Oligo-Alginate with Low Molecular Mass Improves Growth and Physiological Activity of Eucomis autumnalis under Salinity Stress
Abstract
1. Introduction
2. Results and Discussion
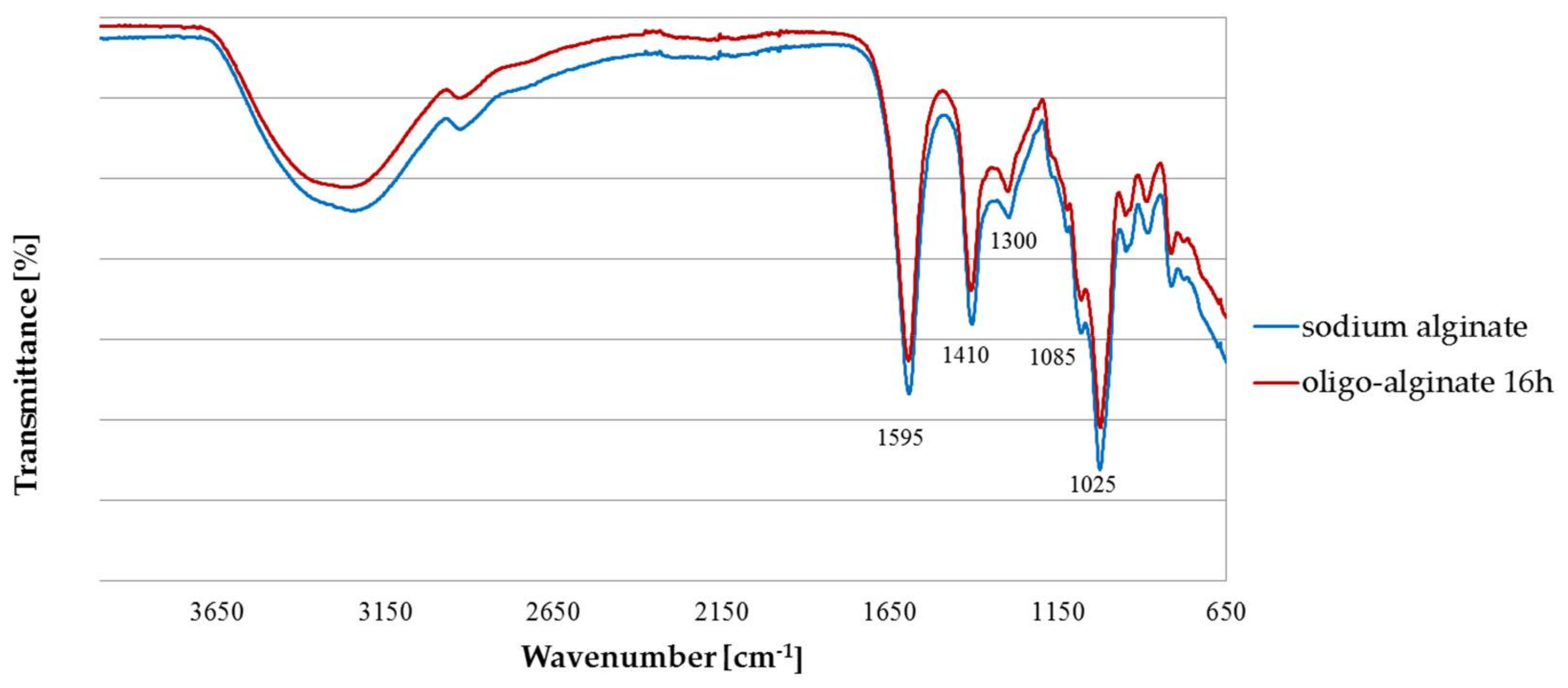
2.1. Fourier Transform Infra Red (FTIR)
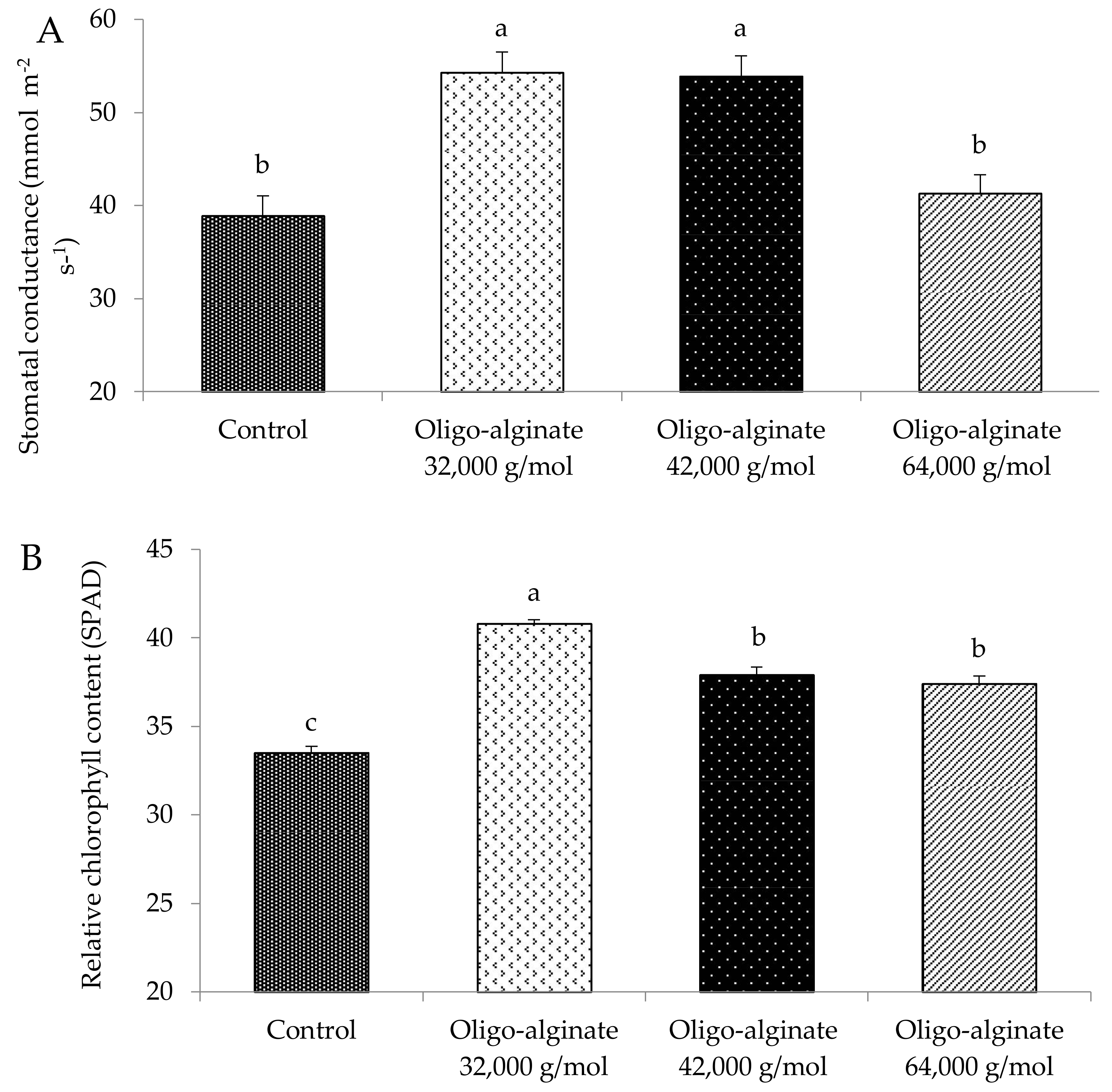
2.2. Effects of Oligo-Alginates with Different Molecular Mass on Growth Attributes of Eucomis autumnalis
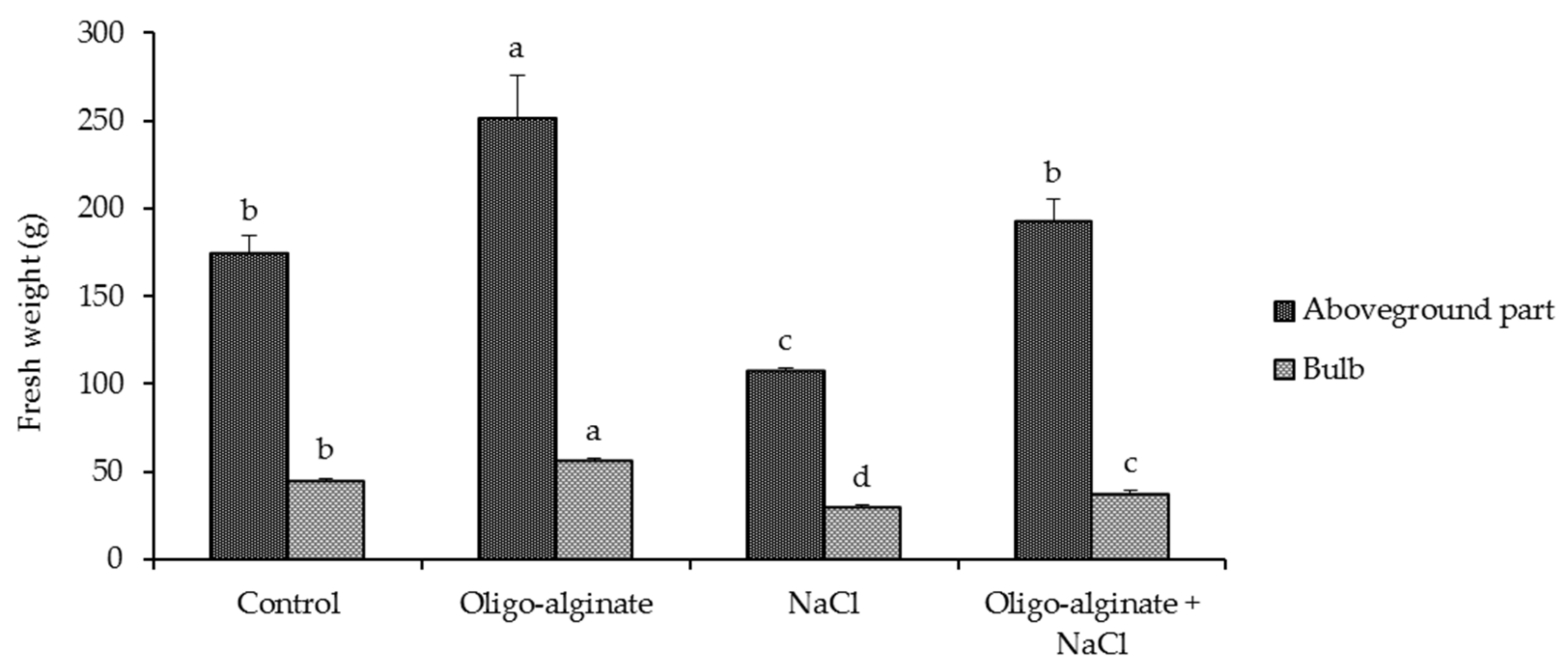
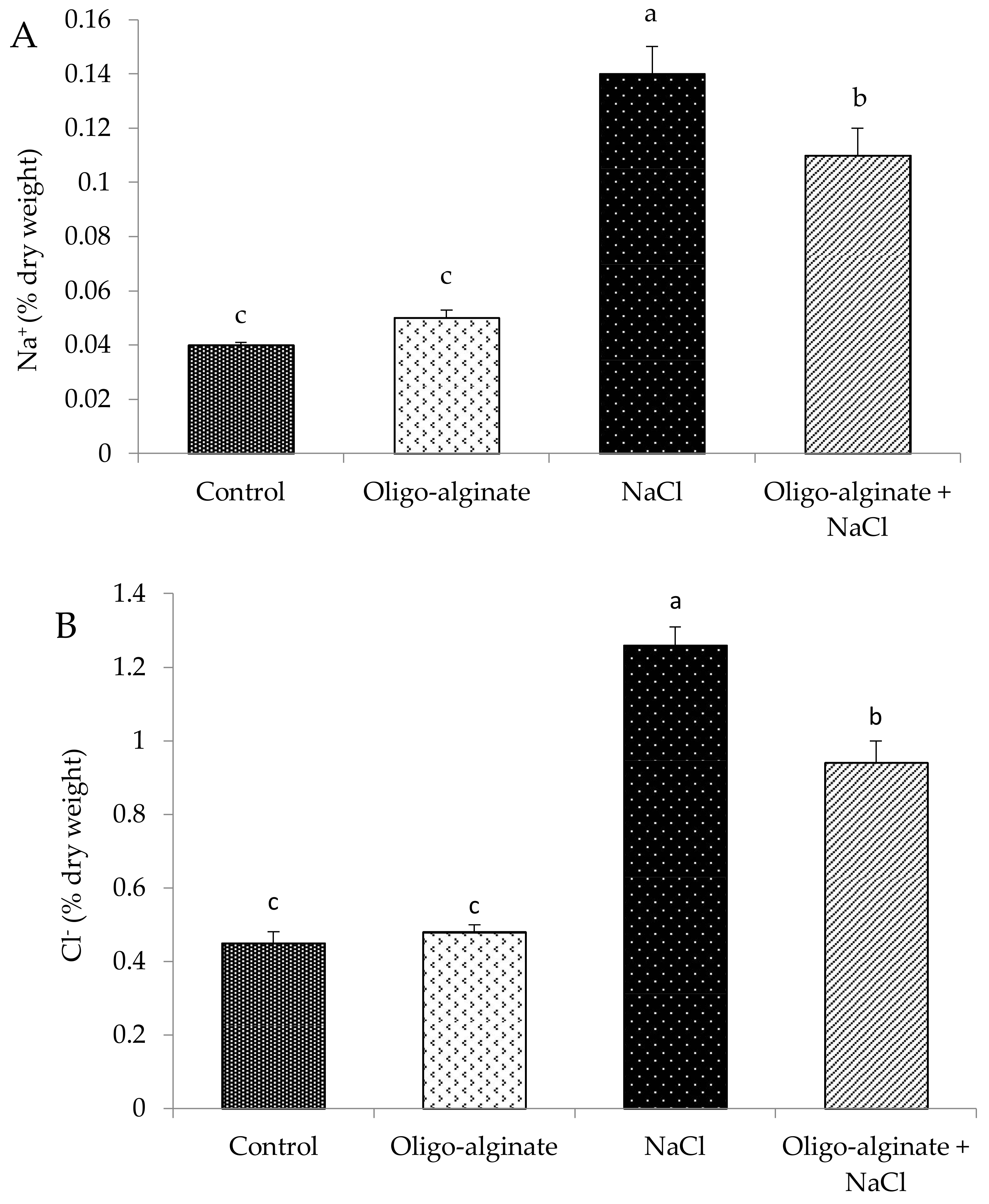
2.3. Effects of Oligo-Alginate and Salinity Stress on Growth and Physiological Attributes of Eucomis autumnalis
3. Materials and Methods
3.1. Plant Materials and Growth Conditions
3.2. Preparation and Determination the Molecular Mass of Oligo-Alginates
3.3. Fourier Transform Infra Red (FTIR) Analysis
3.4. Experiments
3.4.1. Effects of Oligo-Alginates with Different Molecular Mass on Growth of Eucomis autumnalis
Determination of Growth Parameters
3.4.2. Effect of Oligo-Alginate and Salt Stress on Plant Growth and Physiological Activity
Determination of Photosynthetic Pigments
- E—extinction at specific wavelength,
- V—volume of a volumetric flask [cm3],
- m—sample weight in g.
Determination of of l-Ascorbic Acid
Determination of Total Polyphenols
Determination of Antioxidant Activity by 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Free Radical Reduction
- At—absorbance of the test solution
- Ar—absorbance of the reference solution
Determination of Na and Cl
Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in plant science: A global perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48. [Google Scholar] [CrossRef]
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed extracts as biostimulants of plant growth and development. J. Plant Growth Regul. 2009, 28, 386–399. [Google Scholar] [CrossRef]
- Vera, J.; Castro, J.; González, A.; Moenne, A. Seaweed polysaccharides and derived oligosaccharides stimulate defense responses and protection against pathogens in plants. Mar. Drugs 2011, 9, 2514–2525. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D.; Rehman, Z.U.; Moradali, M.F.; Wang, Y.; Rehm, B.H.A. Microbial alginate production, modification and its applications. Microb. Biotechnol. 2013, 6, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Jiang, X.; Hwang, H.; Liu, S.; Guan, H. Promotive effects of alginate-derived oligosaccharide on maize seed germination. J. Appl. Phycol. 2004, 16, 73–76. [Google Scholar] [CrossRef]
- Hien, N.Q.; Nagasawa, N.; Tham, L.X.; Yoshii, F.; Dang, V.H.; Mitomo, H.; Makuuchi, K.; Kume, T. Growth promotion of plants with depolymerized alginates by irradiation. Radiat. Phys. Chem. 2000, 59, 97–101. [Google Scholar] [CrossRef]
- Luan, L.Q.; Hien, N.Q.; Nagasawa, N.; Kume, T.; Yoshii, F.; Nakanishi, T.M. Biological effect of radiation-degraded alginate on flower plants in tissue culture. Biotechnol. Appl. Biochem. 2003, 38, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Luan, L.Q.; Nagasawa, N.; Ha, V.T.T.; Hien, N.Q.; Nakanishi, T.M. Enhancement of plant growth stimulation activity of irradiated alginate by fractionation. Radiat. Phys. Chem. 2009, 78, 796–799. [Google Scholar] [CrossRef]
- Aftab, T.; Khan, M.M.A.; Idrees, M.; Naeem, M.; Hashmi, N.; Varshney, L. Enhancing the growth, photosynthetic capacity and artemisinin content in Artemisia annua L. by irradiated sodium alginate. Radiat. Phys. Chem. 2011, 80, 833–836. [Google Scholar] [CrossRef]
- Ali, A.; Khan, M.M.A.; Uddin, M.; Naeem, M.; Idrees, M.; Hashmi, N.; Dar, T.A.; Varshney, L. Radiolytically depolymerized sodium alginate improves physiological activities, yield attributes and composition of essential oil of Eucalyptus citriodora Hook. Carbohydr. Polym. 2014, 112, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Aftab, T.; Khan, M.M.A.; Naeem, M.; Idrees, M.; Siddiqi, T.O.; Varshney, L. Effect of irradiated sodium alginate and phosphorus on biomass and artemisinin production in Artemisia annua. Carbohydr. Polym. 2014, 110, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Idrees, M.; Naeem, M.; Alam, M.; Aftab, T.; Hashmi, N.; Khan, M.M.A.; Varshney, L. Utilizing the γ-irradiated sodium alginate as a plant growth promoter for enhancing the growth, physiological activities, and alkaloids production in Catharanthus roseus L. Agric. Sci. China 2011, 10, 1213–1221. [Google Scholar] [CrossRef]
- Idrees, M.; Dar, T.A.; Naeem, M.; Aftab, T.; Khan, M.M.A.; Ali, A.; Uddin, M.; Varshney, L. Effects of gamma-irradiated sodium alginate on lemongrass: Field trials monitoring production of essential oil. Ind. Crops Prod. 2015, 63, 269–275. [Google Scholar] [CrossRef]
- Khan, Z.H.; Khan, M.M.A.; Aftab, T.; Idrees, M.; Naeem, M. Influence of alginate oligosaccharides on growth, yield and alkaloid production of opium poppy (Papaver somniferum L.). Front. Agric. China 2011, 5, 122–127. [Google Scholar] [CrossRef]
- Naeem, M.; Idrees, M.; Aftab, T.; Khan, M.M.A.; Varshney, L. Depolymerised carrageenan enhances physiological activities and menthol production in Mentha arvensis L. Carbohydr. Polym. 2012, 87, 1211–1218. [Google Scholar] [CrossRef]
- Sarfaraz, A.; Naeem, M.; Nasir, S.; Idrees, M.; Aftab, T.; Hashmi, N.; Khan, M.A.A.; Varshney, M.; Varshney, L. An evaluation of the effects of irradiated sodium alginate on the growth, physiological activities and essential oil production of fennel (Foeniculum vulgare Mill.). J. Med. Plants Res. 2011, 5, 15–21. [Google Scholar]
- Sano, Y. Antiviral activity of alginate against infection by tobacco mosaic virus. Carbohydr. Polym. 1999, 38, 183–186. [Google Scholar] [CrossRef]
- Khan, N.; Mishra, A.; Chauhan, P.S.; Nautiyal, C.S. Induction of Paenibacillus lentimorbus biofilm by sodium alginate and CaCl2 alleviates drought stress in chickpea. Ann. Appl. Biol. 2011, 159, 372–386. [Google Scholar] [CrossRef]
- Ma, L.J.; Zhang, Y.; Bu, N.; Wang, S.H. Alleviation effect of alginate-derived oligosaccharides on Vicia faba root tip cells damaged by cadmium. Bull. Environ. Contam. Toxicol. 2010, 84, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Sarrocco, S.; Raeta, R.; Vannacci, G. Seeds encapsulation in calcium alginate pellets. Seed Sci. Technol. 2004, 32, 649–661. [Google Scholar] [CrossRef]
- Espino-Díaz, M.; Molina-Corral, F.; Sepulveda, D.; González-Aguilar, G.; Olivas, G. Alginate coatings containing high levels of isoleucine improve aromatic and standard quality in fresh-cut apple. Eur. J. Hortic. Sci. 2016, 81, 175–184. [Google Scholar] [CrossRef]
- Bach, A.; Pawłowska, B.; Malik, M. Plantlets from encapsulated meristems of Gentiana pneumonanthe L. Acta Physiol. Plant. 2004, 26, 53–57. [Google Scholar] [CrossRef]
- Sakhanokho, H.F.; Pounders, C.T.; Blythe, E.K. Alginate encapsulation of Begonia microshoots for short-term storage and distribution. Sci. World J. 2013. [Google Scholar] [CrossRef] [PubMed]
- Darvill, A.; Augur, C.; Bergmann, C.; Carlson, R.W.; Cheong, J.J.; Eberhard, S.; Hahn, M.G.; Lo, V.M.; Marfa, V.; Meyer, B.; et al. Oligosaccharins—Oligosaccharides that regulate growth, development and defence responses in plants. Glycobiology 1992, 2, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Winkler, A.J.; Dominguez-Nuñez, J.A.; Aranaz, I.; Poza-Carrión, C.; Ramonell, K.; Somerville, S.; Berrocal-Lobo, M. Short-chain chitin oligomers: Promoters of plant growth. Mar. Drugs 2017, 15, 40. [Google Scholar] [CrossRef] [PubMed]
- González, A.; Moenne, F.; Gómez, M.; Sáez, C.A.; Contreras, R.A.; Moenne, A. Oligo-carrageenan kappa increases NADPH, ascorbate and glutathione syntheses and TRR/TRX activities enhancing photosynthesis, basal metabolism, and growth in Eucalyptus trees. Front. Plant Sci. 2014, 5, 512. [Google Scholar] [CrossRef] [PubMed]
- El-Mohdy, H.A. Radiation-induced degradation of sodium alginate and its plant growth promotion effect. Arab. J. Chem. 2017, 10, 431–438. [Google Scholar] [CrossRef]
- Farmer, E.E.; Thomas, D.M.; Michael, J.S.; Clarence, A.R. Oligosaccharide signaling in plants. J. Biol. Chem. 1991, 266, 3140–3145. [Google Scholar] [PubMed]
- Shabbir, A.; Khan, M.M.A.; Sadiq, Y.; Jaleel, H.; Ahmad, B.; Uddin, M. Regulation of functional activities and essential oil production in Vetiveria zizanioides L. Nash after γ-irradiated sodium alginate elicitation. Turk. J. Biol. 2017, 41, 661–672. [Google Scholar] [CrossRef]
- Salachna, P.; Wilas, J.; Zawadzińska, A. The effect of chitosan coating of bulbs on the growth and flowering of Ornithogalum saundersiae. Acta Hortic. 2015, 1104, 115–118. [Google Scholar] [CrossRef]
- Zou, P.; Tian, X.; Dong, B.; Zhang, C. Size effects of chitooligomers with certain degrees of polymerization on the chilling tolerance of wheat seedlings. Carbohydr. Polym. 2017, 160, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Salachna, P.; Mizielińska, M.; Soból, M. Exopolysaccharide gellan gum and derived oligo-gellan enhance growth and antimicrobial activity in Eucomis plants. Polymers 2018, 10, 242. [Google Scholar] [CrossRef]
- Bartkowiak, A.; Startek, L.; Zurawik, P.; Salachna, P. Method of Hydrogel Coating Formation on the Surface of Plant Organs. Patent PL 197101, 29 February 2008. [Google Scholar]
- Bartkowiak, A.; Hunkeler, D. New microcapsules based on oligoelectrolyte complexation. Ann. N. Y. Acad. Sci. 1999, 875, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Startek, L.; Bartkowiak, A.; Salachna, P.; Kaminska, M.; Mazurkiewicz-Zapalowicz, K. The influence of new methods of corm coating on freesia growth, development and health. Acta Hortic. 2005, 673, 611–616. [Google Scholar] [CrossRef]
- Salachna, P.; Zawadzińska, A.; Wilas, J. The use of natural polysaccharides in Eucomis autumnalis (Mill.) Chitt. propagation by twin-scale cuttings. Acta Hortic. 2015, 1104, 225–227. [Google Scholar] [CrossRef]
- Salachna, P.; Grzeszczuk, M.; Soból, M. Effects of chitooligosaccharide coating combined with selected ionic polymers on the stimulation of Ornithogalum saundersiae growth. Molecules 2017, 22, 1903. [Google Scholar] [CrossRef] [PubMed]
- Salachna, P.; Zawadzińska, A. Effect of daminozide and flurprimidol on growth, flowering and bulb yield of Eucomis autumnalis (Mill.) Chitt. Folia Hort. 2017, 29, 33–38. [Google Scholar] [CrossRef][Green Version]
- Masondo, N.A.; Finnie, J.F.; Van Staden, J. Pharmacological potential and conservation prospect of the genus Eucomis (Hyacinthaceae) endemic to southern Africa. J. Ethnopharmacol. 2014, 151, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.S.; Dole, J.M. Eucomis for specialty cut flower production. Greenh. Prod. News 2011, 5, 18–22. [Google Scholar]
- Bisi-Johnson, M.A.; Obi, C.L.; Hattori, T.; Oshima, Y.; Li, S.; Kambizi, L.; Vasaikar, S.D. Evaluation of the antibacterial and anticancer activities of some South African medicinal plants. BMC Complement. Altern. Med. 2011, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Koorbanally, C.; Crouch, N.R.; Mulholland, D.A. The phytochemistry and ethnobotany of the southern African genus Eucomis (Hyacinthaceae: Hyacinthoideae). In Phytochemistry: Advances in Research; Imperato, F., Ed.; Research Signpost: Trivandrum, India, 2006; pp. 69–85. ISBN 81-308-0034-9. [Google Scholar]
- Mizielińska, M.; Salachna, P.; Ordon, M.; Łopusiewicz, Ł. Antimicrobial activity of water and acetone extracts of some Eucomis taxa. Asian Pac. J. Trop. Med. 2017, 10, 892–895. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.L.S.; Rabe, T.; McGaw, L.J.; Jäger, A.K.; Van Staden, J. Towards the scientific validation of traditional medicinal plants. Plant Growth Regul. 2001, 34, 23–37. [Google Scholar] [CrossRef]
- Masondo, N.A.; Aremu, A.O.; Rengasamy, K.R.R.; Amoo, S.O.; Gruz, J.; Šubrtová, M.; Doležal, K.; Van Staden, J. Growth and phytochemical response in Eucomis autumnalis (Mill.) Chitt. treated with phenolic biostimulants from brown alga, Ecklonia maxima. S. Afr. J. Bot. 2015, 98, 211. [Google Scholar] [CrossRef]
- Salachna, P.; Zawadzińska, A.; Wierzbiński, Ł.; Senderek, W. Enhancing growth in Eucomis autumnalis (Mill.) Chitt. seedlings with exogenous application of nitric oxide. J. Hortic. Res. 2016, 24, 13–17. [Google Scholar] [CrossRef][Green Version]
- Hura, T.; Szewczyk-Taranek, B.; Hura, K.; Nowak, K.; Pawłowska, B. Physiological responses of Rosa rubiginosa to saline environment. Water Air Soil Pollut. 2017, 228, 81. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Munns, R.; Termaat, A. Whole-plant responses to salinity. Funct. Plant Biol. 1986, 13, 143–160. [Google Scholar] [CrossRef]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Parihar, P.; Singh, S.; Singh, R.; Singh, V.P.; Prasad, S.M. Effect of salinity stress on plants and its tolerance strategies: A review. Environ. Sci. Pollut. R. 2015, 22, 4056–4075. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.S.; Fleming, C.; Selby, C.; Rao, J.R.; Martin, T. Plant biostimulants: A review on the processing of macroalgae and use of extracts for crop management to reduce abiotic and biotic stresses. J. Appl. Phycol. 2014, 26, 465–490. [Google Scholar] [CrossRef]
- Hernandez-Hernandez, H.; Gonzalez-Morales, S.; Benavides-Mendoza, A.; Ortega-Ortiz, H.; Cadenas-Pliego, G.; Juarez-Maldonado, A. Effects of chitosan–PVA and Cu nanoparticles on the growth and antioxidant capacity of tomato under saline stress. Molecules 2018, 23, 178. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Li, K.; Liu, S.; He, X.; Zhang, X.; Xing, R.; Li, P. Effect of sulfated chitooligosaccharides on wheat seedlings (Triticum aestivum L.) under salt stress. J. Agric. Food Chem. 2016, 64, 2815–2821. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Khan, M.M.A.; Uddin, M.; Naeem, M.; Qureshi, M.I. Proliferating effect of radiolytically depolymerized carrageenan on physiological attributes, plant water relation parameters, essential oil production and active constituents of Cymbopogon flexuosus Steud. under drought stress. PLoS ONE 2017, 12, e0180129. [Google Scholar] [CrossRef] [PubMed]
- Lawrie, G.; Keen, I.; Drew, B.; Chandler-Temple, A.; Rintoul, L.; Fredericks, P.; Grøndahl, L. Interactions between alginate and chitosan biopolymers characterized using FTIR and XPS. Biomacromolecules 2007, 8, 2533–2541. [Google Scholar] [CrossRef] [PubMed]
- Sakugawa, K.; Ikeda, A.; Takemura, A.; Ono, H. Simplified method for estimation of composition of alginates by FTIR. J. Appl. Polym. Sci. 2004, 93, 1372–1377. [Google Scholar] [CrossRef]
- González-Pérez, L.; Vázquez-Glaría, A.; Perrotta, L.; Acosta, A.; Scriven, S.A.; Herbert, R.; Cabrera, J.C.; Francis, D.; Rogers, H.J. Oligosaccharins and Pectimorf® stimulate root elongation and shorten the cell cycle in higher plants. Plant Growth Regul. 2012, 68, 211–221. [Google Scholar] [CrossRef]
- Dzung, P.D.; Phu, D.V.; Du, B.D.; Ngoc, L.S.; Duy, N.N.; Hiet, H.D.; Hien, N.Q. Effect of foliar application of oligochitosan with different molecular weight on growth promotion and fruit yield enhancement of chili plant. Plant Prod. Sci. 2017, 20, 389–395. [Google Scholar] [CrossRef]
- Shillo, R.; Ding, M.; Pasternak, D.; Zaccai, M. Cultivation of cut flower and bulb species with saline water. Sci. Hortic. 2002, 92, 41–54. [Google Scholar] [CrossRef]
- Salachna, P.; Zawadzińska, A.; Podsiadło, C. Response of Ornithogalum saundersiae Bak. to salinity stress. Acta Sci. Pol.-Hortorum Cultus 2016, 15, 123–134. [Google Scholar]
- Cassaniti, C.; Romano, D.; Flowers, T.J. The response of ornamental plants to saline irrigation water. In Irrigation-Water Management, Pollution and Alternative Strategies; Garcia-Garizabal, I., Ed.; InTech Europe: Rijeka, Croatia, 2012; pp. 131–158. [Google Scholar]
- Akimoto, C.; Aoyagi, H.; Tanaka, H. Endogenous elicitor-like effects of alginate on physiological activities of plant cells. Appl. Microbiol. Biotechnol. 1999, 52, 429–436. [Google Scholar] [CrossRef]
- Ma, L.J.; Li, X.M.; Bu, N.; Li, N. An alginate-derived oligosaccharide enhanced wheat tolerance to cadmium stress. Plant Growth Regul. 2010, 62, 71–76. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.H.; Yin, H.; Wang, W.X.; Zhao, X.M.; Du, Y.G. Alginate oligosaccharides enhanced Triticum aestivum L. tolerance to drought stress. Plant Physiol. Biochem. 2013, 62, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Zou, P.; Li, K.; Liu, S.; Xing, R.; Qin, Y.; Yu, H.; Zhou, M.; Li, P. Effect of chitooligosaccharides with different degrees of acetylation on wheat seedlings under salt stress. Carbohydr. Polym. 2015, 126, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Veatch-Blohm, M.E.; Sawch, D.; Elia, N.; Pinciotti, D. Salinity tolerance of three commonly planted Narcissus cultivars. HortScience 2014, 49, 1158–1164. [Google Scholar]
- Carter, C.T.; Grieve, C.M. Salt tolerance of floriculture crops. In Ecophysiology of High Salinity Tolerant Plants; Khan, M.A., Weber, D.J., Eds.; Springer: Amsterdam, The Netherlands, 2006; pp. 279–287. [Google Scholar]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lin, F.; Yang, H.; Yue, L.; Hu, F.; Wang, J.; Luo, Y.; Cao, F. Effect of varying NaCl doses on flavonoid production in suspension cells of Ginkgo biloba: Relationship to chlorophyll fluorescence, ion homeostasis, antioxidant system and ultrastructure. Acta Physiol. Plant. 2014, 36, 3173–3187. [Google Scholar] [CrossRef]
- Deinlein, U.; Stephan, A.B.; Horie, T.; Luo, W.; Xu, G.; Schroeder, J.I. Plant salt-tolerance mechanisms. Trends Plant Sci. 2014, 19, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Karaki, N.; Aljawish, A.; Humeau, C.; Muniglia, L.; Jasniewski, J. Enzymatic modification of polysaccharides: Mechanisms, properties, and potential applications: A review. Enzyme Microb. Technol. 2016, 90, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Albersheim, P.; Darvill, A.G. Oligosaccharins. Sci. Am. 1985, 253, 58–65. [Google Scholar] [CrossRef]
- Zhang, Y.; Yin, H.; Zhao, X.; Wang, W.; Du, Y.; He, A.; Sun, K. The promoting effects of alginate oligosaccharides on root development in Oryza sativa L. mediated by auxin signaling. Carbohydr. Polym. 2014, 113, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tang, W.; Jiang, L.; Hou, Y.; Yang, F.; Chen, W.; Li, X. Elicitor activity of algino-oligosaccharide and its potential application in protection of rice plant (Oryza sativa L.) against Magnaporthe grisea. Biotechnol. Biotechnol. Equip. 2015, 29, 646–652. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. AOAC Official Methods of Analysis of the Association of Official Analytical Chemists; Association of Official Analytical Chemists: Washington, DC, USA, 1990. [Google Scholar]
- Salachna, P.; Grzeszczuk, M.; Wilas, J. Total phenolic content, photosynthetic pigment concentration and antioxidant activity of leaves and bulbs of selected Eucomis L’Hér. taxa. Fresenius Environ. Bull. 2015, 24, 4220–4225. [Google Scholar]
- Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food Chem. 1995, 43, 27–32. [Google Scholar] [CrossRef]
- Rossi, M.; Giussani, E.; Morelli, R.; Scalzo, R.; Nani, R.C.; Torreggiani, D. Effect of fruit blanching on phenolics and radical scavenging activity of highbush blueberry juice. Food Res. Int. 2003, 36, 999–1005. [Google Scholar] [CrossRef]
- Ostrowska, A.; Gawliński, S.; Szczubiałka, Z. Methods for Analyzing and Assessing the Properties of Soil and Plants; Instytut Ochrony Środowiska: Warsaw, Poland, 1991; pp. 1–333. (In Polish) [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Parameters | Oligo-Alginate Molecular Mass (g mol−1) | |||
|---|---|---|---|---|
| Control | 32,000 | 42,000 | 64,000 | |
| Days to anthesis | 81.6 ± 0.78 a | 75.2 ± 0.61 b | 76.0 ± 0.50 b | 77.2 ± 0.52 b |
| Plant height (cm) | 31.2 ± 1.59 c | 40.8 ± 1.01 a | 37.5 ± 1.04 a,b | 34.3 ± 1.64 b,c |
| Plant width (cm) | 28.3 ± 1.04 b | 36.4 ± 1.68 a | 32.0 ± 1.80 a,b | 32.5 ± 0.76 a,b |
| Leaf length (cm) | 23.7 ± 1.29 b | 32.0 ± 1.99 a | 30.3 ± 0.79 a | 32.4 ± 1.84 a |
| Number of leaves | 5.33 ± 0.17 a | 5.50 ± 0.29 a | 5.50 ± 0.29 a | 5.50 ± 0.29 a |
| Number of inflorescences | 1.10 ± 0.06 a | 1.10 ± 0.03 a | 1.08 ± 0.04 a | 1.03 ± 0.58 a |
| Inflorescence length (cm) | 15.3 ± 0.88 b | 20.3 ± 0.33 a | 19.8 ± 0.33 a | 21.2 ± 1.36 a |
| Number of florets | 68.6 ± 4.03 c | 88.7 ± 2.23 a | 81.4 ± 1.99 b | 81.4 ± 2.45 b |
| Parameters | Treatment | |||
|---|---|---|---|---|
| Control | Oligo-Alginate | NaCl | Oligo-Alginate + NaCl | |
| Chlorophyll a (mg kg−1 FW) | 434.2 ± 6.90 b | 497.0 ± 5.50 a | 342.3 ± 15.41 c | 396.0 ± 4.51 b |
| Chlorophyll b (mg kg−1 FW) | 166.6 ± 3.91 a,b | 186.8 ± 0.96 a | 146.5 ± 8.29 b | 160.3 ± 1.72 b |
| Chlorophyll a/b ratio | 2.61 ± 0.06 a,b | 2.66 ± 0.04 a | 2.34 ± 0.03 c | 2.47 ± 0.02 b,c |
| Chlorophyll a + b (mg kg−1 FW) | 600.8 ± 9.24 b | 683.8 ± 5.12 a | 488.8 ± 23.66 c | 556.3 ± 6.02 b |
| Carotenoid (mg kg−1 FW) | 170.2 ± 5.68 a,b | 187.8 ± 2.05 a | 153.3 ± 9.22 b | 176.1 ± 2.53 a,b |
| Chlorophyll/carotenoid ratio | 3.54 ± 0.06 a | 3.64 ± 0.01 a | 3.19 ± 0.06 b | 3.16 ± 0.03 b |
| Parameters | Treatment | |||
|---|---|---|---|---|
| Control | Oligo-Alginate | NaCl | Oligo-Alginate + NaCl | |
| Total polyphenols (mg GAE 100 g−1 FW) | 39.6 ± 1.56 c | 57.6 ± 1.19 a | 46.4 ± 2.29 b | 49.1 ± 1.75 b |
| l-ascorbic acid (mg 100 g−1 FW) | 27.4 ± 1.49 c | 42.6 ± 2.02 a | 30.8 ± 2.27 b | 36.0 ± 2.25 a,b |
| Antioxidant activity (% DPPH) | 1.64 ± 0.11 d | 2.94 ± 0.25 a | 2.18 ± 0.09 c | 2.41 ± 0.08 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salachna, P.; Grzeszczuk, M.; Meller, E.; Soból, M. Oligo-Alginate with Low Molecular Mass Improves Growth and Physiological Activity of Eucomis autumnalis under Salinity Stress. Molecules 2018, 23, 812. https://doi.org/10.3390/molecules23040812
Salachna P, Grzeszczuk M, Meller E, Soból M. Oligo-Alginate with Low Molecular Mass Improves Growth and Physiological Activity of Eucomis autumnalis under Salinity Stress. Molecules. 2018; 23(4):812. https://doi.org/10.3390/molecules23040812
Chicago/Turabian StyleSalachna, Piotr, Monika Grzeszczuk, Edward Meller, and Marcin Soból. 2018. "Oligo-Alginate with Low Molecular Mass Improves Growth and Physiological Activity of Eucomis autumnalis under Salinity Stress" Molecules 23, no. 4: 812. https://doi.org/10.3390/molecules23040812
APA StyleSalachna, P., Grzeszczuk, M., Meller, E., & Soból, M. (2018). Oligo-Alginate with Low Molecular Mass Improves Growth and Physiological Activity of Eucomis autumnalis under Salinity Stress. Molecules, 23(4), 812. https://doi.org/10.3390/molecules23040812







