Abstract
Phytochemical investigation of the acetone extract from the roots of Aphanamixis polystachya resulted in isolation of four new tetranortriterpenes (1–4) in addition to one protolimonoid (methyl-1ξ,7R-diacetoxy-23R,25-dihydroxy-20S,24R-21,24-epoxy-3,4-seco-apotirucall-4(28),14(15)-diene-3-oate (5)), five known limonoids (rohituka 3 (6), rohituka 7 (7), nymania 1 (8), rubrin G (9), prieurianin (10)) and a steroid (2,3-dihydroxy-5-pregnan-16-one (11)). Their structures were determined by spectroscopic analyses, including 2D-NMR (COSY, HMQC, HMBC, and NOESY) and high-resolution electrospray ionization mass spectrometry (HRESIMS). Cytotoxic and anti-inflammatory activities of these compounds were evaluated. Compounds 4 and 5 showed significant inhibition against superoxide generation and elastase release by human neutrophils in response to (formyl-l-methionyl-l-leucyl-l-phenylalanine/cytochalasin B) (FMLP/CB).
1. Introduction
Aphanamixis polystachya (Wall.) R.N. Parker (Meliaceae) is a tropical neem tree native to Asia, especially China and India. According to previous studies, the meliaceaeous plants are famous for the limonoid-producing source and the pest control function in agriculture [1,2]. Phytochemical investigations of this plant have resulted in isolation of many limonoids with diverse skeletons [3,4,5,6,7]. Other structures such as guaiane sesquiterpenes from the seeds [8], lignans from the stem barks [9], chromone, and flavonoid glycosides from the roots [10] were also reported. In biological studies, it has been used as traditional Bangladesh medicine for the treatment of cancers, diabetes, and liver diseases [11] in addition to insecticide applications. Besides, the leaf extract showed CNS (central nervous system) depressant and analgesic activity in vivo [12], as well as antimicrobial, antioxidant, cytotoxic, and thrombolytic activities in vitro [13]. Currently, some limonoids from A. polystachya attract much attention due to their interesting anticancer [14], antifungal [15], and antifeedant activities [16,17].
Continuing our phytochemical investigation on the new anti-cancer and anti-inflammatory agents from terrestrial source, the crude extract from the roots of A. polystachya showed significant inhibition on superoxide anion generation and elastase release by human neutrophils in response to FMLP/CB at 10 μg/mL. Fractionation of the active components from the roots of this species was thus initiated. Herein, we report the isolation and structural elucidation of four new and six known triterpenoids, together with one known steroid from the roots of A. polystachya. The evaluation against human tumor cell lines and inhibitory activities on superoxide anion generation and elastase release are also discussed.
2. Materials and Methods
2.1. General Experimental Procedures
Optical rotations were recorded on a JASCO DIP-1000 polarimeter (JASCO, Tokyo, Japan). IR spectra were taken on a HORIBA FT-720 spectrophotometer (HORIBA, Kyoto, Japan). The 1H- and 13C-NMR spectra as well as 2D NMR spectra (COSY, HMQC, HMBC, and NOESY) were recorded in CDCl3 on a Bruker AVX NMR spectrometer (Bruker, Karlsruhe, Germany) operating at 400 MHz for 1H and 100 MHz for 13C using the CDCl3 solvent peak as internal standard (δH 7.265, δC 77.0 ppm). Low-resolution ESIMS were recorded on a VG Quattro 5022 mass spectrometer (VG Instruments, Altrincham, UK). HRESIMS were measured on a JEOL HX 110 mass spectrometer (JEOL, Peabody, MA, USA). LiChrospher® Si 60 (5 μm, 250-10, Merck, Darmstadt, Germany) and LiChrospher® 100 RP-18e (5 μm, 250-10, Merck) were used for NP-HPLC and RP-HPLC (Hitachi, L-6250, Kyoto, Japan; flow rate 2 mL/min, UV detection at 254 nm), respectively. HepG 2, A549 and MCF-7 cancer cells were purchased from Bioresource Collection and Research Center, Food Industry Research and Development Institute (Hsinchu, Taiwan), and Hep-2 was purchased from ATCC (American Type Culture Collectio, Manassas, VA, USA).
2.2. Plant Material
The roots of Aphanamixis polystachya (Wall.) R.N. Parker were collected in Ping-Tong County, Taiwan, in April 2012. The plant material was identified by one of the authors (C.T.C.). A voucher specimen (code No. TP 98-1) has been deposited at the School of Pharmacy, National Taiwan University, Taipei, Taiwan.
2.3. Extraction and Isolation
The air-dried roots (6.6 kg) of A. polystachya were ground and extracted thrice with acetone at room temperature and concentrated under reduced pressure to obtain a crude extract (250 g). The extract was partitioned between EtOAc:H2O (1:1) to give an EtOAc-soluble layer. After evaporating the organic solvent, the EtOAc residue (90 g) was partitioned between n-hexane:MeOH:H2O (4:3:1) to afford a MeOH/H2O extract. The MeOH/H2O extract (60 g) was subjected to pass over a Sephadex LH-20 column eluted with MeOH to afford fractions A–F. Fraction C (38 g) was separated on a Si gel (63–200 μm, 90 × 120 mm) column (n-hexane:EtOAc:MeOH, 100:0:0 to 0:0:100, 1 L for each gradient solvent) to afford fraction C-14 (7.1 g), which was further chromatographed on Si gel (<63 μm, 65 × 600 mm) column (n-hexane:EtOAc, 1:1, 5 L) to furnish three fractions, C-14-C, C-14-D, and C-14-E. Fraction C-14-C (1.7 g) was separated with a Si gel column and eluted with n-hexane:EtOAc:CH2Cl2 (1:1:1, 2.5 L) to give three subfractions, C-14-C-4, C-14-C-5 and C-14-C-6. Subfraction C-14-C-4 (385 mg) was subjected to RP-HPLC (5 μm, 10 mm × 250 mm) (MeOH:H2O, 7:1) to give 1 (7.6 mg, Rt = 20.3 min) and 2 (11.5 mg, Rt = 16.4 min). Subfraction C-14-C-6 (802 mg) was washed with MeOH and filtrated to yield 10 (443 mg). Fraction C-14-D (1.8 g) was separated by a reverse-phase column (20–50 μm, 35 × 35 mm) and eluted with MeOH/H2O (7:3, 2 L) to afford ten subfractions, C-14-D-1 to C-14-D-10. Subfraction C-14-D-6 (174 mg) was separated with an RP-HPLC (CH3CN/H2O, 55:45) to yield methyl-1ξ,7R-diacetoxy-23R,25-dihydroxy-20S,24R-21,24-epoxy-3,4-seco-apotirucall-4(28),14(15)-diene-3-oate (5, 15.6 mg, Rt = 35.4 min). Separation of fraction C-14-E (1.1 g) by a Si gel (<63 μm, 35 cm × 30 mm) column (n-hexane–acetone, 2:1, 2 L) obtained three subfractions, C-14-E-3, C-14-E-4 and C-14-E-6. Subfraction C-14-E-3 (236 mg) was applied on an RP-HPLC (CH3CN–H2O, 3:2) to yield 3 (18.1 mg, Rt = 39.1 min) and 2β,3β-dihydroxy-5α-pragnan-16-one (11, 17.8 mg, Rt = 22.2 min). Subfraction C-14-E-4 (187 mg) was purified by reverse-phase HPLC (MeOH–H2O, 7:1) to obtain 4 (26 mg, Rt = 11.1 min). Subfraction C-14-E-6 (358 mg) was separated by RP-HPLC (MeOH:H2O, 3:2) to furnish rohituka 3 (6, 30.7 mg, Rt = 37.7 min), rohituka 7 (7, 20.8 mg, Rt = 33.6 min), nymania 1 (8, 19.8 mg, Rt = 55.8 min), and rubrin G (9, 11.5 mg, Rt = 76.7 min).
3. Spectral Data
Aphataiwanin A (1). Colorless amorphous solid; −60 (c 0.1, MeOH); IR (CH2Cl2) vmax 3479, 3078, 1740, 1645, 977, 900 cm−1; 1H-NMR (CDCl3) and 13C-NMR (CDCl3) spectroscopic data, see Table 1 and Table 2, respectively; HRESIMS m/z 653.3684 [M + Na]+ (calcd for C36H54O9Na, 653.3660).

Table 1.
1H-NMR spectroscopic data (400 MHz) of compounds 1–4 a.

Table 2.
13C-NMR spectroscopic data (100 MHz) of compounds 1–4 a.
Aphataiwanin B (2). Colorless amorphous solid; −90 (c 0.1, MeOH); IR (CH2Cl2) vmax 3484, 3071, 1740, 1643, 983, 902 cm−1; 1H-NMR (CDCl3) and 13C-NMR (CDCl3) spectroscopic data, see Table 1 and Table 2, respectively; HRESIMS m/z 653.3681 [M + Na]+ (calcd for C36H54O9Na, 653.3660).
4. Biological Activities
4.1. Cytotoxicity Assay
Cytotoxicity was tested against human Hep-G2 (hepatocellular carcinoma), HEp-2 (laryngeal carcinoma), A549 (lung carcinoma) and MCF-7 (breast adenocarcinoma) tumor cell lines. The assay procedure using MTT (3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide) was carried out as previously described [18,19]. The cells were cultured in RPMI-1640 medium. After seeding of cells in a 96-well microplate for 4 h, 20 μL of sample was placed in each well and incubated at 37 °C for 3 days, and then 20 μL MTT was added for 5 h. After removing the medium and putting DMSO (200 μL/well) into the microplate with shaking for 10 min, the formazan crystals were redissolved and their absorbance was measured on a microtiter plate reader (MR 7000, Dynatech, Missouri City, TX, USA) at a wavelength of 550 nm. Mitomycin C was used as a positive control.
4.2. Anti-Inflammatory Assays, Inhibitory Effect on Superoxide Anion Generation and Elastase Release by Human Neutrophils
Neutrophils were obtained by means of dextran sedimentation and Ficoll centrifugation. Superoxide generation and elastase release were carried out according to a procedure described previously [20]. Superoxide anion production was assayed by monitoring the superoxide dismutase-inhibitable reduction of ferricytochrome c. Elastase release experiments were performed using MeO-Suc-Ala-Ala-Pro-Valp-nitroanilide as the elastase substrate. Genistein was used as a standard compound, which showed inhibition of 65.0 ± 5.7 and 51.6 ± 5.9 at 10 μg/mL, respectively, on superoxide anion generation and elastase release.
5. Results and Discussion
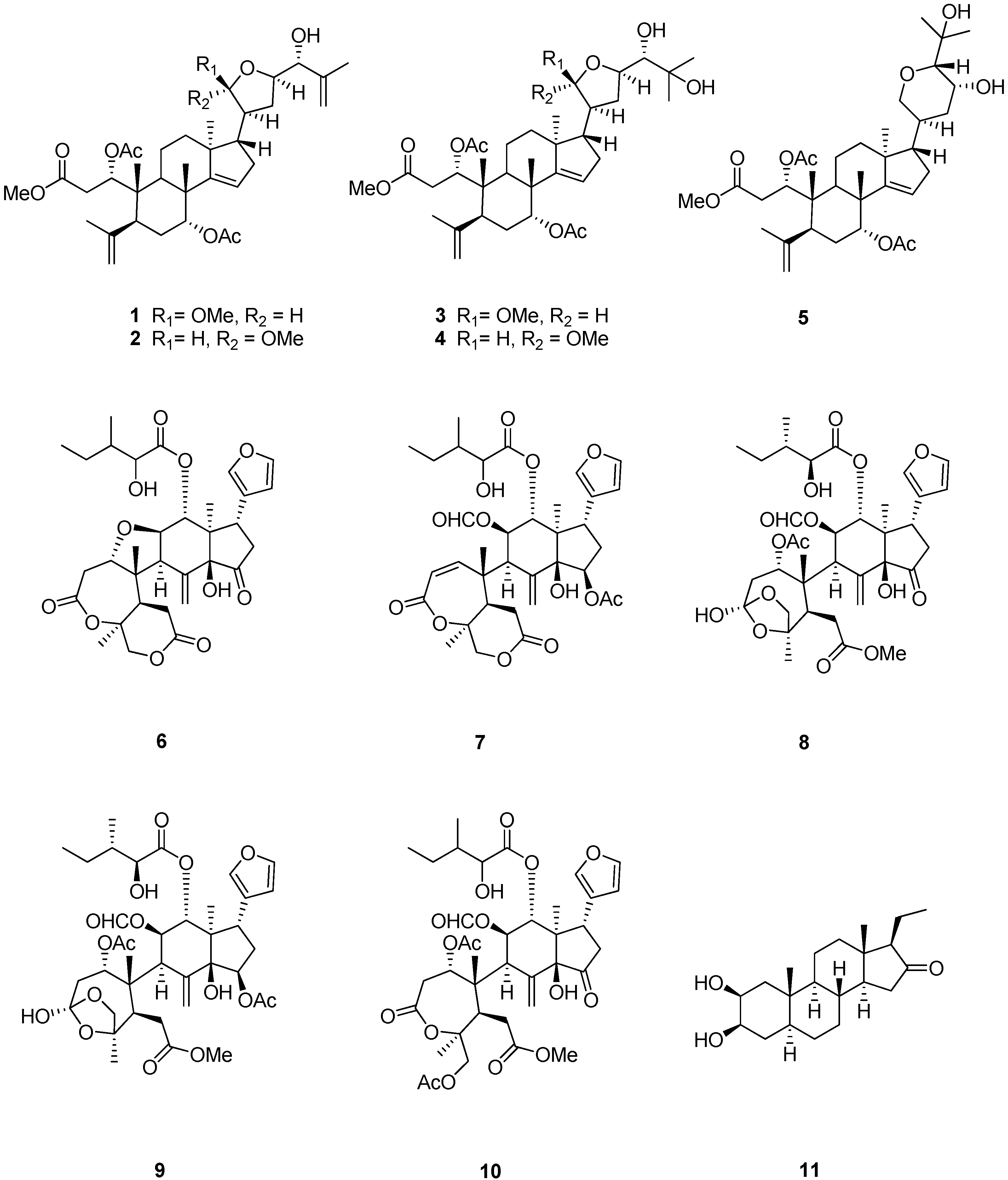
Sephadex LH-20 and extensive column chromatography furnished four new apotirucallane-type triterpenoids 1–4 (Figure 1), together with a protolimonoid, methyl-1ξ,7R-diacetoxy-23R,25-dihydroxy-20S,24R-21,24-epoxy-3,4-seco-apotirucall-4(28),14(15)-diene-3-oate (5) [21]; five limonoids, rohituka 3 (6) [22], rohituka 7 (7) [22], nymania 1 (8) [23], rubrin G (9) [24], prieurianin (10) [25,26]; and one steroid, 2β,3β-dihydroxy-5α-pragnan-16-one (11) [27,28]. The structures of the new compounds were established by interpretation of their spectroscopic data, especially 2D NMR. The relative configurations of compounds 1–4 were determined by NOESY and comparison of NMR data with those published in reference papers.
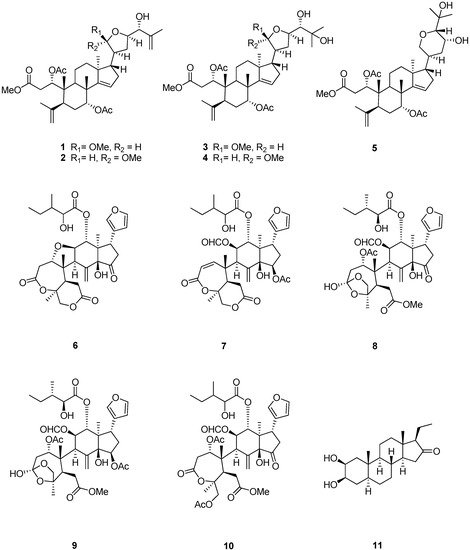
Figure 1.
Chemical structures of compounds 1–11.
Compound 1, −60 (MeOH), had the molecular formula of C36H54O9 and 10 degrees of unsaturation, as deduced from the HRESIMS (m/z 653.3684 [M + Na]+) and 13C-NMR/DEPT spectra. The IR absorption bands revealed the presence of hydroxy (3479 cm−1), ester (1740 cm−1), and double bond (1645 cm−1) functionalities in 1. The 1H-NMR data of 1 (Table 1) exhibited seven methyl singlets (δH 0.96, 1.00, 1.14, 1.78, 1.79, 1.96 and 2.04), two methoxy singlets (δH 3.39, 3.66), exomethylene (δH 4.85, 4.93, 5.02 and 5.05), an olefinic methine doublet (δH 5.27, J = 3.0 Hz), and five oxygen-bearing methine signals (δH 3.83, 4.23, 4.78, 5.16 and 5.49). The 13C-NMR (Table 2) and DEPT spectra of 1 showed 36 carbon signals, consisting of three ester carbonyls (δC 170.2, 170.5, and 172.0), two olefinic methylenes (δC 112.8, 116.6), an olefinic methine (δC 119.1), three olefinic quaternary carbons (δC 144.9, 145.1, and 159.2), five oxymethines (δC 74.7, 77.2, 78.1, 80.7 and 104.6), four aliphatic methines (δC 34.5, 44.3, 45.8, and 52.9), six aliphatic methylenes (δC 18.5, 29.3, 31.0, 33.5, 35.2, and 35.5), two methoxyls (δC 52.3, 54.9), and seven methyls (δC 15.2, 18.7, 20.8, 21.2, 21.5, 23.0, and 26.9). This accounted for 6 of the 10 degrees of unsaturation, indicating that 1 is a tetracyclic triterpenoid with two acetyl and two methoxyl moieties.
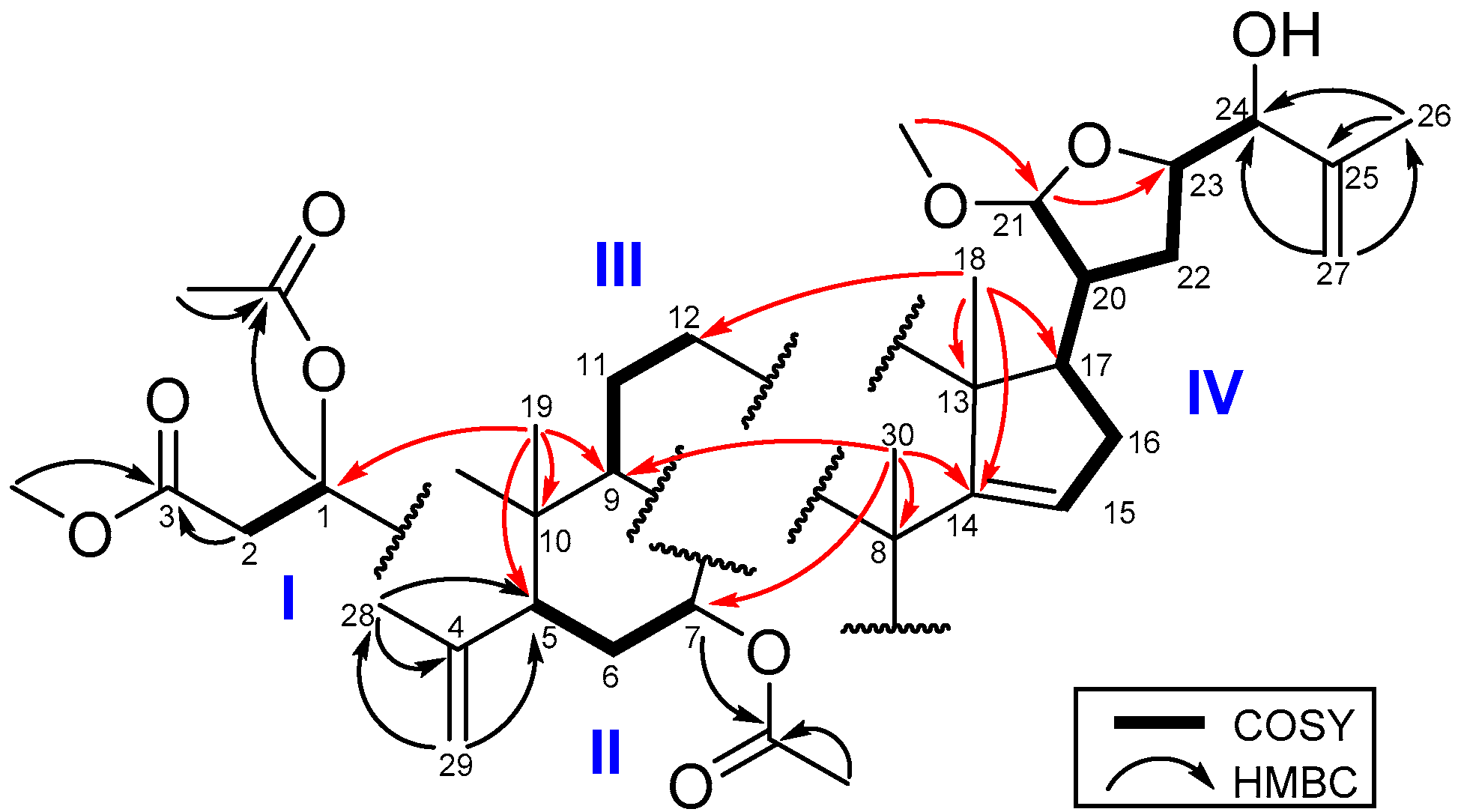
The COSY spectrum (Figure 2) of 1 exhibited four proton spin systems of H-1 (δH 5.49)/H2-2 (δH 2.47, 2.81) and H-5 (δH 2.44)/H2-6 (δH 1.63, 2.14)/H-7 (δH 5.16); H-9 (δH 2.18)/H2-11 (δH 1.67, and 1.89)/H2-12 (δH 1.54, and 1.65); and a proton sequence between H-15 (δH 5.27)/H2-16 (δH 1.93, 2.10)/H-17 (δH 1.92)/H-20 (δH 2.21)/H-21 (δH 4.78) and between H-20/H2-22 (δH 1.70, 1.87)/H-23 (δH 4.23) /H-24 (δH 3.83). In the HMBC spectrum (Figure 2), the H3-19 signal showed correlations to C-1 (δC 77.2), C-5 (δC 44.3), C-9 (δC 34.5) and C-10 (δC 44.3), and the H2-29 had correlations with C-4 (δC 145.1), C-5 and C-28 (δC 23.0) suggesting the ring B. In addition, the HMBC correlations of H3-18 (δH 1.00) with C-13 (δC 46.3), C-12 (δC 33.5), C-14 (δC 159.2), C-17 (δC 52.9), and of H3-30 (δH 1.14) with C-7 (δC 74.7), C-8 (δC 42.5), C-9 (δC 34.5) and C-14 (δC 159.2) established rings C and D. Moreover, the HMBC correlations of both H3-26 (δH 1.78) and H2-27 (δH 4.93, 5.05) with C-24 (δC 78.1) and C-25 (δC 144.9) indicated a methyl vinyl group to be attached at the oxygenated C-24. The HMBC correlations of the methoxy protons (δC 3.39) with C-21 (δC 104.6) and of H-21 (δH 4.78) with C-23 (δC 80.7) revealed the tetrahydrofuran ring with a methoxy group at C-21. The remaining methoxy (δH 3.36) group was found to be connected with C-3 (δC 172.0), and the H-1 (δH 5.49) and H-7 (δH 5.16) were connected with the acetyl carbonyl carbons (δC 170.5, 170.2) indicated that the acetyl groups were attached at C-1 and C-7, respectively. This 2D NMR spectroscopic analysis was used to identify 1 as an A-seco apo-tirucallane triterpenoid [15].
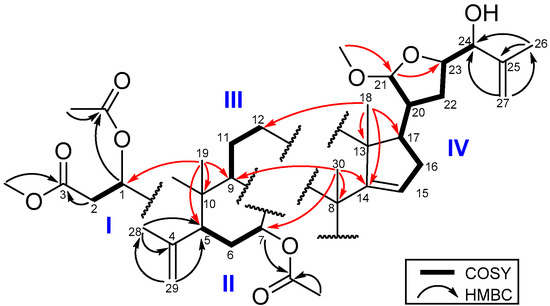
Figure 2.
COSY and HMBC of 1.
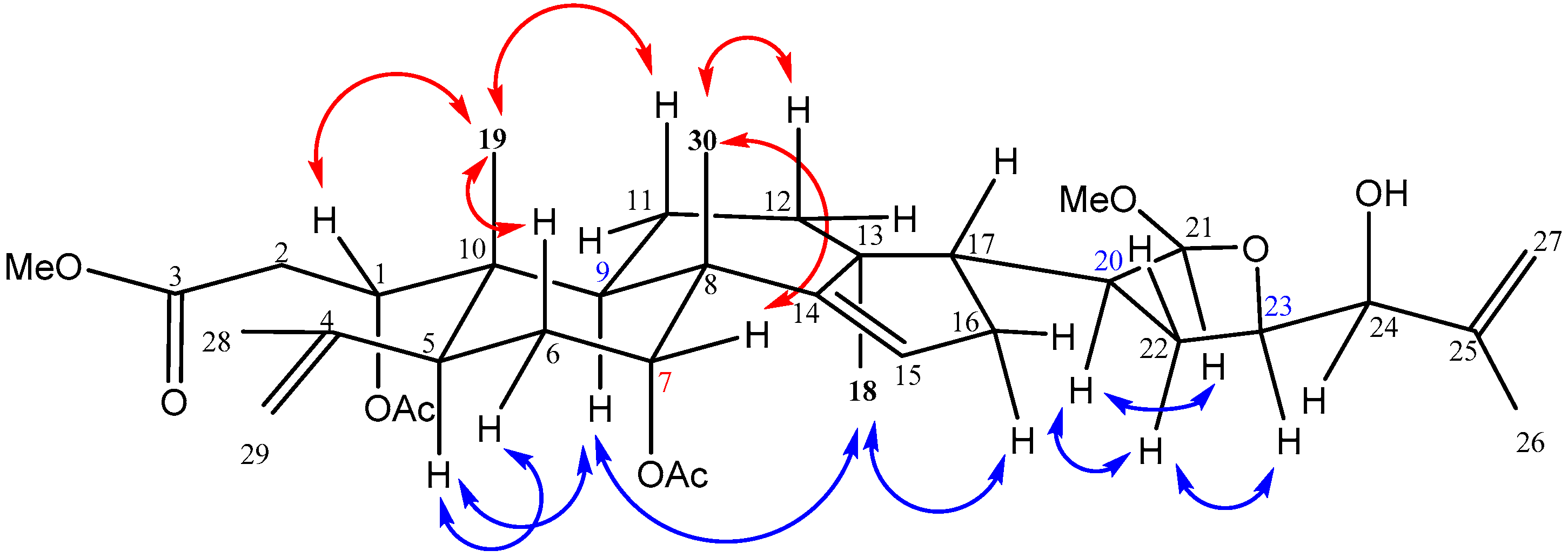
The relative configuration of 1 was determined by the analysis of NOESY correlations (Figure 3). Assuming that H-5 of 1 was α-oriented similar to that of the A-seco apotirucallane tetranortriterpenes [15,29], the NOESY correlations between H-6α/H-5/H-9/H3-18/H-16α revealed that these protons were on the #x3B1;-face. The NOESY correlations of H3-19 with H-1, H-6β and H-11β, as well as Me-30 with H-7 indicating these protons were on the β-face. In addition, NOESY correlations of H-20/H-21/H-22α/H-23 suggested that these protons were all in α-face and the C-21 methoxy was β-oriented. On comparing the 1H- and 13C-NMR spectra of 1 with those of chisopanin C [30] and polystanins C and D [31], it was noted that the configurations of C-20, C-21 and C-23 were assigned S, S and R, respectively, the same as those of polystanin D [31], and the hydroxy at C-24 was assigned as α-disposition, the same as that of chisopanin C [30]. Therefore, structure 1 was established and a name aphataiwanin A was given.
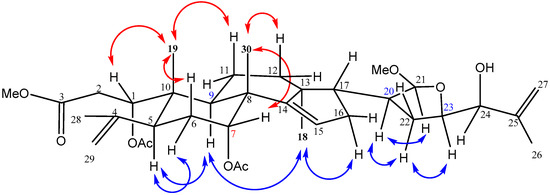
Figure 3.
Key NOESY correlations of 1.
Compound 2, −90 (MeOH), had the same molecular formula C36H54O9 as 1, as deduced from HRESIMS (m/z 653.3681 [M + Na]+) and DEPT spectra. The IR absorption bands revealed the presence of OH (3484 cm−1), ester (1740 cm−1) and double bond (1643 cm−1) in 2. The 1D and 2D NMR spectra of 2 were similar to those of 1, suggesting that 2 was an analogue of 1. On comparing the 13C-NMR spectra of 1 and 2, it was found that the C-21 and C-17 chemical shifts of 2 (δc 109.3, 58.1) were downfield compared to the same carbons of 1 (δ 104.6, 52.9). Thus it was suggested that the configuration of the C-21 methoxy group in 2 was different from that in 1. Also, both the COSY and HMBC correlations showed that all structural fragments were similar to those of 1, confirming that compound 2 is an epimer of 1, in which the C-21 methoxy was α-oriented.
The relative configuration of 2 was determined by NOESY experiment, in which the H3-19 and H3-30 were assigned to be β-oriented while H3-18, H-9, and H-5 were α-orientation. NOESY correlations between H-21/H-22β and H-20/H-23, and H-23/H-22β indicated that the methoxy group was α-oriented. Comparing the 13C-NMR data with those of chisopanin C [30] assigned the configuration of C-13, C-17, C-20, C-23 and C-24 as S, S, S, R, R and R, respectively. Therefore, the structure of compound 2 was determined and it was named aphataiwanin B.
Compound 3, −60 (MeOH), was obtained as an amorphous solid and found to possess the molecular formula C36H56O10, (one more oxygen atom than 1) as inferred from its HRESIMS (m/z 671.3785 [M + Na]+). The similar 1H- and 13C-NMR spectroscopic data of 3 and 1 suggested that they are close analogues. However, the 1H-NMR spectrum of 3 exhibited eight methyl singlets (δH 0.95, 1.00, 1.14, 1.26, 1.27, 1.78, 1.96 and 2.04) instead of seven in 1 and only three olefinic protons (δH 4.84, 5.02 and 5.27) instead of five in 1, implying that a double bond was missing in 3. In the 13C-NMR spectrum, compound 3 was found possessing an additional methyl (δC 26.5) and lacking an olefinic methylene, in comparison to 1. The additional methyl group was assigned at C-27 by observation of the HMBC correlations from H3-27 (δH 1.26) and H3-26 (δH 1.27) to C-25 (δC 73.1) and C-24 (δC 76.8). The relative configuration of 3 was determined on the basis of the NOESY experiment and comparing the J values of 3 with those of 1. The result was identical to 1, suggesting the same configuration. On the basis of above interpretations, the structure of 3 was categorized into the group of ring A-seco apotirucallol and a name aphataiwanin C was given.
Compound 4 was isolated as an amorphous solid, −170 (MeOH). It had the same molecular formula C36H56O10 as 3, as derived from HRESIMS at m/z 671.3763 ([M + Na]+). The IR spectrum revealed that 4 contained a hydroxyl (3491 cm−1), ester (1741 cm−1), and double bond (1642 cm−1) functionalities. The 1H- and 13C-NMR spectra of 4 (Table 1 and Table 2) were similar to those of 3 except for the hemiacetal carbon shifted downfield to δC 109.7 (C-21) in 3 and the oxygenated carbon C-23 shifted upfield to δC 76.9. The 1H-1H COSY and HMBC of 4 revealed cross peaks similar to those of 3, suggesting that compound 4 was an epimer of 3. The configuration at C-21 was determined by NOESY experiment. NOESY correlations between H-21/H-22β, H-20/H-22α and H-22α/H-23 indicated that the methoxy group was α-oriented. Other NOESY correlations were the same as those of 3. Thus, the structure of compound 4 was elucidated and the name aphataiwanin D was given.
The 11 isolates were evaluated for their cytotoxic activities against human hepatocellular carcinoma (Hep-G2), lung carcinoma (A549), epithelial type 2 (HEp-2), and breast adenocarcinoma (MCF-7) cell lines in vitro. Among these compounds, 5 possessed significant activity (ED50 value of 6.83 ± 0.63 μg/mL) against MCF-7 cells and mild activity against Hep-G2 and A549 cells (ED50 value of 11.38 ± 0.98 and 15.49 ± 0.76 μg/mL). Simultaneously, compounds 1 and 10 also showed mild activities against Hep-G2 and HEp-2, respectively, with IC50 values of 16–17 μg/mL (Table 3). In terms of anti-inflammation, compounds 1–11 were tested on superoxide anion generation and elastase release by human neutrophils in the presence of FMLP/CB (Table 4). Only compounds 4 and 5 showed significant anti-inflammatory activity, as tested on superoxide anion generation with IC50 at 5.79 ± 0.88 and 1.25 ± 0.17 μg/mL, as well as the significant inhibition on elastase release with IC50 at 5.22 ± 0.24 and 2.26 ± 0.05 μg/mL, respectively.

Table 3.
Cytotoxic activities of compounds 1–11 a.

Table 4.
Anti-inflammatory data of compounds 1–11 a.
6. Conclusions
In the present study, 10 tetranortriterpenoids, including four new compounds and a steroid, were isolated; compounds 1–5 belong to a group of protolimonoids, ring A-seco apotirucallol, while 6–10 can be classified as rings A,B-seco prieurianins. Among these secondary metabolites, liaphataiwanins A (1) and D (4), 5, and prieurianin (10) were found to have mild cytotoxicities against cancer cells and moderate to potent anti-inflammatory activities. Compound 5, which has the moiety of six-member ether ring, showed the best biological function. The chemical constituents and the evaluation of cytotoxicities against human cancer cells and anti-inflammatory activities reported herein may provide beneficial information for further phytotherapy research.
Acknowledgments
The authors thank the Ministry of Science and Technology, Republic of China (NSC 98-2320-B002-027-MY3) for financial support.
Author Contributions
Ching-Jie Lin performed the experiments. I-Wen Lo and Yu-Chi Lin contributed to manuscript preparation and analysis of data. Shun-Ying Chen and Ching-Te Chien contributed material collection. Yao-Haur Kuo, Tsong-Long Hwang and Shorong-Shii Liou performed the biological assays. Ya-Ching Shen designed the experiment and wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ascher, K.R.S. Nonconventional insecticidal effects of pesticides available from the Neem tree, Azadirachta indica. Arch. Insect Biochem. Physiol. 1993, 22, 433–449. [Google Scholar] [CrossRef]
- Tan, Q.G.; Luo, X.D. Meliaceous limonoids: Chemistry and biological activities. Chem. Rev. 2011, 111, 7437–7522. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.A.; Taylor, D.A.H. Limonoid extractives from Aphanamixis polystachya. Phytochemistry 1978, 17, 1995–1999. [Google Scholar] [CrossRef]
- Kundu, A.B.; Ray, S.; Chatterjee, A. Aphananin, a triterpene from Aphanamixis polystachya. Phytochemistry 1985, 24, 2123–2125. [Google Scholar] [CrossRef]
- Mulholland, D.A.; Naidoo, N. Limonoids from Aphanamixis polystachya. Phytochemistry 1999, 51, 927–930. [Google Scholar] [CrossRef]
- Zhang, H.P.; Wu, S.H.; Luo, X.D.; Ma, Y.B.; Wu, D.G. A new limonoid from the seeds of Aphanamixis polystachya. Chin. Chem. Lett. 2002, 13, 341–342. [Google Scholar]
- Zhang, Y.; Wang, J.S.; Wei, D.D.; Wang, X.B.; Luo, J.; Luo, J.G.; Kong, L.Y. Cytotoxic tirucallane C26 triterpenoids from the stem barks of Aphanamixis polystachya. Phytochemistry 2010, 71, 2199–2204. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Hasan, C.M.; Rashid, M.A. Guaiane sesquiterpenes from Amoora rohituka. Phytochemistry 2003, 62, 1213–1216. [Google Scholar] [CrossRef]
- Sadhu, S.K.; Phattanawasin, P.; Choudhuri, M.S.K.; Ohtsuki, T.; Ishibashi, M. A new lignan from Aphanamixis polystachya. J. Nat. Med. 2006, 60, 258–260. [Google Scholar] [CrossRef]
- Jain, S.A.; Srivastava, S.K. 8-C-Methyl-quercetin-3-O-β-d-xylopyranoside, a new flavone glycoside from the roots of Amoora rohituka. J. Nat. Prod. 1985, 48, 299–301. [Google Scholar] [CrossRef]
- Ghani, A. Medicinal Plants of Bangladesh with Chemical Constituents and Uses, 2nd ed.; Asiatic Society of Bangladesh: Dhaka, Bangladesh, 2003. [Google Scholar]
- Hossain, M.M.; Biva, I.J.; Jahangir, R.; Vhuiyan, M.M.I. Central nervous system depressant and analgesic activity of Aphanamixis polystachya (Wall.) parker leaf extract in mice. Afr. J. Pharm. Pharmacol. 2009, 3, 282–286. [Google Scholar]
- Apu, A.S.; Pathan, A.H.; Jamaluddin, A.T.M.; Ara, F.; Bhuyan, S.H.; Islam, M.R. Phytochemical analysis and bioactivities of Aphanamixis polystachya (Wall.) R. Parker leaves from Bangladesh. J. Biol. Sci. 2013, 13, 393–399. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.S.; Wang, X.B.; Wei, D.D.; Luo, J.G.; Luo, J.; Yang, M.H.; Kong, L.Y. Aphanapolynins A and B, two new limonoids from the fruits of Aphanamixis polystachya. Tetrahedron Lett. 2011, 52, 2590–2593. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.S.; Wang, X.B.; Gu, Y.C.; Wei, D.D.; Guo, C.; Yang, M.H.; Kong, L.Y. Limonoids from the fruits of Aphanamixis polystachya (Meliaceae) and their biological activities. J. Agric. Food Chem. 2013, 61, 2171–2182. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.Y.; Zhang, Y.; Luo, S.H.; Chen, D.Z.; Tang, G.H.; Yuan, C.M.; Di, Y.T.; Li, S.H.; Hao, X.J.; He, H.P. Aphanamixoid A, a potent defensive limonoid, with a new carbon skeleton from Aphanamixis polystachya. Org. Lett. 2012, 14, 2524–2527. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.Y.; Chen, D.Z.; Luo, S.H.; Kong, N.C.; Zhang, Y.; Di, Y.T.; Zhang, Q.; Hua, J.; Jing, S.X.; Li, S.L.; et al. Limonoids from Aphanamixis polystachya and their antifeedant activity. J. Nat. Prod. 2014, 77, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.C.; Chang, Y.T.; Lin, C.L.; Liaw, C.C.; Kuo, Y.H.; Tu, L.C.; Yeh, S.F.; Chern, J.W. Synthesis of 1-Substituted Carbazolyl-1,2,3,4-Tetrahydro- and Carbazolyl-3,4-Dihydro-β-Carboline Analogs as Potential Antitumor Agents. Mar. Drugs 2011, 9, 256–277. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.C.; Wang, S.S.; Pan, Y.L.; Lo, K. L.; Chakraborty, R.; Chien, C.T.; Kuo, Y.H.; Lin, Y.C. New Taxane Diterpenoids from the Leaves and Twigs of Taxus sumatrana. J. Nat. Prod. 2002, 65, 1848–1852. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.C.; Chung, P.J.; Ho, C.M.; Kuo, C.Y.; Hung, M.F.; Huang, Y.T.; Chang, W.Y.; Chang, Y.W.; Chan, K.H.; Hwang, T.L. Propofol Inhibits Superoxide Production, Elastase Release, and Chemotaxis in Formyl Peptide-Activated Human Neutrophils by Blocking Formyl Peptide Receptor 1. J. Immunol. 2013, 190, 6511–6519. [Google Scholar] [CrossRef] [PubMed]
- Garcez, F.R.; Garcez, W.S.; Rodrigues, E.D.; Pott, B.J.; Roque, N.F. Seco-protolimonoids from Trichilia elegans sp. elegans. Phytochemistry 1996, 42, 1399–1403. [Google Scholar] [CrossRef]
- Zhang, H.P.; Chen, F.; Wang, X.; Wu, D.G.; Chen, Q. Spectral assignments and reference data. Complete assignments of 1H and 13C-NMR data for rings A, B-seco limonoid from the seed of Aphanamixis polystachya. Magn. Reson. Chem. 2007, 45, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Maclachlan, L.K.; Taylor, D.A.H. Limonoids from Nymania capensis. Phytochemistry 1982, 21, 1701–1703. [Google Scholar] [CrossRef]
- Musza, L.L.; Killar, L.M.; Speight, P.; Mcelhiney, S.; Barrow, C.J.; Gillum, A.M.; Cooper, R. Potent new cell adhesion inhibitory compounds from the root of Trichilia rubra. Tetrahedron 1994, 50, 11369–11378. [Google Scholar] [CrossRef]
- Gullo, V.P.; Miura, I.; Nakanishi, K.; Cameron, A.F.; Connolly, J.D.; Duncanson, F.D.; Harding, A.E.; Mccrindle, R.; Taylor, D.A.H. Structure of Prieurianin, a complex tetranortriterpenoid; nuclear magnetic resonance analysis at nonambient temperatures and X-ray structure determination. J. Chem. Soc. Chem. Commun. 1975, 345–346. [Google Scholar] [CrossRef]
- Sarker, S.D.; Savchenko, T.; Whiting, P.; Sik, V.; Dinan, L. Two limonoids from Turraea obtusifolia (Meliaceae), prieurianin and rohitukin, antagonise 20-hydroxyecdysone action in a Drosophila cell line. Arch. Insect Biochem. Physiol. 1997, 35, 211–217. [Google Scholar] [CrossRef]
- Inada, A.; Murata, H.; Inatomi, Y.; Nakanishi, T.; Darnaedi, D. Pregnanes and triterpenoid hydroperoxides from the leaves of Aglaia grandis. Phtochemistry 1997, 45, 1225–1228. [Google Scholar] [CrossRef]
- Pupo, M.T.; Vieira, P.C.; Fernandes, J.B.; Silva, M.F.G.F.; Fo, E.R. Androstane and pregnan 2β,19-hemiketal steroids from Trichilia claussenii. Phytochemistry 1997, 45, 1495–1500. [Google Scholar] [CrossRef]
- Yang, S.P.; Chen, H.D.; Liao, S.G.; Xie, B.J.; Miao, Z.H.; Yue, J.M. Aphanamolide A, a new limonoid from Aphanamixis polystachya. Org. Lett. 2011, 13, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.H.; Wang, J.S.; Luo, J.G.; Wang, X.B.; Kong, L.Y. Chisopanins A–K, 11 New protolimonoids from Chisocheton paniculatus and their anti-inflammatory activities. Bioorg. Med. Chem. 2011, 19, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, J.S.; Wang, X.B.; Gu, Y.C.; Kong, L.Y. Polystanins A–D, four new protolimonoids from the fruits of Aphanamixis polystachya. Chem. Pharm. Bull. 2013, 61, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).