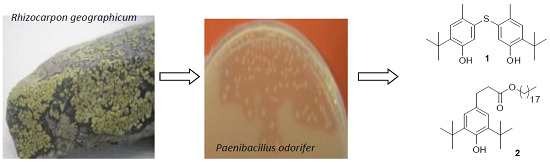
tert-Butylphenolic Derivatives from Paenibacillus odorifer—A Case of Bioconversion
Abstract
1. Introduction
2. Results
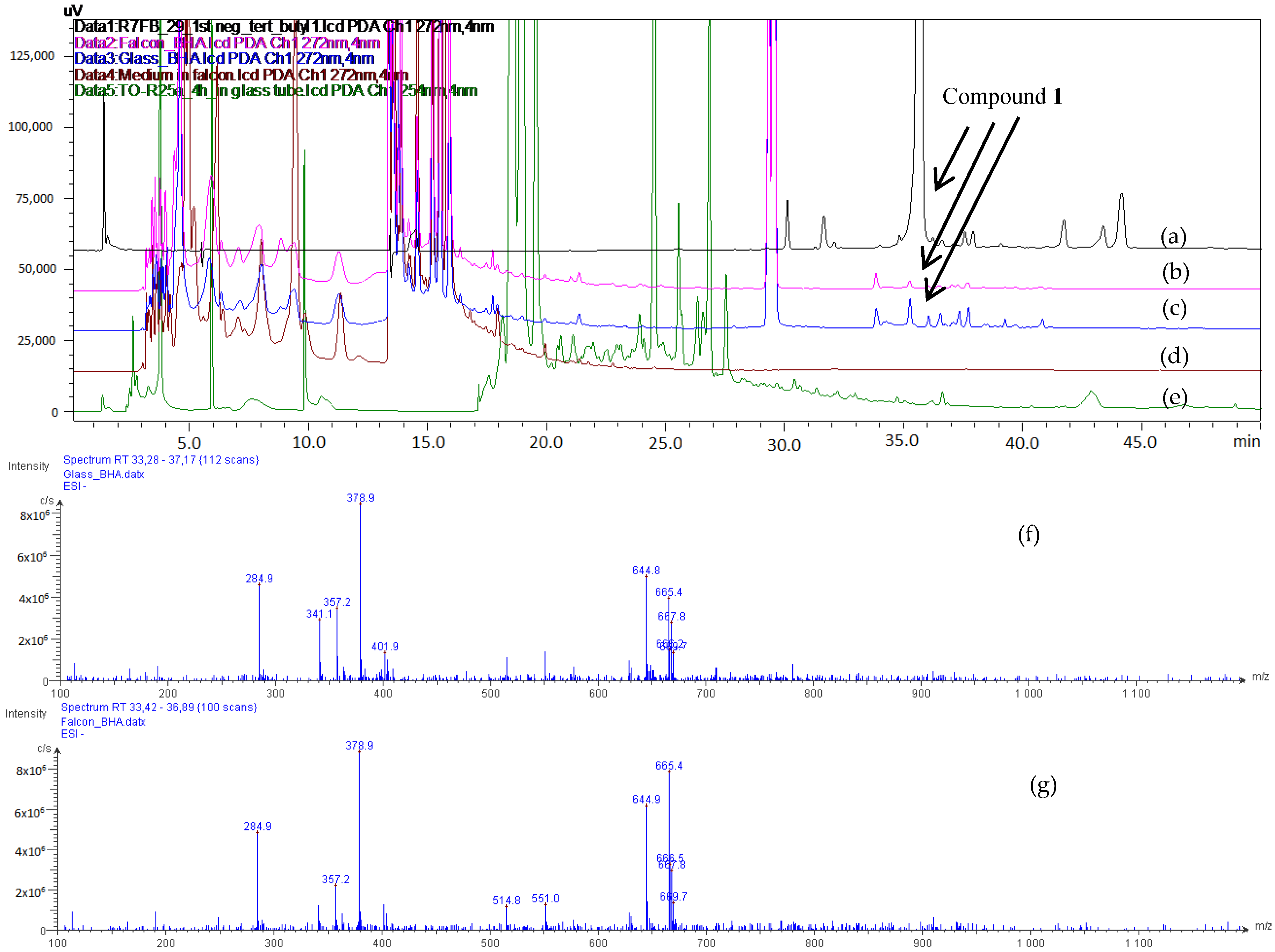
2.1. Process of Purification
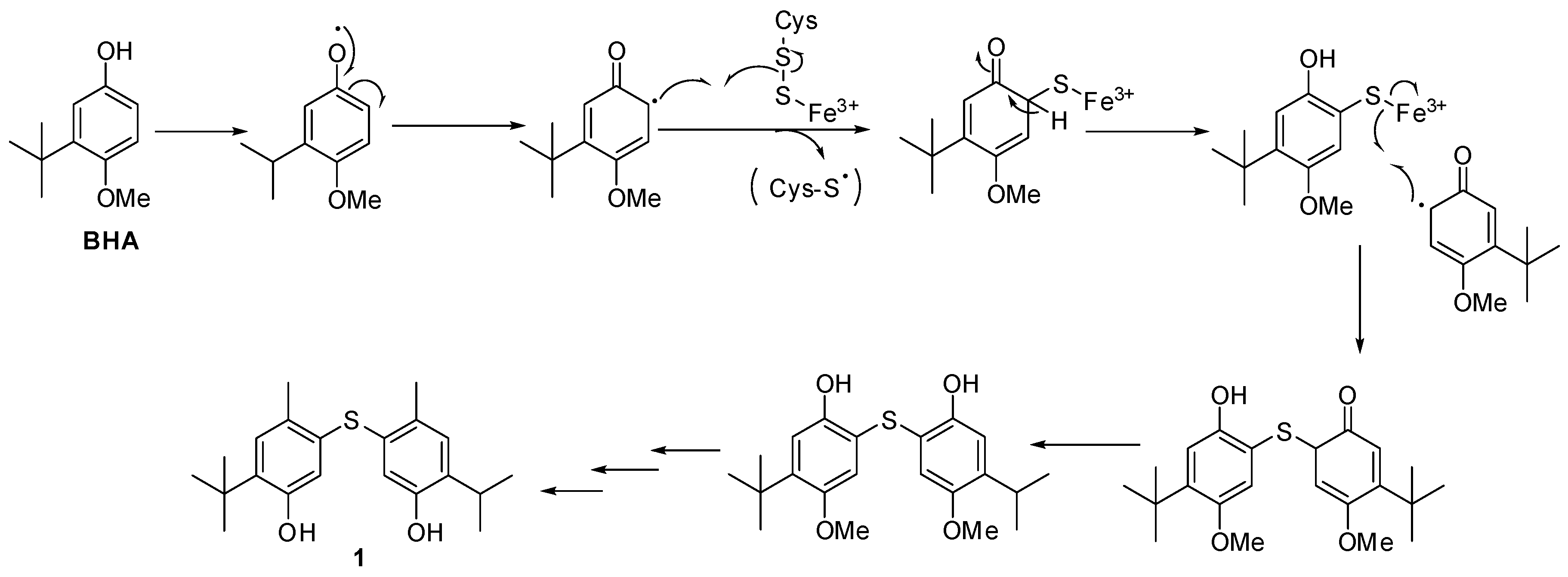
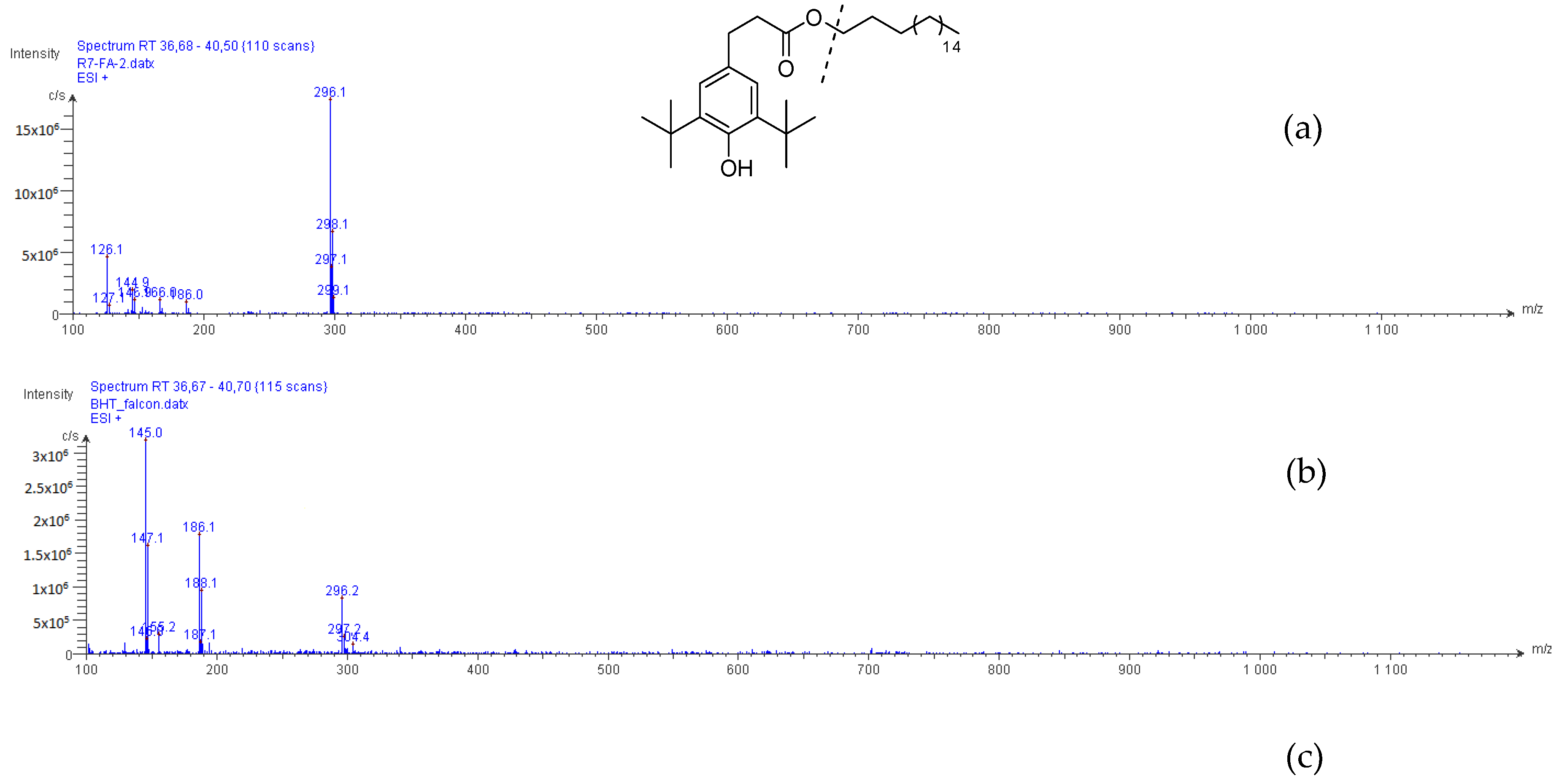
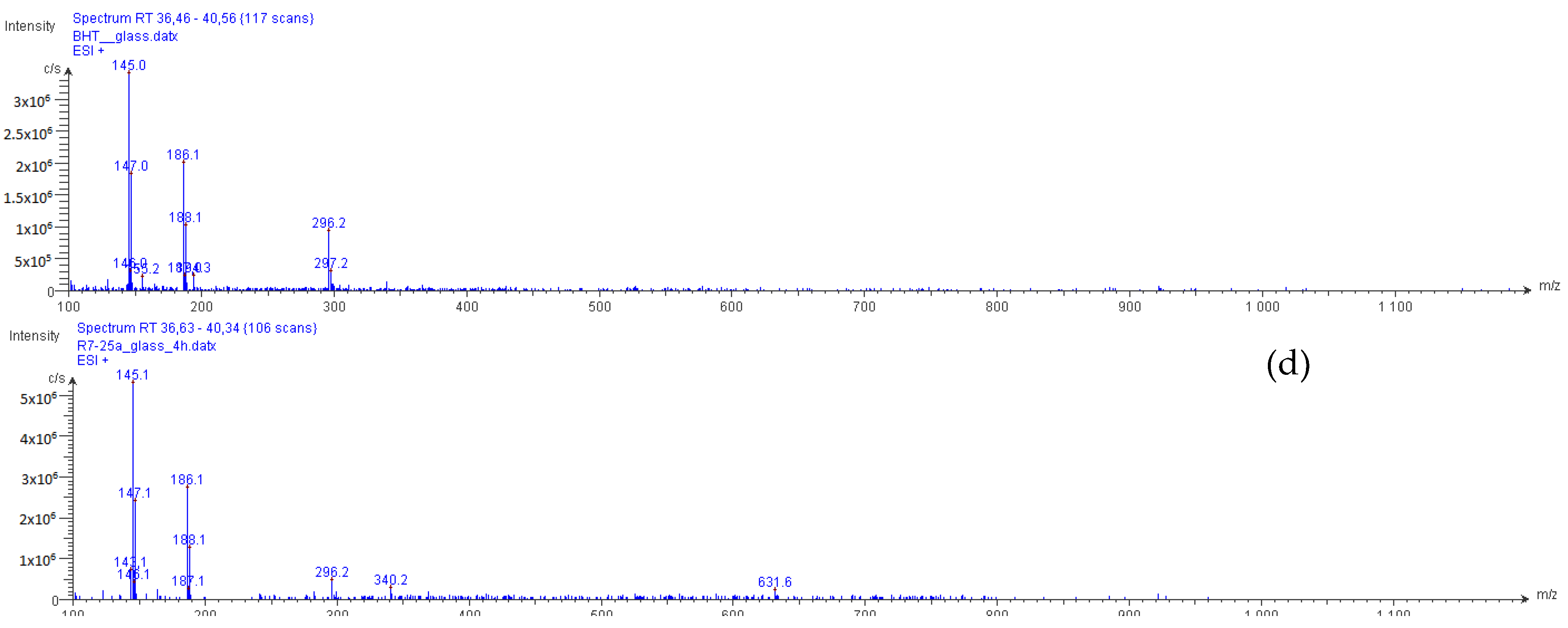
2.2. Structural Elucidation
2.3. Supplementation Assays
2.4. Cytotoxic Activity
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Collection and Phylogenetic Analysis of PC-GYM-TO Strain
3.3. Cultivation and Extraction
3.4. Purification and Isolation
3.5. Molecular Models and Dynamic Simulations
3.6. Cytotoxicity Assays
3.7. DNA Damage Assays
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, Y.; Schnell, B.; Baumann, S.; Müller, R.; Begley, T.P. Biosynthesis of Branched Alkoxy Groups: Iterative Methyl Group Alkylation by a Cobalamin-Dependent Radical SAM Enzyme. J. Am. Chem. Soc. 2017, 139, 1742–1745. [Google Scholar] [CrossRef] [PubMed]
- Bisel, P.; Al-Momani, L.; Müller, M. The tert-Butyl Group in Chemistry and Biology. Org. Biomol. Chem. 2008, 6, 2655–2665. [Google Scholar] [CrossRef] [PubMed]
- Dembitsky, V.M. Natural Neo Acids and Neo Alkanes: Their Analogs and Derivatives. Lipids 2006, 41, 309–340. [Google Scholar] [CrossRef] [PubMed]
- Luesch, H.; Yoshida, W.Y.; Moore, R.E.; Paul, V.J.; Corbett, T.H. Total Structure Determination of Apratoxin A, a Potent Novel Cytotoxin from the Marine Cyanobacterium Lyngbya majuscula. J. Am. Chem. Soc. 2001, 123, 5418–5423. [Google Scholar] [CrossRef] [PubMed]
- Lopanik, N.; Gustafson, K.R.; Lindquist, N. Structure of Bryostatin 20: A Symbiont-Produced Chemical Defense for Larvae of the Host Bryozoan, Bugula Neritina. J. Nat. Prod. 2004, 67, 1412–1414. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.R.; Cao, Z.; Engene, N.; Soria-Mercado, I.E.; Murray, T.F.; Gerwick, W.H. Palmyrolide A, an Unusually Stabilized Neuroactive Macrolide from Palmyra Atoll Cyanobacteria. Org. Lett. 2010, 12, 4490–4493. [Google Scholar] [CrossRef] [PubMed]
- Matthew, S.; Salvador, L.A.; Schupp, P.J.; Paul, V.J.; Luesch, H. Cytotoxic Halogenated Macrolides and Modified Peptides from the Apratoxin-Producing Marine Cyanobacterium Lyngbya bouillonii from Guam. J. Nat. Prod. 2010, 73, 1544–1552. [Google Scholar] [CrossRef] [PubMed]
- Tidgewell, K.; Engene, N.; Byrum, T.; Media, J.; Doi, T.; Valeriote, F.A.; Gerwick, W.H. Evolved Diversification of a Modular Natural Product Pathway: Apratoxins F and G, Two Cytotoxic Cyclic Depsipeptides from a Palmyra Collection of Lyngbya bouillonii. ChemBioChem 2010, 11, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Toyama, T.; Momotani, N.; Ogata, Y.; Miyamori, Y.; Inoue, D.; Sei, K.; Mori, K.; Kikuchi, S.; Ike, M. Isolation and Characterization of 4-tert-Butylphenol-Utilizing Sphingobium fuliginis Strains from Phragmites australis Rhizosphere Sediment. Appl. Environ. Microbiol. 2010, 76, 6733–6740. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.T.; Parrot, D.; Berg, G.; Grube, M.; Tomasi, S. Lichens as Natural Sources of Biotechnologically Relevant Bacteria. Appl. Microbiol. Biotechnol. 2015, 100, 583–595. [Google Scholar] [CrossRef] [PubMed]
- Varsha, K.K.; Devendra, L.; Shilpa, G.; Priya, S.; Pandey, A.; Nampoothiri, K.M. 2,4-Di-tert-Butyl Phenol as the Antifungal, Antioxidant Bioactive Purified from a Newly Isolated Lactococcus Sp. Int. J. Food Microbiol. 2015, 211, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Jaivel, N.; Uvarani, C.; Rajesh, R.; Velmurugan, D.; Marimuthu, P. Natural Occurrence of Organofluorine and Other Constituents from Streptomyces Sp. TC1. J. Nat. Prod. 2014, 77, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Belghit, S.; Driche, E.H.; Bijani, C.; Zitouni, A.; Sabaou, N.; Badji, B.; Mathieu, F. Activity of 2,4-Di-tert-Butylphenol Produced by a Strain of Streptomyces mutabilis Isolated from a Saharan Soil against Candida albicans and Other Pathogenic Fungi. J. Med. Mycol. 2016, 26, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.Z.; Yaniger, S.I.; Jordan, V.C.; Klein, D.J.; Bittner, G.D. Most Plastic Products Release Estrogenic Chemicals: A Potential Health Problem That Can Be Solved. Environ. Health Perspect. 2011, 119, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, Z.-W.; Lin, Q.-B.; Hu, C.-Y.; Su, Q.-Z.; Wu, Y.-M. Determination of Polymer Additives-Antioxidants, Ultraviolet Stabilizers, Plasticizers and Photoinitiators in Plastic Food Package by Accelerated Solvent Extraction Coupled with High-Performance Liquid Chromatography. J. Chromatogr. Sci. 2015, 53, 1026–1035. [Google Scholar] [CrossRef] [PubMed]
- Babich, H. Butylated Hydroxytoluene (BHT): A Review. Environ. Res. 1982, 29, 1–29. [Google Scholar] [CrossRef]
- Fontecave, M.; Ollagnier-de-Choudens, S.; Mulliez, E. Biological Radical Sulfur Insertion Reactions. Chem. Rev. 2003, 103, 2149–2166. [Google Scholar] [CrossRef] [PubMed]
- Fontecave, M.; Py, B.; Ollagnier de Choudens, S.; Barras, F. From Iron and Cysteine to Iron-Sulfur Clusters: The Biogenesis Protein Machineries. EcoSal Plus 2008, 3, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Millot, M.; Tomasi, S.; Studzinska, E.; Rouaud, I.; Boustie, J. Cytotoxic Constituents of the Lichen Diploicia canescens. J. Nat. Prod. 2009, 72, 2177–2180. [Google Scholar] [CrossRef] [PubMed]
- Krieger, E.; Darden, T.; Nabuurs, S.B.; Finkelstein, A.; Vriend, G. Making optimal use of empirical energy functions: Force-field parameterization in crystal space. Proteins 2004, 57, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Van-der-Spoel, D.; Lindahl, E.; Hess, B.; Groenhof, G.; Mark, A.E.; Berendsen, H.J. GROMACS: Fast, flexible, and free. J. Comput. Chem. 2005, 26, 1701–1718. [Google Scholar] [CrossRef] [PubMed]
- Bigot, N.; Mouche, A.; Preti, M.; Loisel, S.; Renoud, M.L.; Le Guével, R.; Sensebé, L.; Tarte, K.; Pedeux, R. Hypoxia Differentially Modulates the Genomic Stability of Clinical-Grade ADSCs and BM-MSCs in Long-Term Culture. Stem Cells 2015, 33, 3608–3620. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the Compounds 1, 2 are available from the authors. |
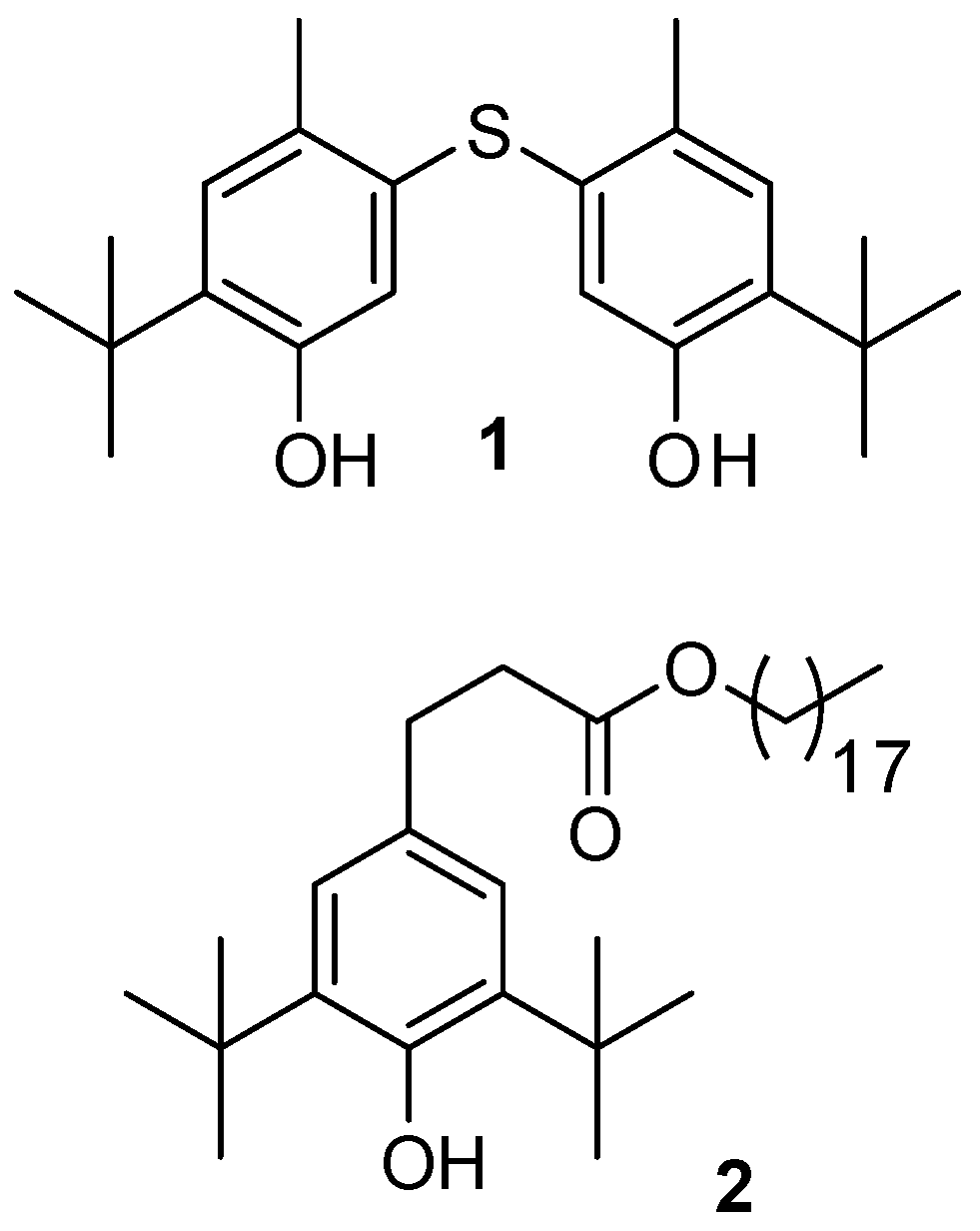
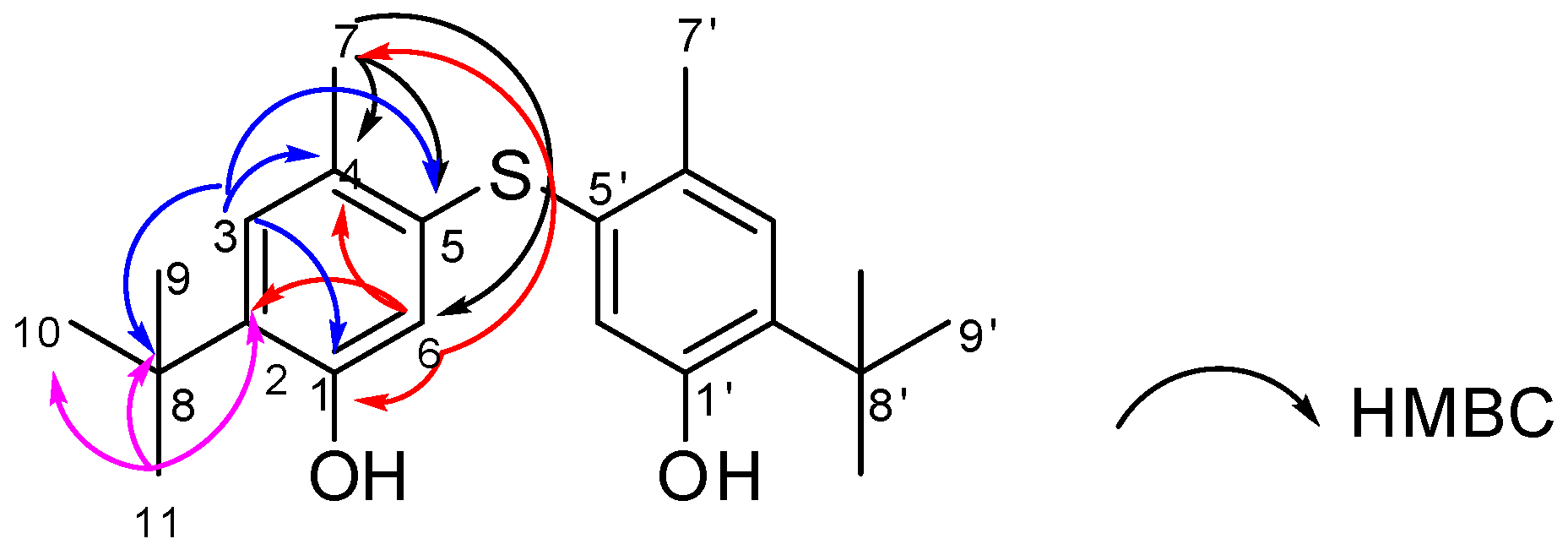
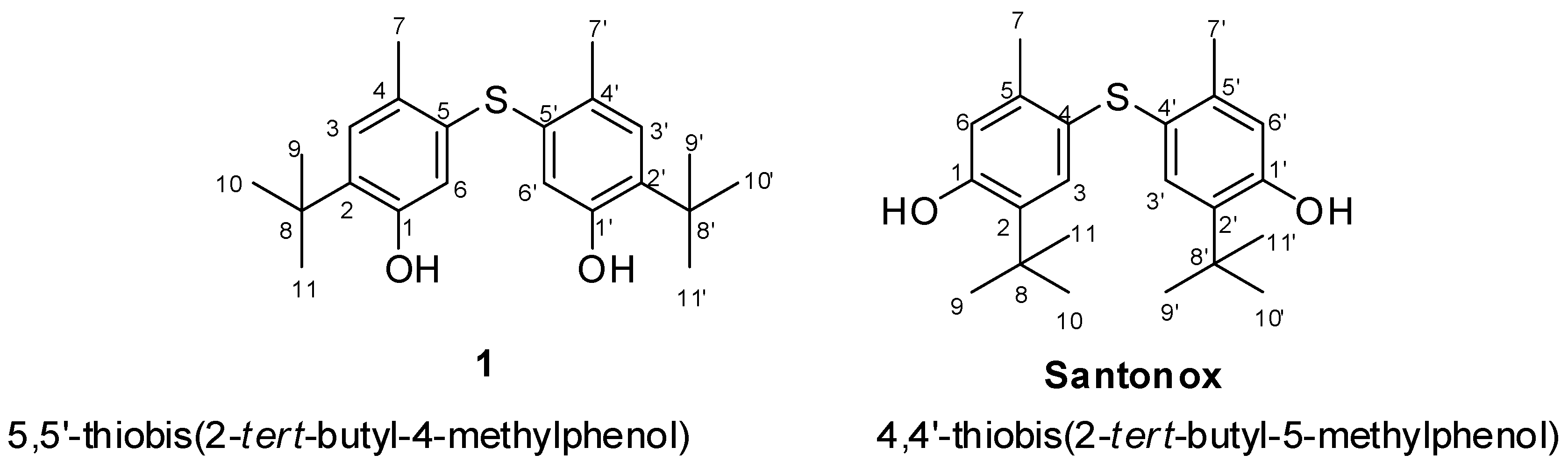
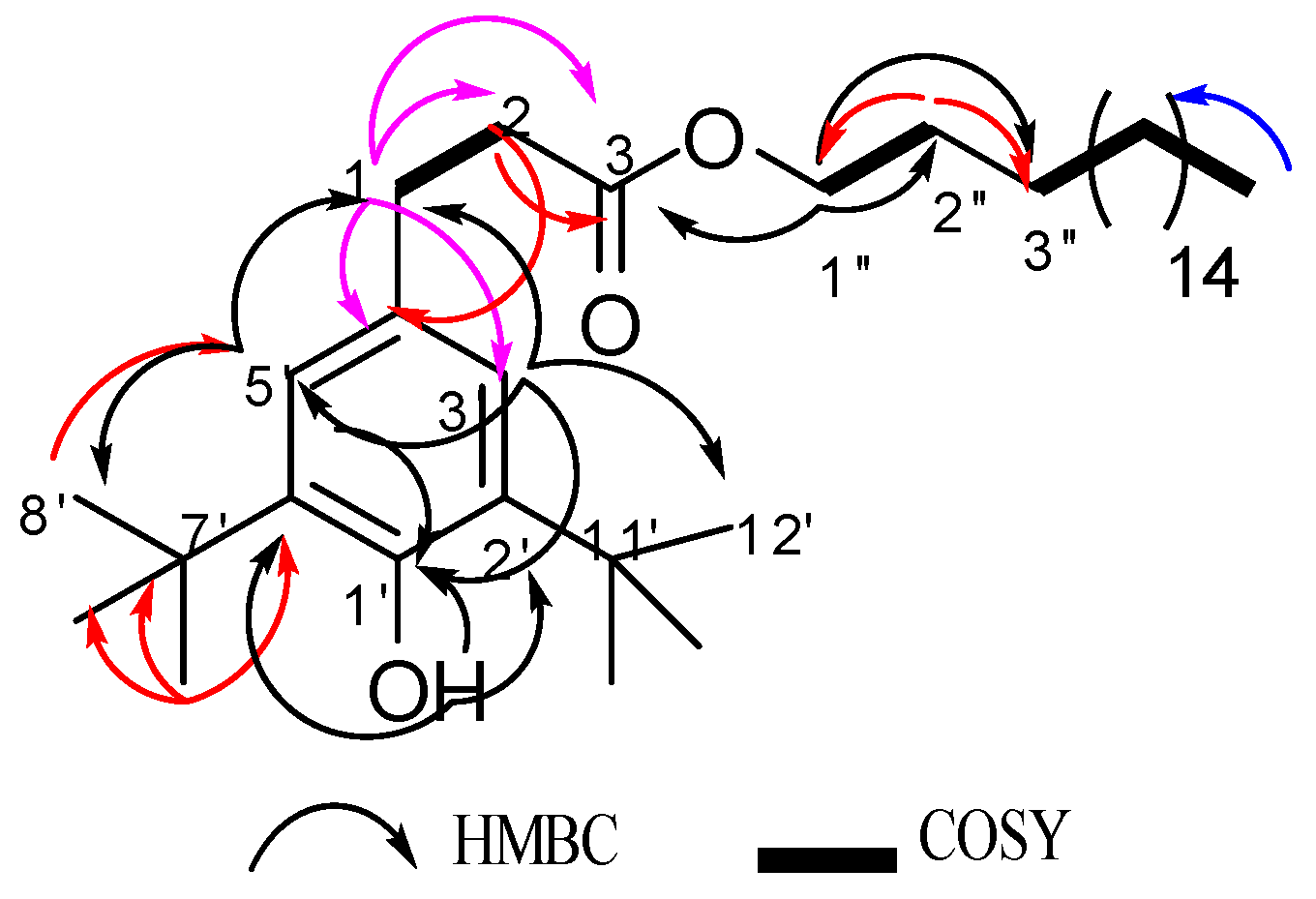
| Compound 1 | Standard Santonox | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Position | δC, ppm | δH, (ppm) mult. (J in Hz) | HMBC (H → C) | COSY | NOESY | Position | δC, ppm | δH, (ppm) mult. (J in Hz) | HMBC (H → C) | COSY | NOESY |
| 1/1′ | 153.4 | - | - | - | 1/1′ | 153.2 | - | - | - | - | |
| 2/2′ | 134.6 | - | - | - | 2/2′ | 134.5 | - | - | - | - | |
| 3/3′ | 130.8 | 7.00, s | 1, 4, 5, 8 | 7, 9/10/11 | 9/10/11 | 3/3′ | 130.6 | 7.00, s | 1, 4, 5, 6, 7, 8 | 7, 9/10/11 | 9/10/11 |
| 4/4′ | 125.5 | - | - | - | 4/4′ | 137.7 | - | - | - | - | |
| 5/5′ | 137.8 | - | - | - | 5/5′ | 125.4 | - | - | - | - | |
| 6/6′ | 118.8 | 6.56, s | 1, 2, 4, 7 | 7 | 6/6′ | 118.7 | 6.54, s | 1, 4, 5, 7, 8 | 7 | 7 | |
| 7/7′ | 19.8 | 2.28, s | 4, 5, 6 | 3, 6 | 7/7′ | 19.7 | 2.27, s | 4, 5, 6 | 3, 6 | 6 | |
| 8/8′ | 34.4 | - | - | 8/8′ | 34.3 | - | - | - | |||
| 9, 10, 11 /9′,10, 11′ | 29.7 | 1.28, s | 9/10/11, 2, 8 | 3 | 3 | 9, 10, 11/9′,10, 11′ | 29.5 | 1.28, s | 9/10/11, 2, 8 | 3 | 3 |
| OH | 4.72, br | - | OH | ||||||||
| Compound 2 | |||||
|---|---|---|---|---|---|
| Position | δC | Type | δH, mult. (J in Hz) | COSY | HMBC (H → C) |
| 1′ | 152.1 | C | - | - | |
| 2′ | 135.8 | C | - | - | |
| 3′ | 124.8 | CH | 6.99, s | 1′, 5′, 12′ | |
| 4′ | 131.1 | C | - | ||
| 5′ | 124.8 | CH | 6.99, s | 1′, 3′, 1, 8′ | |
| 6′ | 135.8 | C | - | ||
| 1 | 31.0 | CH2 | 2.85, dd (9.1, 6.9) | 2 | 3′/5′, 4′, 2, 3 |
| 2 | 36.5 | CH2 | 2.60, dd (9.1, 6.9) | 1 | 5′, 1, 3 |
| 3 | 173.4 | C | - | ||
| 1″ | 64.6 | CH2 | 4.07, t (6.8) | 2″ | 3, 2″, 3″ |
| 2″ | 29.7 | CH2 | 1.56–1.61, m | 1″, 3″ | 1″, 3″ |
| 3″ | 29.7 | CH2 | 1.56–1.61,m | 2″ | 2″ |
| 4″–17″ | 22.7–32.0 | CH2 | 1.24, m | 18″ | 5″–17″, 2″, 3″, 18″ |
| 18″ | 14.1 | CH3 | 0.88, t (6.7) | 17″ | 17″ |
| 7′/11′c | 34.3 | C | - | ||
| 8′,9′,10′/12′,13′,14′ a | 30.3 | CH3 | 1.43, s | 8′,9′,10′/12′,13′,14′, 7′/11′, 2′, 6′, 3′/5′ | |
| OH | 5.07, bs | 1′, 2′, 6′ | |||
| Compound | IC50 (µM) | |
|---|---|---|
| HaCaT | B16 | |
| 1 | 8.38 | 4.75 |
| 2 | >377.4 | 169.8 ± 1 |
| Doxorubicin | 0.096 ± 0.009 | 0.034 ± 0.001 |
| Concentration (µM) | γH2AX Foci/Nuclei |
|---|---|
| 0 | 12.9 ± 0.4 |
| 0.1 | 12.8 ± 0.2 |
| 1 | 3.6 ± 0.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.-B.-L.; Delalande, O.; Rouaud, I.; Ferron, S.; Chaillot, L.; Pedeux, R.; Tomasi, S. tert-Butylphenolic Derivatives from Paenibacillus odorifer—A Case of Bioconversion. Molecules 2018, 23, 1951. https://doi.org/10.3390/molecules23081951
Nguyen T-B-L, Delalande O, Rouaud I, Ferron S, Chaillot L, Pedeux R, Tomasi S. tert-Butylphenolic Derivatives from Paenibacillus odorifer—A Case of Bioconversion. Molecules. 2018; 23(8):1951. https://doi.org/10.3390/molecules23081951
Chicago/Turabian StyleNguyen, Thi-Bach-Le, Olivier Delalande, Isabelle Rouaud, Solenn Ferron, Laura Chaillot, Rémy Pedeux, and Sophie Tomasi. 2018. "tert-Butylphenolic Derivatives from Paenibacillus odorifer—A Case of Bioconversion" Molecules 23, no. 8: 1951. https://doi.org/10.3390/molecules23081951
APA StyleNguyen, T.-B.-L., Delalande, O., Rouaud, I., Ferron, S., Chaillot, L., Pedeux, R., & Tomasi, S. (2018). tert-Butylphenolic Derivatives from Paenibacillus odorifer—A Case of Bioconversion. Molecules, 23(8), 1951. https://doi.org/10.3390/molecules23081951