Chemical Modification of Chitosan for Efficient Vaccine Delivery
Abstract
:1. Introduction
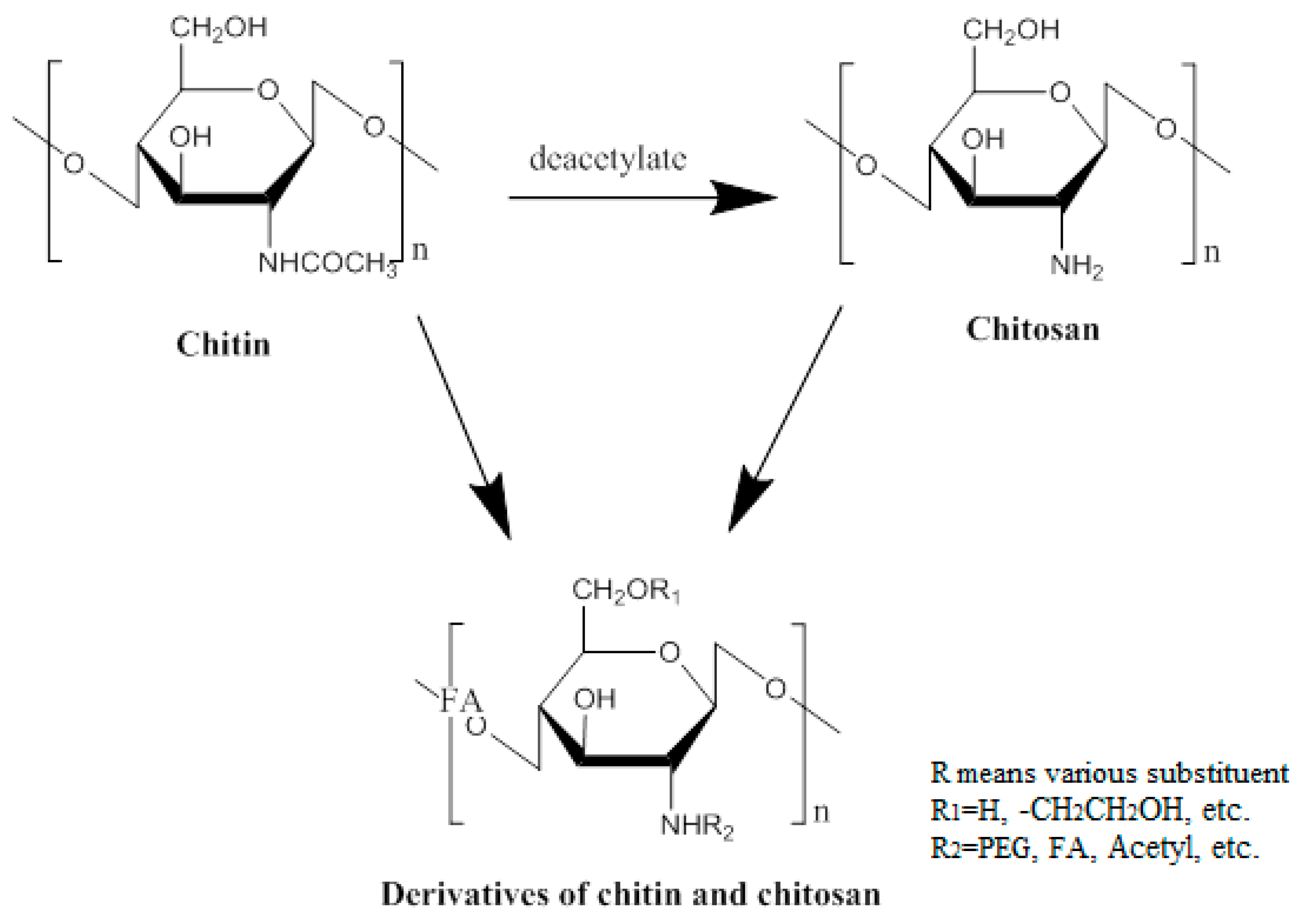
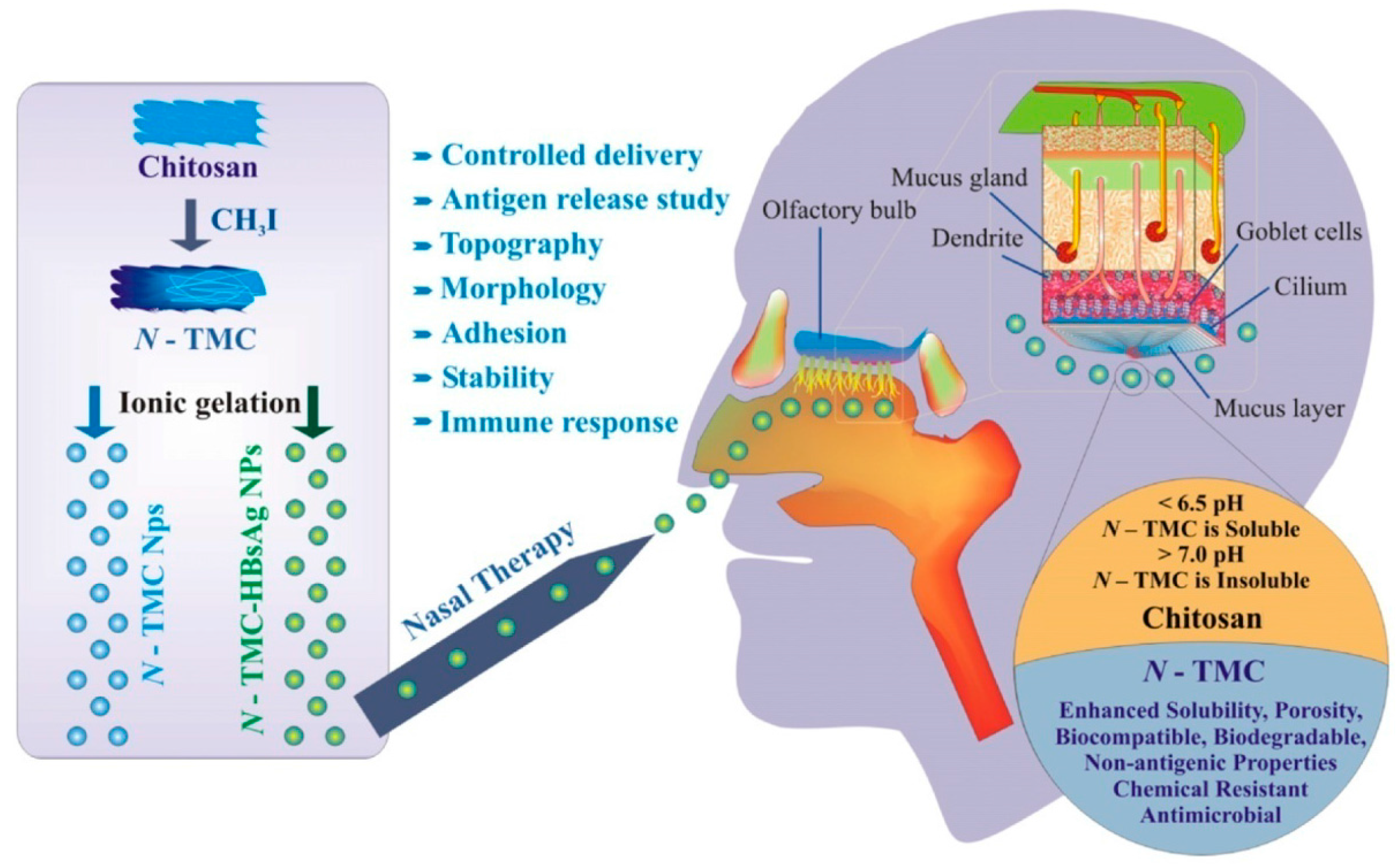
2. Chemical Modification
2.1. Hydrophilic Modification
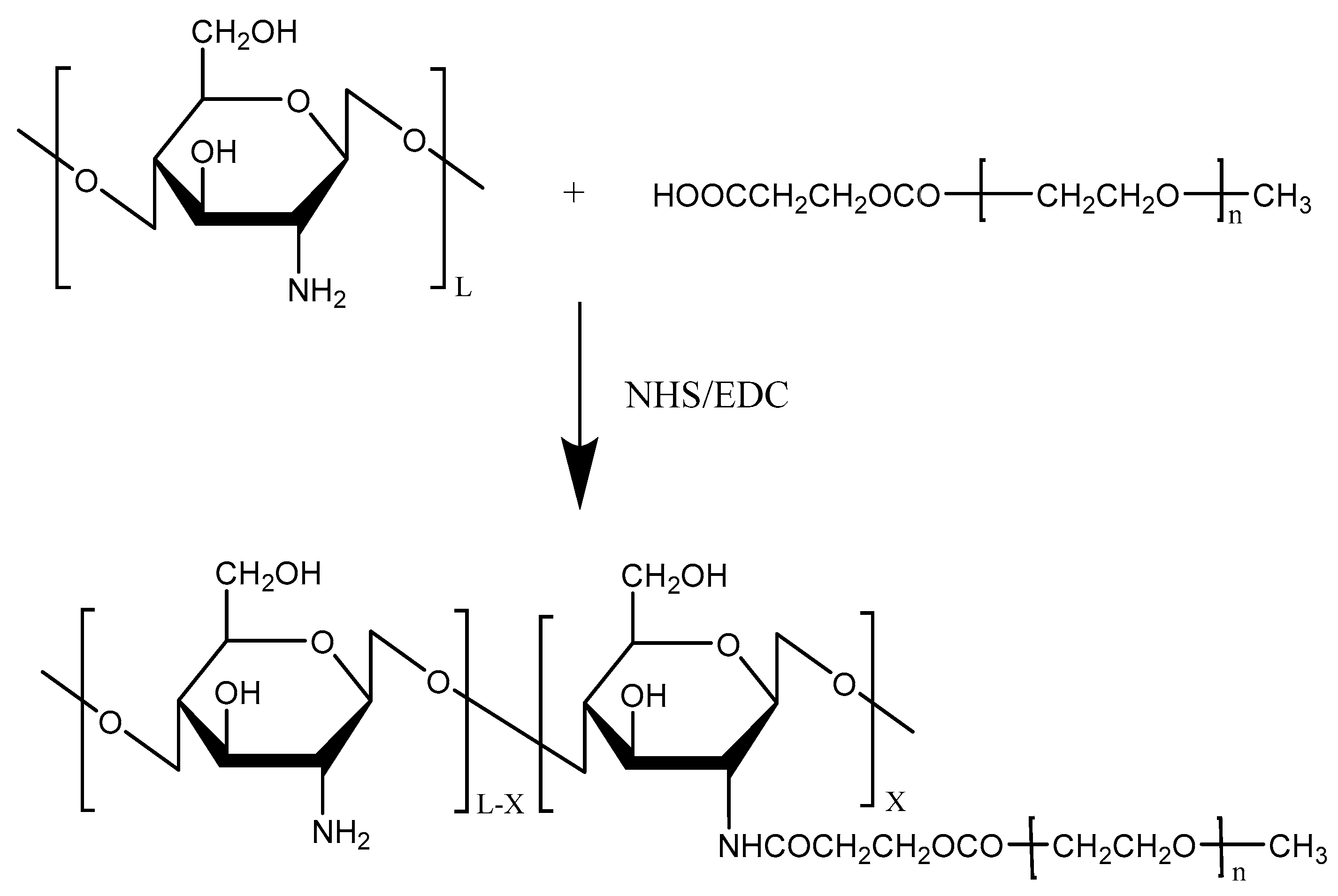
2.1.1. Poly (Ethylene Glycol) (PEG)
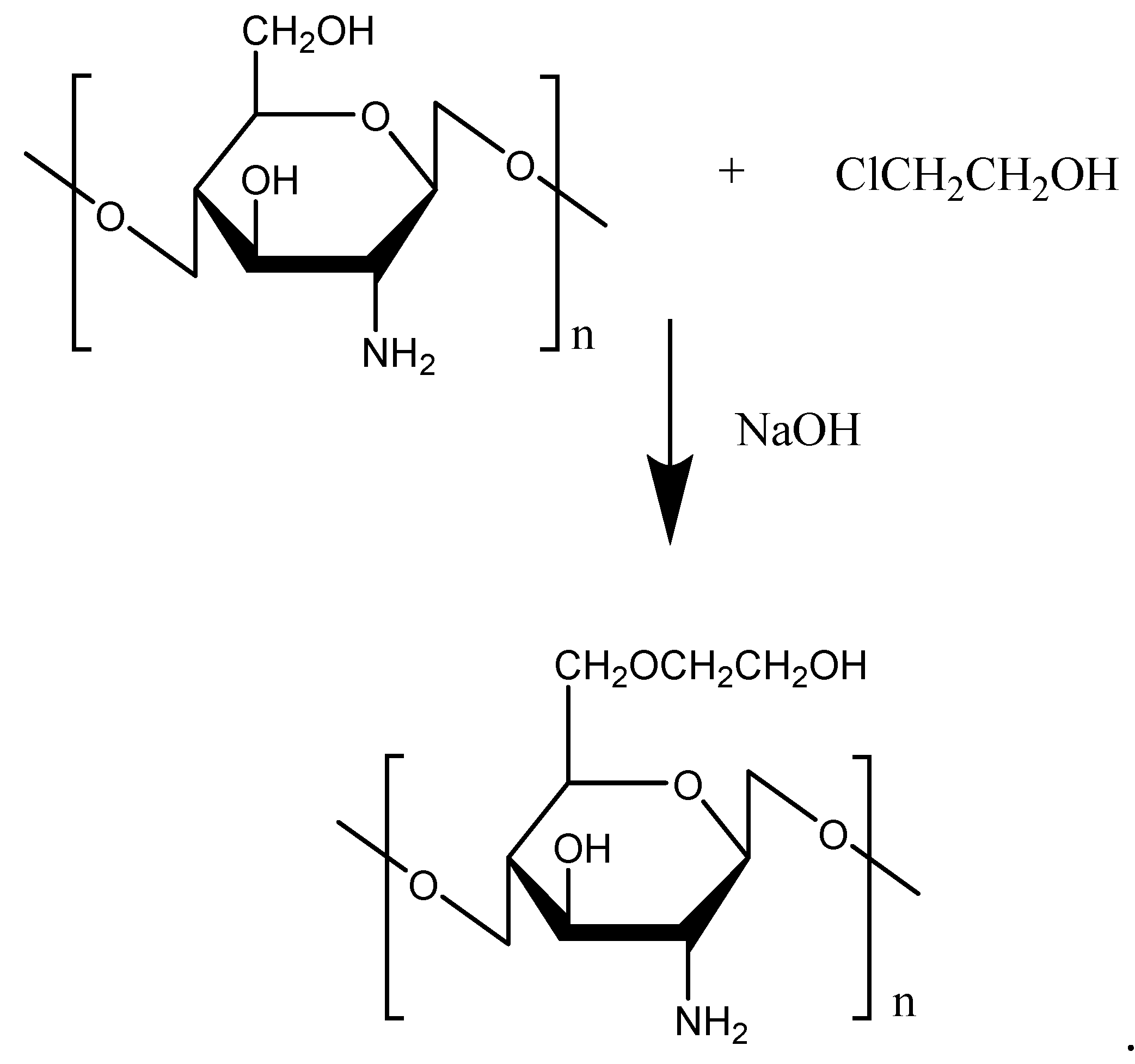
2.1.2. Glycol
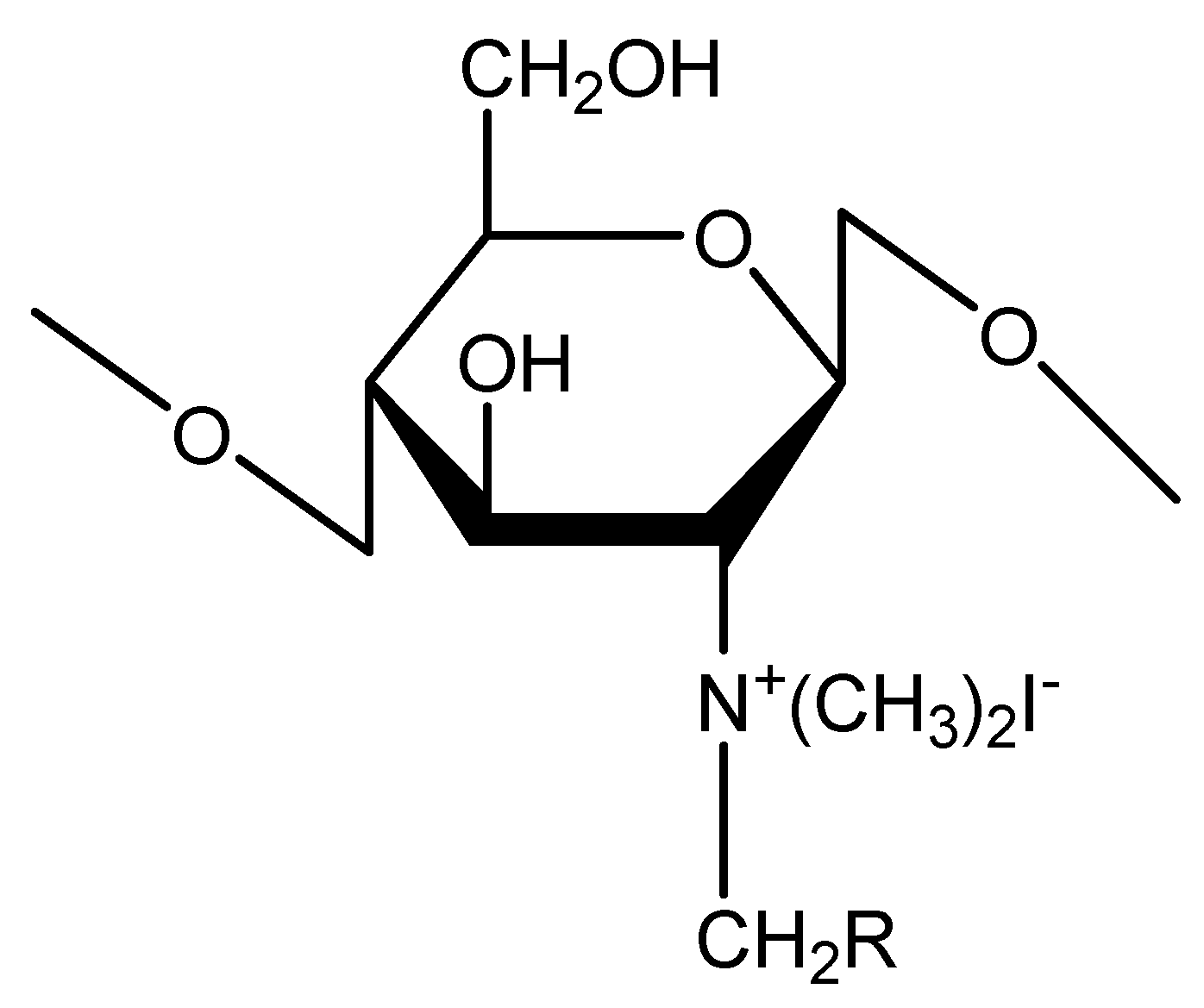
2.1.3. Poloxamer
2.2. Hydrophobic Modification
2.3. Specific Ligand Modification
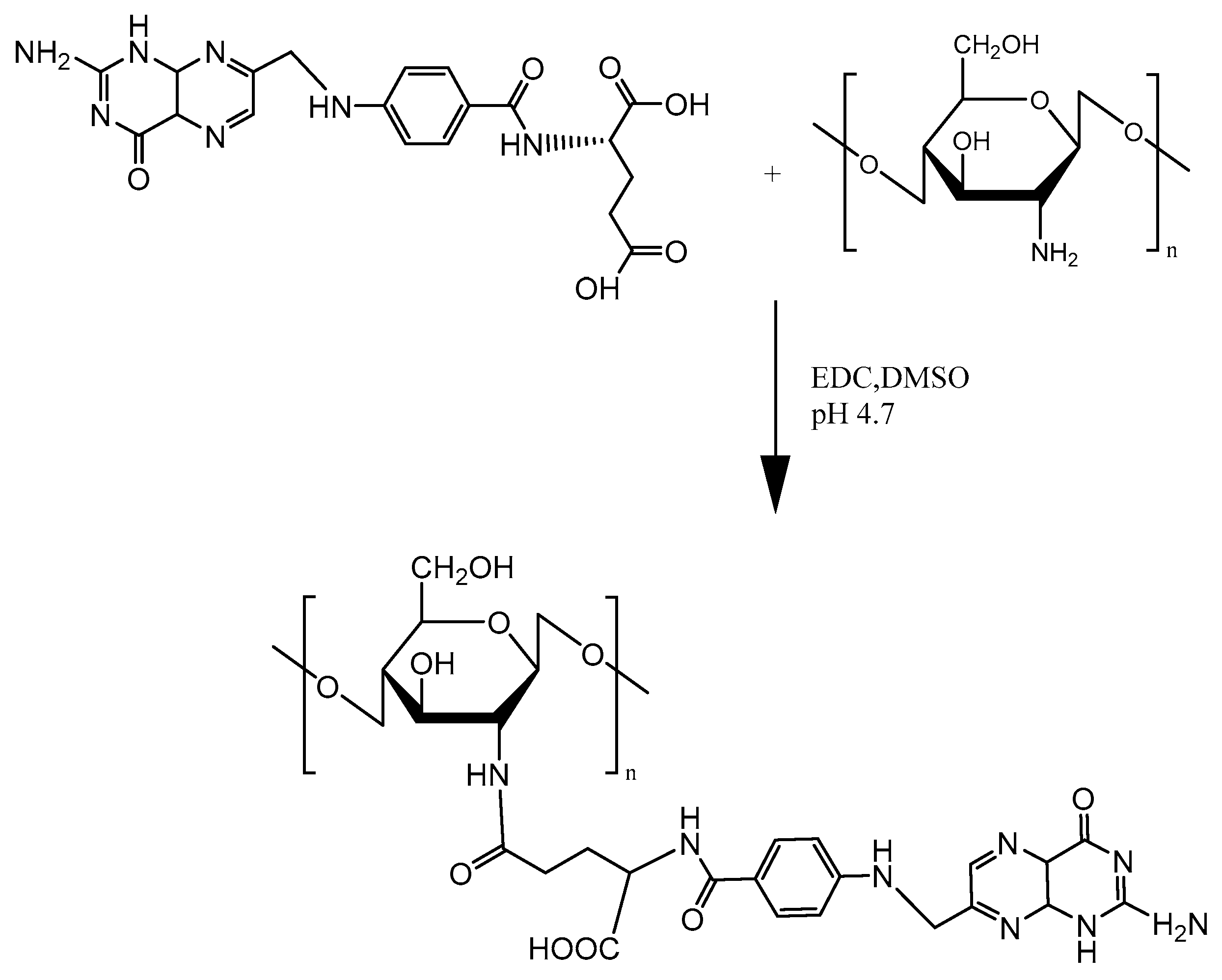
2.3.1. Folate
2.3.2. Mannose
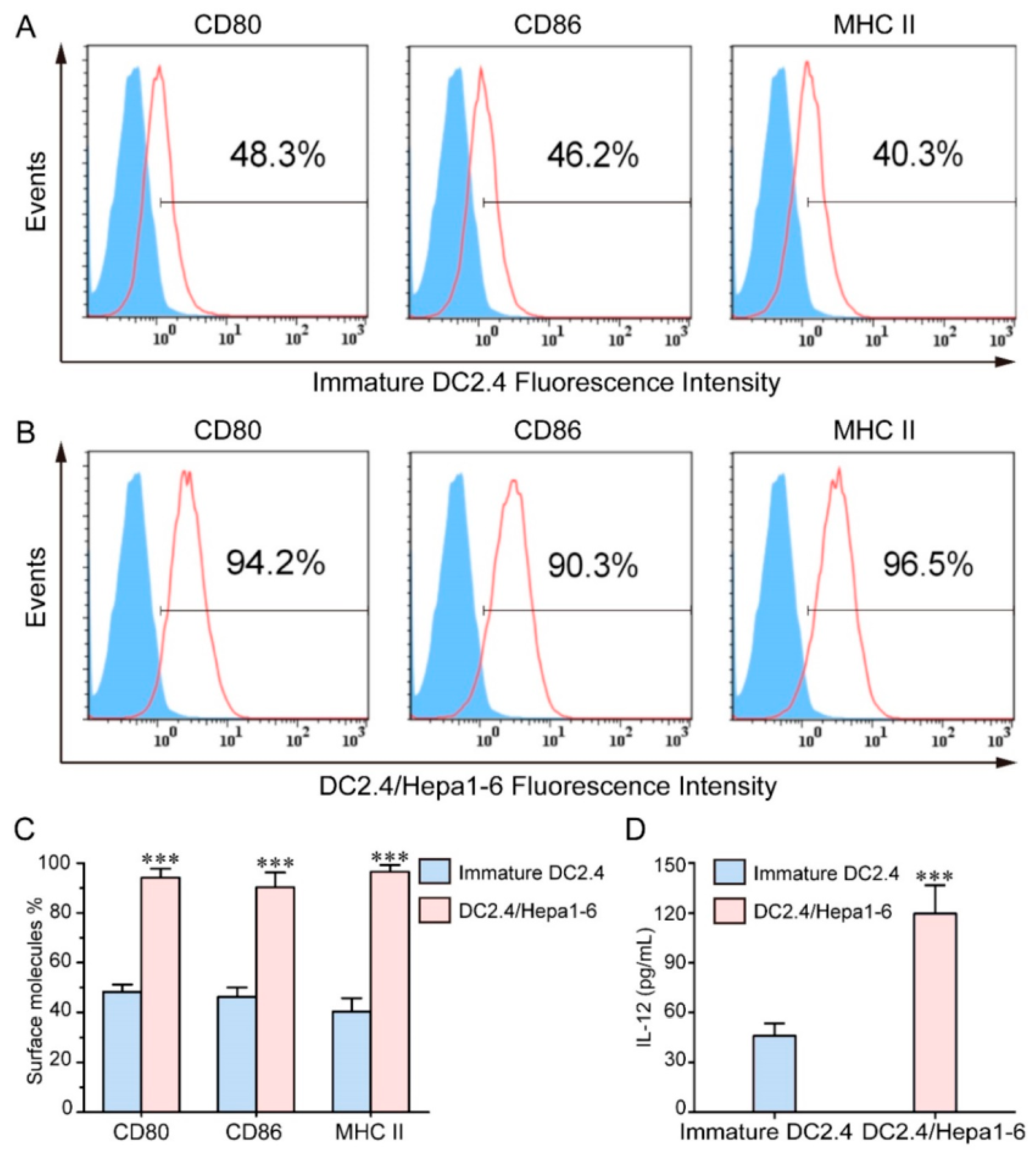
2.4. M-Cell Targeting Modification
2.4.1. Characteristics of M Cells
2.4.2. Targeting Ligand
Peptide Ligand
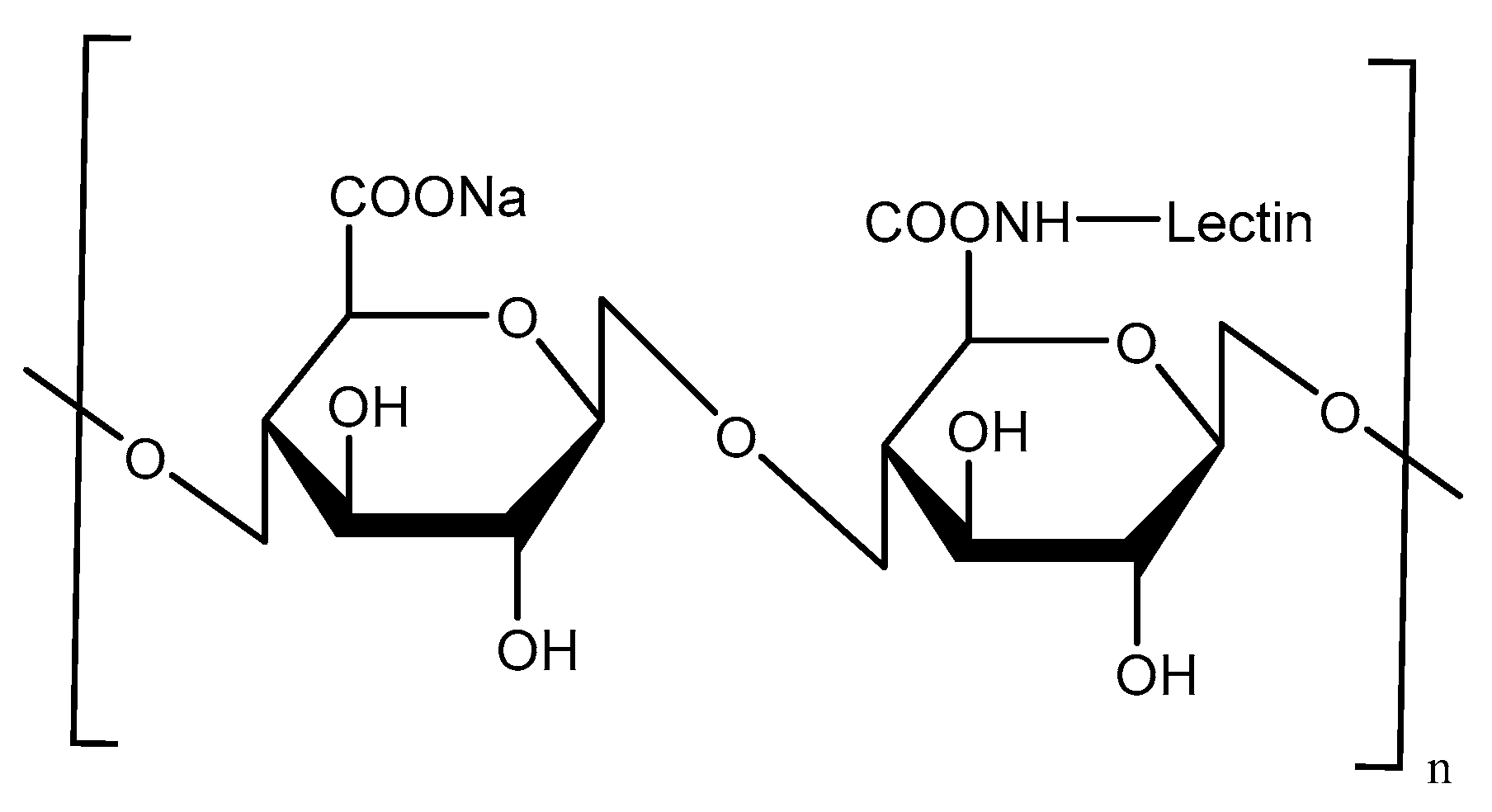
Lectin-Mediated Targeting
3. DNA Vaccine
4. Conclusions and Future Perspectives
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Keitel, W.A.; Cate, T.R.; Couch, R.B.; Huggins, L.L.; Hess, K.R. Efficacy of repeated annual immunization with inactivated influenza virus vaccines over a five year period. Vaccine 1997, 15, 1114–1122. [Google Scholar] [CrossRef]
- Arakawa, T.; Chong, D.K.; Langridge, W.H. Efficacy of a food plant-based oral cholera toxin B subunit vaccine. Nat. Biotechnol. 1998, 16, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Blisnick, T.; Ave, P.; Huerre, M.; Carniel, E.; Demeure, C.E. Oral vaccination against bubonic plague using a live avirulent Yersinia pseudotuberculosis strain. Infect. Immun. 2008, 76, 3808–3816. [Google Scholar] [CrossRef] [PubMed]
- Walter, E.B.; Hornick, R.B.; Poland, G.A.; Tucker, R.; Bland, C.L.; Clements, D.A.; Rhamstine, C.C.; Jacobson, R.M.; Brown, L.; Gress, J.O. Concurrent administration of inactivated hepatitis A vaccine with immune globulin in healthy adults. Vaccine 1999, 17, 1468–1473. [Google Scholar] [CrossRef]
- Denis, G.; René, T.; Nadia, K.; Angermann, B.R.; Geneviève, B.; Abdelali, F.M.; Moser, J.M.; Mehta, R.S.; Iii, D.D.R.; Erika, C. Yellow fever vaccine induces integrated multilineage and polyfunctional immune responses. J. Exp. Med. 2008, 205, 3119–3131. [Google Scholar]
- Dilraj, A.; Sukhoo, R.; Cutts, F.T.; Bennett, J.V. Aerosol and subcutaneous measles vaccine: Measles antibody responses 6 years after re-vaccination. Vaccine 2007, 25, 4170–4174. [Google Scholar] [CrossRef] [PubMed]
- White, C.J.; Stinson, D.; Staehle, B.; Cho, I.; Matthews, H.; Ngai, A.; Keller, P.; Eiden, J.; Kuter, B. The MMRV Vaccine Study Group. Measles, Mumps, Rubella, and Varicella Combination Vaccine: Safety and Immunogenicity Alone and in Combination with Other Vaccines Given to Children. Clin.Infectious Dis. 1997, 24, 925–931. [Google Scholar] [CrossRef]
- Cusi, M.G.; Metelli, R.; Valensin, P.E. Immune responses to wild and vaccine rubella viruses after rubella vaccination. Arch. Virol. 1989, 106, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Halperin, S.A.; Barreto, L.; Eastwood, B.J.; Medd, L.; Guasparini, R.; Mills, E. Safety and immunogenicity of an acellular pertussis diphtheria tetanus vaccine given as a single injection with Haemophilus influenzae b conjugate vaccine. Vaccine 1997, 15, 295–300. [Google Scholar] [CrossRef]
- Anonymous. Diphtheria vaccine: WHO position paper. Wkly. Epidemiol. Rec. 2006, 81, 24–32. [Google Scholar]
- Lee, H.B.; Yoon, S.Y.; Singh, B.; Oh, S.H.; Cui, L.; Yan, C.; Kang, S.K.; Choi, Y.J.; Cho, C.S. Oral Immunization of FMDV Vaccine Using pH-Sensitive and Mucoadhesive Thiolated Cellulose Acetate Phthalate Microparticles. Tissue Eng. Regener. Med. 2017. [Google Scholar] [CrossRef]
- Rappuoli, R. Reverse vaccinology, a genome-based approach to vaccine development. Vaccine 2001, 19, 2688–2691. [Google Scholar] [CrossRef]
- Barocchi, M.A.; Rappuoli, R. Delivering vaccines to the people who need them most. Philos. Trans. R. Soc. Lond. 2015, 370, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Medical Advisory Committee of the Immune Deficiency Foundation; Shearer, W.T.; Fleisher, T.A.; Buckley, R.H.; Ballas, Z.; Ballow, M.; Blaese, R.M.; Bonilla, F.A.; Conley, M.E.; Cunningham-Rundles, C.; et al. Recommendations for live viral and bacterial vaccines in immunodeficient patients and their close contacts. J. Allergy Clin. Immunol. 2014, 133, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Muthumani, K.; Falzarano, D.; Reuschel, E.L.; Tingey, C.; Flingai, S.; Villarreal, D.O.; Wise, M.; Patel, A.; Izmirly, A.; Aljuaid, A. A synthetic consensus anti-spike protein DNA vaccine induces protective immunity against Middle East respiratory syndrome coronavirus in nonhuman primates. Sci. Transl. Med. 2015, 7, 301ra132. [Google Scholar] [CrossRef] [PubMed]
- Stachyra, A.; Rak, M.; Redkiewicz, P.; Madeja, Z.; Gawarecka, K.; Chojnacki, T.; Świeżewska, E.; Masnyk, M.; Chmielewski, M.; Sirko, A. Effective usage of cationic derivatives of polyprenols as carriers of DNA vaccines against influenza virus. Virol. J. 2017, 14, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Lynn, D.J.; Pulendran, B. The potential of the microbiota to influence vaccine responses. J. Leukocyte Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Shen, W.; Shu, J.; Kou, Z.; Jin, X. A novel polyepitope vaccine elicited HIV peptide specific CD4+T cell responses in HLA-A2/DRB1 transgenic mice. PLoS ONE 2017, 12, e0184207. [Google Scholar] [CrossRef] [PubMed]
- Steiner, H.H.; Bonsanto, M.M.; Beckhove, P.; Brysch, M.; Geletneky, K.; Ahmadi, R.; Schuele-Freyer, R.; Kremer, P.; Ranaie, G.; Matejic, D. Antitumor vaccination of patients with glioblastoma multiforme: A pilot study to assess feasibility, safety, and clinical benefit. J. Clin. Oncol. 2004, 22, 4272–4281. [Google Scholar] [CrossRef] [PubMed]
- Takamura-Ishii, M.; Miura, T.; Nakaya, T.; Hagiwara, K. Induction of antitumor response to fibrosarcoma by Newcastle disease virus-infected tumor vaccine. Environ. Sci. Pollut. Res. 2017, 34, 171. [Google Scholar] [CrossRef] [PubMed]
- Sahdev, P.; Ochyl, L.J.; Moon, J.J. Biomaterials for Nanoparticle Vaccine Delivery Systems. Pharm. Res. 2014, 31, 2563–2582. [Google Scholar] [CrossRef] [PubMed]
- Schwendener, R.A. Liposomes as vaccine delivery systems: A review of the recent advances. Ther. Adv. Vaccines 2014, 2, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.E.; Titball, R.; Williamson, D. Vaccine delivery using nanoparticles. Front. Cell. Infect. Microbiol. 2013, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Illum, L. Chitosan and its use as a pharmaceutical excipient. Pharm. Res. 1998, 15, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.L.; Park, I.K.; Kang, M.L.; Yoo, H.S.; Choi, Y.J.; Akaike, T.; Cho, C.S. Immune stimulating activity of an atrophic rhinitis vaccine associated to pegylated chitosan microspheres in vitro. Polym. Adv. Technol. 2007, 18, 220–225. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, J.; Cui, L.; Zhang, Y.; Li, W.; Li, C.; Shi, N.; Chen, Y.; Kong, W. Investigation into Efficiency of a Novel Glycol Chitosan-Bestatin Conjugate to Protect Thymopoietin Oligopeptides from Enzymatic Degradation. J. Pharm. Sci. 2016, 105, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Pawar, D.; Mangal, S.; Goswami, R.; Jaganathan, K.S. Development and characterization of surface modified PLGA nanoparticles for nasal vaccine delivery: Effect of mucoadhesive coating on antigen uptake and immune adjuvant activity. Eur. J. Pharm. Biopharm. 2013, 85, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Chen, Y.; Li, F.; Cui, L.; Shi, N.; Kong, W.; Zhang, Y. Synthesis, Characterization and In Vitro Evaluation of a Novel Glycol Chitosan-EDTA Conjugate to Inhibit Aminopeptidase-Mediated Degradation of Thymopoietin Oligopeptides. Molecules 2017, 22, 1253. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.W.; Hsu, S.H.; Tsai, J.S.; Chen, F.R.; Huang, P.J.; Ke, C.J.; Liao, Z.X.; Hsiao, C.W.; Lin, H.J.; Sung, H.W. Multifunctional core-shell polymeric nanoparticles for transdermal DNA delivery and epidermal Langerhans cells tracking. Biomaterials 2010, 31, 2425–2434. [Google Scholar] [CrossRef] [PubMed]
- Pawar, D.; Jaganathan, K.S. Mucoadhesive glycol chitosan nanoparticles for intranasal delivery of hepatitis B vaccine: Enhancement of mucosal and systemic immune response. Drug Deliv. 2016, 23, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Xuan, J.J.; Balakrishnan, P.; Dong, H.O.; Yeo, W.H.; Park, S.M.; Yong, C.S.; Choi, H.G. Rheological characterization and in vivo evaluation of thermosensitive poloxamer-based hydrogel for intramuscular injection of piroxicam. Int. J. Pharm. 2010, 395, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.L.; Jiang, H.L.; Kang, S.G.; Guo, D.D.; Lee, D.Y.; Cho, C.S.; Yoo, H.S. Pluronic F127 enhances the effect as an adjuvant of chitosan microspheres in the intranasal delivery of Bordetella bronchiseptica antigens containing dermonecrotoxin. Vaccine 2007, 25, 4602–4610. [Google Scholar] [CrossRef] [PubMed]
- Im, V.D. L.; Verhoef, J.C.; Borchard, G.; Junginger, H.E. Chitosan for mucosal vaccination. Adv. Drug Deliv. Rev. 2001, 52, 139–144. [Google Scholar]
- Subbiah, R.; Ramalingam, P.; Ramasundaram, S.; Kim, D.Y.; Park, K.; Ramasamy, M.K.; Choi, K.J. N,N,N-Trimethyl chitosan nanoparticles for controlled intranasal delivery of HBV surface antigen. Carbohydr. Polym. 2012, 89, 1289–1297. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Steenekamp, J.H.; Hamman, J.H. Cross-linked cationic polymer microparticles: Effect of N-trimethyl chitosan chloride on the release and permeation of ibuprofen. Drug Dev. Ind. Pharm. 2005, 31, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Maaden, K.V.D.; Sekerdag, E.; Schipper, P.; Kersten, G.; Jiskoot, W.; Bouwstra, J.A. Layer-by-layer assembly of inactivated poliovirus and N-trimethyl chitosan on pH-sensitive microneedles for dermal vaccination. Langmuir ACS J. Surf. Colloids 2015, 31, 8654–8660. [Google Scholar] [CrossRef] [PubMed]
- Suksamran, T.; Kowapradit, J.; Ngawhirunpat, T.; Rojanarata, T.; Sajomsang, W.; Pitaksuteepong, T.; Opanasopit, P. Oral Methylated N-Aryl Chitosan Derivatives for Inducing Immune Responses to Ovalbumin. Trop. J. Pharm. Res. 2012, 11, 899–908. [Google Scholar] [CrossRef]
- Li, Z.; Xiong, F.; He, J.; Dai, X.; Wang, G. Surface-functionalized, pH-responsive poly(lactic-co-glycolic acid)-based microparticles for intranasal vaccine delivery: Effect of surface modification with chitosan and mannan. Eur. J. Pharm. Biopharm. 2016, 109, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, S.; Cuie, Y.; Winnik, F.; Shi, Q.; Lavigne, P.; Benderdour, M.; Beaumont, E.; Fernandes, J.C. Characterization of folate-chitosan-DNA nanoparticles for gene therapy. Biomaterials 2006, 27, 2060–2065. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Chen, J.; Zhou, S.; Yang, N.; Duan, S.; Zhang, Z.; Su, J.; He, J.; Lu, X.; Zhao, Y. Mouse IP-10 Gene Delivered by Folate-modified Chitosan Nanoparticles and Dendritic/tumor Cells Fusion Vaccine Effectively Inhibit the Growth of Hepatocellular Carcinoma in Mice. Theranostics 2017, 7, 1942–1952. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.-L.; Kang, M.L.; Quan, J.-S.; Kang, S.G.; Akaike, T.; Yoo, H.S.; Cho, C.-S. The potential of mannosylated chitosan microspheres to target macrophage mannose receptors in an adjuvant-delivery system for intranasal immunization. Biomaterials 2008, 29, 1931–1939. [Google Scholar] [CrossRef] [PubMed]
- Harde, H.; Agrawal, A.K.; Jain, S. Tetanus toxoids loaded glucomannosylated chitosan based nanohoming vaccine adjuvant with improved oral stability and immunostimulatory response. Pharm. Res. 2015, 32, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Peng, Y.; Du, M.; Luo, J.; Zong, L. Preventative vaccine-loaded mannosylated chitosan nanoparticles intended for nasal mucosal delivery enhance immune responses and potent tumor immunity. Mol. Pharm. 2013, 10, 2904–2914. [Google Scholar] [CrossRef] [PubMed]
- Miller, H.; Zhang, J.; Kuolee, R.; Patel, G.B.; Chen, W. Intestinal M cells: The fallible sentinels? World J. Gastroenterol. 2007, 13, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Brandtzaeg, P. Mucosal immunity: Induction, dissemination, and effector functions. Scand. J. Immunol. 2009, 70, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Hobson, P.; Barnfield, C.; Barnes, A.; Klavinskis, L.S. Mucosal immunization with DNA vaccines. Methods 2003, 31, 217–224. [Google Scholar] [CrossRef]
- Jepson, M.A.; Clark, M.A. Studying M cells and their role in infection. Trends Microbiol. 1998, 6, 359–365. [Google Scholar] [CrossRef]
- Buda, A.; Sands, C.; Jepson, M.A. Use of fluorescence imaging to investigate the structure and function of intestinal M cells. Adv. Drug Deliv. Rev. 2005, 57, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T.; Gonda, T. Cellular kinetics of villous epithelial cells and m cells in rabbit small intestine. J. Vet. Med. Sci. 2004, 66, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Blanchinroland, S.; Masson, J.M. Mucosal Delivery of Vaccine by M Cell Targeting Strategies. Curr. Drug Ther. 2014, 9, 9–20. [Google Scholar]
- Yun, Y.; Cho, Y.W.; Park, K. Nanoparticles for oral delivery: Targeted nanoparticles with peptidic ligands for oral protein delivery. Adv. Drug Deliv. Rev. 2013, 65, 822–832. [Google Scholar] [CrossRef] [PubMed]
- Yoo, M.K.; Kang, S.K.; Choi, J.H.; Park, I.K.; Na, H.S.; Lee, H.C.; Kim, E.B.; Lee, N.K.; Nah, J.W.; Choi, Y.J. Targeted delivery of chitosan nanoparticles to Peyer’s patch using M cell-homing peptide selected by phage display technique. Biomaterials 2010, 31, 7738–7747. [Google Scholar] [CrossRef] [PubMed]
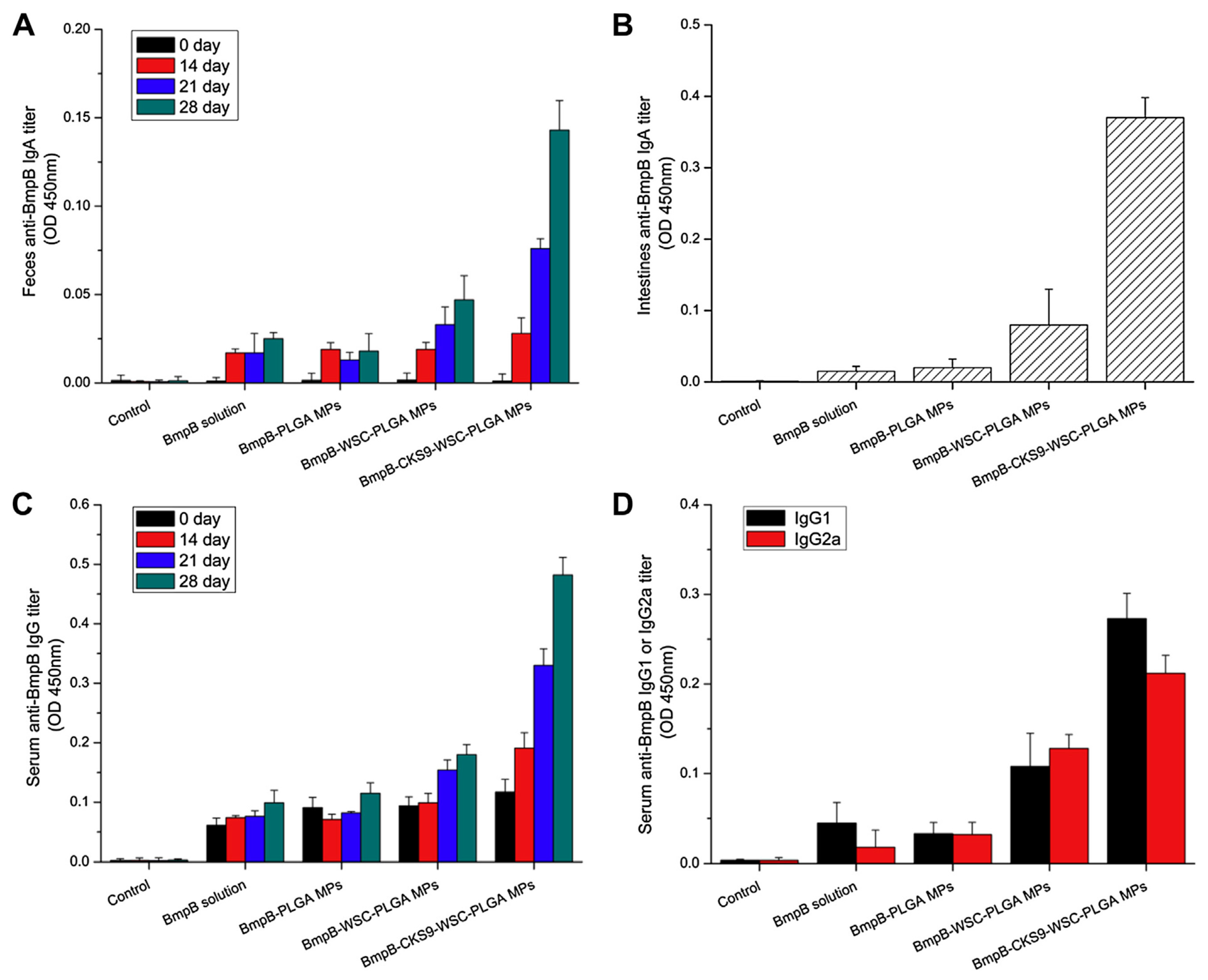
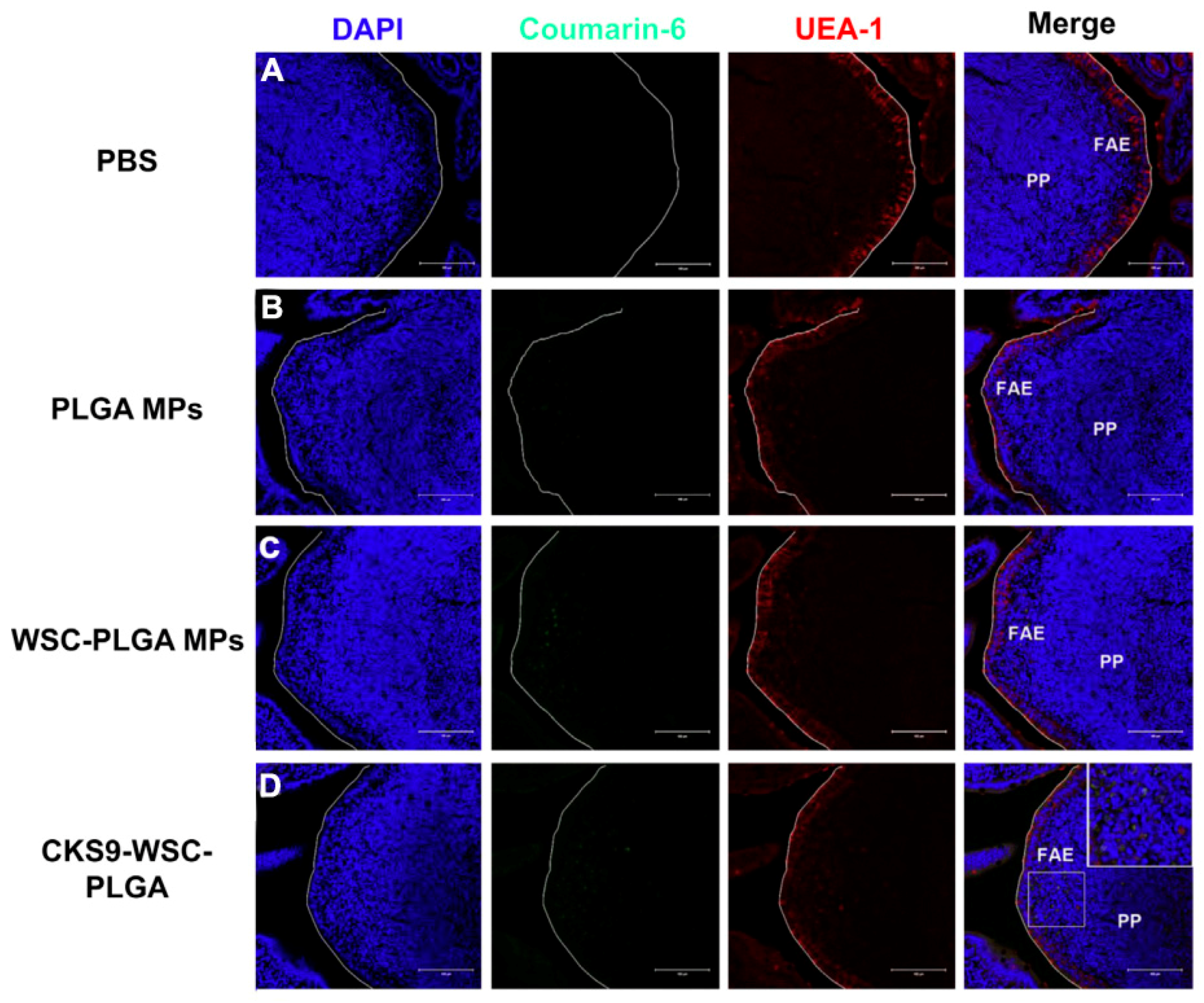
- Jiang, T.; Singh, B.; Li, H.S.; Kim, Y.K.; Kang, S.K.; Nah, J.W.; Choi, Y.J.; Cho, C.S. Targeted oral delivery of BmpB vaccine using porous PLGA microparticles coated with M cell homing peptide-coupled chitosan. Biomaterials 2014, 35, 2365–2373. [Google Scholar] [CrossRef] [PubMed]
- Ye, T.; Yue, Y.; Fan, X.; Dong, C.; Xu, W.; Xiong, S. M cell-targeting strategy facilitates mucosal immune response and enhances protection against CVB3-induced viral myocarditis elicited by chitosan-DNA vaccine. Vaccine 2014, 32, 4457–4465. [Google Scholar] [CrossRef] [PubMed]
- Holmgren, J.; Czerkinsky, C. Mucosal immunity and vaccines. Nat. Med. 2005, 11 (Suppl. 4), 45–53. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.N.; Khatri, K.; Goyal, A.K.; Mishra, N.; Vyas, S.P. M-cell targeted biodegradable PLGA nanoparticles for oral immunization against hepatitis B. J. Drug Target. 2007, 15, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Malik, B.; Goyal, A.K.; Markandeywar, T.S.; Rath, G.; Zakir, F.; Vyas, S.P. Microfold-cell targeted surface engineered polymeric nanoparticles for oral immunization. J. Drug Target. 2012, 20, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Sodhi, R.K.; Katyal, A.; Aneja, R.; Jain, U.K.; Katare, O.P.; Madan, J. Wheat germ agglutinin anchored chitosan microspheres of reduced brominated derivative of noscapine ameliorated acute inflammation in experimental colitis. Colloids Surf. B 2015, 132, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Torchilin, V.; Langer, R. Lectin-bearing polymerized liposomes as potential oral vaccine carriers. Pharm. Res. 1996, 13, 1378–1383. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Shen, Y.; Jiang, Z.; Wang, Y.; Chu, Y.; Xiong, S. Intranasal delivery of chitosan-DNA vaccine generates mucosal SIgA and anti-CVB3 protection. Vaccine 2004, 22, 3603–3612. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Liu, B.; Yu, X.; Zha, X.; Zhang, X.; Chen, Y.; Wang, X.; Jin, Y.; Wu, Y.; Chen, Y. Controlled release of PEI/DNA complexes from mannose-bearing chitosan microspheres as a potent delivery system to enhance immune response to HBV DNA vaccine. J. Control. Release 2007, 121, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Heuking, S.; Borchard, G. Toll-like receptor-7 agonist decoration enhances the adjuvanticity of chitosan-DNA nanoparticles. J. Pharm. Sci. 2012, 101, 1166–1177. [Google Scholar] [CrossRef] [PubMed]
- Pawar, S.N.; Edgar, K.J. Alginate derivatization: A review of chemistry, properties and applications. Biomaterials 2012, 33, 3279–3305. [Google Scholar] [CrossRef] [PubMed]
- Borges, O.; Silva, M.; De, S.A.; Borchard, G.; Junginger, H.E.; Cordeirodasilva, A. Alginate coated chitosan nanoparticles are an effective subcutaneous adjuvant for hepatitis B surface antigen. Int. Immunopharmacol. 2008, 8, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
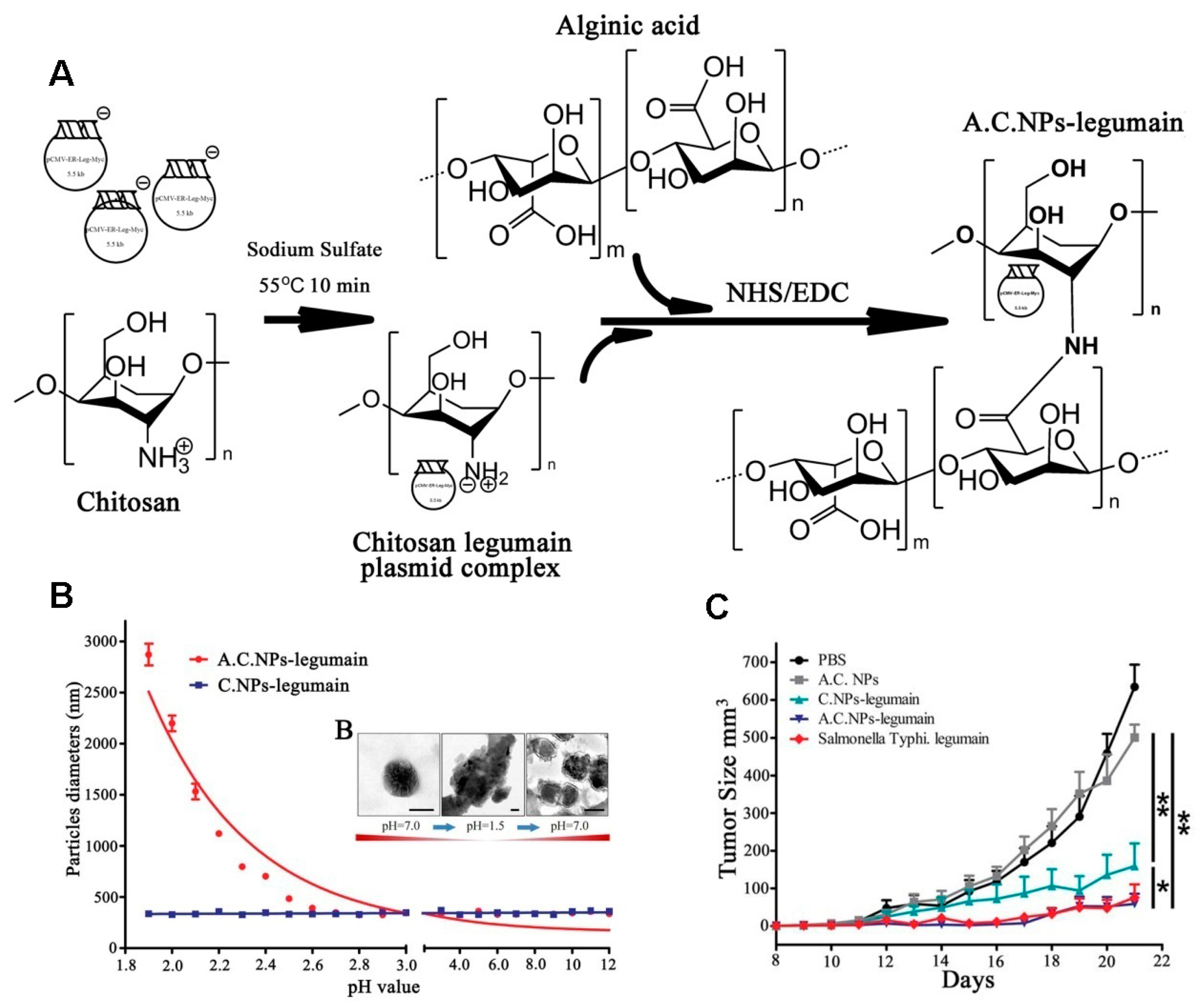
- Liu, Z.; Dan, L.; Shu, L.; Gong, J.; Da, W.; Min, X.; Chen, X.; Rong, X.; Tan, X. Alginic Acid-Coated Chitosan Nanoparticles Loaded with Legumain DNA Vaccine: Effect against Breast Cancer in Mice. PLoS ONE 2013, 8, e60190. [Google Scholar] [CrossRef] [PubMed]







© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xing, L.; Fan, Y.-T.; Zhou, T.-J.; Gong, J.-H.; Cui, L.-H.; Cho, K.-H.; Choi, Y.-J.; Jiang, H.-L.; Cho, C.-S. Chemical Modification of Chitosan for Efficient Vaccine Delivery. Molecules 2018, 23, 229. https://doi.org/10.3390/molecules23020229
Xing L, Fan Y-T, Zhou T-J, Gong J-H, Cui L-H, Cho K-H, Choi Y-J, Jiang H-L, Cho C-S. Chemical Modification of Chitosan for Efficient Vaccine Delivery. Molecules. 2018; 23(2):229. https://doi.org/10.3390/molecules23020229
Chicago/Turabian StyleXing, Lei, Ya-Tong Fan, Tian-Jiao Zhou, Jia-Hui Gong, Lian-Hua Cui, Ki-Hyun Cho, Yun-Jaie Choi, Hu-Lin Jiang, and Chong-Su Cho. 2018. "Chemical Modification of Chitosan for Efficient Vaccine Delivery" Molecules 23, no. 2: 229. https://doi.org/10.3390/molecules23020229
APA StyleXing, L., Fan, Y.-T., Zhou, T.-J., Gong, J.-H., Cui, L.-H., Cho, K.-H., Choi, Y.-J., Jiang, H.-L., & Cho, C.-S. (2018). Chemical Modification of Chitosan for Efficient Vaccine Delivery. Molecules, 23(2), 229. https://doi.org/10.3390/molecules23020229



