Alkaloids from Marine Invertebrates as Important Leads for Anticancer Drugs Discovery and Development
Abstract
:1. Introduction
2. β-Pyridopyrrolopyrimidine Alkaloids: Variolins, a New Family of Cyclin-Dependent Kinases (CDKs) Inhibitors

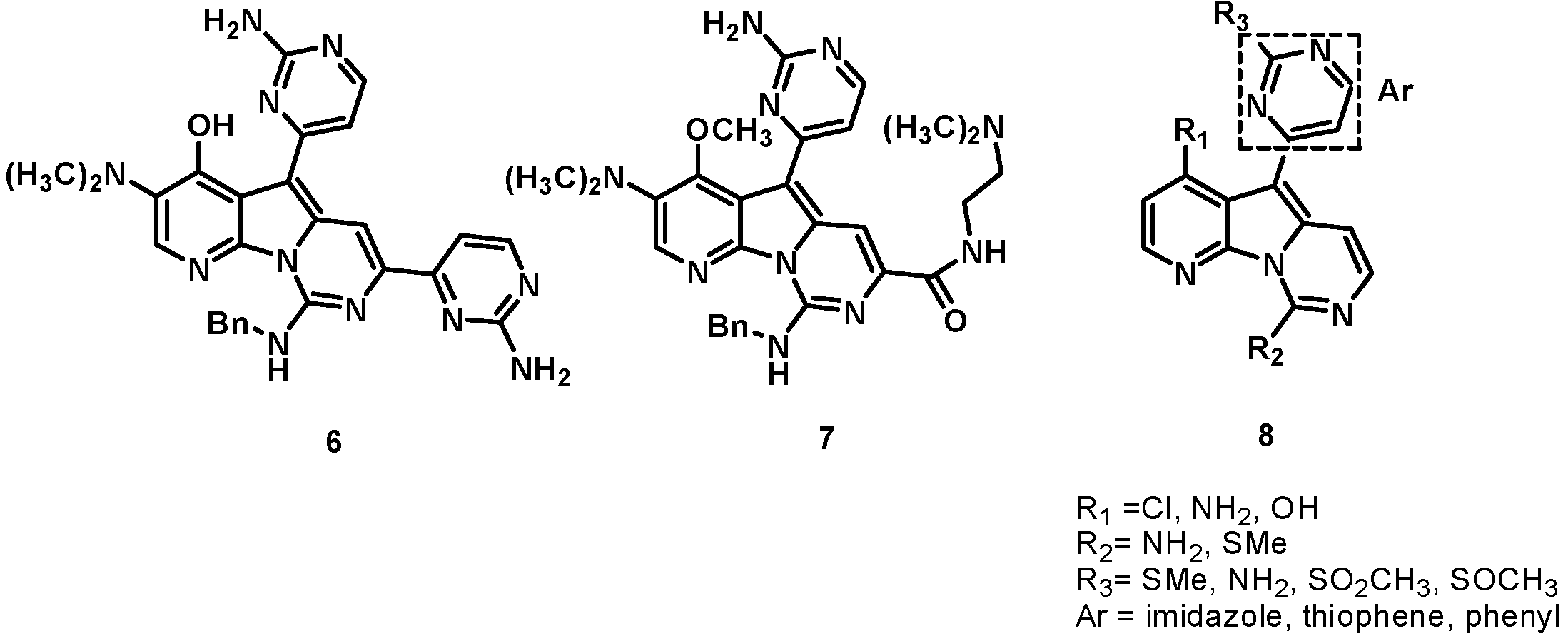
3. Indole Alkaloids: Meridianins, a New Scaffold for the Design of Novel CDK Inhibitors
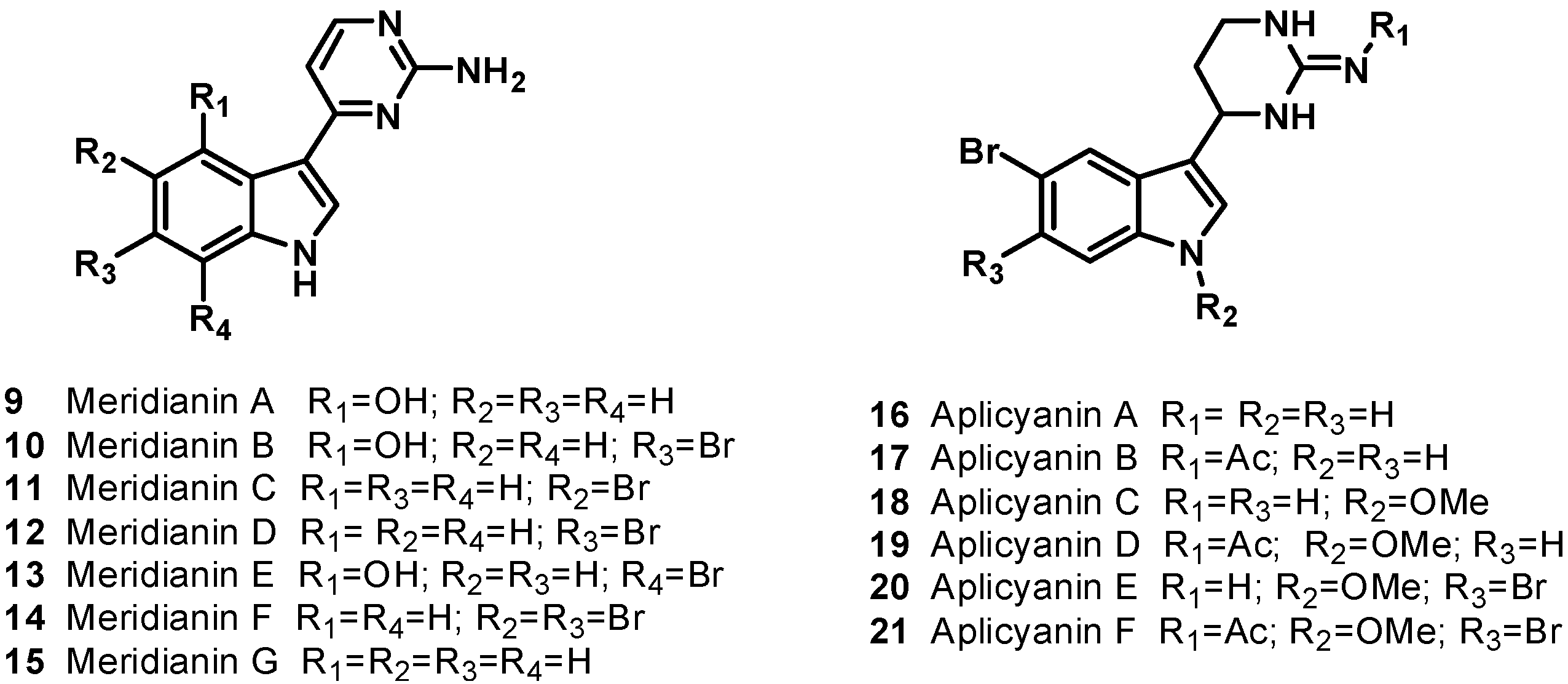

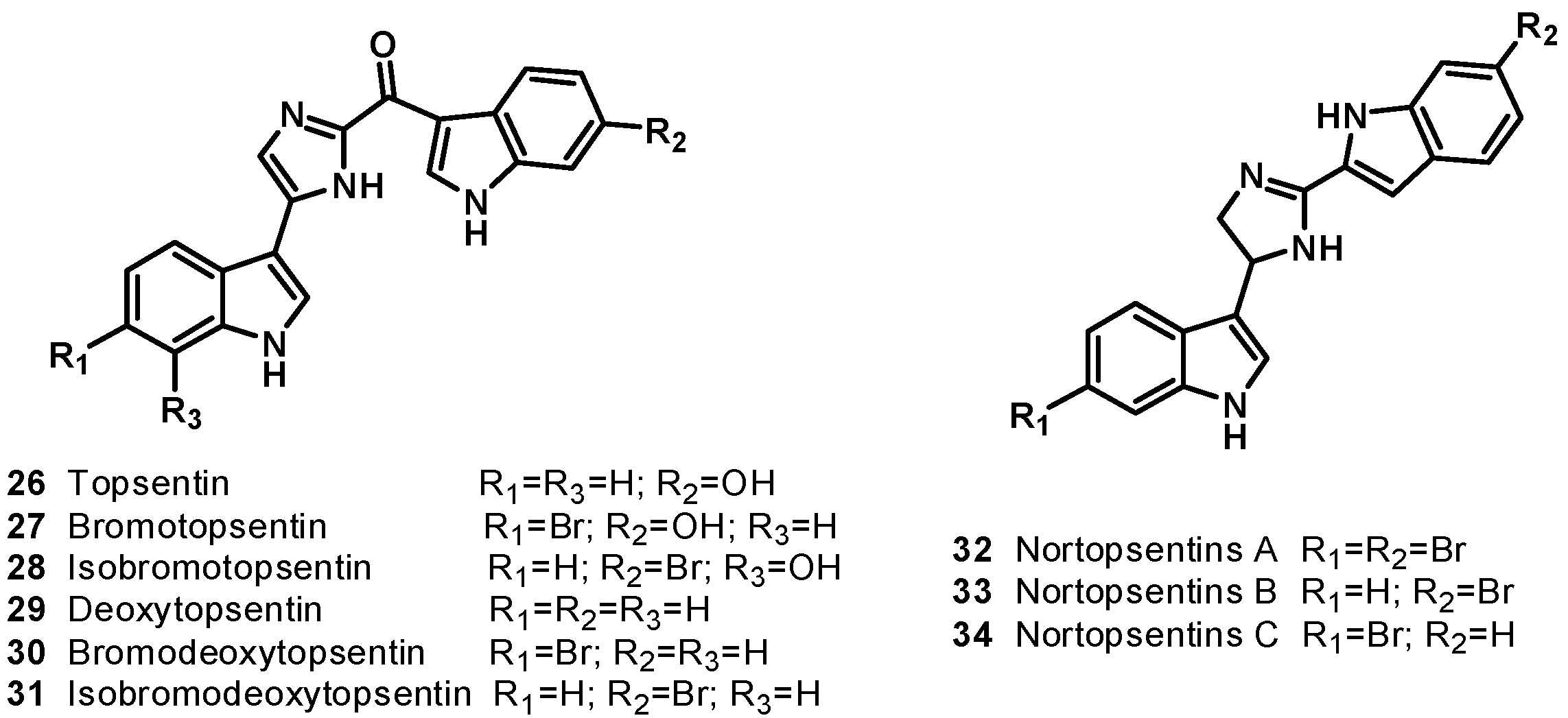

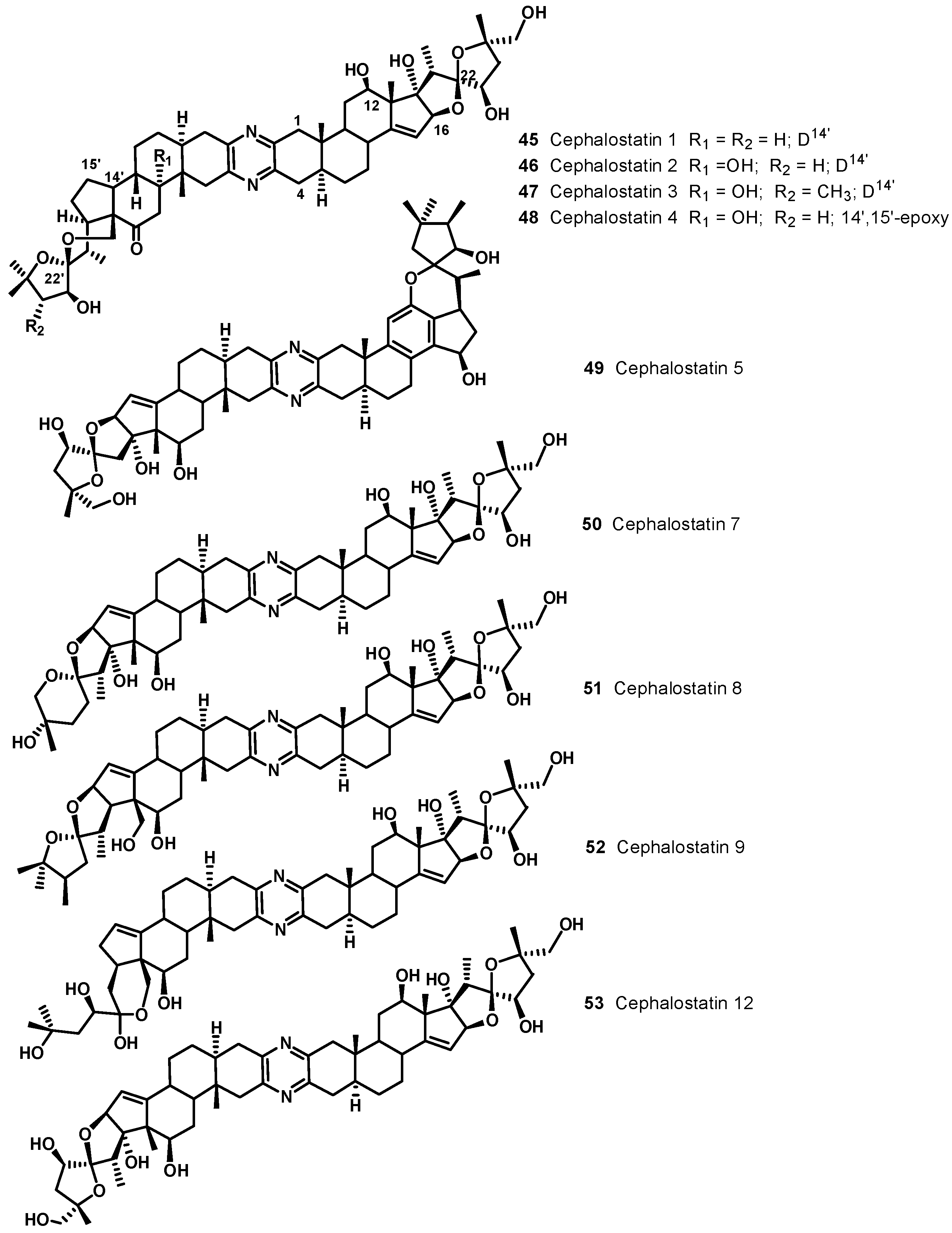
4. Bis-Steroidal Pyrazines: Ritterazines and Cephalostatins, Potent Inducers of Apoptosis Acting via a Novel Antimitotic Mechanism
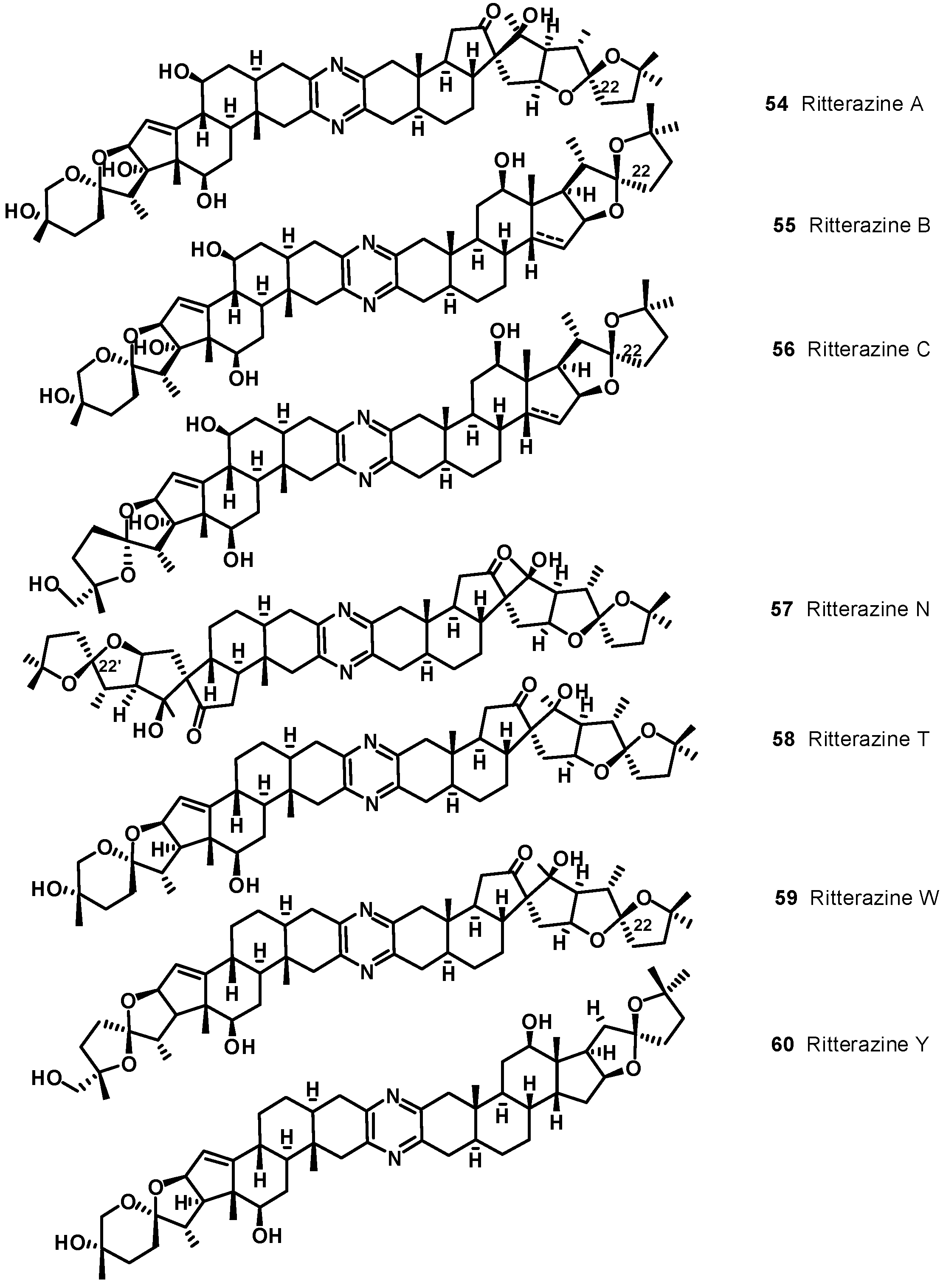
| Ritterazine | (ED50 nM) | Cephalostatin | (ED50 nM) | Ritterazine | (ED50 nM) | Cephalostatin | (ED50 nM) |
|---|---|---|---|---|---|---|---|
| A | l4.2 | 1 | 10−4–10−6 | N | 522 | 14 | 4.4 |
| B | 0.17 | 2 | 10−4–10−6 | O | 2383 | 15 | 26.2 |
| C | 102.3 | 3 | 10−4–10−6 | P | 819 | 16 | <1.1 |
| D | 17.5 | 4 | 10−4–10−6 | Q | 657 | 17 | 4.4 |
| E | 3.8 | 5 | 42.5 | R | 2461 | 18 | 4.6 |
| F | 0.81 | 6 | 2.3 | S | 539 | 19 | 7.9 |
| G | 0.81 | 7 | 1–<0.1 | T | 522 | ||
| H | 17.8 | 8 | 1–<0.1 | U | 2341 | ||
| I | 15.3 | 9 | 1–<0.1 | V | 2341 | ||
| J | 14.0 | 10 | 3.2 | W | 3631 | ||
| K | 10.4 | 11 | 2.7 | X | 3404 | ||
| L | 11.1 | 12 | 76.2 | Y | 4.0 | ||
| M | 16.7 | 13 | 47.9 | Z | 2200 |
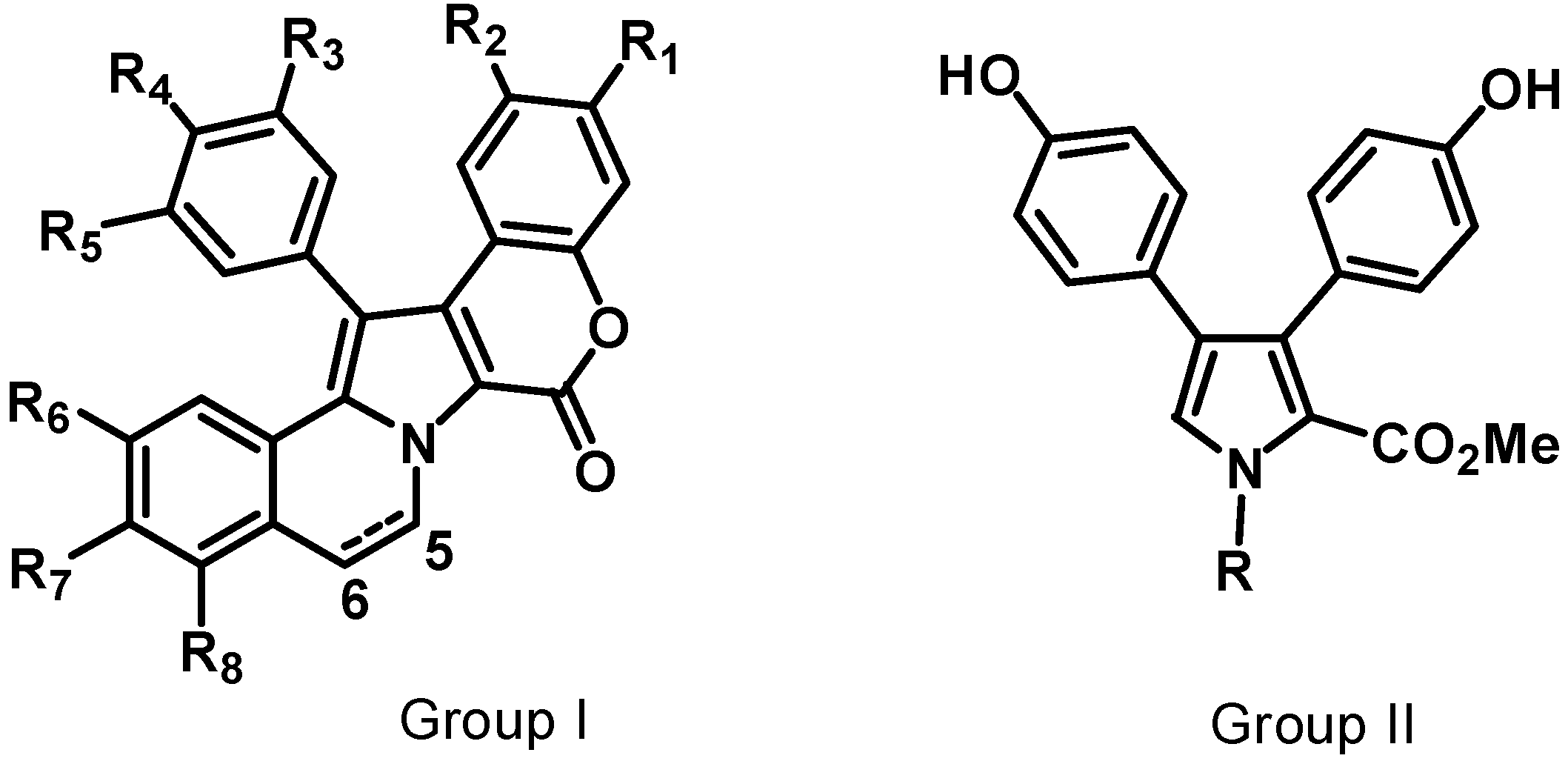
5. Dopa [2-Amino,3-(3',4'-dihydroxyphenyl) Propionic Acid] Derived Pyrrole Alkaloids: Lamellarins, Potent Inhibitors of Topoisomerase (TOPO) I with Multi-Drug Resistance (MDR) Modulating Activity

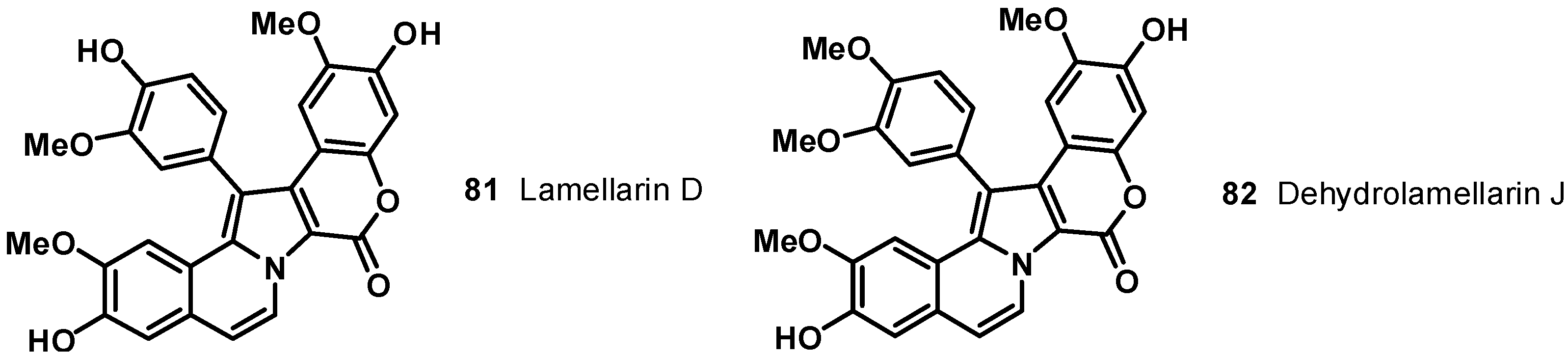
6. Tetrahydroisoquinoline Alkaloids: Ecteinascidins, a New Class of DNA Binding Agents
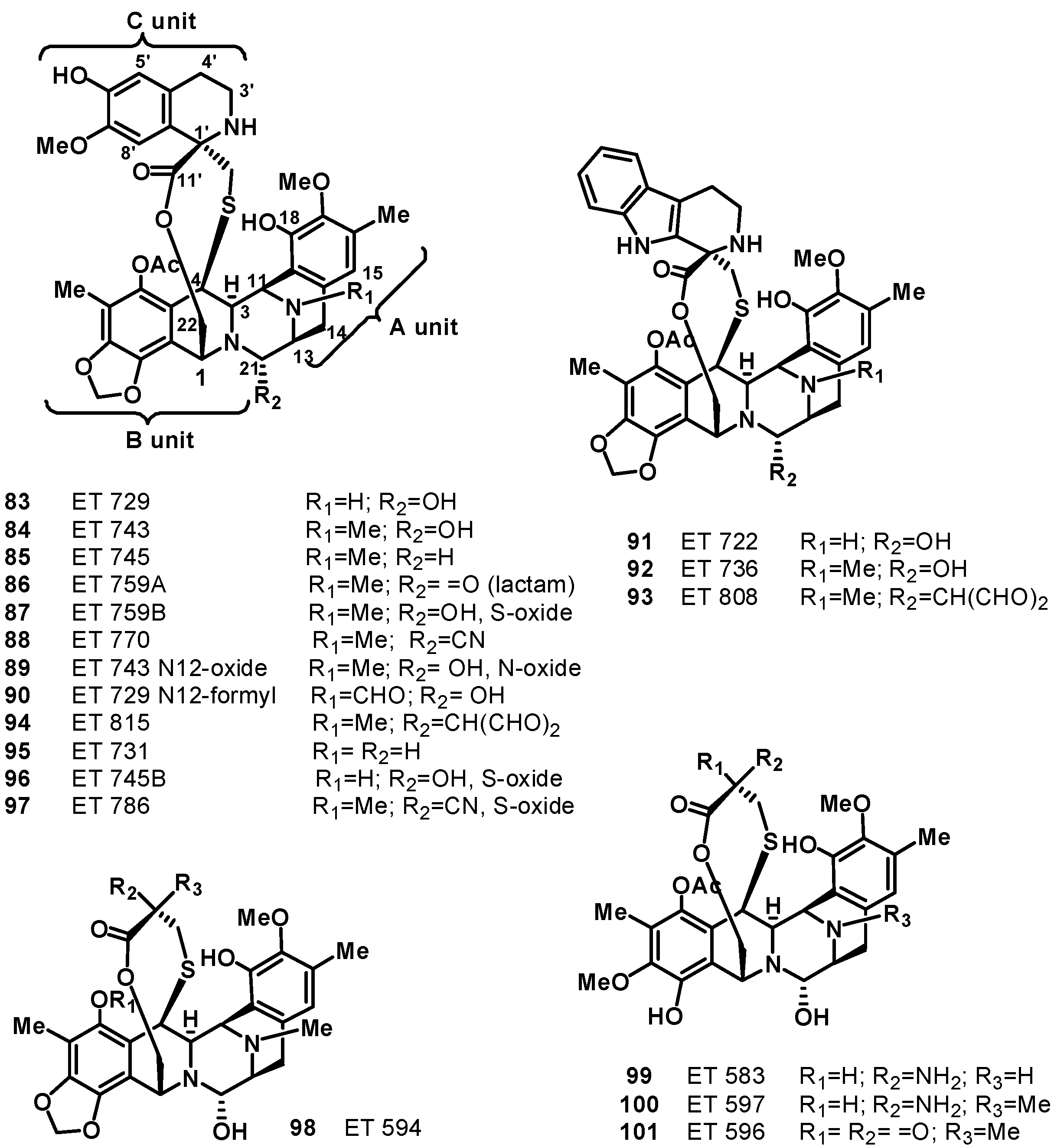
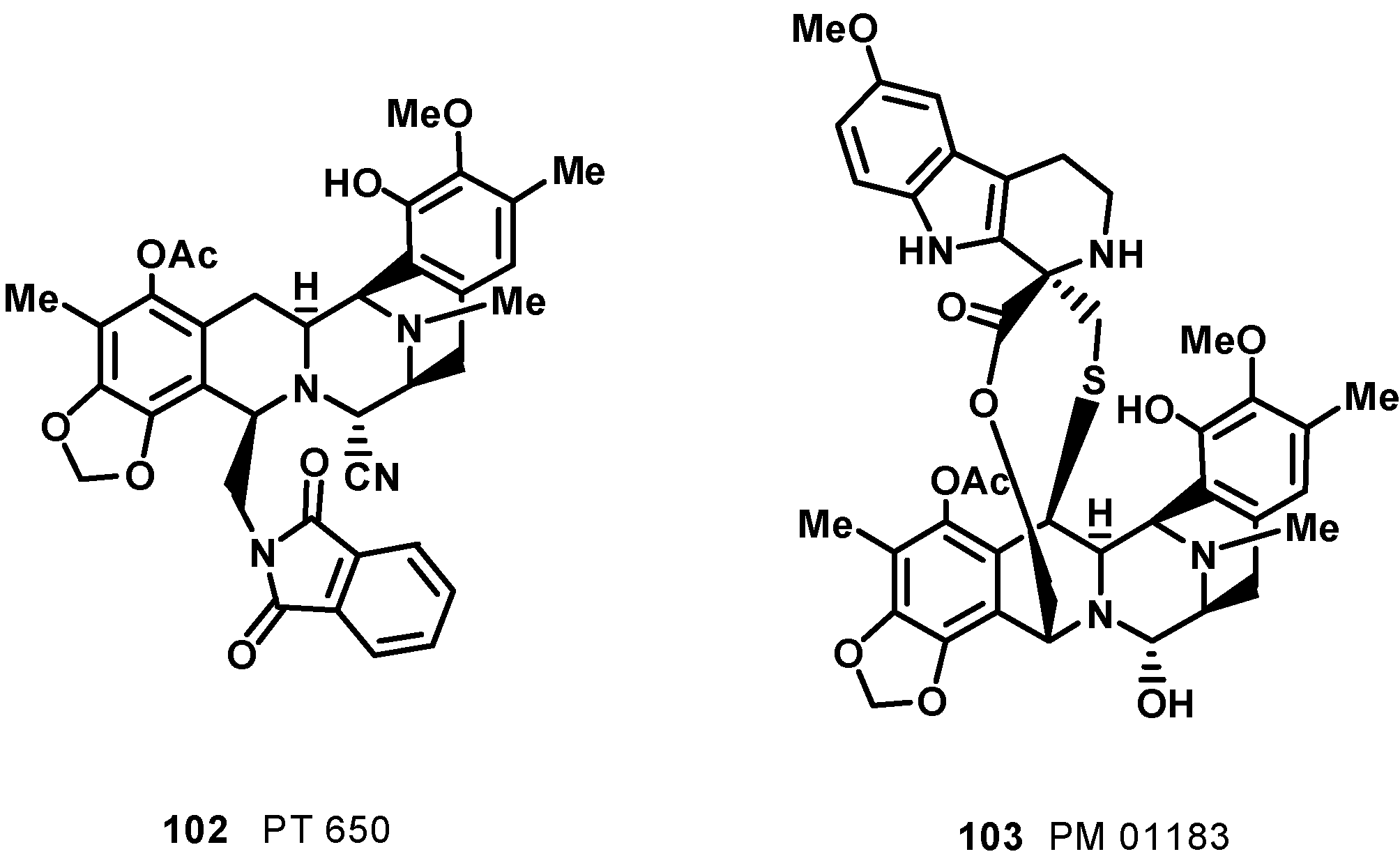
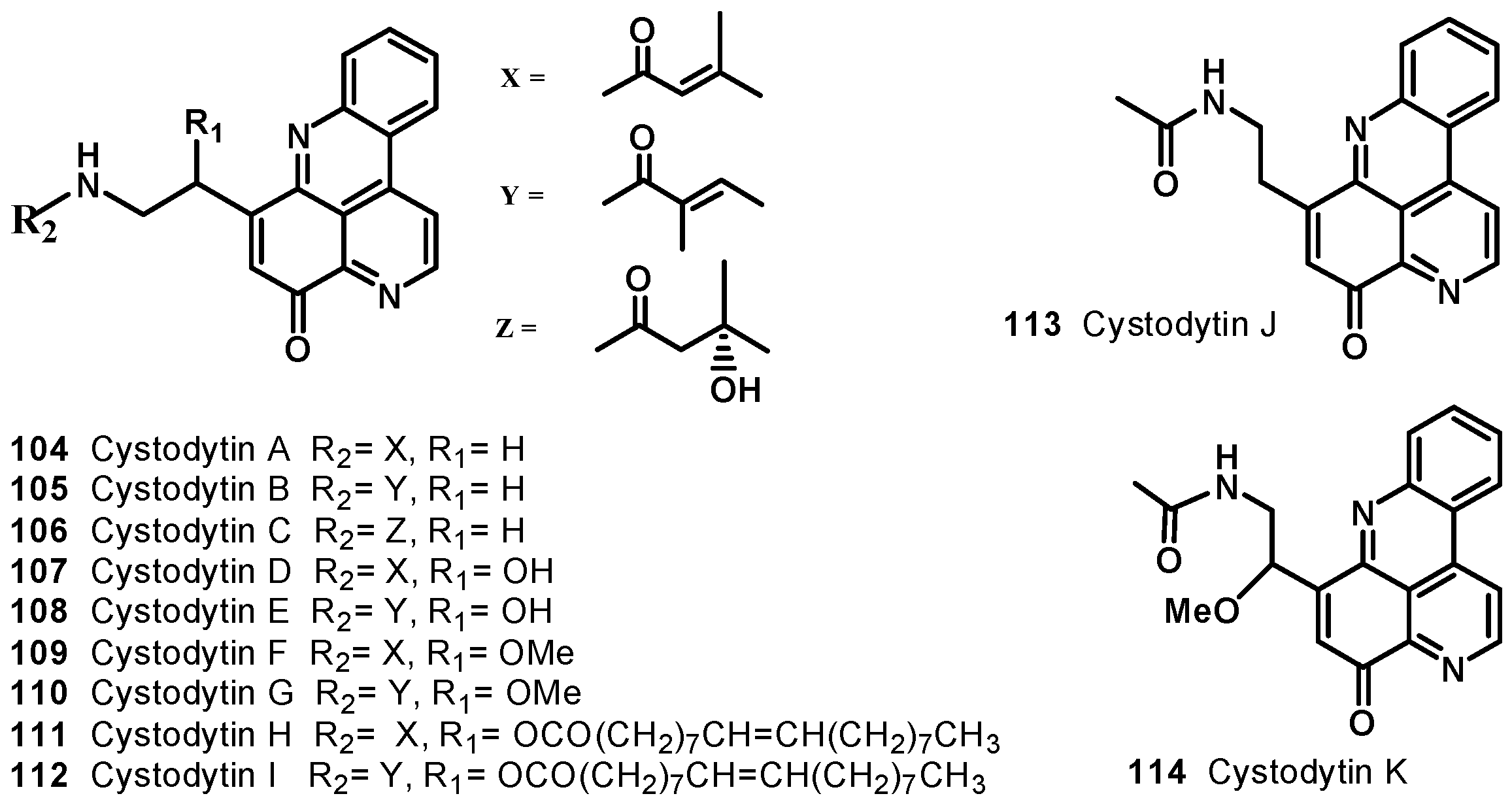
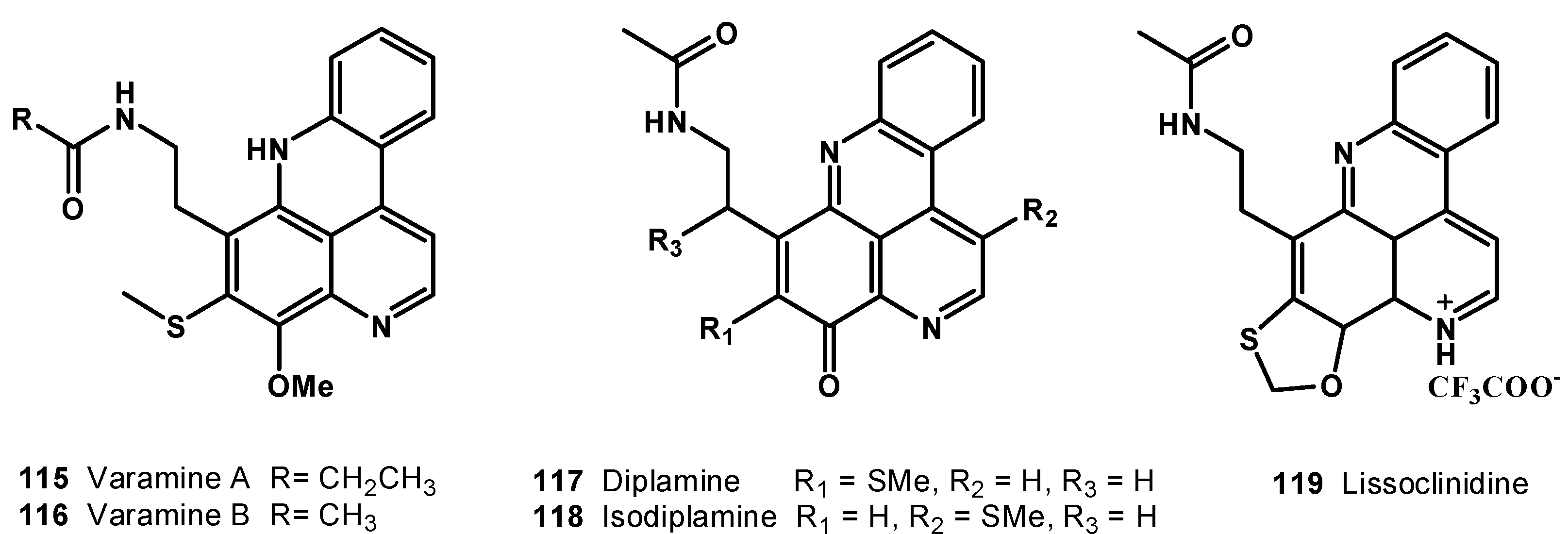
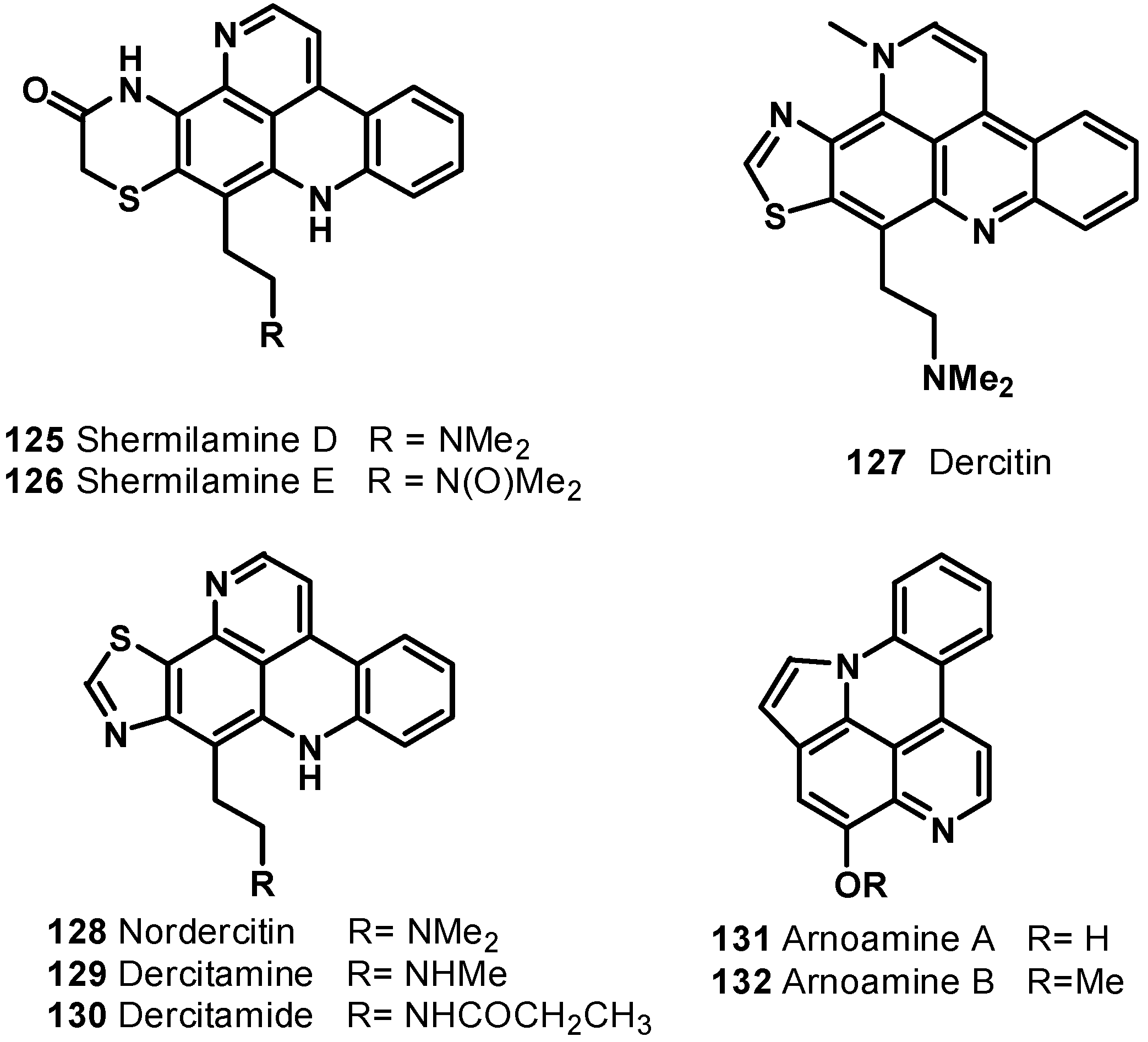
7. Pyridoacridine Alkaloids: DNA Intercalating Agents with Reactive Oxygen Species (ROS) Producing and TOPO-Inhibiting Activities


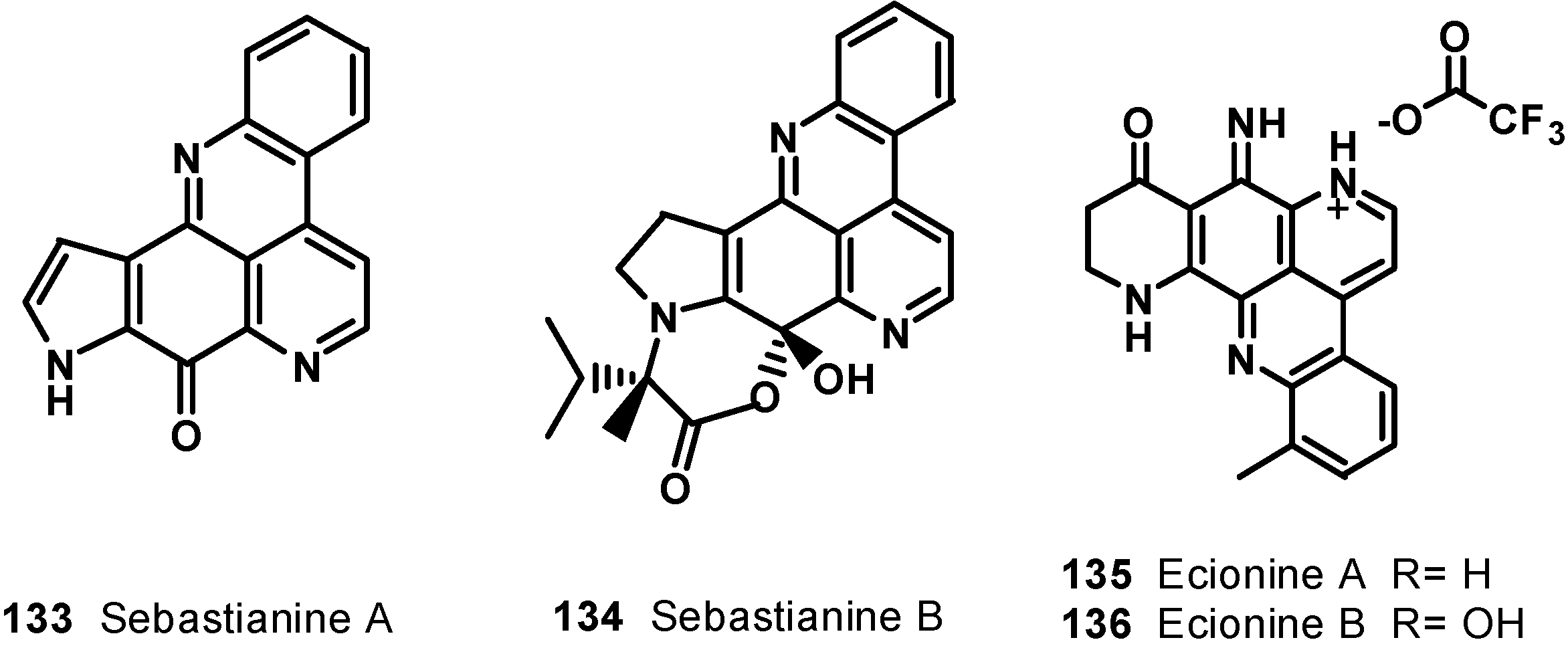
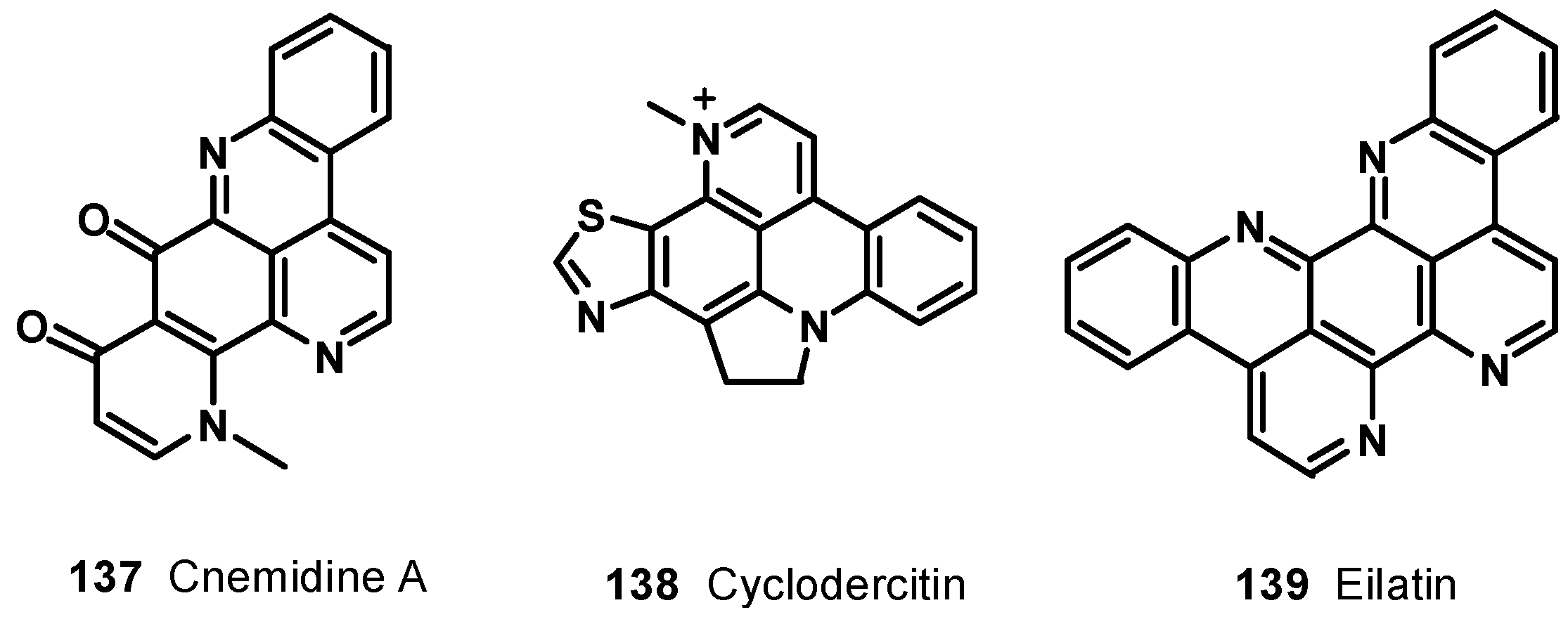
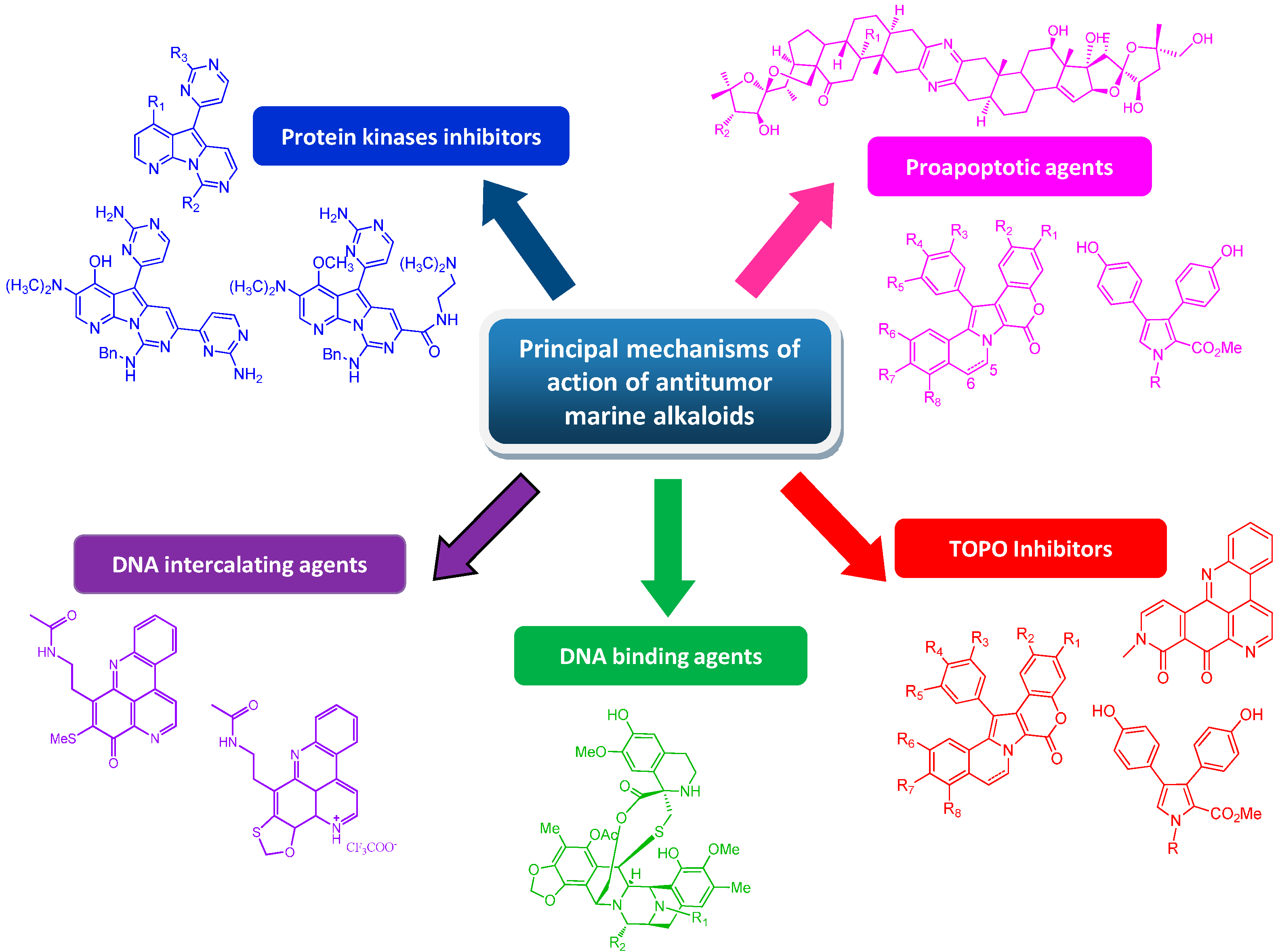
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Perry, N.B.; Ettoouati, L.; Litaudon, M.; Blunt, J.W.; Munro, M.H.G.; Parkin, S.; Hope, H. Alkaloids from the antarctic sponge Kirkpatrickia varialosa. Part 1: Variolin B, a new antitumor and antiviral compound. Tetrahedron 1994, 50, 3987–3992. [Google Scholar] [CrossRef]
- Trimurtulu, G.; Faulkner, D.J.; Perry, N.B.; Ettouati, L.; Litaudon, M.; Blunt, J.W.; Munro, M.H.G.; Jameson, G.B. Alkaloids from the antarctic sponge Kirkpatrickia varialosa. Part 2: Variolin A and N(3')-methyl tetrahydrovariolin B. Tetrahedron 1994, 50, 3993–4000. [Google Scholar] [CrossRef]
- Walker, S.R.; Carter, E.J.; Huff, B.C.; Morris, J.C. Variolins and Related Alkaloids. Chem. Rev. 2009, 109, 3080–3098. [Google Scholar] [CrossRef]
- Anderson, R.J.; Morris, J.C. Total synthesis of variolin B. Tetrahedron Lett. 2001, 42, 8697–8699. [Google Scholar] [CrossRef]
- Anderson, R.J.; Hill, J.B.; Morris, J.C. Concise Total Syntheses of Variolin B and Deoxyvariolin B. J. Org. Chem. 2005, 70, 6204–6212. [Google Scholar] [CrossRef]
- Simone, M.; Erba, E.; Damia, G.; Vikhanskaya, F.; Francesco, A.M.D.; Riccardi, R.; Bailly, C.; Cuevas, C.; Sousa-Faro, J.M.F.; D’Incalci, M. Variolin B and its derivate deoxy-variolin B: New marine natural compounds with cyclin-dependent kinase inhibitor activity. Eur. J. Cancer 2005, 41, 2366–2377. [Google Scholar] [CrossRef]
- Bettayeb, K.; Tirado, O.M.; Marionneau-Lambot, S.; Ferandin, Y.; Lozach, O.; Morris, J.C.; Mateo-Lozano, S.; Drueckes, P.; Schächtele, C.; Kubbutat, M.H.G.; et al. Meriolins, a new class of cell death-inducing Kinase inhibitors with enhanced selectivity for cyclin-dependent Kinases. Cancer Res. 2007, 67, 8325–8334. [Google Scholar] [CrossRef]
- Echalier, A.; Bettayeb, K.; Ferandin, Y.; Lozach, O.; Clement, M.; Valette, A.; Liger, F.; Marquet, B.; Morris, J.C.; Endicott, J.A.; et al. Meriolins (3-(Pyrimidin-4-yl)-7-azaindoles): Synthesis, Kinase Inhibitory Activity, Cellular Effects, and Structure of a CDK2/Cyclin A/Meriolin Complex. J. Med. Chem. 2008, 51, 737–751. [Google Scholar] [CrossRef]
- Anderson, R.J.; Manzanares, I.; Morris, J.C.; Remuinan, M. Variolin Derivatives as Anti-Cancer Agents. WO 0204447 A1, 17 January 2002. [Google Scholar]
- Remuinan, M.; Gonzalez, J.; del Pozo, C.; Francesch, A.; Cuevas, C.; Munt, S.; Manzanares, I.; Morris, J.C.; Anderson, R. Variolin Derivatives and Their Use as Antitumour Agents. WO 2003006457, 23 January 2003. [Google Scholar]
- Alvarez, M.; Fernandez Bleda, D.; Fernandez Puentes, J.L. Preparation of Variolin B Derivatives as Antitumor Agents. WO 2002012240 A1, 14 February 2002. [Google Scholar]
- Fresneda, P.M.; Delgado, S.; Francesch, A.; Manzanares, I.; Cuevas, C.; Molina, P. Synthesis and Cytotoxic Evaluation of New Derivatives of the Marine Alkaloid Variolin B. J. Med. Chem. 2006, 49, 1217–1221. [Google Scholar] [CrossRef]
- Remuinan, M.; Gonzalez, J.; del Pozo, C.; Francesh, A.; Cuevas, C.; Munt, S.; Manzanares, I.; Anderson, R.J.; Morris, J.C. Variolin Derivatives and their Use as Antitumor Agents. U.S. Patent 7772241, 10 August 2010. [Google Scholar]
- Franco, L.H.; Joffe, E.B.; Puricelli, L.; Tatian, M.; Seldes, A.M.; Palermo, J.A. Indole alkaloids from the tunicate Aplidium meridianum. J. Nat. Prod. 1998, 61, 1130–1132. [Google Scholar] [CrossRef]
- Seldes, A.M.; Brasco, M.F.R.; Franco, L.H.; Palermo, J.A. Identification of two meridianins from the crude extract of the tunicate Aplidium meridianum by tandem mass spectrometry. Nat. Prod. Res. 2007, 2, 555–563. [Google Scholar] [CrossRef]
- Gompel, M.; Leost, M.; de Kier, J.E.B.; Puricelli, L.; Franco, L.H.; Palermo, J.; Meijer, L. Meridianins, a new family of protein kinase inhibitors isolated from the ascidian Aplidium meridianum. Bioorg. Med. Chem. Lett. 2004, 14, 1703–1707. [Google Scholar] [CrossRef]
- Reyes, F.; Fernandez, R.; Rodriguez, A.; Francesch, A.; Taboada, S.; Avila, C.; Cuevas, C. Aplicyanins A-F, new cytotoxic bromoindole derivatives from the marine tunicate Aplidium cyaneum. Tetrahedron 2008, 64, 5119–5123. [Google Scholar] [CrossRef]
- Bartik, K.; Braekman, J.-C.; Daloze, D.; Stoller, C.; Huysecom, J.; Vandevyver, G.; Ottinger, R. Topsentins, new toxic bis-indole alkaloids from the marine sponge Topsentia genitrix. Can. J. Chem. 1987, 65, 2118–2121. [Google Scholar] [CrossRef]
- Murray, L.M.; Lim, T.K.; Hooper, J.N.A.; Capon, R.J. Isobromotopsentin: A new bis(indole) alkaloid from a deep-water marine sponge Spongosorites sp. Aust. J. Chem. 1995, 48, 2053–2058. [Google Scholar] [CrossRef]
- Tsujii, S.; Rinehart, K.L.; Gunasekera, S.P.; Kashman, Y.; Cross, S.S.; Lui, M.S.; Pomponi, S.A.; Diaz, M.C. Topsentin, bromotopsentin, and dihydrodeoxybromotopsentin: Antiviral and antitumor bis(indolyl)imidazoles from Caribbean deep-sea sponges of the family Halichondriidae. Structural and synthetic studies. J. Org. Chem. 1988, 53, 5446–5453. [Google Scholar] [CrossRef]
- Casapullo, A.; Bifulco, G.; Bruno, I.; Riccio, R. New bisindole alkaloids of the Topsentin and Hamacanthin classes from the Mediterranean marine sponge Rhaphisia lacazei. J. Nat. Prod. 2000, 63, 447–451. [Google Scholar] [CrossRef]
- Morris, S.A.; Andersen, R.J. Nitrogenous metabolites from the deep water sponge Hexadella sp. Can. J. Chem. 1989, 67, 677–681. [Google Scholar] [CrossRef]
- Shin, J.; Seo, Y.; Cho, K.W.; Rho, J.-R.; Sim, C.J. New Bis(Indole) Alkaloids of the Topsentin class from the sponge Spongosorites genitrix. J. Nat. Prod. 1999, 62, 647–649. [Google Scholar] [CrossRef]
- Sakemi, S.; Sun, H.H. Nortopsentins A, B, and C. Cytotoxic and antifungal imidazolediylbis[indoles] from the sponge Spongosorites ruetzleri. J. Org. Chem. 1991, 56, 4304–4307. [Google Scholar]
- Sun, H.H.; Sakemi, S.; Gunasekera, S.; Kashman, Y.; Lui, M.; Burres, N.; Mc Carthy, P. Bisindole Compounds which are Useful Antitumor and Antimicrobial Agents. U.S. Patent 4970226, 13 November 1990. [Google Scholar]
- Mancini, I.; Guella, G.; Debitus, C.; Waikedre, J.; Pietra, F. From inactive nortopsentin D, a novel bis(indole) alkaloid isolated from the axinellid sponge Dragmacidon sp. from deep waters south of new caledonia, to a strongly cytotoxic derivative. Helv. Chim. Acta 1996, 79, 2075–2082. [Google Scholar] [CrossRef]
- Kawasaki, I.; Yamashita, M.; Ohta, S. Total synthesis of nortopsentins A-D, marine alkaloids. Chem. Pharm. Bull. 1996, 44, 1831–1839. [Google Scholar] [CrossRef]
- Jiang, B.; Gu, X.-H. Syntheses and cytotoxicity evaluation of bis(indolyl)thiazole, bis(indolyl)pyrazinone and bis(indolyl)pyrazine: Analogues of cytotoxic marine bis(indole) alkaloid. Bioorg. Med. Chem. 2000, 8, 363–371. [Google Scholar] [CrossRef]
- Moser, B.R. Review of cytotoxic cephalostatins and ritterazines: Isolation and synthesis. J. Nat. Prod. 2008, 71, 487–491. [Google Scholar] [CrossRef]
- Lee, S.; LaCour, T.G.; Fuchs, P.L. Chemistry of trisdecacyclic pyrazine antineoplastics: The cephalostatins and ritterazines. Chem. Rev. 2009, 109, 2275–2314. [Google Scholar] [CrossRef]
- Menna, M.; Fattorusso, E.; Imperatore, C. Alkaloids from Marine Ascidians. Molecules 2011, 16, 8694–8732. [Google Scholar] [CrossRef]
- Komiya, T.; Fusetani, N.; Matsunaga, S.; Kubo, A.; Kaye, F.J.; Kelley, M.J.; Tamura, K.; Yoshida, M.; Fukuoka, M.; Nakagawa, K. Ritterazine B, a new cytotoxic natural compound, induces apoptosis in cancer cells. Cancer Chemother. Pharmacol. 2003, 51, 202–208. [Google Scholar]
- Ganesan, A. The dimeric steroid-pyrazine marine alkaloids: Challenges for isolation, synthesis, and biological studies. Angew. Chem. Int. Ed. Engl. 1996, 35, 611–615. [Google Scholar] [CrossRef]
- López-Antón, N.; Rudy, A.; Barth, N.; Schmitz, L.M.; Pettit, G.R.; Schulze-Osthoff, K.; Dirsch, V.M.; Vollmar, A.M. The marine product cephalostatin 1 activates and ER stress-specific and apoptosome- independent apoptotic signaling pathway. J. Biol. Chem. 2006, 281, 33078–33086. [Google Scholar]
- Rudy, A.; López-Antón, N.; Dirsch, V.M.; Vollmar, A.M. The Cephalostatin Way of Apoptosis. J. Nat. Prod. 2008, 71, 482–486. [Google Scholar] [CrossRef]
- Rudy, A.; López-Antón, N.; Barth, N.; Pettit, G.R.; Dirsch, V.M.; Schulze-Osthoff, K.; Rehm, M.; Prehn, J.H.M.; Vogler, M.; Fulda, S.; et al. Role of Smac in cephalostatin-induced cell death. Cell Death Differ. 2008, 15, 1930–1940. [Google Scholar] [CrossRef]
- Cironi, P.; Albericio, F.; Alvarez, M. Lamellarins: Isolation, activity and synthesis. Prog. Heterocycl. Chem. 2004, 16, 1–26. [Google Scholar]
- Bailly, C. Lamellarins, from A to Z: A family of anticancer marine pyrrole alkaloids. Curr. Med. Chem. Anti-Cancer Agents 2004, 4, 363–378. [Google Scholar] [CrossRef]
- Fan, H.; Peng, J.; Hamann, M.T.; Hu, J.F. Lamellarins and related pyrrole-derived alkaloids from marine organisms. Chem. Rev. 2008, 108, 264–287. [Google Scholar] [CrossRef]
- Kluza, J.; Marchetti, P.; Bailly, C. Lamellarin alkaloids: Structure and pharmacological properties. In Modern Alkaloids: Structure, Isolation, Synthesis and Biology; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008; pp. 171–187. [Google Scholar]
- Pla, D.; Albericio, F.; Alvarez, M. Progress on lamellarins. Med. Chem. Commun. 2011, 2, 689–697. [Google Scholar] [CrossRef]
- Andersen, R.J.; Faulkner, D.J.; He, C.H.; van Duyne, G.D.; Clardy, J. Metabolites of the marine prosobranch mollusk Lamellaria sp. J. Am. Chem. Soc. 1985, 107, 5492–5495. [Google Scholar] [CrossRef]
- Lindquist, N.; Fenical, W.; van Duyne, G.D.; Clardy, J. New alkaloids of the Lamellarin class from the marine ascidian Didemnum chartaceum. J. Org. Chem. 1988, 53, 4570–4574. [Google Scholar] [CrossRef]
- Carroll, A.R.; Bowden, B.F.; Coll, J.C. Studies of Australian ascidians. I. Six new lamellarin class alkaloids from a colonial ascidian, Didemnum sp. Aust. J. Chem. 1993, 46, 489–501. [Google Scholar] [CrossRef]
- Davis, R.A.; Carroll, A.R.; Pierens, G.K.; Quinn, R.J. New lamellarin alkaloids from the Australian ascidian Didemnum chartaceum. J. Nat. Prod. 1999, 62, 419–424. [Google Scholar] [CrossRef]
- Reddy, M.V.R.; Rao, M.R.; Rhodes, D.; Hansen, M.S.T.; Rubins, K.; Bushman, F.D.; Venkateswarlu, Y.; Faulkner, D.J. Lamellarin α-20 sulfate, an inhibitor of HIV-1 integrase active against HIV-1 virus in cell culture. J. Med. Chem. 1999, 42, 1901–1907. [Google Scholar] [CrossRef]
- Urban, S.; Capon, R.J. Lamellarin-S: A new aromatic metabolite from an Australian Tunicate, Didemnum sp. Aust. J. Chem. 1996, 49, 711–713. [Google Scholar]
- Reddy, S.M.; Srinivasulu, M.; Satyanarayana, N.; Kondapi, A.K.; Venkateswarlu, Y. New potent cytotoxic lamellarin alkaloids from Indian ascidian Didemnum obscurum. Tetrahedron 2005, 61, 9242–9247. [Google Scholar] [CrossRef]
- Urban, S.; Butler, M.S.; Capon, R.J. Lamellarins O and P: New aromatic metabolites from the Australian marine sponge Dendrilla cactos. Aust. J. Chem. 1994, 47, 1919–1924. [Google Scholar] [CrossRef]
- Urban, S.; Hobbs, L.; Hooper, J.N.A.; Capon, R.J. Lamellarins Q and R: New aromatic metabolites from an Australian marine Sponge Dendrilla cactos. Aust. J. Chem. 1995, 48, 1491–1494. [Google Scholar] [CrossRef]
- Quesada, A.R.; Garcia Gravalos, M.D.; Fernandez Puentes, J.L. Polyaromatic alkaloids from marine invertebrates as cytotoxic compounds and inhibitors of multidrug resistance caused by Pglycoprotein. Br. J. Cancer 1996, 74, 677–682. [Google Scholar] [CrossRef]
- Urban, S.; Hickford, S.J.H.; Blunt, J.W.; Munro, M.H.G. Bioactive marine alkaloids. Curr. Org. Chem. 2000, 4, 765–807. [Google Scholar] [CrossRef]
- Facompre, M.; Tardy, C.; Bal-Mahieu, C.; Colson, P.; Perez, C.; Manzanares, I.; Cuevas, C.; Bailly, C. Lamellarin D: A novel potent inhibitor of topoisomerase I. Cancer Res. 2003, 63, 7392–7399. [Google Scholar]
- Tardy, C.; Facompre, M.; Laine, W.; Baldeyrou, B.; Garcia-Gravalos, D.; Francesch, A.; Mateo, C.; Pastor, A.; Jimenez, J.A.; Manzanares, I.; et al. Topoisomerase I-mediated DNA cleavage as a guide to the development of antitumor agents derived from the marine alkaloid lamellarin D: Trimethylester derivatives incorporating amino acid residues. Bioorg. Med. Chem. 2004, 12, 1697–1712. [Google Scholar] [CrossRef]
- Marco, E.; Laine, W.; Tardy, C.; Lansiaux, A.; Iwao, M.; Ishibashi, F.; Bailly, C.; Gago, F. Molecular determinants of topoisomerase I poisoning by lamellarins: Comparison with camptothecin and structure-activity relationships. J. Med. Chem. 2005, 48, 3796–3807. [Google Scholar] [CrossRef]
- Vanhuyse, M.; Kluza, J.; Tardy, C.; Otero, G.; Cuevas, C.; Bailly, C.; Lansiaux, A. Lamellarin D: A novel pro-apoptotic agent from marine origin insensitive to P-glycoprotein-mediated drug efflux. Cancer Lett. 2005, 22, 165–175. [Google Scholar] [CrossRef]
- Kluza, J.; Gallego, M.A.; Loyens, A.; Beauvillain, J.C.; Sousa- Faro, J.M.; Cuevas, C.; Marchetti, P.; Bailly, C. Cancer cell mitochondria are direct proapoptotic targets for the marine antitumor drug lamellarin D. Cancer Res. 2006, 66, 3177–3187. [Google Scholar] [CrossRef]
- Gallego, M.A.; Ballot, C.; Kluza, J.; Hajji, N.; Martoriati, A.; Castéra, L.; Cuevas, C.; Formstecher, P.; Joseph, B.; Kroemer, G.; et al. Overcoming chemoresistance of non-small cell lung carcinoma through restoration of an AIF-dependent apoptotic pathway. Oncogene 2008, 27, 1981–1992. [Google Scholar] [CrossRef]
- Pla, D.; Marchal, A.; Olsen, C.A.; Francesch, A.; Cuevas, C.; Albericio, F.; Alvarez, M. Synthesis and Structure Activity Relationship Study of Potent Cytotoxic Analogues of the Marine Alkaloid Lamellarin D. J. Med. Chem. 2006, 49, 3257–3268. [Google Scholar] [CrossRef]
- Chittchang, M.; Batsomboon, P.; Ruchirawat, S.; Ploypradith, P. Cytotoxicities and structure—Activity relationships of natural and unnatural lamellarins toward cancer cell Lines. Chem. Med. Chem. 2009, 4, 457–465. [Google Scholar] [CrossRef]
- Khiati, S.; Seol, Y.; Agama, K.; Dalla Rosa, I.; Agrawal, S.; Fesen, K.; Zhang, H.; Neuman, K.C.; Pommie, Y. Poisoning of mitochondrial topoisomerase I by lamellarin D. Mol. Pharmacol. 2014, 86, 193–199. [Google Scholar] [CrossRef]
- Chittchang, M.; Gleeson, M.P.; Ploypradith, P.; Ruchirawat, S. Assessing the drug-likeness of lamellarins, a marine-derived natural product class with diverse oncological activities. Eur. J. Med. Chem. 2010, 45, 2165–2172. [Google Scholar] [CrossRef]
- Le, V.H.; Inai, M.; Williams, R.M.; Kan, T. Ecteinascidins. A review of the chemistry, biology and clinical utility of potent tetrahydroisoquinoline antitumor antibiotics. Nat. Prod. Rep. 2014. [Google Scholar] [CrossRef]
- Garcia-Carbonero, R.; Supko, J.G.; Maki, R.G.; Manola, J.; Ryan, D.P.; Harmon, D.; Puchalski, T.A.; Goss, G.; Seiden, M.V.; Waxman, A.; et al. Ecteinascidin-743 (ET-743) for chemotherapy-naive patients with advanced soft tissue sarcomas: Multicenter phase II and pharmacokinetic study. J. Clin. Oncol. 2005, 23, 5484–5492. [Google Scholar] [CrossRef]
- Cuevas, C.; Francesch, A. Development of Yondelis® (trabectedin, ET-743). A semisynthetic process solves the supply problem. Nat. Prod. Rep. 2009, 26, 322–337. [Google Scholar] [CrossRef]
- Krasner, C.N.; McMeekin, D.S.; Chan, S.; Braly, P.S.; Renshaw, F.G.; Kaye, S.; Provencher, D.M.; Campos, S.; Gore, M.E. A Phase II study of trabectedin single agent in patients with recurrent ovarian cancer previously treated with platinum-based regimens. Br. J. Cancer 2007, 97, 1618–1624. [Google Scholar] [CrossRef]
- Rinehart, K.L.; Holt, T.G.; Fregeau, N.L.; Stroh, J.G.; Keifer, P.A.; Sun, F.; Li, L.H.; Martin, D.G. Ecteinascidins 729, 743, 745, 759A. 759B, and 770: Potent antitumor agents from the Caribbean tunicate Ecteinascidia turbinata. J. Org. Chem. 1990, 55, 4512–4515. [Google Scholar] [CrossRef]
- Wright, A.E.; Forleo, D.A.; Gunawardana, G.P.; Gunasekera, S.P.; Koehn, F.E.; McConnell, O.J. Antitumor tetrahydroisoquinoline alkaloids from the colonial ascidian Ecteinascidia turbinata. J. Org. Chem. 1990, 55, 4508–4512. [Google Scholar] [CrossRef]
- Guan, Y.; Sakai, R.; Rinehart, K.L.; Wang, A.H.J. Molecular and crystal structures of ecteinascidins: Potent antitumor compounds from the Caribbean tunicate Ecteinascidia turbinata. J. Biomol. Struct. Dyn. 1993, 10, 793–818. [Google Scholar]
- Sakai, R.; Rinehart, K.L.; Guan, Y.; Wang, A.H.J. Additional antitumor ecteinascidins from a Caribbean tunicate: Crystal structures and activities in vivo. Proc. Natl. Acad. Sci. USA 1992, 89, 11456–11460. [Google Scholar] [CrossRef]
- Rinehart, K.L.; Sakai, R. Isolation, Structure Elucidation, and Bioactivities of Novel Ecteinascidins from Ecteinascidia turbinata. U.S. Patent 20040059112 Al, 25 March 2004. [Google Scholar]
- Suwanborirux, K.; Charupant, K.; Amnuoypol, S.; Pummangura, S.; Kubo, A.; Saito, N. Ecteinascidins 770 and 786 from the Thai Tunicate Ecteinascidia thurstoni. J. Nat. Prod. 2002, 65, 935–937. [Google Scholar] [CrossRef]
- Sakai, R.; Jares-Erijman, E.A.; Manzanares, I.; Elipe, M.V.S.; Rinehart, K.L. Ecteinascidins: Putative biosynthetic precursors and absolute stereochemistry. J. Am. Chem. Soc. 1996, 118, 9017–9023. [Google Scholar] [CrossRef]
- Scott, J.D.; Williams, R.M. Chemistry and Biology of the Tetrahydroisoquinoline Antitumor Antibiotics. Chem. Rev. 2002, 102, 1669–1730. [Google Scholar] [CrossRef]
- Soares, D.G.; Larsen, A.K.; Escargueil, A.E. The DNA damage response to monofunctional anticancer DNA binders. Drug Discov. Today: Dis. Models 2012, 9, e59–e67. [Google Scholar] [CrossRef]
- Aune, G.J.; Furuta, T.; Pommier, Y. Ecteinascidin 743: A novel anticancer drug with a unique mechanism of action. Anticancer Drugs 2002, 13, 545–555. [Google Scholar] [CrossRef]
- Zewail-Foote, M.; Hurley, L.H. Ecteinascidin 743: A minor groove alkylator that bends DNA toward the major groove. J. Med. Chem. 1999, 42, 2493–2497. [Google Scholar] [CrossRef]
- Takebayashi, Y.; Pourquier, P.; Zimonjic, D.B.; Nakayama, K.; Emmert, S.; Ueda, T.; Urasaki, Y.; Kanzaki, A.; Akiyama, S.-I.; Popescu, N.; et al. Antiproliferative activity of ecteinascidin 743 is dependent upon transcription- coupled nucleotide-excision repair. Nat. Med. 2001, 7, 961–966. [Google Scholar] [CrossRef]
- D’Incalci, M.; Erba, E.; Damia, G.; Galliera, E.; Carrassa, L.; Marchini, S.; Mantovani, R.; Tognon, G.; Fruscio, R.; Jimeno, J.; et al. Unique features of the mode of action of ET-743. Oncologist 2002, 7, 210–216. [Google Scholar] [CrossRef]
- Fayette, J.; Coquard, I.R.; Alberti, L.; Boyle, H.; Meeus, P.; Decouvelaere, A.V.; Thiesse, P.; Sunyach, M.P.; Ranchere, D.; Blay, J.Y. ET-743: A novel agent with activity in soft-tissue sarcomas. Curr. Opin. Oncol. 2006, 18, 347–353. [Google Scholar] [CrossRef]
- Christinat, A.; Leyvraz, S. Role of trabectedin in the treatment of soft tissue sarcoma. Onco Targets Ther. 2009, 2, 105–113. [Google Scholar]
- Gajdos, C.; Elias, A. Trabectedin: Safety and efficacy in the treatment of advanced sarcoma. Clin. Med. Insight Oncol. 2011, 5, 35–43. [Google Scholar] [CrossRef]
- Pharmamar. Available online: http://www.yondelis.com/yondelis.aspx (accessed on 18 December 2013).
- Carballo, J.L.; Naranho, S.; Kukurtzu, B.; de La Calle, F.; Hernandez-Zanuy, A. Production of Ecteinascidia turbinate (Ascidiacea: Perophoridae) for obtaining anticancer compounds. J. World Aquac. Soc. 2000, 31, 481–490. [Google Scholar] [CrossRef]
- Corey, E.J.; Gin, D.Y.; Kania, R.S. Enantioselective total synthesis of Ecteinascidin 743. J. Am. Chem. Soc. 1996, 118, 9202–9203. [Google Scholar] [CrossRef]
- Martinez, E.; Corey, E.J. A New, More Efficient, and Effective Process for the Synthesis of a Key Pentacyclic Intermediate for Production of Ecteinascidin and Phthalascidin Antitumor Agents. Org. Lett. 2000, 2, 993–996. [Google Scholar] [CrossRef]
- Endo, A.; Yanagisawa, A.; Abe, M.; Tohma, S.; Kan, T.; Fukuyama, T. Total Synthesis of Ecteinascidin 743. J. Am. Chem. Soc. 2002, 124, 6552–6554. [Google Scholar] [CrossRef]
- Chen, J.; Chen, X.; Bois-Choussy, M.; Zhu, J. Total Synthesis of Ecteinascidin 743. J. Am. Chem. Soc. 2006, 128, 87–89. [Google Scholar] [CrossRef]
- Zheng, S.; Chan, C.; Furuuchi, T.; Wright, B.J.D.; Zhou, B.; Gio, Z.J.; Danishefsky, S.J. Stereospecific Formal Total Synthesis of Ecteinascidin 743. Angew. Chem. Int. Ed. 2006, 45, 1754–1759. [Google Scholar] [CrossRef]
- Fishlock, D.; Williams, R.M. Synthetic Studies on Et-743. Assembly of the Pentacyclic core and a formal total synthesis. J. Org. Chem. 2008, 73, 9594–9600. [Google Scholar] [CrossRef]
- Avendaño, C.; de la Cuesta, E. Recent synthetic approaches to 6,15-inoisoquino[3,2-b]3-benzazocine compounds. Chem. Eur. J. 2010, 16, 9722–9734. [Google Scholar] [CrossRef]
- Cuevas, C.; Perez, M.; Martin, M.J.; Chicharro, J.L.; Fernandez-Rivas, C.; Flores, M.; Francesch, A.; Gallego, P.; Zarzuelo, M.; de la Calle, F.; et al. Synthesis of ecteinascidin ET-743 and phthalascidin Pt-650 from cyanosafracin B. Org Lett. 2000, 2, 2545–2548. [Google Scholar] [CrossRef]
- Menchaca, R.; Martínez, V.; Rodríguez, A.; Rodríguez, N.; Flores, M.; Gallego, P.; Manzanares, I.; Cuevas, C. Synthesis of natural Ecteinascidins (ET-729, ET-745, ET-759B, ET-736, ET-637, ET-594) from Cyanosafracin B. J. Org. Chem. 2003, 68, 8859–8866. [Google Scholar] [CrossRef]
- Martinez, E.J.; Owa, T.; Schreiber, S.L.; Corey, E.J. Phtalascidin, a synthetic antitumor agent with potency and mode of action comparable to ecteinascidin 743. Proc. Natl. Acad. Sci. USA 1999, 96, 3496–3501. [Google Scholar] [CrossRef]
- Garcia-Nieto, R.; Manzanares, I.; Cuevas, C.; Gago, F. Bending of DNA upon binding of ecteinascidin 743 and phthalascidin 650 studied by unrestrained molecular dynamics simulations. J. Am. Chem. Soc. 2000, 122, 7172–7182. [Google Scholar] [CrossRef]
- Martinez, E.J.; Corey, E.J.; Owa, T. Antitumor activity- and gene expression-based profiling of ecteinascidin Et743 and phtalascidin Pt 650. Chem. Biol. 2001, 8, 1151–1160. [Google Scholar] [CrossRef]
- Leal, J.F.M.; Martínez-Díez, M.; García-Hernández, V.; Moneo, V.; Domingo, A; Bueren-Calabuig, J.A.; Negri, A.; Gago, F.; Guillén-Navarro, M.J.; Avilés, P.; et al. PM01183, a new DNA minor groove covalent binder with potent in vitro and in vivo anti-tumour activity. Br. J. Pharmacol. 2010, 161, 1099–1110. [Google Scholar] [CrossRef]
- Soares, D.G.; Machado, M.S.; Rocca, C.J.; Poindessous, V.; Ouaret, D.; Sarasin, A.; Galmarini, C.M.; Henriques, G.A.P.; Escargueil, A.E.; Larsen, A.K. Trabectedin and Its C Subunit Modified Analogue PM01183 Attenuate Nucleotide Excision Repair and Show Activity toward Platinum-Resistant Cells. Mol. Cancer. Ther. 2011, 10, 1481–1489. [Google Scholar] [CrossRef]
- Pharmamar. Available online: http://www.pharmamar.com/busqueda.aspx (accessed on 29 August 2012).
- Delfourne, E.; Bastide, J. Marine pyridoacridine alkaloids and synthetic analogues as antitumor agents. Med. Res. Rev. 2003, 23, 234–252. [Google Scholar] [CrossRef]
- Lederer, E.; Teissier, G.; Huttrer, C. The isolation and chemical composition of calliactine, pigment of the sea anemone “Sagartia parasitica” (Calliactis effoeta). Bull. Soc. Chim. Fr. 1940, 7, 608–615. [Google Scholar]
- Schmitz, F.J.; Agarwal, S.K.; Gunasekera, S.P.; Schmidt, P.G.; Shoolery, J.N. Amphimedine, new aromatic alkaloid from a pacific sponge, Amphimedon sp. Carbon connectivity determination from natural abundance carbon-13-carbon-13 coupling constants. J. Am. Chem. Soc. 1983, 105, 4835–4836. [Google Scholar]
- Molinski, T.F. Marine pyridoacridine alkaloids: Structure, synthesis, and biological chemistry. Chem. Rev. 1993, 93, 1825–1838. [Google Scholar] [CrossRef]
- Bontemps, N.; Gattacceca, F.; Long, C.; Thomas, O.P.; Banaigs, B. Additional Cytotoxic Pyridoacridine Alkaloids from the Ascidian Cystodytes violatinctus and Biogenetic Considerations. J. Nat. Prod. 2013, 76, 1801–1805. [Google Scholar] [CrossRef]
- Marshall, K.M.; Barrows, L.R. Biological activities of pyridoacridines. Nat. Prod. Rep. 2004, 21, 731–751. [Google Scholar] [CrossRef]
- De Guzman, F.S.; Carte, B.; Troupe, N.; Faulkner, D.J.; Harper, M.K.; Concepcion, G.P.; Mangalindan, G.C.; Matsumoto, S.S.; Barrows, L.R.; Ireland, C.M. Neoamphimedine: A new pyridoacridine topoisomerase II inhibitor which catenates DNA. J. Org. Chem. 1999, 64, 1400–1402. [Google Scholar] [CrossRef]
- Feng, Y.R.; Davis, A.; Sykes, M.L.; Avery, V.M.; Carroll, A.R.; Camp, D.; Quinn, R.J. Antitrypanosomal pyridoacridine alkaloids from the Australian ascidian Polysyncraton echinatum. Tetrahedron Lett. 2010, 51, 2477–2479. [Google Scholar] [CrossRef]
- Fuente, J.A.D.L.; Martın, M.J.; Blanco, M.D.M.; Alfonso, E.P.; Avendano, C.; Menendez, J.C. A C-Ring regioisomer of the marine alkaloid meridine exhibits selective in vitro cytotoxicity for solid tumours. Bioorg. Med. Chem. 2001, 9, 1807–1814. [Google Scholar] [CrossRef]
- Schmitz, F.J.; Deguzman, F.S.; Hossain, M.B.; Helm, D.V.D. Cytotoxic aromatic alkaloids from the ascidian Amphicarpa meridiana and Leptoclinides sp.: Meridine and 11-hydroxyascididemin. J. Org. Chem. 1991, 56, 804–808. [Google Scholar] [CrossRef]
- McCarthy, P.J.; Pitts, T.P.; Gunawardana, G.P.; Borges, M.K.; Pomponi, S.A. Antifungal activity of meridine, a natural product from the marine sponge Corticium sp. J. Nat. Prod. 1992, 55, 1664–1668. [Google Scholar] [CrossRef]
- Carroll, A.R.; Ngo, A.; Quinn, R.J.; Redburn, J.; Hooper, J.N.A. Petrosamine B, an inhibitor of the Helicobacter pylori enzyme aspartyl semialdehyde dehydrogenase from the Australian sponge Oceanapia sp. J. Nat. Prod. 2005, 68, 804–806. [Google Scholar] [CrossRef]
- Skyler, D.; Heathcock, C.H. The Pyridoacridine Family Tree: A Useful Scheme for Designing Synthesis and Predicting Undiscovered Natural Products. J. Nat. Prod. 2002, 65, 1573–1581. [Google Scholar] [CrossRef]
- Antunes, E.M.; Copp, B.R.; Coleman, M.T.D.; Samaai, T. Pyrroloiminoquinone and related metabolites from marine sponges. Nat. Prod. Rep. 2005, 22, 62–72. [Google Scholar] [CrossRef]
- Kobayashi, J.; Cheng, J.F.; Walchli, M.R.; Nakamura, H.; Hirata, Y.; Sasaki, T.; Ohizumi, Y. Cystodytins A, B, and C, novel tetracyclic aromatic alkaloids with potent antineoplastic activity from the Okinawan tunicate Cystodytes dellechiajei. J. Org. Chem. 1988, 53, 1800–1804. [Google Scholar] [CrossRef]
- Kobayashi, J.; Tsuda, M.; Tanabe, M.; Ishibashi, M. Cystodytins D-I, new cytotoxic tetracyclic aromatic alkaloids from the Okinawan marine tunicate Cystodytes dellechiajei. J. Nat. Prod. 1991, 54, 1634–1638. [Google Scholar] [CrossRef]
- McDonald, L.A.; Eldredge, G.S.; Barrows, L.R.; Ireland, C.M. Inhibition of topoisomerase II catalytic activity by pyridoacridine alkaloids from a Cystodytes sp. ascidian: A mechanism for the apparent intercalator-induced inhibition of topoisomerase II. J. Med. Chem. 1994, 37, 3819–3827. [Google Scholar] [CrossRef]
- Appleton, D.R.; Pearce, A.N.; Lambert, G.; Babcockc, R.C.; Copp, B.R. Isodiplamine, cystodytin K and lissoclinidine: Novel bioactive alkaloids from the New Zealand ascidian Lissoclinum nott. Tetrahedron 2002, 58, 9779–9783. [Google Scholar] [CrossRef]
- Molinski, T.F.; Ireland, C.M. Varamines A and B, new cytotoxic thioalkaloids from Lissoclinum vareau. J. Org. Chem. 1989, 54, 4256–4259. [Google Scholar] [CrossRef]
- Charyulu, G.A.; McKee, T.C.; Ireland, C.M. Diplamine, a cytotoxic polyaromatic alkaloid from the tunicate Diplosoma sp. Tetrahedron Lett. 1989, 30, 4201–4202. [Google Scholar] [CrossRef]
- Tasdemir, D.; Marshall, K.M.; Mangalindan, G.C.; Concepcion, G.P.; Barrows, L.R.; Harper, M.K.; Ireland, C.M. Deoxyamphimedine, a new pyridoacridine alkaloid from two tropical Xestospongia sponges. J. Org. Chem. 2001, 66, 3246–3248. [Google Scholar] [CrossRef]
- Marshall, K.M.; Matsumoto, S.S.; Holden, J.A.; Concepcion, G.P.; Tasdemir, D.; Ireland, C.M.; Barrows, L.R. The anti-neoplastic and novel topoisomerase II-mediated cytotoxicity of neoamphimedine, a marine pyridoacridine. Biochem. Pharmacol. 2003, 66, 447–458. [Google Scholar] [CrossRef]
- Marshall, K.M.; Andjelic, C.D.; Tasdemir, D.; Concepción, G.P.; Ireland, C.M.; Barrows, L.R. Deoxyamphimedine, a pyridoacridine alkaloid, damages DNA via the production of reactive oxygen species. Mar. Drugs 2009, 7, 196–209. [Google Scholar] [CrossRef]
- Kobayashi, J.; Cheng, J.F.; Nakamura, H.; Ohizumi, Y.; Hirata, Y.; Sasaki, T.; Ohta, T.; Nozoe, S. Ascididemin, a novel pentacyclic aromatic alkaloid with potent antileukemic activity from the Okinawan tunicate Didemnum sp. Tetrahedron Lett. 1988, 29, 1177–1180. [Google Scholar] [CrossRef]
- Delfourne, E.; Kiss, R.; le Corre, L.; Merza, J.; Bastide, J.; Frydman, A.; Darro, F. Synthesis and in vitro antitumor activity of an isomer of the marine pyridoacridine alkaloid ascididemin and related compounds. Bioorg. Med. Chem. 2003, 11, 4351–4356. [Google Scholar] [CrossRef]
- Koren-Goldshlager, G.; Aknin, M.; Gaydou, E.M.; Kashman, Y. Three New Alkaloids from the Marine Tunicate Cystodytes violatinctus. J. Org. Chem. 1998, 63, 4601–4603. [Google Scholar] [CrossRef]
- Kashman, Y.; Koren-Goldshlager, G.; Aknin, M.; Garcia Gravalos, D. Isolation and Characterization of a Cytotoxic Pyridoacridine Alkaloid, Shermilamine D, from Cystodytes violatinctus, and its Antitumor Activity. WO 9923099 A1, 14 May 1999. [Google Scholar]
- Gunawardana, G.P.; Kohmoto, S.; Gunasekera, S.P.; McConnell, O.J.; Koehn, F.E. Dercitine, a new biologically active acridine alkaloid from a deep water marine sponge, Dercitus sp. J. Am. Chem. Soc. 1988, 110, 4856–4858. [Google Scholar] [CrossRef]
- Gunawardana, G.P.; Kohmoto, S.; Burres, N.S. New cytotoxic acridine alkaloids from two deep water marine sponges of the family Pachastrellidae. Tetrahedron Lett. 1989, 30, 4359–4362. [Google Scholar] [CrossRef]
- Plubrukarn, A.; Davidson, B.S. Arnoamines A and B, new cytotoxic pentacyclic pyridoacridine alkaloids from the ascidian Cystodytes sp. J. Org. Chem. 1998, 63, 1657–1659. [Google Scholar] [CrossRef]
- Torres, Y.R.; Bugni, T.S.; Berlinck, R.G.S.; Ireland, C.M.; Magalhaes, A.; Ferreira, A.G.; Moreira da Rocha, R. Sebastianines A and B, novel biologically active pyridoacridine alkaloids from the Brazilian ascidian Cystodytes dellechiajei. J. Org. Chem. 2002, 67, 5429–5432. [Google Scholar] [CrossRef]
- Barnes, E.C.; Akmarina, S.N.; Elizabeth, D.W.; Hooper, J.N.A.; Davis, R.A. Ecionines A and B, two new cytotoxic pyridoacridine alkaloids from the Australian marine sponge, Ecionemia geodides. Tetrahedron 2010, 66, 283–287. [Google Scholar] [CrossRef]
- Tran, T.D.; Pham, N.B.; Quinn, R.J. Structure determination of pentacyclic pyridoacridine alkaloids from the Australian marine organisms Ancorina geodides and Cnemidocarpa stolonifera. Eur. J. Org. Chem. 2014, 4805–4816. [Google Scholar]
- Rudi, A.; Benayahu, Y.; Goldberg, I.; Kashman, Y. Eilatin, a novel alkaloid from the marine tunicate Eudistoma sp. Tetrahedron Lett. 1988, 29, 6655–6656. [Google Scholar] [CrossRef]
- Rudi, A.; Kashman, Y. Six new alkaloids from the purple Red Sea tunicate Eudistoma sp. J. Org. Chem. 1989, 54, 5331–5337. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov. 2005, 4, 206–220. [Google Scholar] [CrossRef]
- Witting, K.; Süssmuth, R.D. Discovery of antibacterials and other bioactive compounds from microorganisms-evaluating methodologies for discovery and generation of non-ribosomal peptide antibiotics. Curr. Drug Targets 2011, 12, 1547–1559. [Google Scholar] [CrossRef]
- Kennedy, J. Mutasynthesis, chemobiosynthesis, and back to semi-synthesis. Combining synthetic chemistry and biosynthetic engineering for diversifying natural products. Nat. Prod. Rep. 2008, 25, 25–34. [Google Scholar] [CrossRef]
- Kirschning, A.; Hahn, F. Merging chemical synthesis and biosynthesis: A new chapter in the total synthesis of natural products and natural product libraries. Angew. Chem. Int. Ed. 2012, 51, 4012–4022. [Google Scholar] [CrossRef]
- Kirschning, A.; Taft, F.; Knobloch, T. Total synthesis approaches to natural product derivatives based on the combination of chemical synthesis and metabolic engineering. Org. Biomol. Chem. 2007, 5, 3245–3259. [Google Scholar] [CrossRef]
- Bauer, A.; Bronstrup, M. Industrial natural product chemistry for drug discovery and development. Nat. Prod. Rep. 2014, 31, 35–60. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imperatore, C.; Aiello, A.; D'Aniello, F.; Senese, M.; Menna, M. Alkaloids from Marine Invertebrates as Important Leads for Anticancer Drugs Discovery and Development. Molecules 2014, 19, 20391-20423. https://doi.org/10.3390/molecules191220391
Imperatore C, Aiello A, D'Aniello F, Senese M, Menna M. Alkaloids from Marine Invertebrates as Important Leads for Anticancer Drugs Discovery and Development. Molecules. 2014; 19(12):20391-20423. https://doi.org/10.3390/molecules191220391
Chicago/Turabian StyleImperatore, Concetta, Anna Aiello, Filomena D'Aniello, Maria Senese, and Marialuisa Menna. 2014. "Alkaloids from Marine Invertebrates as Important Leads for Anticancer Drugs Discovery and Development" Molecules 19, no. 12: 20391-20423. https://doi.org/10.3390/molecules191220391
APA StyleImperatore, C., Aiello, A., D'Aniello, F., Senese, M., & Menna, M. (2014). Alkaloids from Marine Invertebrates as Important Leads for Anticancer Drugs Discovery and Development. Molecules, 19(12), 20391-20423. https://doi.org/10.3390/molecules191220391








