Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania
Abstract
1. Introduction
2. Results and Discussion
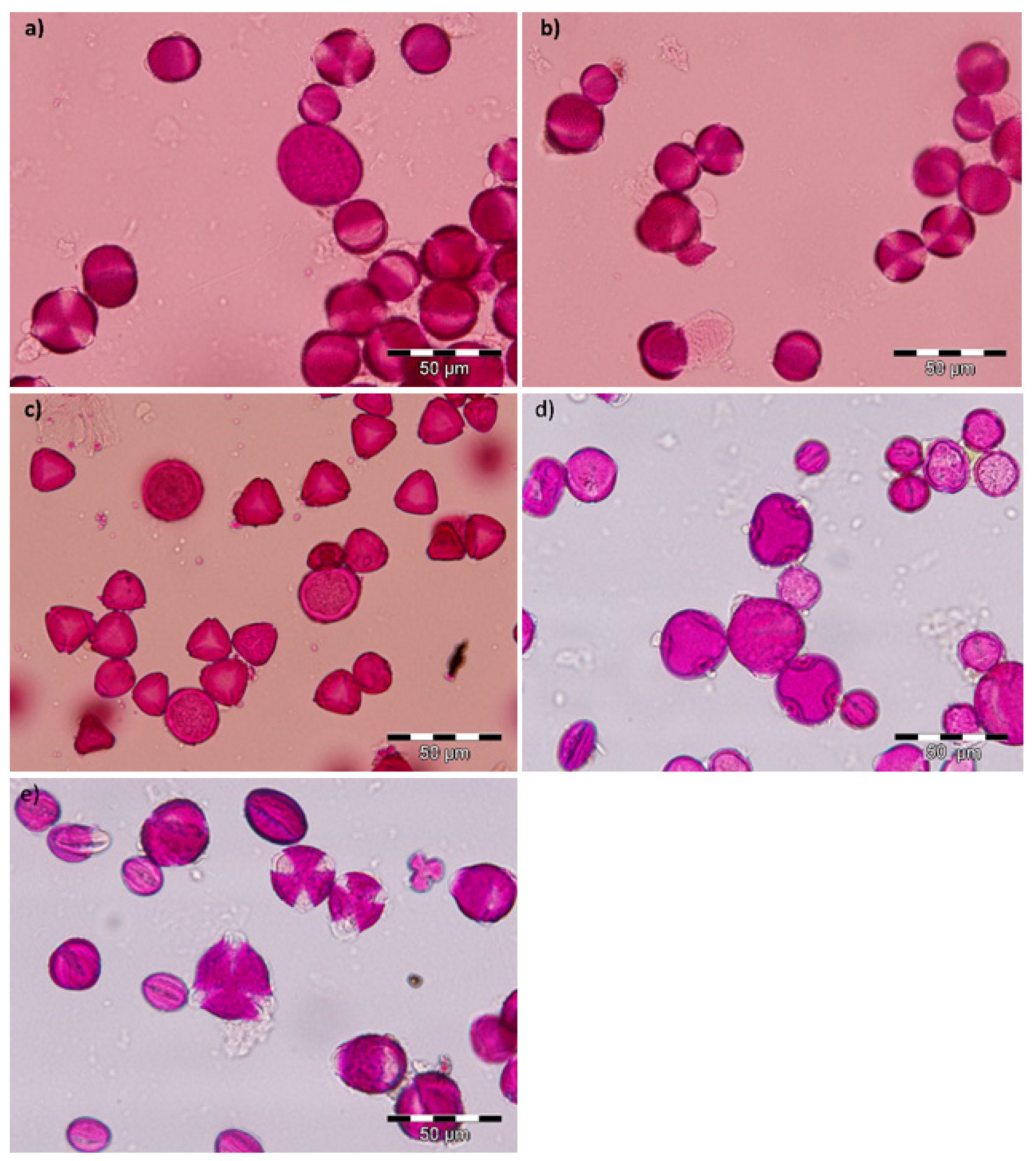
2.1. Microscopy Results
2.2. Sugars Content
2.3. Total Phenolic Content
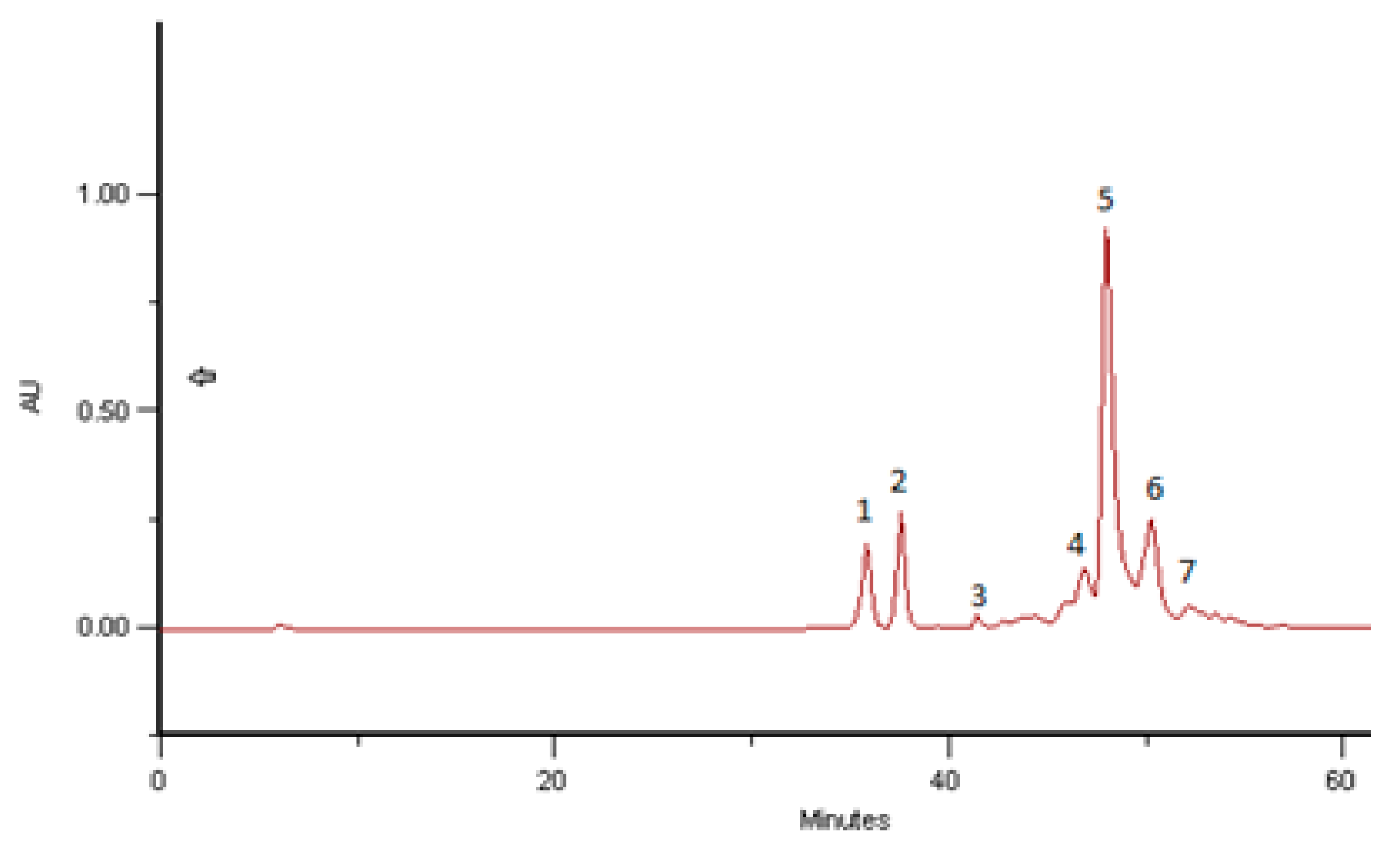
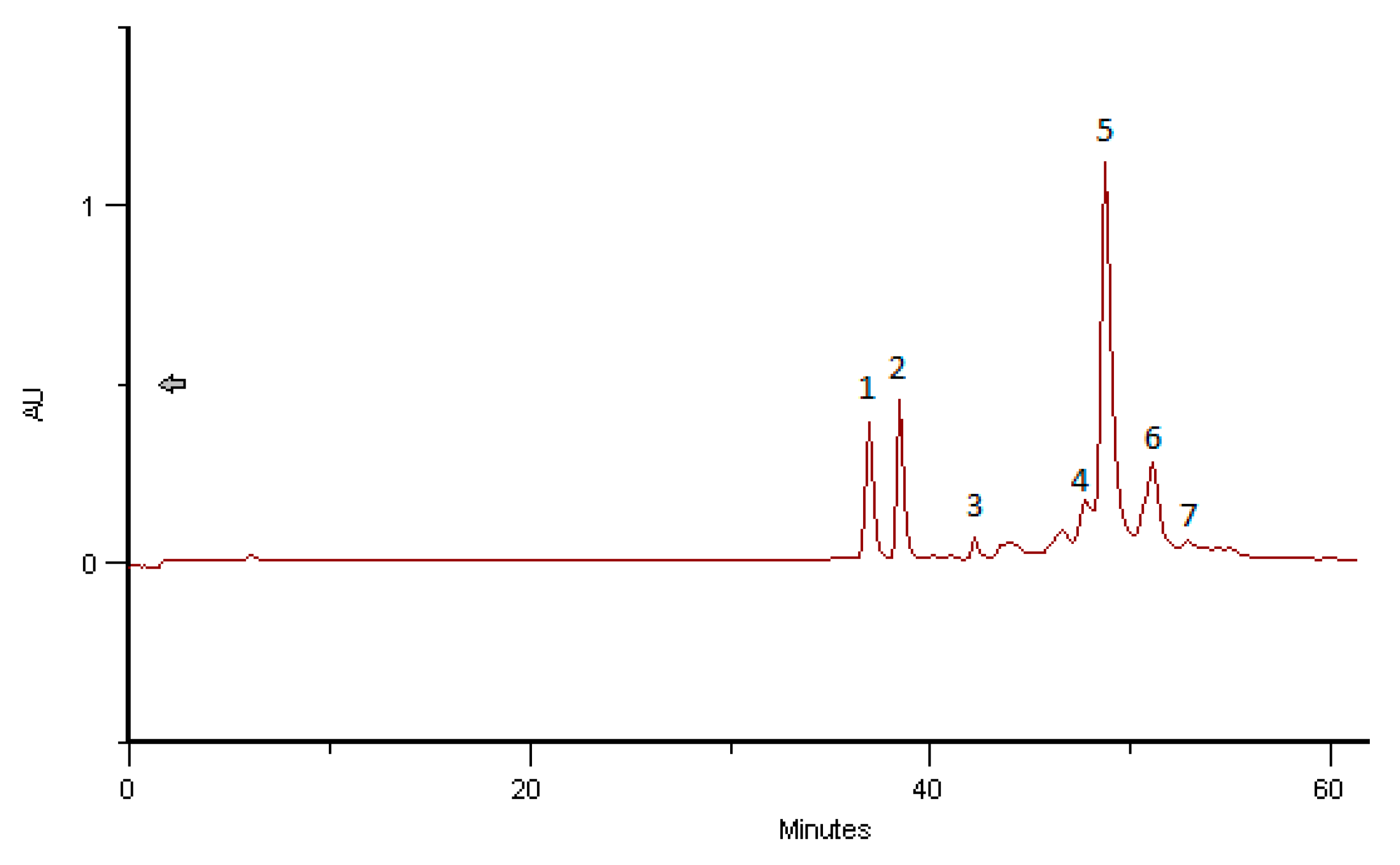
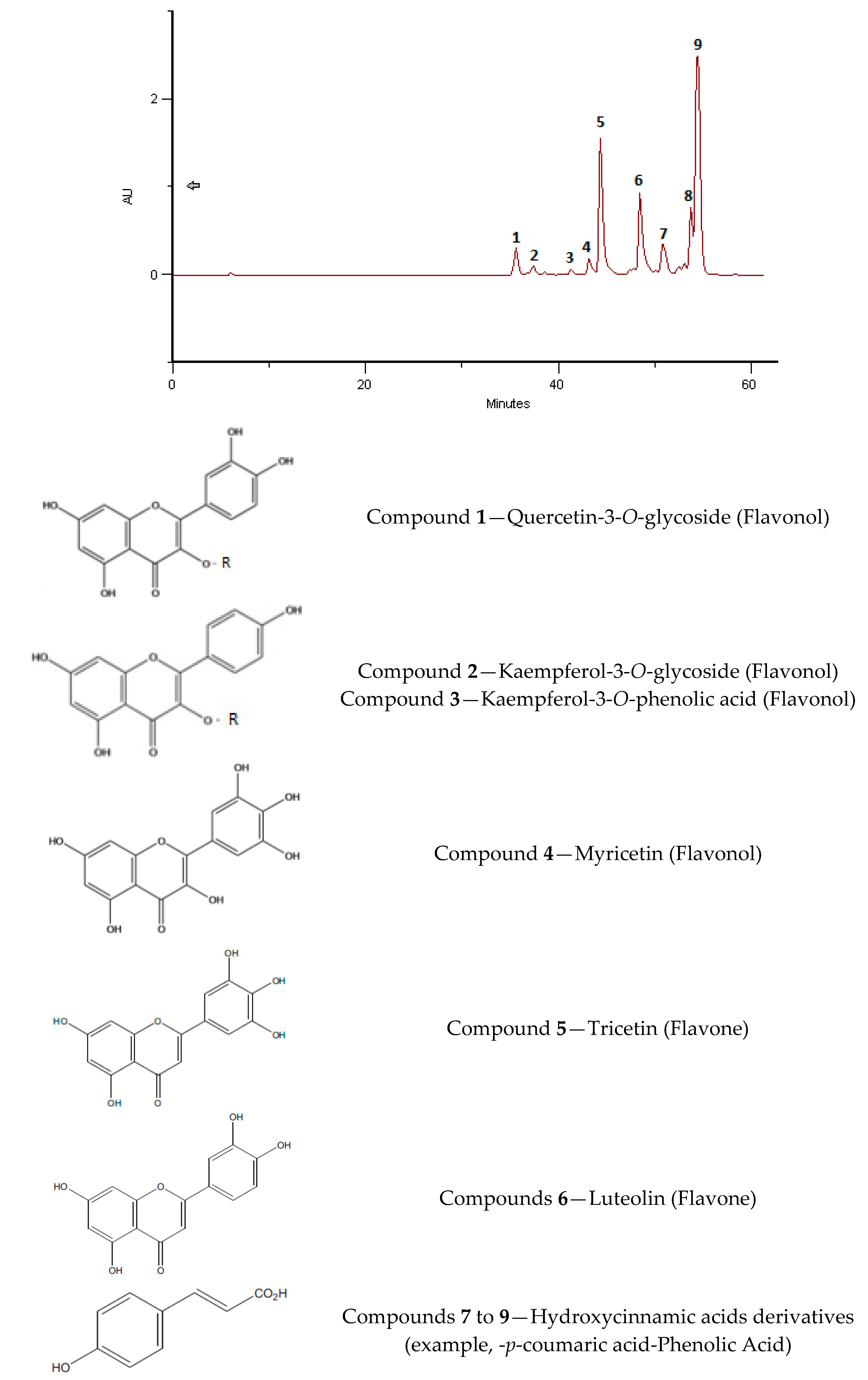
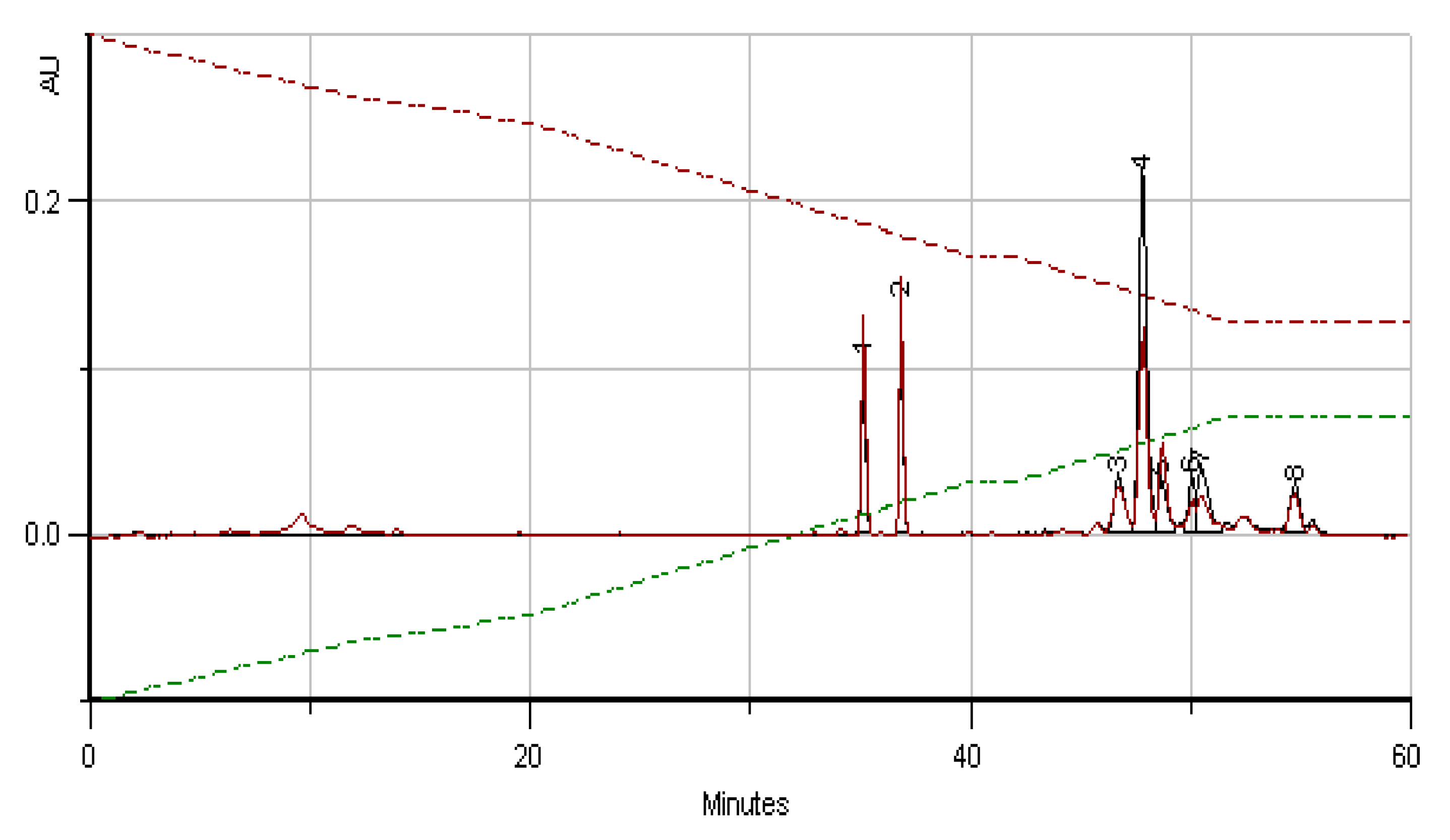
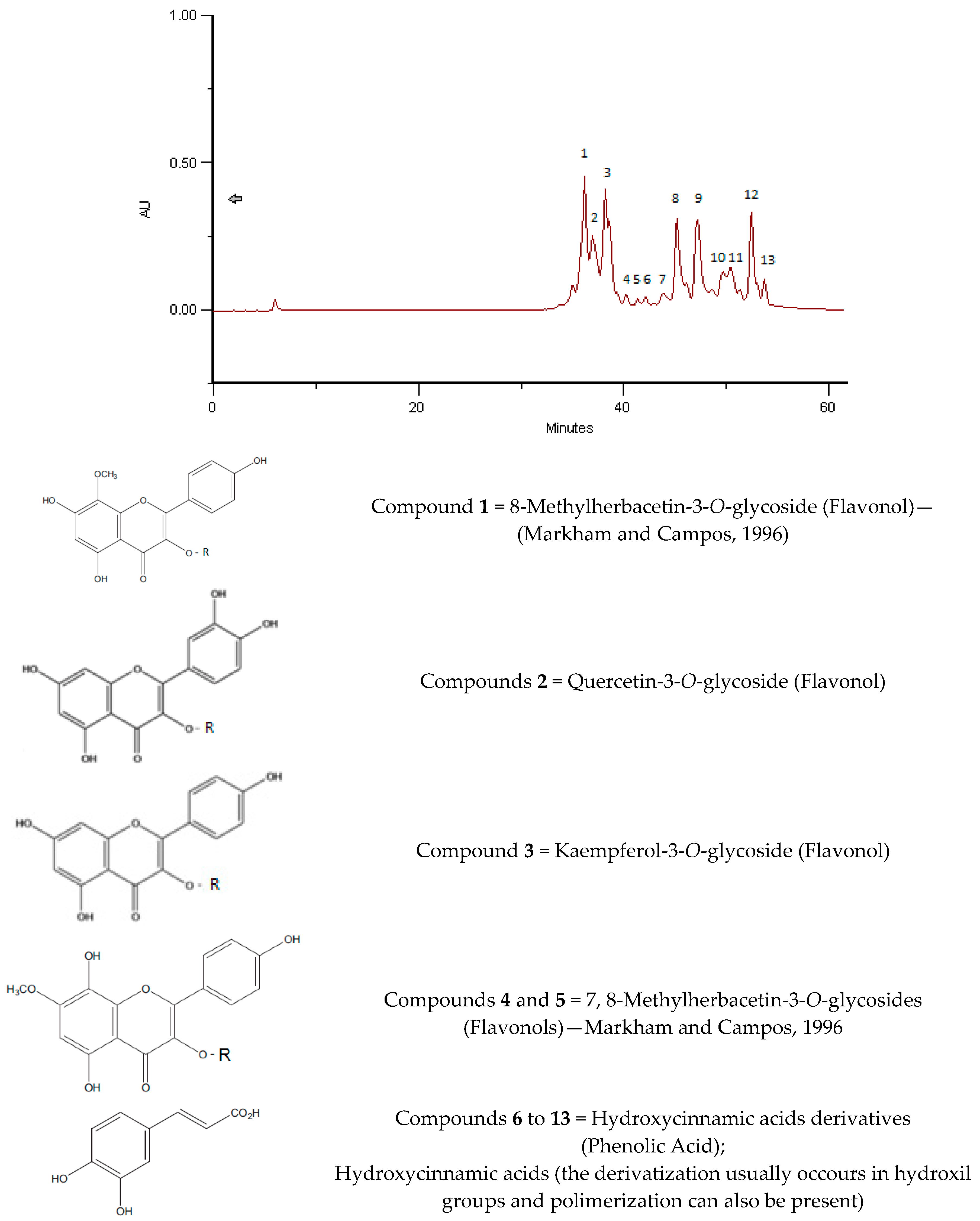
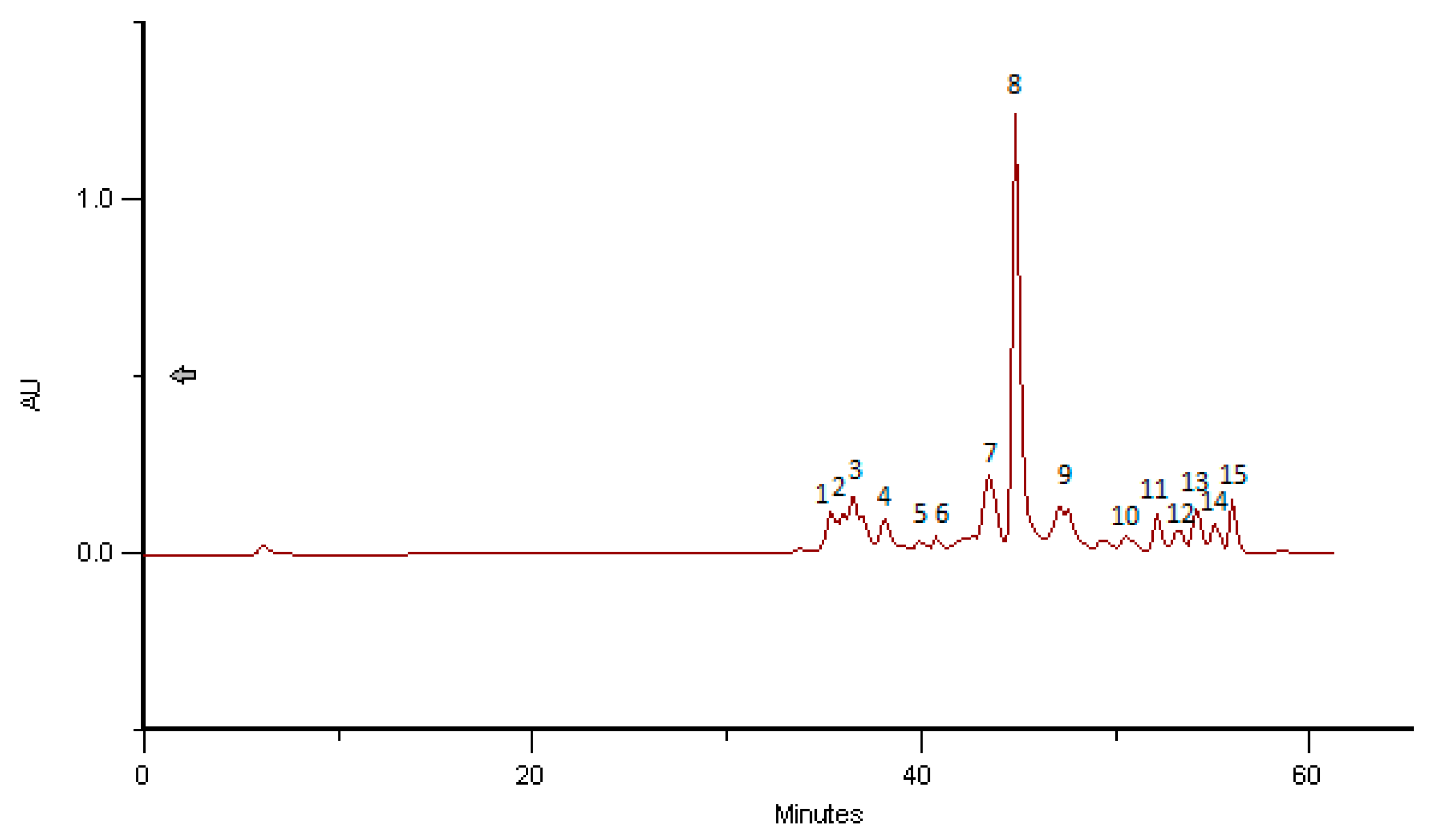
2.4. Phenolic and Polyphenolic Fingerprint by HPLC/DAD
3. Materials and Methods
3.1. Samples Collection
3.2. Standards and Reagents
3.3. Microscopic Analysis
3.4. Sugar Profiles
3.5. Total Phenolic and Flavonoids Content
3.5.1. Preparation of Extracts
3.5.2. Total Phenolic Content by Folin–Ciocateau Method
3.5.3. Total Flavonoid Content
3.6. Phenolic and Polyphenolic Fingerprint by HPLC/DAD
Sample Extraction
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vásquez, A.; Olofsson, T.C. The Lactic Acid Bacteria Involved in the Production of Bee Pollen and Bee Bread. J. Apic. Res. 2009, 48, 189–195. [Google Scholar] [CrossRef]
- Markiewicz-Zukowska, R.; Naliwajko, S.K.; Bartosiuk, E.; Moskwa, J.; Isidorov, V.; Soroczyńska, J.; Borawska, M.H. Chemical composition and antioxidant activity of beebread, and its influence on the glioblastoma cell line (U87MG). J. Apic. Sci. 2013, 57, 147–157. [Google Scholar] [CrossRef]
- Barene, I.; Daberte, I.; Siksna, S. Investigation of Bee Bread and Development of Its Dosage Forms. Med. Teorija Praktika 2014, 21, 16–22. [Google Scholar] [CrossRef]
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and bee bread as new health-oriented products: A review. Trends Food Sci. Technol. 2017, 71, 170–180. [Google Scholar] [CrossRef]
- Nagai, T.; Nagashima, T.; Myoda, T.; Inoue, R. Preparation and functional properties of extracts from bee bread. Food Nahrung 2004, 48, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Atkin, S.L.; Barrier, S.; Cui, Z.; Fletcher, P.D.; Mackenzie, G.; Panel, V.; Zhang, X. UV and visible light screening by individual sporopollenin exines derived from Lycopodium clavatum (club moss) and Ambrosia trifida (giant ragweed). J. Photochem. Photobiol. B Biol. 2011, 102, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Ivanišová, E.; Kačániová, M.; Frančáková, H.; Petrová, J.; Hutková, J.; Brovarskyi, V.; Musilová, J. Bee bread-perspective source of bioactive compounds for future. Potravin Slovak J. Food Sci. 2015, 9, 592–598. [Google Scholar] [CrossRef]
- Rimpler, M. Von bienen gesammelte blütenpollen: Eigenschaften und verwendung. Arztez. Naturheilverfahren 2003, 44, 158–165. [Google Scholar]
- Campos, M.G.; Frigerio, C.; Lopes, J.; Bogdanov, S. What Is the Future of Bee-Pollen? J. ApiProd. ApiMed. Sci. 2010, 2, 131–144. [Google Scholar] [CrossRef]
- Zuluaga, C.M.; Serrato, J.C.; Quicazan, M.C. Chemical, Nutritional and Bioactive Characterization of Colombian Bee-Bread. Chem. Eng. Trans. 2015, 43, 175–180. [Google Scholar] [CrossRef]
- Anderson, K.E.; Carroll, M.J.; Sheehan, T.I.M.; Mott, B.M.; Maes, P.; Corby-Harris, V. Hive-stored pollen of honey bees: Many lines of evidence are consistent with pollen preservation, not nutrient conversion. Mol. Ecol. 2014, 23, 5904–5917. [Google Scholar] [CrossRef] [PubMed]
- Nicolson, S.W. Bee food: The chemistry and nutritional value of nectar, pollen and mixtures of the two. Afr. Zool. 2011, 46, 197–204. [Google Scholar] [CrossRef]
- Čeksterytė, V.; Kurtinaitienė, B.; Venskutonis, P.R.; Pukalskas, A.; Kazernavičiūtė, R.; Balžekas, J. Evaluation of Antioxidant Activity and Flavonoid Composition in Differently Preserved Bee Products. Czech. J. Food Sci. 2016, 34, 133–142. [Google Scholar] [CrossRef]
- Sobral, F.; Calhelha, R.C.; Barros, L.; Dueñas, M.; Tomás, A.; Santos-Buelga, C.; Ferreira, I.C. Flavonoid composition and antitumor activity of bee bread collected in northeast Portugal. Molecules 2017, 22, 248. [Google Scholar] [CrossRef] [PubMed]
- Baltrušaityte, V.; Venskutonis, P.R.; Čeksteryte, V. Radical Scavenging Activity of Different Floral Origin Honey and Beebread Phenolic Extracts. Food Chem. 2007, 101, 502–514. [Google Scholar] [CrossRef]
- Tomás, A.; Falcão, S.I.; Russo-Almeida, P.; Vilas-Boas, M. Potentialities of Beebread as a Food Supplement and Source of Nutraceuticals: Botanical Origin, Nutritional Composition and Antioxidant Activity. J. Apic. Res. 2017, 56, 219–230. [Google Scholar] [CrossRef]
- Abouda, Z.; Zerdani, I.; Kalalou, I.; Faid, M.; Ahami, M.T. The antibacterial activity of Moroccan bee bread and bee-pollen (fresh and dried) against pathogenic bacteria. Res. J. Microbiol. 2011, 6, 376–384. [Google Scholar] [CrossRef]
- Bakour, M.; Al-Waili, N.S.; El Menyiy, N.; Imtara, H.; Figuira, A.C.; Al-Waili, T.; Lyoussi, B. Antioxidant activity and protective effect of bee bread (honey and pollen) in aluminum-induced anemia, elevation of inflammatory makers and hepato-renal toxicity. J. Food Sci. Technol. 2017, 54, 4205–4212. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.G.; Markham, K.R.; Mitchell, K.A.; da Cunha, P. An Approach to the Characterization of Bee Pollens via Their Flavonoid/Phenolic Profiles. Phytochem. Anal. 1997, 8, 181–185. [Google Scholar] [CrossRef]
- Campos, M.G.; Bogdanov, S.; de Almeida-Muradian, L.B.; Szczesna, T.; Mancebo, Y.; Frigerio, C.; Ferreira, F. Pollen composition and standardisation of analytical methods. J. Apic. Res. 2008, 47, 154–161. [Google Scholar] [CrossRef]
- Stanciu, S.; Mărghitaş, L.A.; Dezmirean, D. Examination of antioxidant capacity of beebread extracts by different complementary assays. Bull. USAMV-CN 2007, 63, 64. [Google Scholar]
- Serra Bonvehı, J.; Escola, J.R. Nutrient Composition and Microbiological Quality of Honeybee-Collected Pollen in Spain. J. Agric. Food Chem. 1997, 45, 725–732. [Google Scholar] [CrossRef]
- Szczesna, T. Honeybee-Collected Pollen. J. Apic. Sci. 2007, 51, 15–22. [Google Scholar]
- Qian, W.L.; Khan, Z.; Watson, D.G.; Fearnley, J. Analysis of sugars in bee pollen and propolis by ligand exchange chromatography in combination with pulsed amperometric detection and mass spectrometry. J. Food Compos. Anal. 2008, 21, 78–83. [Google Scholar] [CrossRef]
- Campos, M.G.; Webby, R.F.; Markham, K.R.; Mitchell, K.A.; da Cunha, A.P. Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J. Agric. Food Chem. 2003, 51, 742–745. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, C.M.Z.; Bermudez, J.C.S.; de Cuenca, M.C.Q. Valorization alternatives of Colombian bee-pollen for its use as food resource—A structured review. Vitae 2014, 21, 237–247. [Google Scholar]
- Čeksterytė, V.; Račys, J.; Kaškonienė, V.; Venskutonis, P.R. Fatty Acid Composition in Beebread. Biologija 2008, 54, 253–257. [Google Scholar] [CrossRef]
- Isidorov, V.A.; Isidorova, A.G.; Sczczepaniak, L.; Czyzewska, U. Gas Chromatographic-Mass Spectrometric Investigation of the Chemical Composition of Beebread. Food Chem. 2009, 115, 1056–1063. [Google Scholar] [CrossRef]
- Araneda Durán, X.; Quezada Mardones, I.; Martínez Gutiérrez, M.; Morales Ulloa, D. Polifenoles Totales En Pan de Abeja (Apis mellifera L.) de Colmenas de la Región de la Araucanía. Idesia 2014, 32, 107–111. [Google Scholar] [CrossRef]
- Mărghitaş, L.A.; Stanciu, O.G.; Dezmirean, D.S.; Bobiş, O.; Popescu, O.; Bogdanov, S.; Campos, M.G. In vitro antioxidant capacity of honeybee-collected pollen of selected floral origin harvested from Romania. Food Chem. 2009, 115, 878–883. [Google Scholar] [CrossRef]
- Carpes, S.T.; Begnini, R.; Alencar, S.M.D.; Masson, M.L. Study of preparations of bee pollen extracts, antioxidant and antibacterial activity. Ciênc. Agrotecnol. 2007, 31, 1818–1825. [Google Scholar] [CrossRef]
- Feás, X.; Vázquez-Tato, M.P.; Estevinho, L.; Seijas, J.A.; Iglesias, A. Organic bee pollen: Botanical origin, nutritional value, bioactive compounds, antioxidant activity and microbiological quality. Molecules 2012, 17, 8359–8377. [Google Scholar] [CrossRef] [PubMed]
- Cocan, O.; Marghitas, L.A.; Dezmirean, D. Total Polyphenols, Flavonoids and Radical Scavenging Activity of Beepollen and Bee Bread Collected from Transylvania Area. Bull. UASVM Anim. Sci. Biotechnol. 2009, 62, 149–152. [Google Scholar]
- Campos, M.G.; Markham, K.R. Structure Information from HPLC and On-Line Measured Absorption Spectra: Flavones, Flavonols and Phenolic Acids; Impresora da Universidade de Coimbra: Coimbra, Portugal, 2007; ISBN 978-989-8074-05-8. [Google Scholar]
- Wang, R.D.; Su, G.H.; Wang, L.; Xia, Q.; Liu, R.; Lu, Q.; Zhang, J.L. Identification and mechanism of effective components from rape (Brassica napus L.) bee pollen on serum uric acid level and xanthine oxidase activity. J. Funct. Foods 2018, 47, 241–251. [Google Scholar] [CrossRef]
- Campos, M.G.; Webby, R.F.; Markham, K.R. The Unique Occurrence of the Flavone Aglycone Tricetin in Myrtaceae Pollen. Z. Naturforschung Sect. C 2002, 57, 944–946. [Google Scholar] [CrossRef]
- Markham, K.R.; Campos, M.G. 7- and 8-O-Methylherbacetin-3-O-Sophorosides from Bee Pollens and Some Structure. Phytochemistry 1996, 43, 763–767. [Google Scholar] [CrossRef]
- Negri, G.; Barreto, L.M.R.C.; Sper, F.L.; Carvalho, C.D.; Campos, M.G. Phytochemical analysis and botanical origin of Apis mellifera bee pollen from the municipality of Canavieiras, Bahia State, Brazil. Braz. J. Food Technol. 2018, 21. [Google Scholar] [CrossRef]
- Silva, F.A.; Borges, F.; Guimarães, C.; Lima, J.L.; Matos, C.; Reis, S. Phenolic acids and derivatives: Studies on the relationship among structure, radical scavenging activity, and physicochemical parameters. J. Agric. Food Chem. 2000, 48, 2122–2126. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; Tomás-Lorente, F.; Ferreres, F.; Garcia-Viguera, C. Flavonoids as biochemical markers of the plant origin of bee pollen. J. Sci. Food Agric. 1989, 47, 337–340. [Google Scholar] [CrossRef]
- Harborne, J.B. Methods of plant analysis. In Phytochemical Methods; Springer: Berlin, Germany, 1984; pp. 1–36. [Google Scholar]
- Tavdidishvili, D.; Khutsidze, T.P.; Khakadze, M.; Vanidze, M.; Kalandia, A. Flavonoids in Georgian bee bread and bee pollen. J. Chem. Chem. Eng. 2014, 8, 676–681. [Google Scholar]
- Corby-Harris, V.; Snyder, L.; Meador, C.; Ayotte, T. Honey bee (Apis mellifera) nurses do not consume pollens based on their nutritional quality. PLoS ONE 2018, 13, e0191050. [Google Scholar] [CrossRef] [PubMed]
- Baum, K.A.; Rubink, W.L.; Coulson, R.N.; Bryant, V.M. Pollen selection by feral honey bee (Hymenoptera: Apidae) colonies in a coastal prairie landscape. Environ. Entomol. 2004, 33, 727–739. [Google Scholar] [CrossRef]
- Carpes, S.T.; Mourão, G.B.; De Alencar, S.M.; Masson, M.L. Chemical composition and free radical scavenging activity of Apis mellifera bee pollen from Southern Brazil. Braz. J. Food Technol. 2009, 12, 220–229. [Google Scholar] [CrossRef]
- Degrandi-Hoffman, G.; Eckholm, B.; Huang, M. Methods for Comparing Nutrients in Beebread Made by Africanized and European Honey Bees and the Effects on Hemolymph Protein Titers. JoVE 2015, 97, 52448. [Google Scholar] [CrossRef] [PubMed]
- Erel, O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin. Biochem. 2004, 37, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of melissopalynology. Bee World 1978, 59, 139–157. [Google Scholar] [CrossRef]
- Almeida-Muradian, L.B.; Pamplona, L.C.; Coimbra, S.; Barth, O.M. Chemical composition and botanical evaluation of dried bee pollen pellets. J. Food Compos. Anal. 2005, 18, 105–111. [Google Scholar] [CrossRef]
- Bonta, V.; Marghitas, L.A.; Stanciu, O.; Laslo, L.; Dezmirean, D.; Bobis, O. High-performance liquid chromatographic analysis of sugars in Transylvanian honeydew honey. Bull. UASVM Anim. Sci. Biotechnol. 2008, 65, 229–232. [Google Scholar]
- Attard, E. A rapid microtitre plate Folin-Ciocalteu method for the assessment of polyphenols. Open Life Sci. 2013, 8, 48–53. [Google Scholar] [CrossRef]
- Dezmirean, D.S.; Mărghitaş, L.A.; Chirilă, F.; Copaciu, F.; Simonca, V.; Bobiş, O.; Erler, S. Influence of Geographic Origin, Plant Source and Polyphenolic Substances on Antimicrobial Properties of Propolis against Human and Honey Bee Pathogens. J. Apic. Res. 2017, 56, 588–597. [Google Scholar] [CrossRef]
- Almaraz-Abarca, N.; Campos, M.G.; Ávila-Reyes, J.A.; Naranjo-Jiménez, N.; Herrera-Corral, J.; González-Valdez, L.S. Variability of antioxidant activity among honeybee-collected pollen of different botanical origin. Interciencia 2004, 29, 574–578. [Google Scholar] [CrossRef]
Sample Availability: BB samples are available from the authors. |

| Sample | Floral Composition (Botanical Name, %) |
|---|---|
| BB Romania—July 2016 | Brassicaceae—Brassica sp. (97%), Poaceae (3%) |
| BB India—June 2016 | Brassicaceae—Brassica sp. (98%), 2% Unknown pollen |
| BB India—June 2016 | Myrtaceae—Eucalyptus sp. (91%), Rutaceae—Citrus sp. (7%), Asteraceae (2%) |
| BB Romania—June 2016 | Fabaceae—Trifolium (34.6%), Tiliaceae—Tilia sp. (22%), Fabaceae (13%), Rosaceae (7.5%), Plantaginaceae—Plantago (5%), Fabaceae—Lotus (7.5%), Asteraceae—Taraxacum officinale (3%), Asteraceae (3%), Lamiaceae (2.9%), Asteraceae—Centaurea montana (1.5%) |
| BB Romania—April 2016 | Salicaceae—Salix sp. (61%), Rosaceae—Prunus sp. (26%), Fagaceae—Quercus sp. (13%) |
| Sample | Fructose (%) | Glucose (%) | Sucrose (%) | Turanose (%) | Maltose (%) | F/G |
|---|---|---|---|---|---|---|
| BB Romania—July 2016 | 19.32 ± 0.01 | 13.19 ± 0.01 | nd | 0.66 ± 0.01 | 0.89 ± 0.01 | 1.46 |
| BB India—June 2016 | 18.79 ± 0.06 | 12.66 ± 0.04 | nd | 0.65 ± 0.01 | 0.82 ± 0.01 | 1.48 |
| BB India—June 2016 | 19.58 ± 0.03 | 15.13 ± 0.02 | nd | 0.70 ± 0.02 | 1.00 ± 0.01 | 1.29 |
| BB Romania—June 2016 | 13.97 ± 0.05 | 6.40 ± 0.010 | nd | 0.56 ± 0.02 | 0.82 ± 0.02 | 2.18 |
| BB Romania—April 2016 | 19.29 ± 0.03 | 10.07 ± 0.02 | nd | 0.87 ± 0.01 | 0.91 ± 0.02 | 1.92 |
| Sample | Total Polyphenols (mg GAE/g) | Total Flavonoids (mg Qe/g) |
|---|---|---|
| BB Romania—July 2016 | 6.30 ± 0.11 | 29.33 ± 0.34 |
| BB India—June 2016 | 6.41 ± 0.12 | 30.23± 0.06 |
| BB India—June 2016 | 5.67 ± 0.09 | 34.46 ± 0.09 |
| BB Romania—June 2016 | 7.57 ± 0.16 | 23.64 ± 0.1 |
| BB Romania—April 2016 | 12.83 ± 0.08 | 36.19 ± 0.05 |
| Sample | Name of the Substance | Time of Retention | Bee Bread mg/g |
|---|---|---|---|
| BB Romania, 2016, Brassica sp. | Kaempferol-3-O-glycoside | 35.83 | 2.68 |
| Kaempferol-3-O-glycoside | 37.64 | 2.33 | |
| Hydroxycinnamic acid derivative 1 | 42.11 | 0.04 | |
| Hydroxycinnamic acid derivative 2 | 47.61 | 0.80 | |
| Hydroxycinnamic acid derivative 3 | 48.69 | 5.59 | |
| Hydroxycinnamic acid derivative 4 | 50.99 | 1.77 | |
| Hydroxycinnamic acid derivative 5 | 54.82 | 0.10 | |
| BB India, 2016, Brassica sp. | Kaempferol-3-O-glycoside | 35.83 | 2.57 |
| Kaempferol-3-O-glycoside | 37.64 | 2.21 | |
| Hydroxycinnamic acid derivative 1 | 42.19 | 0.03 | |
| Hydroxycinnamic acid derivative 2 | 47.74 | 0.58 | |
| Hydroxycinnamic acid derivative 3 | 48.72 | 5.61 | |
| Hydroxycinnamic acid derivative 4 | 51.08 | 1.62 | |
| Hydroxycinnamic acid derivative 5 | 54.94 | 0.08 | |
| BB India, 2016, Eucalyptus sp. | Quercetin-3-O-sophoroside | 35.61 | 2.40 |
| Kaempferol-3-O-glycoside | 37.42 | 0.86 | |
| Kaempferol-3-O-derivative (probably with a Hydroxycinnamic acids derivative in C3) | 38.55 | 0.35 | |
| Myrcetin | 41.3 | 0.44 | |
| Trycetin | 43.18 | 2.33 | |
| Luteolin | 44.35 | 6.98 | |
| Hydroxycinnamic acid derivative 1 | 50.84 | 2.00 | |
| Hydroxycinnamic acid derivative 2 | 53.77 | 3.11 | |
| Hydroxycinnamic acid derivative 3 | 54.44 | 9.35 | |
| BB Romania, April 2016 | Herbacetin-3-O-glycoside | 36.27 | 4.30 |
| Quercetin-3-O-glycoside | 36.98 | 2.98 | |
| Kaempferol-3-O-glycoside | 38.38 | 2.85 | |
| Herbacetin-3-O-glycoside | 40.36 | 0.43 | |
| Herbacetin-3-O-glycoside | 42.17 | 0.27 | |
| Hydroxycinnamic acid derivative 1 | 43.87 | 0.25 | |
| Hydroxycinnamic acid derivative 2 | 45.17 | 1.31 | |
| Hydroxycinnamic acid derivative 3 | 47.10 | 0.80 | |
| Hydroxycinnamic acid derivative 4 | 47.21 | 1.18 | |
| Hydroxycinnamic acid derivative 5 | 49.61 | 1.94 | |
| Hydroxycinnamic acid derivative 6 | 50.75 | 1.74 | |
| Hydroxycinnamic acid derivative 7 | 52.42 | 4.64 | |
| Hydroxycinnamic acid derivative 8 | 53.68 | 0.43 | |
| BB Romania, June 2016 | Kaempferol-3-O-glycoside | 35.73 | 0.90 |
| Quercetin-3-O-glycoside | 36.48 | 1.08 | |
| Quercetin-3-O-glycoside | 38.13 | 0.81 | |
| Isoramnethin-3-O-glycoside | 40.81 | 0.91 | |
| Hydroxycinnamic acid derivative 1 | 43.48 | 0.61 | |
| Hydroxycinnamic acid derivative 2 | 44.85 | 1.72 | |
| Hydroxycinnamic acid derivative 3 | 45.62 | 0.28 | |
| Hydroxycinnamic acid derivative 4 | 47.10 | 0.27 | |
| Hydroxycinnamic acid derivative 5 | 47.57 | 0.24 | |
| Hydroxycinnamic acid derivative 6 | 49.24 | 0.17 | |
| Hydroxycinnamic acid derivative 7 | 50.52 | 0.26 | |
| Hydroxycinnamic acid derivative 8 | 52.11 | 0.53 | |
| Hydroxycinnamic acid derivative 9 | 53.16 | 0.33 | |
| Hydroxycinnamic acid derivative 10 | 54.24 | 0.49 | |
| Hydroxycinnamic acid derivative 11 | 55.10 | 0.44 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urcan, A.C.; Criste, A.D.; Dezmirean, D.S.; Mărgăoan, R.; Caeiro, A.; Graça Campos, M. Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania. Molecules 2018, 23, 2491. https://doi.org/10.3390/molecules23102491
Urcan AC, Criste AD, Dezmirean DS, Mărgăoan R, Caeiro A, Graça Campos M. Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania. Molecules. 2018; 23(10):2491. https://doi.org/10.3390/molecules23102491
Chicago/Turabian StyleUrcan, Adriana Cristina, Adriana Dalila Criste, Daniel Severus Dezmirean, Rodica Mărgăoan, André Caeiro, and Maria Graça Campos. 2018. "Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania" Molecules 23, no. 10: 2491. https://doi.org/10.3390/molecules23102491
APA StyleUrcan, A. C., Criste, A. D., Dezmirean, D. S., Mărgăoan, R., Caeiro, A., & Graça Campos, M. (2018). Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania. Molecules, 23(10), 2491. https://doi.org/10.3390/molecules23102491








