Phenolics Isolated from Aframomum meleguta Enhance Proliferation and Ossification Markers in Bone Cells
Abstract
1. Introduction
2. Results
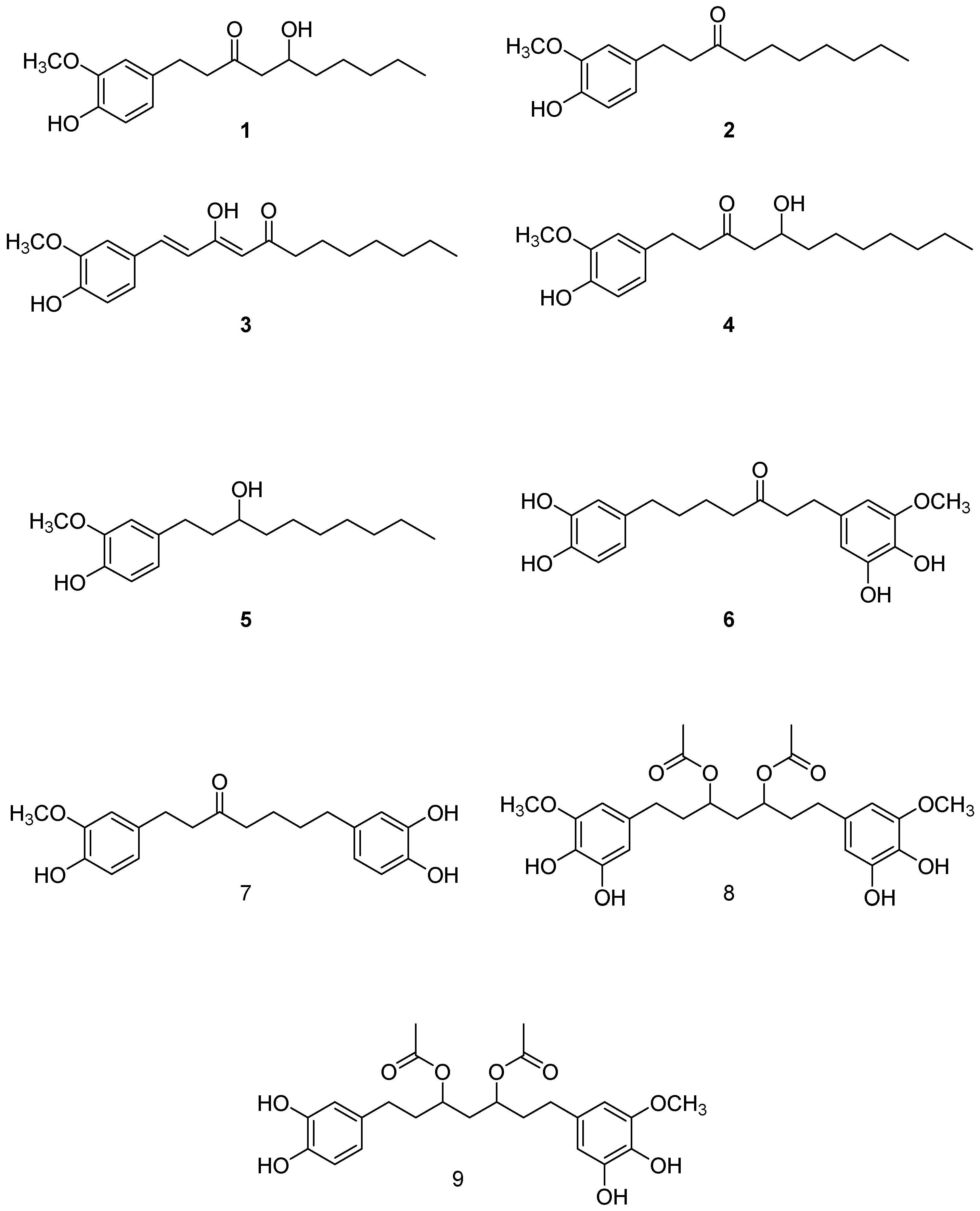
2.1. Isolation and Structural Identification of Major Phenolics from A. melegueta
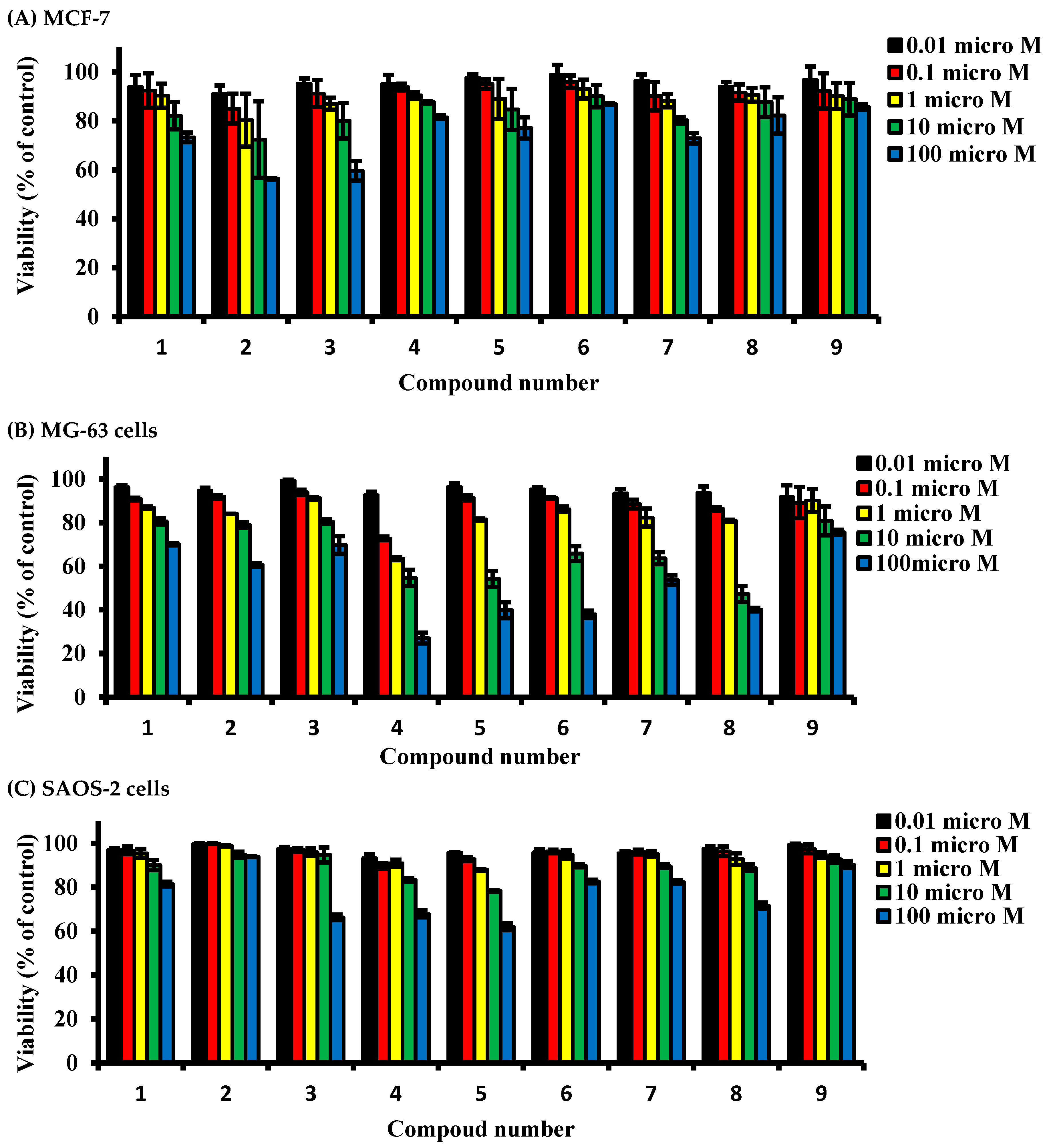
2.2. Determining Cell Killing Effects and Subcytotoxic Concentration of Compounds Isolated from A. meleguta in MCF-7, MG-63, and SAOS-2 Cell Lines
2.3. Determining the Proliferative Properties of Compounds Isolated from A. meleguta in MCF-7, MG-63, and SAOS-2 Cell Lines
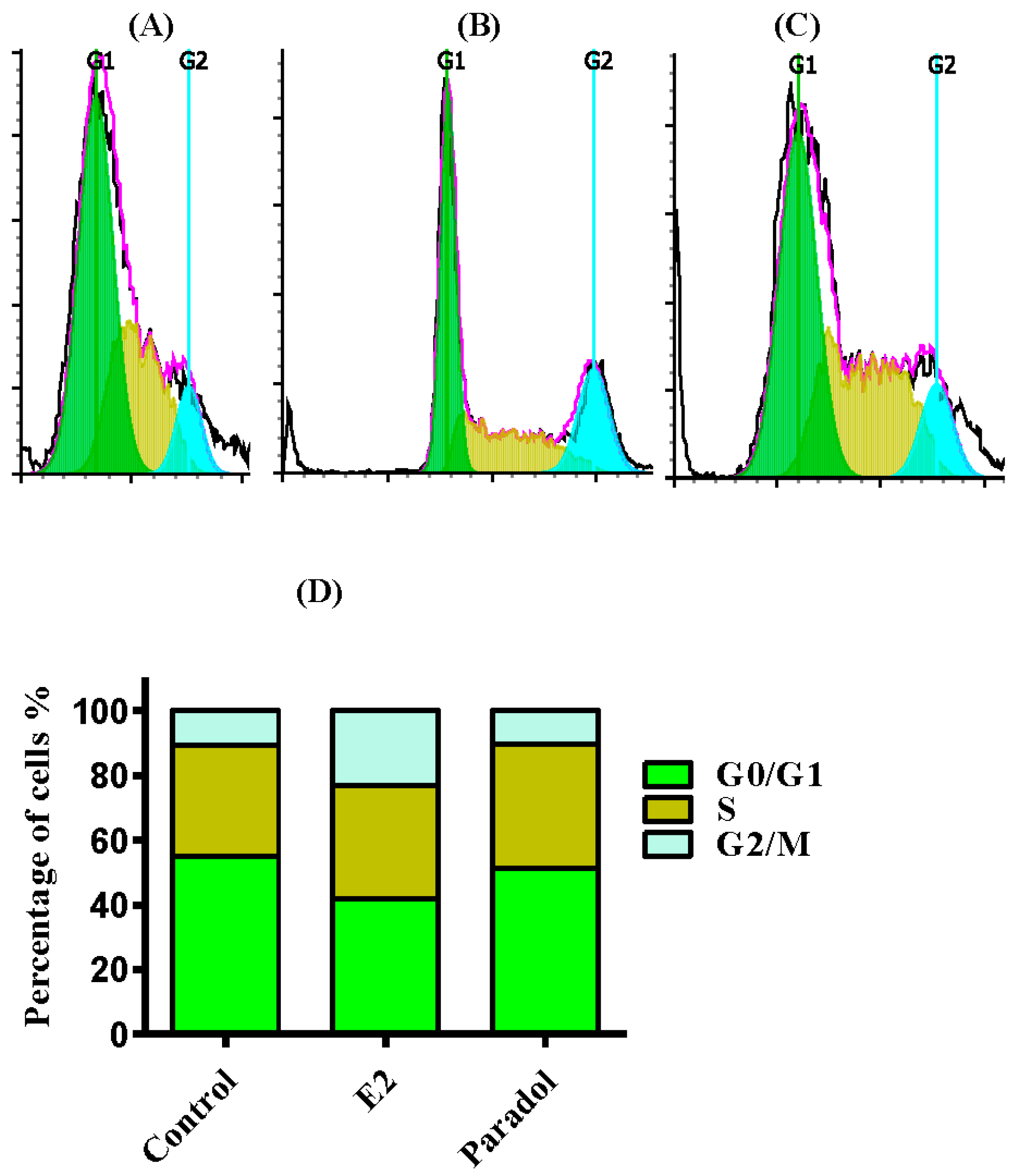
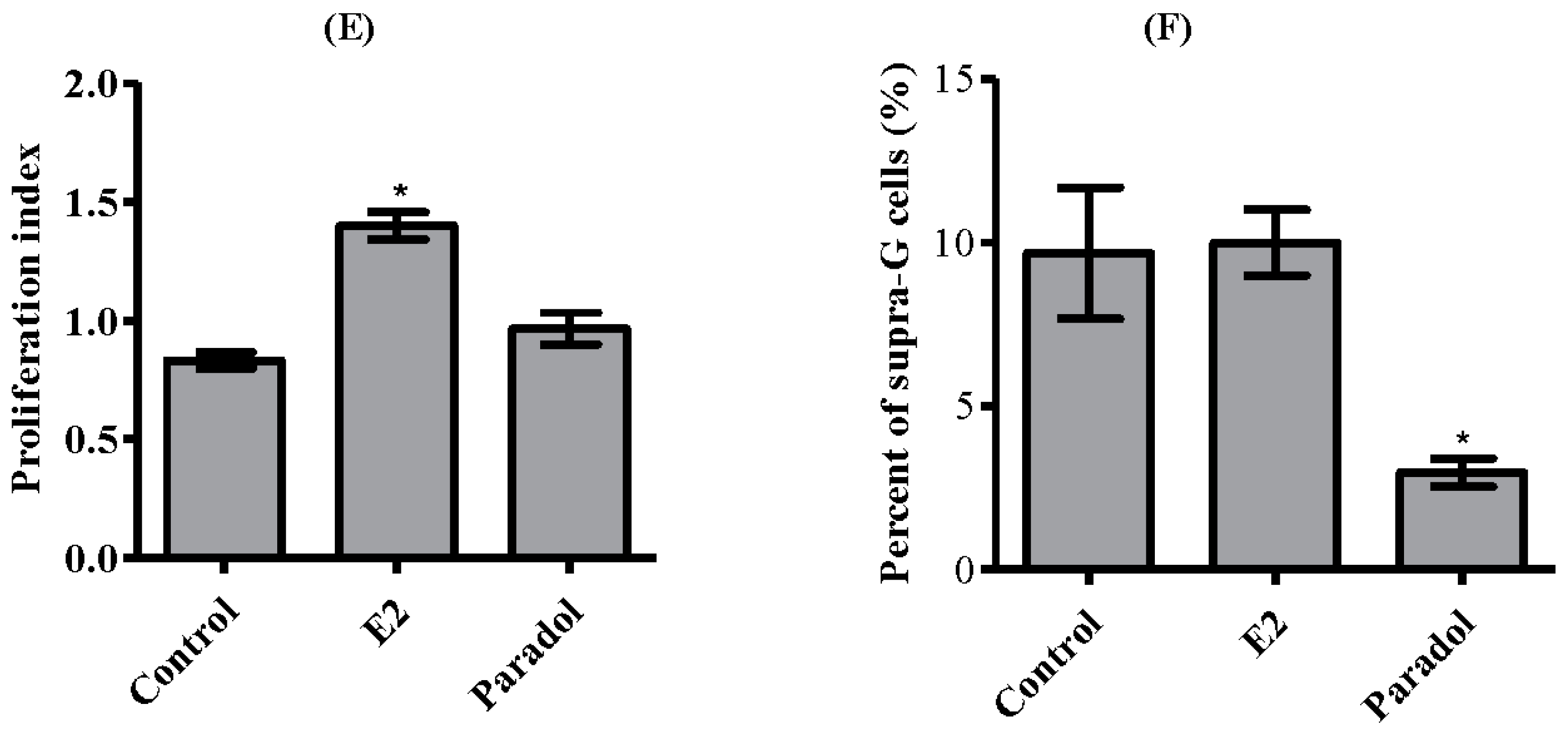
2.4. Determining the Effect of Paradol on Cell Cycle Distribution
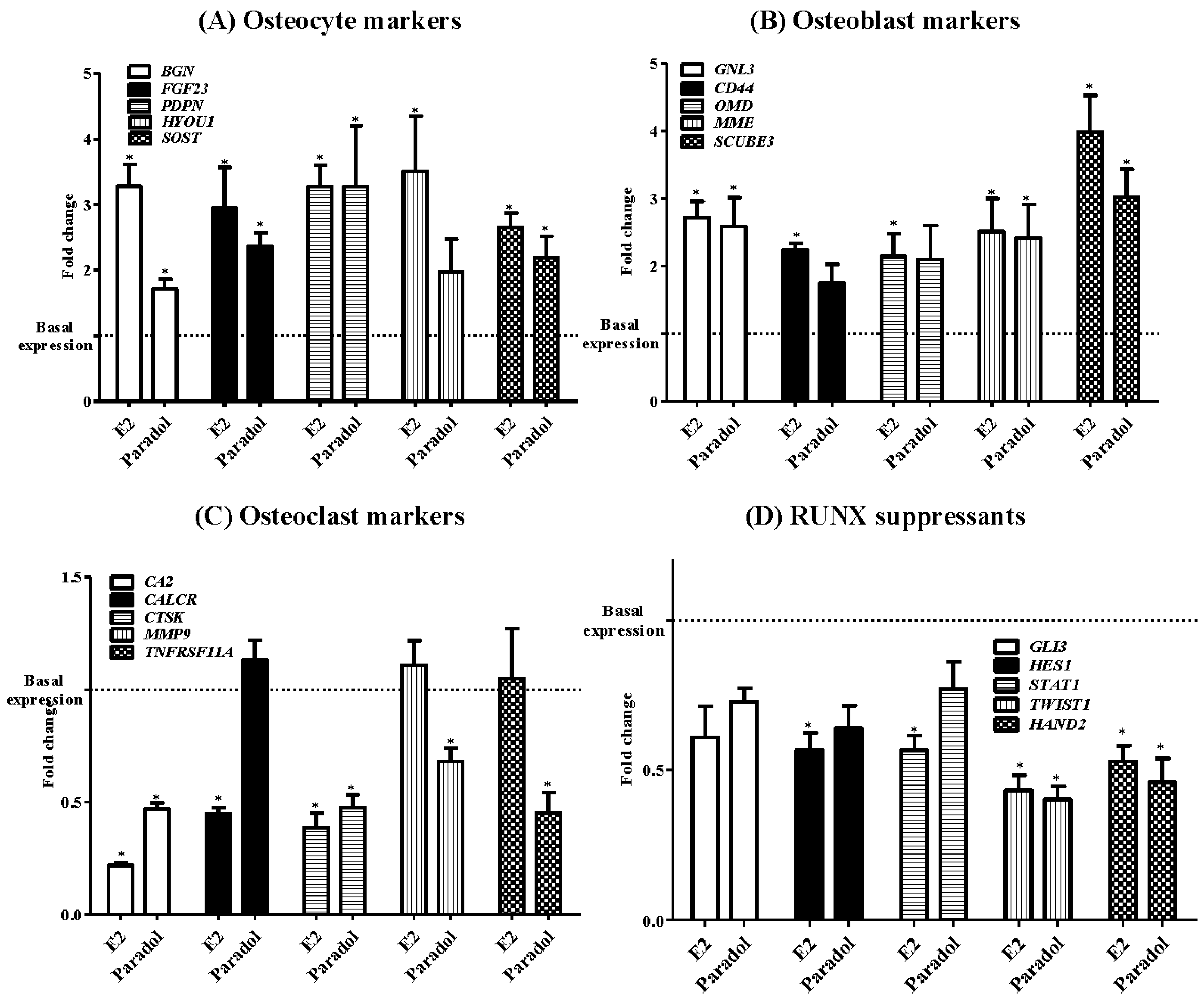
2.5. Assessment of Osteogenic Gene Expression
2.6. Assessment of Antiosteoporotic Biochemical Markers
3. Discussion
4. Materials and Methods
4.1. General
4.2. Plant Material
4.3. Extraction and Isolation
4.4. Chemicals and Media
4.5. Cell Culture
4.6. Cytotoxicity Assessment
4.7. Proliferation Assay (Doubling Time Determination)
4.8. Analysis of Cell Cycle Distribution
4.9. Osteogenic Gene Array Analysis
4.10. Assessment of Biochemical Osteoporosis Marker
4.11. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Reginster, J.-Y.; Burlet, N. Osteoporosis: A still increasing prevalence. Bone 2006, 38, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Sornay-Rendu, E.; Munoz, F.; Garnero, P.; Duboeuf, F.; Delmas, P.D. Identification of osteopenic women at high risk of fracture: The OFELY study. J. Bone Miner. Res. 2005, 20, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.M.; Correa, P.H.S. Bone quality and osteoporosis therapy. Arq. Bras. Endocrinol. Metabol. 2010, 54, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Randell, A.; Sambrook, P.N.; Nguyen, T.V.; Lapsley, H.; Jones, G.; Kelly, P.J.; Eisman, J.A. Direct clinical and welfare costs of osteoporotic fractures in elderly men and women. Osteoporos. Int. 1995, 5, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.S.T. Bone health status of postmenopausal Chinese women. Hong Kong Med. J. 2015, 21, 536–541. [Google Scholar] [PubMed]
- Lufkin, E.G.; Sarkar, S.; Kulkarni, P.M.; Ciaccia, A.V.; Siddhanti, S.; Stock, J.; Plouffe, L. Antiresorptive treatment of postmenopausal osteoporosis: Review of randomized clinical studies and rationale for the Evista alendronate comparison (EVA) trial. Curr. Med. Res. Opin. 2004, 20, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Brass, L.M. Estrogens and Stroke: Use of Oral Contraceptives and postmenopausal use of estrogen: Current recommendations. Curr. Treat. Opt. Neurol. 2004, 6, 459–467. [Google Scholar] [CrossRef]
- Strampel, W.; Emkey, R.; Civitelli, R. Safety considerations with bisphosphonates for the treatment of osteoporosis. Drug Saf. 2007, 30, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Yardley, D.A.; Yardley, D. Pharmacologic management of bone-related complications and bone metastases in postmenopausal women with hormone receptor-positive breast cancer. Breast Cancer 2016, 8, 73–82. [Google Scholar] [PubMed]
- Dean, M.; Murphy, B.T.; Burdette, J.E. Phytosteroids beyond estrogens: Regulators of reproductive and endocrine function in natural products. Mol. Cell. Endocrinol. 2017, 442, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Banu, J.; Varela, E.; Fernandes, G. Alternative therapies for the prevention and treatment of osteoporosis. Nutr. Rev. 2012, 70, 22–40. [Google Scholar] [CrossRef] [PubMed]
- Bharti, A.C.; Takada, Y.; Aggarwal, B.B. Curcumin (diferuloylmethane) inhibits receptor activator of NF-kappa B ligand-induced NF-kappa B activation in osteoclast precursors and suppresses osteoclastogenesis. J. Immunol. 2004, 172, 5940–5947. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.-W.; Wang, T.-H.; Yan, P.-P.; Chu, L.-W.; Yu, J.; Gao, Z.-D.; Li, Y.-Z.; Guo, B.-L. Curcumin improves bone microarchitecture and enhances mineral density in APP/PS1 transgenic mice. Phytomedicine 2011, 18, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.M. Effect of ginger tea on the fetal development of Sprague-Dawley rats. Reprod. Toxicol. 2000, 14, 507–512. [Google Scholar] [CrossRef]
- Ito, S.; Ohmi, A.; Sakamiya, A.; Yano, T.; Okumura, K.; Nishimura, N.; Kagontani, K. Ginger hexane extract suppresses RANKL-induced osteoclast differentiation. Biosci. Biotechnol. Biochem. 2016, 80, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.Z.; Yang, X.; Bi, Z.G. The effects of 6-gingerol on proliferation, differentiation, and maturation of osteoblast-like MG-63 cells. Braz. J. Med. Biol. Res. 2015, 48, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Iwu, M. Handbook of African Medicinal Plants, 2nd ed.; Iwu, M., Ed.; CRC Press, Taylor and Francis Group: New York, NY, USA, 2014. [Google Scholar]
- El-Halawany, A.M.; El Dine, R.S.; Chung, M.H.; Nishihara, T.; Hattori, M. Screening for estrogenic and antiestrogenic activities of plants growing in Egypt and Thailand. Pharmacogn. Res. 2011, 3, 107–113. [Google Scholar]
- Mohammed, A.; Koorbanally, N.A.; Islam, M.S. Phytochemistry, antioxidant activity and inhibition of key enzymes linked to type 2 diabetes by various parts of Aframomum melegueta in vitro. Acta Pol. Pharm. 2016, 73, 403–417. [Google Scholar] [PubMed]
- Adefegha, S.A.; Olasehinde, T.A.; Oboh, G. Essential oil composition, antioxidant, antidiabetic and antihypertensive properties of two Afromomum species. J. Oleo Sci. 2017, 66, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Ogbole, O.O.; Ajaiyeoba, E.O. Traditional management of tuberculosis in Ogun State of Nigeria: The practice and ethnobotanical survey. Afr. J. Tradit. Complement. Altern. Med. 2009, 7, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Gbolade, A. Ethnobotanical study of plants used in treating hypertension in Edo State of Nigeria. J. Ethnopharmacol. 2012, 144, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhang, S.; Duan, W.; Wang, W. An enantioselective synthesis of (+)-(S)-[n]-gingerols via the l-proline-catalyzed aldol reaction. Bioorg. Med. Chem. Lett. 2009, 19, 3909–3911. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Timmermann, B.N.; Gang, D.R. Characterization and identification of diarylheptanoids in ginger (Zingiber officinale Rosc.) using high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2007, 1, 509–518. [Google Scholar] [CrossRef] [PubMed]
- El-Halawany, A.M.; El Dine, R.S.; El Sayed, N.S.; Hattori, M. Protective effect of Aframomum melegueta phenolics against CCl4-induced rat hepatocytes damage; role of Apoptosis and pro-inflammatory cytokines inhibition. Sci. Rep. 2014, 4, 5880. [Google Scholar] [CrossRef] [PubMed]
- Tackie, A.; Dwuma-Badu, D.; Ayim, J.; Dabra, T.; Knapp, J.; Slatkin, D.; Schiff, P.L., Jr. Hydroxyphenylalkanones from Amomum melegueta. Phytochemistry 1975, 14, 853–854. [Google Scholar] [CrossRef]
- El-Halawany, A.M.; Hattori, M. Anti-oestrogenic diarylheptanoids from Aframomum melegueta with in silico oestrogen receptor alpha binding conformation similar to enterodiol and enterolactone. Food Chem. 2012, 134, 219–226. [Google Scholar] [CrossRef]
- Kling, J.M.; Clarke, B.L.; Sandhu, N.P. Osteoporosis prevention, screening, and treatment: A review. J. Women’s Heal. 2014, 23, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Papapetrou, P.D. Bisphosphonate-associated adverse events. Hormones (Athens) 2009, 8, 96–110. [Google Scholar] [CrossRef] [PubMed]
- Sharan, K.; Siddiqui, J.A.; Swarnkar, G.; Maurya, R.; Chattopadhyay, N. Role of phytochemicals in the prevention of menopausal bone loss: Evidence from in vitro and in vivo, human interventional and pharma-cokinetic studies. Curr. Med. Chem. 2009, 16, 1138–1157. [Google Scholar] [CrossRef] [PubMed]
- Mosli, H.A.; Tolba, M.F.; Al-Abd, A.M.; Abdel-Naim, A.B. Catechol estrogens induce proliferation and malignant transformation in prostate epithelial cells. Toxicol. Lett. 2013, 220, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Soto, A.M.; Sonnenschein, C. The role of estrogens on the proliferation of human breast tumor cells (MCF-7). J. Steroid Biochem. 1985, 23, 87–94. [Google Scholar] [CrossRef]
- Weng, C.-J.; Wu, C.-F.; Huang, H.-W.; Ho, C.-T.; Yen, G.-C. Anti-invasion effects of 6-shogaol and 6-gingerol, two active components in ginger, on human hepatocarcinoma cells. Mol. Nutr. Food Res. 2010, 54, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- Shukla, Y.; Singh, M. Cancer preventive properties of ginger: A brief review. Food Chem. Toxicol. 2007, 45, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Mitchison, J.M.; Carter, B.L. Cell cycle analysis. Methods Cell Biol. 1975, 11, 201–219. [Google Scholar] [PubMed]
- Takahashi, M. Theoretical basis for cell cycle analysis: II. Further studies on labelled mitosis wave method. J. Theor. Biol. 1968, 18, 195–209. [Google Scholar] [CrossRef]
- Kanis, J.A.; Glüer, C.C. An update on the diagnosis and assessment of osteoporosis with densitometry. Committee of Scientific Advisors, International Osteoporosis Foundation. Osteoporos. Int. 2000, 11, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Rachner, T.D.; Khosla, S.; Hofbauer, L.C. Osteoporosis: Now and the future. Lancet 2011, 377, 1276–1287. [Google Scholar] [CrossRef]
- Schaffler, M.B.; Kennedy, O.D. Osteocyte signaling in bone. Curr. Osteoporos. Rep. 2012, 10, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Parisuthiman, D.; Mochida, Y.; Duarte, W.R.; Yamauchi, M. Biglycan modulates osteoblast differentiation and matrix mineralization. J. Bone Miner. Res. 2005, 20, 1878–1886. [Google Scholar] [CrossRef] [PubMed]
- Sapir-Koren, R.; Livshits, G. Osteocyte control of bone remodeling: Is sclerostin a key molecular coordinator of the balanced bone resorption-formation cycles? Osteoporos. Int. 2014, 25, 2685–2700. [Google Scholar] [CrossRef] [PubMed]
- Dayong Guo, D.; Bonewald, L.F. Advancing our understanding of osteocyte cell biology. Ther. Adv. Musculoskelet. Dis. 2009, 1, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ominsky, M.S.; Niu, Q.-T.; Sun, N.; Daugherty, B.; D’Agostin, D.; Kurahara, C.; Gao, Y.; Cao, J.; Gong, J.; et al. Targeted deletion of the sclerostin gene in mice results in increased bone formation and bone strength. J. Bone Miner. Res. 2008, 23, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Roles of Runx2 in skeletal development. Adv. Exp. Med. Biol. 2017, 962, 83–93. [Google Scholar] [PubMed]
- Abdallah, H.M.; Al-Abd, A.M.; Asaad, G.F.; Abdel-Naim, A.B.; El-halawany, A.M. Isolation of antiosteoporotic compounds from seeds of Sophora japonica. PLoS ONE 2014, 9, e98559. [Google Scholar] [CrossRef] [PubMed]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, RESEARCH0034. [Google Scholar] [CrossRef] [PubMed]
- Gerhardt, W.; Nielsen, M.L.; Nielsen, O.V.; Olsen, J.S.; Statland, B.E. Clinical evaluation of routine measurement of liver and bone alkaline phosphatases in human serum. Differential inhibition by l-phenylalanine and carbamide (urea) on the LKB 8600 reaction rate analyzer. Clin. Chim. Acta 1974, 53, 291–297. [Google Scholar] [CrossRef]
- Fu, J.Y.; Muller, D. Simple, rapid enzyme-linked immunosorbent assay (ELISA) for the determination of rat osteocalcin. Calcif. Tissue Int. 1999, 64, 229–233. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Naim, A.B.; Alghamdi, A.A.; Algandaby, M.M.; Al-Abbasi, F.A.; Al-Abd, A.M.; Abdallah, H.M.; El-Halawany, A.M.; Hattori, M. Phenolics Isolated from Aframomum meleguta Enhance Proliferation and Ossification Markers in Bone Cells. Molecules 2017, 22, 1467. https://doi.org/10.3390/molecules22091467
Abdel-Naim AB, Alghamdi AA, Algandaby MM, Al-Abbasi FA, Al-Abd AM, Abdallah HM, El-Halawany AM, Hattori M. Phenolics Isolated from Aframomum meleguta Enhance Proliferation and Ossification Markers in Bone Cells. Molecules. 2017; 22(9):1467. https://doi.org/10.3390/molecules22091467
Chicago/Turabian StyleAbdel-Naim, Ashraf B., Abdullah A. Alghamdi, Mardi M. Algandaby, Fahad A. Al-Abbasi, Ahmed M. Al-Abd, Hossam M. Abdallah, Ali M. El-Halawany, and Masao Hattori. 2017. "Phenolics Isolated from Aframomum meleguta Enhance Proliferation and Ossification Markers in Bone Cells" Molecules 22, no. 9: 1467. https://doi.org/10.3390/molecules22091467
APA StyleAbdel-Naim, A. B., Alghamdi, A. A., Algandaby, M. M., Al-Abbasi, F. A., Al-Abd, A. M., Abdallah, H. M., El-Halawany, A. M., & Hattori, M. (2017). Phenolics Isolated from Aframomum meleguta Enhance Proliferation and Ossification Markers in Bone Cells. Molecules, 22(9), 1467. https://doi.org/10.3390/molecules22091467





