Chemical Constituents of Kino Extract from Corymbia torelliana
Abstract
:1. Introduction
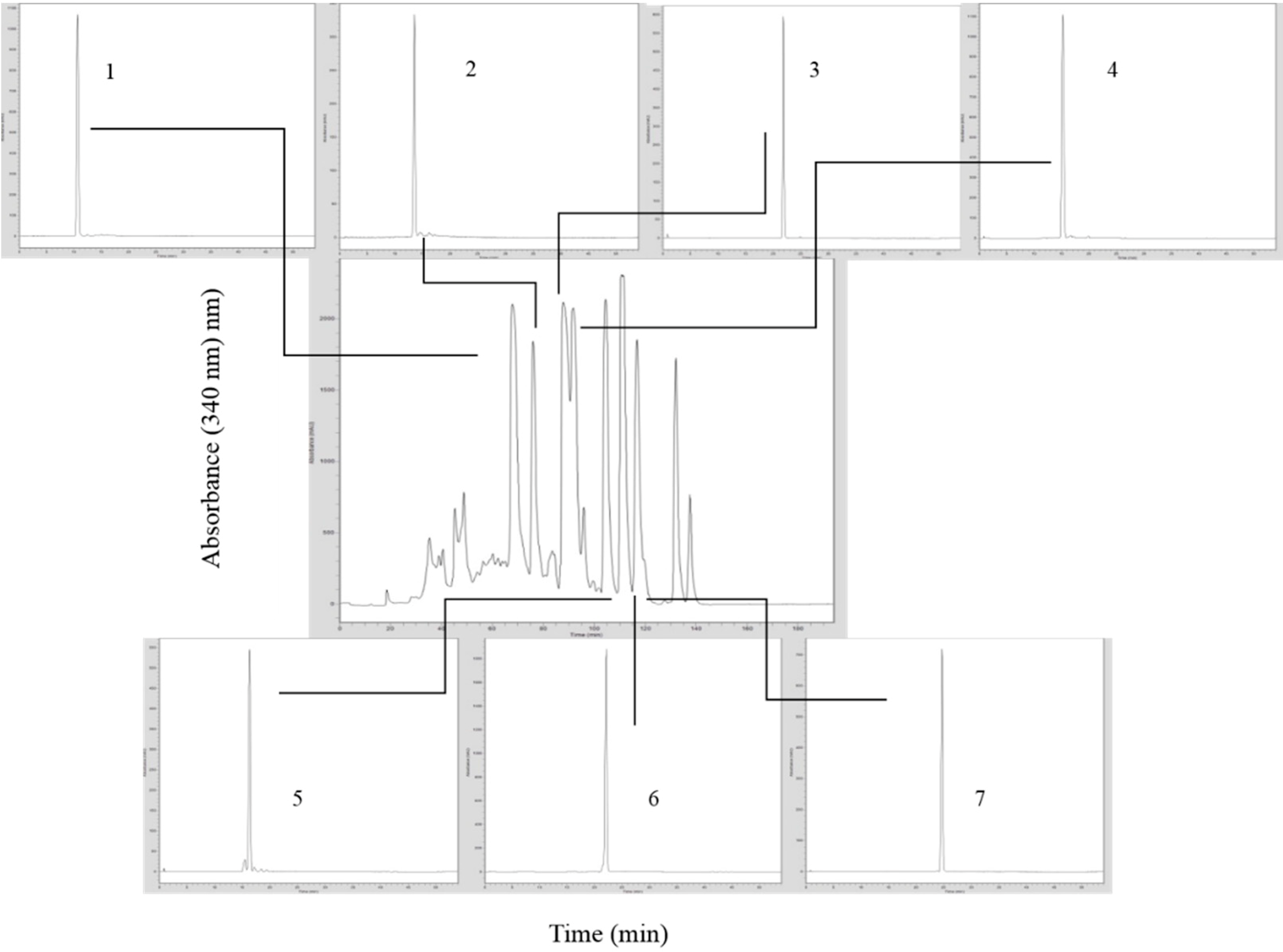
2. Results and Discussion

| Compound | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| 1 | H | OH | H | OH | H |
| 2 | H | OH | H | H | OH |
| 3 | H | OH | H | H | H |
| 4 | H | OMe | H | OH | H |
| 5 | H | OH | Me | H | H |
| 6 | Me | OH | Me | H | H |
| 7 | H | OMe | H | H | H |
| Position | Compound 1 (d6 DMSO) | Compound 2 (CD3OD) | ||||
|---|---|---|---|---|---|---|
| 13C | 1H | HMBC | 13C | 1H | HMBC | |
| 2 | 82.7 (CH) | 5.04 (d,11.4) | 3,4,8a,1',2' | 80.4 (CH) | 5.27 (dd, 12.6, 3.0) | 1',2',6' |
| 3 | 71.3 (CH) | 4.56 (d,11.4) | 2,4,1' | 44.1(CH2) | A 3.05 (dd,17.0, 12.6) | 2,4,1' |
| B 2.69 (dd, 17.0, 3.0) | 4 | |||||
| 4 | 197.2 (C) | 197.2 (C) | ||||
| 4a | 100.1 (C) | 103.3 (C) | ||||
| 5 | 163.1 (C) | 165.1 (C) | ||||
| 5-OH | 11.9 br s | |||||
| 6 | 96.0 (CH) | 5.89 (d, 1.8 Hz) | 4a,5,7,8 | 97.6 (CH) | 5.88 (d, 1.7) | 4a,7 |
| 6-Me | ||||||
| 7 | 167.1 (C) | 169.0 (C) | ||||
| 7-Me | ||||||
| 8 | 95.0 (CH) | 5.83 (d,1.8 Hz) | 4a,6,7,8a | 96.5 (CH) | 5.86 (d, 1.7) | 4a,7,8a |
| 8a | 162.4 (C) | 164.6 (C) | ||||
| 1' | 127.5 (C) | 132.0 (C) | ||||
| 2' | 114.7 (CH) | 6.91 br s | 2,1',3',4',6' | |||
| 3' | 116.3 (CH) | 6.78 s | 1' | |||
| 4' | 157.6 (C) | 146.6 (C) | ||||
| 5' | 143.2 (C) | |||||
| 6' | 119.3 (CH) | 6.78 s | 2,2',4' | |||
| 2'/6' | 129.3 (CH) | 7.31 (d, 8.25) | 2,1',3'/5',4' | |||
| 3'/5' | 114.8 (CH) | 6.78 (d, 8.25) | 1',2'/6',4' | |||
| Position | Compound 3 (d6 DMSO) | Compound 4 (CD3OD) | ||||
|---|---|---|---|---|---|---|
| 13C | 1H | HMBC | 13C | 1H | HMBC | |
| 2 | 78.4 (CH) | 5.43 (dd, 12.9, 2.9) | 3,4,1',2' | 83.7 (CH) | 5.01 (d, 11.4) | 3,4,1',2' |
| 3 | 41.9 (CH2) | A 3.25 (dd, 17.2, 12.9) | 2,4,1' | 72.3 (CH) | 4.57 (d, 11.4) | 2,4,1' |
| B 2.68 (dd, 17.2, 2.9) | 2,1' | |||||
| 4 | 196.8 (C) | 197.6 (C) | ||||
| 4a | 101.7 (C) | 101.2 (C) | ||||
| 5 | 162.9 (C) | 163.7 (C) | ||||
| 5-OH | 12.13 (br s) | 4a,5,6 | ||||
| 6 | 95.8 (CH) | 5.85 (br s) | 5,7,8,8a | 94.6 (CH) | 6.08 (d. 2.4) | 5,7,8,4a |
| 6-Me | ||||||
| 7 | 166.7 (C) | 168.4 (C) | ||||
| 7-Me | 54.9 (CH3) | 3.8 | 7 | |||
| 8 | 94.9 (CH) | 5.85 (br s) | 5,7,8,8a | 93.6 (CH) | 6.04 (d, 2.4) | 4a,6,7,8a |
| 8a | 163.4 (C) | 163.0 (C) | ||||
| 1' | 128.8 (C) | 127.7 (C) | ||||
| 2' | ||||||
| 3' | ||||||
| 4' | 157.6 (C) | 157.8 (C) | ||||
| 5' | 6.8 | |||||
| 6' | 7.35 | |||||
| 2'/6' | 128.3 (CH) | 7.31 (d, 8.5) | 2,1',3'/5',4' | 128.4 (CH) | 7.35 (d, 8.4) | 2,3',4' |
| 3'/5' | 115.1 (CH) | 6.79 (d,8.5) | 1',2'/6',4' | 114.7 (CH) | 6.80 (d, 8.4) | 1',2',4' |
| Position | Compound 5 (d6 DMSO) | Compound 6 (CD3OD) | Compound 7 (CDCl3) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 13C | 1H | HMBC | 13C | 1H | HMBC | 13C | 1H | HMBC | |
| 2 | 77.9 (CH) | 5.32 (dd,12.4,2.8) | 3,4,1',2' | 78.6 (CH) | 5.30 (dd, 12.9, 3.0) | 1',2' | 79.0 (CH) | 5.34 (dd, 13.0, 3.0) | 3,4,1',2' |
| 3 | 42.0 (CH2) | A 3.12(dd,17.0,12.4) B 2.60 (dd,17.0,2.8) | 2,4,1' 2,1' | 42.7(CH2) | A 3.05 (dd,16.8, 12.9) B 2.71 (dd, 16.8, 3.0) | 2,4,1' 4 | 43.2 ( CH2) | A 3.08 (dd,17.3, 13.0) B 2.78 (dd, 17.3, 3.0) | 2,4,1' 2,1' |
| 4 | 194.3 (C) | 196.7 (C) | 196.2 (C) | ||||||
| 4a | 100.6 (C) | 102.8 (C) | 103.9 (C) | ||||||
| 5 | 160.4 (C) | 157.9 (C) | 164.5 (C) | ||||||
| 5-OH | 12.47 (br s) | 4a,5,6 | |||||||
| 6 | 103.3 (C) | 103.5 (C) | 95.1 (CH) | 6.06 (d, 2.4) | 4a,5,7,8 | ||||
| 6-Me | 7.1 (CH3) | 1.83 (s) | 5,6,7 | 6.8 (CH3) | 1.99 (s) | 5,6,7 | |||
| 7 | 168.8 (C) | 163.6 | 168.1 (C) | ||||||
| 7-Me | 55.7 (CH3) | 3.79 (s) | 7 | ||||||
| 8 | 94.9 (CH) | 5.82 (s) | 6,7,8a | 101.6 (C) | 94.2 (CH) | 6.03 (d, 2.4) | 6,7,8a | ||
| 8-Me | 6.0 (CH3) | 1.98 (s) | 7,8,8a | ||||||
| 8a | 160.2 (C) | 158.8 (C) | 163.2 (C) | ||||||
| 1' | 129.2 (C) | 130.2 (C) | 127.7(C) | ||||||
| 2''/6'' | 128.0 (CH) | 7.29 (d, 8.3) | 2,1',3'/5',4' | 127.4 (CH) | 7.32 (d, 8.4) | 2,3',4' | 127.9 (CH) | 7.31 (d, 8.6) | 2,1',3'/5',4' |
| 3''/5' | 115.0 (CH) | 6.77 (d, 8.3) | 1',2'/6',4' | 114.9 (CH) | 6.81 (d, 8.4) | 1',2',4' | 115.7 (CH) | 6.88 (d, 8.6) | 1',2'/6',4' |
| 4' | 157.5 (C) | 157.4 (C) | 156.5 (C) | ||||||
| Compound | Identification | Formula [M–H]− | Calculated Mass [M–H]− (m/z) | Found Mass |
|---|---|---|---|---|
| 1 | 3,4',5,7-tetrahydroxyflavanone | C15H11O6 | 287.05553 | 287.0565 |
| 2 | 3',4',5,7-tetrahydroxyflavanone | C15H11O6 | 287.05553 | 287.0566 |
| 3 | 4',5,7-trihydroxyflavanone | C15H11O5 | 271.06063 | 271.0616 |
| 4 | 3,4',5 -trihydroxy-7-methoxyflavanone | C16H13O6 | 301.07119 | 301.0721 |
| 5 | (+)-(2S)-4',5,7-trihydroxy-6-methylflavanone | C16H13O5 | 285.07629 | 285.0773 |
| 6 | 4',5,7-trihydroxy-6,8-dimethylflavanone | C17H15O5 | 299.09695 | 299.0930 |
| 7 | 4',5-dihydroxy-7-methoxyflavanone | C16H13O5 | 285.07629 | 285.0771 |
3. Experimental Section
3.1. Plant Material
3.2. Extraction
3.3. Chromatography
3.3.1. Analytical HPLC-UV/DAD
3.3.2. Preparative HPLC
3.3.3. UPLC-HR-MS Analysis
3.3.4. Spectroscopic Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wilson, P.G.; O’Brien, M.M.; Gadek, P.A.; Quinn, C.J. Myrtaceae revisited: A reassessment of infrafamilial groups. Am. J. Bot. 2001, 88, 2013–2025. [Google Scholar] [CrossRef] [PubMed]
- Leonhardt, S.D.; Wallace, H.M.; Schmitt, T. The cuticular profiles of Australian stingless bees are shaped by resin of the eucalypt tree Corymbia torelliana. Austral Ecol. 2011, 36, 537–543. [Google Scholar] [CrossRef]
- Wallace, H.M.; Howell, M.G.; Lee, D.J. Standard yet unusual mechanisms of long-distance dispersal: Seed dispersal of Corymbia torelliana by bees. Divers. Distrib. 2008, 14, 87–94. [Google Scholar] [CrossRef]
- Wallace, H.M.; Trueman, S.J. Dispersal of Eucalyptus torelliana seeds by the resin-collecting stingless bee, Trigona carbonaria. Oecologia 1995, 104, 12–16. [Google Scholar] [CrossRef]
- Wallace, H.; Lee, D. Resin-foraging by colonies of Trigona sapiens and T. hockingsi (Hymenoptera: Apidae, Meliponini) and consequent seed dispersal of Corymbia torelliana (Myrtaceae). Apidologie 2010, 41, 428–435. [Google Scholar] [CrossRef]
- Penfold, A. The Eucalypts; Interscience Publishers: New York, NY, USA, 1961. [Google Scholar]
- Maiden, J.H. The gums, resins and other vegetable exudations of Australia. J. R. Soc. N. S. W. 1901, 35, 161–212. [Google Scholar]
- Locher, C.; Currie, L. Revisiting kinos—An Australian perspective. J. Ethnopharmacol. 2010, 128, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Freitas, M.O.; Lima, M.A.; Silveira, E.R. NMR assignments of unusual flavonoids from the kino of Eucalyptus citriodora. Magn. Reson. Chem. 2007, 45, 262–264. [Google Scholar] [CrossRef] [PubMed]
- Hillis, W. The formation of polyphenols in trees 2. The polyphenols of Eucalyptus sieberiana kino. Biochem. J. 1963, 92, 516–521. [Google Scholar]
- Hillis, W.; Carle, A. The formation of phenolic substances in Eucalyptus gigantea and Eucalyptus sieberiana. Biochemistry 1960, 74, 608–615. [Google Scholar]
- Hillis, W.; Carle, A. The chemistry of the eucalypt kinos iv. Eucalyptus hemiphloia kino. Aust. J. Chem. 1962, 1, 147–159. [Google Scholar]
- Hillis, W. The chemistry of the eucalypt kinos. Part i. Chromatographic resolution. Aust. J. Basic Appl. Sci. 1951, 3, 385–397. [Google Scholar]
- Hillis, W. The chemistry of the eucalypt kinos. Part ii. Aromadendrin, kaempferol and ellagic acid. Aust. J. Sci. Res. Ser. A 1952, 2, 379–386. [Google Scholar]
- Hillis, W.; Carle, A. The chemistry of the eucalypt kinos iii. (+)-Afzelechin, pyrogallol, and (+)-catechin from Eucalyptus calophylla kino. Aust. J. Chem. 1960, 13, 390–395. [Google Scholar] [CrossRef]
- Lambert, J.B.; Wu, Y.; Kozminski, M.A. Characterization of Eucalyptus and chemically related exudates by nuclear magnetic resonance spectroscopy. Aust. J. Chem. 2007, 60, 862–870. [Google Scholar] [CrossRef]
- Gell, R.; Pinhey, J.; Ritchie, E. The constituents of the kino of Eucalyptus maculata Hook. Aust. J. Chem. 1958, 11, 372–375. [Google Scholar] [CrossRef]
- Satwalekar, S.S.; Gupta, T.R.; Narasimha Rao, P.L. Chemical and antibacterial properties of kinos from Eucalyptus spp. Citriodorol—The antibiotic principle from the kino of Eucalyptus citriodora. J. Indian Inst. Sci. 1956, 2, 195–212. [Google Scholar]
- Power, F.B.; Tutin, F. Chemical examination of Eriodictyon. Pharm. Rev. 1906, 24, 300–304. [Google Scholar]
- Barton, G.M. A new c-methyl flavanone from diseased (Poria weirii Murr.) Douglas fir (Pseudotsuga menziesii (Mirb.) Franco) roots. Can. J. Chem. 1967, 45, 1020–1022. [Google Scholar] [CrossRef]
- Yi, J.H.; Zhang, G.L.; Li, B.G. Studies on the chemical constituents of Pseudotsuga sinensis. Acta Pharm.Sin. 2002, 37, 352–354. [Google Scholar]
- Youssef, D.T.A.; Ramadan, M.A.; Khalifa, A.A. Acetophenones, a chalcone, a chromone and flavonoids from Pancratium maritimum. Phytochemistry 1998, 49, 2579–2583. [Google Scholar] [CrossRef]
- Wu, Z.B.; Zhao, Y.Y.; Yang, X.Y.; Liang, H. Flavonoids from Bauhinia glauca subsp. pernervosa. Chem. Pharm. Bull. 2009, 57, 628–631. [Google Scholar] [CrossRef]
- Birch, A.J.; Dahl, C.J. Some constituents of the resins of Xanthorrhoea preissii, australis and hastile. Aust. J. Chem. 1974, 27, 331–344. [Google Scholar] [CrossRef]
- Maiden, J.H. Botany bay of eucalyptus kino. Pharm. J. Trans. 1889, 3, 221–321. [Google Scholar]
- Sample Availability: Samples of compounds 1–7 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nobakht, M.; Grkovic, T.; Trueman, S.J.; Wallace, H.M.; Katouli, M.; Quinn, R.J.; Brooks, P.R. Chemical Constituents of Kino Extract from Corymbia torelliana. Molecules 2014, 19, 17862-17871. https://doi.org/10.3390/molecules191117862
Nobakht M, Grkovic T, Trueman SJ, Wallace HM, Katouli M, Quinn RJ, Brooks PR. Chemical Constituents of Kino Extract from Corymbia torelliana. Molecules. 2014; 19(11):17862-17871. https://doi.org/10.3390/molecules191117862
Chicago/Turabian StyleNobakht, Motahareh, Tanja Grkovic, Stephen J. Trueman, Helen M. Wallace, Mohammad Katouli, Ronald J. Quinn, and Peter R. Brooks. 2014. "Chemical Constituents of Kino Extract from Corymbia torelliana" Molecules 19, no. 11: 17862-17871. https://doi.org/10.3390/molecules191117862
APA StyleNobakht, M., Grkovic, T., Trueman, S. J., Wallace, H. M., Katouli, M., Quinn, R. J., & Brooks, P. R. (2014). Chemical Constituents of Kino Extract from Corymbia torelliana. Molecules, 19(11), 17862-17871. https://doi.org/10.3390/molecules191117862





