Chromatographic Separation of Vitamin E Enantiomers
Abstract
:1. Introduction
2. Liquid Chromatography
3. Gas Chromatography
4. Combined Systems
5. Applications
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on Dietary Reference Values for vitamin E as α-tocopherol. EFSA J. 2015, 13, 4149. [Google Scholar]
- Goh, S.H.; Choo, Y.M.; Ong, S.H. Minor constituents of palm oil. J. Am. Oil Chem. Soc. 1985, 62, 237–240. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Sundaram, C.; Prasad, S.; Kannappan, R. Tocotrienols, the vitamin E of the 21st century: Its potential against cancer and other chronic diseases. Biochem. Pharmacol. 2010, 80, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohe, R. Vitamin E and drug metabolism. Biochem. Biophys. Res. Commun. 2003, 305, 737–740. [Google Scholar] [CrossRef]
- Zingg, J.M. Vitamin E: An overview of major research directions. Mol. Asp. Med. 2007, 28, 400–422. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Rader, D.; Acuff, R.V.; Ramakrishnan, R.; Brewer, H.B.; Kayden, H.J. Vitamin E dose-response studies in humans with use of deuterated RRR-α-tocopherol. Am. J. Clin. Nutr. 1998, 68, 847–853. [Google Scholar] [PubMed]
- De Silva, L.; Chuah, L.H.; Meganathan, P.; Fu, J.Y. Tocotrienol and cancer metastasis. BioFactors 2016, 42, 149–162. [Google Scholar] [PubMed]
- Gopalan, Y.; Shuaib, I.L.; Magosso, E.; Ansari, M.A.; Abu Bakar, M.R.; Wong, J.W.; Khan, N.A.; Liong, W.C.; Sundram, K.; Ng, B.H.; et al. Clinical investigation of the protective effects of palm vitamin E tocotrienols on brain white matter. Stroke 2014, 45, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Kamal-Eldin, A.; Appelqvist, L.A. The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 1996, 31, 671–701. [Google Scholar] [CrossRef] [PubMed]
- Netscher, T. Stereoisomers of tocopherols—Syntheses and analytics. Chimia 1996, 50, 563–567. [Google Scholar]
- Netscher, T. Cross-metathesis: Efficient preparation of trialkyl-substituted isoprenoid olefins as key-intermediates for tocopherol synthesis. Curr. Top. Med. Chem. 2005, 5, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Malaisé, G.; Bonrath, W.; Breuninger, M.; Netscher, T. A new route to vitamin E key-intermediates by olefin cross-metathesis. Helv. Chim. Acta 2006, 89, 797–812. [Google Scholar] [CrossRef]
- Jensen, S.K.; Lauridsen, C. Alpha-tocopherol stereoisomers. Vitam Horm 2007, 76, 281–308. [Google Scholar] [PubMed]
- Weiser, H.; Vecchi, M. Stereoisomers of alpha-tocopheryl acetate. II. Biopotencies of all eight stereoisomers, individually or in mixtures, as determined by rat resorption-gestation tests. Int. J. Vitam. Nutr. Res. 1982, 52, 351–370. [Google Scholar] [PubMed]
- Kiyose, C.; Kaneko, K.; Muramatsu, R.; Ueda, T.; Igarashi, O. Simultaneous determination of RRR- and SRR-alpha-tocopherols and their quinones in rat plasma and tissues by using chiral high-performance liquid chromatography. Lipids 1999, 34, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; The National Academy Press: Washington, DC, USA, 2000; p. 529. [Google Scholar]
- Zhao, Y.; Monahan, F.J.; McNulty, B.A.; Brennan, L.; Gibney, M.J.; Gibney, E.R. alpha-Tocopherol Stereoisomers in Human Plasma Are Affected by the Level and Form of the Vitamin E Supplement Used. J. Nutr. 2015, 145, 2347–2354. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.W.; Bizzarro, F.T.; Parrish, D.R.; Saucy, G. Syntheses of (2R,4′R,8′R)-alpha-tocopherol and (2R,3′E,7′E)-alpha-tocotrienol. Helv. Chim. Acta 1976, 59, 290–306. [Google Scholar] [CrossRef] [PubMed]
- Couladouros, E.A.; Moutsos, V.I.; Lampropoulou, M.; Little, J.L.; Hyatt, J.A. A short and convenient chemical route to optically pure 2-methyl chromanmethanols. Total asymmetric synthesis of beta-, gamma-, and delta-tocotrienols. J. Org. Chem. 2007, 72, 6735–6741. [Google Scholar] [CrossRef] [PubMed]
- Abidi, S.L. Chromatographic analysis of tocol-derived lipid antioxidants. J. Chromatogr. A 2000, 881, 197–216. [Google Scholar] [CrossRef]
- De Leenheer, A.P.; Nelis, H.J.; Lambert, W.E.; Bauwens, R.M. Chromatography of fat-soluble vitamins in clinical chemistry. J. Chromatogr. 1988, 429, 3–58. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Itakura, Y.; Kunihiro, K. Analysis of the stereoisomers of alpha-tocopheryl acetate by HPLC. Iyakuhin Kenkyu 1984, 15, 536–540. [Google Scholar]
- Vecchi, M.; Walther, W.; Glinz, E.; Netscher, T.; Schmid, R.; Lalonde, M.; Walter, V. Chromatographische Trennung und quantitative Bestimmung aller acht Stereoisomeren von a-Tocopherol. Helv. Chim. Acta 1990, 73, 782–789. [Google Scholar] [CrossRef]
- Ueda, T.; Ichikawa, H.; Igarashi, O. Determination of alpha-tocopherol stereoisomers in biological specimens using chiral phase high-performance liquid chromatography. J. Nutr. Sci. Vitaminol. (Tokyo) 1993, 39, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Nitta, C.; Hayashi, K.; Ueda, T.; Igarashi, O. Distribution of α-Tocopherol Stereoisomers in Rats. Biosci. Biotechnol. Biochem. 1993, 57, 1406–1407. [Google Scholar] [CrossRef]
- Nitta-Kiyose, C.; Hayashi, K.; Ueda, T.; Igarashi, O. Distribution of α-Toc Stereoisomers in Rats. Biosci. Biotechnol. Biochem. 1994, 58, 2000–2003. [Google Scholar] [CrossRef]
- Kiyose, C.; Muramatsu, R.; Kameyama, Y.; Ueda, T.; Igarashi, O. Biodiscrimination of alpha-tocopherol stereoisomers in humans after oral administration. Am. J. Clin. Nutr. 1997, 65, 785–789. [Google Scholar] [PubMed]
- Nakamura, T.; Reicher, H.; Sattler, W. Comparison of RRR-alpha- and all-rac-alpha-tocopherol uptake by permanent rat skeletal muscle myoblasts (L6 cells): Effects of exogenous lipoprotein lipase. Lipids 1998, 33, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Klaczkow, G.; Anuszewska, E.L. Determination of the stereoisomers of racemic alpha-tocopherol in pharmaceutical preparations by high-performance liquid chromatography and gas chromatography. Acta Pol. Pharm. 2008, 65, 715–721. [Google Scholar] [PubMed]
- Amazan, D.; Cordero, G.; Lopez-Bote, C.J.; Lauridsen, C.; Rey, A.I. Effects of oral micellized natural vitamin E (d-alpha-tocopherol) v. synthetic vitamin E (dl-alpha-tocopherol) in feed on alpha-tocopherol levels, stereoisomer distribution, oxidative stress and the immune response in piglets. Animal 2014, 8, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Rey, A.I.; Lopez-Bote, C.J. Alpha-tocopherol stereoisomer analysis as discriminant method for distinguishing Iberian pig feed intake during the fattening phase. Food Chem. 2014, 142, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Lauridsen, C.; Jensen, S.K. Influence of supplementation of all-rac-alpha-tocopheryl acetate preweaning and vitamin C postweaning on alpha-tocopherol and immune responses of piglets. J. Anim. Sci. 2005, 83, 1274–1286. [Google Scholar] [CrossRef] [PubMed]
- Cortinas, L.; Barroeta, A.; Galobart, J.; Jensen, S.K. Distribution of alpha-tocopherol stereoisomers in liver and thigh of chickens. Br. J. Nutr. 2004, 92, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Meglia, G.E.; Jensen, S.K.; Lauridsen, C.; Persson Waller, K. Alpha-tocopherol concentration and stereoisomer composition in plasma and milk from dairy cows fed natural or synthetic vitamin E around calving. J. Dairy Res. 2006, 73, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Weiss, W.P.; Hogan, J.S.; Wyatt, D.J. Relative bioavailability of all-rac and RRR vitamin E based on neutrophil function and total alpha-tocopherol and isomer concentrations in periparturient dairy cows and their calves. J. Dairy Sci. 2009, 92, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Röhrle, F.T.; Moloney, A.P.; Black, A.; Osorio, M.T.; Sweeney, T.; Schmidt, O.; Monahan, F.J. α-Tocopherol stereoisomers in beef as an indicator of vitamin E supplementation in cattle diets. Food Chem. 2011, 124, 935–940. [Google Scholar] [CrossRef]
- Mazzini, F.; Betti, M.; Netscher, T.; Galli, F.; Salvadori, P. Configuration of the vitamin E analogue garcinoic acid extracted from Garcinia Kola seeds. Chirality 2009, 21, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Hsieh, K.H. Nanochitosan crosslinked with polyacrylamide as the chiral stationary phase for open-tubular capillary electrochromatography. Electrophoresis 2011, 32, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Drotleff, A.M.; Ternes, W. Determination of RS,E/Z-tocotrienols by HPLC. J. Chromatogr. A 2001, 909, 215–223. [Google Scholar] [CrossRef]
- Drotleff, A.M.; Ternes, W. Separation and characterization of cis-trans isomers of a-tocotrienol by HPLC using a permethylated b-cyclodextrin phase. Z. Lebensm. Unters. Forsch. A 1998, 206, 9–13. [Google Scholar] [CrossRef]
- Mayer, H.; Metzger, J.; Isler, O. The stereochemistry of natural gamma-tocotrienol (plastochromanol-3), plastochromanol-8 and plastochromenol-8. Helv. Chim. Acta 1967, 50, 1376–1393. [Google Scholar] [CrossRef] [PubMed]
- Slover, H.T.; Thompson, R.H. Chromatographic separation of the stereoisomers of α-tocopherol. Lipids 1981, 16, 268–275. [Google Scholar] [CrossRef]
- Cohen, N.; Scott, C.G.; Neukom, C.; Lopresti, R.J.; Weber, G.; Saucy, G. Total Synthesis of All Eight Stereoisomers of α-Tocopheryl Acetate. Determination of their diastereoisomeric and enantiomeric purity by gas chromatography. Helv. Chim. Acta 1981, 64, 1158–1173. [Google Scholar] [CrossRef]
- Weiser, H.; Vecchi, M. Stereoisomers of alpha-tocopheryl acetate—Characterization of the samples by physico-chemical methods and determination of biological activities in the rat resorption-gestation test. Int. J. Vitam. Nutr. Res. 1981, 51, 100–113. [Google Scholar] [PubMed]
- Piironen, V.I.; Liljeroos, A.I.; Koivistoinen, P.E. Transfer of a-Tocopherol Stereoisomers from Feeds to Eggs. J. Agric. Food Chem. 1991, 39, 99–101. [Google Scholar] [CrossRef]
- Riss, G.; Kormann, A.W.; Glinz, E.; Walther, W.; Ranalder, U.B. Separation of the eight stereoisomers of all-rac-alpha-tocopherol from tissues and plasma: Chiral phase high-performance liquid chromatography and capillary gas chromatography. Methods Enzymol. 1994, 234, 302–310. [Google Scholar] [PubMed]
- Weiser, H.; Riss, G.; Kormann, A.W. Biodiscrimination of the eight alpha-tocopherol stereoisomers results in preferential accumulation of the four 2R forms in tissues and plasma of rats. J. Nutr. 1996, 126, 2539–2549. [Google Scholar] [PubMed]
- Dersjant-Li, Y.; Jensen, S.K.; Bos, L.W.; Peisker, M.R. Bio-discrimination of alpha-tocopherol stereoisomers in rearing and veal calves fed milk replacer supplemented with all-rac-alpha-tocopheryl acetate. Int. J. Vitam. Nutr. Res. 2009, 79, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.K.; Norgaard, J.V.; Lauridsen, C. Bioavailability of alpha-tocopherol stereoisomers in rats depends on dietary doses of all-rac- or RRR-alpha-tocopheryl acetate. Br. J. Nutr. 2006, 95, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Burton, G.W.; Ingold, K.U.; Kayden, H.J. RRR- and SRR-alpha-tocopherols are secreted without discrimination in human chylomicrons, but RRR-alpha-tocopherol is preferentially secreted in very low density lipoproteins. J. Lipid Res. 1990, 31, 675–685. [Google Scholar] [PubMed]
- Lauridsen, C.; Theil, P.K.; Jensen, S.K. Composition of -tocopherol and fatty acids in porcine tissues after dietary supplementation with vitamin E and different fat sources. Anim. Feed Sci. Technol. 2013, 179, 93–102. [Google Scholar] [CrossRef]
- Burton, G.W.; Traber, M.G.; Acuff, R.V.; Walters, D.N.; Kayden, H.; Hughes, L.; Ingold, K.U. Human plasma and tissue alpha-tocopherol concentrations in response to supplementation with deuterated natural and synthetic vitamin E. Am. J. Clin. Nutr. 1998, 67, 669–684. [Google Scholar] [PubMed]
- Lauridsen, C.; Engel, H.; Jensen, S.K.; Craig, A.M.; Traber, M.G. Lactating sows and suckling piglets preferentially incorporate RRR- over all-rac-alpha-tocopherol into milk, plasma and tissues. J. Nutr. 2002, 132, 1258–1264. [Google Scholar] [PubMed]
- Kumar, A.P.; Jin, D.; Lee, Y.-I. Recent development on spectroscopic methods for chiral analysis of enantiomeric compounds. Appl. Spectrosc. Rev. 2009, 44, 267–316. [Google Scholar] [CrossRef]
- Nie, Y.; Liu, X.; Yang, X.; Zhao, X. Review: Recent application of chiral liquid chromatography-tandem mass spectrometric methods for enantiomeric pharmaceutical and biomedical determinations. J. Chromatogr. Sci. 2013, 51, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Barba, C.; Santa-María, G.; Herraiz, M.; Martínez, R.M. Direct enantiomeric analysis of Mentha essential oils. Food Chem. 2013, 141, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Cretin, B.N.; Dubourdieu, D.; Marchal, A. Development of a quantitation method to assay both lyoniresinol enantiomers in wines, spirits, and oak wood by liquid chromatography-high resolution mass spectrometry. Anal. Bioanal. Chem. 2016, 408, 3789–3799. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.F.; Enders, J.R.; Bell, D.S.; Cramer, H.M.; Wallace, F.N.; McIntire, G.L. Improved Chiral Separation of Methamphetamine Enantiomers Using CSP-LC-MS-MS. J. Anal. Toxicol. 2016, 40, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Janssen, M.H.; Powis, I. Detecting chirality in molecules by imaging photoelectron circular dichroism. Phys. Chem. Chem. Phys. 2014, 16, 856–871. [Google Scholar] [CrossRef] [PubMed]
- Fanood, M.M.; Ram, N.B.; Lehmann, C.S.; Powis, I.; Janssen, M.H. Enantiomer-specific analysis of multi-component mixtures by correlated electron imaging–ion mass spectrometry. Nat. Commun. 2015, 6, 7511. [Google Scholar] [CrossRef] [PubMed]
- Boesl, U.; Kartouzian, A. Mass-Selective Chiral Analysis. Annu. Rev. Anal. Chem. 2016, 9, 343–364. [Google Scholar] [CrossRef] [PubMed]
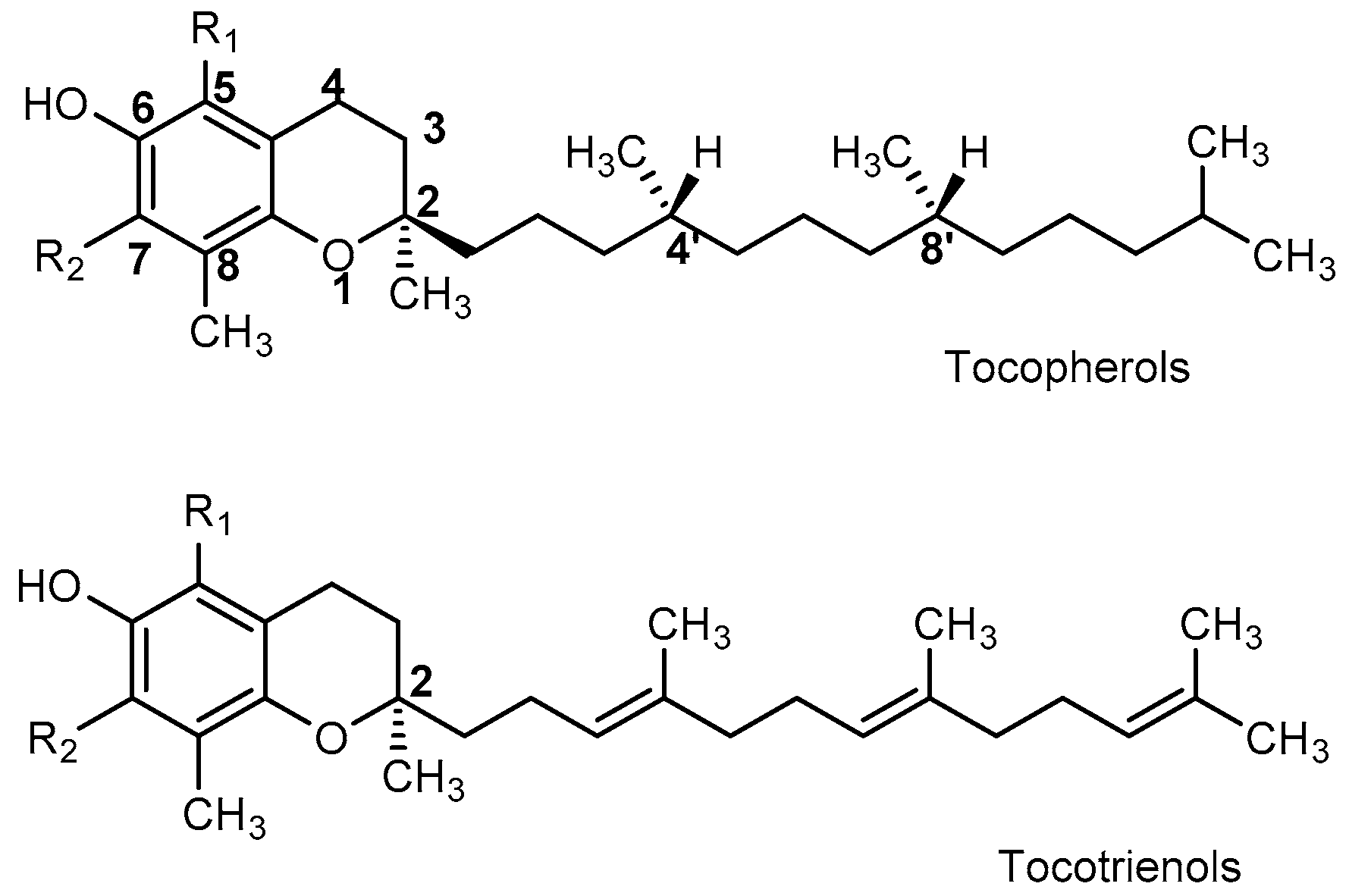
| Tocopherols | Tocotrienols | R1 | R2 |
|---|---|---|---|
| -Tocopherol | -Tocotrienol | CH3 | CH3 |
| β-Tocopherol | -Tocotrienol | CH3 | H |
| -Tocotrienol | -Tocotrienol | H | CH3 |
| -Tocopherol | -Tocotrienol | H | H |
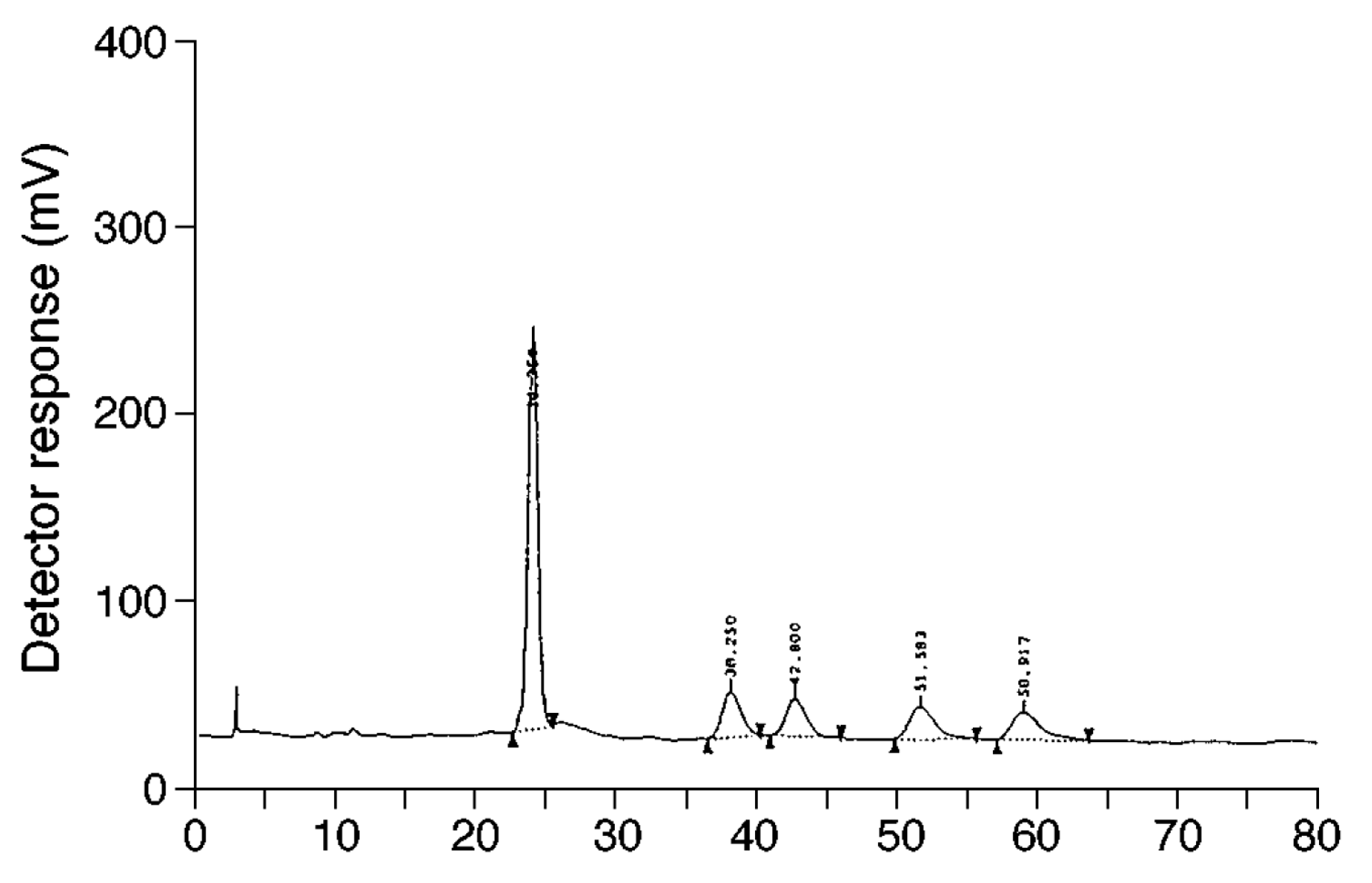

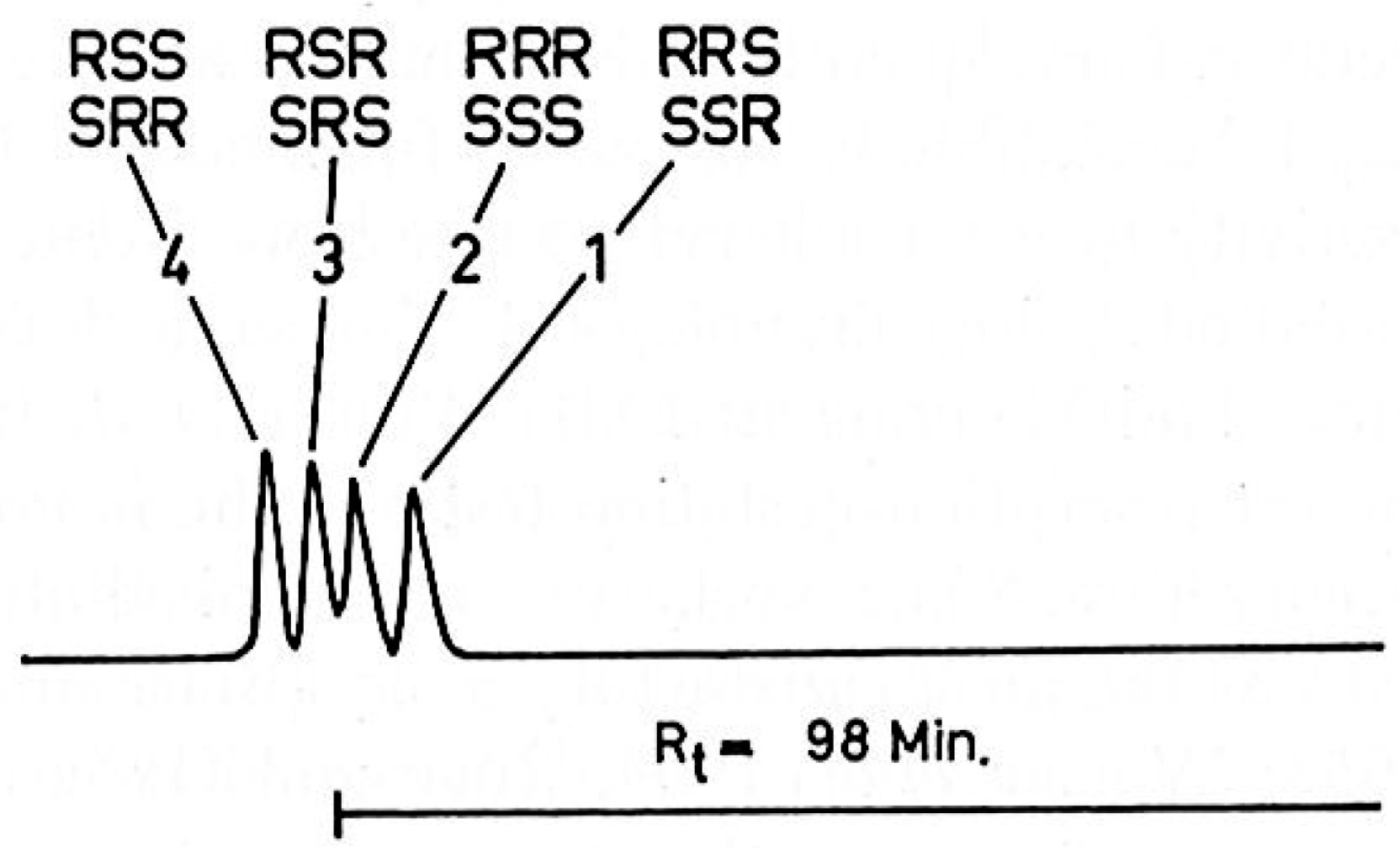
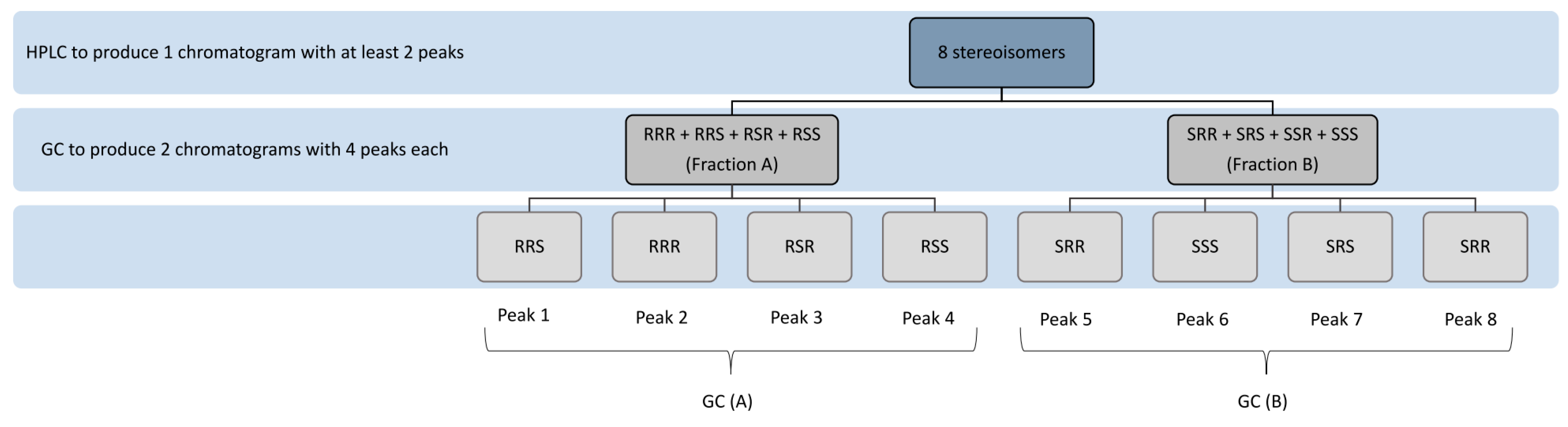
| Column | Mobile Phase | Analytes | Detection Wavelength | Stereoisomer/Diastereomers Separation | Application | Reference |
|---|---|---|---|---|---|---|
| Chiralcel OD-H (250 × 4.6 mm I.D.) | n-hexane | α-Toc methyl ether | FL: 284 nm (Ex) | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Cow feed and muscle, human plasma | [17,36] |
| 326 nm (Em) | ||||||
| Chiralpak OP (+) (250 × 4.6 mm I.D.) | Acetonitrile | α-Toc acetate | UV: 284 nm | (RRR + SSS, RRS + SSR, RSR + SRS), (RRS + SSR, RSR + SRS), (SSR + RSS) | Commercial product | [22] |
| Nucleosil1000-5 coated with (+)-PTMA) (250 × 4 mm I.D.) | Acetonitrile/H2O | α-Toc acetate | UV: 200 nm | (RSR + RSS), (RRR + RRS), (SSS + SSR), (SRS + SRR) | Commercial product, rat blood and tissue | [23] |
| Chiralpak OP (+) (250 × 4.6 mm I.D.) | Methanol/H2O | α-Toc acetate | UV: 284 nm | (RRR + RSR + RRS + RSS), (SSS + SSR), SRR, SRS | Rat tissue, blood, plasma and tissue, human serum and lipoproteins | [24,25,26,27] |
| Chiralcel OD (250 × 4.6 mm I.D.) | n-hexane | α-Toc methyl ether | UV: 283 nm | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Cellular lipid extracts | [28] |
| Chiralcel OD-H (250 × 4.6 mm I.D.) | n-hexane | α-Toc methyl ether | FL: 295 nm (Ex) | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Pharmaceutical preparations of Vitamin E | [29] |
| 330 nm (Em) | ||||||
| Chiralcel OD-H (250 × 4.6 mm I.D.) | n-heptane/isopropanol | α-Toc methyl ether | FL: 290 nm (Ex) | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Pig milk from lactation, blood, subcutaneous fat and piglet Longissimus dorsi muscle, rat plasma, tissues and faeces, cow milk and blood | [30,31,32,34] |
| 327 nm (Em) | ||||||
| Chiralcel OD-H (250 × 4.6 mm I. D.) | n-heptane /isopropanol | α-Toc methyl ether | FL: 295 nm (Ex) | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Chicken feed, liver and thigh | [33] |
| 330 nm (Em) | ||||||
| Chiralcel OD-H (250 × 4.6 mm I.D.) | n-heptane/isopropanol | α-Toc methyl ether | FL: 296 nm (Ex) | (SSS + SSR + SRR + SRS), RSS, RRS, RRR, RSR | Cow plasma, colostrum, milk and blood neutrophils | [35] |
| 372 nm (Em) | ||||||
| Chiralcel OD-H (250 × 4.6 mm I.D.) | Hexane/ethanol | α-Toc | UV: 220 nm | (SSS + SSR + SRR + SRS), (RSS + RRS + RRR + RSR) | Garcinia Kola seeds | [37] |
| MAA-CS capillary (52 cm (47 cm) × 75 mm I.D.) | Background electrolyte: borate buffer modified with acetonitrile | α-Toc | UV: 220 nm | (SSS + SSR + SRR + SRS), (RSS + RRS + RRR + RSR) | Pharmaceutical preparation of Vitamin E | [38] |
| Chiralcel OD-H (250 × 4.6 mm I.D.) | Isohexane/isopropanol | α-tocotrienol | FL: 295 nm (Ex) | (RS,Z-Z), (RS,Z-E-), (RS,E-Z-), (RS,E-E) | Pharmaceutical preparation of Vitamin E | [39] |
| 339 nm (Em) | ||||||
| Nucleodex β-PM (200 × 4 mm I.D.) | Acetonitrile/H2O | α-tocotrienol | UV: 230 nm | (RS, Z-Z- + RS,E/Z- diastereomer), (RS,Z-Z- enantiomer), (RS,E/Z-diasteromers ), (RSS,E-E), (RSR,E-E + RS,E/Z- diastereomer) | Pharmaceutical preparation of Vitamin E | [40] |
| Column | Carrier Gas | Injector Condition | FID Temperature | Analytes | Stereoisomer/Diastereomers Separation | Application | Reference |
|---|---|---|---|---|---|---|---|
| 100 m × 0.3 mm glass capillary column coated with Silar 10 C; temperature 185 °C | Hydrogen at 25 cm/s; split ratio 1/50 | Sample concentration 1 mg/mL; temperature 270 °C | 270 °C | Different preparations of all-rac-α-tocopheryl acetate from large scale production, 4’-ambo-8’-ambo-α-tocopheryl acetate, RRR-α-tocopheryl acetate. All samples were derivatised into methyl ethers. | (RRS + SSR), (RRR + SSS), (RSR + SRS), (RSS + SRR) | Commercial product | [14] |
| Glass capillary column (115 m × 0.25 mm, coated with highly polar liquid phase SP2340; temperature 195 °C) | Hydrogen at 19 cm/s; split ratio 1/50 to 1/100 | Sample size 1.7 μL at concentration 2 mg/mL; temperature 280 °C | 300 °C | RRR-α-tocopherol, 2-ambo-α-tocopherol, all-rac-α-tocopherol and 4’-ambo-8’-ambo-α-tocopherol. All samples were derivatised into TMS ethers. | 2-ambo-α-tocopherol: RRR, SRR. All-rac-α-tocopherol: (RRR + SSS), (RSS + SRR). 4’-ambo-8’-ambo-α-tocopherol: RRR, RSS | Commercial products | [42] |
| Glass capillary column (100 m × 0.3 mm, coated with Silar 10 C; temperature 185 °C) | Hydrogen at 17cm/s; split ratio 1/200 | Sample size 2 μL at concentration 1 mg/mL; temperature 250 °C | 300 °C | All-rac-α-tocopherol and α-tocopheryl acetate. All samples were derivatised into methyl ethers. | (RRS + SSR), (RRR + SSS), (RSR + SRS), (RSS + SRR) | Commercial products | [43] |
| 100 m × 0.3 mm glass capillary column coated with Silar 10 C; temperature 185 °C | Hydrogen at 25 cm/s; split ratio 1/50 | Sample concentration 1 mg/mL; temperature 270 °C | 270 °C | All-rac-α-tocopheryl acetate, 2-ambo-α-tocopheryl acetate, RRR-α-tocopheryl acetate. All samples were derivatised into methyl ethers. | (RRS + SSR), (RRR + SSS), (RSR + SRS), (RSS + SRR) | Commercial product | [44] |
| Fused silica capillary column (50 m × 0.22 mm, CP-Sil 88; temperature programmed from 150 to 210 °C at 2 °C/min) (with a 2-min hold at 150 °C and 10 min hold at 210 °C) and from 210 to 230 °C at 1 °C/min (with 20 min hold at 230 °C) | Helium at 1.8 mL/min; split ratio 1/30 | Sample size 0.7–2.0 μL; temperature 240 °C | 260 °C | α-tocopherol in chicken feed and eggs, derivatised into methyl ethers. | (RRS + SSR), (RRR + SSS), (RSR + SRS), (RSS + SRR) | Animal feeds and products | [45] |
| Analytes | HPLC | GC | Stereoisomer/Diastereomers Separation | Application | Reference | ||||
|---|---|---|---|---|---|---|---|---|---|
| Column | Mobile Phase | Detection Wavelength | Column | Injector Condition | FID Temperature | ||||
| α-Toc ethyl ether | Nucleosil1000-5 coated with (+)-PTMA) (25 × 0.4 cm) | Acetonitrile/H2O (9:1, v/v) | 200 nm (UV) | Silar 10 C coated glass capillary tube (100 m × 0.30 mm), isothermal at 165 °C | Splitless mode at 260 °C | 220 °C | RRS, RRR, RSR, RSS, SRR, SSS, SRS, SRR | Commercial product | [23] |
| α-Toc acetate (HPLC), α-Toc methyl ether (GC) | Chiralcel OD (25 × 0.46 cm) | Acetonitrile/H2O (9:1, v/v) | 200 nm (UV) | Silar 10 C coated glass capillary tube (100 m × 0.30 mm), isothermal at 165 °C | Splitless mode at 260 °C | 220 °C | RRS, RRR, RSR, RSS, SRR, SSS, SRS, SRR | Rat blood and tissue | [47] |
| α-Toc methyl ether | Nucleosil1000-5 coated with (+)-PTMA) (25 × 0.4 cm) Chiralcel OD (25 × 0.46 cm) | n-hexane | 200 nm (UV) | Silar 10 C coated glass capillary tube (100 m × 0.30 mm), isothermal at 165 °C | Splitless mode at 260 °C | 220 °C | RRS, RRR, RSR, RSS, SRR, SSS, SRS, SRR | Rat blood and tissue | [46] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, J.-Y.; Htar, T.-T.; De Silva, L.; Tan, D.M.-Y.; Chuah, L.-H. Chromatographic Separation of Vitamin E Enantiomers. Molecules 2017, 22, 233. https://doi.org/10.3390/molecules22020233
Fu J-Y, Htar T-T, De Silva L, Tan DM-Y, Chuah L-H. Chromatographic Separation of Vitamin E Enantiomers. Molecules. 2017; 22(2):233. https://doi.org/10.3390/molecules22020233
Chicago/Turabian StyleFu, Ju-Yen, Thet-Thet Htar, Leanne De Silva, Doryn Meam-Yee Tan, and Lay-Hong Chuah. 2017. "Chromatographic Separation of Vitamin E Enantiomers" Molecules 22, no. 2: 233. https://doi.org/10.3390/molecules22020233
APA StyleFu, J.-Y., Htar, T.-T., De Silva, L., Tan, D. M.-Y., & Chuah, L.-H. (2017). Chromatographic Separation of Vitamin E Enantiomers. Molecules, 22(2), 233. https://doi.org/10.3390/molecules22020233






