Synthesis and Antimicrobial Evaluation of a New Series of Heterocyclic Systems Bearing a Benzosuberone Scaffold
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
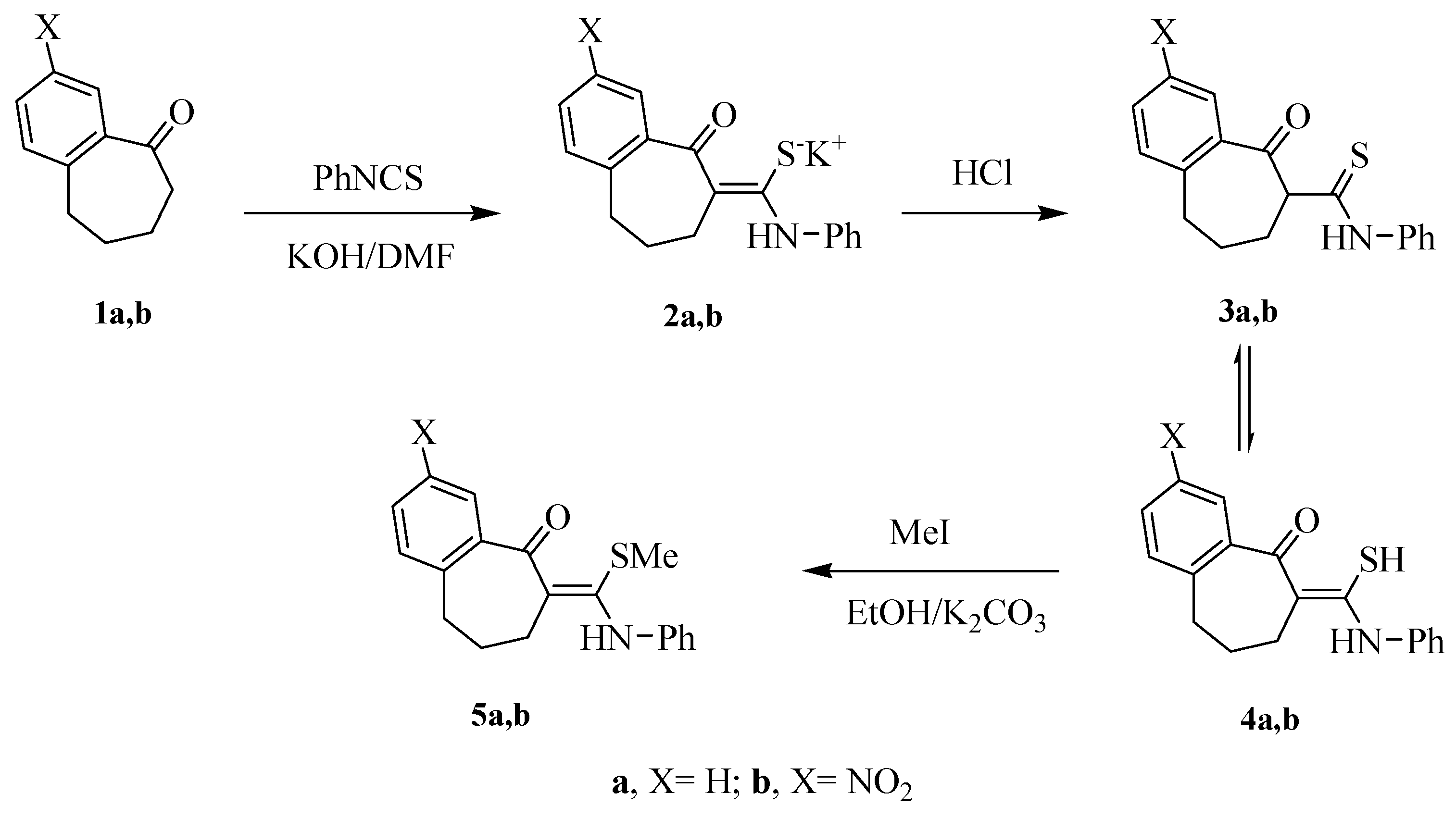
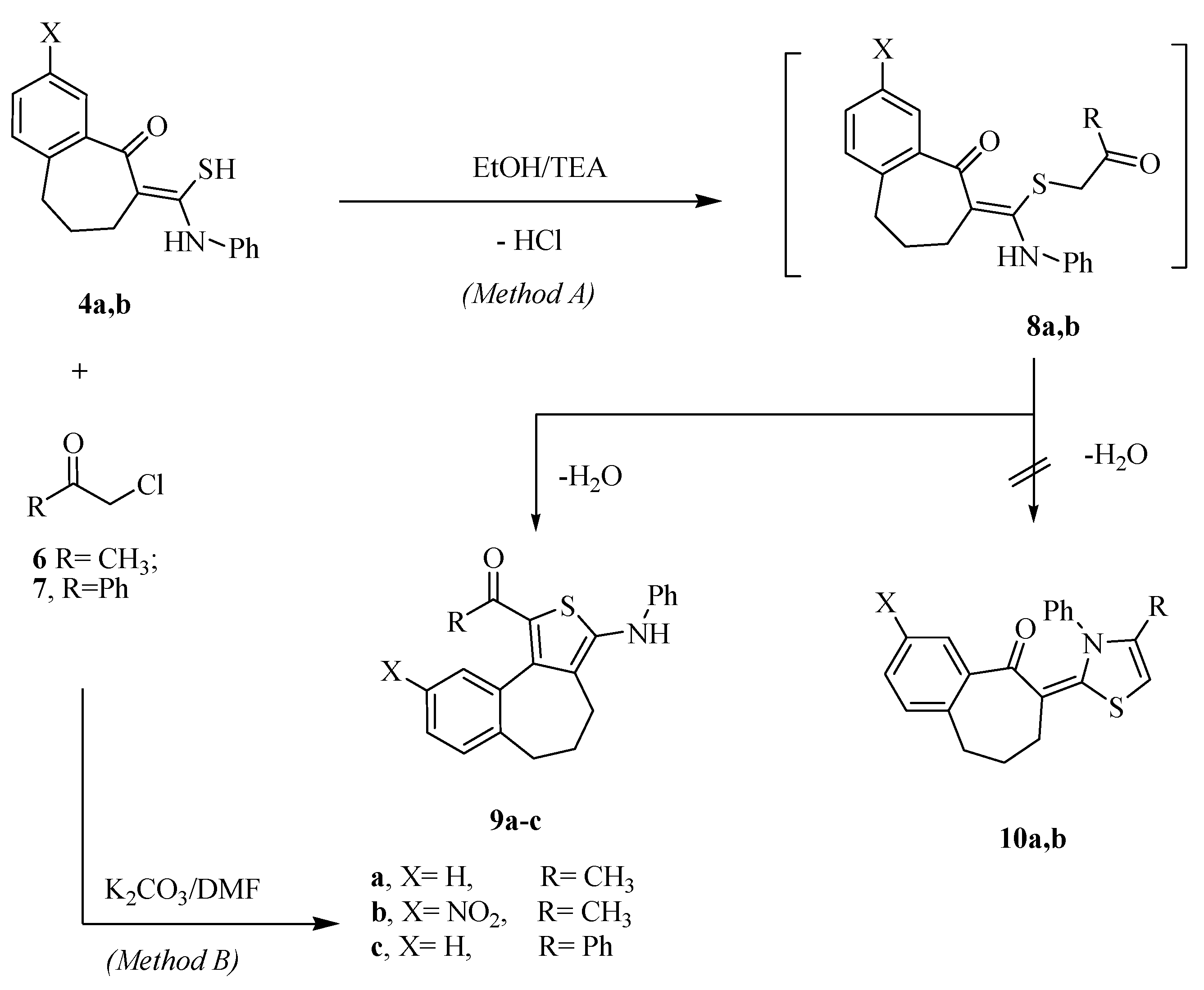
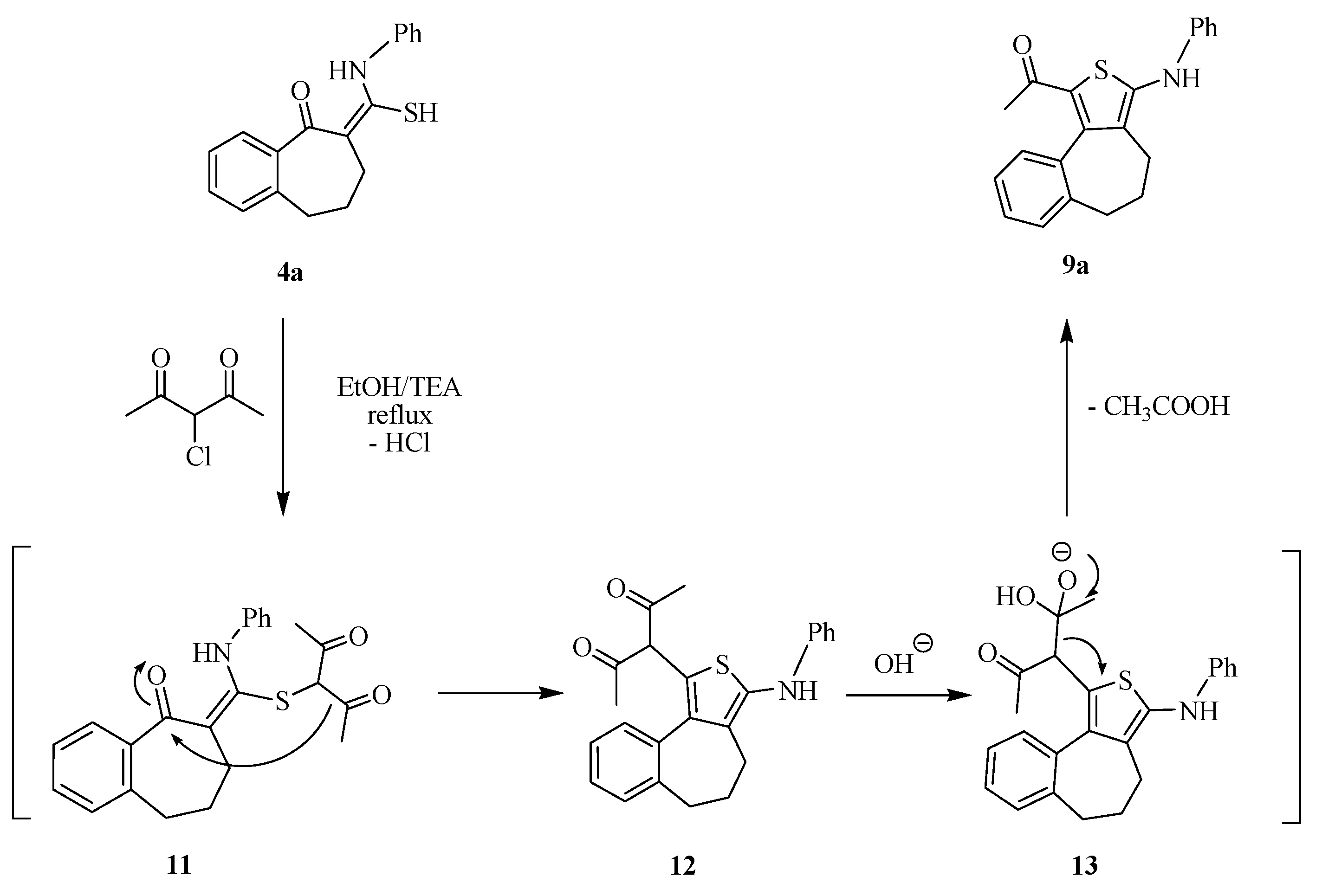
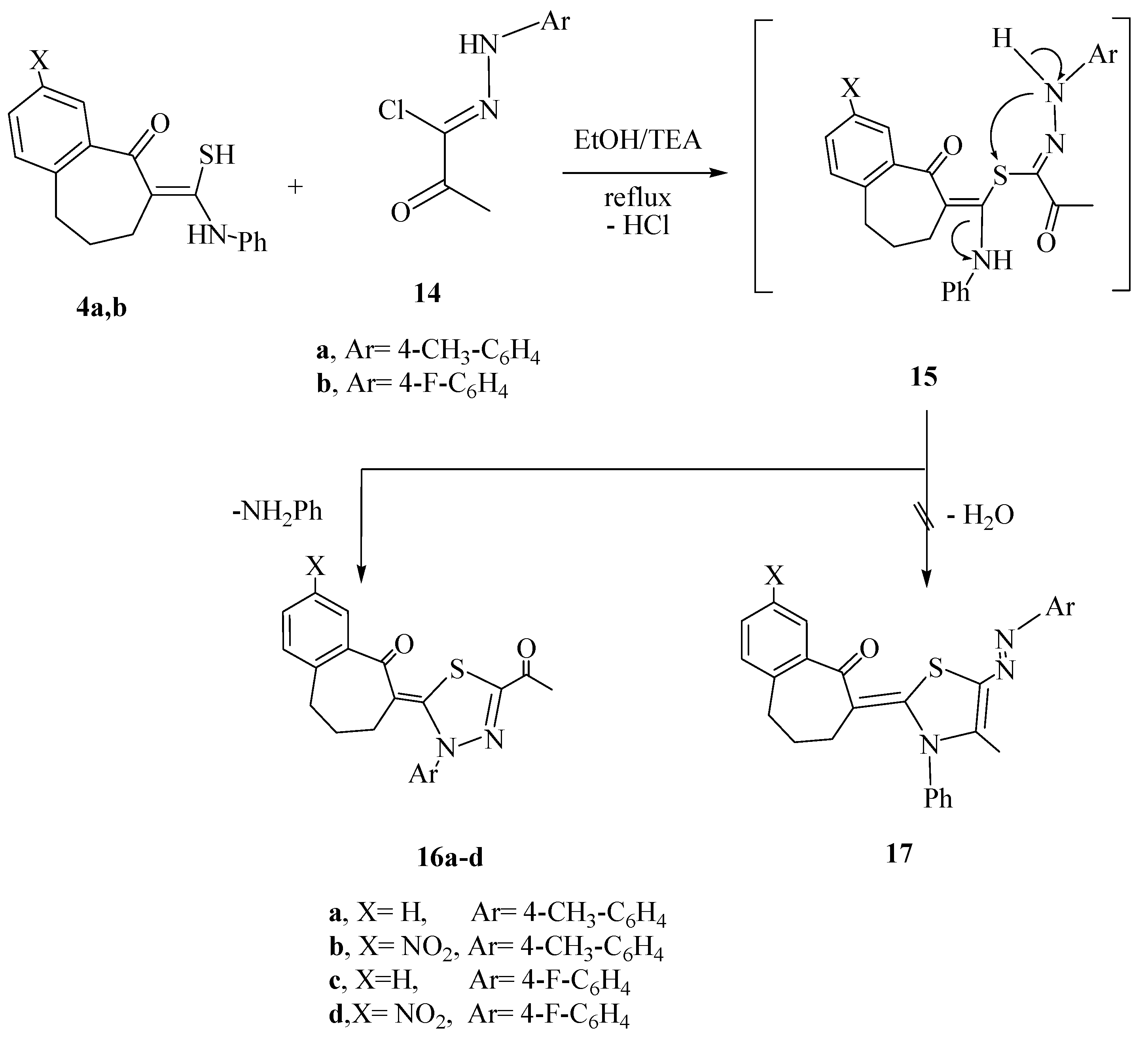
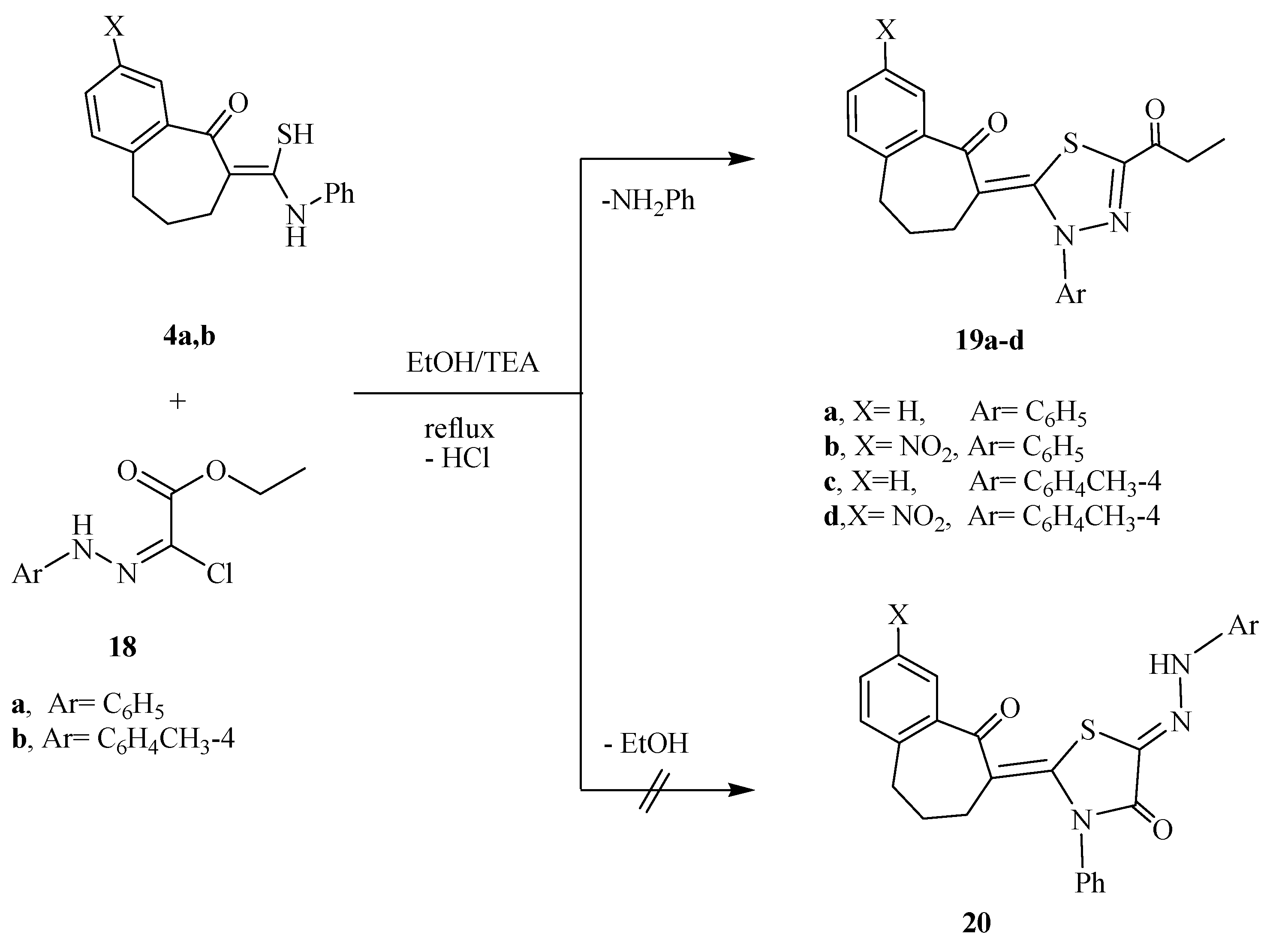

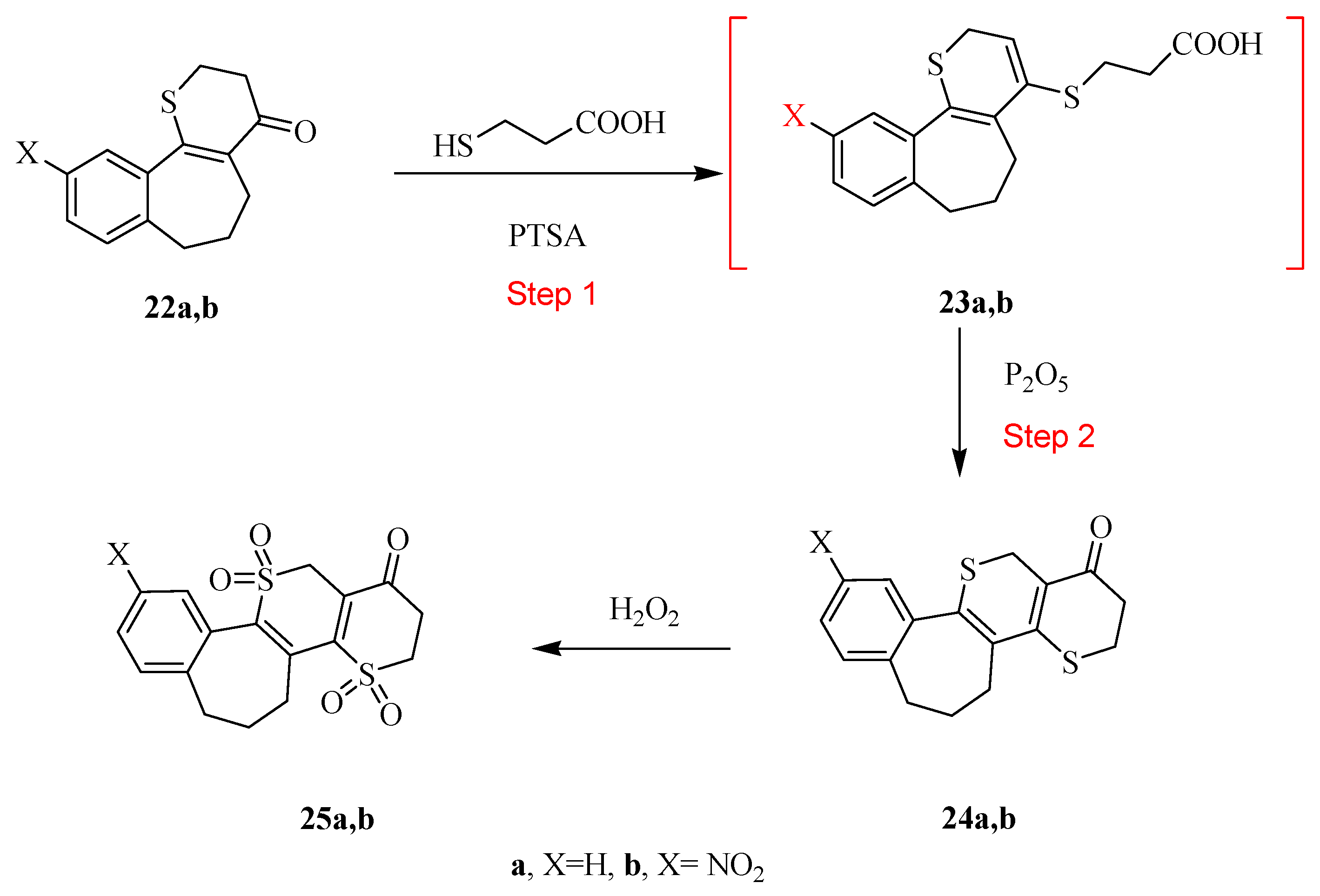
2.2. Antimicrobial Screening
| Comp. No. | Bacteria | Fungi | |||||||
|---|---|---|---|---|---|---|---|---|---|
| G + ve | G − ve | unicellular | filamentous | ||||||
| B. subtilits | S. aureus | Ent. faecalis | E. coli | P. aeruginosa | Proteus sp. | C. albicans | A. niger | A. flavus | |
| 4a | 17 | 17 | 14 | 17 | 14 | 15 | 15 | 12 | 11 |
| 4b | 15 | 15 | 12 | 15 | 12 | 13 | 00 | 20 | 18 |
| 5a | 00 | 18 | 15 | 18 | 15 | 14 | 15 | 20 | 19 |
| 5b | 00 | 12 | 11 | 12 | 11 | 12 | 15 | 20 | 15 |
| 9a | 12 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 9b | 12 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 9c | 22 | 20 | 17 | 20 | 18 | 17 | 17 | 20 | 16 |
| 16a | 20 | 16 | 14 | 16 | 14 | 15 | 00 | 00 | 00 |
| 16b | 12 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 16c | 15 | 15 | 13 | 15 | 12 | 13 | 00 | 00 | 00 |
| 16d | 14 | 13 | 12 | 13 | 12 | 11 | 00 | 00 | 00 |
| 19a | 12 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 19b | 22 | 20 | 17 | 20 | 17 | 18 | 17 | 00 | 00 |
| 19c | 12 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 19d | 20 | 17 | 14 | 17 | 14 | 15 | 00 | 20 | 17 |
| 21a | 17 | 15 | 13 | 15 | 12 | 13 | 20 | 00 | 00 |
| 21b | 14 | 20 | 18 | 20 | 15 | 17 | 00 | 00 | 00 |
| 22a | 25 | 00 | 00 | 00 | 00 | 00 | 30 | 40 | 20 |
| 22b | 15 | 00 | 00 | 00 | 00 | 00 | 15 | 18 | 16 |
| 24a | 15 | 00 | 00 | 00 | 00 | 00 | 15 | 18 | 15 |
| 24b | 12 | 00 | 00 | 00 | 00 | 00 | 15 | 00 | 00 |
| 25a | 17 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| 25b | 27 | 25 | 19 | 25 | 19 | 20 | 25 | 29 | 20 |
| S * =10 µg | 14 | 00 | 00 | 00 | 00 | 00 | 12 | 00 | 00 |
| TE * =30 µg | 10 | 00 | 00 | 00 | 00 | 00 | 23 | 00 | 00 |
| N * =30 µg | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 16 | 14 |
| T * =30 µg | 30 | 00 | 00 | 00 | 00 | 00 | 00 | 00 | 00 |
| Comp. No. | Inhibition Zone Diameters (mm) | MIC (µg/mL) | ||||||
|---|---|---|---|---|---|---|---|---|
| B. sutbtilis | E. coli | C. albicans | A. niger | B. sutbtilis | E. coli | C. albicans | A. niger | |
| 25b | 11 | 11 | 11 | 11 | 75 | 50 | 50 | 100 |
| 22a | 11 | - | - | - | 100 | - | - | - |
| 9c | 11 | - | - | - | 200 | - | - | - |
3. Experimental Section
3.1. Chemistry
3.1.1. Preparation of the Thioamide Derivatives 4a,b
3.1.2. General Procedure for Preparation of the S-Methylated Thioamide Derivatives 5a,b
3.1.3. Reaction of Thioamide Derivatives 4 with α-Halo Carbonyl Compounds: General Procedure for the Preparation of 9a–c
3.1.4. Reactions of Thioamide Derivatives 4a,b with C-Acetyl-N-arylhydrazonoyl Chlorides 14a,b and C-Ethoxycarbonyl-N-arylhydrazonoyl chlorides 18a,b
3.1.5. General Procedure for the Preparation of Compounds 21a,b
3.1.6. General Procedure for the Preparation of Compounds 22a,b
3.1.7. General Procedure for the Preparation of Compounds 24a,b
3.1.8. General Procedure for the Preparation of Compounds 25a,b
3.2. Biological Evaluation
3.2.1. Antimicrobial Activity
Sample Preparation
Strains and Media Used
Bioassay
3.2.2. Determination of Minimum Inhibitory Concentration (MIC)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rida, S.M.; Habib, N.S.; Badawey, E.A.; Fahmy, H.T.; Ghozlan, H.A. Synthesis of novel thiazolo[4,5-d]pyrimidine derivatives for antimicrobial, anti-HIV and anticancer investigation. Pharmazie 1996, 51, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Bahashwan, S.A.; Fayed, A.A.; Ramadan, M.A.; Amr, A.E.; Al-Harbi, N.O. Androgen receptor antagonists and anti-prostate cancer activities of some newly synthesized substituted fused pyrazolo-, triazolo- and thiazolo-pyrimidine derivatives. Int. J. Mol. Sci. 2014, 15, 21587–21602. [Google Scholar] [CrossRef] [PubMed]
- Abou-Ghalia, M.H.; Amr, A.E. Synthesis and investigation of a new cyclo-(Nα-ipicolinoyl)pentapeptide of a breast and CNS cytotoxic activity and an ionophoric specifity. Amino Acids 2004, 26, 283–289. [Google Scholar] [PubMed]
- Yadagiri, B.; Holagunda, U.D.; Bantu, R.; Nagarapu, L.; Kumar, C.G.; Pombala, S.; Sridhar, B. Synthesis of novel building blocks of benzosuberone bearing coumarin moieties and their evaluation as potential anticancer agents. Eur. J. Med. Chem. 2014, 79, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Nagarapu, L.; Yadagiri, B.; Bantu, R.; Kumar, G.; Pombala, S.; Nanubolu, J. Studies on the synthetic and structural aspects of benzosuberones bearing 2,4-thiazolidenone moiety as potential anti-cancer agents. Eur. J. Med. Chem. 2014, 71, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, M.; Groshan, K.; Blakely, R.D.; Richelson, E. Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur. J. Pharmacol. 1997, 340, 249–258. [Google Scholar] [CrossRef]
- Mutschler, E. Drug Actions: Basic Principles and Therapeutic Aspects; ISBN 0-8493-7774-9. Medpharm Scientific Pub.: Stuttgart, Germany, 1995; p. 127, Retrieved 30 January 2013. [Google Scholar]
- Rostom, S.A.F.; El-Ashmawy, I.M.; Abd El Razik, H.A.; Badr, M.H.; Ashour, M.A. Design and synthesis of some thiazolyl and thiadiazolyl derivatives of antipyrine as potential non-acidic anti-inflammatory, analgesic and antimicrobial agents. Bioorg. Med. Chem. 2009, 17, 882–895. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Saber, G.E.; Mahfouz, N.M.; El-Gendy, M.A.; Radwan, A.A.; Hamid, M.A. Synthesis and three-dimensional qualitative structure selectivity relationship of 3,5-disubstituted-2,4-thiazolidinedione derivatives as COX2 inhibitors. Arch. Pharm. Res. 2007, 30, 1186–1204. [Google Scholar] [CrossRef] [PubMed]
- Ouf, N.H.; Amr, A.E.; Sakran, M.I. Anticancer activity of some newly synthesized pyrano[2,3-d][1,2,3]triazine derivatives using 1-(7-hydroxy-2,2-dimethylchroman-6-yl)ethanone as synthon. Med. Chem. Res. 2015, 24, 1514–1526. [Google Scholar] [CrossRef]
- Abd El-Salam, O.I.; Abou El-Ella, D.A.; Ismail, N.S.M.; Abdullah, M. Synthesis, docking studies and anti-inflammatory activity of some 2-amino-5,6,7,8-tetrahydroquinoline-3-carbonitriles and related compounds. Pharmazie 2009, 64, 147–155. [Google Scholar] [PubMed]
- Abdel Salam, O.I.; Khalifa, N.M.; Said, S.A.; Amr, A.E. Synthesis and antimicrobial activities of some newly 2,4,6-tri-substituted pyridine derivatives. Res. Chem. Intermed. 2014, 40, 1147–1155. [Google Scholar] [CrossRef]
- Amr, A.E.; Abd El-Salam, O.I.; Al-Omar, M.A. Synthesis of Chiral Macrocycles: I. Synthesis and Study of Cyclo (Nα-Dinicotinoyl)pentapeptide Candidates. Russ. J. Gen. Chem. 2015, 85, 1161–1166. [Google Scholar] [CrossRef]
- Amr, A.E.; Ali, K.A.; Abdalla, M.M. Cytotoxic, antioxidant activities and structure activity relationship of some newly synthesized terpenoidaloxaliplatin analogs. Eur. J. Med. Chem. 2009, 44, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Ali, K.A.; Abdalghfar, H.S.; Mahmoud, K.; Ragab, E.A. Synthesis and antitumor activity of new poly-substituted thiophenes and 1,3,4-thiadiazoles incorporating 2,6-pyridine moieties. J. Heterocycl. Chem. 2013, 50, 1157–1164. [Google Scholar]
- Ali, K.A.; Hosni, H.M.; Elngar, D.H.; Amr, A.E. Synthesis and reactions of 2-(phenylthiocarbamoyl)-N-(benzothiazol-2-yl)-3-phenyl-3-oxopropanamide with activated chloro-compounds. Phosphorus Sulfur Silicon 2014, 189, 1831–1840. [Google Scholar] [CrossRef]
- Connell, S.R.; Tracz, D.M.; Nierhaus, K.H.; Taylor, D.E. Ribosomal protection proteins and their mechanism of tetracycline resistance. Antimicrob. Agents Chemother. 2003, 47, 3675–3681. [Google Scholar] [CrossRef] [PubMed]
- Hinman, M.M.; Rosenberg, T.A.; Balli, D.; Black-Schaefer, C.; Chovan, L.E.; Kalvin, D.; Merta, P.J.; Nilius, A.M.; Pratt, S.D.; Soni, N.B.; et al. Novel antibacterial class: A series of tetracyclic derivatives. J. Med. Chem. 2006, 49, 4842–4856. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.A.S.; Berry, W.L. Some Exploratory Syntheses of Benzosuberans and Tetrahydrobenzazepinones and Some Related Diazoöxides. J. Org. Chem. 1961, 26, 27–36. [Google Scholar] [CrossRef]
- Amr, A.E.; Mohamed, A.M.; Mohamed, S.F.; Abdel-Hafez, N.A.; Hammam, A.G. Anticancer activities of some newly synthesized pyridine, pyrane, and pyrimidine derivatives. Bioorg. Med. Chem. 2006, 14, 5481–5488. [Google Scholar] [CrossRef] [PubMed]
- Dieckmann, W.; Platz, L. Ueber eineneue Bildungs weise von Osotetrazonen. Chem. Ber. 1905, 38, 2986–2990. [Google Scholar] [CrossRef]
- Shawali, A.S.; Albar, H.A. Kinetics and mechanism of dehydrochlorination of N-aryl-C-ethoxycarbonylformohydrazidoyl chlorides. Can. J. Chem. 1986, 64, 871–875. [Google Scholar] [CrossRef]
- Williams, S.T.; Goodfellow, M.; Wellington, E.M.H.; Vickers, J.C.; Alderson, G.; Sneath, P.H.A.; Sackins, S.M.J.; Mortimer, A.M. A Probability Matrix for Identification of some Streptomycetes. J. Gen. Microbiol. 1983, 129, 1815–1830. [Google Scholar] [CrossRef] [PubMed]
- Bonev, B.; Hooper, J.; Parisot, J. Principles of assessing bacterial susceptibility to antibiotics using the agar diffusion method. J. Antimicrob. Chemother. 2008, 61, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Cruickshank, R.; Duguid, J.P.; Marmion, B.P.; Swain, R.H.A. The practice of medicinal microbiology. In Medical Microbiology, 12th ed.; Churchill Livingstone: Edinburgh, UK, 1975; Volume 11, pp. 196–202. [Google Scholar]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Salam, O.I.A.; Alsayed, A.S.; Ali, K.A.; Elwahab, A.A.A.; Amr, A.E.-G.E.; Awad, H.M. Synthesis and Antimicrobial Evaluation of a New Series of Heterocyclic Systems Bearing a Benzosuberone Scaffold. Molecules 2015, 20, 20434-20447. https://doi.org/10.3390/molecules201119701
El-Salam OIA, Alsayed AS, Ali KA, Elwahab AAA, Amr AE-GE, Awad HM. Synthesis and Antimicrobial Evaluation of a New Series of Heterocyclic Systems Bearing a Benzosuberone Scaffold. Molecules. 2015; 20(11):20434-20447. https://doi.org/10.3390/molecules201119701
Chicago/Turabian StyleEl-Salam, Osama I. Abd, Ali S. Alsayed, Korany A. Ali, Ahmed A. Abd Elwahab, Abd El-Galil E. Amr, and Hassan M. Awad. 2015. "Synthesis and Antimicrobial Evaluation of a New Series of Heterocyclic Systems Bearing a Benzosuberone Scaffold" Molecules 20, no. 11: 20434-20447. https://doi.org/10.3390/molecules201119701
APA StyleEl-Salam, O. I. A., Alsayed, A. S., Ali, K. A., Elwahab, A. A. A., Amr, A. E.-G. E., & Awad, H. M. (2015). Synthesis and Antimicrobial Evaluation of a New Series of Heterocyclic Systems Bearing a Benzosuberone Scaffold. Molecules, 20(11), 20434-20447. https://doi.org/10.3390/molecules201119701







