | 14.6 | 33.5 |
| AVG** | 4.7 | 42.4 | 7.22 | 13.7 | 32 |
| Microgreens | |||||
| Tpr | 5.82 ± 0.25 bA | 45.7 ± 0.43 bcA | 4.84 ± 0.23 bcB | 17.5 ± 1.06 aA | 26.1 |
| Tme | na | na | na | na | na |
| Msa | 5.34 ± 0.28 bcA | 44.0 ± 0.93 cA | 6.48 ± 0.21 aC | 14.5 ± 0.32 bA | 29.7 |
| Mlu | 6.03 ± 0.18 bA | 48.0 ± 0.14 bA | 4.13 ± 0.24 cB | 14.4 ± 0.69 bA | 27.4 |
| Ovi | 7.31 ± 0.40 aA | 58.7 ± 1.04 aA | 5.26 ± 0.16 bB | 8.7 ± 0.47 cB | 20.0 |
| Agl | na | na | na | na | na |
| Aci | na | na | na | na | na |
| AVG** | 6.13 | 49.9 | 5.18 | 13.8 | 25.8 |
| Sample | K | Ca | Mg | P | Ca:P (% from Ca:P in Seeds) | Fe | Zn |
|---|---|---|---|---|---|---|---|
| g/100 g | mg/100 g | ||||||
| Raw seeds | |||||||
| Tpr | 1.35 ± 0.01 bA | 0.491 ± 0.017 cA | 0.403 ± 0.015 aB | 0.702 ± 0.015 abB | 0.699 | 17.8 ± 0.013 cA | 7.11 ± 0.767 aC |
| Tme | 1.10 ± 0.09 cB | 0.495 ± 0.053 cA | 0.309 ± 0.013 bB | 0.732 ± 0.070 aA | 0.676 | 10.2 ± 0.002 dB | 5.74 ± 0.611 bB |
| Msa | 1.27 ± 0.03 bB | 0.716 ± 0.089 abA | 0.304 ± 0.001 bC | 0.749 ± 0.038 aC | 0.956 | 10.5 ± 0.603 dB | 5.71 ± 0.450 bC |
| Mlu | 1.29 ± 0.01 bA | 0.789 ± 0.053 aA | 0.289 ± 0.013 cB | 0.554 ± 0.010 cC | 1.424 | 9.20 ± 0.310 dB | 3.35 ± 0.349 cB |
| Ovi | 1.77 ± 0.26 aAB | 0.716 ± 0.053 abA | 0.243 ± 0.025 dB | 0.673 ± 0.003 bC | 1.064 | 10.5 ± 0.811 dC | 6.32 ± 0.590 abB |
| Agl | 1.32 ± 0.17 bB | 0.703 ± 0.053 bA | 0.288 ± 0.009 cB | 0.592 ± 0.017 cA | 1.188 | 73.4 ± 1.57 aA | 5.01 ± 0.979 bcC |
| Aci | 1.37 ± 0.05 bC | 0.669 ± 0.053 bA | 0.254 ± 0.013 dB | 0.745 ± 0.029 aA | 0.898 | 28.6 ± 1.04 bA | 6.08 ± 0.527 bC |
| AVG* | 1.35 | 0.654 | 0.299 | 0.678 | 0.986 | 25.00 | 5.62 |
| AVG** | 1.42 | 0.678 | 0.310 | 0.670 | 1.036 | 12.00 | 5.62 |
| Sprouted seeds | |||||||
| Tpr | 1.00 ± 0.00 dB | 0.352 ± 0.004 cB | 0.437 ± 0.059 aA | 0.689 ± 0.018 cB | 0.511 (−27.0) | 16.4 ± 1.435 cA | 20.1 ± 0.401 aB |
| Tme | 1.52 ± 0.07 abA | 0.330 ± 0.004 cB | 0.374 ± 0.014 bA | 0.729 ± 0.007 bcA | 0.453 (−33.1) | 21.3 ± 0.334 bA | 20.3 ± 1.68 aA |
| Msa | 1.31 ± 0.04 cB | 0.555 ± 0.004 bB | 0.319 ± 0.035 bcB | 0.858 ± 0.004 aB | 0.647 (−32.3) | 16.6 ± 0.482 cA | 11.8 ± 0.768 cB |
| Mlu | 1.22 ± 0.04 cdAB | 0.671 ± 0.005 aB | 0.307 ± 0.016 cA | 0.717 ± 0.012 cB | 0.936 (−34.3) | 13.9 ± 0.598 dA | 16.2 ± 1.35 bA |
| Ovi | 1.67 ± 0.18 aB | 0.631 ± 0.003 aB | 0.227 ± 0.031 eB | 0.790 ± 0.014 bB | 0.799 (−24.9) | 13.4 ± 0.314 dB | 13.6 ± 1.02 bcA |
| Agl | 1.40 ± 0.06 bB | 0.580 ± 0.001 bB | 0.284 ± 0.018 dB | 0.603 ± 0.006 dA | 0.962 (−19.0) | 34.5 ± 1.02 aB | 11.4 ± 0.810 cB |
| Aci | 1.48 ± 0.02 bB | 0.584 ± 0.023 bB | 0.246 ± 0.010 deB | 0.746 ± 0.014 bcA | 0.783 (−12.8) | 17.1 ± 0.60 cC | 9.50 ± 0.924 cB |
| AVG* | 1.37 | 0.529 | 0.313 | 0.733 | 0.727 (−26.3) | 18.65 | 14.70 |
| AVG** | 1.30 | 0.552 | 0.323 | 0.764 | 0.723 (−28.6) | 15.08 | 15.43 |
| Microgreens | |||||||
| Tpr | 1.34 ± 0.06 cdA | 0.238 ± 0.012 eC | 0.358 ± 0.016 bC | 0.819 ± 0.008 cA | 0.291 (−58.5) | 16.5 ± 0.434 bcA | 29.9 ± 1.50 aA |
| Tme | na | na | na | na | na | na | |
| Msa | 1.49 ± 0.01 cA | 0.611 ± 0.019 bAB | 0.373 ± 0.068 aA | 0.878 ± 0.006 bA | 0.696 (−27.2) | 17.8 ± 0.902 bcA | 27.3 ± 1.69 aA |
| Mlu | 1.13 ± 0.04 dB | 0.702 ± 0.024 aB | 0.293 ± 0.004 cAB | 0.852 ± 0.001 bA | 0.824 (−42.1) | 14.8 ± 1.13 cA | 17.9 ± 0.921 bA |
| Ovi | 1.98 ± 0.01 aA | 0.585 ± 0.038 bC | 0.299 ± 0.001 cA | 1.001 ± 0.009 aA | 0.584 (−45.1) | 19.8 ± 0.511 bA | 13.3 ± 1.24 cA |
| Agl | 1.79 ± 0.03 bA | 0.446 ± 0.038 cC | 0.340 ± 0.022 bA | na | 27.2 ± 0.733 aC | 19.6 ± 1.45 bA | |
| Aci | 1.81 ± 0.03 bA | 0.339 ± 0.038 dC | 0.326 ± 0.022 bcA | na | 21.5 ± 0.697 bB | 30.0 ± 1.57 aA | |
| AVG* | 1.59 | 0.487 | 0.332 | 0.888 | 0.599 (−60.6) | 19.60 | 23.00 |
| AVG** | 1.49 | 0.534 | 0.331 | 0.888 | 0.599 (−40.8) | 17.23 | 22.10 |
| Sample | Formononetin | Biochanin A | Daidzein | Genistein | Sum of Isoflavones |
|---|---|---|---|---|---|
| Raw seeds | |||||
| Tpr | 2.0 ± 3.3 bC | 1.0 ± 0.1 bC | ˂LOQ nc C | ˂LOQ nc C | 3.0 |
| Tme | 10.7 ± 0.8 aC | 7.9 ± 0.8 aC | ˂LOQ nc C | ˂LOQ nc C | 18.6 |
| Msa | ˂LOQ cC | ˂LOQ cC | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| Mlu | ˂LOQ cC | ˂LOQ cC | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| Ovi | ˂LOQ cC | ˂LOQ cC | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| Agl | ˂LOQ cC | ˂LOQ cC | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| Aci | ˂LOQ cC | ˂LOQ cC | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| AVG* | 6.4 | 4.5 | nc | nc | 10.8 |
| Sprouted seeds | |||||
| Tpr | 15.2 ± 1.3 bB | 4.9 ± 0.5 bB | 1.0 ± 0.1 bB | 3.5 ± 0.4 bB | 24.6 |
| Tme | 71.3 ± 4.5 aB | 16.5 ± 1.3 aB | 1.6 ± 0.2 aB | 7.8 ± 0.7 aB | 97.2 |
| Msa | 3.3 ± 0.3 cB | 1.6 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 4.9 |
| Mlu | 1.8 ± 0.9 cA | 1.4 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 3.2 |
| Ovi | 2.6 ± 1.7 cA | 2.3 ± 0.3 cA | ˂LOQ nc NC | ˂LOQ nc NC | 4.9 |
| Agl | 1.3 ± 0.2 cB | 0.8 ± 0.1 cB | ˂LOQ nc NC | ˂LOQ nc NC | 2.1 |
| Aci | 1.6 ± 1.0 cB | 1.1 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 2.7 |
| AVG* | 43.3 | 10.7 | 1.3 | 5.7 | 60.9 |
| AVG** | 2.14 | 1.42 | nc | nc | 3.56 |
| Microgreens | |||||
| Tpr | 89.9 ± 5.3 bA | 19.9 ± 1.3 bA | 1.4 ± 0.2 bA | 1.3 ± 1.0 bA | 112.5 |
| Tme | 117.5 ± 6.3 aA | 33.4 ± 2.1 aA | 2.7 ± 0.3 aA | 17.4 ± 1.4 aA | 171.0 |
| Msa | 4.4 ± 0.4 cA | 1.5 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 5.9 |
| Mlu | 2.2 ± 0.3 cA | 1.8 ± 0.3 cA | ˂LOQ nc NC | ˂LOQ nc NC | 4.0 |
| Ovi | 2.9 ± 0.4 cA | 2.1 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 5.0 |
| Agl | 2.3 ± 0.3 cA | 1.5 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 3.8 |
| Aci | 2.1 ± 0.3 cA | 1.2 ± 0.2 cA | ˂LOQ nc NC | ˂LOQ nc NC | 3.3 |
| AVG* | 103.7 | 26.7 | 2.1 | 9.4 | 141.8 |
| AVG** | 2.56 | 1.84 | nc | nc | 4.4 |
| Compounds | Sample | M. sativa | M. lupulina | O. viciifolia |
|---|---|---|---|---|
| Concentration, mg/100 g ± SD | ||||
| Coumestrol | Raw seeds | ˂LOQ nc NC | ˂LOQ nc NC | ˂LOQ |
| Sprouted seeds | 0.41 ± 0.06 bB | 1.12 ± 0.14 aB | ˂LOQ | |
| Microgreens | 1.27 ± 0.14 bA | 4.56 ± 0.33 aA | ˂LOQ | |
| Triterpene saponins | Raw seeds | 121 ± 21.1 aC | 68.0 ± 15.6 bC | ˂LOQ |
| Sprouted seeds | 286 ± 34.9 aB | 305 ± 47.8 aA | ˂LOQ | |
| Microgreens | 484 ± 66.0 aA | 411 ± 5.27 bA | ˂LOQ | |
| Condensed tannins | Raw seeds | ˂LOQ | ˂LOQ | ˂LOQ NC |
| Sprouted seeds | ˂LOQ | ˂LOQ | 388 ± 45.5 B | |
| Microgreens | ˂LOQ | ˂LOQ | 449 ± 51.7 A | |
| Tribe | Genus | Scientific Name (Abbreviation) | English Name | Cultivar/Wild Ecotype | Country of Origin, Geographic Coordinates |
|---|---|---|---|---|---|
| Trifolieae | Trifolium | T. pratense (Tpr) | Red clover | ‘Sadūnai’ | Lithuania, 55°23′49″ N; 23°51′40″ E |
| Trifolieae | Trifolium | T. medium (Tme) | Zigzag clover | Wild ecotype | Lithuania 55°32′42″ N; 25°02′23″ E |
| Trifolieae | Medicago | M. sativa (Msa) | Alfalfa | ‘Birutė’ | Lithuania, 55°23′49″ N; 23°51′40″ E |
| Trifolieae | Medicago | M. lupulina (Mlu) | Black medick | ‘Arka’ | Lithuania, 55°23′49″ N; 23°51′40″ E |
| Hedysareae | Onobrychis | O. viciifolia (Ovi) | Sainfoin | ‘Meduviai’ | Lithuania, 55°23′49″ N; 23°51′40″ E |
| Galegeae | Astragalus | A. glycyphyllos (Agl) | Liquorice milkvetch | Wild ecotype | Lithuania, 55°22′51″ N; 23°50′35″ E |
| Galegeae | Astragalus | A. cicer (Aci) | Cicer milkvetch | Wild ecotype | Latvia, 57°01′45″ N; 21°25′23″ E |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butkutė, B.; Taujenis, L.; Norkevičienė, E. Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens. Molecules 2019, 24, 133. https://doi.org/10.3390/molecules24010133
Butkutė B, Taujenis L, Norkevičienė E. Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens. Molecules. 2019; 24(1):133. https://doi.org/10.3390/molecules24010133
Chicago/Turabian StyleButkutė, Bronislava, Lukas Taujenis, and Eglė Norkevičienė. 2019. "Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens" Molecules 24, no. 1: 133. https://doi.org/10.3390/molecules24010133
APA StyleButkutė, B., Taujenis, L., & Norkevičienė, E. (2019). Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens. Molecules, 24(1), 133. https://doi.org/10.3390/molecules24010133