Investigation of the In Vivo Metabolism of Sibirioside A and Angoroside C in Rats by HPLC-ESI-IT-TOF-MSn
Abstract
1. Introduction
2. Results
2.1. Fragmentation Behaviors of Sibirioside A and Angoroside C
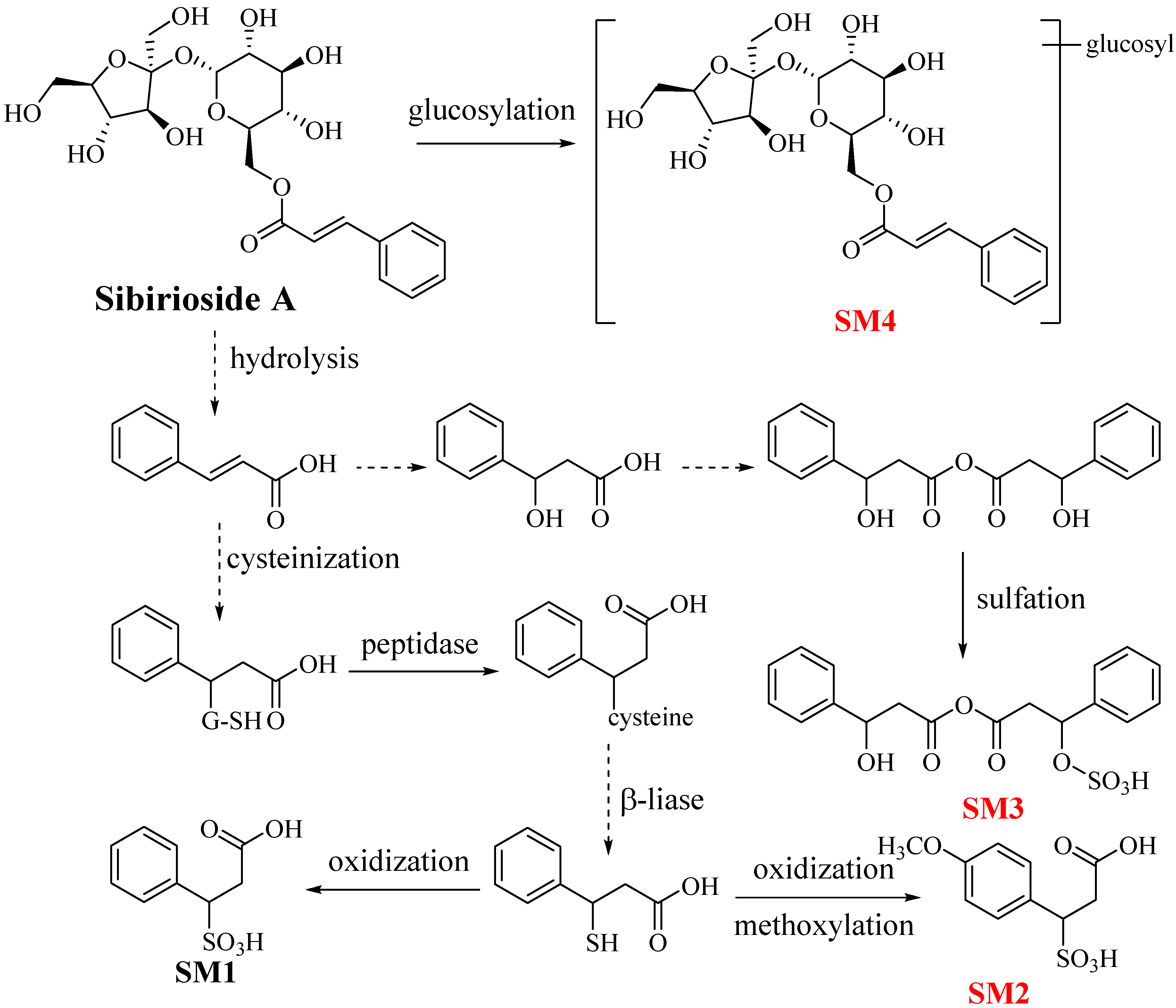
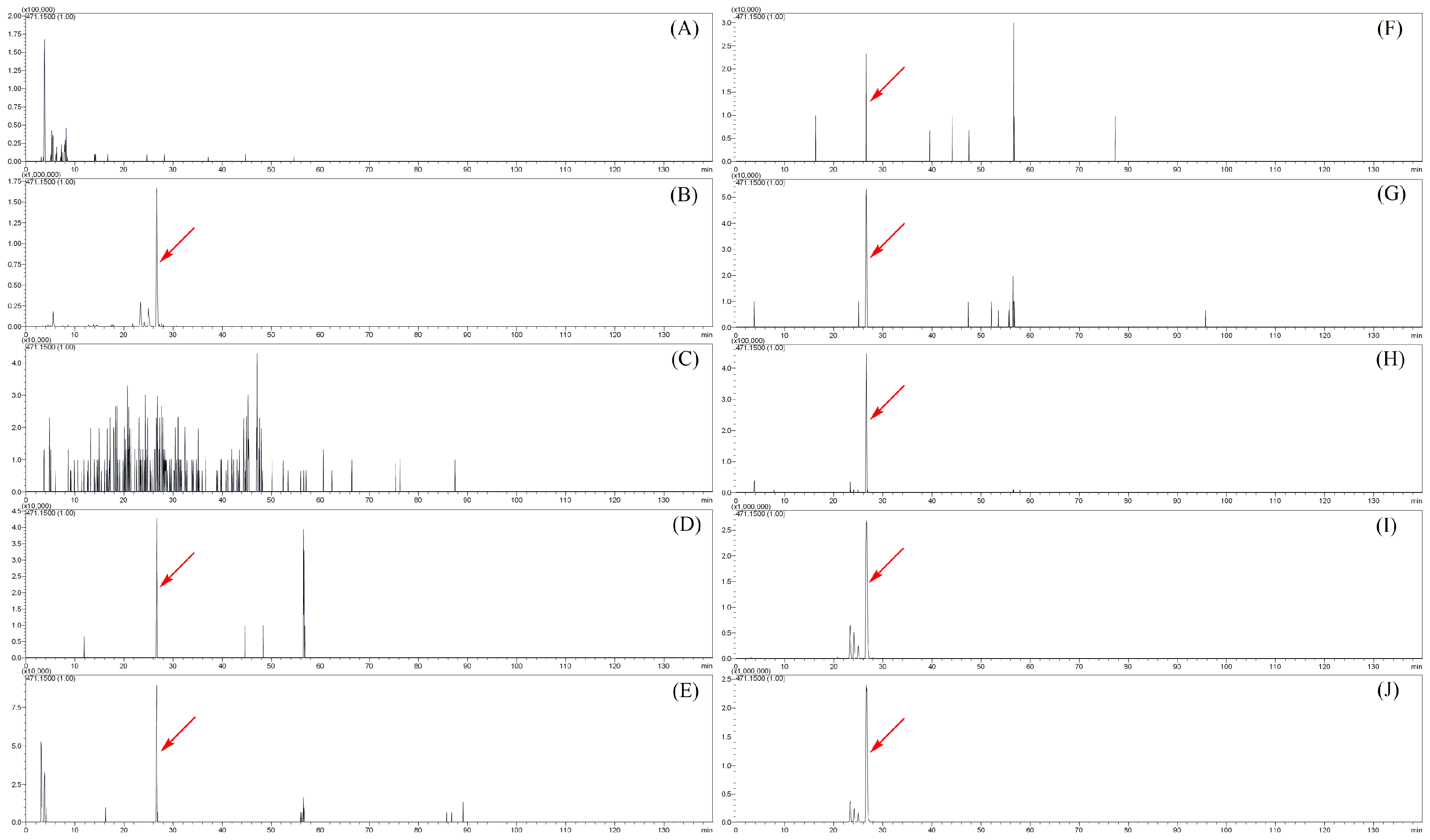
2.2. Metabolites of Sibirioside A: SM1–SM4
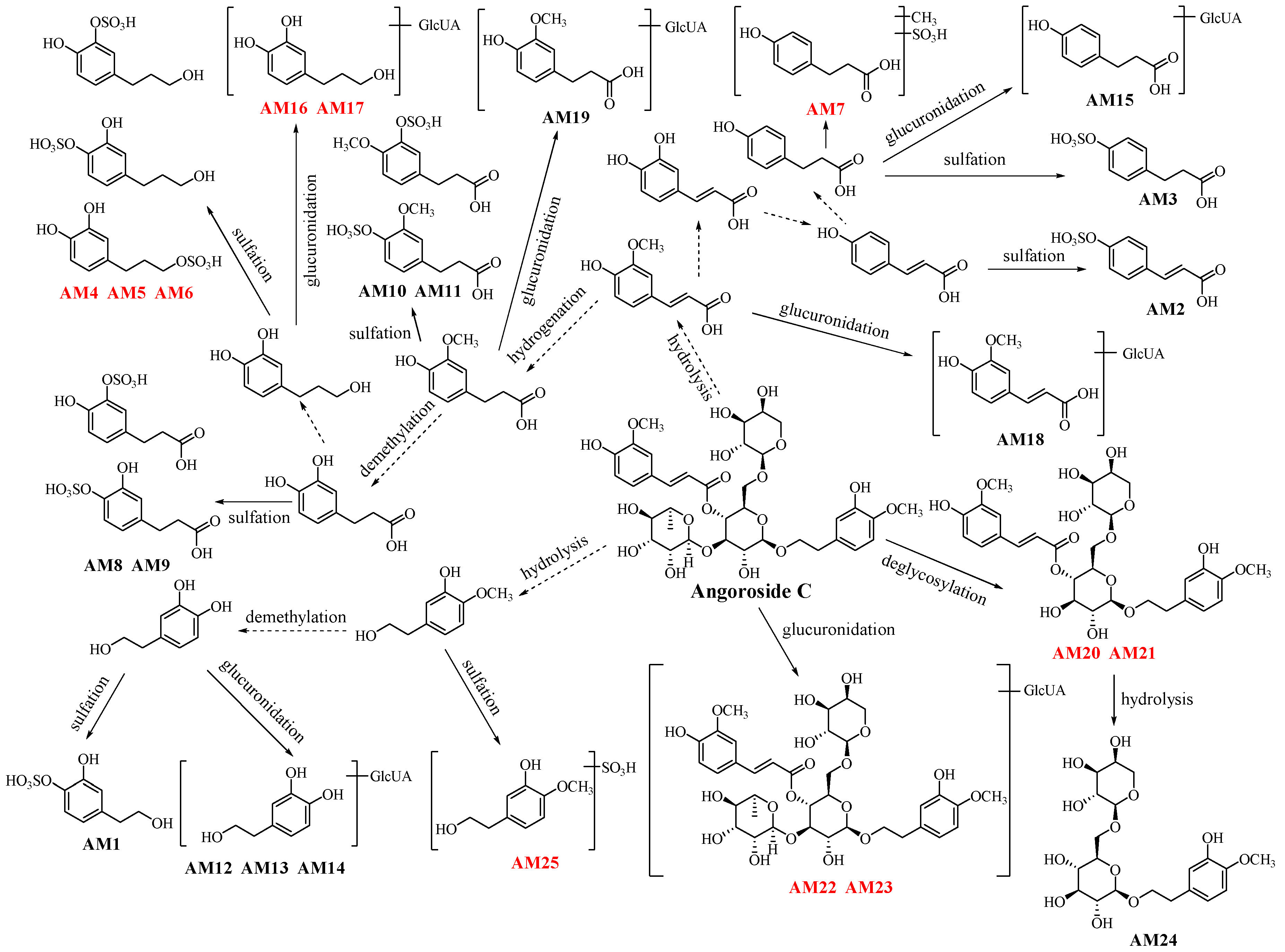
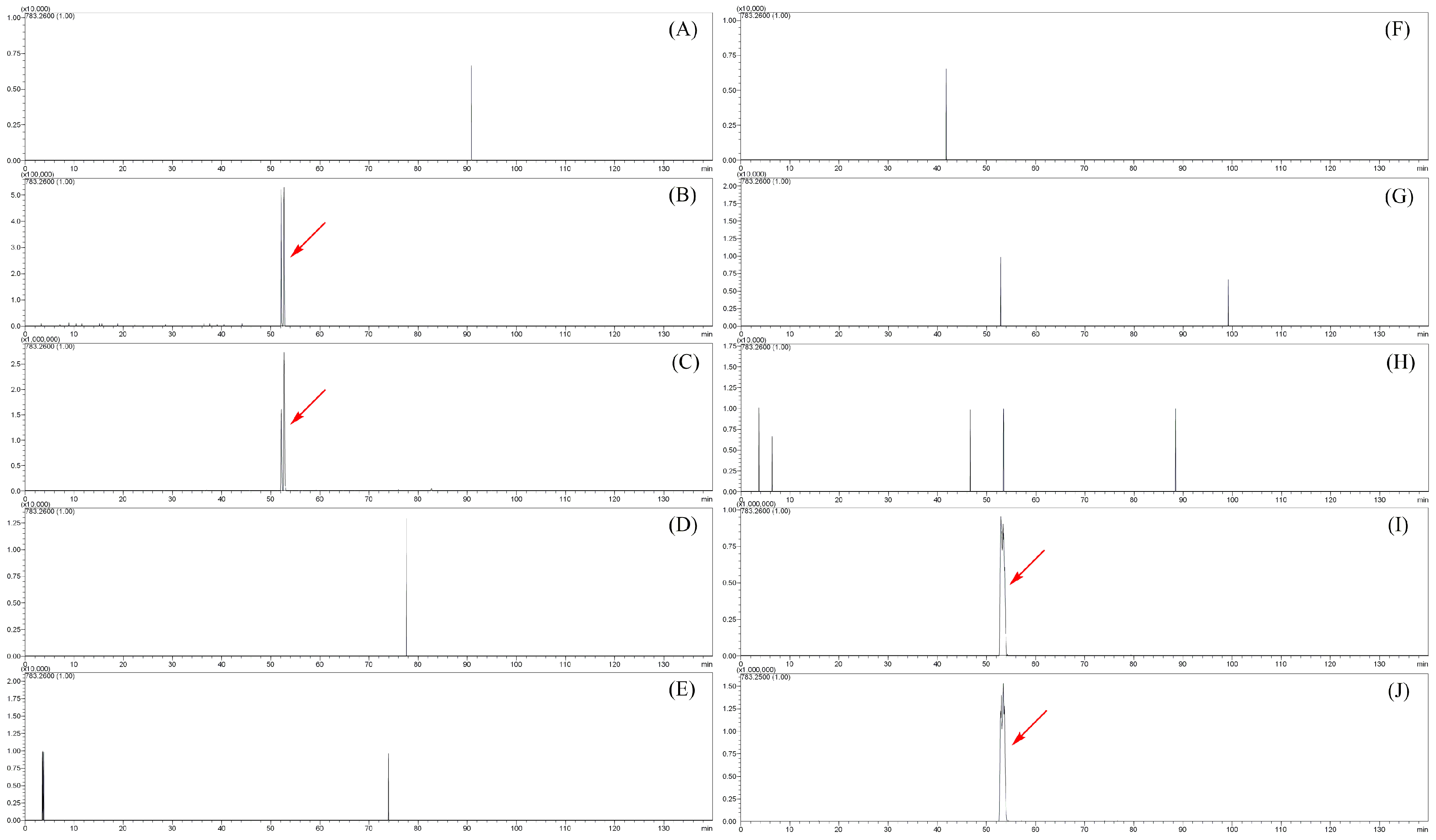
2.3. Metabolites of Angoroside C: AM1–AM25
2.4. The Distribution of Sibirioside A and Angoroside C in Rats
2.5. Biological Activity Prediction of the Metabolites of Sibirioside A and Angoroside C
3. Discussion
4. Materials and Methods
4.1. Chemicals and Materials
4.2. Animal Experiments
4.3. Preparation of Rat Biological Samples
4.4. Instrumental and Analytical Conditions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Xu, F.Q.; Xu, X.D.; Chen, S.L. Progress in chemical constituents and bioactivities of Scrophularia ningpoensis. Mod. Chin. Med. 2013, 9, 752–759. [Google Scholar]
- Li, Y.M.; Zeng, H.W.; He, X.; Jiang, Y.Y.; Jiang, S.H.; Zhu, D.Y. Iridoid and phenylpropanoid glycosides of Scrophularia ningpoensis inhibit the formation of LTB4 and platelet aggregation. Acad. J. Second Mil. Med. Univ. 1999, 20, 301–303. [Google Scholar]
- Díaz, A.M.; Abad, M.J.; Femández, L.; Silván, A.M.; Santos, J.D.; Bemejo, P. Phenylpropanoid glycosides from Scrophularia scorodonia: In vitro anti-inflammatory activity. Life Sci. 2004, 74, 2515–2526. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zheng, Y.F.; Li, C.Y.; Zheng, Y.Y.; Wang, D.Q.; Wu, Z.; Huang, L.; Wang, Y.G.; Li, P.B.; Peng, W.; et al. Discovery of Anti-inflammatory Ingredients in Chinese Herbal Formula Kouyanqing Granule based on Relevance Analysis between Chemical Characters and Biological Effects. Sci. Rep. 2015, 5, 18080. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.M.; Han, Z.H.; Jiang, S.H.; Yao, S.D.; Zhu, D.Y. Fast repairing of oxidized OH radical adducts of dAMP and dGMP by phenylpropanoid glycosides from Scrophularia ningpoensis. Acta Pharmacol. Sin. 2000, 21, 1125–1128. [Google Scholar] [PubMed]
- Gu, W.L.; Chen, C.X.; Huang, X.Y.; Gao, J.P. The effect of angoroside C on pressure overload-induced ventricular remodeling in rats. Phytomedicine 2015, 22, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.M.; Jiang, S.H.; Gao, W.Y.; Zhu, D.Y. Phenylpropanoid glycosides from Scrophularia ningpoensis. Phytochemistry 2000, 54, 923–925. [Google Scholar] [CrossRef]
- Jing, J.; Chan, C.O.; Xu, L.; Jin, D.; Cao, X.; Mok, D.K.; Parekh, H.S.; Chen, S. Development of an in-line HPLC fingerprint ion-trap mass spectrometric method for identification and quality control of Radix Scrophulariae. J. Pharm. Biomed. Anal. 2011, 56, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fanny, C.F.; Liang, Y.; Nancy, Y.; Zhong, B.L.; Lai, C.W.; Xu, S.H. A new iridiod glycoside and a new cinnamoyl glycoside from Scrophularia ningpoensis Hemsl. Nat. Prod. Res. 2017, 31, 2361–2368. [Google Scholar] [CrossRef] [PubMed]
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the Peoples Republic of China; Chemical Industry Press: Beijing, China, 2015. [Google Scholar]
- Zhang, L.; Zhang, L.; Zhang, Y.D. Comparative study on the pharmacognosy and anti-inflammatory activities of Scrophularia buergeriana Miq. Var. tsinglingensis Tsoong and S. ninpoensis Hemsl. Northwest Pharm. J. 2014, 29, 264–267. [Google Scholar]
- Bermejo, B.P.; Diaz, L.A.; Silvan, S.A.; De Santos, G.Z.; Fernandez, M.L.; Sanz, G.A.; Abad, M.J. Effects of some iridoids from plant origin on arachidonic acid metabolism in cellular systems. Planta Med. 2000, 66, 324–328. [Google Scholar]
- Liu, G.L.; Fu, P.Y.; Wang, Z.Y.; Xing, D.Y. Effects of water extract of four Chinese herbal drugs on the binding of insulin with human erythroeyte insulin receptor. Chin. J. Integr. Trad. West. Med. 1991, 10, 606–607. [Google Scholar]
- Ni, Z.; Cai, X.Z.; Huang, Y.P.; Wang, D.J.; Bian, H.M. Effect of extracts of Scrophularia ningpoensis Hemsl. on hemorrheology, coagulation and fibrinolysis in rats. J. Chin. Microcirc. 2004, 3, 152–153. [Google Scholar]
- Sohn, S.; Ko, E.; Jeon, S.; Lee, B.J.; Kim, S.H.; Dong, M.S.; Lee, D.U.; Kwak, J.H.; Kim, Y.S.; Shin, M.K.; et al. The genome-wide expression profile of Scrophularia ningpoensis-treated thapsigargin-stimulated U-87MG cells. Neurotoxicology 2009, 30, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Z.; Liu, Z.; Ma, L.M.; Li, J.; Shang, M.Y.; Liu, G.X.; Xu, F.; Cai, S.Q. Changes of chemical constituents in Scrophulariae Radix during processing based on UPLC-Q-TOF MS. J. Chin. Mass Spectrom. Soc. 2016, 37, 1–9. [Google Scholar]
- Wang, J.Z.; Zhang, Y.F.; Xu, F.; Shang, M.Y.; Liu, G.X.; Cai, S.Q. Investigation of the in vivo metabolism of harpagoside and distribution of its metabolites in rats by HPLC-IT-TOF-MSn. Biomed. Chromatogr. 2018, e4218. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Xu, F.; Wang, J.Z.; Liu, G.X.; Shang, M.Y.; Cai, S.Q. Identification and analysis of harpagide metabolites in rats in vivo. Chin. Pharm. 2017, 28, 1310–1315. [Google Scholar]
- Ounnas, F.; Privé, F.; Salen, P.; Gaci, N.; Tottey, W.; Calani, L.; Bresciani, L.; Lopez-Gutierrez, N.; Hazane-Puch, F.; Laporte, F.; et al. Whole rye consumption lmproves blood and liver n-3 fatty acid profile and gut microbiota composition in rats. PLoS ONE 2016, 11, e0148118. [Google Scholar] [CrossRef] [PubMed]
- Tuck, K.L.; Hayball, P.J.; Stupans, I. Structural characterization of the metabolites of hydroxytyrosol, the principal phenolic component in olive oil, in rats. J. Agric. Food Chem. 2002, 50, 2404–2409. [Google Scholar] [CrossRef] [PubMed]
- Zaklo, D.; Costagliola, R.; Dorio, C.; Rathahao, E.; Cravedi, J.P. In vivo metabolic fate of the xeno-estrogen 4-N-nonylphenol in Wistar rats. Drug Metab. Dispos. 2003, 31, 168–178. [Google Scholar]
- Wusteman, F.S.; Dodgson, K.S.; Lloyd, A.G.; Rose, F.A.; Tudball, N. Thin-layer chromatography in the study of ester sulfates. J. Chromatogr. A 1964, 16, 334–339. [Google Scholar] [CrossRef]
- Stalmach, A.; Mullen, W.D. Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: Identification of biomarkers of coffee consumption. Drug Metab. Dispos. 2009, 37, 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Fumeaux, R.; Menozzi, C.; Stalmach, A.; Munari, C.; Kraehenbuehl, K. First synthesis, characterization, and evidence for the presence of hydroxycinnamic acid sulfate and glucuronide conjugates in human biological fluids as a result of coffee consumption. Org. Biomol. Chem. 2010, 8, 5199–5211. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, S.; Sakurai, S.; Sugimoto, I.; Awata, N. Absorption and metabolism of γ-oryzanol in rats. Chem. Pharm. Bull. 2008, 31, 645–652. [Google Scholar] [CrossRef]
- De Karina, I.T.; Jauregui, O.; Castellote, A.I.; Lamuelaraventós, R.M.; Covas, M.I. Rapid high-performance liquid chromatography-electrospray ionization tandem mass spectrometry method for qualitative and quantitative analysis of virgin olive oil phenolic metabolites in human low-density lipoproteins. J. Chromatogr. A 2006, 1116, 69–75. [Google Scholar]
- Khymenets, O.; Joglar, J.; Clapés, P.; Parella, T.; Covas, M. Biocatalyzed synthesis and structural characterization of monoglucuronides of hydroxytyrosol, tyrosol, homovanillic alcohol, and 3-(4′-hydroxyphenyl)propanol. Adv. Synth. Catal. 2010, 348, 2155–2162. [Google Scholar] [CrossRef]
- Thibaut, R.; Debrauwer, L.; Rao, D.; Cravedi, J.P. Characterization of biliary metabolites of 4-n-nonylphenol in rainbow trout (Oncorhynchus mykiss). Xenobiotica 1998, 28, 745–757. [Google Scholar] [CrossRef] [PubMed]
- Kajimoto, T.; Hidaka, M.; Shoyama, K.; Nohara, T. Iridoids from Scrophularia ningpoensis. Phytochemistry 1989, 28, 2701–2704. [Google Scholar] [CrossRef]
- Liu, X.F.; Ouyang, S.S.; Yu, B.; Liu, B.; Liu, Y.B.; Huang, K.; Gong, J.Y.; Zheng, S.Y.; Li, Z.H.; Li, H.L.; et al. PharmMapper server: A web server for potential drug target identification using pharmacophore mapping approach. Nucleic Acids Res. 2010, 38, W609–W614. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Pan, C.X.; Gong, J.Y.; Liu, X.F.; Li, H.L. Enhancing the enrichment of pharmacophore-based target prediction for the polypharmacological profiles of drugs. J. Chem. Inf. Model. 2016, 56, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Shen, Y.H.; Wang, S.W.; Li, S.L.; Zhang, W.L.; Liu, X.F.; Lai, L.H.; Pei, J.F.; Li, H.L. PharmMapper 2017 update: A web server for potential drug target identification with a comprehensive target pharmacophore database. Nucleic Acids Res. 2017, 45, W356–W360. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
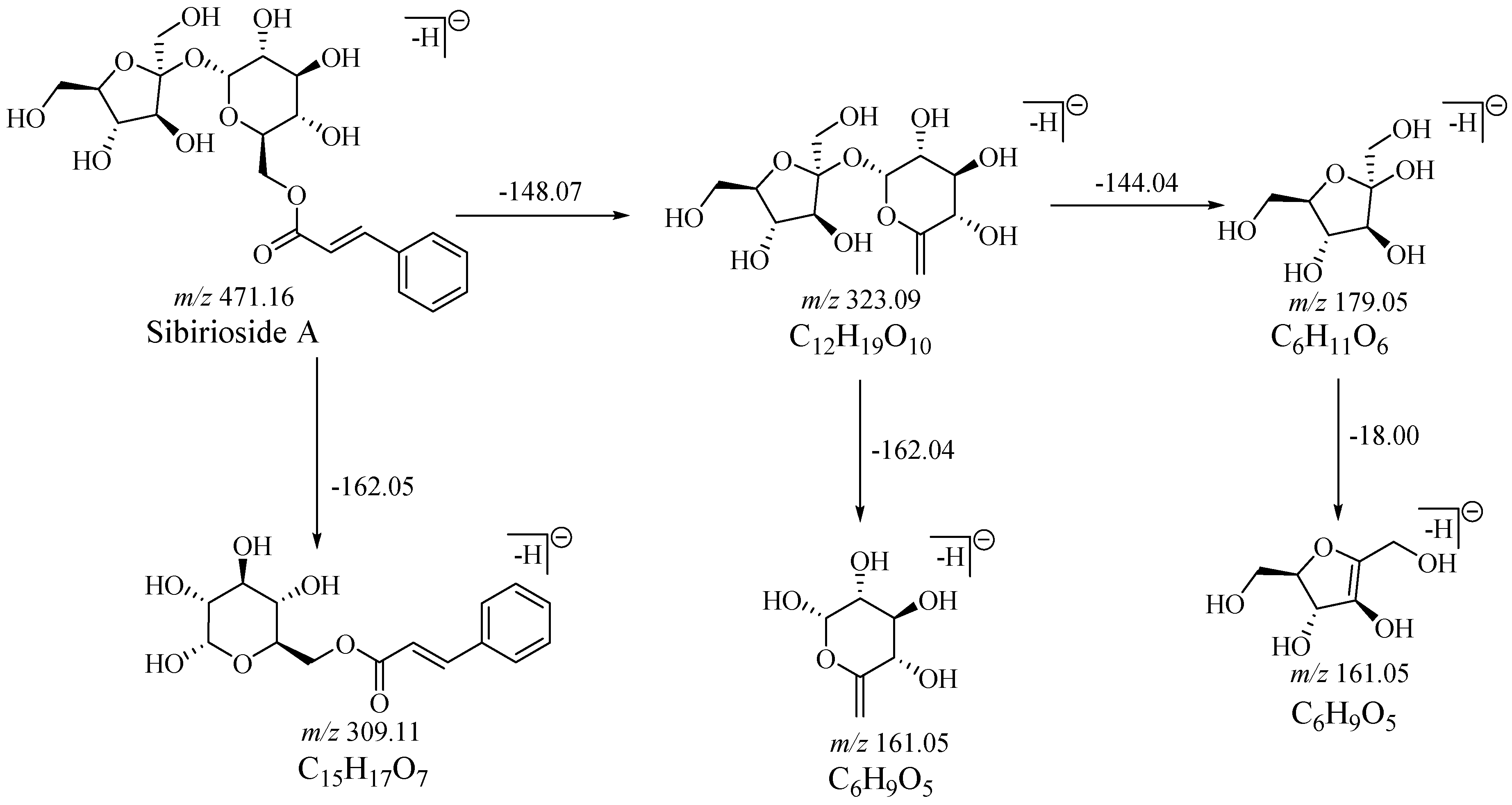
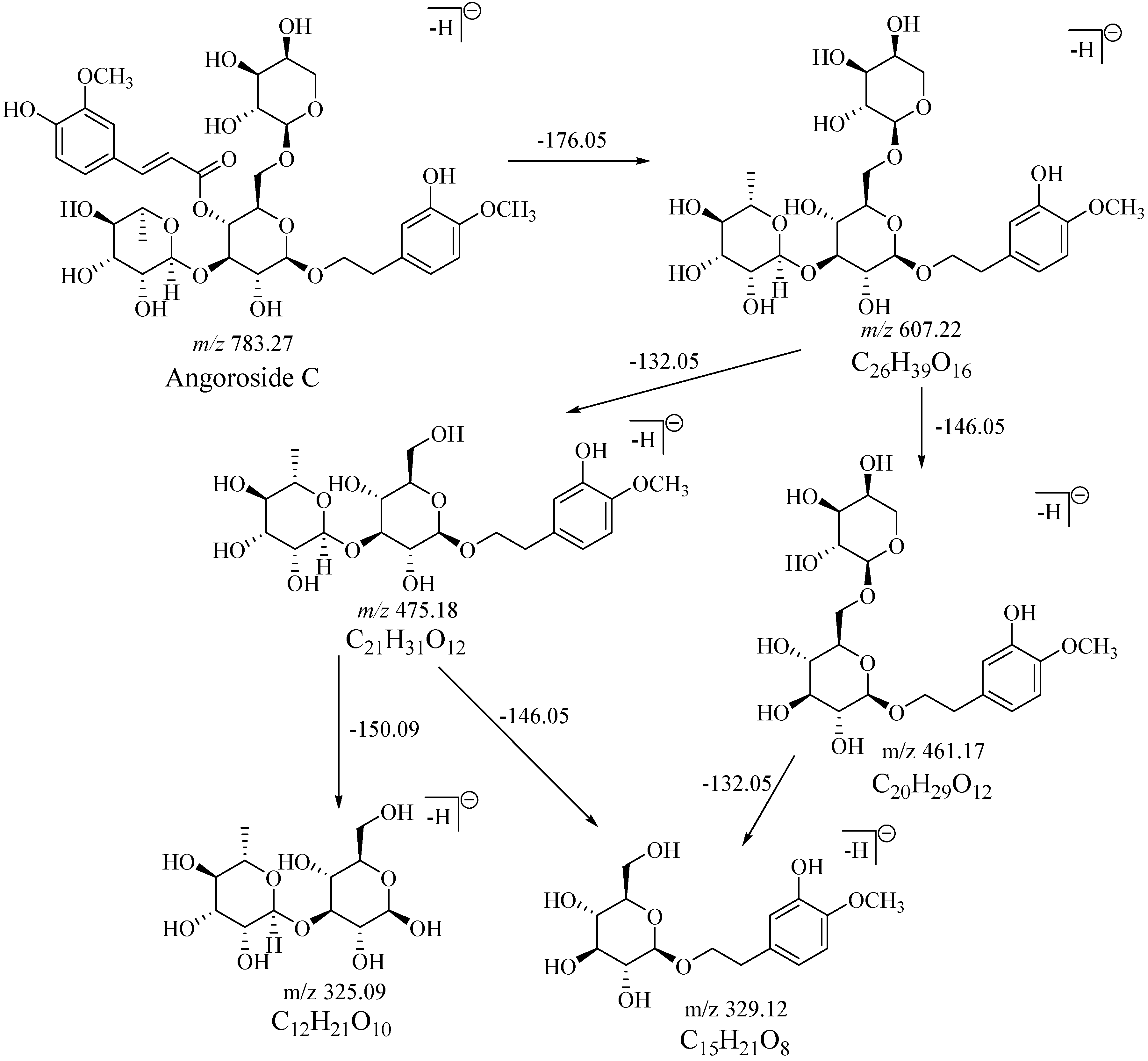
| No. | tR/min | Formula | m/z | Diff | DBE | Fragment | Identification |
|---|---|---|---|---|---|---|---|
| SM1 | 7.813 | C9H10O5S | 229.0155 | −9.17 | 5 | 147.0459, 103.0348 | phenylpropanoic acid sulphate [19] |
| SM2 | 10.328 | C10H12O6S | 259.0293 | 4.25 | 5 | 177.0540, 133.0983 | 4-methoxyphenylpropanoic acid sulphate * |
| SM3 | 7.277 | C18H18O8S | 393.0755 | 10.50 | 10 | 311.1019, 163.0583, 147.0454 | β-hydroxybenzenepropanoic anhydride sulphate * |
| SM4 | 24.513 | C27H38O17 | 633.2014 | −3.47 | 9 | 485.1416, 323.0932 | sibirioside A glucose conjugation * |
| AM1 | 15.602 | C8H10O6S | 233.0119 | −2.57 | 4 | 153.0583, 123.0492 | 4-(2-hydroxyethyl)-1,2-benzenediol sulphate [20] |
| AM2 | 32.993 | C9H8O6S | 242.9965 | −7.82 | 6 | 163.0415, 119.0532 | p-coumaric acid sulphate [21] |
| AM3 | 29.057 | C9H10O6S | 245.0109 | −6.53 | 5 | 165.0574, 121.0680 | 4-hydroxybenzenepropanoic acid sulphate [22] |
| AM4 | 17.995 | C9H12O6S | 247.0265 | −6.88 | 4 | 167.0745, 137.0642 | 3,4-hydroxyphenylpropanol sulphate 1 * |
| AM5 | 20.260 | C9H12O6S | 247.0271 | −4.45 | 4 | 167.0745, 137.0642 | 3,4-hydroxyphenylpropanol sulphate 2 * |
| AM6 | 22.548 | C9H12O6S | 248.0282 | 0 | 4 | 167.0745, 137.0642 | 3,4-hydroxyphenylpropanol sulphate 3 * |
| AM7 | 42.685 | C10H12O6S | 259.0271 | −4.25 | 5 | 179.0726, 163.0385, 147.0473, 119.0507 | β-methoxybenzenepropanoic acid sulphate * |
| AM8 | 18.630 | C9H10O7S | 261.0063 | −4.21 | 5 | 217.0175, 181.0522, 137.0633 | 3,4-dihydroxybenzenepropanoic acid-3-O-sulphate [23] |
| AM9 | 26.887 | C9H10O7S | 261.0051 | −8.81 | 5 | 217.0175, 181.0522, 137.0633 | 3,4-dihydroxybenzenepropanoic acid-3-O-sulphate [24] |
| AM10 | 30.600 | C10H12O7S | 275.0210 | −7.64 | 5 | 195.0665, 177.0545, 151.0799 | 3-methoxyl-4-hydroxybenzenepropanoic acid sulphate [25] |
| AM11 | 38.278 | C10H12O7S | 275.0214 | −6.18 | 5 | 195.0665, 177.0545, 151.0799 | 3-hydroxyl-4- methoxyl benzenepropanoic acid sulphate [24] |
| AM12 | 15.712 | C14H18O9 | 329.0929 | 5.1 | 6 | 175.0282, 153.0568 | 4-(2-hydroxyethyl)-1,2-benzenediol glucuronide [26] |
| AM13 | 16.507 | C14H18O9 | 329.0871 | −2.13 | 6 | 175.0282, 153.0568 | 4-(2-hydroxyethyl)-1,2-benzenediol glucuronide [27] |
| AM14 | 17.792 | C14H18O9 | 329.0857 | −6.38 | 6 | 175.0282, 153.0568 | 4-(2-hydroxyethyl)-1,2-benzenediol glucuronide [20] |
| AM15 | 33.103 | C15H18O9 | 341.0848 | −8.80 | 7 | 323.0709, 175.0244, 165.052, 121.0628 | 4-hydroxybenzenepropanoic acid glucuronide [28] |
| AM16 | 24.020 | C15H20O9 | 343.1017 | −5.25 | 6 | 325.097, 175.0265, 167.0713, 147.0301 | 3,4-hydroxyphenylpropanol glucuronide * |
| AM17 | 29.170 | C15H20O9 | 343.1017 | −5.25 | 6 | 325.097, 175.0265, 167.0713, 147.0301 | 3,4-hydroxyphenylpropanol glucuronide * |
| AM18 | 35.262 | C16H18O10 | 369.0794 | −8.94 | 8 | 193.0508, 175.0169 | 3-methoxyl-4-hydroxycinnamic acid glucuronide [25] |
| AM19 | 32.448 | C16H20O10 | 371.0988 | 1.08 | 7 | 195.0658, 175.0265 | 3-methoxyl-4-hydroxybenzenepropanoic acid glucuronide [25] |
| AM20 | 52.445 | C30H38O15 | 637.2131 | −1.10 | 12 | 461.1636, 443.1533, 311.1151, 275.0889, 193.0548, 167.1269, 137.0759 | deglycosylated angoroside C 1 * |
| AM21 | 53.108 | C30H38O15 | 637.2177 | 6.12 | 12 | 461.1636, 443.1533, 311.1151, 275.0889, 193.0548, 167.1269, 137.0759 | deglycosylated angoroside C 2 * |
| AM22 | 44.933 | C42H56O25 | 959.3007 | −3.23 | 15 | 783.2594, 607.2227, 461.1681, 443.1608 | angoroside C glucuronide 1 * |
| AM23 | 47.378 | C42H56O25 | 959.2941 | −0.31 | 15 | 783.2594, 607.2227, 461.1681, 443.1608 | angoroside C glucuronide 2 * |
| AM24 | 33.343 | C20H30O12 | 461.1701 | 7.81 | 6 | 461.1701, 443.1711, 329.1180, 191.0574 | 2-(3-hydroxy-4-methoxyphenyl)ethyl 6-O-arabinopyranosyl glucopyranoside [29] |
| AM25 | 27.377 | C9H12O6S | 247.0266 | −6.48 | 4 | 167.0750, 137.0627, 121.0264 | 4-(2-hydroxyethyl)-1,2-benzenediol sulphate * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.-F.; Liu, L.-J.; Xu, F.; Shang, M.-Y.; Liu, G.-X.; Cai, S.-Q. Investigation of the In Vivo Metabolism of Sibirioside A and Angoroside C in Rats by HPLC-ESI-IT-TOF-MSn. Molecules 2018, 23, 2702. https://doi.org/10.3390/molecules23102702
Zhang Y-F, Liu L-J, Xu F, Shang M-Y, Liu G-X, Cai S-Q. Investigation of the In Vivo Metabolism of Sibirioside A and Angoroside C in Rats by HPLC-ESI-IT-TOF-MSn. Molecules. 2018; 23(10):2702. https://doi.org/10.3390/molecules23102702
Chicago/Turabian StyleZhang, Yi-Fan, Li-Jia Liu, Feng Xu, Ming-Ying Shang, Guang-Xue Liu, and Shao-Qing Cai. 2018. "Investigation of the In Vivo Metabolism of Sibirioside A and Angoroside C in Rats by HPLC-ESI-IT-TOF-MSn" Molecules 23, no. 10: 2702. https://doi.org/10.3390/molecules23102702
APA StyleZhang, Y.-F., Liu, L.-J., Xu, F., Shang, M.-Y., Liu, G.-X., & Cai, S.-Q. (2018). Investigation of the In Vivo Metabolism of Sibirioside A and Angoroside C in Rats by HPLC-ESI-IT-TOF-MSn. Molecules, 23(10), 2702. https://doi.org/10.3390/molecules23102702





