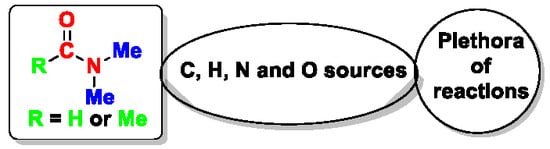
Recent Uses of N,N-Dimethylformamide and N,N-Dimethylacetamide as Reagents
Abstract
1. Introduction
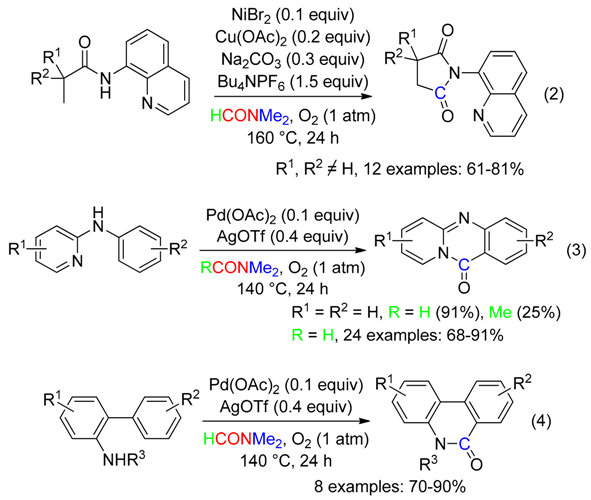
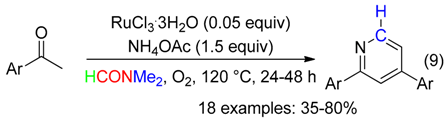
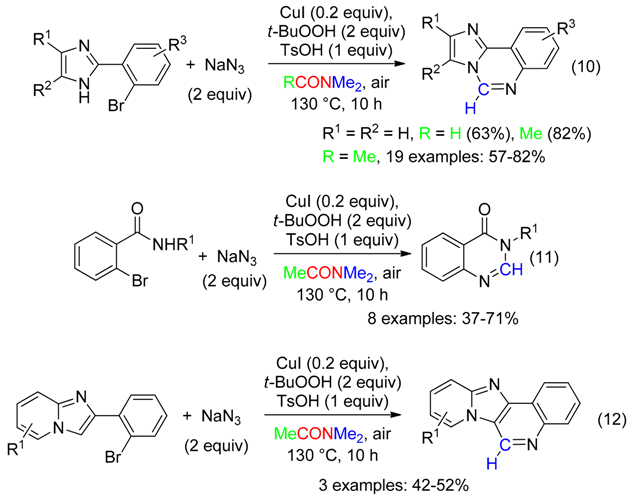
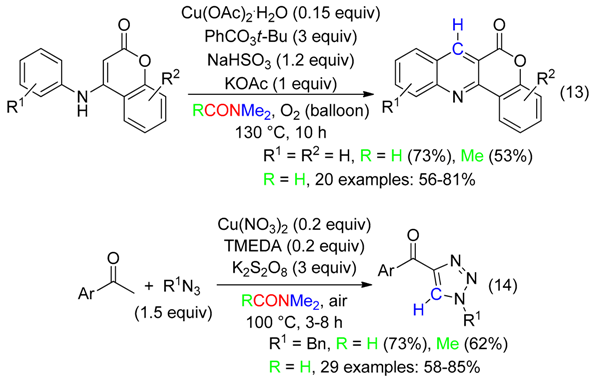
2. C Fragment


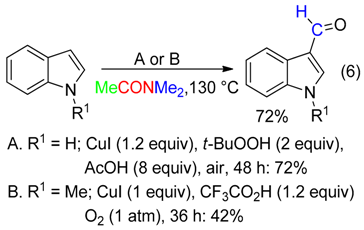
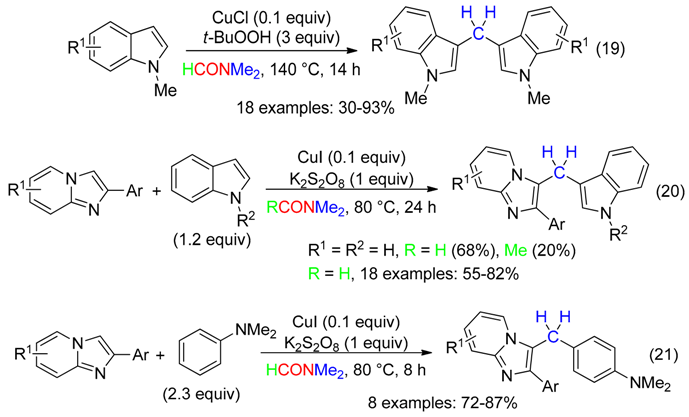
3. CH Fragment
- -
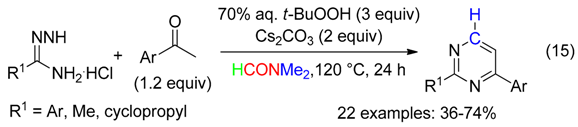
- pyrimidines from t-BuOOH-mediated reaction between acetophenones, amidines and DMF (Equation (15)) [47],
- -
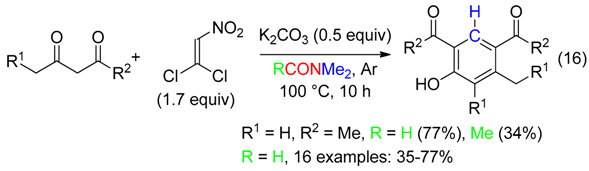
- substituted phenols also from three components cycloadditions (Equation (16)) [48],
- -
- 3-acylindoles from 2-alkenylanilines (Equation (17)) [49],
- -
- benzimidazoles and benzothiazole from o-phenylenediamine or 2-aminobenzenethiol through carbon dioxide-mediated cyclization (Equation (18)) [50].
4. CH2 Fragment
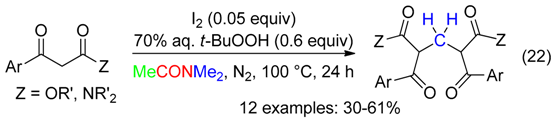
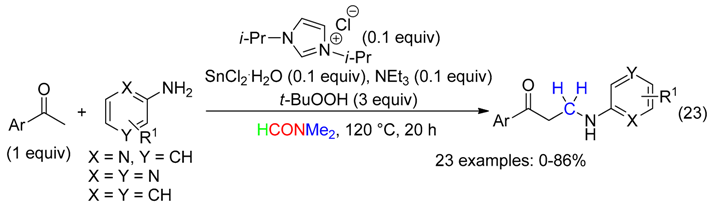
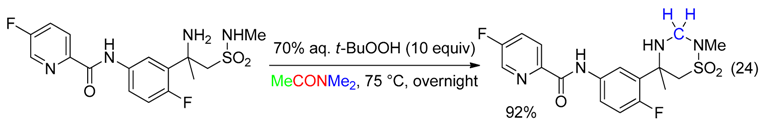
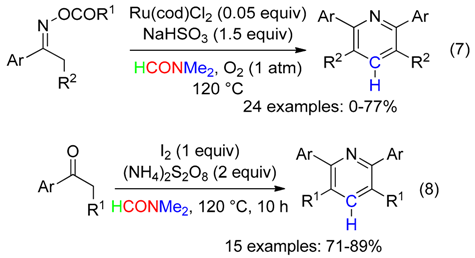
5. NC Fragment
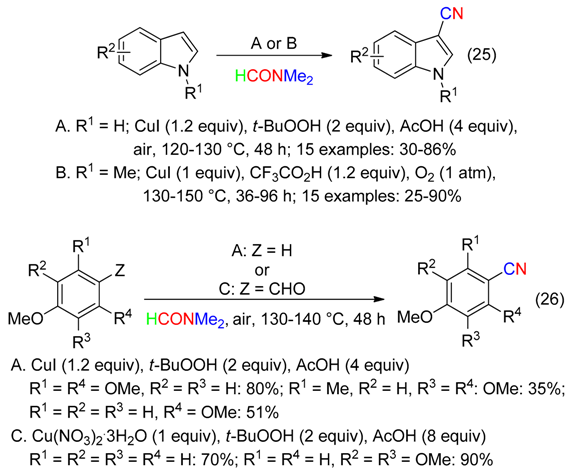

6. NMe2 Fragment
6.1. Aryl Halides
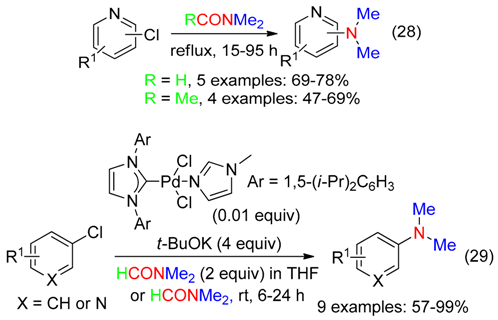
6.2. Alkylarenes
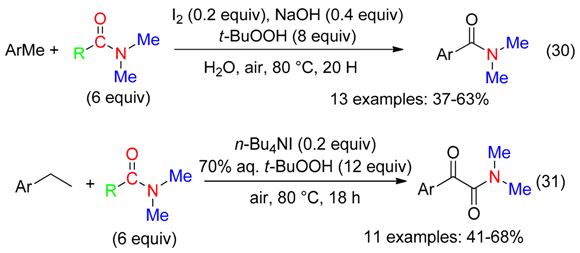
6.3. Alkenes

6.4. Acids
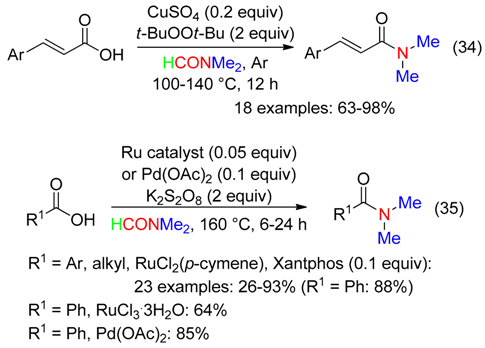
- -
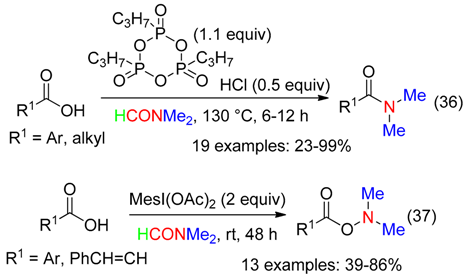
- the amidation of acids promoted with propylphosphonic anhydride associated to HCl at 130 °C (Equation (36)) [15],
- -
- the amination of acids employing a hypervalent iodine reagent at room temperature (Equation (37)) [35]. Mesityliodine diacetate was superior to the other hypervalent iodine reagents, while oxidants such as I2, t-BuOOH, NaIO4 or K2S2O8 did not mediate the amidation reaction [35].

6.5. Carbonylated Compounds
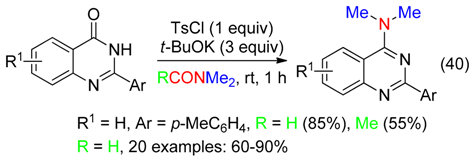
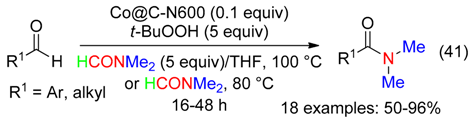
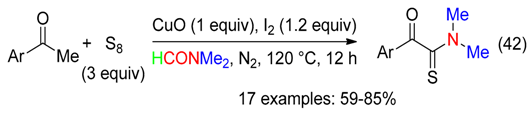

6.6. Benzyl Amines

6.7. Nitriles

6.8. Sulfur Compounds
7. O Fragment

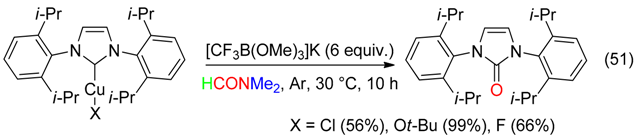

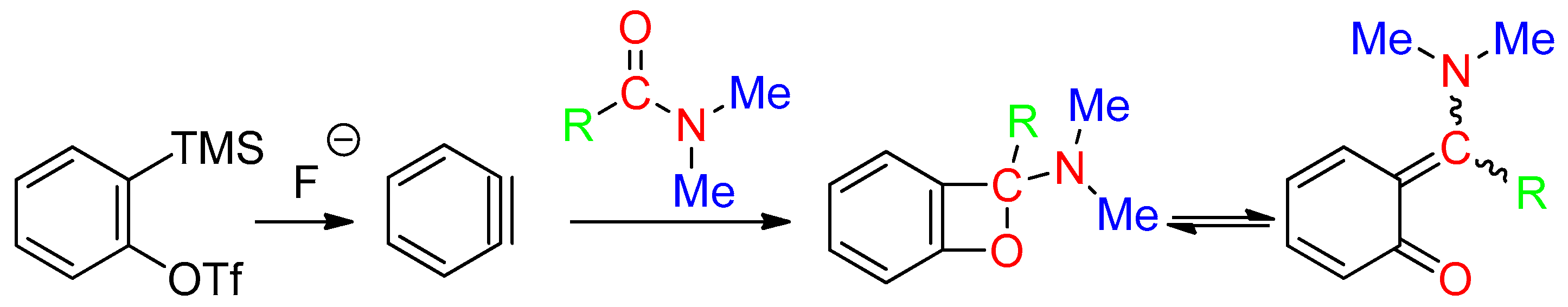
8. C=O Fragment
- -
- via C(sp2)-H bond activation and annulation using Pd/C [67] or Pd(OAc)2 [8], in the presence of K2S2O8, CF3CO2H and O2 (Equation (53)),or carbon dioxide-mediated cyclization of 2-aminobenzonitrile (Equation (54)) [50]. This latter reaction would involve a Vilsmeier-Haack type intermediate and did not occur with DMAc.
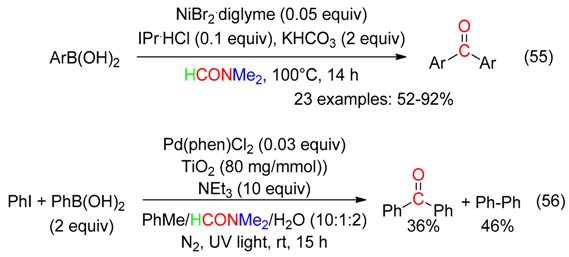
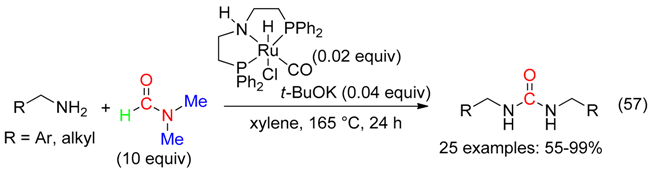

9. C=ONMe2 Fragment

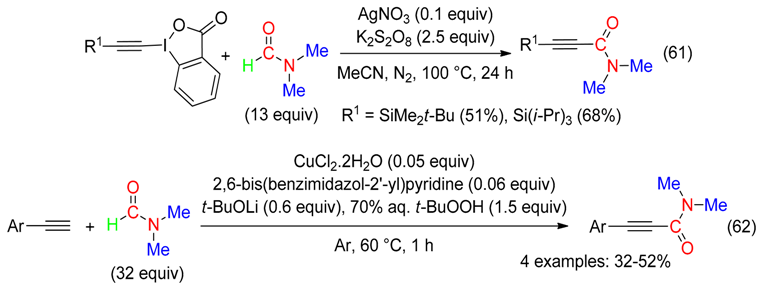

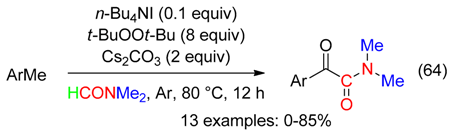

10. H Fragment
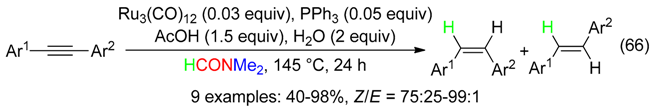
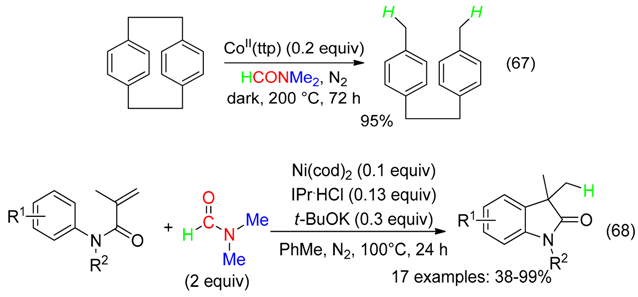
11. RC Fragment
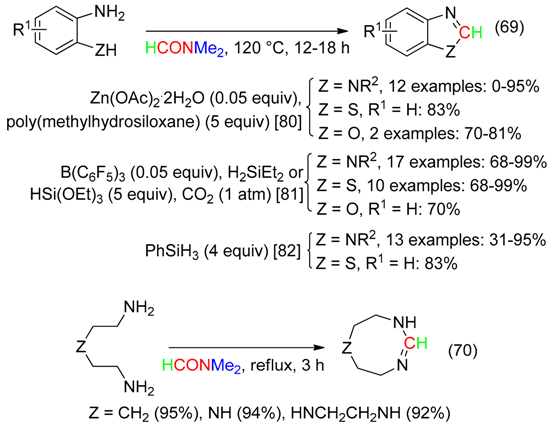
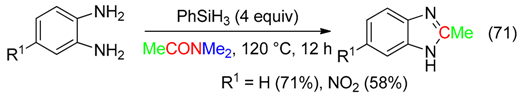

12. RCNMe2 Fragment
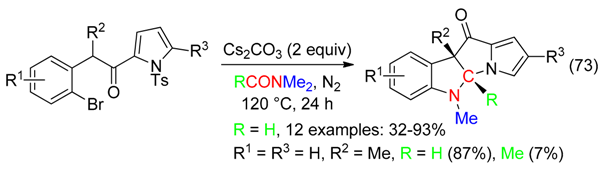
13. RC-O Fragment
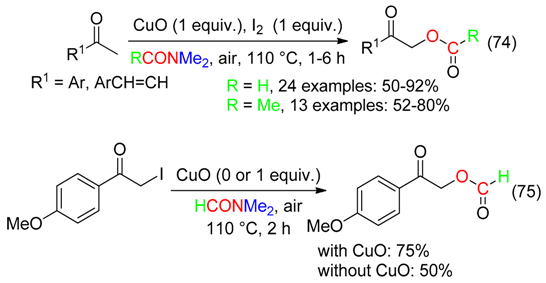




14. RC=O Fragment

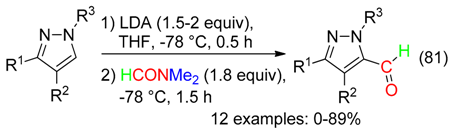
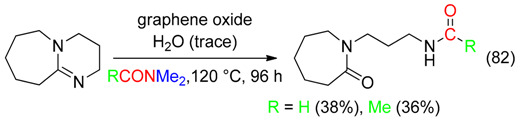
15. RC=ON(CH2)Me Fragment
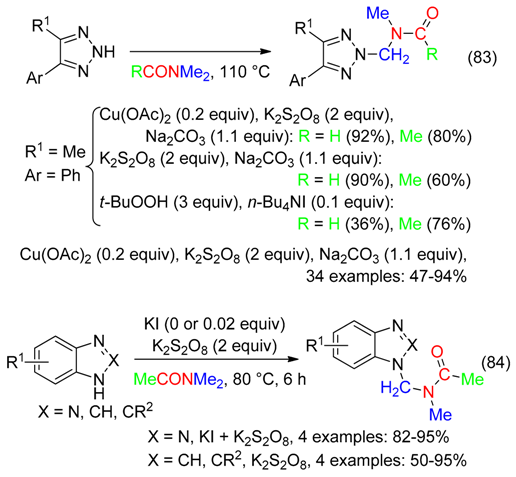
16. HC-ONMe2 Fragment

17. RC and O Fragment
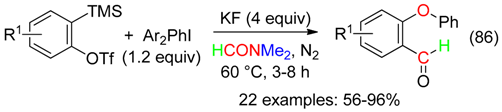
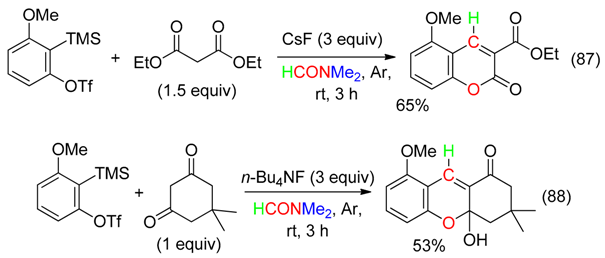
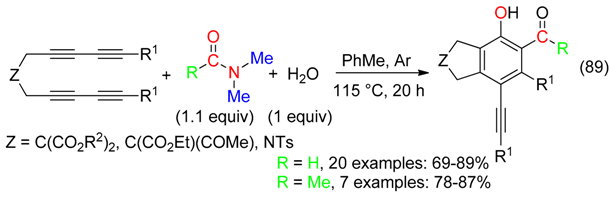
18. RCNMe2 and O Fragment
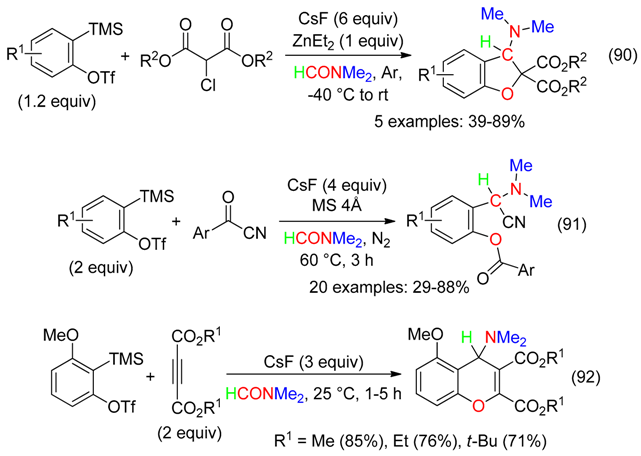
19. HC=O and HC Fragment

20. H and NMe2 Fragment
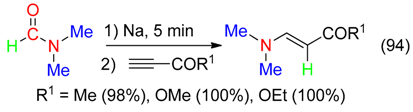
21. H and C=ONMe2 Fragment

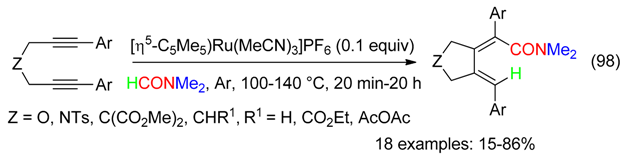
22. H, C=ONMe2 and NMe2 Fragment

23. C=ONMe2 and CH Fragment

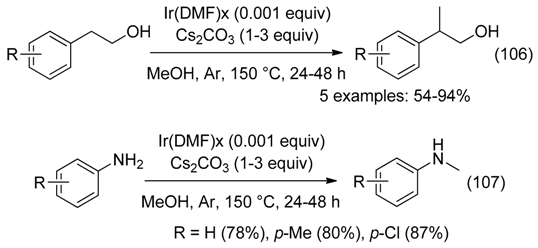
24. Reducing or Stabilizing Agent
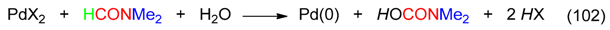
- -
- on iron for the hydrosilylation of alkenes (Equation (103)) [109],
- -
- on palladium for the synthesis of 2,3-disubstituted indoles from 2-halooanilines and alkynes (Equation (104)) [110],
- -
- on copper for Sonogashira–Hagihara cross-coupling reactions (Equation (105)) [111],
- -
- on iridium for methylation of alcohols (Equation (106)) and amines (Equation (107)), using methanol as the C1 source [112].

25. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Muzart, J. N,N-Dimethylformamide: Much more than a solvent. Tetrahedron 2009, 65, 8313–8323. [Google Scholar] [CrossRef]
- Ding, S.; Jiao, N. N,N-Dimethylformamide: A multipurpose building block. Angew. Chem. Int. Ed. 2012, 51, 9226–9231. [Google Scholar] [CrossRef] [PubMed]
- Batra, A.; Singh, P.; Singh, K.N. Cross dehydrogenative coupling (CDC) reactions of N,N-disubstituted formamides, benzaldehydes and cycloalkanes. Eur. J. Org. Chem. 2016, 2016, 4927–4947. [Google Scholar] [CrossRef]
- Le Bras, J.; Muzart, J. N,N-Dimethylformamide and N,N-dimethylacetamide as carbon, hydrogen, nitrogen and/or oxygen sources. In Solvents as Reagents in Organic Synthesis, Reactions and Applications; Wu, X.-F., Ed.; Wiley-VCH: Weinheim, Germany, 2017; pp. 199–314. [Google Scholar]
- Kolosov, M.A.; Shvets, E.H.; Manuenkov, D.A.; Vlasenko, S.A.; Omelchenko, I.V.; Shishkina, S.V.; Orlov, V.D. A synthesis of 6-functionalized 4,7-dihydro[1,2,4]triazolo[1,5-a]pyrimidines. Tetrahedron Lett. 2017, 58, 1207–1210. [Google Scholar] [CrossRef]
- Borah, A.; Goswami, L.; Neog, K.; Gogoi, P. DMF dimethyl acetal as carbon source for α-methylation of ketones: A hydrogenation-hydrogenolysis strategy of enaminones. J. Org. Chem. 2015, 80, 4722–4728. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Vanjari, R.; Guntreddi, T.; Singh, K.N. Sulfur promoted decarboxylative thioamidation of carboxylic acids using formamides as amine proxy. Tetrahedron 2016, 72, 2012–2017. [Google Scholar] [CrossRef]
- Chen, J.; Feng, J.-B.; Natte, K.; Wu, X.-F. Palladium-catalyzed carbonylative cyclization of arenes by C-H bond activation with DMF as the carbonyl source. Chem. Eur. J. 2015, 21, 16370–16373. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Zhu, Z.; Lu, L.; Zhang, W.; Gong, J.; Cai, H. Metal-free Csp3-N bond cleavage of amides using tert-butyl hydroperoxide as oxidant. Synlett 2015, 26, 543–546. [Google Scholar] [CrossRef]
- Zhu, Z.; Wang, Y.; Yang, M.; Huang, L.; Gong, J.; Guo, S.; Cai, H. A metal-free cross-dehydrogenative coupling reaction of amides to access N-alkylazoles. Synlett 2016, 27, 2705–2708. [Google Scholar]
- Alanthadka, A.; Devi, E.S.; Selvi, A.T.; Nagarajan, S.; Sridharan, V.; Maheswari, C.U. N-Heterocyclic carbene-catalyzed Mannich reaction for the synthesis of β-amino ketones: N,N-dimethylformamide as carbon source. Adv. Synth. Catal. 2017, 359, 2369–2374. [Google Scholar] [CrossRef]
- Zewge, D.; Bu, X.; Sheng, H.; Liu, Y.; Liu, Z.; Harman, B.; Reibarkh, M.; Gong, X. Mechanistic insight into oxidized N,N-dimethylacetamide as a source of formaldehyde related derivatives. React. Chem. Eng. 2018, 3, 146–150. [Google Scholar] [CrossRef]
- Li, Y.; Tu, D.-H.; Wang, B.; Lu, J.-Y.; Wang, Y.-Y.; Liu, Z.-T.; Liu, Z.-W.; Lu, J. Nickel-catalyzed carbonylation of arylboronic acids with DMF as CO source. Org. Chem. Front. 2017, 4, 569–572. [Google Scholar] [CrossRef]
- Lu, K.; Han, X.-W.; Yao, W.-W.; Luan, Y.-X.; Wang, Y.-X.; Chen, H.; Xu, X.-T.; Zhang, K.; Ye, M. DMF-promoted redox-neutral Ni-catalyzed intramolecular hydroarylation of alkene with simple arene. ACS Catal. 2018, 8, 3913–3917. [Google Scholar] [CrossRef]
- Bannwart, L.; Abele, S.; Tortoioli, S. Metal-free amidation of acids with formamides and T3P®. Synthesis 2016, 48, 2069–2078. [Google Scholar]
- Zhu, Y.-P.; Gao, Q.-H.; Lian, M.; Yuan, J.-J.; Liu, M.-C.; Zhao, Q.; Yang, Y.; Wu, A.-X. A sustainable byproduct catalyzed domino strategy: Facile synthesis of α-formyloxy and acetoxy ketones via iodination/nucleophilic substitution/hydrolyzation/oxidation sequences. Chem. Commun. 2011, 47, 12700–12702. [Google Scholar] [CrossRef] [PubMed]
- Mata, L.; Avenoza, A.; Busto, J.H.; Peregrina, J.M. Chemoselectivity control in the reactions of 1,2-cyclic sulfamidates with amines. Chem. Eur. J. 2013, 19, 6831–6839. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, C.; Zhou, P. N,N-Dimethylformamide (DMF) as a source of oxygen to access α-hydroxy arones via the α-hydroxylation of arones. J. Org. Chem. 2017, 82, 2219–2222. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Yang, H.; Hu, X.; Jiang, G. Metal-free synthesis of ortho-CHO diaryl ethers by a three-component sequential coupling. Org. Lett. 2014, 16, 6408–6411. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, E.; Kohtani, S.; Miyabe, H. Three-component coupling reactions of arynes for the synthesis of benzofurans and coumarins. Molecules 2014, 19, 863–880. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, E.; Kohtani, S.; Miyabe, H. 2,3,4,9-Tetrahydro-9-(3-hydroxy-1,4-dioxo-1H-dihydronaphthalen-2-yl)-8-methoxy-3,3-dimethyl-1H-xanthen-1-one. Molbank 2015, 2015, M841. [Google Scholar] [CrossRef]
- Yoshioka, E.; Tanaka, H.; Kohtani, S.; Miyabe, H. Straightforward synthesis of dihydrobenzofurans and benzofurans from arynes. Org. Lett. 2013, 15, 3938–3941. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Wang, J.; Jin, J.; Lu, P.; Wang, Y. Three-component synthesis of α-amino-α-aryl carbonitriles from arynes, aroyl cyanides, and N,N-dimethylformamide. Eur. J. Org. Chem. 2014, 2014, 1832–1835. [Google Scholar] [CrossRef]
- Yoshioka, E.; Tamenaga, H.; Miyabe, H. [4+2] Cycloaddition of intermediates generated from arynes and DMF. Tetrahedron Lett. 2014, 55, 1402–1405. [Google Scholar] [CrossRef]
- Kodimuthali, A.; Mungara, A.; Prasunamba, P.L.; Pal, M. A simple synthesis of aminopyridines: Use of amides as amine source. J. Braz. Chem. Soc. 2010, 21, 1439–1445. [Google Scholar] [CrossRef]
- Zhang, Q.; Song, C.; Huang, H.; Zhang, K.; Chang, J. Cesium carbonate promoted cascade reaction involving DMF as a reactant for the synthesis of dihydropyrrolizino[3,2-b]indol-10-ones. Org. Chem. Front. 2018, 5, 80–87. [Google Scholar] [CrossRef]
- Chen, W.-X.; Shao, L.-X. N-Heterocyclic carbene–palladium(II)-1-methylimidazole complex catalyzed amination between aryl chlorides and amides. J. Org. Chem. 2012, 77, 9236–9239. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.-J.; Sun, Q.; Rastogi, N.; Koenig, B. Intermolecular formyloxyarylation of alkenes by photoredox Meerwein reaction. ACS Catal. 2015, 5, 2935–2938. [Google Scholar] [CrossRef]
- Yan, H.; Yang, H.; Lu, L.; Liu, D.; Rong, G.; Mao, J. Copper-catalyzed synthesis of α,β-unsaturated acylamides via direct amidation from cinnamic acids and N-substituted formamides. Tetrahedron 2013, 69, 7258–7263. [Google Scholar] [CrossRef]
- Salamone, M.; Milan, M.; DiLabio, G.A.; Bietti, M. Reactions of the cumyloxyl and benzyloxyl radicals with tertiary amides. Hydrogen abstraction selectivity and the role of specific substrate-radical hydrogen bonding. J. Org. Chem. 2013, 78, 5909–5917. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Sun, P. Syntheses of amides via iodine-catalyzed multiple sp3 C-H bonds oxidation of methylarenes and sequential coupling with N,N-dialkylformamides. Sci. China Chem. 2014, 57, 1176–1182. [Google Scholar] [CrossRef]
- Du, B.; Jin, B.; Sun, P. The syntheses of α-ketoamides via n-Bu4NI-catalyzed multiple sp3C-H bond oxidation of ethylarenes and sequential coupling with dialkylformamides. Org. Biomol. Chem. 2014, 12, 4586–4589. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.; Yao, X.; Li, Y. Easy access to amides through aldehydic C-H bond functionalization catalyzed by heterogeneous Co-based catalysts. ACS Catal. 2015, 5, 884–891. [Google Scholar] [CrossRef]
- Wu, J.-J.; Li, Y.; Zhou, H.-Y.; Wen, A.-H.; Lun, C.-C.; Yao, S.-Y.; Ke, Z.; Ye, B.-H. Copper-catalyzed carbamoylation of terminal alkynes with formamides via cross-dehydrogenative coupling. ACS Catal. 2016, 6, 1263–1267. [Google Scholar] [CrossRef]
- Zhang, C.; Yue, Q.; Xiao, Z.; Wang, X.; Zhang, Q.; Li, D. Synthesis of O-aroyl-N,N-dimethylhydroxylamines through hypervalent iodine-mediated amination of carboxylic acids with N,N-dimethylformamide. Synthesis 2017, 49, 4303–4308. [Google Scholar]
- Wu, X.; Zhao, Y.; Ge, H. Direct aerobic carbonylation of C(sp2)-H and C(sp3)-H bonds through Ni/Cu synergistic catalysis with DMF as the carbonyl source. J. Am. Chem. Soc. 2015, 137, 4924–4927. [Google Scholar] [CrossRef] [PubMed]
- Rao, D.N.; Rasheed, S.; Das, P. Palladium/silver synergistic catalysis in direct aerobic carbonylation of C(sp2)–H bonds using DMF as a carbon source: Synthesis of pyrido-fused quinazolinones and phenanthridinones. Org. Lett. 2016, 18, 3142–3145. [Google Scholar]
- Boosa, V.; Bilakanti, V.; Gutta, N.; Velisoju, V.K.; Medak, S.; Ramineni, K.; Jorge, B.; Muxina, K.; Akula, V. C–H bond cyanation of arenes using N,N-dimethylformamide and NH4HCO3 as a CN source over a hydroxyapatite supported copper catalyst. Catal. Sci. Technol. 2016, 6, 8055–8062. [Google Scholar]
- Zhang, L.; Lu, P.; Wang, Y. Copper-mediated cyanation of indoles and electron-rich arenes using DMF as a single surrogate. Org. Biomol. Chem. 2015, 13, 8322–8329, Correction in Org. Biomol. Chem. 2016, 14, 1840. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Li, Q.; Chen, T.; Han, L.-B. Copper-mediated selective aerobic oxidative C3-cyanation of indoles with DMF. Tetrahedron Lett. 2015, 56, 5937–5940. [Google Scholar] [CrossRef]
- Zhao, M.-N.; Hui, R.-R.; Ren, Z.-H.; Wang, Y.-Y.; Guan, Z.-H. Ruthenium-catalyzed cyclization of ketoxime acetates with DMF for synthesis of symmetrical pyridines. Org. Lett. 2014, 16, 3082–3085. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Tan, H.; Chen, C.; Pan, Y. A method to access symmetrical tetrasubstituted pyridines via iodine and ammonium persulfate mediated [2+2+1+1]-cycloaddition reaction. Adv. Synth. Catal. 2017, 359, 1594–1598. [Google Scholar] [CrossRef]
- Bai, Y.; Tang, L.; Huang, H.; Deng, G.-J. Synthesis of 2,4-diarylsubstituted-pyridines through a Ru-catalyzed four component reaction. Org. Biomol. Chem. 2015, 13, 4404–4407. [Google Scholar] [CrossRef] [PubMed]
- Nandwana, N.K.; Dhiman, S.; Saini, H.K.; Kumar, I.; Kumar, A. Synthesis of quinazolinones, imidazo[1,2-c]quinazolines and imidazo[4,5-c]quinolines through tandem reductive amination of aryl halides and oxidative amination of C(sp3)–H bonds. Eur. J. Org. Chem. 2017, 2017, 514–522. [Google Scholar] [CrossRef]
- Weng, Y.; Zhou, H.; Sun, C.; Xie, Y.; Su, W. Copper-catalyzed cyclization for access to 6H-chromeno[4,3-b]quinolin-6-ones employing DMF as the carbon source. J. Org. Chem. 2017, 82, 9047–9053. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Nie, G.; Zhou, Z.; Jia, L.; Chen, Y. Copper-catalyzed oxidative cross-dehydrogenative coupling/oxidative cycloaddition: Synthesis of 4-acyl-1,2,3-triazoles. J. Org. Chem. 2017, 82, 9198–9203. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.-Y.; Guo, W.; Fan, X.-L. Metal-free, TBHP-mediated, [3+2+1]-type intermolecular cycloaddition reaction: Synthesis of pyrimidines from amidines, ketones, and DMF through C(sp3)–H activation. Asian J. Org. Chem. 2017, 6, 837–840. [Google Scholar] [CrossRef]
- Wang, F.; Wu, W.; Xu, X.; Shao, X.; Li, Z. Synthesis of substituted phenols via 1,1-dichloro-2-nitroethene promoted condensation of carbonyl compounds with DMF. Tetrahedron Lett. 2018, 59, 2506–2510. [Google Scholar] [CrossRef]
- Wang, J.-B.; Li, Y.-L.; Deng, J. Metal-free activation of DMF by dioxygen: A cascade multiple-bond-formation reaction to synthesize 3-acylindoles from 2-alkenylanilines. Adv. Synth. Catal. 2017, 359, 3460–3467. [Google Scholar] [CrossRef]
- Rasal, K.B.; Yadav, G.D. Carbon dioxide mediated novel synthesis of quinazoline-2,4(1H,3H)-dione in water. Org. Process Res. Dev. 2016, 20, 2067–2073. [Google Scholar] [CrossRef]
- Pu, F.; Li, Y.; Song, Y.-H.; Xiao, J.; Liu, Z.-W.; Wang, C.; Liu, Z.-T.; Chen, J.-G.; Lu, J. Copper-catalyzed coupling of indoles with dimethylformamide as a methylenating reagent. Adv. Synth. Catal. 2016, 358, 539–542. [Google Scholar] [CrossRef]
- Mondal, S.; Samanta, S.; Santra, S.; Bagdi, A.K.; Hajra, A. N,N-Dimethylformamide as a methylenating reagent: Synthesis of heterodiarylmethanes via copper-catalyzed coupling between imidazo[1,2-a]pyridines and indoles/N,N-dimethylaniline. Adv. Synth. Catal. 2016, 358, 3633–3641. [Google Scholar] [CrossRef]
- Lee, S.; Jung, H.J.; Choi, H.C.; Hwang, Y.S.; Choi, M.Y. Solvent acting as a precursor: Synthesis of AgCN from AgNO3 in N,N-DMF solvent by laser ablation. Bull. Korean Chem. Soc. 2017, 38, 136–139. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, G.; Gao, B.; Huang, H. Palladium-catalyzed hydrocarbonylative C-N coupling of alkenes with amides. Org. Lett. 2018, 20, 2208–2212. [Google Scholar] [CrossRef] [PubMed]
- Bi, X.; Li, J.; Shi, E.; Wang, H.; Gao, R.; Xiao, J. Ru-catalyzed direct amidation of carboxylic acids with N-substituted formamides. Tetrahedron 2016, 72, 8210–8214. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Li, P.; Huang, Q.-Q.; Zhang, J.-J.; Itoh, S. Dioxygenation of flavonol catalyzed by copper(II) complexes supported by carboxylate-containing ligands: Structural and functional models of quercetin 2,4-dioxygenase. Eur. J. Inorg. Chem. 2017, 2017, 1845–1854. [Google Scholar] [CrossRef]
- Chen, X.; Yang, Q.; Zhou, Y.; Deng, Z.; Mao, X.; Peng, Y. Synthesis of 4-(dimethylamino)quinazoline via direct amination of quinazolin-4(3H)-one using N,N-dimethylformamide as a nitrogen source at room temperature. Synthesis 2015, 47, 2055–2062. [Google Scholar] [CrossRef]
- Gupta, S.S.R.; Nakhate, A.V.; Rasal, K.B.; Deshmukh, G.P.; Mannepalli, L.K. Oxidative amidation of benzaldehydes and benzylamines with N-substituted formamides over a Co/Al hydrotalcite-derived catalyst. New J. Chem. 2017, 41, 15268–15276. [Google Scholar] [CrossRef]
- Liu, W.; Chen, C.; Zhou, P. Concise access to α-arylketothioamides by redox reaction between acetophenones, elemental sulfur and DMF. ChemistrySelect 2017, 2, 5532–5535. [Google Scholar] [CrossRef]
- Liu, W.; Chen, C.; Liu, H. Dimethylamine as the key intermediate generated in situ from dimethylformamide (DMF) for the synthesis of thioamides. Beilstein J. Org. Chem. 2015, 11, 1721–1726. [Google Scholar] [CrossRef] [PubMed]
- Amupitan, J.O. An extension of the Willgerodt-Kindler reaction. Synthesis 1983, 1983, 730. [Google Scholar] [CrossRef]
- Nooshabadi, M.; Aghapoor, K.; Darabi, H.R.; Mojtahedi, M.M. The rapid synthesis of thiomorpholides by Willgerodt-Kindler reaction under microwave heating. Tetrahedron Lett. 1999, 40, 7549–7552. [Google Scholar] [CrossRef]
- Wei, E.; Liu, B.; Lin, S.; Liang, F. Multicomponent reaction of chalcones, malononitrile and DMF leading to γ-ketoamides. Org. Biomol. Chem. 2014, 12, 6389–6392. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, J.; Ni, Z.; Wang, S.; Pan, Y. Copper-mediated S–N formation via an oxygen-activated radical process: A new synthesis method for sulfonamides. Chem. Commun. 2014, 50, 4582–4584. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Rong, X.; Liu, Z.; Gu, Y.; Liang, G.; Xia, Q. Potassium tert-butoxide-mediated metal-free synthesis of sulfonamides from sodium sulfinates and N,N-disubstituted formamides. Tetrahedron Lett. 2018, 59, 2853–2858. [Google Scholar] [CrossRef]
- Zeng, W.; Wang, E.Y.; Qiu, R.; Sohail, M.; Wu, S.; Chen, F.X. Oxygen-atom insertion of NHC-copper complex: The source of oxygen from N,N-dimethylformamide. J. Organomet. Chem. 2013, 743, 44–48. [Google Scholar] [CrossRef]
- Chen, J.; Natte, K.; Wu, X.-F. Pd/C-catalyzed carbonylative C-H activation with DMF as the CO source. Tetrahedron Lett. 2015, 56, 6413–6416. [Google Scholar] [CrossRef]
- Feng, X.; Li, Z. Photocatalytic promoting dimethylformamide (DMF) decomposition to in-situ generation of self-supplied CO for carbonylative Suzuki reaction. J. Photochem. Photobiol. A Chem. 2017, 337, 19–24. [Google Scholar] [CrossRef]
- Krishnakumar, V.; Chatterjee, B.; Gunanathan, C. Ruthenium-catalyzed urea synthesis by N-H activation of amines. Inorg. Chem. 2017, 56, 7278–7284. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, X.; Zeng, W.; Huang, H.; Liang, Y. Copper catalyzed three-component synthesis of benzothiazolones from o-iodoanilines, DMF, and potassium sulfide. RSC Adv. 2014, 4, 6090–6093. [Google Scholar] [CrossRef]
- Mete, T.B.; Singh, A.; Bhat, R.G. Transition-metal-free synthesis of primary to tertiary carboxamides: A quick access to prodrug-pyrazinecarboxamide. Tetrahedron Lett. 2017, 58, 4709–4712. [Google Scholar] [CrossRef]
- Yao, B.; Deng, C.-L.; Liu, Y.; Tang, R.-Y.; Zhang, X.-G.; Li, J.-H. Palladium-catalyzed oxidative carbamoylation of isoquinoline N-oxides with formylamides by means of dual C-H oxidative coupling. Chem. Commun. 2015, 51, 4097–4100. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Guo, L.-N.; Wang, S.; Duan, X.-H. Decarboxylative alkynylation of α-keto acids and oxamic acids in aqueous media. Org. Lett. 2015, 17, 3054–3057. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.J.; Mir, B.A.; Patel, B.K. A TBPB-mediated C-3 cycloalkylation and formamidation of 4-arylcoumarin. Eur. J. Org. Chem. 2018, 2018, 1026–1033. [Google Scholar] [CrossRef]
- Fan, W.; Shi, D.; Feng, B. TBAI-catalyzed synthesis of α-ketoamides via sp3 C-H radical/radical cross-coupling and domino aerobic oxidation. Tetrahedron Lett. 2015, 56, 4638–4641. [Google Scholar] [CrossRef]
- Chen, W.-T.; Bao, W.-H.; Ying, W.-W.; Zhu, W.M.; Liang, H.; Wei, W.-T. Copper-promoted tandem radical reaction of 2-oxindoles with formamides: Facile synthesis of unsymmetrical urea derivatives. Asian J. Org. Chem. 2018, 7, 1057–1060. [Google Scholar] [CrossRef]
- Li, J.; Hua, R. Stereodivergent ruthenium-catalyzed transfer semihydrogenation of diaryl alkynes. Chem. Eur. J. 2011, 17, 8462–8465. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hua, R.; Liu, T. Highly chemo- and stereoselective palladium-catalyzed transfer semihydrogenation of internal alkynes affording cis-alkenes. J. Org. Chem. 2010, 75, 2966–2970. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.M.; To, C.T.; Chan, K.S. Carbon-carbon σ-bond transfer hydrogenation with DMF catalyzed by cobalt porphyrins. Organometallics 2016, 35, 2174–2177. [Google Scholar] [CrossRef]
- Nale, D.B.; Bhanage, B.M. N-substituted formamides as C1-sources for the synthesis of benzimidazole and benzothiazole derivatives by using zinc catalysts. Synlett 2015, 26, 2835–2842. [Google Scholar] [CrossRef]
- Gao, X.; Yu, B.; Mei, Q.; Yang, Z.; Zhao, Y.; Zhang, H.; Hao, L.; Liu, Z. Atmospheric CO2 promoted synthesis of N-containing heterocycles over B(C6F5)3 catalyst. New J. Chem. 2016, 40, 8282–8287. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, Z.; Miao, C.; Liu, W.; Sun, W. Synthesis of benzimidazoles from o-phenylenediamines and DMF derivatives in the presence of PhSiH3. Tetrahedron 2017, 73, 3458–3462. [Google Scholar] [CrossRef]
- Simion, C.; Gherase, D.; Sima, S.; Simion, A.M. Serendipitous synthesis of a cyclic formamidine. Application to the synthesis of polyazamacrocycles. C. R. Chim. 2015, 18, 611–613. [Google Scholar] [CrossRef]
- Kobayashi, E.; Togo, H. Facile one-pot preparation of 5-aryltetrazoles and 3-arylisoxazoles from aryl bromides. Tetrahedron 2018, 74, 4226–4235. [Google Scholar] [CrossRef]
- Ochiai, H.; Niwa, T.; Hosoya, T. Stereoinversion of stereocongested carbocyclic alcohols via triflylation and subsequent treatment with aqueous N,N-dimethylformamide. Org. Lett. 2016, 18, 5982–5985. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Zhang, Q.; Wang, J.; Huang, P.; Yan, P.; Zhang, R.; Dong, D. Triflic anhydride mediated ring-opening/recyclization reaction of α-carbamoyl α-oximyl cyclopropanes with DMF: Synthetic route to 5-aminoisoxazoles. Synthesis 2016, 48, 1934–1938. [Google Scholar] [CrossRef]
- Liu, L.; Zhang-Negrerie, D.; Du, F.; Zhao, K. PhICl2 and wet DMF: An efficient system for regioselective chloroformyloxylation/α-chlorination of alkenes/α,β-unsaturated compounds. Org. Lett. 2014, 16, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, J.; Liu, J.; Zhang, C.; Zhang, Z.; Xu, J.; Xu, S.; Wang, F.; Wang, F. C-N and N-H bond metathesis reactions mediated by carbon dioxide. ChemSusChem 2015, 8, 2066–2072. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, F.; Zhang, C.; Zhang, J.; Li, M.; Xu, J. Transformylating amine with DMF to formamide over CeO2 catalyst. Chem. Commun. 2014, 50, 2438–2441. [Google Scholar] [CrossRef] [PubMed]
- Sheng, H.; Zeng, R.; Wang, W.; Luo, S.; Feng, Y.; Liu, J.; Chen, W.; Zhu, M.; Guo, Q. An efficient heterobimetallic lanthanide alkoxide catalyst for transamidation of amides under solvent-free conditions. Adv. Synth. Catal. 2017, 359, 302–313. [Google Scholar] [CrossRef]
- Sonawane, R.B.; Rasal, N.K.; Jagtap, S.V. Nickel-(II)-catalyzed N-formylation and N-acylation of amines. Org. Lett. 2017, 19, 2078–2081. [Google Scholar] [CrossRef] [PubMed]
- Lindsay-Scott, P.J.; Charlesworth, N.G.; Grozavu, A. A flexible strategy for the regiocontrolled synthesis of pyrazolo[1,5-a]pyrazines. J. Org. Chem. 2017, 82, 11295–11303. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa-Goto, K.; Kobayashi, T.; Kawasaki, T.; Somei, M. Lithiation of 1-alkoxyindole derivatives. Heterocycles 2018. [Google Scholar] [CrossRef]
- Ramírez-Jiménez, R.; Franco, M.; Rodrigo, E.; Sainz, R.; Ferritto, R.; Lamsabhi, A.M.; Aceña, J.L.; Cid, M.B. Unexpected reactivity of graphene oxide with DBU and DMF. J. Mater. Chem. A 2018, 6, 12637–12646. [Google Scholar] [CrossRef]
- Deng, X.; Lei, X.; Nie, G.; Jia, L.; Li, Y.; Chen, Y. Copper-catalyzed cross-dehydrogenative N2-coupling of NH-1,2,3-triazoles with N,N-dialkylamides: N-amidoalkylation of NH-1,2,3-triazoles. J. Org. Chem. 2017, 82, 6163–6171. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, P.E.; Sharma, H.K.; Chakrabarty, S.; Metta-Magaña, A.; Pannell, K.H. Triethylsiloxymethyl-N,N-dimethylamine, Et3SiOCH2NMe2: A dimethylaminomethylation (Mannich) reagent for O–H, S–H, P–H and aromatic C–H systems. Eur. J. Org. Chem. 2017, 2017, 5610–5616. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, Y.; Hu, Q.; Ma, J.; Lv, S.; Liu, B.; Wang, S. Direct access to fused salicylaldehydes and salicylketones from tetraynes. Chem. Eur. J. 2017, 23, 4065–4072. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Liu, Z.; Chen, J.; Xiong, X.; Wang, Y.; Huang, L.; Bai, J.; Dang, Y.; Huang, J. Preparation of thioanisole biscarbanion and C–H lithiation/annulation reactions for the access of five-membered heterocycles. Org. Lett. 2018, 20, 3161–3165. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Sheng, H.; Rao, B.; Feng, Y.; Wang, H.; Sun, Y.; Chen, M.; Zhu, M. An efficient and green approach to synthesizing enamines by intermolecular hydroamination of activated alkynes. Chem. Res. Chin. Univ. 2015, 31, 212–217. [Google Scholar] [CrossRef]
- Liu, D.; Mao, J.; Rong, G.; Yan, H.; Zheng, Y.; Chen, J. Concise synthesis of semicarbazides and formylhydrazines via direct addition reaction between aromatic azoarenes and N-substituted formamides. RSC Adv. 2015, 5, 19301–19305. [Google Scholar] [CrossRef]
- Mori, S.; Shibuya, M.; Yamamoto, Y. Ruthenium-catalyzed hydrocarbamoylative cyclization of 1,6-diynes with formamides. Chem. Lett. 2017, 46, 207–210. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Okude, Y.; Mori, S.; Shibuya, M. Combined experimental and computational study on ruthenium(II)-catalyzed reactions of diynes with aldehydes and N,N-dimethylformamide. J. Org. Chem. 2017, 82, 7964–7973. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y. Theoretical study on ruthenium-catalyzed hydrocarbamoylative cyclization of 1,6-diyne with dimethylformamide. Organometallics 2017, 36, 1154–1163. [Google Scholar] [CrossRef]
- Adams, R.D.; Dhull, P. Formyl C-H activation in N,N-dimethylformamide by a dirhenium carbonyl complex. J. Organomet. Chem. 2017, 849–850, 228–232. [Google Scholar] [CrossRef]
- Guo, W.; Liao, J.; Liu, D.; Li, J.; Ji, F.; Wu, W.; Jiang, H. A four-component reaction strategy for pyrimidine carboxamide synthesis. Angew. Chem. Int. Ed. 2017, 56, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Pastoriza-Santos, I.; Liz-Marzán, L.M. N,N-Dimethylformamide as a reaction medium for metal nanoparticle synthesis. Adv. Funct. Mater. 2009, 19, 679–688. [Google Scholar] [CrossRef]
- Akbari, S.; Mokhtari, J.; Mirjafari, Z. Solvent-free and melt aerobic oxidation of benzyl alcohols using Pd/Cu2(BDC)2DABCO–MOF prepared by one-step and through reduction by dimethylformamide. RSC Adv. 2017, 7, 40881–40886. [Google Scholar] [CrossRef]
- Tahmasebi, S.; Mokhtari, J.; Naimi-Jamal, M.R.; Khosravi, A.; Panahi, L. One-step synthesis of Pd-NPs@Cu2(BDC)2DABCO as efficient heterogeneous catalyst for the Suzuki-Miyaura cross-coupling reaction. J. Organomet. Chem. 2017, 853, 35–41. [Google Scholar] [CrossRef]
- Azuma, R.; Nakamichi, S.; Kimura, J.; Yano, H.; Kawasaki, H.; Suzuki, T.; Kondo, R.; Kanda, Y.; Shimizu, K.-I.; Kato, K.; et al. Solution synthesis of N,N-dimethylformamide-stabilized iron-oxide nanoparticles as an efficient and recyclable catalyst for alkene hydrosilylation. ChemCatChem 2018, 10, 2378–2382. [Google Scholar] [CrossRef]
- Onishi, K.; Oikawa, K.; Yano, H.; Suzuki, T.; Obora, Y. N,N-Dimethylformamide-stabilized palladium nanoclusters as a catalyst for Larock indole synthesis. RSC Adv. 2018, 8, 11324–11329. [Google Scholar] [CrossRef]
- Oka, H.; Kitai, K.; Suzuki, T.; Obora, Y. N,N-Dimethylformamide-stabilized copper nanoparticles as a catalyst precursor for Sonogashira–Hagihara cross coupling. RSC Adv. 2017, 7, 22869–22874. [Google Scholar] [CrossRef]
- Oikawa, K.; Itoh, S.; Yano, H.; Kawasaki, H.; Obora, Y. Preparation and use of DMF-stabilized iridium nanoclusters as methylation catalysts using methanol as the C1 source. Chem. Commun. 2017, 53, 1080–1083. [Google Scholar] [CrossRef] [PubMed]
- Rasal, K.B.; Yadav, G.D. One-pot synthesis of benzimidazole using DMF as a multitasking reagent in presence CuFe2O4 as catalyst. Catal. Today 2018, 309, 51–60. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le Bras, J.; Muzart, J. Recent Uses of N,N-Dimethylformamide and N,N-Dimethylacetamide as Reagents. Molecules 2018, 23, 1939. https://doi.org/10.3390/molecules23081939
Le Bras J, Muzart J. Recent Uses of N,N-Dimethylformamide and N,N-Dimethylacetamide as Reagents. Molecules. 2018; 23(8):1939. https://doi.org/10.3390/molecules23081939
Chicago/Turabian StyleLe Bras, Jean, and Jacques Muzart. 2018. "Recent Uses of N,N-Dimethylformamide and N,N-Dimethylacetamide as Reagents" Molecules 23, no. 8: 1939. https://doi.org/10.3390/molecules23081939
APA StyleLe Bras, J., & Muzart, J. (2018). Recent Uses of N,N-Dimethylformamide and N,N-Dimethylacetamide as Reagents. Molecules, 23(8), 1939. https://doi.org/10.3390/molecules23081939