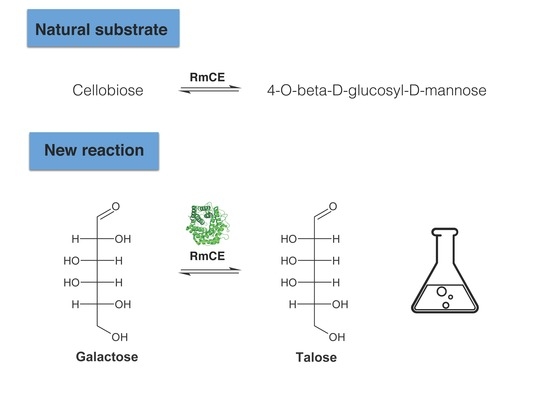
Converting Galactose into the Rare Sugar Talose with Cellobiose 2-Epimerase as Biocatalyst
Abstract
1. Introduction
2. Results
2.1. Substrate Specificity Analysis of Rhodothermus marinus (RmCE) Revealed One-Step Conversion from d-Galactose to d-Talose
2.2. Production of d-Talose
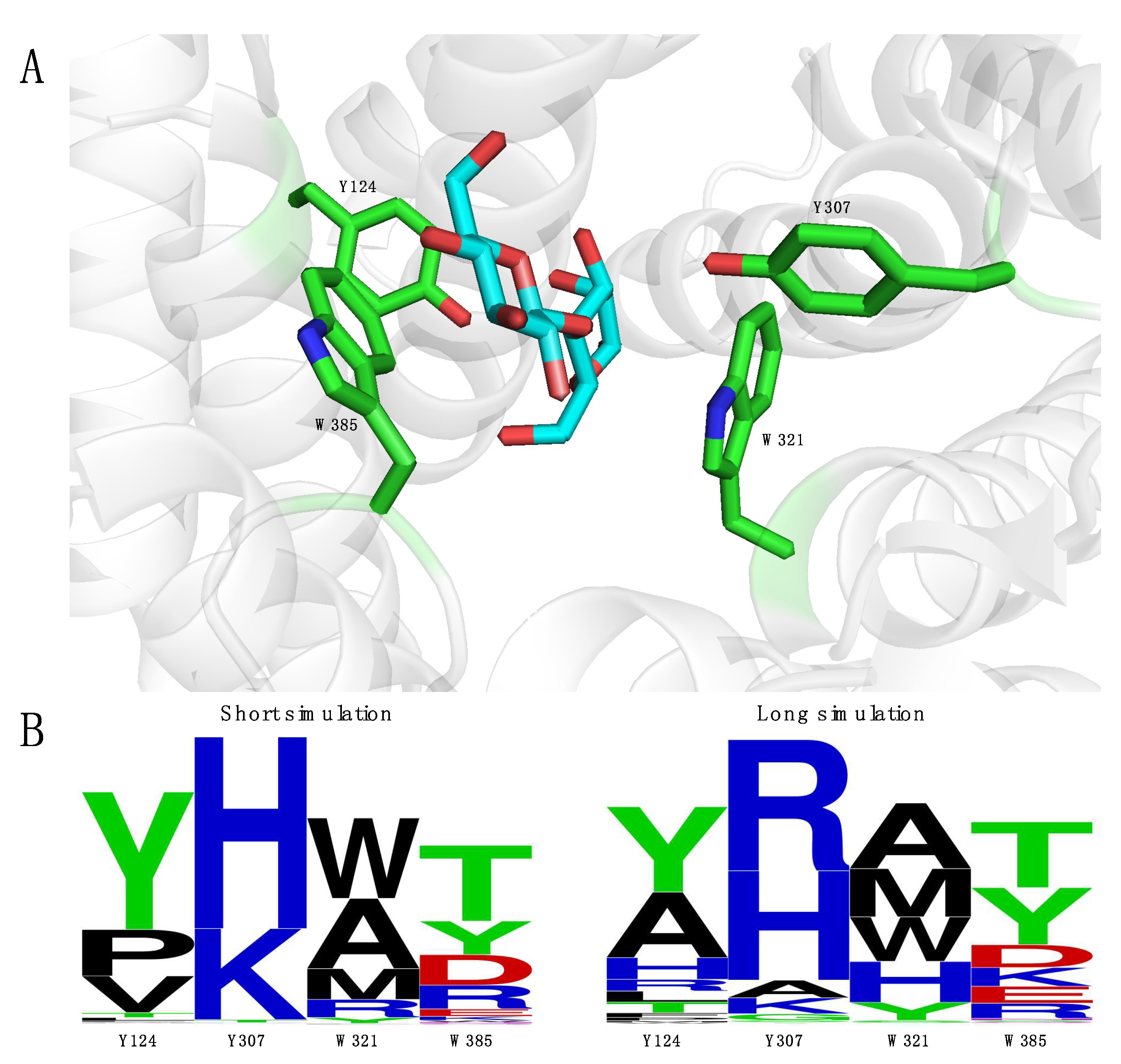
2.3. Engineering of RmCE to Increase Activity on Monosaccharide Substrates
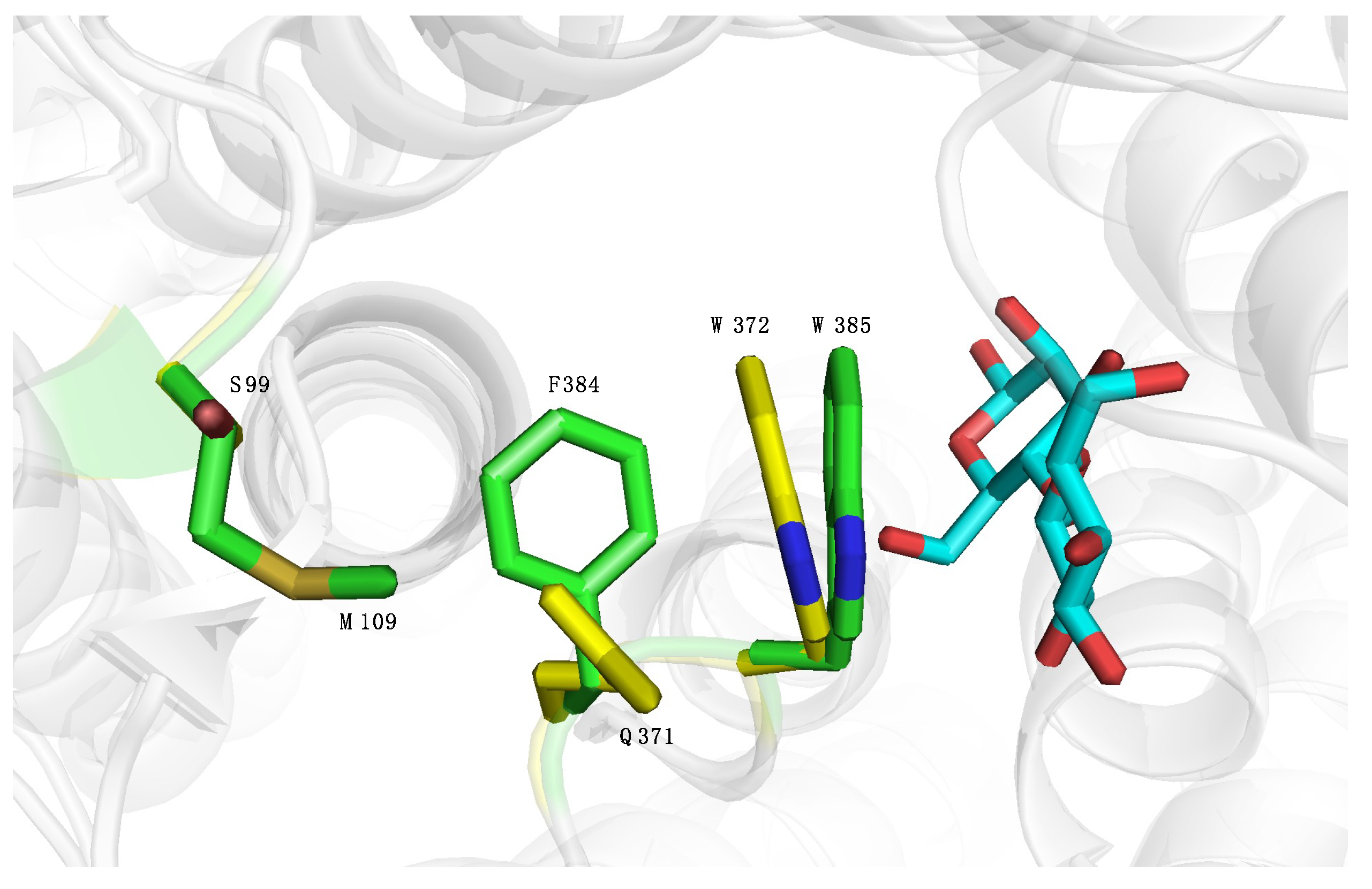
2.4. Introducing Two RmCE Residues in Caldicellulosiruptor saccharolyticus (CsCE) Led to A Twofold kcat Increase
3. Discussion
4. Materials and Methods
4.1. Gene Cloning of Cellobiose 2-Epimerase
4.2. Enzyme Production and Purification
4.3. Substrate Specificity Analysis
4.4. Kinetic Parameters of RmCE
4.5. Talose Production
4.6. Developing and Screening Two Mutant Libraries to Increase Activity on Monosaccharide Substrates
4.6.1. Generation of Mutant Libraries
4.6.2. Screening of Mutant Libraries
4.7. Cloning of CsCE and S99M/Q371F Variant, Enzyme Expression, Purification and Activity Tests
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| 50 mM Std | 100 mM Std | 200 mM Std | 300 mM Std | 400 mM Std | 750 mM Std | 1 M Std |
|---|---|---|---|---|---|---|
| 50:0 | 100:0 | 200:0 | 300:0 | 400:0 | 750:0 | 1000:0 |
| 49:1 | 98:2 | 198:2 | 295:5 | 395:5 | 740:10 | 990:10 |
| 48:2 | 96:4 | 195:5 | 290:10 | 390:10 | 730:20 | 980:20 |
| 47:3 | 94:6 | 192:8 | 280:20 | 380:20 | 710:40 | 970:30 |
| 46:4 | 92:8 | 190:10 | 275:25 | 370:30 | 700:50 | 950:50 |
| 45:5 | 90:10 | 180:20 | 270:30 | 360:40 | 690:60 | 930:70 |
Appendix B. Employing Rosetta EnzDes to Determine Useful Amino Acid Substitutions on Target Positions
Appendix C
Appendix C.1. Primers for the Amplification of Prefragments
| Primer Name | Primer |
| RmCE_bib_A125_Fw | GCCCAGAGTTTTGCTATC |
| RmCE_bib_Y306_Rv | ATACAGAGAGCCATCCG |
| RmCE_bib_W322_Fw | TGGCCGCAAGCCGAA |
| RmCE_bib_F384_Rv | GAAATCGACTTTGTCATCCG |
| RmCE_bib_K386_Fw | AAAGGTCCGTATCATAATGGC |
| RmCE_bib_V123_Rv | AACATGTTTACGGGTGTC |
Appendix C.2. Primers for the Long Library Fragments
| Primer Name | Primer |
| RmCE_bib_Y124Y_Fw | CCGCTGGACACCCGTAAACATGTTTATGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y124P_Fw | CCGCTGGACACCCGTAAACATGTTCCGGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y124V_Fw | CCGCTGGACACCCGTAAACATGTTGTGGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y307YH_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCATRATACAGAGAGCCATCCG |
| RmCE_bib_Y307K_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCTTTATACAGAGAGCCATCCG |
| RmCE_bib_W321WR_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACYGGTGGCCGCAAGCCGAA |
| RmCE_bib_W321A_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACGCGTGGCCGCAAGCCGAA |
| RmCE_bib_W321M_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACATGTGGCCGCAAGCCGAA |
| RmCE_bib_W385WR_Rv | CGCGGCCATTATGATACGGACCTTTCCRGAAATCGACTTTGTCATCCG |
| RmCE_bib_W385T_Rv | CGCGGCCATTATGATACGGACCTTTGGTGAAATCGACTTTGTCATCCG |
| RmCE_bib_W385YD_Rv | CGCGGCCATTATGATACGGACCTTTATMGAAATCGACTTTGTCATCCG |
Appendix C.3. Primers for the Short Library Fragments
| Primer Name | Primer |
| RmCE_bib_Y124YH_Fw | CCGCTGGACACCCGTAAACATGTTYATGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y124A_Fw | CCGCTGGACACCCGTAAACATGTTGCGGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y124L_Fw | CCGCTGGACACCCGTAAACATGTTCTGGCCCAGAGTTTTGCTATC |
| RmCE_bib_Y307YH_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCATRATACAGAGAGCCATCCG |
| RmCE_bib_Y307K_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCTTTATACAGAGAGCCATCCG |
| RmCE_bib_Y307R_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCGCGATACAGAGAGCCATCCG |
| RmCE_bib_Y307A_Rv | GTCGGTATCCAGATGGCCCTGTTCACCAATTTCCGCATACAGAGAGCCATCCG |
| RmCE_bib_W321A_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACGCGTGGCCGCAAGCCGAA |
| RmCE_bib_W321M_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACATGTGGCCGCAAGCCGAA |
| RmCE_bib_W321YH_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACYATTGGCCGCAAGCCGAA |
| RmCE_bib_W321W_Fw | GTGAACAGGGCCATCTGGATACCGACCGTCACTGGTGGCCGCAAGCCGAA |
| RmCE_bib_W385KE_Rv | CGCGGCCATTATGATACGGACCTTTTTYGAAATCGACTTTGTCATCCG |
| RmCE_bib_W385WR_Rv | CGCGGCCATTATGATACGGACCTTTCCRGAAATCGACTTTGTCATCCG |
| RmCE_bib_W385T_Rv | CGCGGCCATTATGATACGGACCTTTGGTGAAATCGACTTTGTCATCCG |
| RmCE_bib_W385YD_Rv | CGCGGCCATTATGATACGGACCTTTATMGAAATCGACTTTGTCATCCG |
References
- Van Overtveldt, S.; Verhaeghe, T.; Joosten, H.-J.; van den Bergh, T.; Beerens, K.; Desmet, T. A structural classification of carbohydrate epimerases: From mechanistic insights to practical applications. Biotechnol. Adv. 2015, 33, 1814–1828. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Kim, Y.S.; Kang, L.W.; Oh, D.K. Characterization of a recombinant cellobiose 2-epimerase from Dictyoglomus turgidum that epimerizes and isomerizes beta-1,4- and alfa-1,4-gluco-oligosaccharides. Biotechnol. Lett. 2012, 34, 2061–2068. [Google Scholar] [CrossRef] [PubMed]
- Uechi, K.; Takata, G.; Fukai, Y.; Yoshihara, A.; Morimoto, K. Gene cloning and characterization of L-ribulose 3-epimerase from Mesorhizobium loti and its application to rare sugar production. Biosci. Biotechnol. Biochem. 2013, 77, 511–515. [Google Scholar] [CrossRef] [PubMed]
- Park, C.-S.; Kim, J.-E.; Choi, J.-G.; Oh, D.-K. Characterization of a recombinant cellobiose 2-epimerase from Caldicellulosiruptor saccharolyticus and its application in the production of mannose from glucose. Appl. Microbiol. Biotechnol. 2011, 92, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Ojima, T.; Saburi, W.; Sato, H.; Yamamoto, T.; Mori, H.; Matsui, H. Biochemical characterization of a thermophilic cellobiose 2-epimerase from a thermohalophilic bacterium, Rhodothermus marinus JCM9785. Biosci. Biotechnol. Biochem. 2011, 75, 2162–2168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, T.; Jiang, B.; Mu, W. Biochemical characterization of a d-psicose 3-epimerase from Treponema primitia ZAS-1 and its application on enzymatic production of d-psicose. J. Sci. Food Agric. 2015, 96, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Beerens, K.; Desmet, T.; Soetaert, W. Enzymes for the biocatalytic production of rare sugars. J. Ind. Microbiol. Biotechnol. 2012, 39, 823–834. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.H.; Park, S.J.; Lee, H.G. d-Psicose, a Sweet Monosaccharide, Ameliorate Hyperglycemia, and Dyslipidemia in C57BL/6J db/db Mice. J. Food Sci. 2010, 75, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Wagner, N.; Bosshart, A.; Failmezger, J.; Bechtold, M.; Panke, S. A Separation-Integrated Cascade Reaction to Overcome Thermodynamic Limitations in Rare-Sugar Synthesis. Angew. Chem. Int. Ed. 2015, 127. [Google Scholar] [CrossRef]
- Fujiwara, T.; Saburi, W.; Matsui, H.; Mori, H.; Yao, M. Structural insights into the epimerization of β-1,4-linked oligosaccharides catalyzed by cellobiose 2-epimerase, the sole enzyme epimerizing non-anomeric hydroxyl groups of unmodified sugars. J. Biol. Chem. 2014, 289, 3405–3415. [Google Scholar] [CrossRef] [PubMed]
- Krewinkel, M.; Kaiser, J.; Merz, M.; Rentschler, E.; Kuschel, B.; Hinrichs, J.; Fischer, L. Novel cellobiose 2-epimerases for the production of epilactose from milk ultrafiltrate containing lactose. J. Dairy Sci. 2015, 98, 3665–3678. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Xu, C.; Li, S.; Liang, J.; Xu, H.; Xu, Z. Efficient production of lactulose from whey powder by cellobiose 2-epimerase in an enzymatic membrane bioreactor. Bioresour. Technol. 2017, 233, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Linington, R.G.; Robertson, M.; Gauthier, A.; Finlay, B.B.; MacMillan, J.B.; Molinski, T.F.; Van Soest, R.; Andersen, R.J. Caminosides B-D, antimicrobial glycolipids isolated from the marine sponge Caminus sphaeroconia. J. Nat. Prod. 2006, 69, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Haukaas, M.H.; O’Doherty, G.A. Enantioselective synthesis of N-Cbz-protected 6-amino-6-deoxymannose, -talose, and -gulose. Org. Lett. 2001, 3, 3899–3902. [Google Scholar] [CrossRef] [PubMed]
- Sznaidman, M.L.; Hecht, S.M. Studies on the total synthesis of tallysomycin. Synthesis of the threonylbithiazole moiety containing a structurally unique glycosylcarbinolamide. Org. Lett. 2001, 3, 2811–2814. [Google Scholar] [CrossRef] [PubMed]
- Beerens, K.; Van Overtveldt, S.; Desmet, T. The “epimerring” highlights the potential of carbohydrate epimerases for rare sugar production. Biocatal. Biotransform. 2017, 35, 230–237. [Google Scholar] [CrossRef]
- Öberg, C.T.; Blanchard, H.; Leffler, H.; Nilsson, U.J. Protein subtype-targeting through ligand epimerization: Talose-selectivity of galectin-4 and galectin-8. Bioorg. Med. Chem. Lett. 2008, 18, 3691–3694. [Google Scholar] [CrossRef] [PubMed]
- Hodosi, G.; Kováč, P. Epimerization of carbohydrates via stannylene acetals. A practical synthesis of D-Talose. J. Carbohydr. Chem. 1998, 17, 557–565. [Google Scholar] [CrossRef]
- Xiao, H.; Wang, G.; Wang, P.; Li, Y. Convenient synthesis of D-talose from D-galactose. Chinese J. Chem. 2010, 28, 1229–1232. [Google Scholar] [CrossRef]
- Fonseca, G.G.; De Carvalho, N.M.B.; Gombert, A.K. Growth of the yeast Kluyveromyces marxianus CBS 6556 on different sugar combinations as sole carbon and energy source. Appl. Microbiol. Biotechnol. 2013, 97, 5055–5067. [Google Scholar] [CrossRef] [PubMed]
- Beniwal, A.; Saini, P.; Kokkiligadda, A.; Vij, S. Physiological growth and galactose utilization by dairy yeast Kluyveromyces marxianus in mixed sugars and whey during fermentation. 3 Biotech 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Kuboki, A.; Okazaki, H.; Sugai, T.; Ohta, H. An expeditious route to N-glycolylneuraminic acid based on enzyme-catalyzed reaction. Tetrahedron 1997, 53, 2387–2400. [Google Scholar] [CrossRef]
- Arbor, A.; Rackham, T.; Rackham, H. Glucosamine metabolism I. N-acetylglucosamine deacetylase. J. Biol. Chem. 1957, 226, 115–124. [Google Scholar]
- Trinder, P. Determination of blood glucose using an oxidase-peroxidase system with a non-carcinogenic chromogen. J. Clin. Pathol. 1969, 22, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Reetz, M.T.; Kahakeaw, D.; Lohmer, R. Addressing the numbers problem in directed evolution. Chembiochem 2008, 9, 1797–1804. [Google Scholar] [CrossRef] [PubMed]
- Röthlisberger, D.; Khersonsky, O.; Wollacott, A.M.; Jiang, L.; DeChancie, J.; Betker, J.; Gallaher, J.L.; Althoff, E.A; Zanghellini, A.; Dym, O.; et al. Kemp elimination catalysts by computational enzyme design. Nature 2008, 453, 190. [Google Scholar] [CrossRef] [PubMed]
- Zanghellini, A.; Jiang, L.I.N.; Wollacott, A.M.; Cheng, G.; Meiler, J.; Althoff, E.A.; Ro, D. New algorithms and an in silico benchmark for computational enzyme design. Protein Sci. 2006, 15, 2785–2794. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Althoff, E.A.; Clemente, F.R.; Doyle, L.; Zanghellini, A.; Gallaher, J.L.; Betker, J.L.; Iii, C.F.B.; Hilvert, D.; Houk, K.N.; et al. De novo computational design of retro-aldo enzymes. Science 2008, 319, 1387–1391. [Google Scholar] [CrossRef] [PubMed]
- Crooks, G.; Hon, G.; Chandonia, J.; Brenner, S. WebLogo: A sequence logo generator. Genome Res 2004, 14, 1188–1190. [Google Scholar] [CrossRef] [PubMed]
- Mu, W.; Li, Q.; Fan, C.; Zhou, C.; Jiang, B. Recent advances on physiological functions and biotechnological production of epilactose. Appl. Microbiol. Biotechnol. 2013, 97, 1821–1827. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Taguchi, H.; Hamada, S.; Kawauchi, S.; Ito, H.; Senoura, T.; Watanabe, J.; Nishimukai, M.; Ito, S.; Matsui, H. Enzymatic properties of cellobiose 2-epimerase from Ruminococcus albus and the synthesis of rare oligosaccharides by the enzyme. Appl. Microbiol. Biotechnol. 2008, 79, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Levin, R.; Zhang, W.; Zhang, T.; Jiang, B.; Stressler, T.; Fischer, L.; Mu, W. Characterisation of a novel cellobiose 2-epimerase from thermophilic Caldicellulosiruptor obsidiansis for lactulose production. J. Sci. Food Agric. 2017, 97, 3095–3105. [Google Scholar] [CrossRef] [PubMed]
- De Castro, C.; Gargiulo, V.; Lanzetta, R.; Parrilli, M. Agrobacterium rubiT DSM 6772 produces a lipophilic polysaccharide capsule whose degree of acetylation is growth modulated. Biomacromolecules 2007, 8, 1047–1051. [Google Scholar] [CrossRef] [PubMed]
- Haukaas, M.H.; O’Doherty, G.A. Synthesis of D- and L-Deoxymannojirimycin via an Asymmetric Aminohydroxylation of Vinylfuran. Org. Lett. 2001, 3, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Czechura, P.; Tam, R.Y.; Dimitrijevic, E.; Murphy, A.V.; Ben, R.N. The importance of hydration for inhibiting ice recrystallization with C-linked antifreeze glycoproteins. J. Am. Chem. Soc. 2008, 130, 2928–2929. [Google Scholar] [CrossRef] [PubMed]
- Hicks, K.B. Preparative HPLC of Carbohydrates. J. Chromatogr. Libr. 1995, 58, 361–387. [Google Scholar] [CrossRef]
- Stead, P. Isolation by preparative HPLC. Methods Biotechnol. 1998, 4, 165–208. [Google Scholar]
- Maru, I.; Ohnishi, J.; Ohta, Y.; Tsukada, Y. Simple and large-scale production of N-acetylneuraminic acid from N-acetyl-d-glucosamine and pyruvate using N-acyl-dglucosamine 2-epimerase and n-acetylneuraminate lyase. Carbohydr. Res. 1998, 306, 575–578. [Google Scholar] [CrossRef]
- Engler, C.; Kandzia, R.; Marillonnet, S. A one pot, one step, precision cloning method with high throughput capability. PLoS ONE 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Verhaeghe, T.; Aerts, D.; Diricks, M.; Soetaert, W.; Desmet, T. The quest for a thermostable sucrose phosphorylase reveals sucrose 6’-phosphate phosphorylase as a novel specificity. Appl. Microbiol. Biotechnol. 2014, 98, 7027–7037. [Google Scholar] [CrossRef] [PubMed]
- Aerts, D.; Verhaeghe, T.F.; Roman, B.I.; Stevens, C.V; Desmet, T.; Soetaert, W. Transglucosylation potential of six sucrose phosphorylases toward different classes of acceptors. Carbohydr. Res. 2011, 346, 1860–1867. [Google Scholar] [CrossRef] [PubMed]
- Quan, J.; Tian, J. PROTOCOL Circular polymerase extension cloning for high- throughput cloning of complex and combinatorial DNA libraries. Nat. Protoc. 2011, 6, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, J.; Fernández, L.; Carballeira, J.D.; Drone, J.; Gumulya, Y.; Höbenreich, H.; Kahakeaw, D.; Kille, S.; Lohmer, R.; Peyralans, J.J.-P.; et al. Improved PCR method for the creation of saturation mutagenesis libraries in directed evolution: Application to difficult-to-amplify templates. Appl. Microbiol. Biotechnol. 2008, 81, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Richter, F.; Leaver-Fay, A.; Khare, S.D.; Bjelic, S.; Baker, D. De novo enzyme design using Rosetta3. PLoS ONE 2011, 6, e19230. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
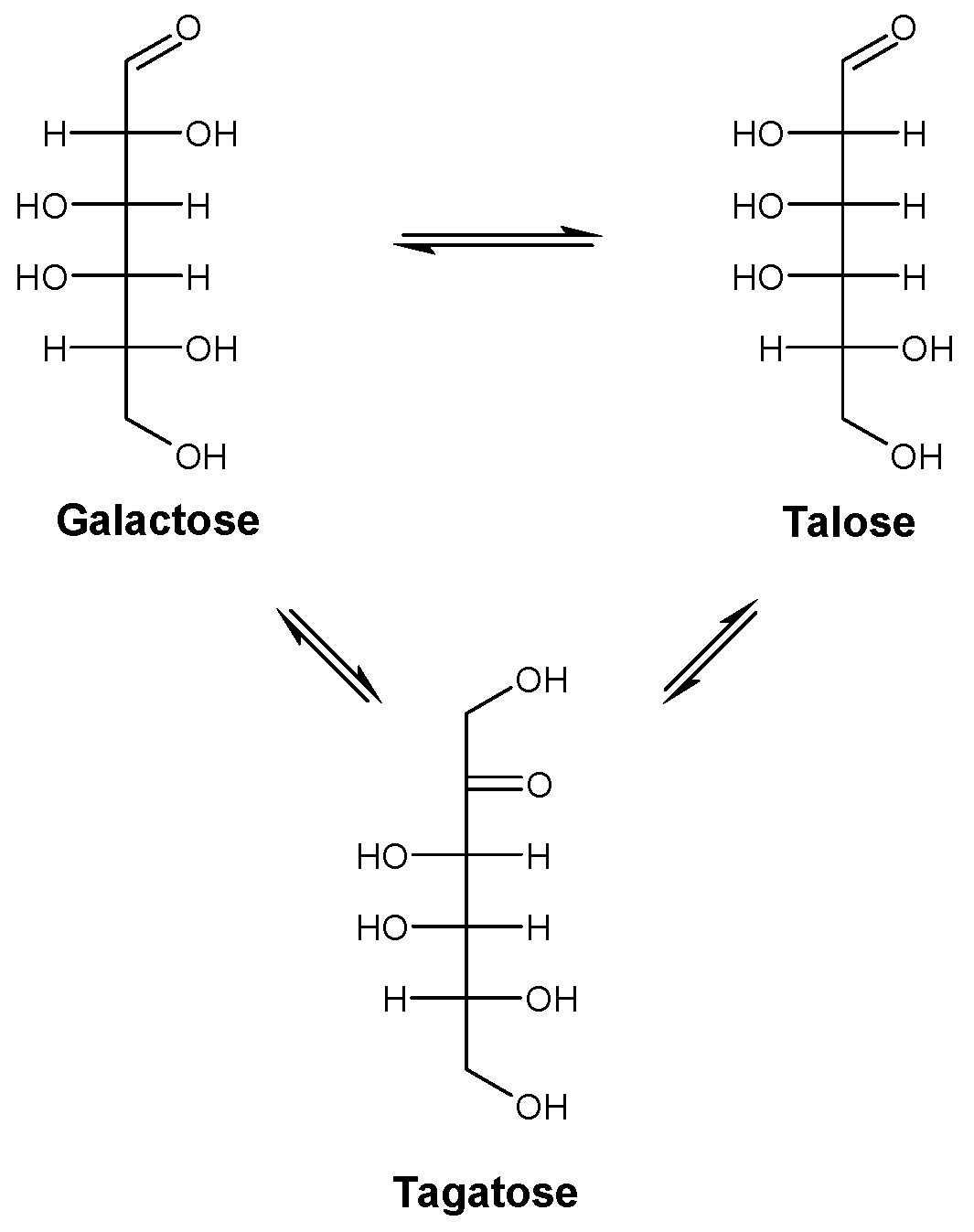

| Substrate | Product | RmCE Specific Activity (mU/mg) | CsCE Specific Activity (mU/mg) 1 |
|---|---|---|---|
| Cellobiose | 4-O-β-d-glucopyranosyl-d-mannose | 87,500 2 | 3600 ± 2.1 |
| d-mannose | d-glucose | 1130 ± 25 | 150 ± 0.5 |
| d-glucose | d-mannose | 440 ± 24 | 35 ± 0.5 |
| d-galactose | d-talose | 1020 ± 83 | / |
| d-xylose | d-lyxose | 38 ± 5 | 40 ± 0.7 |
| d-lyxose | d-xylose | 18 ± 0.2 | 60 ± 0.3 |
| l-arabinose | l-ribose | / | / |
| Substrate | Km (M) | kcat (s−1) | kcat/Km (s−1/M−1) |
|---|---|---|---|
| d-mannose | 0.45 ± 0.18 | 5.4 ± 1 | 12.0 ± 4.4 |
| d-galactose | 1.04 ± 0.13 | 5.0 ± 0.3 | 4.8 ± 0.7 |
| Time (h) | Purity (%) | Yield (%) | d-talose (mM) |
|---|---|---|---|
| 0.5 | >99 | 0.50 ± 0.30 | 8.07 ± 4.85 |
| 1 | >99 | 1.90 ± 0.16 | 30.36 ± 2.49 |
| 1.5 | 97.01 ± 2.59 | 2.68 ± 0.23 | 42.81 ± 3.76 |
| 3 | 93.65 ± 1.92 | 5.43 ± 0.08 | 86.90 ± 1.36 |
| 4.5 | 92.47 ± 1.26 | 8.97 ± 0.66 | 143.50 ± 10.50 |
| 7 | 91.22 ± 0.86 | 12.01 ± 0.82 | 192.14 ± 13.18 |
| 18 | 89.78 ± 1.53 | 19.28 ± 1.28 | 308.40 ± 20.44 |
| CsCE | Km (mM) | kcat (s−1) |
|---|---|---|
| WT | 412 ± 91 | 3.8 ± 0.2 |
| S99M/Q371F | 430 ± 94 | 7.5 ± 0.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Overtveldt, S.; Gevaert, O.; Cherlet, M.; Beerens, K.; Desmet, T. Converting Galactose into the Rare Sugar Talose with Cellobiose 2-Epimerase as Biocatalyst. Molecules 2018, 23, 2519. https://doi.org/10.3390/molecules23102519
Van Overtveldt S, Gevaert O, Cherlet M, Beerens K, Desmet T. Converting Galactose into the Rare Sugar Talose with Cellobiose 2-Epimerase as Biocatalyst. Molecules. 2018; 23(10):2519. https://doi.org/10.3390/molecules23102519
Chicago/Turabian StyleVan Overtveldt, Stevie, Ophelia Gevaert, Martijn Cherlet, Koen Beerens, and Tom Desmet. 2018. "Converting Galactose into the Rare Sugar Talose with Cellobiose 2-Epimerase as Biocatalyst" Molecules 23, no. 10: 2519. https://doi.org/10.3390/molecules23102519
APA StyleVan Overtveldt, S., Gevaert, O., Cherlet, M., Beerens, K., & Desmet, T. (2018). Converting Galactose into the Rare Sugar Talose with Cellobiose 2-Epimerase as Biocatalyst. Molecules, 23(10), 2519. https://doi.org/10.3390/molecules23102519