Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms
Abstract
1. Introduction
2. Results and Discussions
2.1. Proximate Composition
2.2. Fatty Acid Composition
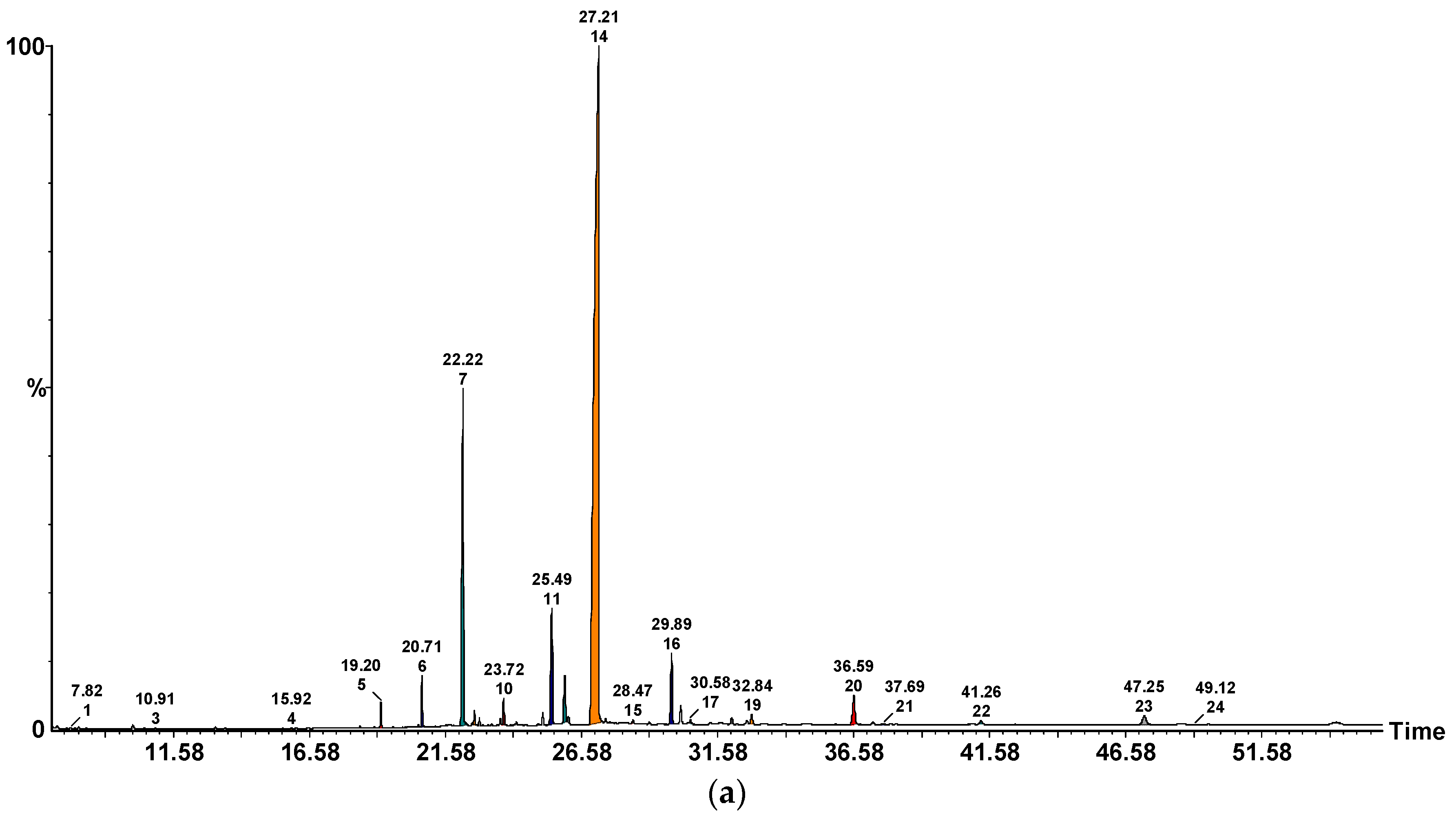
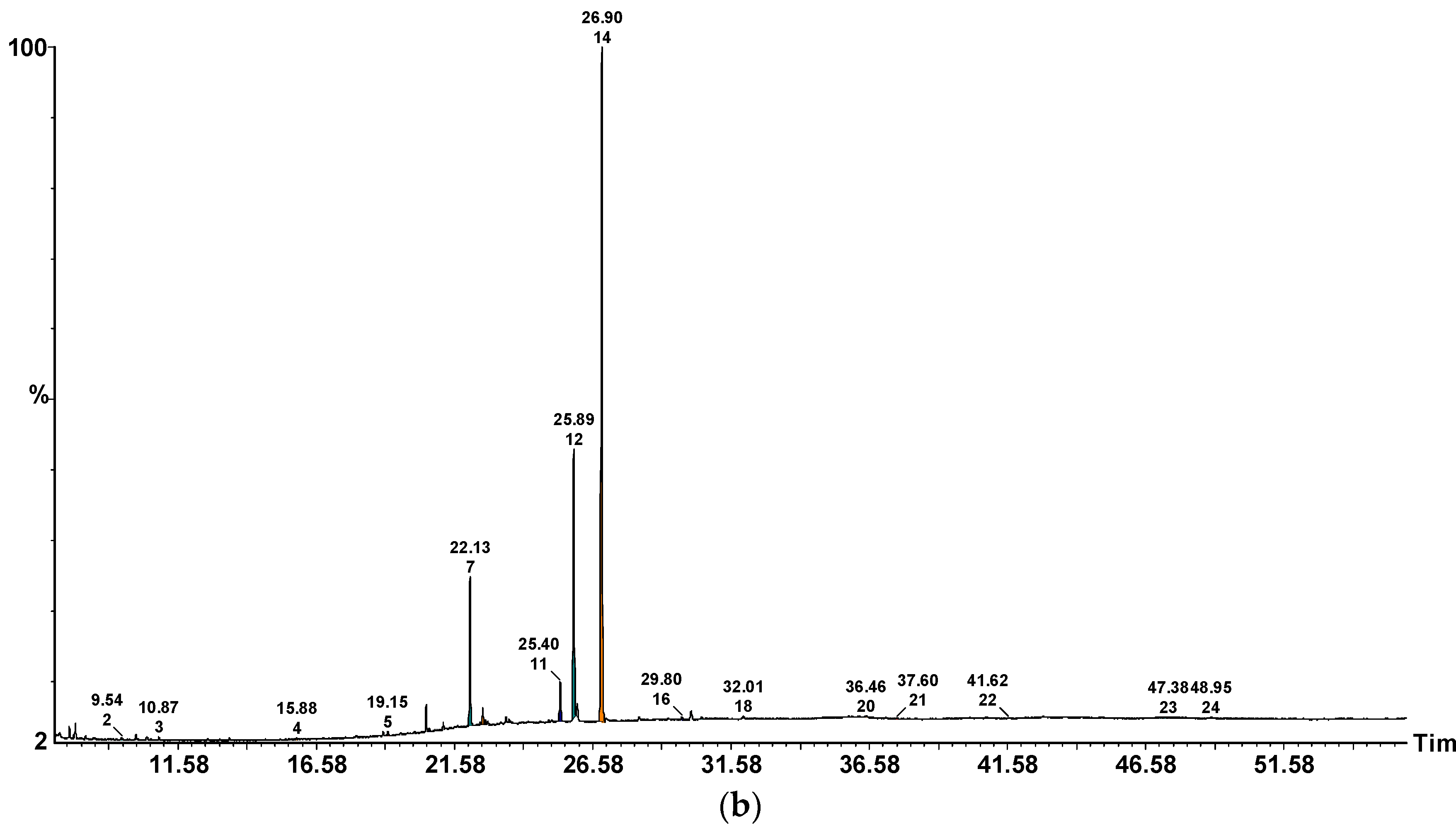
2.3. ITEX/GC-MS Profile of Volatile Compounds
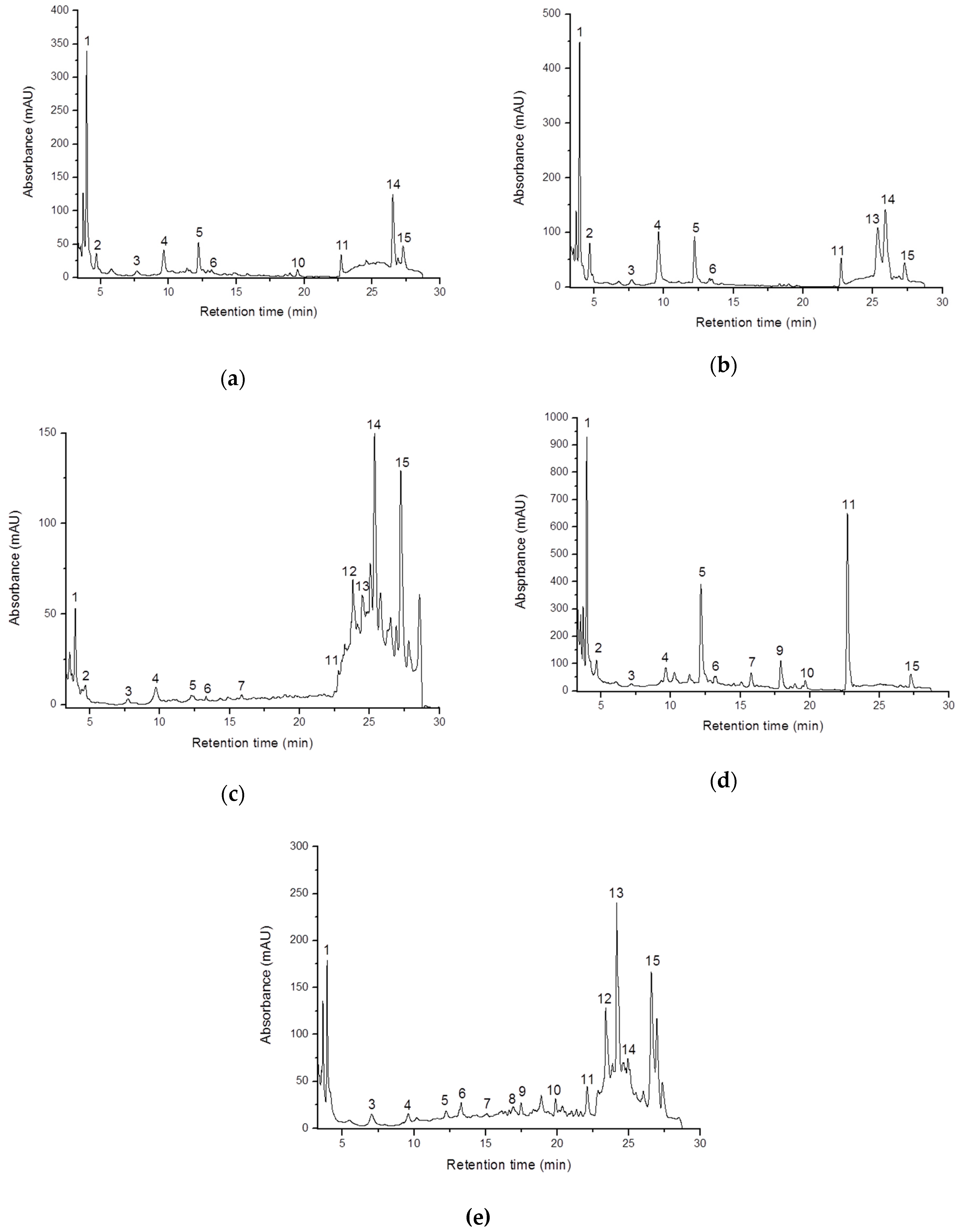
2.4. Phenolic Content
3. Materials and Methods
3.1. Experimental Material
3.2. Proximate Composition
3.3. Extraction of Lipids and Fatty Acid Composition
3.4 Extraction and Analysis of Volatile Compounds
3.5 HPLC–ESI/MS Identification Analysis of Phenolic Compounds
3.6. RP-HPLC-PDA Quantification Analysis of Phenolic Compounds
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Obodai, M.; Ferreira, I.C.; Fernandes, A.; Barros, L.; Mensah, D.L.; Dzomeku, M.; Urben, A.F.; Prempeh, J.; Takli, R.K. Evaluation of the chemical and antioxidant properties of wild and cultivated mushrooms of Ghana. Molecules 2014, 19, 19532–19548. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.S.; Pereira, E.; Barros, L.; Sousa, M.J.; Martins, A.; Ferreira, I.C. Biomolecule profiles in inedible wild mushrooms with antioxidant value. Molecules 2011, 16, 4328–4338. [Google Scholar] [CrossRef] [PubMed]
- Vamanu, E.; Nita, S. Antioxidant capacity and the correlation with major phenolic compounds, anthocyanin, and tocopherol content in various extracts from the wild edible Boletus edulis mushroom. BioMed Res. Int. 2013, 2013, 313905. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.S.; Martins, A.; Barros, L.; Ferreira, I.C. Antioxidant properties and phenolic profile of the most widely appreciated cultivated mushrooms: A comparative study between in vivo and in vitro samples. Food Chem. Toxicol. 2012, 50, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Giannenas, I.; Pappas, I.S.; Mavridis, S.; Kontopidis, G.; Skoufos, J.; Kyriazakis, I. Performance and antioxidant status of broiler chickens supplemented with dried mushrooms (Agaricus bisporus) in their diet. Poult. Sci. 2010, 89, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Giannenasa, I.; Tsalieb, E.; Chronisc, E.; Mavridisd, S.; Tontisb, D.; Kyriazakis, I. Consumption of Agaricus bisporus mushroom affects the performance, intestinal microbiota composition and morphology, and antioxidant status of turkey poults. Anim. Feed Sci. Technol. 2011, 165, 218–229. [Google Scholar] [CrossRef]
- Beluhan, S.; Ranogajec, A. Chemical composition and non-volatile components of Croatian wild edible mushrooms. Food Chem. 2011, 124, 1076–1082. [Google Scholar] [CrossRef]
- Kalac, P. Chemical Composition and Nutritional Value of European Species of Wild Growing Mushrooms; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2012. [Google Scholar]
- Colak, A.; Faiz, Ö.; Sesli, E. Nutritional composition of some wild edible mushrooms. Turk. J. Biochem. 2009, 34, 25–31. [Google Scholar]
- Heleno, S.A.; Barros, L.; Martins, A.; Morales, P.; Fernández-Ruiz, V.; Glamoclija, J.; Sokovic, M.; Ferreira, I.C.F.R. Nutritional value, bioactive compounds, antimicrobial activity and bioaccessibility studies with wild edible mushrooms. LWT–Food Sci. Technol. 2015, 63, 799–806. [Google Scholar] [CrossRef]
- Mattila, P.; Salo-Vaananen, P.; Konko, K.; Aro, H.; Jalava, T. Basic composition and amino acid contents of mushrooms cultivated in Finland. J. Agric. Food Chem. 2002, 50, 6419–6422. [Google Scholar] [CrossRef]
- Barros, L.; Venturini, B.A.; Baptista, P.; Estevinho, L.M.; Ferreira, I. Chemical composition and biological properties of Portuguese wild mushrooms: A comprehensive study. J. Agric. Food Chem. 2008, 56, 3856–3862. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.M.; Wu, W.W.; Li, G.K. AGC–MS study of the volatile organic composition of straw and oyster mushrooms during maturity and its relation to antioxidant activity. J. Chromatogr. Sci. 2008, 46, 690–696. [Google Scholar] [CrossRef] [PubMed]
- Leffingwell, J.C.; Alford, E.D. Volatile constituents of the giant Puffball mushroom (Calvatia gigantea). Leffingwell Rep. 2011, 4, 1–17. [Google Scholar]
- Palacios, I.; Lozano, M.; Moro, C.; D’Arrigo, M.; Rostagno, M.A.; Martínez, J.A.; García-Lafuente, A.; Guillamón, E.; Villares, A. Antioxidant properties of phenolic compounds occurring in edible mushrooms. Food Chem. 2011, 128, 674–678. [Google Scholar] [CrossRef]
- Reis, F.S.; Martins, A.; Vasconcelos, M.H.; Morales, P.; Ferreira, I.C.F.R. Functional foods based on extracts or compounds derived from mushrooms. Trends Food Sci. Technol. 2017, 66, 48–62. [Google Scholar] [CrossRef]
- Taofiq, O.; Calhelha, R.C.; Heleno, S.; Barros, L.; Martins, A.; Santos-Buelga, C.; Queiroz, M.; Ferreira, I. The contribution of phenolic acids to the anti-inflammatory activity of mushrooms: Screening in phenolic extracts, individual parent molecules and synthesized glucuronated and methylated derivatives. Food Res. Int. 2015, 76, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.Y.; Seguin, P.; Ahn, J.K.; Kim, J.J.; Chun, S.C.; Kim, E.H.; Seo, S.H.; Kang, E.Y.; Kim, S.L.; Park, Y.J.; et al. Phenolic compound concentration and antioxidant activities of edible and medicinal mushrooms from Korea. J. Agric. Food Chem. 2008, 56, 7265–7270. [Google Scholar] [CrossRef]
- Muszynska, B.; Katarzyna, K.; Szewczyk, A.; Łojewski, M.; Sulkowska-Ziaja, K.; Rojowski, J. Analysis of the content of phenolic compounds in in vitro culture of some edible mushrooms (Basidiomycota). Med. Int. Rev. 2015, 26, 146–152. [Google Scholar]
- Barros, L.; Duenas, M.; Ferreira, I.C.; Baptista, P.; Santos-Buelga, C. Phenolic acids determination by HPLC-DAD-ESI/MS in sixteen different Portuguese wild mushrooms species. Food Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Jakobek, L.; García-Villalba, R.; Tomás-Barberán, F.A. Polyphenolic characterisation of old local apple varieties from Southeastern European region. J. Food Compost. Anal. 2013, 31, 199–211. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 20 th Edition (2016). Available online: http://www.aoac.org/aoac_prod_imis/AOAC/Publications/Official_Methods_of_Analysis/AOAC_Member/Pubs/OMA/AOAC_Official_Methods_of_Analysis.aspx, (accessed on 5 December 2018).
- Mocan, A.; Fernandes, A.; Barros, L.; Crisan, G.; Smiljkovic, M.; Sokovic, M.; Ferreira, I. Chemical composition and bioactive properties of the wild mushroom Polyporus squamosus (Huds.) Fr: A study with samples from Romania. Food Funct. 2018, 9, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.; Mureşan, C.; Socaci, S.A.; Tofană, M.; Fărcaș, A.; Biriş, S.E. Study on influence of different types of meat on the quality of meat products. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Food Sci. Technol. 2015, 72, 215–218. [Google Scholar] [CrossRef]
- Dulf, F.; Andrei, S.; Bunea, A.; Socaciu, C. Fatty acid and phytosterol contents of some Romanian wild and cultivated berry pomaces. Chem. Pap. 2012, 66, 925–934. [Google Scholar] [CrossRef]
- Christie, W.W. Preparation of Methyl Ester and Other Derivatives. In Gas Chromatography and Lipids. A Practical Guide; Oily Press: Glasgow, UK, 1989; Volume I, pp. 36–47. [Google Scholar]
- Dulf, F.V.; Oroian, I.; Vodnar, D.C.; Socaciu, C.; Pintea, A. Lipid classes and fatty acid regiodistribution in triacylglycerols of seed oils of two Sambucus species (S. nigra L. and S. ebulus L.). Molecules 2013, 18, 11768–11782. [Google Scholar] [CrossRef]
- Socaci, S.A.; Socaciu, C.; Muresan, C.; Farcas, A.; Tofana, M.; Vicas, S.; Pintea, A. Chemometric discrimination of different tomato cultivars based on their volatile fingerprint in relation to lycopene and total phenolics content. Phytochem. Anal. PCA 2014, 25, 161–169. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Samples | Moisture (g/100 g dw) | Total Fat (g/100 g dw) | Crude Protein (g/100 g dw) | Ash (g/100 g dw) | Carbohydrates (g/100 g dw) | Energy (kcal/100 g dw) |
|---|---|---|---|---|---|---|
| P. ostreatus | 7.26 ± 0.08 | 1.26 ± 0.08 | 17.92 ± 0.18 | 11.11 ± 0.22 | 62.45 ± 0.58 | 332.80 ± 0.82 |
| A. bisporus | 5.35 ± 0.07 | 2.41 ± 0.01 | 34.68 ± 0.17 | 7.24 ± 0.05 | 50.33 ± 0.31 | 361.70 ± 0.44 |
| C. cibarius | 2.17 ± 0.02 | 2.17 ± 0.04 | 21.03 ± 0.04 | 9.57 ± 0.08 | 65.06 ± 0.11 | 363.9 ± 0.66 |
| B. edulis | 7.23 ± 0.18 | 1.92 ± 0.09 | 36.24 ± 0.12 | 8.38 ± 0.07 | 46.23 ± 0.22 | 347.5 ± 0.52 |
| L. piperatus | 14.20 ± 0.27 | 2.69 ± 0.05 | 31.81 ± 0.15 | 8.30 ± 0.21 | 43.01 ± 0.70 | 323.50 ± 1.67 |
| No | Fatty Acid | P. ostreatus | A. bisporus | C. cibarius | B. edulis | L. piperatus |
|---|---|---|---|---|---|---|
| 1 | (6:0) | 0.02 ± 0.001 | n.d. | 0.02 ± 0.001 | 0.15 ± 0.002 | 0.25 ± 0.02 |
| 2 | (8:0) | 0.02 ± 0.001 | 0.01 ± 0.001 | 0.01 ± 0.001 | 0.12 ± 0.01 | 0.07 ± 0.005 |
| 3 | (10:0) | 0.06 ± 0.002 | 0.02 ± 0.001 | 0.06 ± 0.006 | 0.16 ± 0.003 | 0.17 ± 0.002 |
| 4 | (12:0) | 0.03 ± 0.001 | 0.01 ± 0.01 | 0.03 ± 0.002 | 0.07 ± 0.002 | 0.05 ± 0.001 |
| 5 | (14:0) | 0.47 ± 0.001 | 0.55 ± 0.005 | 0.13 ± 0.001 | 0.22 ± 0.001 | 0.15 ± 0.001 |
| 6 | (15:0) | 1.87 ± 0.01 | 1.17 ± 0.02 | 0.49 ± 0.01 | 0.26 ± 0.001 | 0.33 ± 0.02 |
| 7 | (16:0) | 12.40 ± 0.07 | 12.73 ± 0.07 | 5.35 ± 0.5 | 9.93 ± 0.01 | 15.11 ± 0.04 |
| 8 | (16:1)n-9 | 0.16 ± 0.01 | 0.04 ± 0.002 | 0.13 ± 0.002 | 0.14 ± 0.001 | 0.21 ± 0.04 |
| 9 | (16:1)n-7 | 0.47 ± 0.002 | 0.28 ± 0.001 | 0.24 ± 0.004 | 0.96 ± 0.02 | 0.07 ± 0.001 |
| 10 | (17:0) | 0.19 ± 0.02 | 0.71 ± 0.01 | 0.06 ± 0.002 | 0.04 ± 0.001 | 0.11 ± 0.001 |
| 11 | (18:0) | 2.18 ± 0.01 | 5.00 ± 0.5 | 43.23 ± 0.04 | 3.32 ± 0.5 | 9.01 ± 0.4 |
| 12 | (18:1)n-9 | 15.96 ± 0.04 | 1.80 ± 0.02 | 36.29 ± 0.01 | 22.06 ± 0.01 | 9.84 ± 0.6 |
| 13 | (18:1)n-7 | 0.12 ± 0.005 | 0.24 ± 0.001 | 0.12 ± 0.001 | 0.96 ± 0.002 | 18.58 ± 0.02 |
| 14 | (18:2)n-6 | 63.68 ± 0.001 | 71.41 ± 0.04 | 11.72 ± 0.04 | 59.88 ± 0.04 | 44.51 ± 0.06 |
| 15 | (18:3)n-3 | 0.12 ± 0.001 | 0.10 ± 0.002 | 0.03 ± 0.001 | 0.05 ± 0.002 | 0.12 ± 0.001 |
| 16 | (20:0) | 0.20 ± 0.003 | 2.55 ± 0.01 | 1.14 ± 0.03 | 0.23 ± 0.02 | 0.26 ± 0.004 |
| 17 | (20:1)n-9 | 0.16 ± 0.01 | 0.12 ± 0.001 | n.d. | 0.21 ± 0.001 | 0.74 ± 0.001 |
| 18 | (20:2)n-6 | 0.24 ± 0.006 | 0.25 ± 0.001 | 0.03 ± 0.002 | 0.22 ± 0.001 | n.d. |
| 19 | (21:0) | 0.04 ± 0.03 | 0.38 ± 0.004 | n.d. | n.d. | n.d. |
| 20 | (22:0) | 0.64 ± 0.02 | 1.60 ± 0.02 | 0.39 ± 0.004 | 0.18 ± 0.002 | n.d. |
| 21 | (22:1)n-9 | 0.18 ± 0.01 | 0.04 ± 0.006 | n.d. | 0.12 ± 0.002 | n.d. |
| 22 | (23:0) | 0.06 ± 0.004 | 0.20 ± 0.001 | n.d. | 0.07 ± 0.004 | 0.15 ± 0.001 |
| 23 | (24:0) | 0.21 ± 0.03 | 0.77 ± 0.01 | 0.52 ± 0.005 | 0.52 ± 0.001 | 0.25 ± 0.001 |
| 24 | (24:1) | 0.53 ± 0.02 | 0.02 ± 0.001 | n.d. | 0.14 ± 0.01 | n.d. |
| Total SFA | 18.38 ± 0.04 | 25.69 ± 0.01 | 51.43 ± 0.03 | 15.26 ± 0.07 | 25.93 ± 0.06 | |
| Total MUFA | 17.58 ± 0.01 | 2.55 ± 0.05 | 36.78 ± 0.01 | 24.59 ± 0.01 | 29.44 ± 0.01 | |
| Total PUFA | 64.04 ± 0.03 | 71.75 ± 0.03 | 11.79 ± 0.01 | 60.14 ± 0.04 | 44.63 ± 0.06 | |
| PUFAs/SFAs | 3.49 ± 0.002 | 2.79 ± 0.03 | 0.23 ± 0.001 | 3.94 ± 0.02 | 1.72 ± 0.002 |
| No. | Volatile Compound | Odour Perception | P. ostreatus | A. bisporus | C. cibarius | B. edulis | L. piperatus |
|---|---|---|---|---|---|---|---|
| Alcohols | |||||||
| 1. | 2-Methyl-1-butanol | Malty, alcoholic, fruity, whiskey, burnt | 1.39 ± 0.33 | n.d. | n.d. | n.d. | 1.26 ± 0.07 |
| 2. | 1-Pentanol | Sweet, balsamic, fruity | n.d. | n.d. | 1.3 ± 0.12 | n.d. | n.d. |
| 3. | 1-Hexanol | resin, flower, green | n.d. | n.d. | 0.86 ± 0.30 | n.d. | n.d. |
| 4. | 1-Octen-3-ol | mushroom | n.d. | n.d. | 1.36 ± 0.06 | 4.87 ± 0.07 | n.d. |
| 5. | 1-Dodecanol | waxy, fatty, honey | n.d. | 2.09 ± 0.46 | 0.66 ± 0.14 | 7.09 ± 0.23 | n.d. |
| Total | 1.39 ± 0.33 | 2.09 ± 0.46 | 4.18 ± 0.62 | 11.96 ± 0.30 | 1.26 ± 0.07 | ||
| Aldehydes | |||||||
| 1. | 2-Methylpentanal | ethereal fruity green | 19.26 ± 0.35 | 7.56 ± 0.16 | 4.45 ± 0.10 | 4.45 ± 0.23 | 8.83 ± 0.48 |
| 2. | Hexanal | Green, grass, fat | 8.45 ± 0.25 | 19.12 ± 0.31 | 55.48 ± 0.29 | 11.13 ± 0.03 | 5.04 ± 0.68 |
| 3. | Heptanal | Fat, citrus, rancid | n.d. | 2.06 ± 0.16 | 1.82 ± 0.02 | 4.01 ± 0.17 | n.d. |
| 4. | Benzaldehyde | Almond, burnt sugar | 43.36 ± 1.28 | 36.72 ± 0.31 | 6.12 ± 0.81 | 13.12 ± 0.29 | 6.12 ± 0.81 |
| 5. | Octanal | Fat, soap, lemon, green | 1.43 ± 0.21 | 2.98 ± 0.22 | 0.99 ± 0.07 | 4.13 ± 0.31 | 1.88 ± 0.23 |
| 6. | Benzeneacetaldehyde | Harsh, green, honey, cocoa | n.d. | 1.28 ± 0.10 | n.d. | n.d. | n.d. |
| 7. | (E)-2-Octenal | Green, nut, fat | n.d. | n.d. | 0.66 ± 0.07 | n.d. | n.d. |
| 8. | Nonanal | Fat, citrus, green | 2.34 ± 0.09 | 2.33 ± 0.33 | 0.71 ± 0.11 | 3.34 ± 0.02 | 2.33 ± 0.39 |
| 9. | Decanal | Soap, orange peel, tallow | n.d. | n.d. | n.d. | 1.09 ± 0.07 | n.d. |
| Total | 74.84 ± 2.18 | 72.05 ± 1.59 | 70.23 ± 1.47 | 41.27±1.12 | 24.20 ± 2.59 | ||
| Ketones | n.d. | n.d. | |||||
| 1. | 2-Hexanone | fruity, fungal, meaty, buttery | n.d. | n.d. | 0.6 ± 0.06 | n.d. | |
| 2. | 2,3-Dimethylcyclopentanone | n.d. | n.d. | n.d. | n.d. | 3.85 ± 0.18 | |
| 3. | 2-Heptanone | Sulfur, pungent, green, fruity | 12.61 ± 0.59 | n.d. | 3.83 ± 0.09 | n.d. | 2.19 ± 0.03 |
| 4. | 1-Octen-3-one | herbal, mushroom, earthy, musty | n.d. | n.d. | 0.56 ± 0.06 | n.d. | n.d. |
| 5. | 3-Octanone | Musty, mushroom, herbal | n.d. | n.d. | 0.13 ± 0.02 | 6.46 ± 0.51 | n.d. |
| 6. | Acetophenone | Sweet, flower, almond | n.d. | 1.72 ± 0.08 | 0.42 ± 0.09 | 2.79 ± 0.42 | 0.65 ± 0.04 |
| 7. | Benzophenone | balsam rose metallic powdery geranium | n.d. | 3.09 ± 0.12 | 0.92 ± 0.18 | 7.81 ± 0.76 | n.d. |
| Total | 12.61 ± 0.59 | 4.81 ± 0.20 | 6.46 ± 0.50 | 17.06 ± 1.69 | 6.69 ± 0.25 | ||
| Terpenoids | |||||||
| 1. | α-Pinene | pine, turpentine | n.d. | n.d. | 0.40 ± 0.08 | n.d. | n.d. |
| 2. | β-Phellandrene | mint, terpentine | n.d. | 1.42 ± 0.03 | 0.53 ± 0.05 | n.d. | n.d. |
| 3. | β-Pinene | pine, resin, turpentine | n.d. | 1.71 ± 0.29 | 1.16 ± 0.03 | n.d. | 1.52 ± 0.27 |
| 4. | Β-Myrcene | balsamic, must, spice | n.d. | n.d. | n.d. | 4.56 ± 0.55 | n.d. |
| 5. | D-Limonene | citrus, mint, fruity | n.d. | 4.55 ± 0.61 | 1.05 ± 0.38 | n.d. | 3.07 ± 0.12 |
| Total | n.d. | 7.68 ± 0.93 | 3.14 ± 0.54 | 4.56 ± 0.55 | 4.59 ± 0.39 | ||
| Sulphur compounds | |||||||
| 1. | Methyl disulfide | onion, cabbage, sulfurous, vegetable | n.d. | n.d. | 0.58 ± 0.10 | 11.56 ± 0.48 | 1.31 ± 0.54 |
| 2. | Methyl 2-propenyl disulfide | alliaceous, garlic, green, onion | n.d. | n.d. | 2.15 ± 0.38 | n.d. | 22.19 ± 2.05 |
| 3. | Dimethyl trisulfide | sulfurous, cooked onion, savory, meaty | n.d. | n.d. | n.d. | 5.59 ± 0.29 | n.d. |
| 4. | Diallyl disulphide | alliaceous, onion, garlic, metallic | n.d. | n.d. | 2.7 ± 0.25 | n.d. | 27.89 ± 1.21 |
| Total | n.d. | n.d. | 5.43 ± 0.73 | 17.15 ± 0.77 | 51.39 ± 3.80 | ||
| Acids | |||||||
| 1. | Nonanoic acid | green, fat | n.d. | n.d. | n.d. | n.d. | n.d. |
| 2. | n-Decanoic acid | fat | n.d. | n.d. | 0.54 ± 0.18 | 2.96 ± 0.45 | n.d. |
| 3. | Dodecanoic acid | fatty, waxy | 5.14 ± 0.34 | 6.53 ± 0.27 | 1.99 ± 0.06 | 5.07 ± 0.44 | 6.86 ± 1.17 |
| Total | 5.14 ± 0.34 | 6.53 ± 0.27 | 2.53 ± 0.24 | 8.03 ± 0.89 | 6.86 ± 1.17 | ||
| Others | |||||||
| 1 | 2-n-Pentylfuran | fruity, green, earthy, beany | 6.05 ± 0.37 | 6.88 ± 0.36 | 11.29 ± 0.4 | n.d. | 5.05 ± 0.57 |
| Total | 6.05 ± 0.37 | 6.88 ± 0.36 | 11.29 ± 0.4 | n.d. | 5.05 ± 0.57 |
| Peak No. | Compound | Phenolic Compound in Analyzed Mushrooms Extracts (mg/100 g fw) | ||||
|---|---|---|---|---|---|---|
| P. ostreatus | A. bisporus | C. cibarius | B. edulis | L. piperatus | ||
| 1 | 4-Hydroxybenzoic acid | 75.04 ± 0.20 | 79.50 ± 0.02 | 16.16 ± 0.12 | 209.87 ± 0.35 | 42.93 ± 0.22 |
| 2 | 2,4-Dihydroxybenzoic acid | 11.84 ± 0.20 | 19.62 ± 0.06 | 4.96 ± 0.02 | 69.13 ± 0.15 | n.d. |
| 3 | 4-Hydroxyphenylacetic acid | 4.02 ± 0.05 | 5.06 ± 0.005 | 1.60 ± 0.01 | 25.30 ± 0.28 | 7.38 ± 0.03 |
| 4 | Protocatechuic acid | 17.28 ± 0.6 | 46.11 ± 0.05 | 5.17 ± 0.02 | 43.58 ± 0.25 | 5.48 ± 0.01 |
| 5 | Catechin | 14.86 ± 0.10 | 31.29 ± 0.02 | 2.43±0.06 | 145.57 ± 0.40 | 6.47 ± 0.05 |
| 6 | Gallocatechin | 5.04 ± 0.15 | 5.27 ± 0.01 | 1.03±0.02 | 26.63 ± 0.25 | 10.95 ± 0.2 |
| 7 | p-Coumaric acid | n.d. | n.d. | 1.47±0.01 | 23.11 ± 0.20 | 5.19 ± 0.06 |
| 8 | Ferulic acid | n.d. | n.d. | n.d. | n.d. | 9.15 ± 0.03 |
| 9 | Sinapic acid | n.d. | n.d. | n.d. | 27.38 ± 0.08 | 8.66 ± 0.01 |
| 10 | o-Coumaric acid | 3.63 ± 0.20 | n.d. | n.d. | 11.42 ± 0.06 | 6.37 ± 0.24 |
| 11 | Cinnamic acid | 10.09 ± 0.15 | 14.36 ± 0.02 | 2.38 ± 0.01 | 168.61 ± 0.45 | 14.54 ± 0.15 |
| 12 | 3-Feruloylquinic acid | n.d. | n.d. | 9.49 ± 0.08 | n.d. | 66.73 ± 0.40 |
| 13 | 4-Feruloylquinic acid | n.d. | 60.46 ± 0.06 | 6.31 ± 0.06 | n.d. | 87.62 ± 0.35 |
| 14 | 5-Feruloylquinic acid | 35.04 ± 0.08 | 71.01 ± 0.04 | 55.33 ± 0.25 | n.d. | 38.19 ± 0.10 |
| 15 | 3,5 Dicaffeoylquinic acid | 14.60 ± 0.10 | 14.00 ± 0.11 | 54.21 ± 0.13 | 31.55 ± 0.45 | 61.14 ± 0.30 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fogarasi, M.; Socaci, S.A.; Dulf, F.V.; Diaconeasa, Z.M.; Fărcaș, A.C.; Tofană, M.; Semeniuc, C.A. Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms. Molecules 2018, 23, 3272. https://doi.org/10.3390/molecules23123272
Fogarasi M, Socaci SA, Dulf FV, Diaconeasa ZM, Fărcaș AC, Tofană M, Semeniuc CA. Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms. Molecules. 2018; 23(12):3272. https://doi.org/10.3390/molecules23123272
Chicago/Turabian StyleFogarasi, Melinda, Sonia Ancuţa Socaci, Francisc Vasile Dulf, Zorița Maria Diaconeasa, Anca Corina Fărcaș, Maria Tofană, and Cristina Anamaria Semeniuc. 2018. "Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms" Molecules 23, no. 12: 3272. https://doi.org/10.3390/molecules23123272
APA StyleFogarasi, M., Socaci, S. A., Dulf, F. V., Diaconeasa, Z. M., Fărcaș, A. C., Tofană, M., & Semeniuc, C. A. (2018). Bioactive Compounds and Volatile Profiles of Five Transylvanian Wild Edible Mushrooms. Molecules, 23(12), 3272. https://doi.org/10.3390/molecules23123272