Abstract
The weevil Aegorhinus superciliosus Guérin (Coleoptera: Curculionidae), which is endemic to Central-Southern Chile and Argentina, is one of the major berry pests in Chile and the most important pest in the La Araucanía Region (38°44′9″S, 72°35′25″W). Due to the poor effectiveness and problems surrounding the implementation of the traditional control methods using organophosphate and carbamate insecticides, new strategies for controlling this pest are needed. In this communication, we evaluated the behavioral responses of male and female A. superciliosus to volatile compounds released from the essential oil (EO) obtained from the heartwood of Pilgerodendron uviferum (D. Don) Florin using olfactometric bioassays. The composition of the EO was analyzed using gas chromatography (GC) and gas chromatography/mass spectrometry (GC/MS). According to these analyses, δ-cadinol (24.16%), cubenol (22.64%), 15-copaenol (15.46%) and δ-cadinene (10.81%) were the principal components of the EO. The Pilgerodendron uviferum EO, which is almost exclusively composed of sesquiterpenes (99.5%), exhibited a repellent effect against A. superciliosus adults, regardless of the sex or concentration used (56.6 mg/cm3 and 1.58 × 10−2 mg/cm3). The EO has low volatility and greater persistence than the EOs composed of monoterpenes and is considered a good model in the search for raspberry weevil repellents.
1. Introduction
Aegorhinus superciliosus Guérin (Coleoptera: Curculionidae), commonly known as the raspberry weevil, is endemic to Central-Southern Chile and Neuquén, Río Negro, Argentina. A. superciliosus is associated with deciduous forests and Valdivian temperate forests from the Maule to Los Lagos Regions (41°55′11″S, 72°8′29″W) [1] and is the most important pest in the La Araucanía Region because they feed on valuable berries including raspberries, blueberries and strawberries [2]. The larvae bore into the lateral roots with their powerful jaws, finally boring into the primary roots to build chambers to pupate, which may cause the death of the hosts. After pupal eclosion, the emerged adults cause damage to leaves, buds and fruits [3,4,5].
Only two parasitoids of A. superciliosus have been found in Chile—Centistes spp., a parasitoid wasp for the larvae, and a tachinid (Diptera), a parasitoid of adults—and they are unable to control the pest populations. Therefore, insecticide treatment is the main control strategy, followed to a lesser extent by biological, mechanical-physical and cultural control. Chemical control has limited effectiveness in adult insects, and it is ineffective against the larvae that populate the roots. Organophosphate and carbamate insecticides have been used to control this pest; however, these kind of compounds have negative effects on the environment [6,7], such as a long residual effect, exclusion period and toxicity to pollinators when applied close to the flowering period. The entomopathogenic fungi Beauveria and Metarhizium (Moniliales: Moniliaceae) have been used as biological treatments. These fungi are harmless to plants and animals [8], but so far, their use has not spread. Another control method investigated in recent years is the behavioral control of weevils using semiochemicals, reorted by Parra et al. [9] and Mutis et al. [5]. This promising line of work is currently being investigated.
The low efficiency and problems arising from the application of current control methods determined the urgent need for new strategies for controlling A. superciliosus. In this scenario, the use of natural products with properties that modify the behavior of A. superciliosus, such as essential oils (EOs), is viewed as an important alternative [6]. EOs have a wide spectrum of biological activities against insects: they can act as insecticides [10], repellents, antifeedants, growth regulators and oviposition deterrents [11]. Additionally, EOs and their constituents are not toxic and qualify as “low-risk pesticides” [6,12].
The examination of the literature on the repellent activity of EOs against Coleoptera species shows that the principal components of the active oils tend to be monoterpenoids, and little is known about the repellent activity of sesquiterpene-rich essential oils [13,14]. In this communication we report the behavioral response of A. superciliosus males and females to volatile compounds released from Pilgerodendron uviferum (D. Don) Florin (Cupressaceae) EO, which is almost exclusively composed of sesquiterpenes [15].
Pilgerodendron uviferum (D. Don) Florin (Cupressaceae) is a tree native to southern Chile and Argentina. Its wood is resistant to degradation, and it has been postulated that its resistance to microorganisms and insects is related to its chemical composition [15,16]. In this communication, we report the composition of the EO obtained by hydrodistillation from the fresh heartwood of P. uviferum, which was determined using gas chromatography (GC) and gas chromatography/mass spectrometry (GC/MS), and the behavioral responses of male and female A. superciliosus to the P. uviferum EO at two different concentrations using a four-arm olfactometer.
2. Results and Discussion
After fresh milling of P. uviferum heartwood (2.0 kg), 26.31 g of EO was obtained (1.4%). The EO was analyzed by GC/MS (Table 1). Twenty compounds were identified in the EO, corresponding to 87.05% of all detected compound. Three monoterpenes (0.43%)—two non-oxygenated (0.36%) and one oxygenated (0.06%)—and 17 sesquiterpenes (86.62%)—12 non-oxygenated and five oxygenated—were present in the EO. The sesquiterpenes δ-cadinene (10.81%), δ-cadinol (24.16%), cubenol (22.64%) and 15-copaenol (15.46%) were the most abundant compounds in the EO. Identification was corroborated by studying the fragmentation patterns in the obtained mass spectra. Although the relative abundance of the monoterpenes is low, this is the first report of the presence of monoterpenes in the EO from P. uviferum heartwood, highlighting the usefulness and advantages of GC/MS for studying volatile compounds compared to other extractives techniques that involved successive evaporations.

Table 1.
Composition of the essential oil from P. uviferum heartwood.
The sesquiterpene nature of the heartwood EO agrees with the report of Oyarzún and Garbarino [17], who isolated the sesquiterpenes (−)-trans-calamenene, (+)-δ-cadinene, (−)-caryophyllene-4,5-epoxide, (−)-humulene-1,2-epoxide, (−)-cubenol, (−)-epi-cubenol, (−)-torreyol and (−)-15-copaenol from the heartwood of P. uviferum collected in Chile. However, the composition of the EO from the heartwood of P. uviferum that we studied is very different from the composition of the EO from the leaves of P. uviferum collected in Argentina reported by Malizia et al. [18], which was rich in monoterpenes (54.1%), although with a large presence of sesquiterpenes (40.4%).
The olfactometric response of A. superciliosus adults toward the EO from P. uviferum was evaluated. The average time spent by individuals in the control zone was significantly higher (12.5 ± 1.3 min) than in the stimulus zone corresponding to diluted EO (1.58 × 10−2 mg/cm3) (6.9 ± 1.3 min) (p = 0.044) and was very significantly higher than in the decision zone (0.5 ± 0.1 min) (p < 0.0001). Compared with the pure EO (56.6 mg/cm3), the average time spent by individuals in the control zone was higher (12.1 ± 1.6 min) than in the stimulus zone (7.2 ± 1.6 min) and was significantly higher than in the decision zone (0.7 ± 0.4 min) (p < 0.0001). No differences were observed in the insect repellent response toward pure or diluted EO, although the dilute concentration was approximately 3500 times lower. Insects exposed to the pure EO sample decreased their movement and groomed their antennae continuously with their front legs. No signs of toxicity were observed. Therefore, the EO from the heartwood of P. uviferum repelled A. superciliosus adults.
Additionally, the olfactometric responses of male and female A. superciliosus toward the EO from the heartwood of P. uviferum were evaluated. The tendency of the insects to stay in the control zone longer than in the stimulus zone or decision zone remained (Figure 1). The insect responses were independent of sex and EO concentration (Figure 1). The EO from the heartwood of P. uviferum repelled both male and female A. superciliosus.
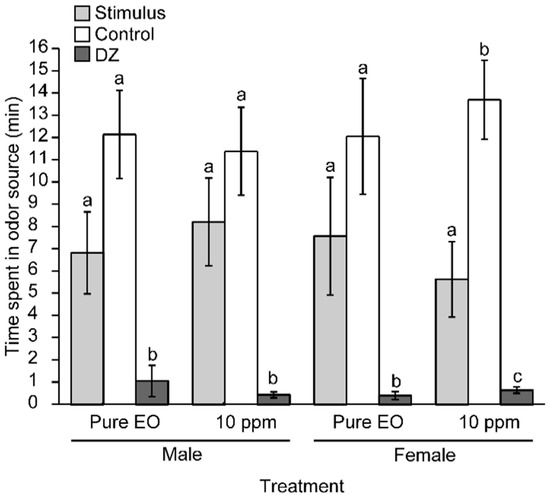
Figure 1.
Average time spent (min ± SE) by A. superciliosus in the olfactometer arms containing pure EO and diluted P. uviferum heartwood EO (2 µL and 50 µL of 10 mg/L EO, i.e., 56.6 mg/cm3 and 1.58 × 10−2 mg/cm3, respectively). Control: purified air, DZ: decision zone. The lines on the bars indicate the standard error. Different letters indicate significant differences based on the non-parametric Friedman test followed by the Conover-Inman test (p ≤ 0.05) N = 24.
The behavior of A. superciliosus exposed to P. uviferum EO follows. In general, insects walked around the four arms of the olfactometer during the first half of the bioassay. Then, individuals chose and remained in one of the control arms until the end of the bioassay. No abdominal movements alluding to mating nor exposition of the aedeagus from male weevils were observed [19].
This is the first report of the behavioral response of male and female A. superciliosus to volatile compounds released from P. uviferum EO, which is almost exclusively composed of sesquiterpenes.
Among the few EOs rich in sesquiterpenoids that have been tested on Coleptera, we will mention the following: the EOs from the leaves and flowers of Teucrium polium, Eupatorium inulaefolium and Eupatorium arnotti that contain sesquiterpene-rich EOs show repellent and fumigant toxicity properties against the stored-product Tribolium castaneum [20,21]. Additionally, the EO from Zanthoxylum dissitum roots exhibited moderate contact toxicity against tree species of stored-product insects, including Lasioderma serricorne adults, Tribolium castaneum adults and Attagenus piceus larvae [22]. The EO from Z. dissitum leaves showed lower contact toxicity than the root EO. The major components in the EOs from Z. dissitum roots and leaves were humulene epoxide, caryophyllene oxide, δ-cadinol and caryophyllene. All of them are present in the EO from P. uviferum. The EO from Chrysanthemum macrotum caused significant mortality in larvae, adults and nymphs of Tribolium confusum by contact fumigant toxicity. The EO is rich in sesquiterpenes with cubenol, T-cadinol, α-cadinol and α-humulene being the major constituents. Cubenol is one of the major components in P. uviferum EO. Additionally, the EO from Zanthoxylum dissitum roots exhibited moderate contact toxicity against 3 species of storage pests, i.e., Lasioderma serricorne adults, Tribolium castaneum adults and Attagenus piceus larvae, with LD50 values of 13.8, 43.7 and 96.8 μg/adult, respectively. The major components of Z. dissitum roots and leaves were humulene epoxide II (29.4%), caryophyllene oxide (24.0%), δ-cadinol (12.8%) and caryophyllene (12.7%) [22]. All of them are present in the EO of P. uviferum.
Our results are in agreement with the few reports on EOs, and some of the sesquiterpenes present in P. uviferum EO are also present in EOs that shown repellency and toxicity toward Coleoptera. Essential oils containing sesquiterpenes are less volatile than EOs containing monoterpenes because their constituents have lower vapor pressures [23,24]. Sesquiterpenes have average boiling points 100 °C higher than monoterpenes [25]. The resulting longer action and lower associated cost is promising for their application as biocides [26]. In addition, insects have difficulty building up resistance because EOs are complex mixtures of many compounds [27,28].
3. Experimental Section
3.1. Insects
The colonies of A. superciliosus used in this study were hand collected in the same phenological state at an experimental blueberry plantation in Collipulli, La Araucanía Region (37°57′29″S, 72°25′55″W), Chile, in November 2014. The individual weevils were transferred to a small cage and then reared in entomological cages (30 cm × 30 cm × 30 cm) at 26 ± 1 °C under a 16:8 light:dark cycle and 70% humidity. The adults were provided with fresh blueberry twigs and leaves.
3.2. Plant Material
P. uviferum heartwood was collected in February 2012 in the south of the Baker River Basin (47°1′23″S, 72°49′46″W), Aysén Region, Chile. Voucher specimens were deposited at Laboratorio de Química de la Madera, Facultad de Ciencias Forestales y de la Conservación de la Naturaleza, Universidad de Chile. The samples were identified by forestry engineer René Carmona.
3.3. Essential Oil Extraction and Analysis by GC/MS
The EO was extracted from 2 kg of milled P. uviferum heartwood by hydrodistillation for 6 h in a Clevenger-type apparatus. The EO was dried over anhydrous sodium sulfate. Analysis of the EO components was performed by gas chromatography and gas chromatography/mass spectrometry (GC/MS) using the instrumentation described below. Qualitative analysis was performed using a Thermo Scientific Trace GC Ultra linked to an ISQ quadrupole mass spectrometric detector with an integrated data system (Xcalibur 2.0, Thermo Fisher Scientific Inc., Waltham, MA, USA); quantitative analysis was carried out using a Shimadzu GC-9A gas chromatograph fitted with an FID-9 detector (Shimadzu Corporation, Kyoto, Japan). The same capillary column (Rtx-5 MS, film thickness 0.25 μm, 60 m × 0.25 mm, Restek Corporation, Bellefonte, PA, USA) was used in both instruments. The operating conditions were as follows: On-column injection; injector temperature, 250 °C; detector temperature, 280 °C; carrier gas, He at 1.25 mL/min; oven temperature program: 40 °C increase to 260 °C at 4 °C/min, and then 260 °C for 5 min. The mass spectra were obtained at an ionization voltage of 70 eV. Recording conditions employed a scan time of 1.5 s and a mass range of 40 to 400 amu. The identification of compounds in the chromatographic profiles was achieved by comparison of their mass spectra with a library database (NIST08, NIST, Gaithersburg, MD, USA) and by comparison of their calculated retention indices with those reported in the literature [29] for the same type of column.
3.4. Olfactometer Bioassays
The behavioral responses of male and female A. superciliosus to the P. uviferum EO at two different concentrations were tested using a four-arm olfactometer [9,30]. The olfactometer consists of three acrylic plates held together with four metal screws; the top plate has a hole in the center to introduce the insect, which is subsequently connected to a vacuum pump adapter. One hole in each of the four arms is connected to a glass tube containing filter paper impregnated with the odor stimulus or the control (See Supplementary Figure S1). The olfactometric assay was conducted according to the procedure of Tapia et al. [31], which consists of observing the movements of the insect in an area called the arena. The arena was divided into five zones: a central square zone called the decision zone and four zones corresponding to the olfactometer arms. Two of the arms were enriched with air containing the volatile EO as the stimuli, and as control zones, the other two arms contained dichloromethane (HPLC grade, Sigma Aldrich, Steinheim, Germany) if the EO sample was diluted or purified air if the EO sample was pure. The samples were prepared by applying either 2 μL of pure P. uviferum EO or 50 μL of the EO diluted in dichloromethane (10 mg/L) on Whatman no. 1 filter paper (0.5 cm wide by 3.5 cm long). The filter papers, which were impregnated with stimuli, were placed in glass tubes (7 cm long; 1.5 cm i.d.) in different arms of the olfactometer. The insects were placed in the center of the olfactometer, and a purified air flow (800 mL/min) was generated to carry the volatile stimuli from the arms to a central hole in the olfactometer. The air was purified through a column of activated carbon. The bioassay was performed during the morning, according to the observed period of more activity of A. superciliosus. The olfactometric response of each insect to the stimulus was registered for 20 min, and the time spent in each arm was processed using EthoVision 3.1 software [32]. Each treatment was replicated 24 times for each sex, first with the males and then with the females. A different individual was used in each replicate of the experiments and then was discarded. After each replicate the olfactometer was cleaned with dichloromethane, and dried for 20 min at 55 °C in a forced-air oven.
3.5. Data Analyses
The times spent by A. superciliosus in the stimulus and control zones of the olfactometer were compared through the non-parametric Friedman test (p ≤ 0.05) followed by the Conover-Inman test (p ≤ 0.05) using StatsDirect 3.1 [33] statistics software.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/21/4/533/s1.
Acknowledgments
The authors would like to acknowledge CONICYT-PCHA scholarship No 2012-21120365 awarded to JE and Grant, which provided support for this research at Universidad de Santiago de Chile, Proyectos Basales (Vicerrectoría de Investigación, Desarrollo e Innovación Usach).
Author Contributions
A.U., J.E. and A.Q. conceived and designed the experiments; J.E. performed the experiments; A.U. and J.E. analyzed the chemical data; J.E., L.P. and J.T. analyzed the behavioral and statistical data; A.U. and J.E. wrote the paper; all authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zavala, A.; Elgueta, M.; Abarzúa, J.; Aguilera, A.; Quiroz, A.; Rebolledo, R. Diversity and distribution of the Aegorhinus genus in the La Araucanía Region of chile, with special reference to A. superciliosus and A. nodipennis. Cienc. Investig. Agrar. 2011, 38, 367–377. [Google Scholar] [CrossRef]
- Medel, V.; Palma, R.; Mercado, D.; Rebolledo, R.; Quiroz, A.; Mutis, A. The effect of protease inhibitors on digestive proteolytic activity in the Raspberry Weevil, Aegorhinus superciliosus (Guerin) (coleoptera: Curculionidae). Neotrop. Entomol. 2015, 44, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, P.A.; Zampezzi, V.M.; Araneda, D.X.; Klein, K.C.; Rebolledo, R.R. Efectividad de la azadirachtina en la inhibición de la embriogénesis de Aegorhinus superciliosus (Guérin) (Coleoptera: Curculionidae). IDESIA 2009, 27, 47–55. [Google Scholar] [CrossRef]
- Parra, L.; Mutis, A.; Aguilera, A.; Rebolledo, A.; Quiroz, A. Knowledge of the “Cabrito del frambueso” weevil (CF) Aegorhinus superciliosus (Guérin) (Coleoptera: Curculionidae). IDESIA 2009, 27, 57–65. [Google Scholar] [CrossRef]
- Mutis, A.; Parra, L.; Manosalva, L.; Palma, R.; Candia, O.; Lizama, M.; Pardo, F.; Perich, F.; Quiroz, A. Electroantennographic and behavioral responses of adults of raspberry weevil Aegorhinus superciliosus (Coleoptera: Curculionidae) to odors released from conspecific females. Environ. Entomol. 2010, 39, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Adeyemi, M.H. The potential of secondary metabolites in plant material as deterents against insect pests: A review. Afr. J. Pure Appl. Chem. 2010, 4, 243–246. [Google Scholar]
- Kim, S.-I.; Yoon, J.-S.; Jung, J.W.; Hong, K.-B.; Ahn, Y.-J.; Kwon, H.W. Toxicity and repellency of origanum essential oil and its components against Tribolium castaneum (Coleoptera: Tenebrionidae) adults. J. Asia Pac. Entomol. 2010, 13, 369–373. [Google Scholar] [CrossRef]
- Yáñez, M.; France, A. Effects of fungicides on the development of the entomopathogenic fungus Metarhizium anisopliae var. Anisopliae. Chil. J. Agric. Res. 2010, 70, 390–398. [Google Scholar]
- Parra, L.; Mutis, A.; Ceballos, R.; Lizama, M.; Pardo, F.; Perich, F.; Quiroz, A. Volatiles released from Vaccinium corymbosum were attractive to Aegorhinus superciliosus (Coleoptera: Curculionidae) in an olfactometric bioassay. Environ. Entomol. 2009, 38, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Urzúa, A.; Santander, R.; Echeverría, J.; Villalobos, C.; Palacios, S.M.; Rossi, Y. Insecticidal properties of Peumus Boldus Mol. essential oil on the House Fly, Musca domestica L. Bol. Latinoam. Caribe Plant Med. Aromat. 2010, 9, 465–469. [Google Scholar]
- Olivero-Verbel, J.; Tirado-Ballestas, I.; Caballero-Gallardo, K.; Stashenko, E.E. Essential oils applied to the food act as repellents toward Tribolium castaneum. J. Stored Prod. Res. 2013, 55, 145–147. [Google Scholar] [CrossRef]
- Kalita, B.; Bora, S.; Sharma, A.K. Plant essential oils as mosquito repellent—A review. Int. J. Res. Dev. Pharm. Life Sci. 2013, 3, 741–747. [Google Scholar]
- Nerio, L.S.; Olivero-Verbel, J.; Stashenko, E. Repellent activity of essential oils: A review. Bioresour. Technol. 2010, 101, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Stefanazzi, N.; Stadler, T.; Ferrero, A. Composition and toxic, repellent and feeding deterrent activity of essential oils against the stored-grain pests Tribolium castaneum (Coleoptera: Tenebrionidae) and Sitophilus oryzae (Coleoptera: Curculionidae). Pest Manag. Sci. 2011, 67, 639–646. [Google Scholar] [CrossRef] [PubMed]
- Solís, C.; Becerra, J.; Flores, C.; Robledo, J.; Silva, M. Antibacterial and antifungal terpenes from Pilgerodendron uviferum (D. Don) Florin. J. Chil. Chem. Soc. 2004, 49, 157–161. [Google Scholar] [CrossRef]
- Donoso, C.A.; Becerra, J.; Bittner, M.; Elissetche, J.P.; Freer, J.; Mendoza, R.; Sterner, O.; Silva, M. Allelochemicals and natural durability in Chilean Cupressaceae heartwoods. Allelopath. J. 2008, 21, 119–132. [Google Scholar]
- Oyarzún, M.L.; Garbarino, J.A. Sesquiterpenoids from Pilgerodendron uvífera. Phytochemistry 1988, 27, 1121–1123. [Google Scholar] [CrossRef]
- Malizia, R.A.; Cardell, D.A.; Molli, J.S.; González, S.; Guerra, P.E.; Grau, R.J. Volatile constituents of leaf oils from the Cupressacea family: Part II. Austrocedrus chilensis, Fitzroya cupressoides and Pilgerodendron uviferum species growing in Argentina. J. Essent. Oil Res. 2000, 12, 233–237. [Google Scholar] [CrossRef]
- Mutis, A.; Parra, L.; Palma, R.; Pardo, F.; Perich, F.; Quiroz, A. Evidence of contact pheromone use in mating behavior of the raspberry weevil (Coleoptera: Curculionidae). Environ. Entomol. 2009, 38, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Lancelle, H.G.; Giordano, O.S.; Sosa, M.E.; Tonn, C.E. Chemical composition of four essential oils from Eupatorium spp. Biological activities toward Tribolium castaneum (Coleoptera: Tenebrionidae). Rev. Soc. Entomol. Argent 2009, 68, 329–338. [Google Scholar]
- Khani, A.; Heydarian, M. Fumigant and repellent properties of sesquiterpene-rich essential oil from Teucrium polium subsp. Capitatum (L.). Asian Pac. J. Trop. Med. 2014, 7, 956–961. [Google Scholar] [CrossRef]
- Wang, C.-F.; Yang, K.; You, C.-X.; Zhang, W.-J.; Guo, S.-S.; Geng, Z.-F.; Du, S.-S.; Wang, Y.-Y. Chemical composition and insecticidal activity of essential oils from Zanthoxylum dissitum leaves and roots against three species of storage pests. Molecules 2015, 20, 7990–7999. [Google Scholar] [CrossRef] [PubMed]
- Tingey, D.T.; Manning, M.; Grothaus, L.C.; Burns, W.F. Influence of light and temperature on monoterpene emission rates from slash pine. Plant Physiol. 1980, 65, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Fichan, I.; Larroche, C.; Gros, J.B. Water solubility, vapor pressure, and activity coefficients of terpenes and terpenoids. J. Chem. Eng. Data 1999, 44, 56–62. [Google Scholar] [CrossRef]
- Ortega, J.; Helmig, D. Approaches for quantifying reactive and low-volatility biogenic organic compound emissions by vegetation enclosure techniques—Part A. Chemosphere 2008, 72, 343–364. [Google Scholar] [CrossRef] [PubMed]
- Paluch, G.; Grodnitzky, J.; Bartholomay, L.; Coats, J. Quantitative structure-activity relationship of botanical sesquiterpenes: Spatial and contact repellency to the yellow fever mosquito, Aedes aegypti. J. Agric. Food Chem. 2009, 57, 7618–7625. [Google Scholar] [CrossRef] [PubMed]
- Koul, O.; Walia, S.; Dhaliwal, G. Essential oils as green pesticides: Potential and constraints. Biopestic. Int. 2008, 4, 63–84. [Google Scholar]
- Rattan, R.S. Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot. 2010, 29, 913–920. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Tampe, J.; Parra, L.; Huaiquil, K.; Mutis, A.; Quiroz, A. Repellent effect and metabolite volatile profile of the essential oil of Achillea millefolium against Aegorhinus nodipennis (Hope) (Coleoptera: Curculionidae). Neotrop. Entomol. 2015, 44, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Tapia, S.; Pardo, F.; Perich, F.; Quiroz, A. Clover root borer Hylastinus obscurus (Marsham) (Coleoptera: Scolytidae) has no preference for volatiles from root extracts of disease infected red clover. Acta Agric. Scand. B 2005, 55, 158–160. [Google Scholar]
- Noldus, L.P.J.J.; Spink, A.J.; Tegelenbosch, R.A.J. Computerised video tracking, movement analysis and behaviour recognition in insects. Comput. Electron. Agric. 2002, 35, 201–227. [Google Scholar] [CrossRef]
- StatsDirect Ltd. StatsDirect Statistical Software; StatsDirect Ltd.: Cheshire, UK, 2013. [Google Scholar]
- Sample Availability: Samples of the EOs are not available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).