MALDI-TOF MS Analysis of Cellodextrins and Xylo-oligosaccharides Produced by Hindgut Homogenates of Reticulitermes santonensis
Abstract
:1. Introduction
| Cellulase Activity | Substrate | Hydrolysis Products |
|---|---|---|
| Endo-1,4-β-glucanase | Amorphous cellulose, soluble cellulose | Glucose, cellobiose, cellotriose, higher oligomers |
| Cellobiohydrolase | Crystalline cellulose, soluble cellodextrins | Glucose, cellobiose |
| Cellodextrinase | Soluble cellodextrins | Glucose, cellobiose |
| β-1,4-glucosidase | Cellobiose, soluble cellodextrins | Glucose |
2. Results and Discussion
2.1. Oligosaccharides Produced from Cellulose Derivative


2.2. Oligosaccharides Produced from Crystalline Cellulose
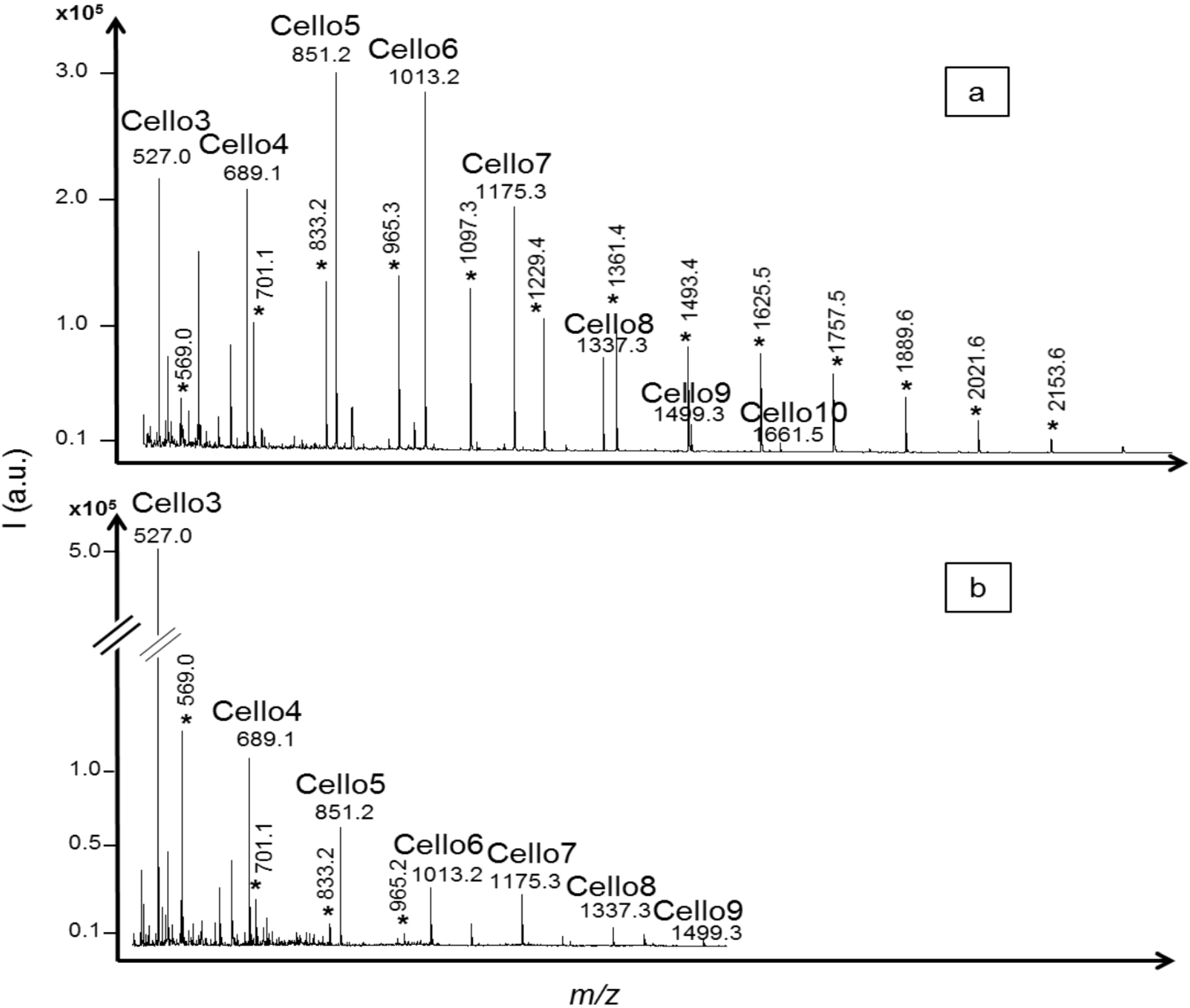
2.3. Oligosaccharides Produced from Xylan
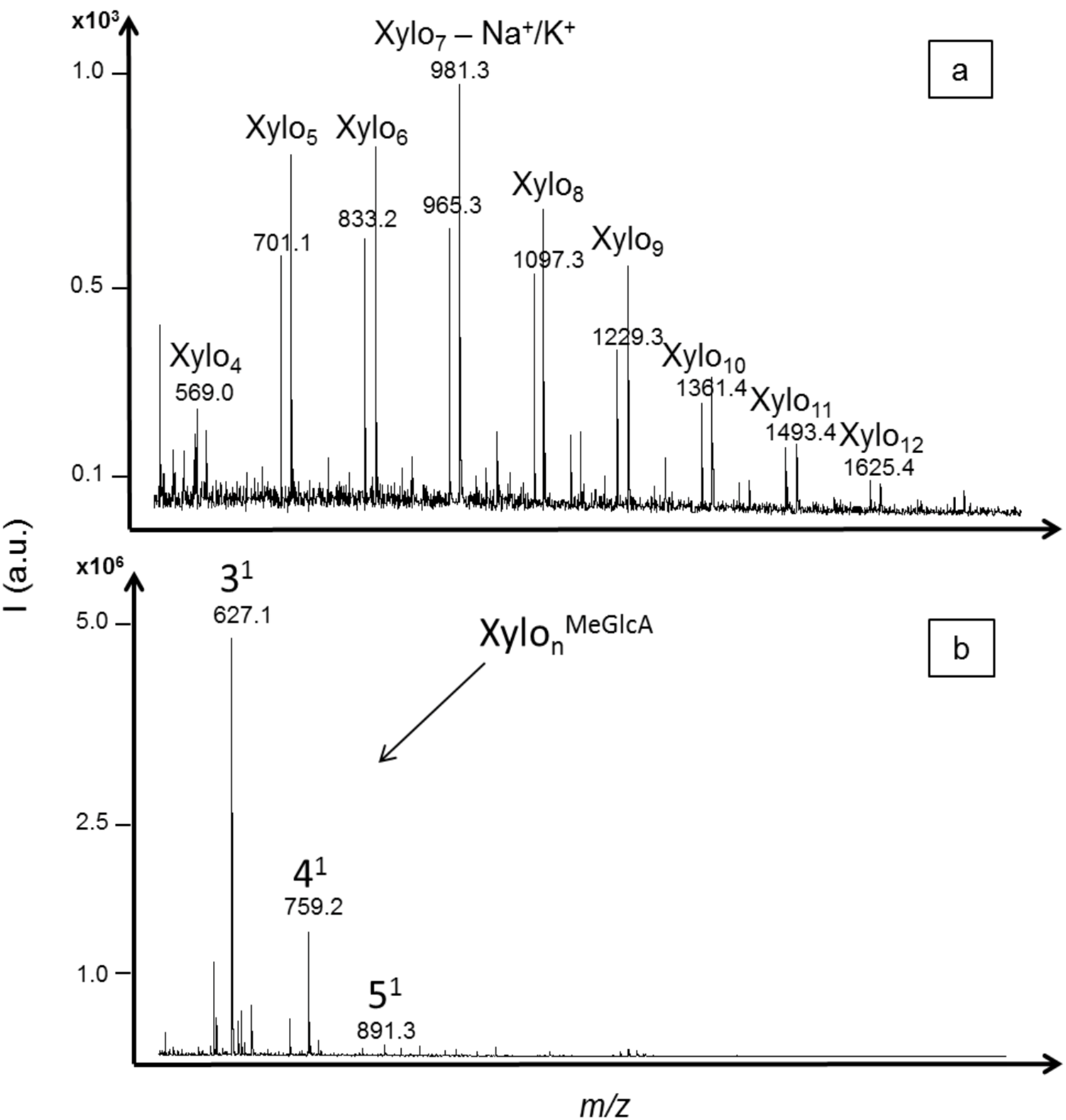
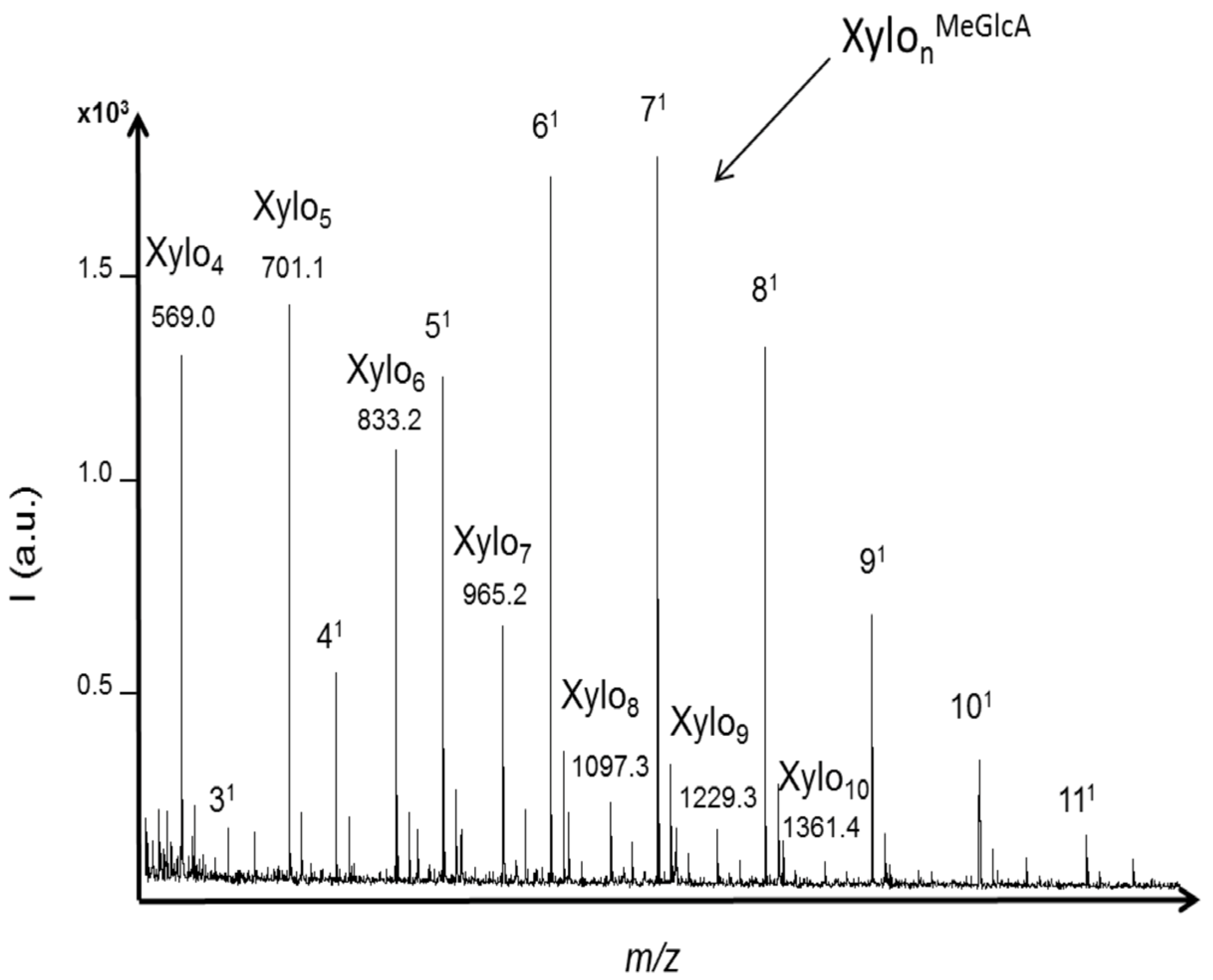
3. Experimental
3.1. Termites
3.2. Hydrolysate Preparation
3.3. MALDI-TOF MS Analysis
3.4. Data Processing
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Breznak, J.A.; Brune, A. Role of microorganisms in the digestion of lignocellulose by termites. Annu. Rev. Entomol. 1994, 39, 453–487. [Google Scholar] [CrossRef]
- König, H.; Li, L.; Fröhlich, J. The cellulolytic system of the termite gut. Appl. Microbiol. Biotechnol. 2013, 97, 7943–7962. [Google Scholar] [CrossRef]
- Merino, S.T.; Cherry, J. Progress and challenges in enzyme development for biomass utilization. Adv. Biochem. Eng. Biotechnol. 2007, 108, 95–120. [Google Scholar]
- Ni, J.; Tokuda, G. Lignocellulose-degrading enzymes from termites and their symbiotic microbiota. Biotechnol. Adv. 2013, 31, 838–850. [Google Scholar] [CrossRef]
- Lucena, S.A.; Lima, L.S.; Cordeiro, L.S., Jr.; Sant’Anna, C.; Constantino, R.; Azambuja, P.; de Souza, W.; Garcia, E.S.; Genta, F.A. High throughput screening of hydrolytic enzymes from termites using a natural substrate derived from sugarcane bagasse. Biotechnol. Biofuels. 2011, 4. [Google Scholar] [CrossRef]
- Oppert, C.; Klingeman, W.E.; Willis, J.D.; Oppert, B.; Jurat-Fuentes, J.L. Prospecting for cellulolytic activity in insect digestive fluids. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2010, 155, 145–154. [Google Scholar] [CrossRef]
- Tokuda, G.; Lo, N.; Watanabe, H. Marked variations in patterns of cellulase activity against crystalline- vs. carboxymethyl-cellulose in the digestive systems of diverse, wood-feeding termites. Physiol. Entomol. 2005, 30, 372–380. [Google Scholar]
- Andrić, P.; Meyer, A.S.; Jensen, P.A.; Dam-Johansen, K. Reactor design for minimizing product inhibition during enzymatic lignocellulosehydrolysis: I. Significance and mechanism of cellobiose and glucose inhibition oncellulolytic enzymes. Biotechnol. Adv. 2010, 28, 308–324. [Google Scholar] [CrossRef]
- Xiao, Z.; Zhang, X.; Gregg, D.J.; Saddler, J.N. Effects of sugar inhibition on cellulases and β-glucosidase during enzymatic hydrolysis of softwood substrates. Appl. Biochem. Biotechnol. 2004, 113–116, 1115–1126. [Google Scholar]
- Harjunpää, V.; Helin, J.; Koivula, A.; Siika-aho, M.; Drakenberg, T. A comparative study of two retaining enzymes of Trichoderma reesei: Transglycosylation of oligosaccharides catalysed by the cellobiohydrolase I, Cel7A, and the β-mannanase, Man5A. FEBS Lett. 1999, 443, 149–153. [Google Scholar] [CrossRef]
- Nouaille, R.; Matulova, M.; Delort, A.M.; Forano, E. Oligosaccharides synthesis in Fibrobacter succinogenes S85 and its modulation by the substrate. FEBS J. 2005, 272, 2416–2427. [Google Scholar] [CrossRef]
- Suzuki, H.; Igarashi, K.; Samejima, M. Cellotriose and cellotetraose as inducers of the genes encoding cellobiohydrolases in the basidiomycete Phanerochaete chrysosporium. Appl. Environ. Microbiol. 2010, 76, 6164–6170. [Google Scholar] [CrossRef]
- Pokusaeva, K.; O’Connell-Motherway, M.; Zomer, A.; MacSharry, J.; Fitzgerald, G.F.; van Sinderen, D. Cellodextrin utilization by Bifidobacterium breve UCC2003. Appl. Environ. Microbiol. 2011, 77, 1681–1690. [Google Scholar] [CrossRef]
- Hu, J.; Arantes, V.; Saddler, J.N. The enhancement of enzymatic hydrolysis of lignocellulosic substrates by the addition of accessory enzymes such as xylanase: Is it an additive or synergistic effect? Biotechnol. Biofuels. 2011, 4. [Google Scholar] [CrossRef]
- Qing, Q.; Yang, B.; Wyman, C.E. Xylooligomers are strong inhibitors of cellulose hydrolysis by enzymes. Bioresour. Technol. 2010, 101, 9624–9630. [Google Scholar] [CrossRef]
- Zhang, J.; Viikari, L. Xylo-oligosaccharides are competitive inhibitors of cellobiohydrolase I from Thermoascus aurantiacus. Bioresour. Technol. 2012, 117, 286–291. [Google Scholar] [CrossRef]
- Aachary, A.A.; Prapulla, S.G. Xylooligosaccharides (XOS) as an emerging prebiotic: microbial synthesis, utilization, structural characterization, bioactive properties, and applications. Compr. Rev. Food. Sci. Food Saf. 2011, 10, 2–16. [Google Scholar] [CrossRef]
- Qing, Q.; Li, H.; Kumar, R.; Wyman, C.E. Xylooligosaccharides Production, Quantification, and Characterization in Context of Lignocellulosic Biomass Pretreatment. In Aqueous Pretreatment of Plant Biomass for Biological and Chemical Conversion to Fuels and Chemicals, 1st ed.; Wyman, C.E., Ed.; John Wiley & Sons, Ltd: Chichester, UK, 2013. [Google Scholar] [CrossRef]
- Andersson, M.; Wittgren, B.; Schagerlof, H.; Momcilovic, D.; Wahlund, K.G. Size and structure characterization of ethylhydroxyethyl cellulose by the combination of field-flow fractionation with other techniques. Investigation of ultra large components. Biomacromolecules 2004, 5, 97–105. [Google Scholar]
- Enebro, J.; Momcilovic, D.; Siika-aho, M.; Karlsson, S. A new approach for studying correlations between the chemical structure and the rheological properties in carboxymethyl cellulose. Biomacromolecules 2007, 8, 3253–3257. [Google Scholar] [CrossRef]
- Momcilovic, D.; Wittgren, B.; Wahlund, K.G.; Karlsson, J.; Brinkmalm, G. Sample preparation effects in matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry of partially depolymerised carboxymethyl cellulose. Rapid Commun. Mass Spectrom. 2003, 17, 1107–1115. [Google Scholar] [CrossRef]
- Cohen, A.; Schagerlof, H.; Nilsson, C.; Melander, C.; Tjerneld, F.; Gorton, L. Liquid chromatography-mass spectrometry analysis of enzyme-hydrolysed carboxymethylcellulose for investigation of enzyme selectivity and substituent pattern. J. Chromatogr. A 2004, 1029, 87–95. [Google Scholar]
- Enebro, J.; Momcilovic, D.; Siika-aho, M.; Karlsson, S. Investigation of endoglucanase selectivity on carboxymethyl cellulose by mass spectrometric techniques. Cellulose 2009, 16, 271–280. [Google Scholar] [CrossRef]
- Karlsson, J.; Momcilovic, D.; Wittgren, B.; Schulein, M.; Tjerneld, F.; Brinkmalm, G. Enzymatic degradation of carboxymethyl cellulose hydrolyzed by the endoglucanases Cel5A, Cel7B, and Cel45A from Humicola insolens and Cel7B, Cel12A and Cel45Acore from Trichoderma reesei. Biopolymers 2002, 63, 32–40. [Google Scholar] [CrossRef]
- Enebro, J.; Karlsson, S. Improved matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry of carboxymethyl cellulose. Rapid Commun. Mass Spectrom. 2006, 20, 3693–3698. [Google Scholar] [CrossRef]
- Park, S.; Baker, J.O.; Himmel, M.E.; Parilla, P.A.; Johnson, D.K. Cellulose crystallinity index: Measurement techniques and their impact on interpreting cellulase performance. Biotechnol. Biofuels. 2010, 3. [Google Scholar] [CrossRef]
- Reese, E.T.; Smakula, E.; Perlin, A.S. Enzymatic production of cellotriose from cellulose. Arch. Biochem. Biophys. 1959, 85, 171–175. [Google Scholar] [CrossRef]
- Huebner, A.; Ladisch, M.R.; Tsao, G.T. Preparation of cellodextrins: An engineering approach. Biotechnol. Bioeng. 1978, 20, 1669–1677. [Google Scholar] [CrossRef]
- Damude, H.G.; Ferro, V.; Whiters, S.G.; Warren, R.A.J. Substrate specificity of endoglucanase A from Cellulomonas fimi: Fundamental differences between endoglucanases and exoglucanases from family 6. Biochem. J. 1996, 315, 467–472. [Google Scholar]
- Gupta, R.; Lee, Y.Y. Mechanism of cellulase reaction on pure cellulosic substrates. Biotechnol. Bioeng. 2009, 102, 1570–1581. [Google Scholar] [CrossRef]
- Polglase, W.J. Polysaccharides associated with wood cellulose. Adv. Carbohydr. Chem. 1955, 10, 283–333. [Google Scholar]
- Kolenova, K.; Vrsanska, M.; Biely, P. Mode of action of endo-beta-1,4-xylanases of families 10 and 11 on acidic xylooligosaccharides. J. Biotechnol. 2006, 121, 338–345. [Google Scholar] [CrossRef]
- Tenkanen, M.; Luonteri, E.; Teleman, A. Effect of side groups on the action of β-xylosidase from Trichoderma reesei against substituted xylo-oligosaccharides. FEBS Lett. 1996, 399, 303–306. [Google Scholar] [CrossRef]
- Jacobs, A.; Larsson, P.T.; Dahlman, O. Distribution of uronic acids in xylans from various species of soft- and hardwood as determined by MALDI mass spectrometry. Biomacromolecules 2001, 2, 979–990. [Google Scholar] [CrossRef]
- Teleman, A.; Tenkanen, M.; Jacobs, A.; Dahlman, O. Characterization of O-acetyl-(4-O-methylglucurono)xylan isolated from birch and beech. Carbohydr. Res. 2002, 337, 373–377. [Google Scholar] [CrossRef]
- Schäfer, A.; Konrad, R.; Kuhnigk, T.; Kämpfer, P.; Hertel, H.; König, H. Hemicellulose-degrading bacteria and yeasts from the termite gut. J. Appl. Bacteriol. 1996, 80, 471–478. [Google Scholar] [CrossRef]
- Bignell, D.E.; Andersson, J.M. Determination of pH and oxygen status in the guts of lower and higher termites. J. Insect. Physiol. 1980, 26, 183–188. [Google Scholar] [CrossRef]
- Mattéotti, C.; Bauwens, J.; Brasseur, C.; Tarayre, C.; Thonart, P.; Destain, J.; Francis, F.; Haubruge, E.; de Pauw, E.; Portetelle, D.; et al. Identification and characterization of a new xylanase from Gram-positive bacteria isolated from termite gut (Reticulitermes santonensis). Protein. Expr. Purify. 2012, 83, 117–127. [Google Scholar] [CrossRef]
- Tarayre, C.; Brognaux, A.; Brasseur, C.; Bauwens, J.; Millet, C.; Mattéotti, C.; Destain, J.; Vandenbol, M.; Portetelle, D.; de Pauw, E.; et al. Isolation and cultivation of a xylanolytic Bacillus subtilis extracted from the gut of the termite Reticulitermes santonensis. Appl. Biochem. Biotechnol. 2013, 171, 225–245. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the produced compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brasseur, C.; Bauwens, J.; Tarayre, C.; Mattéotti, C.; Thonart, P.; Destain, J.; Francis, F.; Haubruge, E.; Portetelle, D.; Vandenbol, M.; et al. MALDI-TOF MS Analysis of Cellodextrins and Xylo-oligosaccharides Produced by Hindgut Homogenates of Reticulitermes santonensis. Molecules 2014, 19, 4578-4594. https://doi.org/10.3390/molecules19044578
Brasseur C, Bauwens J, Tarayre C, Mattéotti C, Thonart P, Destain J, Francis F, Haubruge E, Portetelle D, Vandenbol M, et al. MALDI-TOF MS Analysis of Cellodextrins and Xylo-oligosaccharides Produced by Hindgut Homogenates of Reticulitermes santonensis. Molecules. 2014; 19(4):4578-4594. https://doi.org/10.3390/molecules19044578
Chicago/Turabian StyleBrasseur, Catherine, Julien Bauwens, Cédric Tarayre, Christel Mattéotti, Philippe Thonart, Jacqueline Destain, Frédéric Francis, Eric Haubruge, Daniel Portetelle, Micheline Vandenbol, and et al. 2014. "MALDI-TOF MS Analysis of Cellodextrins and Xylo-oligosaccharides Produced by Hindgut Homogenates of Reticulitermes santonensis" Molecules 19, no. 4: 4578-4594. https://doi.org/10.3390/molecules19044578
APA StyleBrasseur, C., Bauwens, J., Tarayre, C., Mattéotti, C., Thonart, P., Destain, J., Francis, F., Haubruge, E., Portetelle, D., Vandenbol, M., Focant, J.-F., & De Pauw, E. (2014). MALDI-TOF MS Analysis of Cellodextrins and Xylo-oligosaccharides Produced by Hindgut Homogenates of Reticulitermes santonensis. Molecules, 19(4), 4578-4594. https://doi.org/10.3390/molecules19044578






