Synthesis, Structural Characterization and Antimicrobial Activity of Cu(II) and Fe(III) Complexes Incorporating Azo-Azomethine Ligand
Abstract
1. Introduction
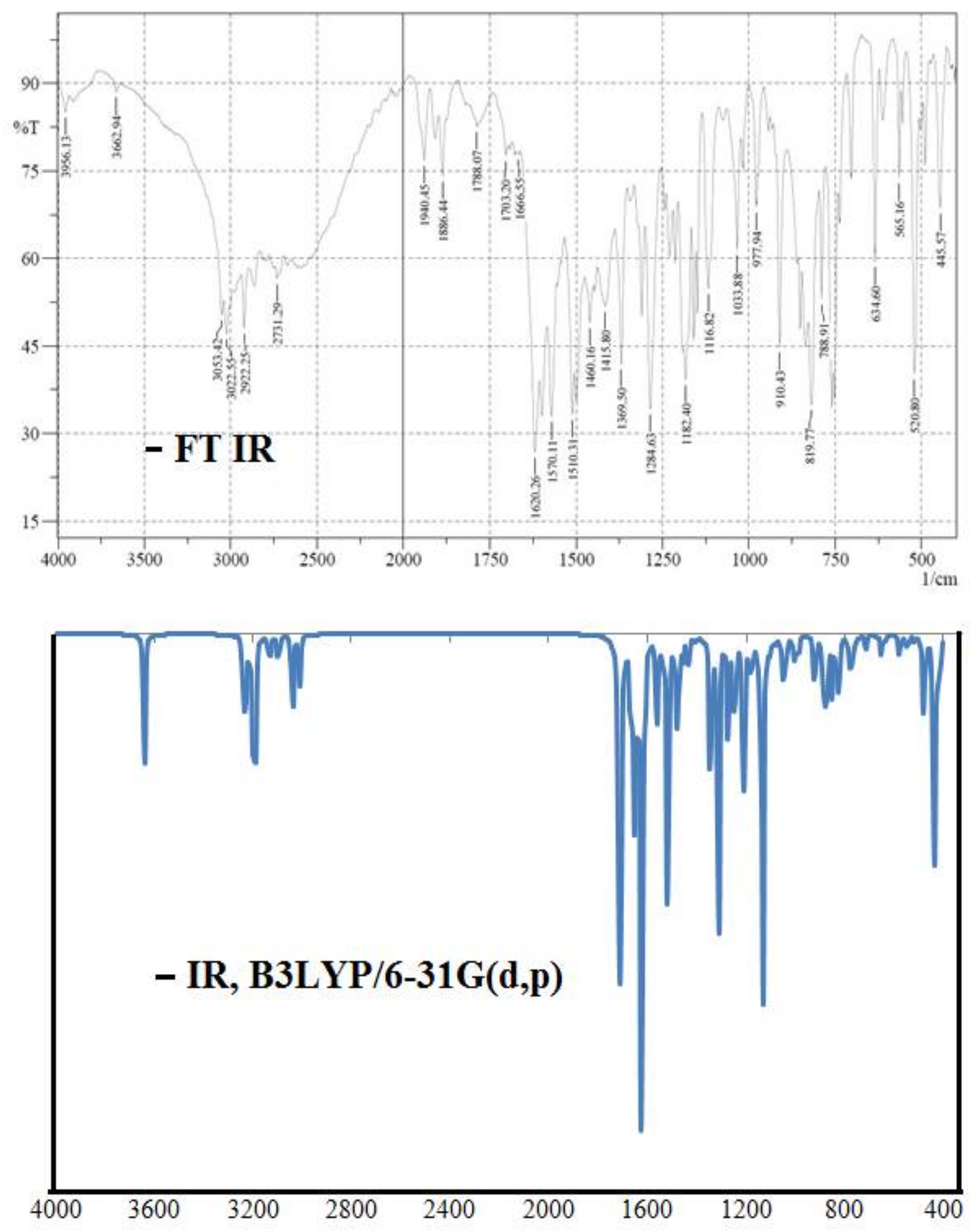
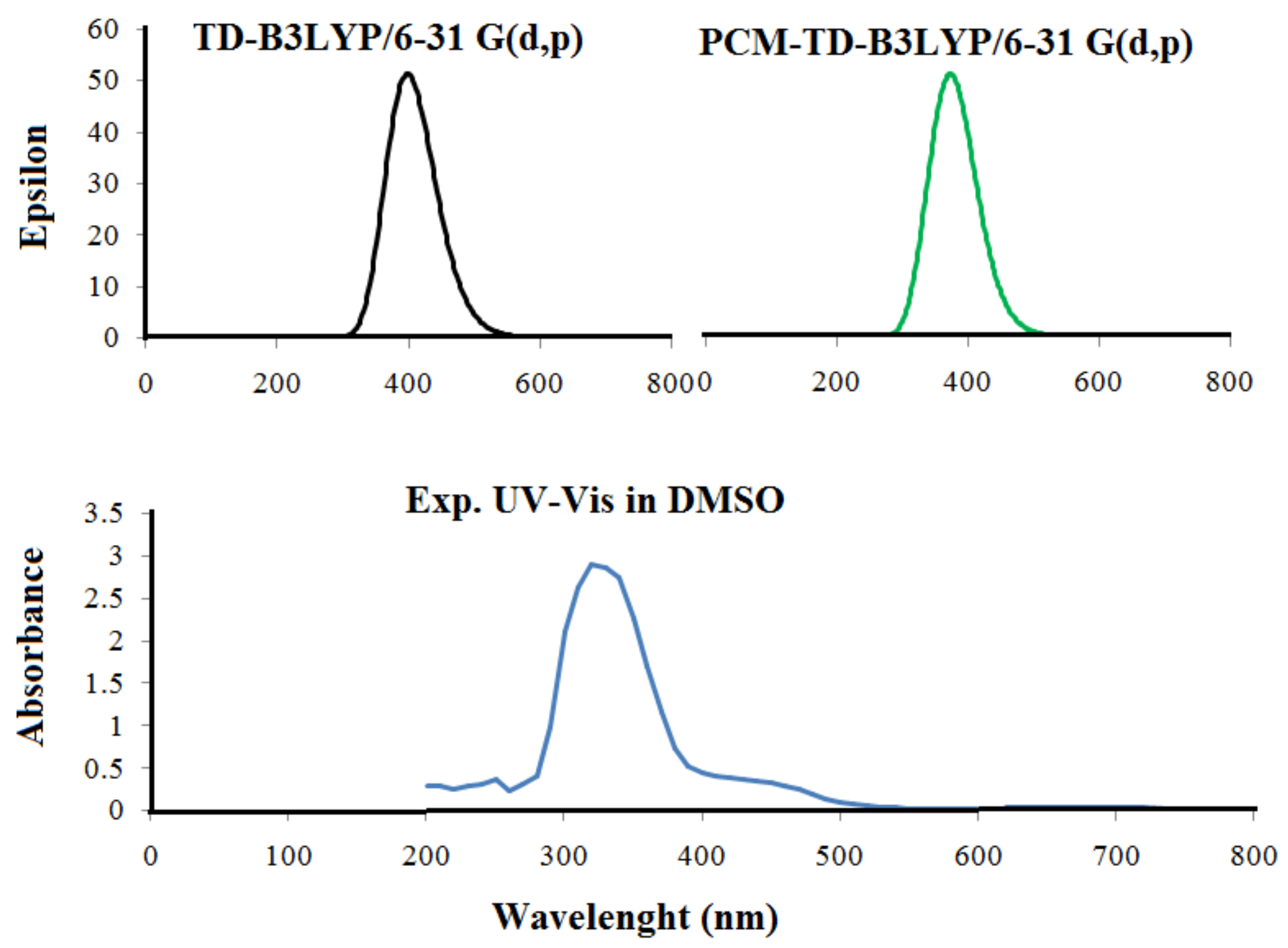
2. Results and Discussion
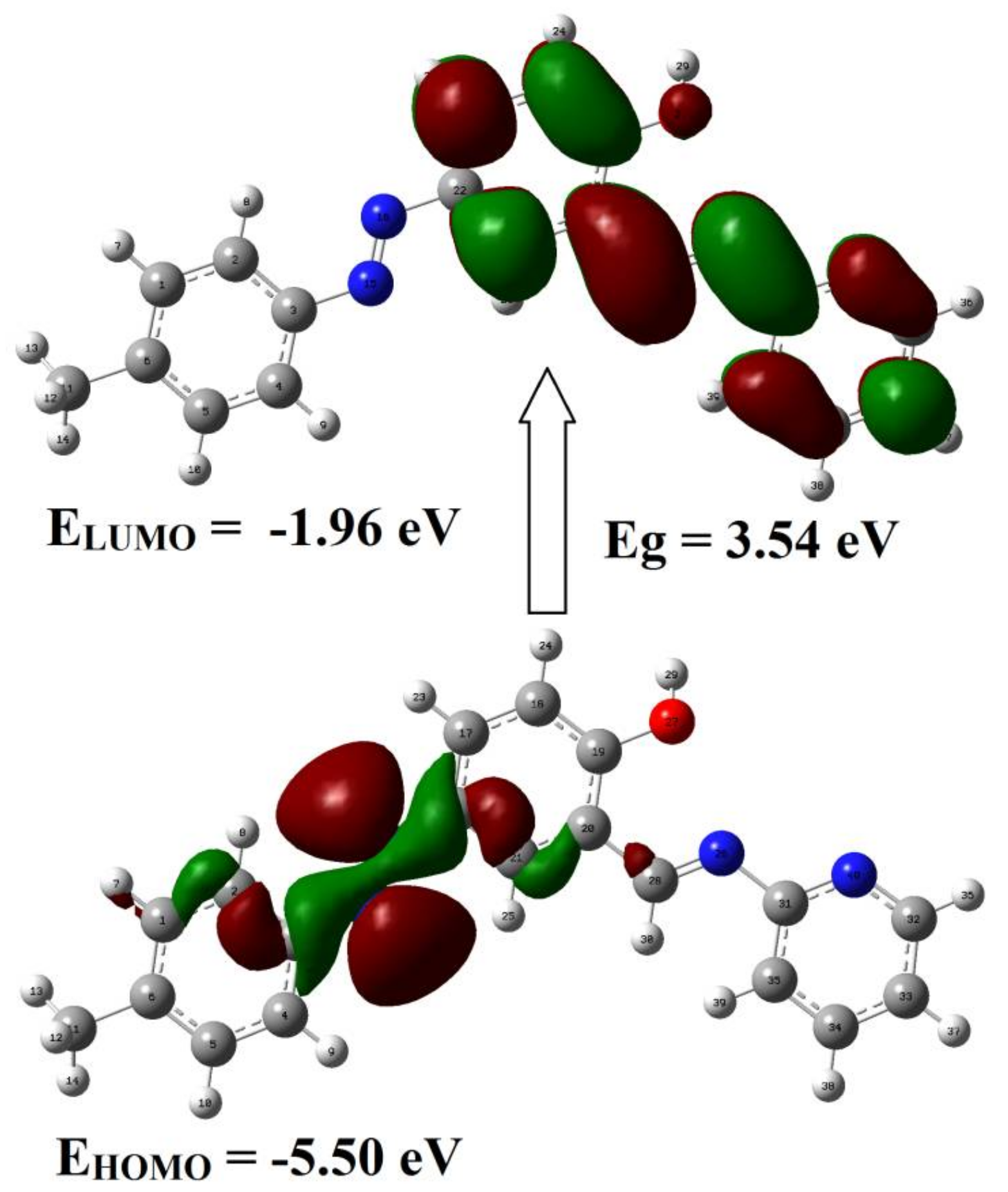
2.1. Frontier Molecular Orbitals—HOMO and LUMO of Ligand
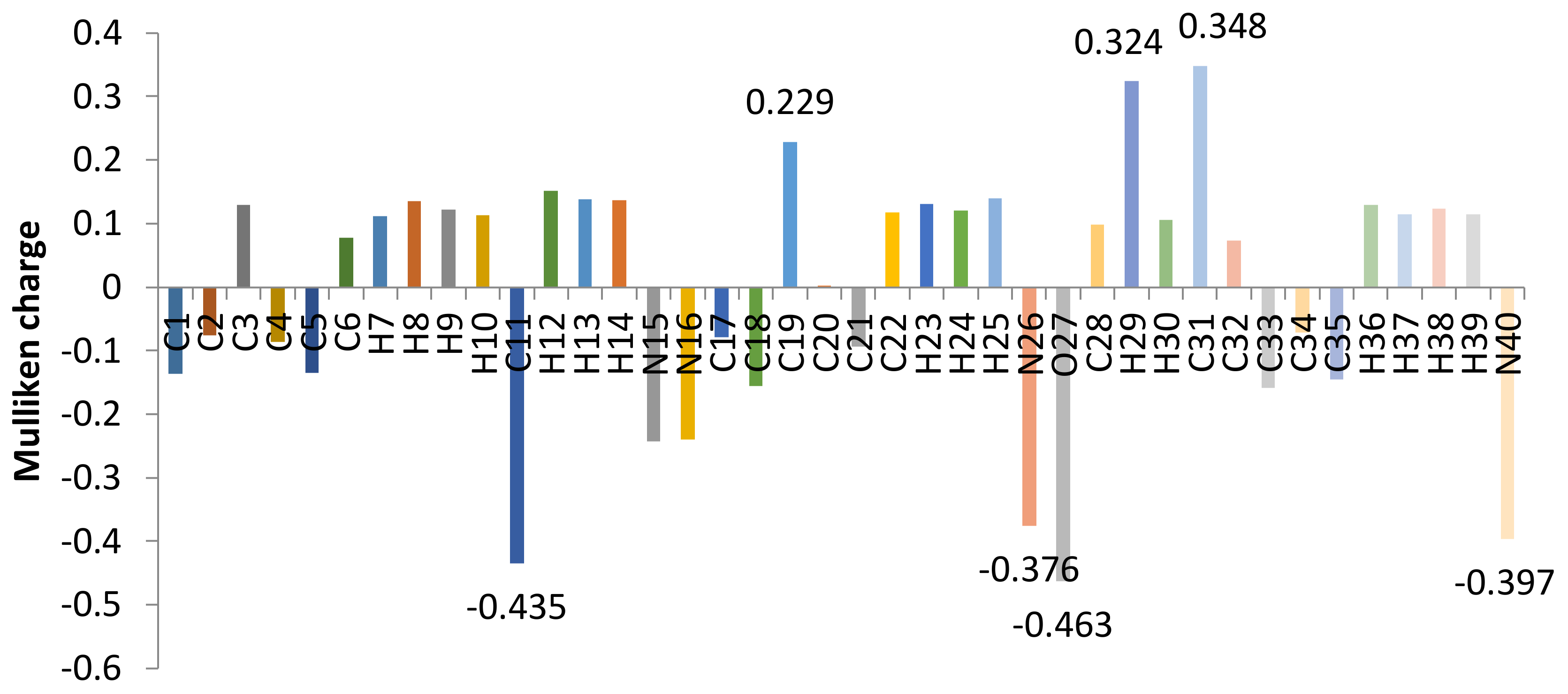
2.2. Mulliken Charge Analysis of Ligand
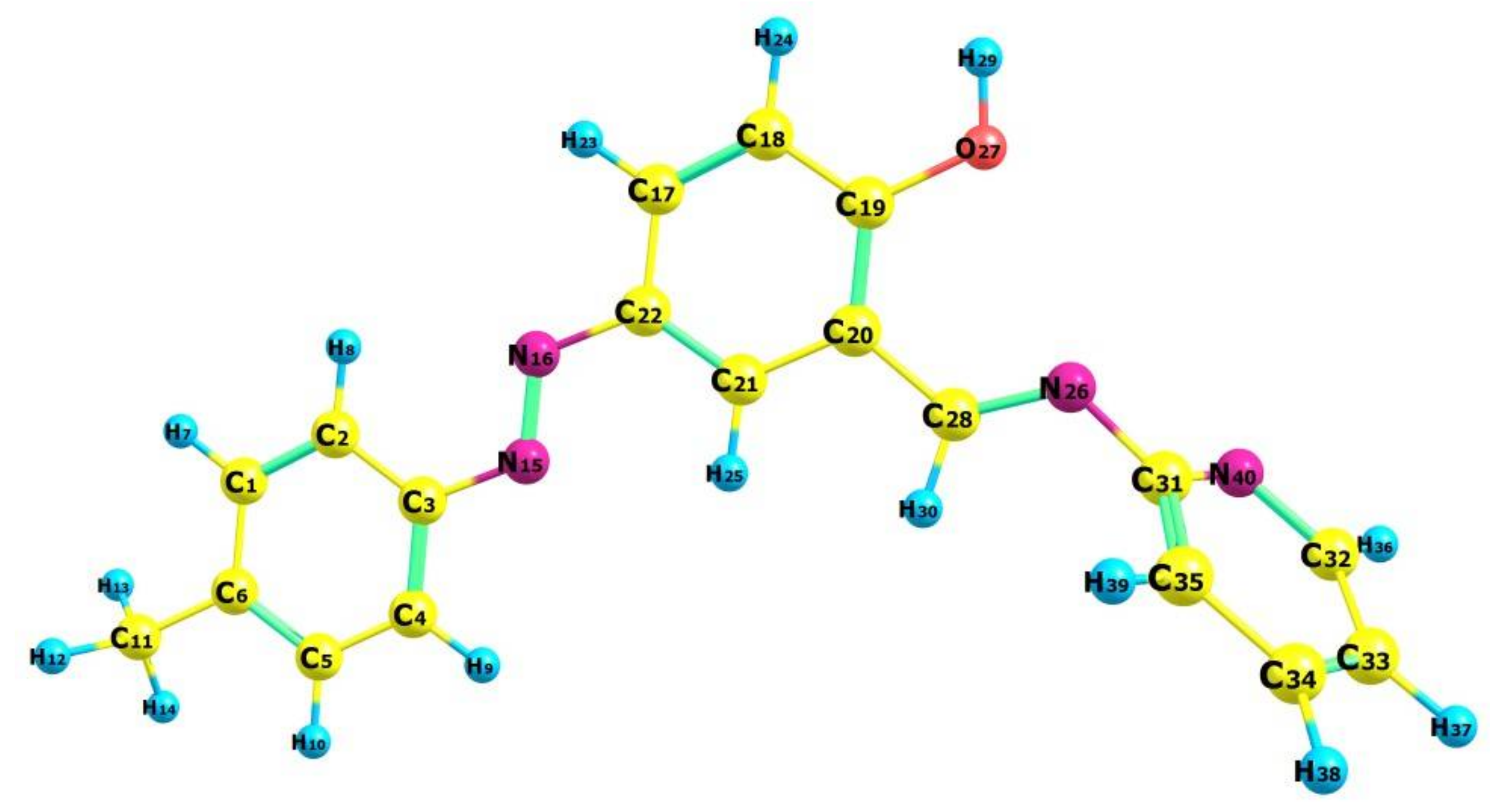
2.3. Molecular Geometry of Ligand
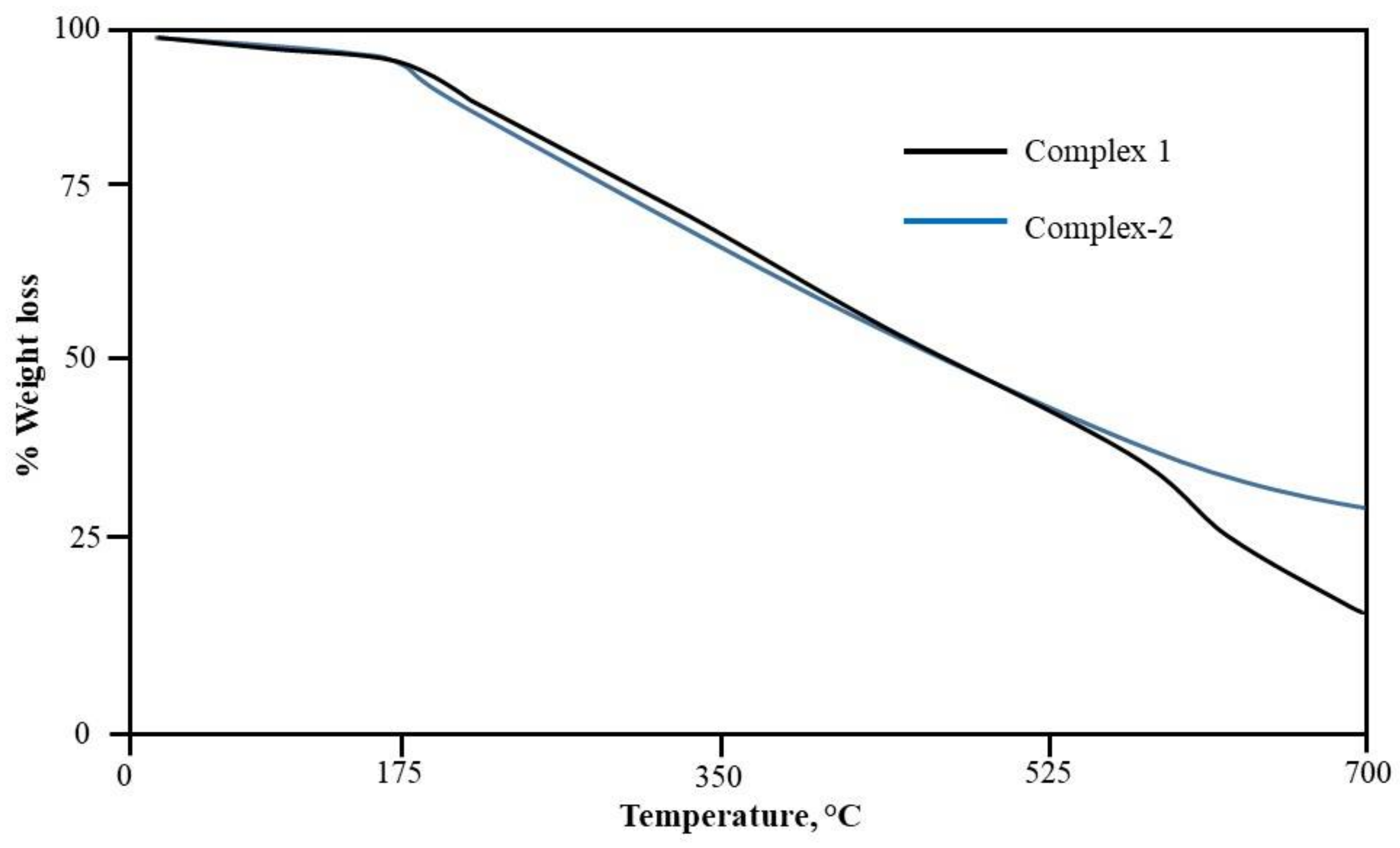
2.4. Thermal Studies
2.5. Antimicrobial Activity
3. Materials and Methods
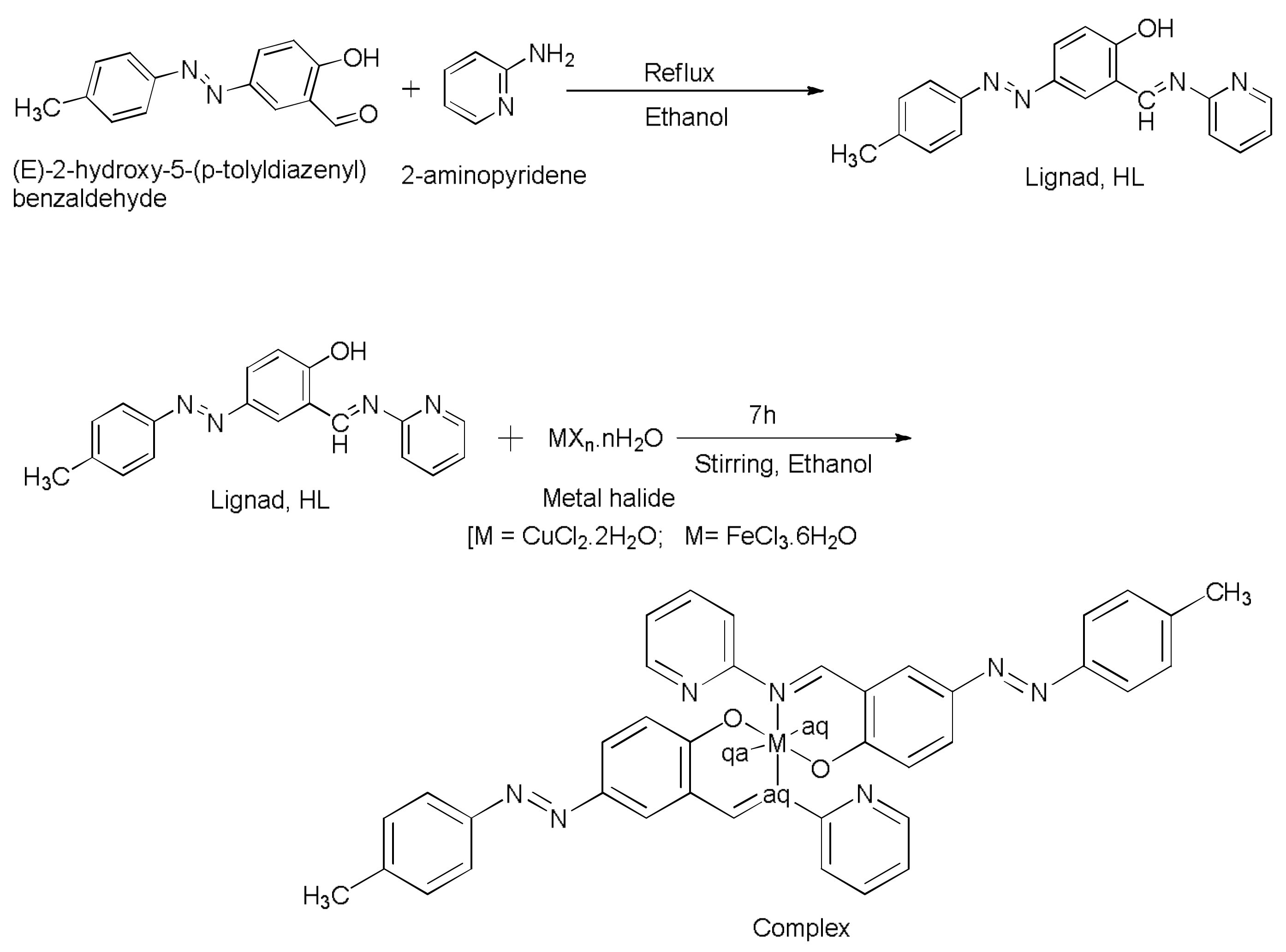
3.1. Synthesis of Ligand, (4E)-4-(2-p-tolyldiazenyl)-2-((E)-(pyridin-2-ylimino)methyl)phenol, HL
3.2. Synthesis of Complexes
3.2.1. Synthesis of Complex 1, [Cu(L2)(H2O)2]
3.2.2. Synthesis of Complex 2, [Fe(L)22H2O]
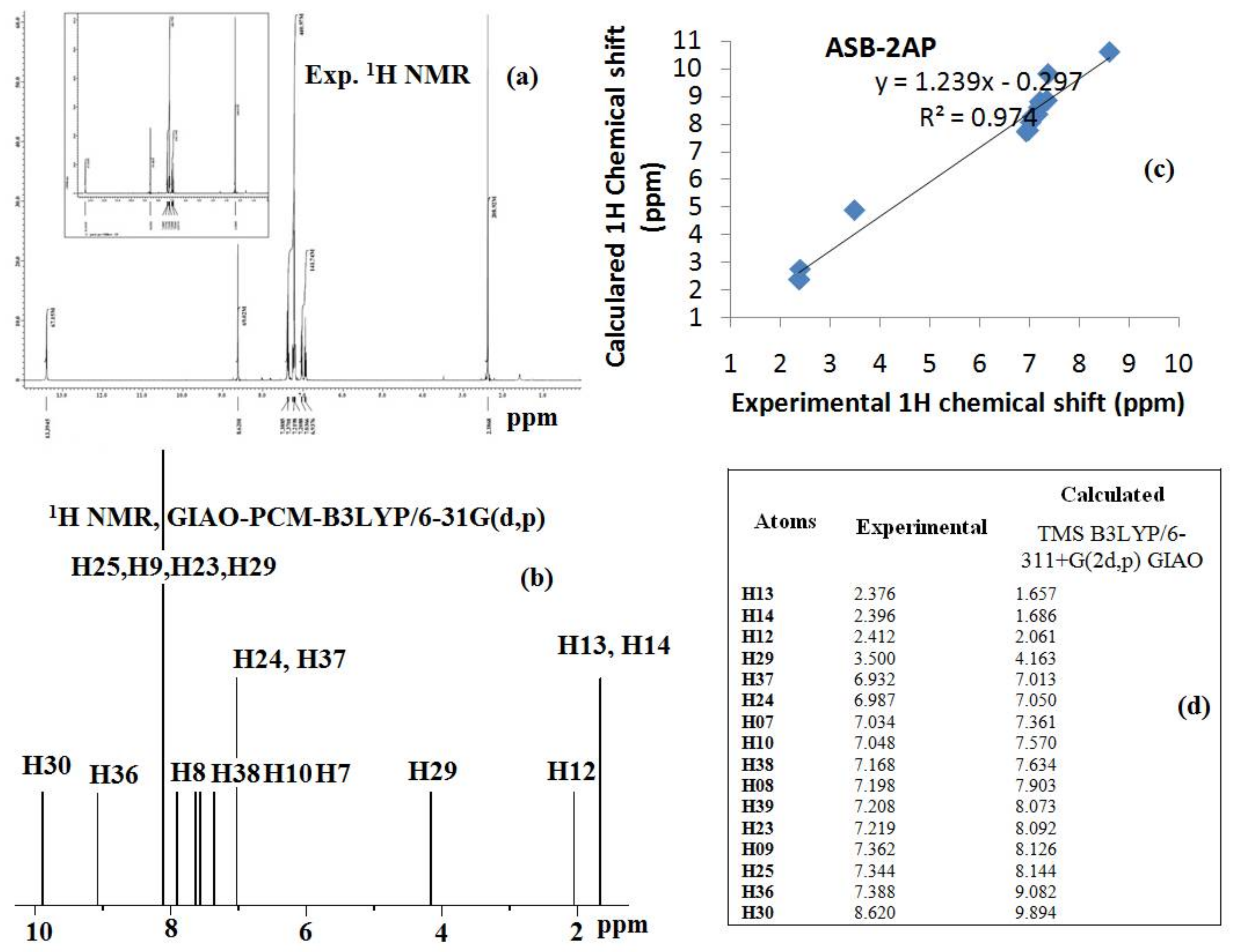
3.3. DFT Calculations for Ligand, HL
3.4. Antimicrobial Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hunger, K. Industrial Dyes. Chemistry, Properties, Applications; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Sarigul, M.; Kariper, S.E.; Deveci, P.; Atabey, H.; Karakas, D.; Kurtoglu, M. Multi-properties of a new azo-Schiff base and its binuclear copper(II) chelate: Preparation, spectral characterization, electrochemical, potentiometric and modeling studies. J. Mol. Struct. 2017, 1149, 520–529. [Google Scholar] [CrossRef]
- Eren, T.; Kose, M.; Kurtoglu, N.; Ceyhan, G.; McKee, V.; Kurtoglu, M. An azo-azomethine ligand and its copper (II) complex: Synthesis, X-ray crystal structure, spectral, thermal, electrochemical and photoluminescence properties. Inorg. Chim. Acta 2015, 430, 268–279. [Google Scholar] [CrossRef]
- El-Bindary, A.A.; Mohamed, G.G.; El-Sonbati, A.Z.; Diab, M.A.; Hassan, W.M.I.; Morgan, S.M.; Elkholy, A.K. Geometrical structure, potentiometric, molecular docking and thermodynamic studies of azo dye ligand and its metal complexes. J. Mol. Liq. 2016, 218, 138–149. [Google Scholar] [CrossRef]
- Pramanik, A.K.; Sarkar, D.; Mondal, T.K. Electronic structure of thioether containing NSNO donor azo-ligand and its copper(II) complex: Experimental and theoretical studies. J. Mol. Struct. 2015, 1099, 92–98. [Google Scholar] [CrossRef]
- Gup, R.; Giziroglu, E.; Kırkan, B. Synthesis and spectroscopic properties of new azo-dyes and azo-metal complexes derived from barbituric acid and aminoquinoline. Dyes Pigments 2007, 73, 40–46. [Google Scholar] [CrossRef]
- Peters, A.T.; Freeman, H.S. Colour Chemistry, the Design and Synthesis of Organic Dyes and Pigments; Elsevier Applied Science: Essex, UK, 1991; p. 193. [Google Scholar]
- Gregory, P. High-Technology Applications of Organic Colorants; Plenum Press: New York, NY, USA, 1991; p. 1. [Google Scholar]
- Catino, S.C.; Farris, R.E. Concise Encyclopedia of Chemical Technology; Grayson, M., Ed.; John Wiley & Sons: New York, NY, USA, 1985; p. 142. [Google Scholar]
- Ghasemian, M.; Kakanejadifard, A.; Azarbani, F.; Zabardasti, A.; Kakanejadifard, S. Spectroscopy and solvatochromism studies along with antioxidant and antibacterial activities investigation of azo–azomethine compounds 2-(2-hydroxyphenylimino)methyl)-4-phenyldiazenyl)phenol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 124, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Sorochinsky, A.E.; Acena, J.L.; Moriwaki, H.; Sato, T.; Soloshonok, V.A. Asymmetric synthesis of α-amino acids via homologation of Ni(II) complexes of glycine Schiff bases; Part 1: Alkyl halide alkylations. Amino Acids 2013, 45, 691–718. [Google Scholar] [CrossRef] [PubMed]
- Sorochinsky, A.E.; Acena, J.L.; Moriwaki, H.; Sato, T.; Soloshonok, V.A. Asymmetric synthesis of α-amino acids via homologation of Ni(II) complexes of glycine Schiff bases. Part 2: Aldol, Mannich addition reactions, deracemization and (S) to (R) interconversion of α-amino acids. Amino Acids 2013, 45, 1017–1033. [Google Scholar] [CrossRef] [PubMed]
- Acena, J.L.; Sorochinsky, A.E.; Soloshonok, V. Asymmetric synthesis of alpha-amino acids via homologation of Ni(II) complexes of glycine schiff bases. Part 3: Michael addition reactions and miscellaneous transformations. Amino Acids 2014, 46, 2047–2073. [Google Scholar] [CrossRef] [PubMed]
- Acena, J.L.; Sorochinsky, A.E.; Moriwaki, H.; Sato, T.; Soloshonok, V.A. ChemInform Abstract: Synthesis of Fluorine-Containing α-Amino Acids in Enantiomerically Pure Form via Homologation of Ni(II) Complexes of Glycine and Alanine Schiff Bases. J. Fluor. Chem. 2013, 155, 21–38. [Google Scholar] [CrossRef]
- Al-Resayes, S.I.; Shakir, M.; Shahid, N.; Azam, M.; Khan, A.U. Synthesis, spectroscopic characterization and in vitro antimicrobial studies of Schiff base ligand, H2L derived from glyoxalic acid and 1,8-diaminonaphthalene and its Co(II), Ni(II), Cu(II) and Zn(II) complexes. Arab. J. Chem. 2016, 9, 335–343. [Google Scholar] [CrossRef]
- Santosa, A.F.; Brottoa, D.F.; Favarina, L.R.V.; Cabezaa, N.A.; Andrade, G.R.; Batistote, M.; Alberto, A.; AdemirNeves, C.; Rodrigues, D.C.M.; dos Anjos, A. Study of the antimicrobial activity of metal complexes and their ligands through bioassays applied to plant extracts. Rev. Bras. Farmacogn. 2014, 24, 309–315. [Google Scholar] [CrossRef]
- Lewandowski, E.M.; Lethbridge, K.G.; Sanishvili, R.; Skiba, J.; Kowalski, K.; Chen, Y. Mechanisms of proton relay and product release by Class A β-lactamase at ultrahigh resolution. FEBS J. 2018, 285, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, E.M.; Szczupak, L.; Wong, S.; Skiba, J.; Guśpiel, A.; Solecka, J.; Vrček, V.; Kowalski, K.; Chen, Y. Antibacterial Properties of Metallocenyl-7-ADCA Derivatives and Structure in Complex with CTX-M β-Lactamase. Organometallics 2017, 36, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, M.; Patra, M.; Senges, C.H.R.; Ott, I.; Stepanek, J.J.; Pinto, A.; Prochnow, P.; Vuong, C.; Langklotz, S.; Metzler-Nolte, N.; et al. Analysis of the Mechanism of Action of Potent Antibacterial Hetero-tri-organometallic Compounds: A Structurally New Class of Antibiotics. ACS Chem. Biol. 2013, 8, 1442–1450. [Google Scholar] [CrossRef] [PubMed]
- Patra, M.; Gasser, G.; Metzler-Nolte, N. Small organometallic compounds as antibacterial agents. Dalton Trans. 2012, 41, 6350–6358. [Google Scholar] [CrossRef] [PubMed]
- Albada, H.B.; Prochnow, P.; Bobersky, S.; Bandow, J.E.; Metzler-Nolte, N. Highly active antibacterial ferrocenoylated or ruthenocenoylated Arg-Trp peptides can be discovered by an l-to-d substitution scan. Chem. Sci. 2014, 5, 4453–4459. [Google Scholar] [CrossRef]
- Jabłonski, A.; Matczak, K.; Koceva-Chyła, A.; Durka, K.; Steverding, D.; Jakubiec-Krzesniak, K.; Solecka, J.; Trzybinski, D.; Wozniak, K.; Andreu, V.; et al. Cymantrenyl-Nucleobases: Synthesis, Anticancer, Antitrypanosomal and Antimicrobial Activity Studies. Molecules 2017, 22, 2220. [Google Scholar] [CrossRef] [PubMed]
- Khanmohammadi, H.; Darvishpour, M. New azo ligands containing azomethine groups in the pyridazine-based chain: Synthesis and characterization. Dyes Pigments 2009, 81, 167–173. [Google Scholar] [CrossRef]
- El-Baradie, K.; El-Sharkawy, R.; El-Ghamry, H.; Sakai, K. Synthesis and characterization of Cu(II), Co(II) and Ni(II) complexes of a number of sulfadrug azodyes and their application for wastewater treatment. Spectrochim. Acta Part A 2014, 121, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Shakir, M.; Azam, M.; Ullah, M.F.; Hadi, S.M. Synthesis, spectroscopic and electrochemical studies of N,N-bis[(E)-2-thienylmethylidene]-1,8-naphthalenediamine and its Cu(II) complex: DNA cleavage and generation of superoxide anion. J. Photochem. Photobiol. B 2011, 104, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Warad, I.; Khan, A.A.; Azam, M.; Al-Resayes, S.I.; Haddad, S.F. Design and structural studies of diimine/CdX2 (X = Cl, I) complexes based on 2,2-dimethyl-1,3-diaminopropane ligand. J. Mol. Struct. 2014, 1062, 167–173. [Google Scholar] [CrossRef]
- Azam, M.; Al-Resayes, S.I.; Velmurugan, G.; Venuvanalingam, P.; Wagler, J.; Kroke, E. Novel uranyl(vi) complexes incorporating propylene-bridged salen-type N2O2-ligands: A structural and computational approach. Dalton Trans. 2015, 44, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Sheikhshoaie, I.; Saheb, V. A new salen base 5-(phenylazo)-N-(2-amino pyridine) salicyliden Schiff base ligand: Synthesis, experimental and density functional studies on its crystal structure, FTIR, 1H-NMR and 13C NMR spectra. Spectrochim. Acta Part A 2010, 77, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Qian, W.; Xia, Z.; Zou, Y.; Wan, S.; Shen, S. Investigation of third-order nonlinearity of an azo dye and its metal-substituted compounds. Chem. Phys. Lett. 2000, 330, 535–540. [Google Scholar] [CrossRef]
- Manonmani, J.; Kandaswamy, M.; Narayanan, V.; Thirumurugan, R.; Sundura, S.S.; Shanmugam, G.; Ponnuswamy, M.N.; Fun, H.K. Synthesis of copper(II) and nickel(II) complexes using compartmental ligands: X-ray, electrochemical and magnetic studies. Polyhedron 2001, 20, 3039. [Google Scholar] [CrossRef]
- Cotton, F.A.; Wilkinson, G. Advanced Inorganic Chemistry, 5th ed.; Wiley: New York, NY, USA, 1988. [Google Scholar]
- Firdaus, F.; Fatma, K.; Azam, M.; Khan, S.N.; Khan, A.U.; Shakir, M. Template synthesis and physicochemical studies of 14-membered hexaazmacrocyclic complexes with Co(II), Ni(II), Cu(II) and Zn(II): A comparative spectroscopic approach on DNA binding with Cu(II) and Ni(II) complexes. Trans. Met. Chem. 2008, 33, 467–473. [Google Scholar] [CrossRef]
- Masoud, M.S.; El-Dussouky, A.; Ghatwary, E.E. Geometry of New Iron (III) and Copper (II) Azo-Nitroso complexes Trans. Met. Chem. 1986, 11, 161–164. [Google Scholar] [CrossRef]
- Fukui, K.; Yonezawa, T.; Shingu, H.J. A Molecular Orbital Theory of Reactivity in Aromatic Hydrocarbons. J. Chem. Phys. 1952, 20, 722–725. [Google Scholar] [CrossRef]
- Lynam, M.M.; Kuty, M.; Damborsky, J.; Koca, J.; Adriaens, P. Molecular orbital calculations to describe microbial reductive dechlorination of polychlorinated dioxins. Environ. Toxicol. Chem. 1998, 17, 988–997. [Google Scholar] [CrossRef]
- Alam, M.; Alam, M.J.; Azaz, S.; Parveen, M.; Park, S.; Ahmad, S. DFT/TD-DFT calculations, spectroscopic characterizations (FTIR, NMR, UV-vis), molecular docking and enzyme inhibition study of 7-benzoyloxycoumarin. Comput. Biol. Chem. 2018, 73, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Orojloo, M.; Zolgharnein, P.; Solimannejad, M.; Amani, S. Synthesis and characterization of cobalt (II), nickel (II), copper (II) and zinc (II) complexes derived from two Schiff base ligands: Spectroscopic, thermal, magnetic moment, electrochemical and antimicrobial studies. Inorg. Chim. Acta 2017, 467, 227–237. [Google Scholar] [CrossRef]
- Patel, R.N.; Singh, Y.; Singh, Y.P.; Butcher, R.J.; Kamal, A.; Tripathi, I.P. Copper(II) and nickel(II) complexes with N′-[(Z)-phenyl(pyridin-2-yl)methylidene]acetohydrazide: Synthesis, crystal structures, DFT calculations and antioxidant effects. Polyhedron 2016, 117, 20–34. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Higuchi, M. From metal complexes to metallosupramolecular polymers via polycondensation: Synthesis, structure and electrochromic properties of Co(III)- and Fe(III)-based metallosupramolecular polymers with aromatic azo ligands. Eur. Polym. J. 2013, 49, 1688–1697. [Google Scholar] [CrossRef]
- Ferro, R.; Milione, S.; Caruso, T.; Grassi, A. Iron(III) complexes of bidentate nitrogen ligands as catalysts in reverse atom transfer radical polymerization of styrene. J. Mol. Catal. A Chem. 2009, 307, 128–133. [Google Scholar] [CrossRef]
- Frisch, M.J. Gaussian 09, Revision D.01; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Beck, A.D. Density-functional exchange-energy approximation with correct asymptotic behaviour. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the colle-salvetticorrelationenergy formula into a functional of the electron density. Phys. Rev. A 1988, 37, 785–789. [Google Scholar]
- Dennington, R.; Keith, T.; Millam, J. GaussView; Ver. 5; Semichem Inc.: Shawnee Mission, KS, USA, 2009. [Google Scholar]
- Chermette, H. Chemical reactivity indexes in density functional theory. J. Comput. Chem. 1999, 20, 129–154. [Google Scholar] [CrossRef]
- Perez, C.; Pauli, M.; Bazerque, P. An antibiotic assay by the agar well diffusion method. Acta Biol. Med. Exp. 1990, 15, 113–115. [Google Scholar]
- Aqil, F.; Khan, M.S.A.; Ahmed, I. Effect of certain bioactive plant extracts on clinical isolates of beta-lactamase producing methicillin resistant Staphylococcus aureus. J. Basic Microbiol. 2005, 45, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Cruickshank, R.; Duguid, J.P.; Marmion, B.P.; Swain, R.H.A. Medicinal Microbiology, 12th ed.; Churchill Livingstone: London, UK, 1975; Volume 2, pp. 196–202. [Google Scholar]
- Doughari, J.H.; Nuya, S.P. In Vitro antifungal activity of deterium microcarpum. Pak. J. Med. Sci. 2008, 24, 91–95. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Ligand | |
|---|---|
| HOMO | −0.2040 (−5.50 eV) |
| LUMO | −0.0722 (−1.96 eV) |
| ΔE | 0.1302 (3.54 eV) |
| Mulliken Electronegativity χ | 0.1373 |
| Global Hardness η | 0.0651 |
| Absolute softness σ | 15.3609 |
| Chemical Potential μ | −0.1373 |
| Global Softness S | 7.6804 |
| Global Electrophilicity ω | 0.1447 |
| Electronic Charge ΔN | 2.1090 |
| Dipole Moment | 3.4276 |
| Bond Length (Å) | B3LYP/6-31G (d,p) | Ref. [6] | Bond Angles (°) | B3LYP/6-31G (d,p) | Literature |
|---|---|---|---|---|---|
| N15-N16 | 1.232 | 1.25 | N15-N16-C22 | 119.9 | 112.2 |
| C19-O27 | 1.430 | 1.367 | N15-N16-C3 | 120.0 | 113.4 |
| C28-N26 | 1.293 | 1.304 | C19-N20-C21 | 119.9 | 120.0 |
| C31-N40 | 1.343 | 1.341 | C19-N20-C28 | 120.0 | 121.2 |
| C31-N26 | 1.399 | 1.397 | Dihedral angles (°) | B3LYP/6-31G (d,p) | Ref |
| C3-N15 | 1.470 | 1.428 | C3-N15-N16-C22 | −179.9 | −178.1 |
| O27-H29 | 0.960 | 0.840 | C20-C28-N26-C31 | −179.9 | -- |
| C20-C21 | 1.401 | 1.390 | C3-N15-N16-C22 | −179.9 | -- |
| Bond angles (°) | B3LYP/6-31G (d,p) | Ref. [6] | C21-C20-C19-O27 | −179.9 | −178.4 |
| C28-N26-C31 | 119.9 | 116.6 | C28-C20-C19-O27 | 0.0 | −1.1 |
| C31-N40-C32 | 121.4 | 118.1 | C21-C20-C19-C18 | 0.0 | 0.0 |
| C19-O27-H29 | 109.4 | 109.5 | C28-C20-C19-C18 | −179.9 | 178.4 |
| Compound Bacterial Strains | Inhibition Zone (mm) | MIC and MBC (μg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligand HL | Complex 1 | Complex 2 | Cam * | DMSO | MIC (MBC) of HL | MIC (MBC) of complex 1 | MIC (MBC) of complex 2 | MIC (MBC) of Cam | |
| S. Aureus (MRSA +ve) | 7 | 13 | 11 | 17 | No zone | 512 (512) | 128 (128) | 256 (256) | 32 (32) |
| E. Coli (Gram −ve) | 9 | 15 | 17 | 20 | No zone | 256 (>256) | 64 (256) | 256 (>256) | 32 (128) |
| Fungal strains | Ligand HL | Complex 1 | Complex 2 | Nys * | MIC (MBC) of HL | MIC (MBC) of complex 1 | MIC (MBC) of complex 2 | MIC (MFC) of Nys | |
| C. Albicans | No zone | No zone | No zone | 22 | No zone | ND | ND | ND | 50 (50) |
| A. Niger | No zone | No zone | No zone | 20 | No zone | ND | ND | ND | 200 (200) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azam, M.; Al-Resayes, S.I.; Wabaidur, S.M.; Altaf, M.; Chaurasia, B.; Alam, M.; Shukla, S.N.; Gaur, P.; Albaqami, N.T.M.; Islam, M.S.; et al. Synthesis, Structural Characterization and Antimicrobial Activity of Cu(II) and Fe(III) Complexes Incorporating Azo-Azomethine Ligand. Molecules 2018, 23, 813. https://doi.org/10.3390/molecules23040813
Azam M, Al-Resayes SI, Wabaidur SM, Altaf M, Chaurasia B, Alam M, Shukla SN, Gaur P, Albaqami NTM, Islam MS, et al. Synthesis, Structural Characterization and Antimicrobial Activity of Cu(II) and Fe(III) Complexes Incorporating Azo-Azomethine Ligand. Molecules. 2018; 23(4):813. https://doi.org/10.3390/molecules23040813
Chicago/Turabian StyleAzam, Mohammad, Saud I. Al-Resayes, Saikh Mohammad Wabaidur, Mohammad Altaf, Bhaskar Chaurasia, Mahboob Alam, Satyendra Nath Shukla, Pratiksha Gaur, Nader Talmas M. Albaqami, Mohammad Shahidul Islam, and et al. 2018. "Synthesis, Structural Characterization and Antimicrobial Activity of Cu(II) and Fe(III) Complexes Incorporating Azo-Azomethine Ligand" Molecules 23, no. 4: 813. https://doi.org/10.3390/molecules23040813
APA StyleAzam, M., Al-Resayes, S. I., Wabaidur, S. M., Altaf, M., Chaurasia, B., Alam, M., Shukla, S. N., Gaur, P., Albaqami, N. T. M., Islam, M. S., & Park, S. (2018). Synthesis, Structural Characterization and Antimicrobial Activity of Cu(II) and Fe(III) Complexes Incorporating Azo-Azomethine Ligand. Molecules, 23(4), 813. https://doi.org/10.3390/molecules23040813






