Effects of Polysaccharides from Platycodon grandiflorum on Immunity-Enhancing Activity In Vitro
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Preparation of PGPSs
2.3. Peripheral Lymphocyte Proliferation Assay
2.4. Cell Cycle Distribution Analysis
2.5. Detection of CD4+ and CD8+ T Lymphocytes
2.6. Statistical Analysis
3. Results
3.1. Characterization of Platycodon grandiflorum
3.2. Cytotoxicity of PGPSs to Peripheral Lymphocytes
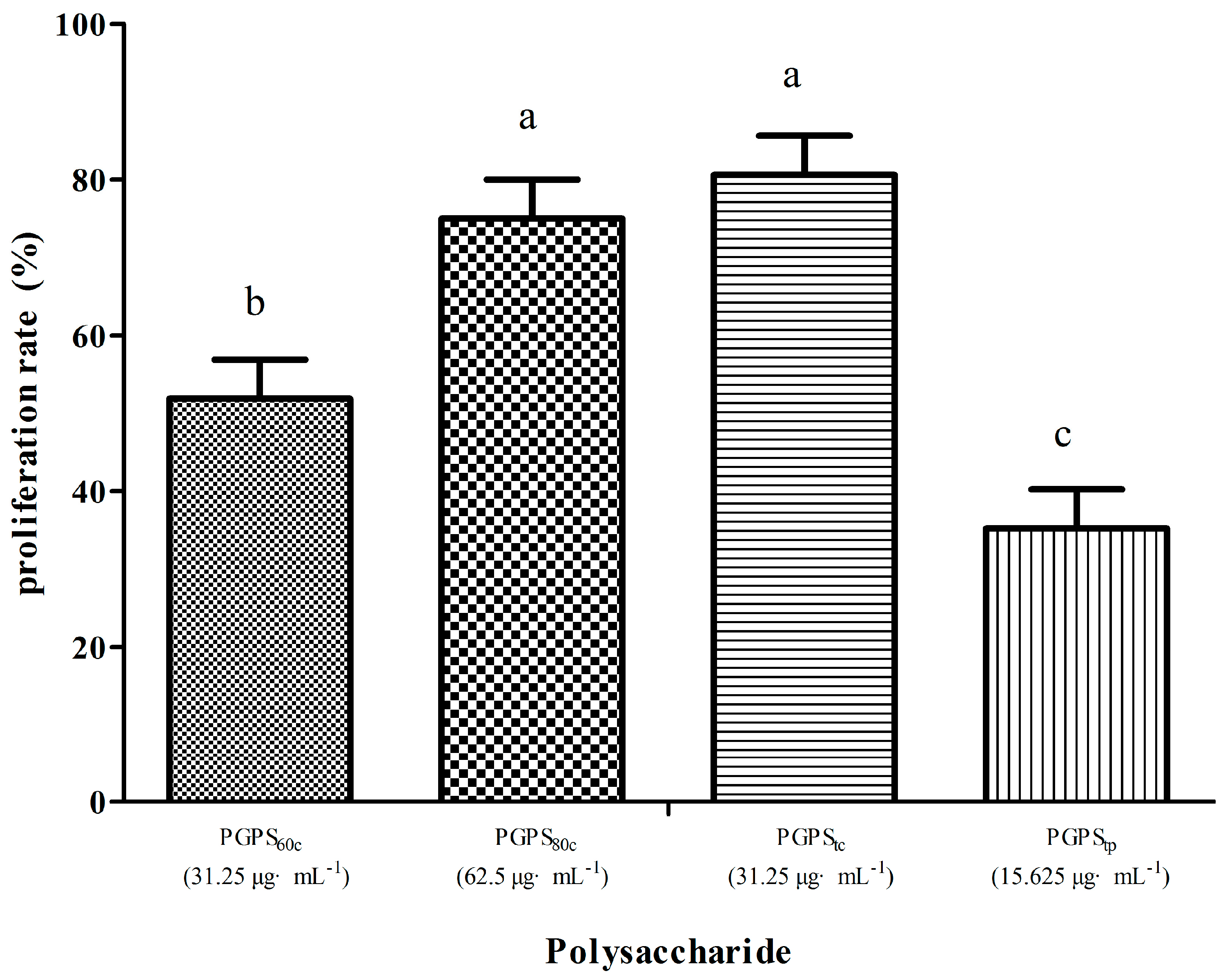
3.3. Peripheral Lymphocyte Proliferation in Single Stimulation with PGPSs
3.4. Peripheral Lymphocyte Proliferation in Synergistic Stimulation of PGPSs with PHA
3.5. Cell Cycle Analysis
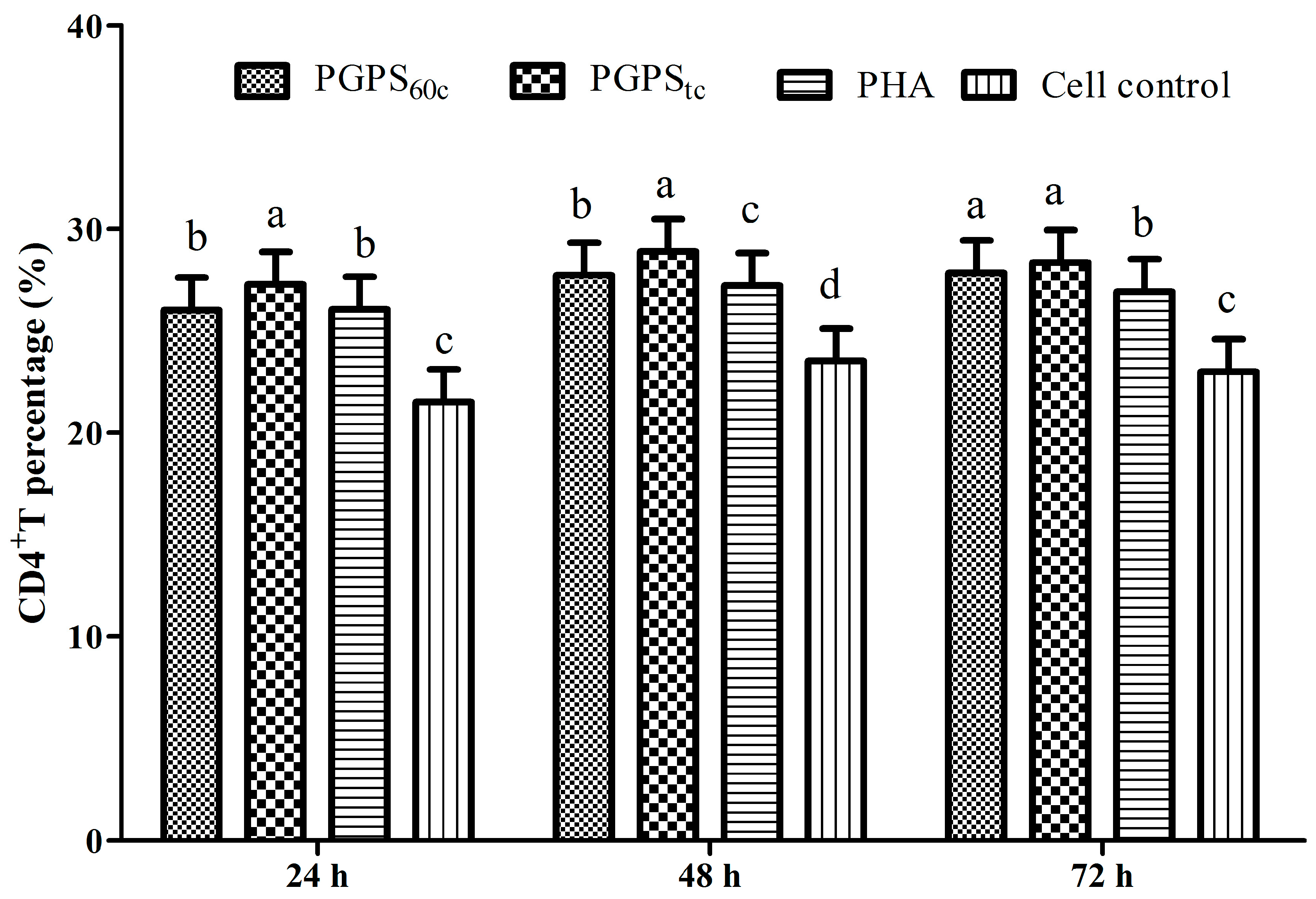
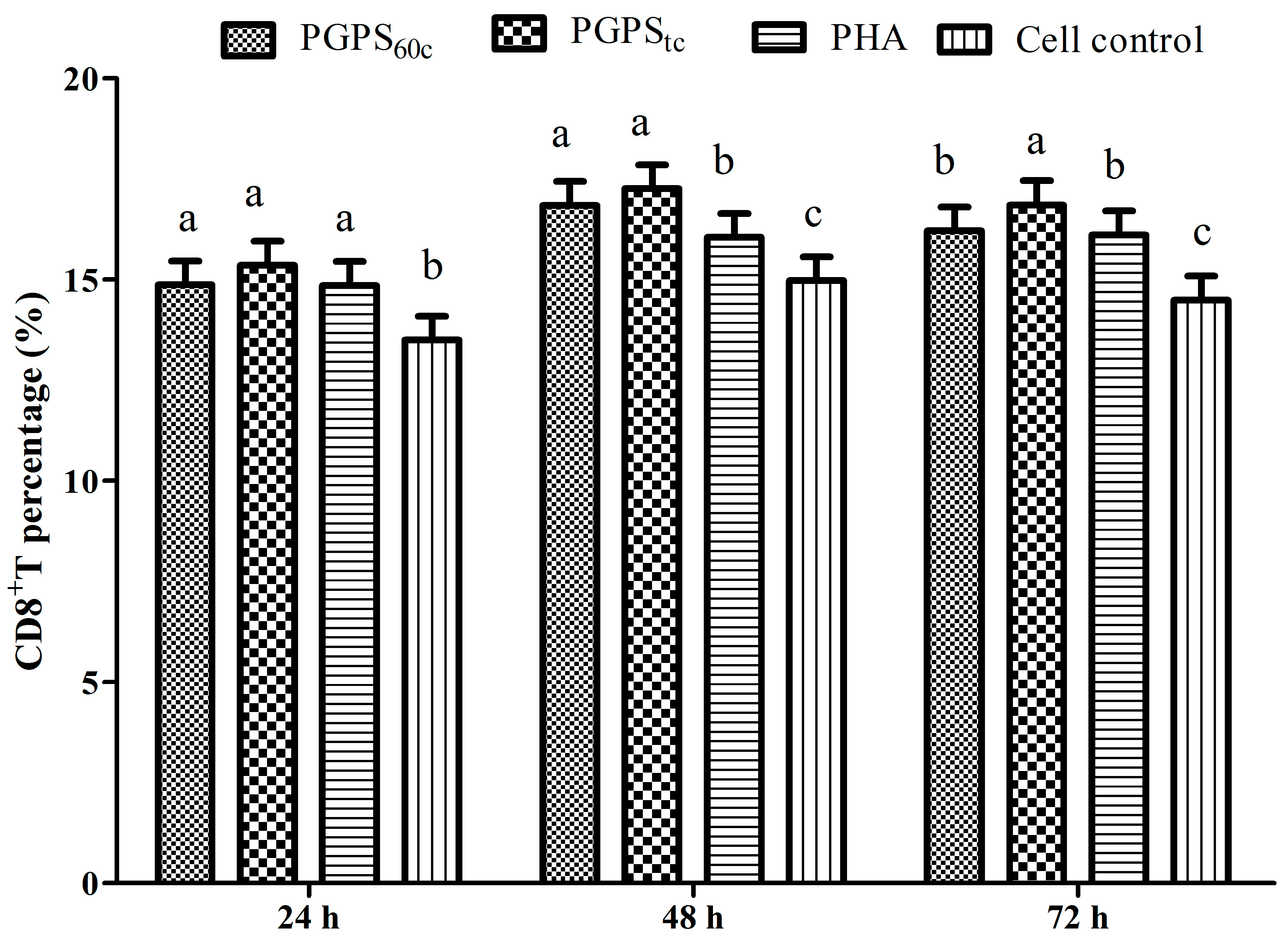
3.6. Changes in Lymphocytes CD4+ and CD8+ T Cells
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Faggio, C.; Pagano, M.; Dottore, A.; Genovese, G.; Morabito, M. Evaluation of anticoagulant activity of two algal polysaccharides. Nat. Prod. Res. 2016, 30, 1934–1937. [Google Scholar] [CrossRef] [PubMed]
- Faggio, C.; Morabito, M.; Minicante, S.A.; Lo Piano, G.; Pagano, M.; Genovese, G. Potential use of polysaccharides from the brown alga Undaria pinnatifida as anticoagulants. Braz. Arch. Biol. Technol. 2015, 3, 759–770. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, S.; Zhang, F.; Hu, R. Adjuvant activity of Chinese herbal polysaccharides in inactivated veterinary rabies vaccines. Int. J. Biol. Macromol. 2012, 50, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Liang, M.-F.; Liu, G.-H.; Zhao, Q.-Y.; Yang, S.-F.; Zhong, S.-X.; Cui, G.-L.; He, X.-H.; Zhao, X.; Guo, F.-X.; Wu, C. Effects of Taishan Robinia pseudoacacia Polysaccharides on immune function in chickens. Int. Immunopharmacol. 2013, 15, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, Y.; Shen, J.; Wang, S.; Yao, J.; Yang, X. Effect of Astragalus polysaccharide and its sulfated derivative on growth performance and immune condition of lipopolysaccharide-treated broilers. Int. J. Biol. Macromol. 2015, 76, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.-W.; Zhou, Q.-C.; Geng, J. Pyrolytic and kinetic characteristics of Platycodon grandiflorum peel and its cellulose extract. Carbohydr. Polym. 2015, 117, 644–649. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Ryu, H.S.; Kim, J.S.; Lee, H.K.; Kang, J.S.; Yun, J.; Kim, S.Y.; Lee, M.K.; Hong, J.T.; Kim, Y. Platycodon grandiflorum polysaccharide induces dendritic cell maturation via TLR4 signaling. Food Chem. Toxicol. 2014, 72, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Yeo Dae, Y.; Sang Bae, H.; Jong Soon, K.; Chang Woo, L.; Song-Kyu, P.; Hyun Sun, L.; Jong Seong, K.; Hwan Mook, K. Toll-like receptor 4-dependent activation of macrophages by polysaccharide isolated from the radix of Platycodon grandiflorum. Int. Immunopharmacol. 2003, 3, 1873–1882. [Google Scholar]
- Qin, T.; Chen, J.; Wang, D.; Hu, Y.; Wang, M.; Zhang, J.; Nguyen, T.L.; Liu, C.; Liu, X. Optimization of selenylation conditions for Chinese angelica polysaccharide based on immune-enhancing activity. Carbohydr. Polym. 2013, 92, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Hu, Y.; Wang, D.; Guo, L.; Yang, S.; Fan, Y.; Zhao, B.; Wang, Y.; Abula, S. Optimization of sulfated modification conditions of tremella polysaccharide and effects of modifiers on cellular infectivity of NDV. Int. J. Biol. Macromol. 2011, 49, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Fan, W.; Wang, S.; Pan, H.; Yang, W.; Wan, H.; Hao, Z.; Liu, J.; Zhao, X. Characterization of polysaccharides extracted from Platycodon grandiflorus (Jacq.) A.DC. affecting activation of chicken peritoneal macrophages. Int. J. Biol. Macromol. 2017, 96, 775–785. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Hu, Y.; Wang, D.; Liu, J.; Guo, L. The comparison of immune-enhancing activity of sulfated polysaccharidses from Tremella and Condonpsis pilosula. Carbohydr. Polym. 2013, 98, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Tang, X.; Wang, X.; Huang, X.; Ye, Y.; Lu, X.; Wei, X.; Zeng, Y. Bererine induces peripheral lymphocytes immune regulations to realize its neuroprotective effects in the cerebral ischemia/reperfusion mice. Cell. Immunol. 2012, 276, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Meng, K.; Qi, C.; Yang, X.; Wang, Y.; Fan, W.; Yan, Z.; Zhao, X.; Liu, J. Immune-enhancing activity of polysaccharides isolated from Atractylodis macrocephalae Koidz. Carbohydr. Polym. 2015, 126, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zhang, L.; Duan, X.; Liao, Z.; Ding, H.; Cheung, P.C. Novel highly branched water-soluble heteropolysaccharides as immunopotentiators to inhibit S-180 tumor cell growth in BALB/c mice. Carbohydr. Polym. 2012, 87, 427–434. [Google Scholar] [CrossRef]
- Yang, Z.; Xu, J.; Fu, Q.; Fu, X.; Shu, T.; Bi, Y.; Song, B. Antitumor activity of a polysaccharide from Pleurotus eryngii on mice bearing renal cancer. Carbohydr. Polym. 2013, 95, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Aceves, M.A.; Cohen, A.; Smith, Y.; Faggio, C. Estrogen regulation of gene expression in the teleost fish immune system. Fish Shellfish Immunol. 2016, 58, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Fan, J.; Qiu, H.; Wang, Z.; Yan, Z.; Yuan, L.; Guan, L.; Du, X.; Song, Z.; Han, X. Chuanminshen violaceum polysaccharides improve the immune responses of foot-and-mouth disease vaccine in mice. Int. J. Biol. Macromol. 2015, 78, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Ding, R.; Jiang, S.; Ji, L.; Pan, M.; Liu, L.; Zhang, W.; Gao, X.; Huang, W.; Zhang, G. The adjuvanticity of Ganoderma lucidum polysaccharide for Newcastle disease vaccine. Int. J. Biol. Macromol. 2014, 65, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, X.; Yue, C.; Hou, R.; Chen, J.; Lu, Y.; Li, X.; Li, R.; Liu, C.; Gao, Z. Effect of selenylation modification on immune-enhancing activity of Atractylodes macrocephala polysaccharide. Int. J. Biol. Macromol. 2015, 72, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; He, C.; Fan, Y.; Si, H.; Wang, Y.; Shi, Z.; Zhao, X.; Zheng, Y.; Liu, Q.; Zhang, H. Immune-enhancing activity of polysaccharides from Cyrtomium macrophyllum. Int. J. Biol. Macromol. 2014, 70, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, M.; Sun, R.; Pan, L. Extraction, characterization of a Ginseng fruits polysaccharide and its immune modulating activities in rats with Lewis lung carcinoma. Carbohydr. Polym. 2015, 127, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Chi, A.; Kang, C.; Zhang, Y.; Tang, L.; Guo, H.; Li, H.; Zhang, K. Immunomodulating and antioxidant effects of polysaccharide conjugates from the fruits of Ziziphus Jujube on Chronic Fatigue Syndrome rats. Carbohydr. Polym. 2015, 122, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Zheng, P.; Wang, Y.; Hao, P.; Liu, J.; Zhao, X. Analysis of immunostimulatory activity of polysaccharide extracted from Yu-Ping-Feng in vitro and in vivo. Biomed. Pharmacother. 2017, 93, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wu, C.; Liu, Z.; Hu, Y.; Shi, C.; Yu, Y.; Zhao, X.; Liu, C.; Liu, J.; Wu, Y. Optimization on preparation conditions of Rehmannia glutinosa polysaccharide liposome and its immunological activity. Carbohydr. Polym. 2014, 104, 118–126. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |



| Concentration (μg·mL−1) | PGPS60c | PGPS80c | PGPStc | PGPStp |
|---|---|---|---|---|
| 2000 | 0.111 ± 0.003 c | 0.101 ± 0.004 c | 0.113 ± 0.001 d | 0.106 ± 0.002 f |
| 1000 | 0.110 ± 0.010 c | 0.120 ± 0.002 b | 0.114 ± 0.001 c,d | 0.109 ± 0.003 e,f |
| 500 | 0.141 ± 0.006 a | 0.125 ± 0.005 c | 0.118 ± 0.002 b,c,d | 0.110 ± 0.003 d,e,f |
| 250 | 0.131 ± 0.009 a,b | 0.128 ± 0.002 b | 0.118 ± 0.001 b,c,d | 0.112 ± 0.003 c,d,e,f |
| 125 | 0.130 ± 0.002 a,b | 0.127 ± 0.003 b | 0.118 ± 0.002 b,c,d | 0.114 ± 0.001 c,d,e |
| 62.5 | 0.126 ± 0.004 b | 0.179 ± 0.002 a | 0.119 ± 0.002 b,c,d | 0.130 ± 0.003 a |
| 31.25 | 0.128 ± 0.001 a,b | 0.125 ± 0.002 b | 0.133 ± 0.002 a | 0.130 ± 0.001 a |
| 15.625 | 0.128 ± 0.004 a,b | 0.126 ± 0.006 b | 0.133 ± 0.001 a | 0.130 ± 0.002 a |
| 7.813 | 0.126 ± 0.001 b | 0.125 ± 0.001 b | 0.138 ± 0.001 a | 0.123 ± 0.003 a,b |
| 3.907 | 0.131 ± 0.003 a,b | 0.126 ± 0.000 b | 0.121 ± 0.004 b,c | 0.118 ± 0.001 b,c |
| 1.953 | 0.128 ± 0.003 a,b | 0.128 ± 0.001 b | 0.119 ± 0.004 b,c,d | 0.117 ± 0.002 b,c,d |
| Cell control | 0.125 ± 0.002 b | 0.125 ± 0.002 b | 0.124 ± 0.001 b | 0.124 ± 0.001 a,b |
| Concentration (μg·mL−1) | PGPS60c | PGPS80c | PGPStc | PGPStp |
|---|---|---|---|---|
| 62.5 | 0.196 ± 0.009 a | 0.224 ± 0.019 a | 0.210 ± 0.007 a | 0.151 ± 0.005 a |
| 31.25 | 0.202 ± 0.005 a | 0.219 ± 0.009 a | 0.224 ± 0.024 a | 0.160 ± 0.013 a |
| 15.625 | 0.200 ± 0.004 a | 0.208 ± 0.010 a,b | 0.219 ± 0.012 a | 0.165 ± 0.009 a |
| 7.813 | 0.192 ± 0.003 a | 0.200 ± 0.010 a,b | 0.213 ± 0.007 a | 0.162 ± 0.008 a,b |
| 3.907 | 0.190 ± 0.015 a | 0.182 ± 0.006 b | 0.218 ± 0.007 a | 0.168 ± 0.003 a,b |
| Cell control | 0.133 ± 0.006 b | 0.128 ± 0.0039 c | 0.122 ± 0.004 b | 0.122 ± 0.004 b |
| Concentration (μg·mL−1) | PGPS60c | PGPS80c | PGPStc | PGPStp |
|---|---|---|---|---|
| 62.5 | 0.122 ± 0.002 a,b,c | 0.123 ± 0.004 a | 0.152 ± 0.029 a,b | 0.135 ± 0.009 b,c |
| 31.25 | 0.132 ± 0.004 a | 0.118 ± 0.004 a | 0.210 ± 0.008 a | 0.130 ± 0.010 b,c |
| 15.625 | 0.125 ± 0.002 a,b | 0.126 ± 0.006 a | 0.187 ± 0.002 a | 0.148 ± 0.009 a,b |
| 7.813 | 0.113 ± 0.000 b,c | 0.125 ± 0.006 a | 0.184 ± 0.005 a | 0.161 ± 0.013 a |
| 3.907 | 0.115 ± 0.002 b,c | 0.118 ± 0.005 a | 0.186 ± 0.007 a | 0.145 ± 0.007 a,b |
| PHA | 0.111 ± 0.003 c | 0.112 ± 0.004 a | 0.120 ± 0.002 b | 0.115 ± 0.002 c |
| Group | SPF | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| PGPS60c | 17.787 ± 0.202 a,b | 20.630 ± 0.052 a | 17.277 ± 0.993 b |
| PGPStc | 18.673 ± 0.511 a | 20.263 ± 0.265 a | 18.800 ± 0.779 a |
| PHA | 17.050 ± 0.309 b | 19.203 ± 0.178 b | 17.767 ± 0.393 a,b |
| Cell control | 14.830 ± 0.432 c | 16.760 ± 0.382 c | 14.613 ± 0.296 c |
| Group | PI | ||
|---|---|---|---|
| 24 h | 48 h | 72 h | |
| PGPS60c | 28.223 ± 0.419 b | 30.330 ± 0.427 b | 28.393 ± 0.435 a,b |
| PGPStc | 29.347 ± 0.077 a | 31.860 ± 0.639 a | 30.490 ± 0.524 a |
| PHA | 27.713 ± 0.999 b | 27.130 ± 0.929 c | 27.2670 ± 0.202 b |
| Cell control | 22.547 ± 0.855 c | 23.603 ± 0.762 d | 21.990 ± 0.743 c |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.; Wang, Y.; Yan, P.; Cheng, G.; Wang, C.; Geng, N.; Wang, X.; Liu, J. Effects of Polysaccharides from Platycodon grandiflorum on Immunity-Enhancing Activity In Vitro. Molecules 2017, 22, 1918. https://doi.org/10.3390/molecules22111918
Zhao X, Wang Y, Yan P, Cheng G, Wang C, Geng N, Wang X, Liu J. Effects of Polysaccharides from Platycodon grandiflorum on Immunity-Enhancing Activity In Vitro. Molecules. 2017; 22(11):1918. https://doi.org/10.3390/molecules22111918
Chicago/Turabian StyleZhao, Xiaona, Yuge Wang, Peng Yan, Guodong Cheng, Cheng Wang, Na Geng, Xuepeng Wang, and Jianzhu Liu. 2017. "Effects of Polysaccharides from Platycodon grandiflorum on Immunity-Enhancing Activity In Vitro" Molecules 22, no. 11: 1918. https://doi.org/10.3390/molecules22111918
APA StyleZhao, X., Wang, Y., Yan, P., Cheng, G., Wang, C., Geng, N., Wang, X., & Liu, J. (2017). Effects of Polysaccharides from Platycodon grandiflorum on Immunity-Enhancing Activity In Vitro. Molecules, 22(11), 1918. https://doi.org/10.3390/molecules22111918






