3. Experimental
3.1. General
All chemicals and solvents were reagent grade and used without purification. Dry THF and extra dry DMF (99.8%) over molecular sieves were purchased from Acros. Melting points were determined on a Büchi 530 apparatus and are uncorrected. Specific rotations were measured at 25 °C on a Perkin–Elmer polarimeter using a 10 cm cell. Nuclear magnetic resonance spectra were obtained on a Varian Mercury spectrometer (1H-NMR recorded at 200 MHz and 13C-NMR recorded at 50 MHz and they are referenced in ppm relative to TMS as an internal standard). Mass spectra were recorded on a Finnigan Surveyor MSQ Plus with only molecular ions and major peaks being reported with intensities quoted as percentages of the base peak. TLC plates (Silica Gel 60 F254) were purchased from Merck. Visualization of spots was effected with UV light and/or phosphomolybdic acid in EtOH stain. All target compounds possessed 95% purity as determined by combustion analysis.
3.2. General Procedure for the Preparation of Compounds 28, 30, 35, 37
To an ice cooled solution of appropriate alcohol 27, 29, 34, 36 (10 mmol) in Et2O (25 mL), PBr3 (1.41 mL, 15 mmol) was added dropwise under argon. The mixture was stirred at room temperature for 3–7 h and the reaction was quenched by the addition of H2O (15 mL) in small portions at 0 °C. The aqueous phase was removed and the organic layer was washed with H2O, dried over Na2SO4 and evaporated in vacuo. In the case of products 30 and 35, purification was achieved by column chromatography using the appropriate solvent systems as it will be defined in each case below. In the case of bromides 28, 37, the oily products were not visible either in UV-light or in iodine stain. They were characterized by NMR and used in the next step without further purification. For this reason the yields of their preparation must be considered as crude yields.
1-Bromo-2-Methoxyethane (28). Prepared from the commercially available alcohol 27; Yield: 58% (colorless oil). 1H-NMR (CDCl3): δ 3.69 (t, J = 6.0 Hz, 2H, OCH2), 3.45 (t, J = 6.0 Hz, 2H, CH2Br), 3.38 (s, 3H, CH3); 13C-NMR: δ 72.3 (OCH2), 58.8 (CH3), 30.3 (CH2Br). Anal. calcd for C3H7BrO: C, 25.92; H, 5.08; Found: C, 25.68; H, 5.12.
1-(Bromomethyl)-4-(2-Methoxyethoxy)benzene (30). Prepared from alcohol 18. Eluent EtOAc–petroleum ether (bp. 40–60 °C), 3:7; white solid; yield 60%; mp. 47–49 °C; Rf 0.50 in EtOAc–petroleum ether (bp. 40–60 °C), 3:7. 1H-NMR (CDCl3): δ 7.30 (d, J = 8.7 Hz, 2H, Ph), 6.87 (d, J = 8.7 Hz, 2H, Ph), 4.48 (s, 2H, CH2Br), 4.09 (t, J = 4.5 Hz, 2H, PhOCH2), 3.73 (t, J = 4.8 Hz, 2H, CH2OCH3), 3.43 (s, 3H, CH3). 13C-NMR: δ 158.7, 130.3, 114.7, 70.8 (CH2OCH3), 67.2 (PhOCH2), 59.1 (CH3), 33.9 (CH2Br). Anal. calcd for C10H13BrO2: C, 49.00; H, 5.35; Found: C, 48.95; H, 5.25.
1-(Bromomethyl)-4-(3-Methoxypropoxy)benzene (35). Prepared from alcohol 34. Eluent EtOAc–petroleum ether (bp. 40–60 °C), 3:7; yield: 70% (colorless oil); Rf 0.35 in EtOAc–petroleum ether (bp. 40–60 °C), 3:7. 1H-NMR (CDCl3): δ 7.31 (d, J = 8.7 Hz, 2H, Ph), 6.86 (d, J = 8.7 Hz, 2H, Ph), 4.50 (s, 2H, CH2Br), 4.05 (t, J = 6.3 Hz, 2H, PhOCH2), 3.55 (t, J = 6.2 Hz, 2H, CH2ΟCH3), 3.35 (s, 3H, CH3), 2.11–1.98 (m, 2H, CH2CH2CH2). 13C-NMR: δ 159.1, 130.4, 129.8, 114.7, 69.1 (CH2OCH3), 64.8 (PhOCH2), 58.7 (CH3), 34.1 (CH2Br), 29.5 (CH2CH2CH2). Anal. calcd for C11H15BrO2: C, 50.98; H, 5.83; Found: C, 50.82; H, 5.93.
1-Bromo-4-Methoxybutane (37). Prepared from the commercially available alcohol 36; Yield: 60% (colorless oil). 1H-NMR (CDCl3): δ 3.47–3.37 (m, 4H, OCH2, CH2Br), 3.33 (s, 3H, CH3), 2.02–1.88 (m, 2H, CH2CH2Br), 1.78–1.64 (m, 2H, CH2CH2O). 13C-NMR: δ 71.7 (OCH2), 58.6 (CH3), 33.7 (CH2Br), 29.6 (CH2CH2Br), 28.2 (CH2CH2O). Anal. calcd for C5H11BrO: C, 35.95; H, 6.64; Found: C, 35.82; H, 6.59.
1-(Bromomethyl)-4-(4-Methoxybutoxy)benzene (39). To a stirred solution containing alcohol 38 (35 mg, 0.166 mmol) and NaBr (17 mg, 0.166 mmol) in CH3CN (0.5 mL), TMS-Cl (21 μL, 0.166 mmol) was added under argon. After stirring for 1.5h at rt, the solvent was evaporated in vacuo and the residue dissolved in Et2O was washed with brine and H2O. The organic layer was dried over Na2SO4 and evaporated under reduced pressure. After purification by column chromatography using EtOAc–petroleum ether (bp. 40–60 °C) 1:9 as eluent, the product was obtained as a colorless oil in 42% yield (19 mg). Rf 0.35 in EtOAc-petroleum ether (bp. 40–60 °C) 1: 9. 1H-NMR (CDCl3): δ 7.27 (d, J = 8.6 Hz, 2H, Ph), 6.84 (d, J = 8.6 Hz, 2H, Ph), 4.50 (d, J = 12.6 Hz, 2H, CH2Br), 3.95 (t, J = 5.9 Hz, 2H, PhOCH2), 3.42 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.33 (s, 3H, CH3), 1.87–1.68 (m, 4H, CH2CH2CH2CH2). 13C-NMR: δ 159.0, 130.3, 114.5, 72.2 (CH2OCH3), 67.5 (PhOCH2), 58.5 (CH3), 34.0 (CH2Br), 26.1, 25.9. Anal. calcd for C12H17BrO2: C, 52.76; H, 6.27; Found: C, 52.82; H, 6.35.
3.3. General Procedure for the Preparation of Compounds 29, 38
To a stirred solution of 4-hydroxybenzylalcohol (1.24 g, 10 mmol) in acetone (45 mL), bromides 28 or 37 (10.5 mmol), K2CO3 (4.14 g, 30 mmol) and 18-crown-6 (0.020 g) were added consecutively. The mixture was refluxed for 3 days. After evaporation of the solvent, the residue was dissolved in EtOAc and washed with brine and H2O. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure. Purification was achieved by column chromatography, eluting with the appropriate solvents as defined for each case below.
[4-(2-Methoxyethoxy)-phenyl]methanol (29). Prepared from bromide 28. Eluent CHCl3–MeOH, 9.5:0.5; Yield: 76% (colorless oil); Rf 0.48 in CHCl3–MeOH, 9.5:0.5. 1H-NMR (CDCl3): δ 7.27 (d, J = 8.7 Hz, 2H, Ph), 6.90 (d, J = 8.7 Hz, 2H, Ph), 4.61 (s, 2H, CH2OH), 4.11 (t, J = 4.6 Hz, 2H, PhOCH2), 3.75 (t, J = 4.9 Hz, 2H, CH2OCH3), 3.44 (s, 3H, CH3); 13C-NMR: δ 158.2, 133.3, 128.5, 114.5, 70.9 (CH2OCH3), 67.2 (PhOCH2), 64.8 (CH2OH), 59.2 (CH3). Anal. calcd for C10H14O3: C, 65.91; H, 7.74; Found: C, 65.79; H, 7.68.
[4-(4-Methoxybutoxy) Phenyl]methanol (38). Prepared from bromide 37. Eluent CHCl3–MeOH, 9:1; Yield: 67% (colorless oil); Rf 0.35 in CHCl3–MeOH, 9:1. 1H-NMR (CDCl3): δ 7.26 (d, J = 8.7 Hz, 2H, Ph), 6.86 (d, J = 8.7 Hz, 2H, Ph), 4.60 (s, 2H, CH2OH), 3.97 (t, J = 6.1 Hz, 2H, PhOCH2), 3.43 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.34 (s, 3H, CH3), 1.88–1.69 (m, 4H, CH2CH2CH2CH2). 13C-NMR: δ 158.6, 132.9, 128.6, 114.5, 72.3 (CH2OCH3), 67.6 (PhOCH2), 65.0 (CH2OH), 58.5 (CH3), 26.2, 26.0. Anal. calcd for C12H18O3: C, 68.54; H, 8.63; Found: C, 68.62; H, 8.59.
4-(3-Bromopropoxy) Benzaldehyde (32). To a stirred solution of 4-hydroxy-benzaldehyde (1 g, 8.2 mmol) in CH3CN (15 mL), K2CO3 (1.70 g, 12.3 mmol) and 1,3-dibromopropane (8.3 mL, 81.9 mmol) were added consecutively. A tube filled with CaCl2 was fixed on the condenser and the mixture was refluxed overnight. After evaporation of the solvent, the residue was dissolved in EtOAc and washed with brine and H2O. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure. Purification was achieved by column chromatography using EtOAc–petroleum ether (bp. 40–60 °C) 1:4 as eluent. The product was obtained as colorless oil in 40% yield (0.8 g) and stored at −4 °C, only for a short time. Rf 0.36 in EtOAc–petroleum ether (bp. 40–60 °C) 1:4. 1H-NMR (CDCl3): δ 9.88 (s, 1H, CHO), 7.83 (d, J = 8.9 Hz, 2H, Ph), 7.00 (d, J = 8.9 Hz, 2H, Ph), 4.19 (t, J = 5.8 Hz, 2H, PhOCH2), 3.60 (t, J = 6.3 Hz, 2H, CH2Br), 2.41–2.29 (m, 2H, CH2CH2CH2). 13C-NMR: δ 190.7 (CHO), 163.6, 131.9, 130.1, 114.7, 65.6 (PhOCH2), 32.0 (CH2Br), 29.6 (CH2CH2CH2). Anal. calcd for C10H11BrO3: C, 49.41; H, 4.56; Found: C, 49.56; H, 4.39.
4-(3-Methoxypropoxy)benzaldehyde (33). To a stirred solution of bromide 32 (46 mg, 0.19 mmol) in MeOH (2 mL), a freshly prepared solution of 1N CH3ONa/MeOH (200 μL) was added and the mixture was left stirring at rt for 2 days. The reaction was quenched by the addition of H2O and the mixture was acidified with aq. 1 N HCl. After evaporation of the solvent, the residue was dissolved in EtOAc and washed with brine and H2O. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure. Purification was achieved by column chromatography using EtOAc–petroleum ether (bp. 40–60 °C) 2:3 as eluent. The product was obtained as a colorless oil in 38% yield (14 mg) and was immediately subjected to the following reaction. Rf 0.38 in EtOAc–petroleum ether (bp. 40–60 °C) 2:3. 1H-NMR (CDCl3): δ 9.87 (s, 1H, CHO), 7.82 (d, J = 8.8 Hz, 2H, Ph), 7.00 (d, J = 8.8 Hz, 2H, Ph), 4.14 (t, J = 6.3 Hz, 2H, PhOCH2), 3.56 (t, J = 6.0 Hz, 2H, CH2Ο), 3.35 (s, 3H, CH3), 2.14–2.01 (m, 2H, CH2CH2CH2). 13C-NMR: δ 190.8 (CHO), 164.0, 131.9, 129.8, 114.7, 68.8 (CH2OCH3), 65.2 (PhOCH2), 58.7 (CH3), 29.4 (CH2CH2CH2). MS (ESI): m/z = 195 (100) (M+H+). Anal. calcd for C11H14O3: C, 68.02; H, 7.27; Found: C, 68.18; H, 7.15.
[4-(3-Methoxypropoxy) Phenyl]methanol (34). To a stirred solution of aldehyde 33 (63 mg, 0.324 mmol) in dry THF (2 mL) at 0 °C NaBH4 (18 mg, 0.487 mmol) was added under argon. The reaction mixture was left stirring at rt overnight. The reaction was quenched by the addition of a 20% solution of AcOH at 0 °C, until the production of the gas was ceased. The organic solvent was evaporated under reduced pressure and the residue was dissolved in EtOAc. The organic phase was washed with 5% aq. NaHCO3, brine and H2O, dried over Na2SO4 and evaporated under reduced pressure. After purification with column chromatography using EtOAc–petroleum ether (bp. 40–60 °C) 2:3 as eluent, the product was obtained as a colorless oil in 75% yield (47 mg). Rf 0.25 in EtOAc-petroleum ether (bp. 40–60 °C) 2:3. 1H-NMR (CDCl3): δ 7.28 (d, J = 8.6 Hz, 2H, Ph), 6.89 (d, J = 8.6 Hz, 2H, Ph), 4.61 (s, 2H, CH2OH), 4.05 (t, J = 6.3 Hz, 2H, PhOCH2), 3.55 (t, J = 6.2 Hz, 2H, CH2ΟCH3), 3.35 (s, 3H, CH3), 2.11–1.98 (m, 2H, CH2CH2CH2). 13C-NMR: δ 158.6, 133.0, 114.5, 69.2 (CH2OCH3), 65.0 (CH2OH), 64.8 (PhOCH2), 58.7 (CH3), 29.5 (CH2CH2CH2). MS (ESI): m/z = 197 (100) (M+H+). Anal. calcd for C11H16O3: C, 67.32; H, 8.22; Found: C, 67.53; H, 8.09.
(5S)-(Hydroxymethyl)-2-Pyrrolidinone (
2). See reference [
17].
5-[[(Triethylsilyl)oxy] Methyl]-2-Pyrrolidinone (
3). See reference [
18].
3.4. General Procedure of for the Preparation of Compounds 4, 8, 16a–c, 20
To a cooled solution of methyl (S)-pyroglutamate (1), O-protected-S-pyroglutaminol (3), or methylester 7 (1 mmol) in dry THF (5 mL) NaH (60% in paraffin oil, 1.5 mmol) was added in small portions, followed by the addition of the appropriate benzyl bromide (or benzyloxycarbonylchloride in case of compound 4) (1.1 mmol). The stirring was continued for 15 min at 0 °C, and for 24 h at rt under argon. The reaction was quenched by the addition of a saturated solution of NH4Cl at 0 °C, the organic solvent was evaporated and the residue was dissolved in ethyl acetate. The organic layer was washed with brine, dried over Na2SO4 and evaporated under reduced pressure. The product was purified by column chromatography (Silica Gel 60) using the appropriate solvent systems as defined in each case below.
Benzyl (5S)-2-oxo-5-{[(triethylsilyl)oxy]methyl}pyrrolidine-1-carboxylate (4). Prepared from compound 3 and benzyloxycarbonyl chloride. Eluent EtOAc–petroleum ether (bp. 40–60 °C), 4:6. The product was obtained as colorless oil in 50% yield. Rf 0.53 in EtOAc–petroleum ether (bp. 40–60 °C), 4:6; [α]D −68.2 (c 1.1, CHCl3). 1H-NMR (CDCl3): δ 7.46–7.32 (m, 5H, Ph), 5.28 (dd, J1 = 12.4 Hz, J2 = 18.4 Hz, 2H, COOCH2), 4.28–4.20 (m, 1H, NCH), 3.88 (dd, J1 = 4.0 Hz, J2 = 10.6 Hz, 1H, CHHOSi), 3.67 (dd, J1 = 2.5 Hz, J2 = 10.7 Hz, 1H, CHHOSi), 2.84–2.03 (m, 4H, 2xCH2), 0.88 (t, J = 7.6 Hz, 9H, 3 × CH3), 0.51 (q, J = 7.6 Hz, 6H, 3 × (CH2CH3)) 13C-NMR: δ 174.7 (CON), 151.3 (COO), 135.2, 128.5, 128.3, 128.2, 67.8 (CH2OSi), 63.7 (OCH2Ph), 58.8 (NCH), 32.1, 21.2, 6.6 (CH2CH3), 4.1 (CH2CH3). MS (ESI): m/z = 364 (100) (M+H+). Anal. calcd for C19H29NO4Si: C, 62.78; H, 8.04; N, 3.85 Found: C, 62.83; H, 7.95; N, 3.92.
Methyl 3-{(2S)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-yl} propanoate (8). Prepared from compound 7 and bromide 30. Eluent EtOAc–petroleum ether (bp. 40–60 °C), 9:1. The product was obtained as colorless oil in 40% yield. Rf 0.23 in EtOAc–petroleum ether (bp. 40–60 °C), 1:9; [α]D +5.2 (c 0.92, CHCl3). 1H-NMR (CDCl3): δ 7.13 (d, J = 8.6 Hz, 2H, Ph), 6.83 (d, J = 8.6 Hz, 2H, Ph), 4.90 (d, J = 14.8 Hz, 1H, NCHHPh), 4.09–4.04 (m, 2H, PhOCH2), 3.85 (d, J = 14.8 Hz, 1H, NCHHPh), 3.73–3.68 (m, 2H, CH2OCH3), 3.62 (s, 3H, COOCH3), 3.42–3.38 (m, 1H, NCH), 3.41 (s, 3H, OCH3), 2.45–1.54 (m, 8H, 4 × CH2). 13C-NMR: δ 174.8 (NCO), 173.0 (COO), 158.1, 129.2, 128.7, 114.6, 70.7 (CH2OCH3), 67.1 (PhOCH2), 59.1 (OCH3), 55.7 (NCH), 51.7 (COOCH3), 43.3 (NCH2Ph), 30.0, 29.0, 27.6, 23.3. MS (ESI): m/z = 358 (100) (M+Na+). Anal. calcd for C18H25NO5: C, 64.46; H, 7.51; N, 4.18 Found: C, 64.51; H, 7.62; N, 4.05.
Methyl 1-[4-(2-Methoxyethoxy)benzyl]-5-oxo-L-prolinate (16a). Prepared from methyl-S-pyroglutamate (1) and bromide 30. Eluent EtOAc. The product was obtained as a colorless oil in 73% yield. Rf 0.42 in EtOAc; [α]D +20.2 (c 1.0, CHCl3). 1H-NMR (CDCl3): δ 7.07 (d, J = 8.5 Hz, 2H, Ph), 6.82 (d, J = 8.5 Hz, 2H, Ph), 4.87 (d, J = 14.7 Hz, 1H, CHHPh), 4.07–4.02 (m, 2H, PhOCH2), 3.93–3.86 (m, 2H, CHHPh, NCHCOO), 3.71–3.66 (m, 2H, CH2OCH3), 3.62 (s, 3H, COOCH3), 3.39 (s, 3H, OCH3), 2.61–1.92 (m, 4H, 2 × CH2). 13C-NMR: δ 174.8 (NCO), 172.1 (COO), 158.2, 129.7, 127.8, 114.6, 70.8 (CH2OCH3), 67.1 (OCH2), 59.0 (NCH), 58.4 (OCH3), 52.2 (COOCH3), 44.8 (CH2Ph), 29.5, 22.6. MS (ESI): m/z = 308 (100) (M+Η+). Anal. calcd for C16H21NO5: C, 62.53; H, 6.89; N, 4.56 Found: C, 62.58; H, 6.75; N, 4.61.
Methyl 1-[4-(3-Methoxypropoxy)benzyl]-5-oxo-L-prolinate (16b). Prepared from methyl-S-pyroglutamate (1) and bromide 35. Eluent EtOAc. The product was obtained as a colorless oil in 72% yield. Rf 0.44 in EtOAc; [α]D +13.5 (c 1.0, CHCl3). 1H-NMR (CDCl3): δ 7.08 (d, J = 8.6 Hz, 2H, Ph), 6.80 (d, J = 8.6 Hz, 2H, Ph), 4.90 (d, J = 14.6 Hz, 1H, NCHHPh), 3.99 (t, J = 6.3 Hz, 2H, PhOCH2), 3.95–3.90 (m, 1H, NCHCOO), 3.89 (d, J = 4.6 Hz, 1H, NCHHPh), 3.65 (s, 3H, COOCH3), 3.51 (t, J = 6.2Hz, 2H, CH2OCH3), 3.31 (s, 3H, OCH3), 2.57–1.93 (m, 6H, 3 × CH2). 13C-NMR: δ 174.8 (NCO), 172.2 (COO), 158.5, 129.7, 127.5, 114.5, 69.0 (CH2OCH3), 64.7 (PhOCH2), 58.6 (NCH), 58.4 (OCH3), 44.8 (CH2Ph), 29.5, 29.4, 22.6. MS (ESI): m/z = 322 (100) (M+Η+). Anal. calcd for C17H23NO5: C, 63.54; H, 7.21; N, 4.36 Found: C, 63.48; H, 7.28; N, 4.18.
Methyl 1-[4-(4-Methoxybutoxy)benzyl]-5-oxo-L-prolinate (16c). Prepared from methyl-S-pyroglutamate (1) and bromide 39. Eluent EtOAc. The product was obtained as a colorless oil in 66% yield. Rf 0.47 in EtOAc; [α]D +11.7 (c 1.0, CHCl3). 1H-NMR (CDCl3): δ 7.08 (d, J = 8.6 Hz, 2H, Ph), 6.79 (d, J = 8.6 Hz, 2H, Ph), 4.90 (d, J = 14.7 Hz, 1H, NCHHPh), 3.97–3.89 (m, 1H, NCHCO), 3.92 (t, J = 6.30 Hz, 2H, PhOCH2), 3.90 (d, J = 14.7 Hz, 1H, NCHHPh), 3.65 (s, 3H, COOCH3), 3.40 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.31 (s, 3H, OCH3), 2.58–1.96 (m, 4H, 2 × CH2), 1.84-1.67 (m, 4H, (OCH2CH2)2). 13C-NMR: δ 174.8 (NCO), 172.2 (COO), 158.6, 129.7, 127.5, 114.5, 72.2 (CH2OCH3), 67.5 (PhOCH2), 58.5 (NCH), 58.4 (OCH3), 52.2 (COOCH3), 44.9 (NCH2Ph), 29.5, 26.1, 25.9, 22.7. MS (ESI): m/z = 336 (100) (M+Η+). Anal. calcd for C18H25NO5: C, 64.46; H, 7.51; N, 4.18 Found: C, 64.38; H, 7.48; N, 4.27.
Methyl 1-[4-(Benzyloxy)benzyl]-5-oxo-L-prolinate (
20). Prepared from methyl-
S-pyroglutamate (
1) and 1-(benzyloxy)-4-(bromomethyl) benzene [
32]. Eluent EtOAc -petroleum ether (bp. 40–60 °C), 7:3. The product was obtained as a colorless oil in 47% yield. Rf 0.35 in EtOAc–petroleum ether (bp. 40–60 °C), 7:3; [α]
D +22.5 (c 1, CHCl
3).
1H-NMR (CDCl
3): δ 7.44–7.28 (m, 5H, Ph), 7.14 (d,
J = 8.5 Hz, 2H, Ph), 6.92 (d,
J = 8.5 Hz, 2H, Ph), 5.05 (s, 2H, OC
H2Ph), 4.93 (d,
J = 14.7 Hz, 1H, NCH
HPh), 4.00-3.92 (m, 2H, NCH
HPh, NC
H), 3.66 (s, 3H, C
H3), 2.53–2.04 (m, 4H, 2 × C
H2).
13C-NMR: δ 174.8 (N
CO), 172.1 (
COOCH
3), 158.2, 136.7, 129.7, 128.4, 127.9, 127.8, 127.3, 114.8, 69.8 (O
CH
2Ph), 58.5 (N
CH), 52.2 (
CH
3), 44.8 (N
CHPh), 29.4, 22.6. MS (ESI):
m/z = 340 (100) (M+Η
+). Anal. calcd for C
20H
21NO
4: C, 70.78; H, 6.24; N, 4.13 Found: C, 70.62; H, 6.27; N, 4.22.
Benzyl (2S)-2-(Hydroxymethyl)-5-oxopyrrolidine-1-carboxylate (5). To a stirred solution of compound 4 (1.20 g, 3.30 mmol) in DCM (20 mL), TFA was added (2.02 mL, 26.4 mmol) at room temperature. Once the reaction was finished (45 min), the solvent was evaporated to dryness. The residue was dissolved in toluene and the solvent was evaporated (twice) for the removal of the acid. Finally the residue was dissolved in ethyl acetate and the organic phase was washed with brine to neutral pH. After drying over Na2SO4 and evaporation of the solvent under reduced pressure, the product was obtained as a pure white solid. Yield: 0.710 g (86%); Rf 0.37 in EtOAc; mp. 83–86 °C; [α]D −54.2 (c 1.0, CHCl3). 1H-NMR (CDCl3): δ 7.37–7.33 (m, 5H, Ph), 5.23 (s, 2H, OCH2Ph), 4.29–4.23 (m, 1H, NCH), 3.95 (dd, J1 = 3.2 Hz, J2 = 11.8 Hz, 1H, CHHOH), 3.66 (dd, J1 = 3.2 Hz, J2 = 11.8 Hz, 1H, CHHOH), 3.09 (bs, 1H, OH), 3.86–1.95 (m, 4H, 2 × CH2). 13C-NMR: δ 175.5 (CON), 151.7 (COO), 135.0, 128.6, 128.4, 128.1, 68.0 (OCH2Ph), 64.1 (CH2OH), 59.4 (NCH), 32.1, 20.9. MS (ESI): m/z = 250 (100) (M+Η+). Anal. calcd for C13H15NO4: C, 62.64; H, 6.07; N, 5.62 Found: C, 62.59; H, 6.12; N, 5.59.
Benzyl (2S)-2-[(1E)-3-Methoxy-3-oxoprop-1-en-1-yl]-5-oxopyrrolidine-1-carboxylate (6). Alcohol 5 (0.22 g, 0.882 mmol) was dissolved in a mixture of dry toluene (5 mL) and dry DMSO (2.5 mL), followed by the addition of EDC·HCl (0.51 g, 2.64 mmol), the dropwise addition of dry pyridine (0.25 mL, 3.04 mmol) and finally TFA (33.2 μL, 0.441 mmol) under argon. After stirring at rt for 1.5 h the reaction was quenched by the addition of CHCl3 and the solution was washed with brine and H2O. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure. The resulting crude aldehyde was subjected to Wittig reaction without further purification. More specifically, it was dissolved in dry THF (6 mL), methyl (triphenylphosphoranylidene)acetate (0.32 g, 0.95 mmol) was added and the mixture was refluxed under argon for 1 h. The reaction was quenched by the addition of saturated solution of NH4Cl. The solvent was evaporated under reduced pressure and the residue dissolved in EtOAc was successively washed with NH4Cl, H2O and brine. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure. After purification by column chromatography using EtOAc as eluent, the product was obtained in 60% yield (0.16 g) as a colorless oil. Rf 0.63 in EtOAc; [α]D −71.1 (c 1.0, CHCl3). 1H-NMR (CDCl3): δ 7.38–7.31 (m, 5H, Ph), 6.89 (dd, J1 = 6.3 Hz, J2 = 6.2 Hz, 1H, CHCHCOOCH3), 5.25 (dd, J1 = 12.3 Hz, J2 = 12.2 Hz, 2H, CH2Ph), 4.87–4.81 (m, 1H, NCH), 3.73 (s, 3H, OCH3), 2.64–1.82 (m, 4H, 2 × CH2). 13C-NMR: δ 173.0 (NCOCH2), 166.0 (COOCH3), 150.8 (COOCH2), 145.1, 134.8, 128.5, 128.4, 128.2, 121.5 (CHCOOCH3), 68.2 (OCH2Ph), 57.8 (NCH), 51.7 (CH3), 30.7, 23.8. MS (ESI): m/z = 326 (100) (M+Na+). Anal. calcd for C17H17NO5: C, 63.36; H, 5.65; N, 4.62 Found: C, 63.29; H, 5.48; N, 4.75.
(5R)-5-(3-Hydroxypropyl)-1-[4-(2-methoxyethoxy)benzyl]-pyrrolidin-2-one (9). To a two necked flask, LiBH4 (14 mg, 0.656 mmol) was suspended in dry THF (0.5 mL) under argon at rt. A solution of compound 8 (0.110g, 0.656 mmol) in dry THF (1.5 mL) was added dropwise and the reaction mixture was stirred for 10 h. The reaction was quenched by the addition of a 20% aq. solution of AcOH at 0 °C until gas production ceased. The excess of acetic acid was neutralized by the addition of a small quantity of Na2CO3. The organic solvent was evaporated under reduced pressure and the residue was dissolved in EtOAc. The organic phase was then washed with brine, dried over Na2SO4 and evaporated under reduced pressure. After purification with column chromatography using EtOAc–MeOH 9:1 as eluent, the product was obtained as a colorless oil in 87% yield (87 mg). Rf 0.34 in EtOAc–MeOH 9:1; [α]D +7.6 (c 0.95, CHCl3). 1H-NMR (CDCl3): δ 7.13 (d, J = 8.6 Hz, 2H, Ph), 6.84 (d, J = 8.6 Hz, 2H, Ph), 4.89 (d, J = 14.8 Hz, 1H, NCHHPh), 4.10–4.05 (m, 2H, PhOCH2), 3.88 (d, J = 14.8 Hz, 1H, NCHHPh), 3.74–3.70 (m, 2H, CH2OCH3), 3.64–3.54 (m, 2H, CH2OH), 3.42 (s, 4H, NCH, CH3), 2.47–1.36 (m, 8H, 4 × CH2). 13C-NMR: δ 175.1 (NCO), 158.1, 129.2, 128.8, 114.6, 70.9 (CH2OCH3), 67.1 (PhOCH2), 62.2 (CH2OH), 59.2 (CH3), 56.6 (NCH), 43.4 (NCH2Ph), 30.2, 28.9, 27.3, 23.7. MS (ESI): m/z = 308 (90) (M+H+).Anal. calcd for C17H25NO4: C, 66.43; H, 8.20; N, 4.56 Found: C, 66.38; H, 8.26; N, 4.42.
3-{(2R)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-yl}propyl 4-Methylbenzenesulfonate (10). To an ice cooled stirred solution of alcohol 9 (35 mg 0.114 mmol) in DCM (2 mL), p-toluenesulfonylchloride (43 mg, 0.228 mmol) was added, followed by the addition of Et3N (17.5 μL). The reaction mixture was stirred at 0 °C for 10 min and overnight at room temperature. The organic layer was subsequently washed with a 1N HCl aq. solution, brine, 5% aq. solution of NaHCO3 and brine. After drying over Na2SO4 and evaporation in vacuo, the residue was purified by column chromatography using EtOAc as eluent, and the product was obtained as a yellowish oil in 72% yield (37 mg). Rf 0.35 in EtOAc; [α]D +5.6 (c 1, CHCl3). 1H-NMR (CDCl3): δ 7.75 (d, J = 8.3 Hz, 2H, Ph), 7.34 (d, J = 8.0 Hz, 2H, Ph), 7.10 (d, J = 8.6 Hz, 2H, Ph), 6.85 (d, J = 8.6 Hz, 2H, Ph), 4.83 (d, J = 14.9 Hz, 1H, NCHHPh), 4.11–4.06 (m, 2H, PhOCH2), 3.95 (t, J = 5.9 Hz, 2H, CH2OSO2), 3.83 (d, J = 14.9Hz, 1H, NCHHPh), 3.75–3.71 (m, 2H, CH2OCH3), 3.43 (s, 3H, OCH3), 3.39-3.31 (m, 1H, NCH), 2.44 (s, 3H, CH3), 2.41–1.50 (m, 8H, 4 × CH2). 13C-NMR: δ 174.9 (NCO), 158.1, 144.9, 132.8, 129.9, 129.2, 128.7, 127.8, 114.7, 70.9 (CH2OCH3), 69.9 (CH2OSO2), 67.2 (PhOCH2), 59.2 (CH3), 56.1 (NCH), 43.5 (NCH2Ph), 30.1, 28.7, 23.9, 23.5, 21.6 (PhCH3). MS (ESI): m/z = 462 (100) (M+H+). Anal. calcd for C24H31NO6S: C, 62.45; H, 6.77; N, 3.03 Found: C, 62.32; H, 6.82; N, 3.18.
3.5. General Procedure of for the Preparation of Compounds 11–13
Imidazole or 1
H-imidazole-4(5)-carboxylic acid phenylmethyl ester [
16], (1.2 mmol) and cesium carbonate (0.39 g, 1.2 mmol) were dissolved in dry DMF (3 mL) under argon. After stirring of the mixture for 30 min at 50 °C, a solution of tosyl ester
10 (1 mmol) dissolved in dry DMF (3 mL) was added and the stirring was continued overnight at 50 °C under argon. After evaporation of DMF in high vacuo, the residue was dissolved in EtOAc and the organic phase was washed with brine to neutral pH, dried over Na
2SO
4 and evaporated under reduced pressure. The residual product was purified by column chromatography (silica gel) using the appropriate solvent systems as it will be defined, in each case, below.
(5R)-5-[3-(1H-Imidazol-1-yl)propyl]-1-[4-(2-methoxyethoxy)benzyl]-pyrrolidin-2-one (11). Prepared from tosyl ester 10 and imidazole as a colorless oil. Eluent EtOAc–MeOH, 7:3. Yield: 58%. Rf 0.29 in EtOAc–MeOH, 7:3. [α]D +4.1 (c 0.8, MeOH). 1H-NMR (CD3OD): δ 7.61 (s, 1H, NCHN), 7.11 (d, J = 8.6 Hz, 2H, Ph), 7.07 (s, 1H, CH2NCHCHN), 6.98 (s, 1H, CH = NCH = CHN), 6.89 (d, J = 8.6 Hz, 2H, Ph), 4.69 (d, J = 14.9 Hz, 1H, NCHHPh), 4.12–4.07 (m, 2H, PhOCH2), 4.03–3.93 (m, 3H, NCHHPh, CH2N), 3.75–3.71 (m, 2H, CH2OCH3), 3.52–3.42 (m, 1H, NCH), 3.41 (s, 3H, CH3), 2.47–1.30 (m, 8H, 4 × CH2). 13C-NMR: δ 177.8 (NCO), 159.8, 138.5, 130.4, 130.1, 129.1, 120.6, 115.8, 72.2 (CH2OCH3), 68.5 (PhOCH2), 59.3 (CH3), 58.7 (CONCH), 47.7 (CH2N), 44.7 (NCH2Ph), 31.2, 30.6, 26.9, 24.5. MS (ESI): m/z = 358 (100) (M+H+). Anal. calcd for C20H27N3O3: C, 67.20; H, 7.61; N, 11.76 Found: C, 67.28; H, 7.52; N, 11.82.
Benzyl 1-(3-{(2R)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-ylpropyl)-1H-imidazole-5-carboxylate (
12). Prepared from tosyl ester
10 and 1
H-imidazole-4(5)-carboxylic acid phenylmethyl ester [
16], as a mixture with its constitutional isomer
13. Eluent EtOAc-MeOH, 9:1. Yield: 55% (colorless oil). Rf 0.42 in EtOAc–MeOH, 7:3. [α]
D +2.8 (c 1, CHCl
3).
1H-NMR (CDCl
3): δ 7.78 (s, 1H, NC
HN), 7.58 (s, 1H, NC
HCOO), 7.40–7.34 (m, 5H, Ph), 7.08 (d,
J = 8.6 Hz, 2H, Ph), 6.85 (d,
J = 8.6 Hz, 2H, Ph), 5.28 (s, 2H, OC
H2Ph), 4.78 (d,
J = 14.9 Hz, 1H, NCH
HPh), 4.20 (t,
J = 6.7 Hz, 2H, C
H2N), 4.10–4.06 (m, 2H, PhO
CH
2), 3.87 (d,
J = 14.9 Hz, 1H, NCH
HPh), 3.74–3.70 (m, 2H, C
H2OCH
3), 3.42 (s, 3H, C
H3), 3.41–3.33 (m, 1H, NC
H), 2.43–1.24 (m, 8H, 4 × C
H2).
13C-NMR: δ 174.9 (N
CO), 159.8 (COO), 158.1, 141.8, 137.9, 135.5, 129.1, 128.8, 128.6, 128.5, 128.3, 128.0, 114.7, 70.9 (
CH
2OCH
3), 67.2 (PhO
CH
2), 66.2 (COO
CH
2Ph), 59.1 (
CH
3), 56.3 (N
CH), 46.6 (
CH
2NCCOO), 43.6 (N
CH
2Ph), 30.1, 29.6, 26.0, 23.6. MS (ESI):
m/z = 492 (100) (M+H
+). Anal. calcd for C
28H
33N
3O
5: C, 68.41; H, 6.77; N, 8.55 Found: C, 68.32; H, 6.60; N, 8.57.
Benzyl 1-(3-{(2R)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-yl}propyl)-1H-imidazole-4-carboxylate (
13). Prepared from tosyl ester
10 and 1
H-imidazole-4(5)-carboxylic acid, phenylmethyl ester [
16], as a mixture with its constitutional isomer
12. After the separation of isomer
12, the elution system (EtOAc–MeOH, 9:1) was gradually changed to the more polar EtOAc–MeOH, 3:2 in order to recover isomer 13. Yield: 30% (colorless oil). Rf 0.56 in EtOAc–MeOH, 3:2. [α]
D +9.9 (c 1, CHCl
3).
1H-NMR (CDCl
3): δ 7.53 (s, 1H, NC
HN), 7.51 (s, 1H, NC
HCOO), 7.46–7.31 (m, 5H, Ph), 7.07 (d,
J = 8.7 Hz, 2H, Ph), 6.84 (d,
J = 8.7 Hz, 2H, Ph), 6.34 (s, 2H, COOC
H2Ph), 4.73 (d,
J = 14.9 Hz, 1H, NCH
HPh), 4.09–4.05 (m, 2H, PhO
CH
2), 3.97–3.82 (m, 3H, NCH
HPh, C
H2N), 3.74–3.69 (m, 2H, C
H2OCH
3), 3.43 (s, 3H, C
H3), 3.41–3.36 (m, 1H, NC
H), 2.45–1.24 (m, 8H, 4 × C
H2).
13C-NMR: δ 174.9 (N
CO), 162.2 (COO), 158.2, 137.9, 136.0, 133.6, 129.1, 128.7, 128.5, 128.4, 128.2, 124.9, 114.7, 70.9 (
CH
2OCH
3), 67.2 (PhO
CH
2), 66.2 (COO
CH
2Ph), 59.2 (
CH
3), 56.4 (N
CH), 47.3 (
CH
2NCHN), 43.9 (N
CH
2Ph), 30.1, 29.7, 25.8, 23.6. MS (ESI):
m/z = 492 (100) (M+H
+). Anal. calcd for C
28H
33N
3O
5: C, 68.41; H, 6.77; N, 8.55 Found: C, 68.38; H, 6.82; N, 8.45.
3.6. General Procedure for the Preparation of Compounds 7, 14, 15, 19a–c, 21, 25
A mixture of a compound possessing a double bond and also the N- benzyloxycarbonyl group (compound 6), benzyl ester (compounds 12,13), benzyl ether (compound 20) or benzyl ester and N-trityl imidazole group (compounds 18a–c, 24) (1 mmol) in MeOH (10 mL) and 10% palladium on activated carbon (0.02 g or 0.04 g for compounds 6, 18a–c, 24) was hydrogenated for 1.5–3 h (overnight for compound 6) under atmospheric conditions. After filtration through a pad of Celite, the solvent was removed in vacuo to give the deprotected final compound in almost quantitative yield and high purity. In the case of compounds 18a–c and 24 the triphenylmethane produced from the hydrogenation of trityl protecting group was completely removed by adding a small quantity of CHCl3 and discounting the supernatant where the polar products (19a–c, 25) were insoluble.
Methyl 3-[(2S)-5-Oxopyrrolidin-2-yl] propanoate (7). Prepared from alkene 6. The product was obtained as a yellowish solid in 94% yield and high purity. Rf 0.14 in EtOAc; mp. 72–75 °C; [α]D +14.3 (c 0.95, CHCl3). 1H-NMR (CDCl3): δ 7.01 (bs, 1H, NH), 3.73–3.64 (m, 1H, NCH), 3.66 (s, 3H, CH3), 2.41–1.60 (m, 8H, 4 × CH2). 13C-NMR: δ 178.6 (NCO), 173.1 (COOCH3), 53.7 (NCH), 51.4 (CH3), 31.3, 30.1, 26.5. MS (ESI): m/z = 172 (100) (M+Η+). Anal. calcd for C8H13NO3: C, 56.13; H, 7.65; N, 8.18 Found: C, 56.22; H, 7.58; N, 8.22.
1-(3-{(2R)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-yl}propyl)-1H-imidazole-5-carboxylic acid (14). Catalytic hydrogenation of compound 12 afforded product 14 as a pure colorless oil in 99% yield. Rf 0.18 in EtOAc–MeOH, 3:2. [α]D +2.1 (c 1, MeOH). 1H-NMR (CD3OD): δ 8.09 (s, 1H, NCHN), 7.80 (s, 1H, NCHCCOO), 7.12 (d, J = 8.5 Hz, 2H, Ph), 6.88 (d, J = 8.5 Hz, 2H, Ph), 4.71 (d, J = 15.0 Hz, 1H, NCHHPh), 4.41–4.34 (m, 2H, CH2NCHCOO), 4.11–3.98 (m, 3H, NCHHPh, PhOCH2), 3.75–3.71 (m, 2H, CH2OCH3), 3.55–3.48 (m, 1H, NCHCH2), 3.41 (s, 3H, OCH3), 2.46–1.28 (m, 8H, 4 × CH2). 13C-NMR: δ 177.7 (NCO), 162.8 (COOH), 159.8, 142.0, 134.2, 130.4, 130.0, 126.5, 115.8, 72.2 (CH2OCH3), 68.4 (PhOCH2), 59.3 (CH3), 58.6 (NCH), 48.1 (CH2NCCOOH), 44.5 (NCH2Ph), 31.2, 30.5, 27.0, 24.5. MS (ESI): m/z = 402 (100) (M+H+). Anal. calcd for C21H27N3O5: C, 62.83; H, 6.78; N, 10.47 Found: C, 62.88; H, 6.73; N, 10.38.
1-(3-{(2R)-1-[4-(2-Methoxyethoxy)benzyl]-5-oxopyrrolidin-2-yl}propyl)-1H-imidazole-4-carboxylic acid (15). Catalytic hydrogenation of compound 13 afforded product 15 as a pure colorless oil in 90% yield. Rf 0.15 in EtOAc–MeOH, 1:1. [α]D +0.9 (c 1, MeOH). 1H-NMR (CD3OD): δ 7.83 (s, 1H, NCHN), 7.68 (s, 1H, NCHCOO), 7.10 (d, J = 8.2 Hz, 2H, Ph), 6.88 (d, J = 8.2 Hz, 2H, Ph), 4.60 (d, J = 14.9 Hz, 1H, NCHHPh), 4.11–3.99 (m, 5H, PhOCH2, NCHHPh, CH2NCHCOO), 3.75–3.71 (m, 2H, CH2OCH3), 3.51–3.47 (m, 1H, NCH), 3.41 (s, 3H, OCH3), 2.47–1.21 (m, 8H, 4 × CH2). 13C-NMR: δ 177.8 (NCO), 165.6 (COOH), 159.8, 139.5, 136.0, 134.7, 130.4, 130.0, 126.4, 115.8, 72.3 (CH2OCH3), 68.4 (PhOCH2), 59.3 (CH3), 58.5 (NCH), 48.4 (CH2NCHN), 44.7 (NCH2Ph), 31.2, 30.6, 26.8, 24.6. MS (ESI): m/z = 402 (100) (M+H+). Anal. calcd for C21H27N3O5: C, 62.83; H, 6.78; N, 10.47 Found: C, 62.74; H, 6.84; N, 10.35.
1-[4-(2-Methoxyethoxy)benzyl]-5-oxo-L-prolyl-L-histidine (19a). Prepared from compound 18a as a colorless oil in 97% yield. Rf 0.1 in EtOAc–MeOH, 1:1. [α]D +3.0 (c 1, MeOH). 1H-NMR (CD3OD): δ 8.85 (s, 1H, NCHN), 7.34 (s, 1H, NCHNCH), 7.05 (d, J = 8.5 Hz, 2H, Ph), 6.88 (d, J = 8.5 Hz, 2H, Ph), 4.84–4.72 (m, 2H, NHCHCOO, NCHHPh), 4.10–4.06 (m, 2H, PhOCH2), 4.01–3.95 (m, 1H, NCHCON), 3.75–3.62 (m, 3H, CH2OCH3, NCHHPh), 3.42 (s, 3H, CH3), 3.15–3.02 (m, 2H, CH2CHN), 2.57–1.96 (m, 4H, 2 × CH2). 13C-NMR: δ 178.3 (NCO), 173.9, 173.2, 160.0, 135.3, 131.7, 130.8, 129.1, 118.3, 115.9, 72.2 (CH2OCH3), 68.5 (OCH2), 61.1 (NCHCON), 59.3 (OCH3), 52.7 (HNCHCO), 45.8 (CH2Ph), 30.8 (CH2CHN), 28.0, 24.2. MS (ESI): m/z = 431 (100) (M+Η+). Anal. calcd for C21H26N4O6: C, 58.59; H, 6.09; N, 13.02 Found: C, 58.65; H, 5.98; N, 13.14.
1-[4-(3-Methoxypropoxy)benzyl]-5-oxo-L-prolyl-L-histidine (19b). Prepared from compound 18b as a colorless oil in 99% yield. Rf 0.11 in EtOAc–MeOH, 1:1. [α]D +11.5 (c 1, MeOH). 1H-NMR (CD3OD): δ 8.75 (s, 1H, NCHN), 7.32 (s, 1H, NCHNC), 7.04 (d, J = 8.5 Hz, 2H, Ph), 6.85 (d, J = 8.5 Hz, 2H, Ph), 4.88 (d, J = 14.8 Hz, 1H, NCHHPh), 4.75–4.65 (m, 1H, NHCHCOO), 4.04–3.98 (m, 1H, NCHCO), 4.01 (t, J = 6.3 Hz, 2H, PhOCH2), 3.63 (d, J = 14.8 Hz, 1H, NCHHPh), 3.54 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.33 (s, 3H, OCH3), 3.28–3.03 (m, 2H, CH2CN), 2.53–1.93 (m, 6H, 3 × CH2). 13C-NMR: δ 178.3 (NCO), 173.7, 160.2, 135.2, 132.1, 131.0, 130.7, 128.9, 118.2, 115.8, 70.3 (CH2OCH3), 65.9 (PhOCH2), 61.2 (NCH), 58.9 (OCH3), 53.4 (CHCOOH), 45.8 (NCH2Ph), 30.9, 30.6, 28.4, 24.2. MS (ESI): m/z = 445 (100) (M+Η+). Anal. calcd for C22H28N4O6: C, 59.45; H, 6.35; N, 12.61 Found: C, 59.52; H, 6.37; N, 12.50.
1-[4-(4-Methoxybutoxy)benzyl]-5-oxo-L-prolyl-L-histidine (19c). Prepared from compound 18c as a colorless oil in 92% yield. Rf 0.16 in EtOAc–MeOH, 1:1. [α]D +6.9 (c 1, MeOH). 1H-NMR (CD3OD): δ 8.84 (s, 1H, HNCHN), 7.36 (s, 1H, NCCHN), 7.04 (d, J = 8.5 Hz, 2H, Ph), 6.85 (d, J = 8.5 Hz, 2H, Ph), 4.89 (d, J = 14.7 Hz, 1H, NCHHPh), 4.80–4.73 (m, 1H, NHCHCOO), 4.02–3.96 (m, 1H, NCH), 3.95 (t, J = 5.4 Hz, 2H, PhOCH2), 3.63 (d, J = 14.7 Hz, 1H, NCHHPh), 3.45 (t, J = 6.1 Hz, 2H, CH2OCH3), 3.33 (s, 3H, CH3), 3.17–3.05 (m, 2H, CH2CNH), 2.49-1.96 (m, 4H, 2 × CH2), 1.84–1.65 (m, 4H, (OCH2CH2)2). 13C-NMR: δ 178.2 (NCO), 173.9, 160.3, 135.2, 131.8, 131.1, 130.7, 128.8, 118.3, 115.8, 73.5 (CH2OCH3), 68.9 (PhOCH2), 61.1 (NCH), 58.8 (OCH3), 52.9 (CHCOOH), 45.8 (NCH2Ph), 30.8, 28.0, 27.3, 27.2, 24.1. MS (ESI): m/z = 459 (100) (M+Η+). Anal. calcd for C23H30N4O6: C, 60.25; H, 6.59; N, 12.12 Found: C, 60.32; H, 6.47; N, 12.30.
Methyl 1-(4-Hydroxybenzyl)-5-oxo-L-prolinate (21). Prepared from compound 20 as a colorless oil in 99% yield. Rf 0.69 in EtOAc. [α]D +35.5 (c 1.1, CHCl3). 1H-NMR (CDCl3): δ 7.03 (d, J = 7.5 Hz, 2H, Ph), 6.80 (d, J = 7.5 Hz, 2H, Ph), 4.89 (d, J = 14.5 Hz, 1H, NCHHPh), 4.02–3.91 (m, 2H, NCHHPh, NCH), 3.67 (s, 3H, CH3), 2.55–2.04 (m, 4H, 2xCH2). 13C-NMR: δ 175.7 (NCO), 172.2 (COOCH3), 156.3, 130.0, 126.5, 115.7, 58.9 (NCH), 52.5 (COOCH3), 45.3 (NCHPh), 29.8, 22.7. MS (ESI): m/z = 250 (97) (M+Η+). Anal. calcd for C13H15NO4: C, 62.64; H, 6.07; N, 5.62 Found: C, 62.59; H, 6.17; N, 5.54.
1-(4-{[(4R)-3-(tert-Butoxycarbonyl)-2,2-dimethyl-1,3-oxazolidin-4-yl]methoxy}benzyl)-5-oxo-L-prolyl-L-histidine (25). Prepared from compound 24 as a colorless oil in 97% yield. Rf 0.18 in EtOAc–MeOH, 1:1. [α]D −24.9 (c 0.8, MeOH). 1H-NMR (CD3OD): δ 8.90 (s, 1H, HNCHN), 7.40 (s, 1H, HNCHNCH), 7.05 (d, J = 8.3 Hz, 2H, Ph), 6.93 (d, J = 8.3 Hz, 2H, Ph), 4.87–4.75 (m, 2H, CHCOOH, NCHHPh), 4.17–3,81 (m, 6H, NCHCO, PhOCH2CHCH2O), 3.64 (d, J = 15.0 Hz, 1H, NCHHPh), 3.41–3.06 (m, 2H,HOOCCHCH2), 2.58–1.94 (m, 4H, 2xCH2), 1.58–1.45 (m, 15H, 5 × CH3). 13C-NMR: δ 173.9 (NCO), 172.8, 159.8, 153.4 (NCOO), 135.3, 131.5, 130.8, 129.5, 129.3, 118.4, 115.9, 95.0 (NC(CH3)2O), 82.2 (C(CH3)3), 67.3, 66.2, 61.1 (NCHCO), 57.5 (PhOCH2), 52.5 (NHCHCOOH), 45.8 (NCH2Ph), 30.8, 28.8 (CH3), 27.9, 27.7, 24.5, 24.2. MS (ESI): m/z = 586 (100) (M+Η+). Anal. calcd for C29H39N5O8: C, 59.47; H, 6.71; N, 11.96 Found: C, 59.52; H, 6.68; N, 12.05.
3.7. General Procedure for the Preparation of Compounds 17a–c, 23
To a stirred solution of methylester (compounds 16a–c, 22) (1 mmol), in MeOH (3 mL), an aq. solution of 2N NaOH (0.5 ml, 1 mmol) was added and the reaction mixture was left stirring at rt for 2–3 h. Upon completion of the reaction, the MeOH was evaporated under reduced pressure and the residual product was diluted with H2O and extracted with Et2O (1 × 10 mL). The aqueous phase was acidified with 1 N HCl aq. solution at pH 2 and extracted with EtOAc (2 × 10 mL). The combined organic phases were neutralized by washing with brine and H2O, dried over Na2SO4 and evaporated under reduced pressure to afford the carboxylic products in quantitative yield and high purity.
1-[4-(2-Methoxyethoxy)benzyl]-5-oxo-L-proline (17a). Prepared from methylester 16a as a colorless oil in 94% yield. Rf 0.36 in EtOAc–MeOH, 3:2. [α]D +43.8 (c 1, CHCl3). 1H-NMR (CDCl3): δ 8.77 (bs, 1H, COOH), 7.13 (d, J = 8.6 Hz, 2H, Ph), 6.85 (d, J = 8.6 Hz, 2H, Ph), 5.02 (d, J = 14.7 Hz, 1H, CHHPh), 4.11–4.06 (m, 2H, PhOCH2), 3.99–3.93 (m, 1H, NCHCOO), 3.91 (d, J = 4.7 Hz, 1H, CHHPh), 3.77–3.72 (m, 2H, CH2OCH3), 3.44 (s, 3H, OCH3), 2.68–2.09 (m, 4H, 2 × CH2). 13C-NMR: δ 176.2 (NCO), 174.1(COOH), 158.4, 129.9, 127.6, 114.7, 70.9 (CH2OCH3), 67.1 (OCH2), 59.1 (NCH), 58.5 (OCH3), 45.0 (CH2Ph), 29.6, 22.8. MS (ESI): m/z = 292 (100) (M−Η+). Anal. calcd for C15H19NO5: C, 61.42; H, 6.53; N, 4.78 Found: C, 61.38; H, 6.57; N, 4.82.
1-[4-(3-Methoxypropoxy)benzyl]-5-oxo-L-proline (17b). Prepared from methylester 16b, as a colorless oil in 99% yield. Rf 0.24 in EtOAc–MeOH, 3:2. [α]D +17.2 (c 1, CHCl3). 1H-NMR (CDCl3): δ 9.62 (bs, 1H, COOH), 7.13 (d, J = 8.6 Hz, 2H, Ph), 6.83 (d, J = 8.6 Hz, 2H, Ph), 5.05 (d, J = 4.7 Hz, 1H, CHHPh), 4.04–3.94 (m, 3H, NCHCOO, PhOCH2), 3.91 (d, J = 14.7 Hz, 1H, CHHPh), 3.56 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.35 (s, 3H, OCH3), 2.66–2.09 (m, 4H, 2 × CH2), 2.09–1.96 (m, 2H, CH2CH2CH2). 13C NMR: δ 176.2 (NCO), 174.2 (COOH), 158.6, 129.9, 127.2, 114.6, 69.2 (CH2OCH3), 64.7 (PhOCH2), 58.6 (NCH), 58.5 (OCH3), 45.0 (CH2Ph), 29.7, 29.4, 22.8. MS (ESI): m/z = 306 (100) (M−Η+). Anal. calcd for C16H21NO5: C, 62.53; H, 6.89; N, 4.56 Found: C, 62.58; H, 6.77; N, 4.53.
1-[4-(4-Methoxybutoxy)benzyl]-5-oxo-L-proline (17c). Prepared from methylester 16c, as a colorless oil in 96% yield. Rf 0.30 in EtOAc–MeOH, 3:2. [α]D +13.3 (c 1, CHCl3). 1H-NMR (CDCl3): δ 9.98 (bs, 1H, COOH), 7.12 (d, J = 8.6 Hz, 2H, Ph), 6.81 (d, J = 8.6 Hz, 2H, Ph), 5.03 (d, J = 14.7 Hz, 1H, NCHHPh), 4.00–3.88 (m, 4H, NCHHPh, NCH, PhOCH2), 3.44 (t, J = 6.2Hz, 2H, CH2OCH3), 3.34 (s, 3H, OCH3), 2.65–2.08 (m, 4H, 2xCH2), 1.85–1.68 (m, 4H, (OCH2CH2)2). 13C-NMR: δ 176.2 (NCO), 173.8 (COO), 158.6, 129.9, 127.1, 114.6, 72.3 (CH2OCH3), 67.5 (PhOCH2), 58.4 (NCH), 58.3 (OCH3), 45.0 (CH2Ph), 29.6, 26.0, 25.8, 22.8. MS (ESI): m/z = 320 (100) (M−Η+). Anal. calcd for C17H23NO5: C, 63.54; H, 7.21; N, 4.36 Found: C, 63.38; H, 7.19; N, 4.42.
1-(4-{[(4R)-3-(tert-Butoxycarbonyl)-2,2-dimethyl-1,3-oxazolidin-4-yl]methoxy}benzyl)-5-oxo-L-proline (23). Prepared from methylester 22, as a colorless oil in 94% yield. Rf 0.43 in EtOAc–MeOH, 1:1. [α]D −11.5 (c 1, CHCl3). 1H-NMR (CDCl3): δ 7.62 (bs, 1H, COOH), 7.13 (d, J = 7.4 Hz, 2H, Ph), 6.88 (d, J = 4 Hz, 2H, Ph), 5.03 (dd, J1 = 14.6 Hz, J2 = 3.1 Hz, 1H, NCHHPh), 4.28–3.80 (m, 7H, NCHHPh, NCHCOOH, OCH2CHCH2O), 4.68–2.15 (m, 4H, 2 × CH2), 1.64-1.52 (m, 15H, 5 × CH3). 13C-NMR: δ 176.1 (NCO), 173.9 (COOH), 158.1, 152.4 (NCOO), 129.9, 127.6, 114.8, 93.6 (OC(CH3)2N), 80.9 (C(CH3)3), 66.0, 65.1, 58.4 (NCHCOOH), 55.9 (PhOCH2), 45.0 (NCH2Ph), 29.6, 28.3 (CH3), 27.4, 24.2, 22.8. MS (ESI): m/z = 447 (100) (M-Η+). Anal. calcd for C23H32N2O7: C, 61.59; H, 7.19; N, 6.25 Found: C, 61.53; H, 7.24; N, 6.32.
3.8. General Procedure for the Preparation of Compounds 18a–c, 24
To a stirred solution of carboxylic acids (compounds 17a–c, 23) (1 mmol) in CH2Cl2 (10 mL) at 0 °C, 1-hydroxybenzotriazole (HOBt) (0.135 g, 1 mmol), freshly prepared benzyl 3-trityl-L-histidinate (42) (0.537 g, 1.1 mmol), Et3N (0.154 mL, 1.1 mmol) and N-ethyl-N-dimethylaminopropyl-carbodiimide hydrochloride (EDC.HCl) (0.210 g, 1.1 mmol) were added consecutively. The reaction mixture was left stirring at 0 °C for 1 h and then warmed to room temperature and left stirring for 18 h. The solvents were evaporated under reduced pressure and the crude product was dissolved in EtOAc (20 mL). The organic layer was washed with 5% aq. H2SO4, H2O, 5% aq. NaHCO3, and brine. After drying over Na2SO4 and evaporation of the solvent in vacuo, the crude ester was purified using column chromatography with the appropriate solvent systems as it will be defined in each case below.
Benzyl 1-[4-(2-Methoxyethoxy)benzyl]-5-oxo-L-prolyl-3-trityl-L-histidinate (18a). Prepared from compound 17a. Eluent EtOAc–MeOH 9:1. Yield: 55% (colorless oil); Rf 0.55 in EtOAc–MeOH 9:1; [α]D +3.8 (c 1, CHCl3). 1H-NMR (CDCl3): δ 8.22 (d, J = 7.8 Hz, 1H, NH), 7.39 (d, J = 1.3 Hz, 1H, NCHN), 7.33–7.03 (m, 22H, Ph), 6.80 (d, J = 8.6 Hz, 2H, Ph), 6.47 (s, 1H, NCHNCHC), 5.01 (d, J = 14.3 Hz, 1H, CHHPh), 5.00 (s, 2H, OCH2Ph), 4.86–4.77 (m, 1H, NHCHCOO), 4.12–4.04 (m, 2H, PhOCH2), 3.89–3.83 (m, 1H, NCHCON), 3.75 (d, J = 14.3 Hz, 1H, CHHPh), 3.74–3.69 (m, 2H, CH2OCH3), 3.43 (s, 3H, OCH3), 3.13–2.91 (m, 2H, COCHCH2CHN), 2.68–1.98 (m, 4H, 2 × CH2). 13C-NMR: δ 175.5 (NCO), 171.6, 170.6, 158.2, 142.0, 138.7, 135.9, 135.2, 129.9, 129.6, 128.5, 128.4, 128.3, 128.2, 128.1, 119.5, 119.4, 114.6, 75.4 (NCPh), 70.9 (CH2OCH3), 67.1 (OCH2), 67.0 (OCH2Ph), 60.0 (NCH), 59.2 (OCH3), 52.5 (HNCHCO), 44.5 (CH2Ph), 29.7, 28.8 (NHCHCH2C), 23.2. MS (ESI): m/z = 763 (100) (M+Η+). Anal. calcd for C47H46N4O6: C, 74.00; H, 6.08; N, 7.34 Found: C, 74.15; H, 6.16; N, 7.27.
Benzyl 1-[4-(3-Methoxypropoxy)benzyl]-5-oxo-L-prolyl-3-trityl-L-histidinate (18b). Prepared from compound 17b. Eluent EtOAc–MeOH 9:1. Yield: 68% (colorless oil); Rf 0.63 in EtOAc–MeOH 9:1; [α]D +8.5 (c 1, CHCl3). 1H-NMR (CDCl3): δ 8.19 (d, J = 7.8 Hz, 1H, NH), 7.40 (d, J = 1.3 Hz, 1H, NCHN), 7.34–7.04 (m, 22H, Ph), 6.78 (d, J = 8.7 Hz, 2H, Ph), 6.48 (d, J = 1.2 Hz, 1H, NCHCN), 5.02 (d, J = 14.5 Hz, 1H, NCHHPh), 5.01 (s, 2H, CH2Ph), 4.00 (t, J = 6.3 Hz, 2H, PhOCH2), 3.89–3.83 (m, 1H, NCHCO), 3.76 (d, J = 14.5 Hz, 1H, NCHHPh), 3.53 (t, J = 6.2 Hz, 2H, CH2OCH3), 3.34 (s, 3H, OCH3), 3.14–2.92 (m, 2H, HNCHCH2), 2.69–2.08 (m, 4H, 2 × CH2), 2.05–1.95 (m, 2H, CH2CH2CH2). 13C-NMR: δ 175.5 (NCO), 171.6, 170.6, 158.4, 142.1, 138.7, 136.0, 135.2, 130.0, 129.6, 128.5, 128.3, 128.2, 128.1, 128.0, 119.5, 114.5, 75.4 (CPh), 69.2 (CH2OCH3), 67.0 (OCH2Ph), 64.7 (PhOCH2), 60.0 (NCH), 58.7 (OCH3), 52.5 (HNCHCO), 44.5 (CH2Ph), 29.8, 29.5 (CH2CH2CH2), 28.9 (NHCHCH2C), 23.2. MS (ESI): m/z = 777 (100) (M+Η+). Anal. calcd for C48H48N4O6: C, 74.21; H, 6.23; N, 7.21 Found: C, 74.11; H, 6.32; N, 7.28.
Benzyl 1-[4-(4-Methoxybutoxy)benzyl]-5-oxo-L-prolyl-3-trityl-L-histidinate (18c). Prepared from compound 17c. Eluent EtOAc–MeOH 9:1. Yield: 63% (colorless oil); Rf 0.56 in EtOAc–MeOH 9:1; [α]D +3.8 (c 1, CHCl3). 1H-NMR (CDCl3): δ 8.21 (d, J = 7.9 Hz, 1H, NH), 7.40 (d, J = 1.3 Hz, 1H, NCHN), 7.35–7.04 (m, 22H, Ph), 6.76 (d, J = 8.6 Hz, 2H, Ph), 6.48 (d, J = 1.1 Hz, 1H, NCHCN), 5.02 (d, J = 14.5 Hz, 1H, NCHHPh), 5.01 (s, 2H, COOCH2Ph), 4.87–4.79 (m, 1H, NCHCOO), 3.92 (t, J = 5.9 Hz, 2H, PhOCH2), 3.88–3.83 (m, 1H, NCH), 3.76 (d, J = 14.5 Hz, 1H, NCHHPh), 3.43 (t, J = 6.1 Hz, 2H, CH2OCH3), 3.34 (s, 3H, OCH3), 3.13–2.92 (m, 2H, CHCH2CN), 2.65–1.98 (m, 4H, 2 × CH2), 1.86–1.65 (m, 4H, (OCH2CH2)2). 13C-NMR: δ 175.4 (NCO), 171.6, 170.5, 158.4, 142.0, 138.7, 135.9, 135.2, 129.9, 129.6, 128.4, 128.3, 128.1, 128.0, 127.9, 119.4, 114.5, 75.3 (CPh), 72.2 (CH2OCH3), 67.5 (OCH2Ph), 66.9 (PhOCH2), 59.9 (NCH), 58.5 (OCH3), 52.5 (HNCHCO), 44.5 (CH2Ph), 29.7, 28.8, 26.1, 25.9, 23.2. MS (ESI): m/z = 791 (100) (M+Η+). Anal. calcd for C49H50N4O6: C, 74.41; H, 6.37; N, 7.08 Found: C, 74.35; H, 6.42; N, 7.18.
Benzyl 1-(4-{[(4R)-3-(tert-Butoxycarbonyl)-2,2-dimethyl-1,3-oxazolidin-4-yl]methoxy}benzyl)-5-oxo-L-prolyl-3-trityl-L-histidinate (24). Prepared from compound 23. Eluent EtOAc-petroleum ether (bp. 40–60 °C) 9:1, followed by EtOAc. Yield: 60% (colorless oil); Rf 0.64 in EtOAc; [α]D −16.4 (c 1, CHCl3). 1H-NMR (CDCl3): δ 8.23 (t, J = 8.3 Hz, 1H, NH), 7.40–6.80 (m, 25H, Ph), 6.47 (s, 1H, Ph), 5.05–4.98 (m, 3H, COOCH2, NCHHPh), 4.86–4.77 (m, 1H, HNCHCOO), 4.26–3.69 (m, 7H, NCHHPh, NCHCONH, PhOCH2CHCH2O), 3.13–2.92 (m, 2H, HNCHCH2C), 2.68–2.03 (m, 4H, 2×CH2), 1.61–1.48 (m, 15H, 5 × CH3). 13C-NMR: δ 175.5 (NCO), 171.6, 170.5, 158.0, 152.2 (NCOO), 142.0, 138.6, 135.8, 135.2, 130.0, 129.9, 129.6, 128.6, 128.5, 128.3, 128.1, 119.6, 114.7, 93.5 (OC(CH3)2N), 80.2 (C(CH3)3), 75.4 (CPh3), 67.0 (COOCH2Ph), 66.1, 65.3, 60.0 (NCHCO), 55.7 (PhOCH2), 52.5 (NHCHCOO), 44.5 (NCH2Ph), 29.7, 28.5 (CH3), 27.5, 24.2, 23.3, 23.0. MS (ESI): m/z = 919 (100) (M+Η+). Anal. calcd for C55H59N5O8: C, 71.95; H, 6.48; N, 7.63 Found: C, 72.07; H, 6.24; N, 7.58.
Μethyl 1-(4-{[(4R)-3-(tert-Butoxycarbonyl)-2,2-dimethyl-1,3-oxazolidin-4-yl]methoxy}benzyl)-5-oxo-L-prolinate (
22). To a stirred solution of alcohol (
tert-butyl (4
R)-4-(hydroxymethyl)-2,2-dimethyl-1,3-oxazolidine-3-carboxylate) [
33], (0.231g, 1 mmol) in dry toluene (5 mL), compound
21 (0.249 g, 1 mmol), DEAD (0.19 mL, 1.2 mmol), and PPh
3 (0.314 g, 1.2 mmol) were added consecutively and the reaction mixture was refluxed for 15 h under argon. After evaporation of the solvent under reduced pressure the product was purified using column chromatography and EtOAc–petroleum ether (bp. 40–60 °C) 7:3 as eluent and isolated as a colorless oil in 60% yield (0.278 g). Rf 0.45 in EtOAc–petroleum ether (bp. 40–60 °C) 7:3; [α]
D −17.7 (c 1, CHCl
3).
1H-NMR (CDCl
3): δ 7.10 (d,
J = 7.9 Hz, 2H, Ph), 6.70 (bs, 2H, Ph), 4.94 (d,
J = 14.6 Hz, 1H, NCH
HPh), 4.21–3.81 (m, 7H, NCH
HPh, NC
HCOOCH
3, PhOC
H2C
HC
H2O), 3.67 (s, 3H, OC
H3), 2.64–2.02 (m, 4H, 2 × C
H2), 1.60–1.48 (m, 15H, 5 × C
H3).
13C-NMR: δ 174.8 (N
CO), 172.2 (
COOCH
3), 158.1, 152.2 (N
COO), 129.8, 127.9, 114.7, 93.4 (N
C(CH
3)
2O), 80.4 (
C(CH
3)
3), 66.0, 65.1, 58.5 (N
CHCOO), 55.6 (PhO
CH
2), 52.3 (COO
CH
3), 44.8 (N
CHPh), 29.5, 28.3 (C(
CH
3)
3), 27.4, 24.2, 22.6. MS (ESI):
m/z = 480 (100) (M+NΗ
4+). Anal. calcd for C
24H
34N
2O
7: C, 62.32; H, 7.41; N, 6.06 Found: C, 62.28; H, 7.39; N, 6.15.
1-(4-{[(2S)-2-Ammonio-3-Hydroxypropyl]oxy}benzyl)-5-oxo-L-prolyl-L-histidine hydrochloride (26). To a stirred solution of compound 25 (21 mg, 0.036 mmol) in dioxane (1 mL) at 0 °C, a 4 M solution of HCl in dioxane (0.108 mL, 0.432 mmol) was added and the reaction mixture was left stirring at room temperature for 1 h. After evaporation of the solvent under reduced pressure, the product was precipitated by the addition of Et2O and recrystallized from dioxane/Et2O twice. It was obtained as a white gummy solid in 90% yield (16 mg). [α]D +3.4 (c 0.7, H2O). 1H-NMR (D2O): δ 8.87 (s, 1H, HNCHN), 7.32 (s, 1H, HNCHNCH), 7.14 (d, J = 8.4 Hz, 2H, Ph), 6.99 (d, J = 8.4 Hz, 2H, Ph), 4.75–4.70 (m, 2H, CHCOOH, NCHHPh), 4.33–4.19 (m, 2H, PhOCH2), 4.16–4.12 (m, 1H, NCH), 3.98–3.80 (m, 4H, NCHHPh, CHNH2, CH2OH), 3.33 (dd, J1 = 5.4 Hz, J2 = 15.6 Hz, 1H, HNCHCHHC), 3.15 (dd, J1 = 9.0 Hz, J2 = 15.5 Hz, 1H, HNCHCHHC), 2.61-1.87 (m, 4H, 2 × CH2). 13C-NMR: δ 179.6 (NCO), 174.2, 173.6, 158.0, 134.2, 130.3, 129.5, 128.7, 117.7, 115.5, 65.6 (PhOCH2), 61.3 (NCHCO), 59.3 (CH2OH), 52.8, 52.3, 45.7 (NCH2Ph), 30.4, 26.6, 23.2. MS (ESI): m/z = 446 (100) (M+Η+). Anal. calcd for C21H28N5O6Cl: C, 52.34; H, 5.86; N, 14.53 Found: C, 52.28; H, 5.92; N, 14.47.
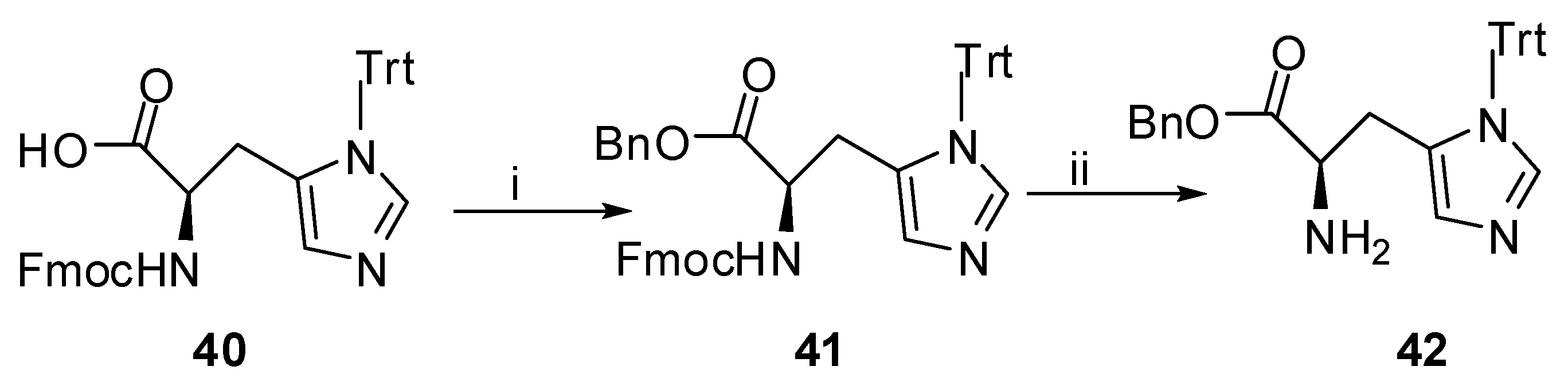
Benzyl N-[(9H-Fluoren-9-ylmethoxy)carbonyl]-1-trityl-L-histidinate (41). To a solution of Fmoc-S-His(Trt)-OH (0.20 g, 0.33 mmol) in DMF (1 mL) Cs2CO3 (53 mg, 0.162 mmol) was added with some drops of H2O to dissolve the inorganic salt. The solvent was evaporated under reduced pressure to dryness and the residue, dissolved in DMF (3 mL) was stirred for 5 min at rt, followed by the addition of benzyl bromide (44 μL, 0.371 mmol). After stirring overnight at rt and evaporation of DMF under reduced pressure, the residue was dissolved in EtOAc and the organic phase was washed with H2O, 5% aq. solution of NaHCO3 and H2O. The organic layer was dried over Na2SO4, evaporated in vacuo and the residue was purified by column chromatography on silica gel, eluting with EtOAc-petroleum ether (bp. 40–60 °C) 1:1. The product was isolated as colorless oil in 87% yield (0.20 g). Rf 0.50 in EtOAc–petroleum ether (bp. 40–60 °C) 1:1; [α]D −2.7 (c 1, CHCl3). 1H-NMR (CDCl3): δ 7.77–7.06 (m, 29H, Ph, NCHN), 6.61 (d, J = 8.2 Hz, 1H, NH), 6.54 (s, 1H, NCHNCHC), 5.07 (dd, J1 = 12.5 Hz, J2 = 14.9 Hz, 2H, OCH2Ph), 4.73–4.64 (m, 1H, HNCHCO), 4.40–4.19 (m, 3H, CHCH2OCO), 3.12 (d, J = 5.0Hz, 2H, CH2CHN). 13C-NMR: δ 171.2 (HNCHCOO), 156.2 (COONH), 144.1, 143.9, 142.0, 141.2, 138.4, 135.8, 135.4, 129.7, 128.5, 128.2, 128.1, 127.6, 127.0, 125.4, 125.3, 119.8, 75.6 (CPh), 67.2 (CH2COONH), 66.9 (OCH2Ph), 54.3 (HNCHCOO), 47.1 (CHCH2COO), 29.7 (CH2CHN). MS (ESI): m/z = 710 (100) (M+Η+). Anal. calcd for C47H39N3O4: C, 79.53; H, 5.54; N, 5.92 Found: C, 79.60; H, 5.58; N, 5.85.
Benzyl-3-trityl-L-histidinate (42). Compound 41 (0.15 g, 0.21 mmol) was dissolved in EtOH (1.5 mL) and Et2NH (153 μL, 1.48 mmol) was added. After leaving overnight at rt the solvent was evaporated under reduced pressure. The crude product was redissolved in water, washed by EtOAc (3 × 5mL) and the combined organic layers were dried over Na2SO4 and evaporated in vacuo. After purification by column chromatography and using EtOAc–MeOH (9:1) as eluent the product was isolated as colorless oil in 82% yield (84 mg). Rf 0.37 in EtOAc–MeOH 9:1; [α]D −11.7 (c 1, CHCl3). 1H-NMR (CDCl3): δ 7.32–7.08 (m, 21H, 4 × Ph, NCHN), 6.54 (s, 1H, NCHNCHC), 5.04 (dd, J1 = 12.3 Hz, J2 = 20.8 Hz, 2H, OCH2Ph), 3.90–3.85 (m, 1H, H2NCH), 3.08–2.82 (m, 2H, CH2CHN). 13C-NMR: δ 174.4 (COO), 142.3, 138.6, 137.0, 135.6, 129.7, 128.5, 128.3, 128.2, 128.1, 128.0, 119.5, 75.2 (CPh), 66.6 (CH2Ph), 54.6 (H2NCH), 32.9 (CH2CHN). MS (ESI): m/z = 488 (100) (M+Η+). Anal. calcd for C32H29N3O2: C, 78.82; H, 5.99; N, 8.62 Found: C, 78.68; H, 5.79; N, 8.74.


 , Cs2CO3 in dry DMF; 58%; (xii)
, Cs2CO3 in dry DMF; 58%; (xii)  Cs2CO3 in dry DMF; 55% for 12; 30% for 13; (xiii) H2, 10% Pd/C in MeOH; 99% for 14; 90% for 15.
Cs2CO3 in dry DMF; 55% for 12; 30% for 13; (xiii) H2, 10% Pd/C in MeOH; 99% for 14; 90% for 15.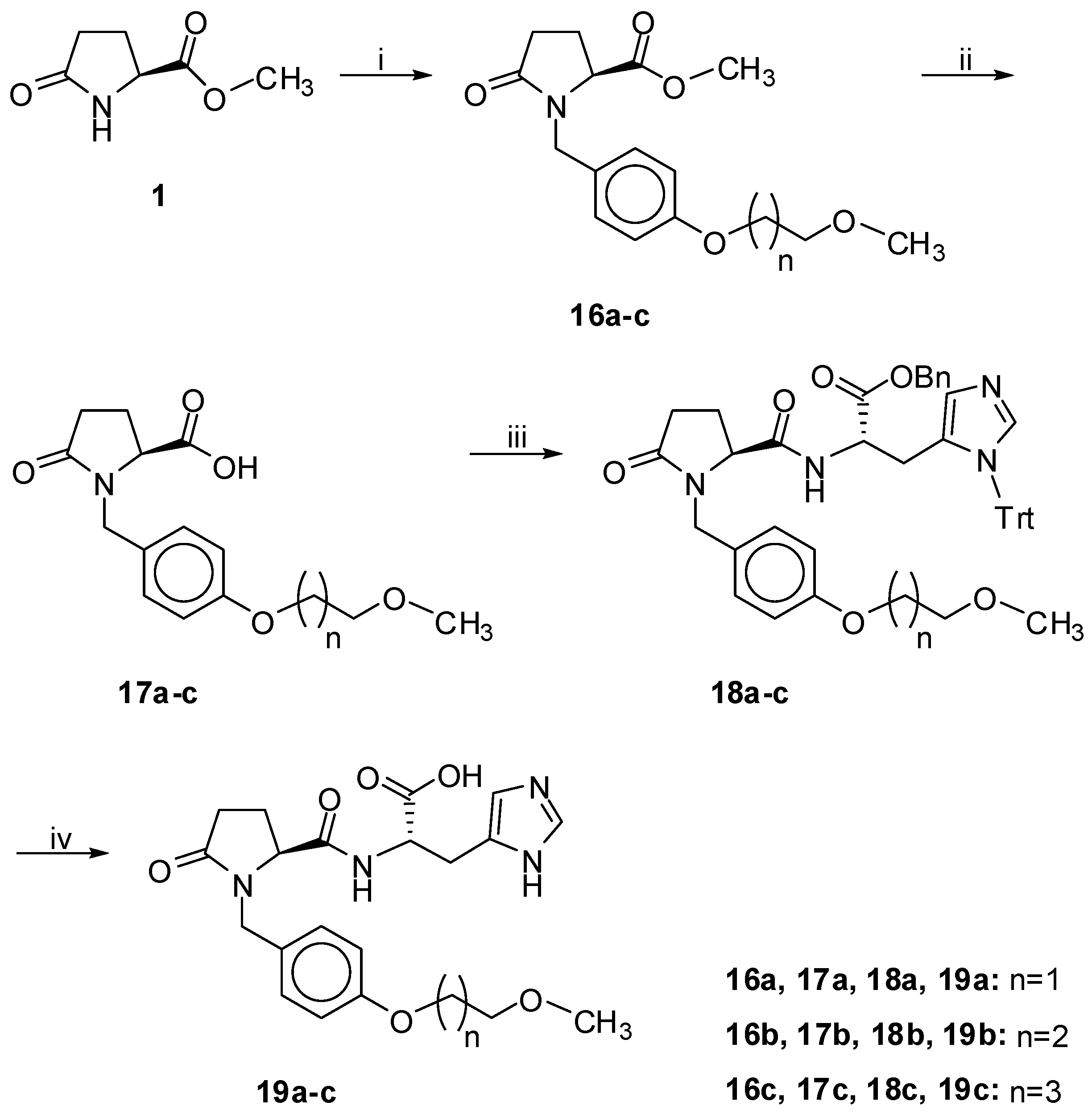

 , NaH in dry THF; 47%; (ii) H2, 10% Pd/C in MeOH; 99%; (iii)
, NaH in dry THF; 47%; (ii) H2, 10% Pd/C in MeOH; 99%; (iii)  , PPh3, DEAD in dry Tol; 60%; (iv) 2N NaOH in MeOH; 94%;(v) 42, Et3N, HOBt, EDC.HCl in CH2Cl2; 48%; (vi) H2, 10% Pd/C in MeOH; 97%; (vii) 4M HCl/dioxane; 90%.
, PPh3, DEAD in dry Tol; 60%; (iv) 2N NaOH in MeOH; 94%;(v) 42, Et3N, HOBt, EDC.HCl in CH2Cl2; 48%; (vi) H2, 10% Pd/C in MeOH; 97%; (vii) 4M HCl/dioxane; 90%.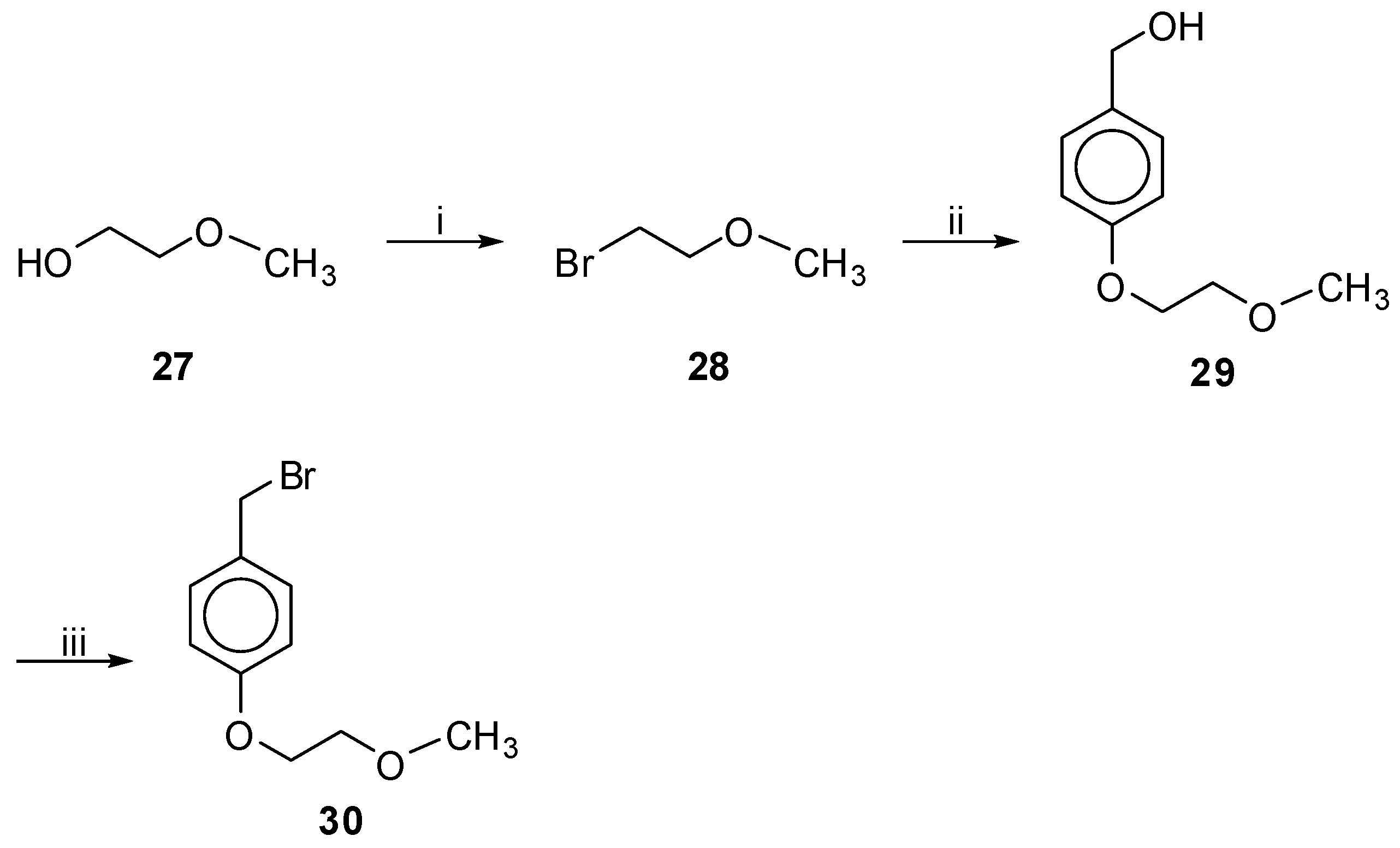
 , K2CO3, 18-Crown-6 in acetone; 76%; (iii) PBr3 in Et2O; 60%.
, K2CO3, 18-Crown-6 in acetone; 76%; (iii) PBr3 in Et2O; 60%.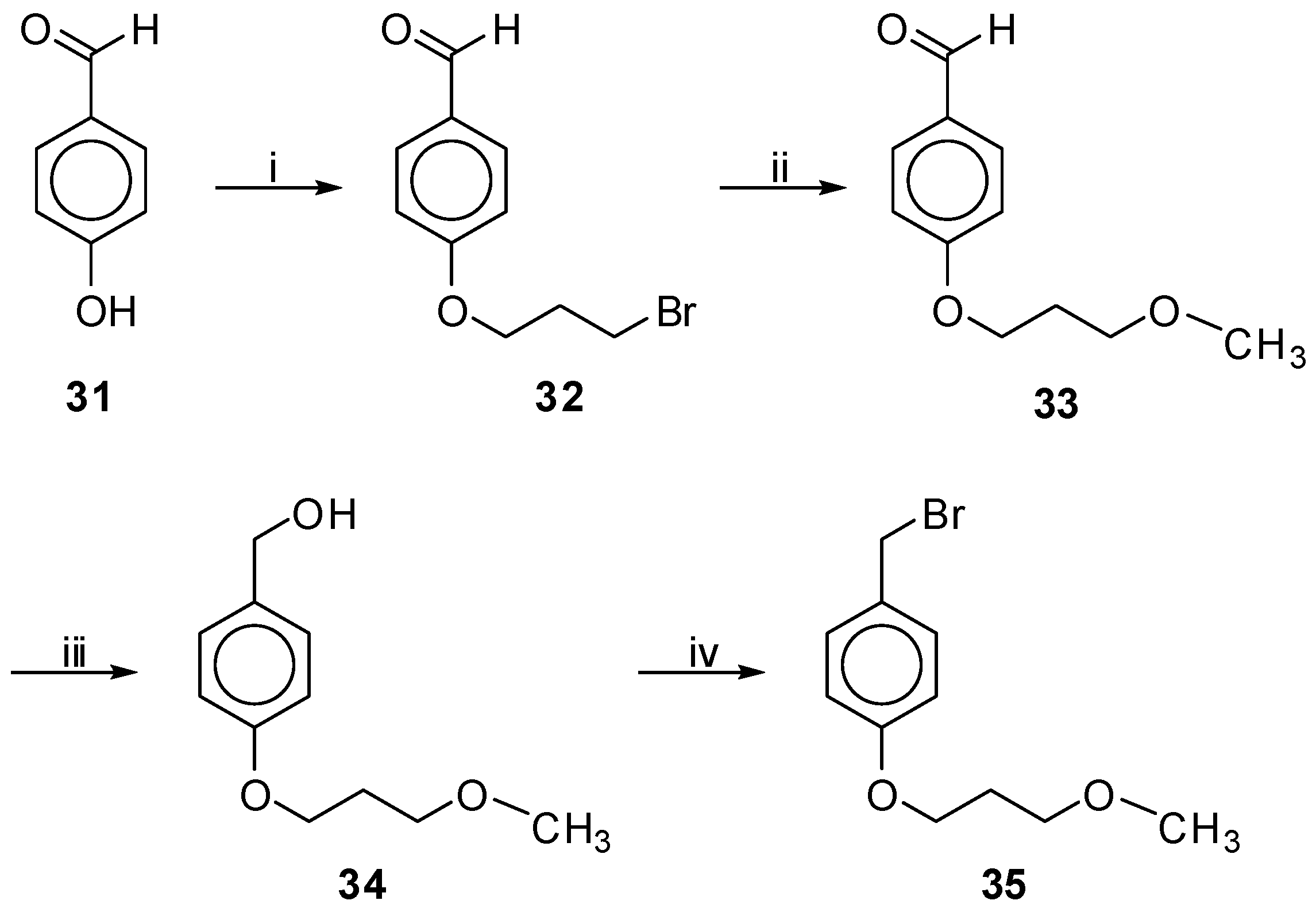
 , K2CO3 in acetone; 40%; (ii) 1N MeONa/MeOH; 38%; (iii) NaBH4 in dry THF; 80%; (iv) PBr3 in Et2O; 70%.
, K2CO3 in acetone; 40%; (ii) 1N MeONa/MeOH; 38%; (iii) NaBH4 in dry THF; 80%; (iv) PBr3 in Et2O; 70%.
 K2CO3, 18-Crown-6 in acetone; 67%; (iii) TMS-Cl, NaBr in CH3CN; 42%.
K2CO3, 18-Crown-6 in acetone; 67%; (iii) TMS-Cl, NaBr in CH3CN; 42%.