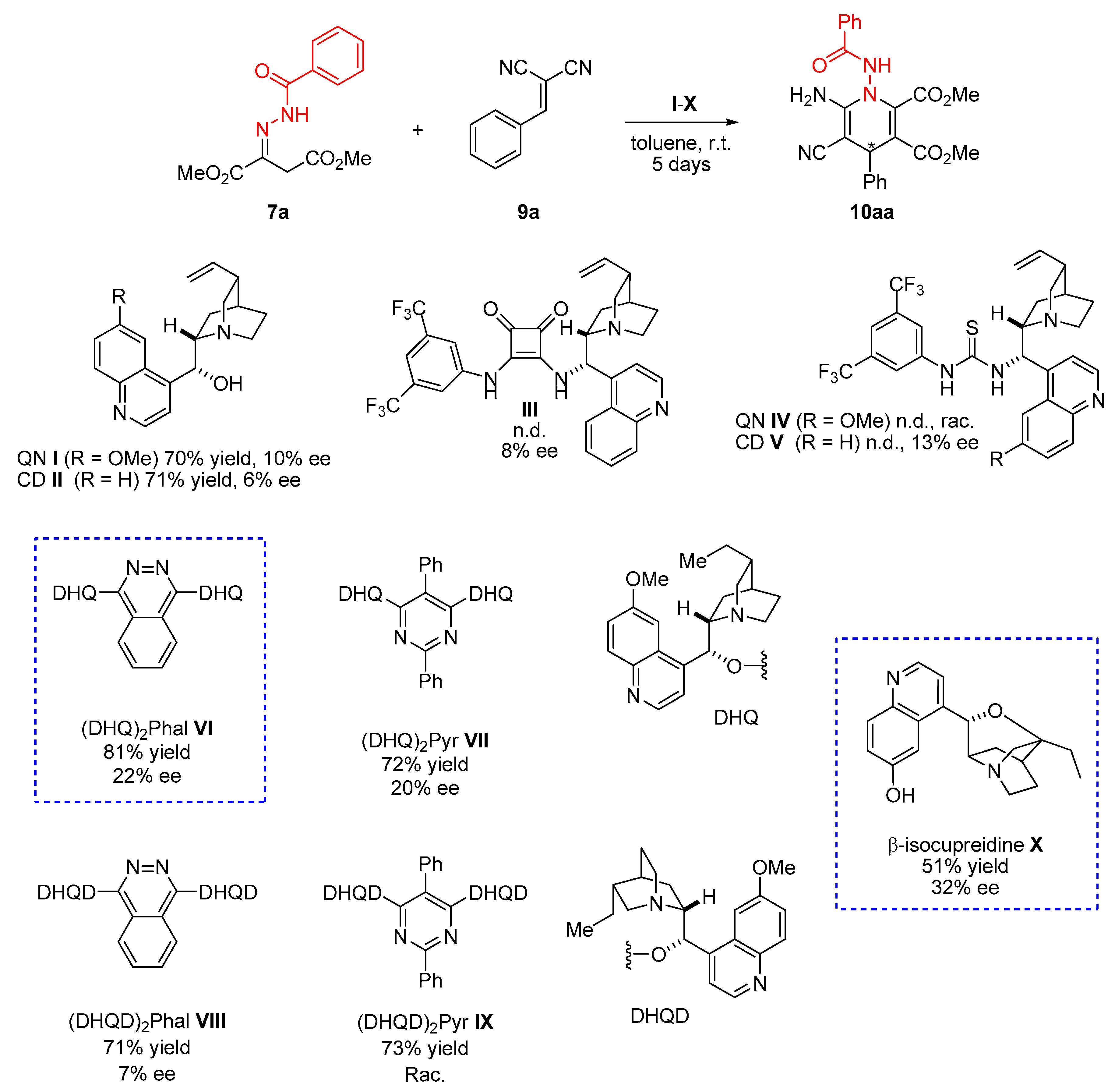
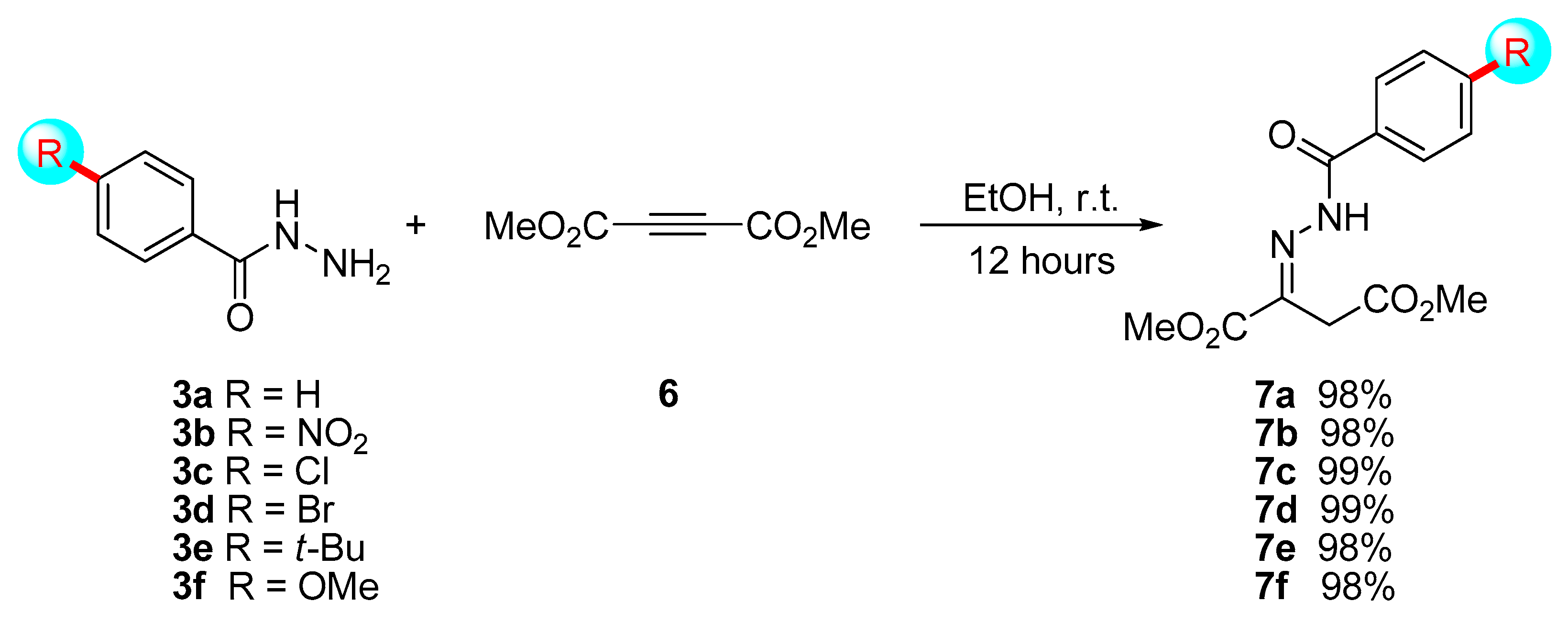3.2. General Procedure for the Synthesis of Hydrazones 7
To a solution of the corresponding hydrazide
3 (5 mmol) in ethanol (15 mL), dimethyl acetylenedicarboxylate
6 (5 mmol, 615 μL) was slowly added due to the exothermic characteristics of the reaction. The reaction mixture was stirred for 12 h at room temperature. Then, the solvent was evaporated under vacuum, and the reaction crude was purified by filtration, washing the white precipitate with small portions of ethanol (3 × 3 mL) giving rise to the corresponding final adduct
7 (
Scheme 3).
(E)-Dimethyl 2-(2-(benzoyl)hydrazono)succinate (
7a) [
26]: Following the general procedure, compound
7a was obtained as a white solid in a 98% yield.
(E)-Dimethyl 2-(2-(4-nitrobenzoyl)hydrazono)succinate (7b): Following the general procedure, compound 7b was obtained as a white solid in a 98% yield. mp 146–148 °C. 1H-NMR (300 MHz, DMSO-d6) δ 3.65 (s, 3H), 3.76 (s, 3H), 3.90 (s, 2H), 8.06 (d, J = 8.1 Hz, 2H), 8.35 (d, J = 8.5 Hz, 2H), 11.70 (s, 1H). 13C-APT-NMR (75 MHz, DMSO-d6) δ 32.6 (1C), 52.2 (1C), 52.6 (1C), 123.3 (2C), 130.3 (2C), 138.8 (1C), 149.3 (1C), 164.4 (1C), 168.2 (3C). IR (neat) (cm–1) ν 3451, 3244, 3113, 1729, 1690, 1671, 1520, 1349, 1241, 1124, 1005, 852, 719. HRMS (ESI+) calcd for C13H13N3NaO7 346.0646; found 346.0649 [M + Na].
(E)-Dimethyl 2-(2-(4-chlorobenzoyl)hydrazono)succinate (7c): Following the general procedure, compound 7c was obtained as a white solid in a 99% yield. mp 138–140 °C. 1H-NMR (300 MHz, DMSO-d6) δ 3.64 (s, 3H), 3.76 (s, 3H), 3.89 (s, 2H), 7.60 (d, J = 8.6 Hz, 2H), 7.87 (d, J = 8.6 Hz, 2H), 11.47 (s, 1H). 13C-APT-NMR (75 MHz, DMSO-d6) δ 32.5 (1C), 52.2 (1C), 52.5 (1C), 128.4 (2C), 130.7 (2C), 131.7 (1C), 137.0 (1C), 164.5 (1C), 168.3 (3C). IR (neat) (cm–1) ν 3441, 3238, 2954, 1741, 1726, 1694, 1670, 1594, 1536, 1440, 1250, 1146, 1123, 1111, 1089, 1003, 889, 846, 756. HRMS (ESI+) calcd for C13H13ClN2NaO5 335.0405; found 335.0385 [M + Na].
(E)-Dimethyl 2-(2-(4-bromobenzoyl)hydrazono)succinate (7d): Following the general procedure, compound 7d was obtained as a white solid in a 99% yield. mp 114–116 °C. 1H-NMR (300 MHz, DMSO-d6) δ 3.64 (s, 3H), 3.76 (s, 3H), 3.89 (s, 2H), 7.77 (q, J = 8.4 Hz, 4H), 11.48 (s, 1H). 13C-APT-NMR (75 MHz, DMSO-d6) δ 30.9 (3C), 32.5 (1C), 52.2 (1C), 52.6 (1C), 125.9 (1C), 130.9 (2C), 131.3 (2C), 132.1 (2C), 164.5 (1C), 168.3 (2C). IR (neat) (cm–1) ν 3256, 2953, 1734, 1716, 1685, 1591, 1434, 1222, 1201, 1127, 1111, 1009, 889, 838, 752. HRMS (ESI+) calcd for C13H13BrN2NaO5 378.9900; found 378.9903 [M + Na].
(E)-Dimethyl 2-(2-(4-tert-butylbenzoyl)hydrazono)succinate (7e): Following the general procedure, compound 7e was obtained as a white solid in a 98% yield. mp 165–167 °C. 1H-NMR (300 MHz, DMSO-d6) δ 1.31(s, 9H), 3.64 (s, 3H), 3.77 (s, 3H), 3.89 (s, 2H), 7.54 (d, J = 8.6 Hz, 2H), 7.78 (d, J = 8.6 Hz, 2H), 11.33 (s, 1H). 13C-APT-NMR (75 MHz, DMSO-d6) δ 30.9 (3C), 32.4 (1C), 34.8 (1C), 52.1 (1C), 52.5 (1C), 125.1 (2C), 128.6 (2C), 130.3 (1C), 155.2(1C), 164.6 (1C), 168.4 (3C). IR (neat) (cm–1) ν 3232, 3201, 2964, 2949, 1741, 1721, 1671, 1609, 1536, 1432, 1246, 1205, 1150, 1126, 1118, 1021, 895, 857, 841, 707. HRMS (ESI+) calcd for C17H22N2NaO5 357.1421; found 357.1419 [M + Na].
(E)-Dimethyl 2-(2-(4-methoxybenzoyl)hydrazono)succinate (7f): Following the general procedure, compound 7f was obtained as a white solid in a 98% yield. mp 144–146 °C. 1H-NMR (300 MHz, DMSO-d6) δ 3.64 (s, 3H), 3.76 (s, 3H), 3.84 (s, 3H), 3.89 (s, 2H), 7.06 (d, J = 8.9 Hz, 2H), 7.87 (d, J = 8.9 Hz, 2H), 11.25 (s, 1H). 13C-APT-NMR (75 MHz, DMSO-d6) δ 32.4 (1C), 52.2 (1C), 52.5 (1C), 55.5 (1C), 113.6 (2C), 124.9 (1C), 130.9 (2C), 162.4 (1C), 164.6 (1C), 168.5 (3C). IR (neat) (cm–1) ν 3419, 3223, 3189, 1737, 1715, 1659, 1601, 1541, 1508, 1436, 1319, 1256, 1205, 1170, 1143, 1109, 1027, 996, 889, 849, 762. HRMS (ESI+) calcd for C14H16N2NaO6 331.0901; found 331.0888 [M + Na].
3.3. General Procedure for the Synthesis of 1,4-Dihydropyridines 10
To a mixture of β-isocupreidine catalyst
X (20 mol%, 6.21 mg) and the corresponding benzylidenemalononitrile
9 (0.15 mmol) in tetrahydrofuran (0.5 mL), hydrazones
7 (0.1 mmol) were added. The reaction mixture was stirred for 5 days at room temperature. Then, the reaction crude was purified by column chromatography (SiO
2,
n-hexane:diethyl ether 20:80 to 0:100), giving rise to the corresponding final chiral adducts
10 (
Scheme 6).
Dimethyl 6-amino-1-benzamido-5-cyano-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10aa): Following the general procedure, compound 10aa was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 61% yield (26.37 mg). mp 106–108 °C. The ee of the product was determined to be 50% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 237.7 nm): τmajor = 30.4 min; τminor = 11.1 min. [α]D24 = −25.4 (c = 0.07, MeOH, 50% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.51 (s, 3H), 3.57 (s, 0.75H), 3.64 (s, 2.25H), 4.36 (s, 0.75H), 4.48 (s, 0.25H), 6.38 (s, 1.5H), 6.46 (s, 0.5H), 7.24 (t, J = 7.3 Hz, 1.5H), 7.35 (t, J = 7.4 Hz, 2H), 7.45–7.59 (m, 3.5H), 7.63 (t, J = 7.1 Hz, 1H), 7.79–7.91 (m, 2H), 11.21 (s, 0.75H), 11.32 (s, 0.25H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.2 (1C), 51.8 (1C), 52.8 (1C), 58.6 (1C), 104.6 (1C), 120.9 (1C), 126.8 (2C), 127.7 (1C), 127.9 (2C), 128.3 (2C), 128.5 (2C), 131.3 (1C), 132.5 (1C), 142.6 (1C), 145.7 (1C), 151.1 (1C), 162.4 (1C), 164.7 (1C), 166.7 (1C). IR (neat) (cm–1) ν 3420, 3334, 3249, 3219, 2957, 2190, 1736, 1707, 1662, 1590, 1479, 1428, 1225, 1110, 717, 699, 689. HRMS (ESI+) calcd for C23H20N4NaO5 455.1326; found 455.1340 [M + Na].
Dimethyl 6-amino-5-cyano-1-(4-nitrobenzamido)-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10ba): Following the general procedure, compound 10ba was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a yellow solid in 56% yield (26.82 mg). mp 238–240 °C. The ee of the product was determined to be 45% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 249.6 nm): τmajor = 47.0 min; τminor = 21.4 min. [α]D24 = −6.6 (c = 0.13, MeOH, 45% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.51 (s, 3H), 3.58 (s, 0.78H), 3.65 (s, 2.22H), 4.37 (s, 0.74H), 4.44 (s, 0.26H), 6.55 (s, 1.48H), 6.60 (s, 0.52H), 7.24 (t, J = 7.3 Hz, 1.48H), 7.35 (t, J = 7.5 Hz, 2H), 7.52 (d, J = 7.2 Hz, 1.52H), 8.04 (d, J = 8.8 Hz, 0.52H), 8.10 (d, J = 8.8 Hz, 1.48H), 8.38 (d, J = 8.8 Hz, 2H), 11.62 (s, 0.74H), 11.68 (s, 0.26H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.2 (1C), 51.8 (1C), 52.9 (1C), 58.1 (1C), 104.9 (1C), 120.8 (1C), 123.5 (min), 123.6 (2C), 126.9 (1C), 127.6 (2C), 128.3 (2C), 128.6 (min), 129.4 (min), 129.5 (2C), 137.0 (1C), 142.2 (1C), 145.6 (1C), 149.7 (1C), 151.0 (1C), 162.4 (1C), 164.6 (1C), 165.4 (1C). IR (neat) (cm–1) ν 3420, 3331, 3208, 2955, 2923, 2852, 2193, 1722, 1710, 1679, 1660, 1589, 1527, 1430, 1346, 1231, 1117, 1081, 697. HRMS (ESI+) calcd for C23H19N5NaO7 500.1177; found 500.1175 [M + Na].
Dimethyl 6-amino-1-(4-chlorobenzamido)-5-cyano-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10ca): Following the general procedure, compound 10ca was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 75% yield (34.88 mg). mp 143–145 °C. The ee of the product was determined to be 43% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 254.0 nm): τmajor = 24.2 min; τminor = 11.6 min. [α]D24 = −26.7 (c = 0.10, MeOH, 43% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.49 (s, 0.9H), 3.51 (s, 2.1H), 3.56 (s, 0.9H), 3.63 (s, 2.1H), 4.35 (s, 0.7H), 4.47 (s, 0.3H), 6.46 (s, 1.4H), 6.50 (s, 0.6H), 7.24 (t, J = 7 Hz, 1.4H), 7.30-7.38 (m, 2.2H), 7.53 (d, J = 7,1 Hz, 1.4H), 7.63 (d, J = 8.6 Hz, 2H), 7.83 (d, J = 8.6 Hz, 0.6H), 7.88 (d, J = 8.6 Hz, 1.4H), 11.33 (s, 0.7H), 11.42 (s, 0.3H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.1 (1C), 51.9 (1C), 52.8 (min), 52.9 (1C), 57.2 (min), 58.3 (1C), 104.7 (1C), 120.9 (1C), 121.0 (min), 126.9 (min), 127.0 (1C), 127.7 (2C), 128.4 (2C), 128.5 (min), 128.6 (2C), 129.9 (2C), 130.2 (1C), 130.3 (min), 137.2 (1C), 137.4 (1C), 142.5 (1C), 143.0 (1C), 145.7 (1C), 151.1 (1C), 151.6 (1C), 162.4 (1C), 164.7 (1C), 164.8 (1C), 165.3 (1C), 165.8 (1C). IR (neat) (cm–1) ν 3413, 3331, 3251, 3025, 2956, 2192, 1727, 1715, 1664, 1590, 1432, 1335, 1230, 1093, 1014, 698. HRMS (ESI+) calcd for C23H19ClN4NaO5 489.0912; found 489.0913 [M + Na].
Dimethyl 6-amino-1-(4-bromobenzamido)-5-cyano-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10da): Following the general procedure, compound 10da was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a yellow solid in 60% yield (30.81 mg). mp 134–136 °C. The ee of the product was determined to be 44% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 330.0 nm): τmajor = 24.6 min; τminor = 10.7 min. [α]D24 = −20.3 (c = 0.15, MeOH, 44% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.49 (s, 0.75H), 3.51 (s, 2.25H), 3.56 (s, 0.75H), 3.63 (s, 2.25H), 4.35 (s, 0.75H), 4.47 (s, 0.25H), 6.45 (s, 1.5 H), 6.50 (s, 0.50H), 7.24 (t, J = 7.3 Hz, 1.5H), 7.34 (t, J = 7.5 Hz, 2H), 7.53 (d, J = 7.1 Hz, 1.5H), 7.73–7.84 (m, 4H), 11.33 (s, 0.75H), 11.42 (s, 0.25H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.2 (1C), 51.9 (1C), 52.8 (1C), 58.3 (1C), 104.7 (1C), 120.9 (1C), 126.3 (1C), 126.8 (1C), 126.9 (min), 127.6 (2C), 128.3 (2C), 128.6 (min), 130.0 (2C), 130.5 (min), 131.4 (2C), 131.5 (1C), 142.4 (1C), 145.6 (1C), 151.1 (1C), 162.4 (1C), 164.6 (1C), 165.9 (1C). IR (neat) (cm–1) ν 3417, 3331, 3240, 2955, 2190, 1716, 1663, 1588, 1431, 1230, 1079, 1010, 698. HRMS (ESI+) calcd for C23H19BrN4NaO5 533.0418; found 533.0418 [M + Na].
Dimethyl 6-amino-1-(4-(tert-butyl)benzamido)-5-cyano-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10ea): Following the general procedure, compound 10ea was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a yellow solid in 58% yield (28.33 mg). mp 141–143 °C. The ee of the product was determined to be 48% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 242.5 nm): τmajor = 13.2 min; τminor = 9.1 min. [α]D24 = −24.7 (c = 0.15, MeOH, 48% ee). 1H-NMR (400 MHz, DMSO-d6) δ 1.31 (s, 9H), 3.51 (s, 3H), 3.58 (s, 0.78H), 3.66 (s, 2.22H), 4.36 (s, 0.74H), 4.48 (s, 0.26H), 6.33 (s, 1.48H), 6.38 (s, 0.52H), 7.24 (t, J = 7.4 Hz, 1.48H), 7.35 (t, J = 7.4 Hz, 2H), 7.56 (d, J = 8.2 Hz, 3.52H), 7.74–7.84 (m, 2H), 11.13 (s, 0.74H), 11.13 (s, 0.26H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 30.9 (1C), 34.8 (1C), 39.2 (1C), 51.9 (1C), 52.8 (1C), 58.7 (1C), 104.5 (1C), 120.9 (1C), 125.2 (min), 125.3 (2C), 126.8 (1C), 127.0 (min), 127.7 (2C), 127.8 (2C), 128.3 (2C), 128.5 (1C), 128.6 (min), 142.7 (1C), 145.7 (1C), 151.1 (1C), 155.6 (1C), 162.4 (1C), 164.7 (1C), 166.4 (1C). IR (neat) (cm–1) ν 3261, 2958, 2192, 1731, 1638, 1608, 1438, 1270, 1238, 1118, 849, 698. HRMS (ESI+) calcd for C27H28N4NaO5 511.1948; found 511.1947 [M + Na].
Dimethyl 6-amino-5-cyano-1-(4-methoxybenzamido)-4-phenyl-1,4-dihydropyridine-2,3-dicarboxylate (10fa): Following the general procedure, compound 10fa was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a yellow solid in 55% yield (25.24 mg). mp 113–115 °C. The ee of the product was determined to be 42% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 253.2 nm): τmajor = 32.7 min; τminor = 14.8 min. [α]D24 = −25.6 (c = 0.08, MeOH, 42% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.51 (s, 3H), 3.55 (s, 0.75H), 3.62 (s, 2.25H), 3.84 (s, 3H), 4.35 (s, 0.75H), 4.47 (s, 0.25H), 6.33 (s, 1.50H), 6.38 (s, 0.5H), 7.07 (d, J = 8.7 Hz, 2H), 7.24 (t, J = 7.1 Hz, 1.5H), 7.34 (t, J = 7.4 Hz, 2H), 7.55 (d, J = 7.5 Hz, 1.5H), 7.77–7.89 (m, 2H), 11.04 (s, 0.75H), 11.16 (s, 0.25H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.3 (1C), 51.8 (1C), 52.7 (1C), 55.5 (1C), 58.6 (1C), 104.5 (1C), 113.6 (min), 113.8 (2C), 120.9 (1C), 123.4 (1C), 126.8 (1C), 127.0 (min), 127.7 (2C), 128.3 (2C), 128.6 (min), 129.9 (2C), 142.8 (1C), 145.7 (1C), 151.2 (1C), 162.4 (1C), 162.6 (1C), 164.7 (1C), 166.0 (1C).IR (neat) (cm–1) ν 3409, 3334, 3247, 3214, 2953, 2186, 1732, 1713, 1663, 1604, 1587, 1489, 1431, 1249, 1228, 1184, 1115, 1076, 1024, 841, 700. HRMS (ESI+) calcd for C24H22N4NaO6 485.1426; found 485.1425 [M+Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(4-nitrophenyl)-1,4-dihydropyridine-2,3-dicarboxylate (10ab): Following the general procedure, compound 10ab was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 95% yield (45.3 mg). mp 158–160 °C. The ee of the product was determined to be 46% by HPLC using a Daicel Chiralpak IA column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 236.6 nm): τmajor = 26.1 min; τminor = 11.2 min. [α]D25 = +24.5 (c = 0.15, MeOH, 46% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.51 (s, 3H), 3.64 (s, 3H), 4.50 (s, 1H), 6.62 (s, 2H), 7.54 (t, J = 7.4 Hz, 2H), 7.59–7.83 (m, 3H), 7.87 (d, J = 7.3 Hz, 2H), 8.24 (d, J = 8.7 Hz, 2H), 11.31 (s, 1H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.1 (1C), 45.7 (1C), 52.1 (1C), 52.9 (1C), 103.8 (1C), 120.5 (1C), 123.8 (2C), 128.0 (2C), 128.5 (2C), 128.7 (2C), 131.2 (1C), 132.6 (1C), 146.5 (1C), 151.6 (1C), 152.9 (1C), 162.2 (1C), 164.4 (1C), 166.8 (1C). IR (neat) (cm–1) ν 3330, 3200, 2953, 2185, 1743, 1708, 1652, 1579, 1516, 1428, 1344, 1225, 1110, 823, 694. HRMS (ESI+) calcd for C23H19N5NaO7 500.1181; found 500.1181 [M + Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(3-nitrophenyl)-1,4-dihydropyridine-2,3-dicarboxylate (
10ac) [
19]: Following the general procedure, compound
10ac was obtained after 120 h of reaction at room temperature and was purified by column chromatography (
n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 89% yield (42.4 mg). The ee of the product was determined to be 51% by HPLC using a Daicel Chiralpak IC column (
n-hexane/
i-PrOH = 70:30, flow rate 1 mL min
−1, λ = 238.9 nm): τ
major = 18.6 min; τ
minor = 15.4 min. [α]
D24 = −2.8 (
c = 0.24, MeOH, 51% ee).
Dimethyl 6-amino-1-benzamido-4-(3-chlorophenyl)-5-cyano-1,4-dihydropyridine-2,3-dicarboxylate (10ad): Following the general procedure, compound 10ad was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 70% yield (32.6 mg). mp 134–136 °C. The ee of the product was determined to be 52% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 80:20, flow rate 1 mL min−1, λ = 236.6 nm): τmajor = 32.3 min; τminor = 18.6 min. [α]D24 = +23.4 (c = 0.13, MeOH, 52% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.53 (s, 3H), 3.65 (s, 3H), 4.41 (s, 1H), 6.49 (s, 2H), 7.31 (d, J = 7.4 Hz, 1H), 7.39 (t, J = 7.7 Hz, 1H), 7.54 (t, J = 7.6 Hz, 3H), 7.60–7.68 (m, 2H), 7.86 (d, J = 7.3 Hz, 2H), 11.27 (s, 1H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.0 (1C), 52.0 (1C), 52.9 (1C), 58.1 (1C), 104.3 (1C), 120.6 (1C), 126.4 (1C), 127.0 (1C), 127.6 (1C), 127.9 (2C), 128.5 (2C), 130.2 (1C), 131.3 (1C), 132.5 (1C), 133.2 (1C), 142.9 (1C), 148.1 (1C), 151.3 (1C), 162.3 (1C), 164.5 (1C), 166.8 (1C). IR (neat) (cm–1) ν 3330, 2953, 2922, 2850, 2184, 1736, 1707, 1654, 1578, 1430, 1227, 1115, 1079, 886, 781, 692. HRMS (ESI+) calcd for C23H19ClN4NaO5 489.0942; found 489.0941 [M + Na].
Dimethyl 6-amino-1-benzamido-4-(4-chlorophenyl)-5-cyano-1,4-dihydropyridine-2,3-dicarboxylate (
10ae) [
19]: Following the general procedure, compound
10ae was obtained after 120 h of reaction at room temperature and was purified by column chromatography (
n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 70% yield (32.7 mg). The ee of the product was determined to be 50% by HPLC using a Daicel Chiralpak IC column (
n-hexane/
i-PrOH = 80:20, flow rate 1 mL min
−1, λ = 237.7 nm): τ
major = 22.5 min; τ
minor = 16.4 min. [α]
D24 = −2.2 (
c = 0.15, MeOH, 50% ee).
Dimethyl 6-amino-1-benzamido-4-(4-bromophenyl)-5-cyano-1,4-dihydropyridine-2,3-dicarboxylate (
10af) [
19]: Following the general procedure, compound
10af was obtained after 120 h of reaction at room temperature and was purified by column chromatography (
n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 61% yield (31.2 mg). The ee of the product was determined to be 52% by HPLC using a Daicel Chiralpak IC column (
n-hexane/
i-PrOH = 80:20, flow rate 1 mL min
−1, λ = 236.6 nm): τ
major = 23.0 min; τ
minor = 18.0 min. [α]
D24 = −19.5 (
c = 0.05, MeOH, 52% ee).
Dimethyl 6-amino-1-benzamido-5-cyano-4-(4-cyanophenyl)-1,4-dihydropyridine-2,3-dicarboxylate (10ag): Following the general procedure, compound 10ag was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 99% yield (45.09 mg). mp 154–156 °C. The ee of the product was determined to be 50% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 234.2 nm): τmajor = 22.2 min; τminor = 18.5 min. [α]D24 = +4.0 (c = 0.07, MeOH, 50% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.51 (s, 3H), 3.65 (s, 3H), 4.49 (s, 1H), 6.55 (s, 2H), 7.35–7.69 (m,4H), 7.70–7.91 (m, 5H), 11.30 (s, 1H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 39.4 (1C), 52.0 (1C), 52.9 (1C), 57.5 (1C), 103.9 (1C), 109.8 (1C), 118.9 (1C), 120.5 (1C), 127.9 (3C), 128.5 (2C), 128.6 (2C), 131.2 (1C), 132.5 (2C), 143.3 (1C), 151.0 (1C), 151.4 (1C), 162.2 (1C), 164.4 (1C), 166.8 (1C). IR (neat) (cm–1) ν 3414, 3313, 3210, 2947, 2231, 2192, 1750, 1704, 1682, 1665, 1590, 1433, 1360, 1272, 1253, 1222, 1115, 929, 846, 686. HRMS (ESI+) calcd for C24H19N5NaO5 480.1258; found 480.1256 [M + Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(naphthalen-1-yl)-1,4-dihydropyridine-2,3-dicarboxylate (10ah): Following the general procedure, compound 10ah was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a yellow solid in 51% yield (24.61 mg). mp 122–124 °C. The ee of the product was determined to be 50% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 221.2 nm): τmajor = 27.9 min; τminor = 16.5 min. [α]D24 = −28.9 (c = 0.10, MeOH, 50% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.33 (s, 3H), 3.60 (s, 0.75H), 3.67 (s, 2.25H), 5.42 (s, 0.75H), 5.47 (s, 0.25H), 6.33 (s, 1.5H), 6.38 (s, 0.5H), 7.41–7.70 (m, 6.25H), 7.78–7.97 (m, 4H), 8.09 (d, J = 6.7 Hz, 0.75H), 8.46 (d, J = 8.7 Hz, 1H), 11.27 (s, 0.75H), 11.39 (s, 0.25H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 40.0 (1C), 51.7 (1C), 52.8 (1C), 59.2 (1C), 64.9 (1C), 105.4 (1C), 120.8 (1C), 123.5 (1C), 125.5 (1C), 125.7 (1C), 126.0 (1C), 127.0 (1C), 127.1 (1C), 127.9 (2C), 128.3 (1C), 128.5 (2C), 130.3 (1C), 131.4 (1C), 132.5 (1C), 133.1 (1C), 143.0 (1C), 151.1 (1C), 162.5 (1C), 164.8 (1C), 166.7 (1C). IR (neat) (cm–1) ν 3202, 2951, 2194, 1715, 1704, 1661, 1592, 1510, 1426, 1334, 1232, 1070, 782. HRMS (ESI+) calcd for C27H22N4NaO5 505.1482; found 505.1454 [M + Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(p-tolyl)-1,4-dihydropyridine-2,3-dicarboxylate (
10ai) [
19]: Following the general procedure, compound
10ai was obtained after 120 h of reaction at room temperature and was purified by column chromatography (
n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 44% yield (19.6 mg). The ee of the product was determined to be 50% by HPLC using a Daicel Chiralpak IC column (
n-hexane/
i-PrOH = 70:30, flow rate 1 mL min
−1, λ = 237.7 nm): τ
major = 15.9 min; τ
minor = 9.6 min. [α]
D24 = −19.4 (
c = 0.12, MeOH, 50% ee).
Dimethyl 6-amino-1-benzamido-5-cyano-4-(4-methoxyphenyl)-1,4-dihydropyridine-2,3-dicarboxylate (10aj): Following the general procedure, compound 10aj was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 40% yield (18.57 mg). mp 137–139 °C. The ee of the product was determined to be 40% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 237.7 nm): τmajor = 48.4 min; τminor = 19.2 min. [α]D24 = −24.0 (c = 0.05, MeOH, 40% ee). 1H-NMR (400 MHz, DMSO-d6) 3.52 (s, 3H), 3.55 (s, 0.75H), 3.63 (s, 2.25H), 3.75 (s, 3H), 4.30 (s, 0.75H), 4.42 (s, 0.25H), 6.32 (s, 1.5H), 6.38 (s, 0.50H), 6.89 (d, J = 8.5 Hz, 2.25H), 7.10–7.22 (m, 0.75H), 7.43–7.68 (m, 4H), 7.79–7.88 (m, 2H), 11.19 (s, 0.75H), 11.30 (s, 0.25H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 38.4 (1C), 51.8 (1C), 52.7 (1C), 55.0 (1C), 58.9 (1C), 104.9 (1C), 113.6 (2C), 113.9 (min), 120.9 (1C), 127.9 (2C), 128.5 (2C), 128.8 (2C), 131.3 (1C), 132.5 (1C), 137.9 (1C), 142.2 (1C), 151.0 (1C), 158.2 (1C), 162.5 (1C), 164.8 (1C), 166.7 (1C). IR (neat) (cm–1) ν 3313, 3201, 2952, 2838, 2186, 1742, 1707, 1651, 1606, 1509, 1428, 1227, 1175, 1110, 1028, 833, 691. HRMS (ESI+) calcd for C24H22N4NaO6 485.1438; found 485.1435 [M + Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(furan-2-yl)-1,4-dihydropyridine-2,3-dicarboxylate (10ak): Following the general procedure, compound 10ak was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 70% yield (29.77 mg). mp 151–153 °C. The ee of the product was determined to be 54% by HPLC using a Daicel Chiralpak IA column (n-hexane/i-PrOH = 80:20, flow rate 1 mL min−1, λ = 236.6 nm): τmajor = 16.4 min; τminor = 12.4 min. [α]D24 = −15.3 (c = 0.10, MeOH, 54% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.55 (s, 1.35H), 3.58 (s, 1.35H), 3.59 (s, 1.65H), 3.65 (s, 1.65H), 4.51 (s, 0.55H), 4.64 (s, 0.45H), 6.11 (d, J = 3.1 Hz, 0.45H), 6.33 (d, J = 3.1 Hz, 0.55H), 6.37–6.45 (m, 1H), 6.50 (s, 1.1H), 6.57 (s, 0.9H), 7.46–7.67 (m, 4H), 7.77–7.86 (m, 2H), 11.20 (s, 0.55H), 11.38 (s, 0.45H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 32.4 (1C), 33.0 (min), 52.0 (min), 52.1 (1C), 52.7 (min), 52.9 (1C), 53.8 (min), 55.5 (1C), 101.8 (min), 102.3 (1C), 105.3 (min), 106.0 (1C), 110.6 (min), 110.8 (1C), 120.8 (1C), 120.9 (min), 127.8 (1C), 128.0 (min), 128.3 (min), 128.5 (1C), 131.3 (1C), 131.4 (min), 132.3 (min), 132.5 (1C), 141.6 (1C), 142.3 (min), 143.1 (1C), 143.7 (min), 152.0 (1C), 152.6 (min), 156.4 (min), 157.6 (1C), 162.1 (min), 162.2 (1C), 164.5 (1C), 164.7 (min), 166.1 (min), 166.2 (1C). IR (neat) (cm–1) ν 3527, 3406, 3328, 2175, 1735, 1707, 1652, 1578, 1431, 1336, 1225, 1013, 939, 754, 705. HRMS (ESI+) calcd for C21H18N4NaO6 445.1125; found 445.1125 [M + Na].
Dimethyl 6-amino-1-benzamido-5-cyano-4-(thiophen-2-yl)-1,4-dihydropyridine-2,3-dicarboxylate (10al): Following the general procedure, compound 10al was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100), as a white solid in 76% yield (33.18 mg). mp 124–126 °C. The ee of the product was determined to be 43% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 236.6 nm): τmajor = 27.1 min; τminor = 16.1 min. [α]D24 = −12.6 (c = 0.10, MeOH, 43% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.55 (s, 1.11H), 3.60 (s, 3H), 3.64 (s, 1.89H), 4.66 (s, 0.63H), 4.82 (s, 0.37H), 6.50 (s, 1.26H), 6.57 (s, 0.74H), 6.88 (s, 0.37H), 7.00 (s, 1H), 7.19 (s, 0.63H), 7.39 (d, J = 4.7 Hz, 1H), 7.52 (t, J = 6.4 Hz, 2H), 7.62 (t, J = 6.7 Hz, 1H), 7.83 (d, J = 7.4 Hz, 2H), 11.21 (s, 0.63H), 11.38 (d, 0.37H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 33.7 (1C), 52.0 (1C), 52.8 (1C), 57.9 (1C), 104.3 (1C), 120.8 (1C), 124.5 (1C), 124.9 (1C), 127.1 (1C), 127.9 (2C), 128.3 (min), 128.5 (2C), 131.4 (1C), 132.4 (1C), 142.3 (1C), 148.7 (1C), 151.6 (1C), 162.2 (1C), 164.5 (1C), 166.2 (1C). IR (neat) (cm–1) ν 3415, 3332, 3248, 3213, 2954, 2191, 1719, 1685, 1660, 1587, 1434, 1337, 1230, 941, 707, 687. HRMS (ESI+) calcd for C21H18N4NaO5S 461.0898; found 461.0897 [M + Na].
Dimethyl 6′-amino-1′-benzamido-5′-cyano-1′,4′-dihydro-[3,4′-bipyridine]-2′,3′-dicarboxylate (10am): Following the general procedure, compound 10am was obtained after 120 h of reaction at room temperature and was purified by column chromatography (n-hexane:diethyl ether 20:80 to 0:100 to ethyl acetate 100), as a white solid in 92% yield (39.66 mg). mp 157–159 °C. The ee of the product was determined to be 40% by HPLC using a Daicel Chiralpak IC column (n-hexane/i-PrOH = 70:30, flow rate 1 mL min−1, λ = 336.0 nm): τmajor = 32.9 min; τminor = 50.6 min. [α]D24 = −12.6 (c = 0.17, MeOH, 40% ee). 1H-NMR (400 MHz, DMSO-d6) δ 3.52 (s, 3H), 3.64 (s, 3H), 4.44 (s, 1H), 6.53 (s, 2H), 7.42 (dd, J = 8 Hz, 5 Hz, 1H), 7.52 (t, J = 7.5 Hz, 2H), 7.63 (t, J = 7.3 Hz, 1H), 7.87 (d, J = 7.3 Hz, 2H), 8.05 (s, 1H), 8.47 (dd, J = 4.7 Hz, 1.6 Hz, 1H), 8.67 (s, 1H), 11.31 (s, 1H). 13C-APT-NMR (100 MHz, DMSO-d6) δ 36.9 (1C), 45.8 (1C), 52.0 (1C), 52.9 (1C), 104.1 (1C), 120.6 (1C), 123.8 (1C), 127.9 (2C), 128.5 (2C), 131.2 (1C), 132.6 (1C), 135.3 (1C), 141.0 (1C), 143.1 (1C), 148.2 (1C), 148.7 (1C), 151.5 (1C), 162.2 (1C), 164.4 (1C), 166.8 (1C). IR (neat) (cm–1) ν 3317, 3226, 2952, 2185, 1749, 1707, 1685, 1654, 1578, 1425, 1330, 1275, 1221, 1115, 1029, 703. HRMS (ESI+) calcd for C22H20N5O5 434.1468; found 434.1467 [M + H].