An Alternative Synthesis of 3′,4′-Diaminoflavones to Evaluate Their Antioxidant Ability and Cell Apoptosis of Zebrafish Larvae
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
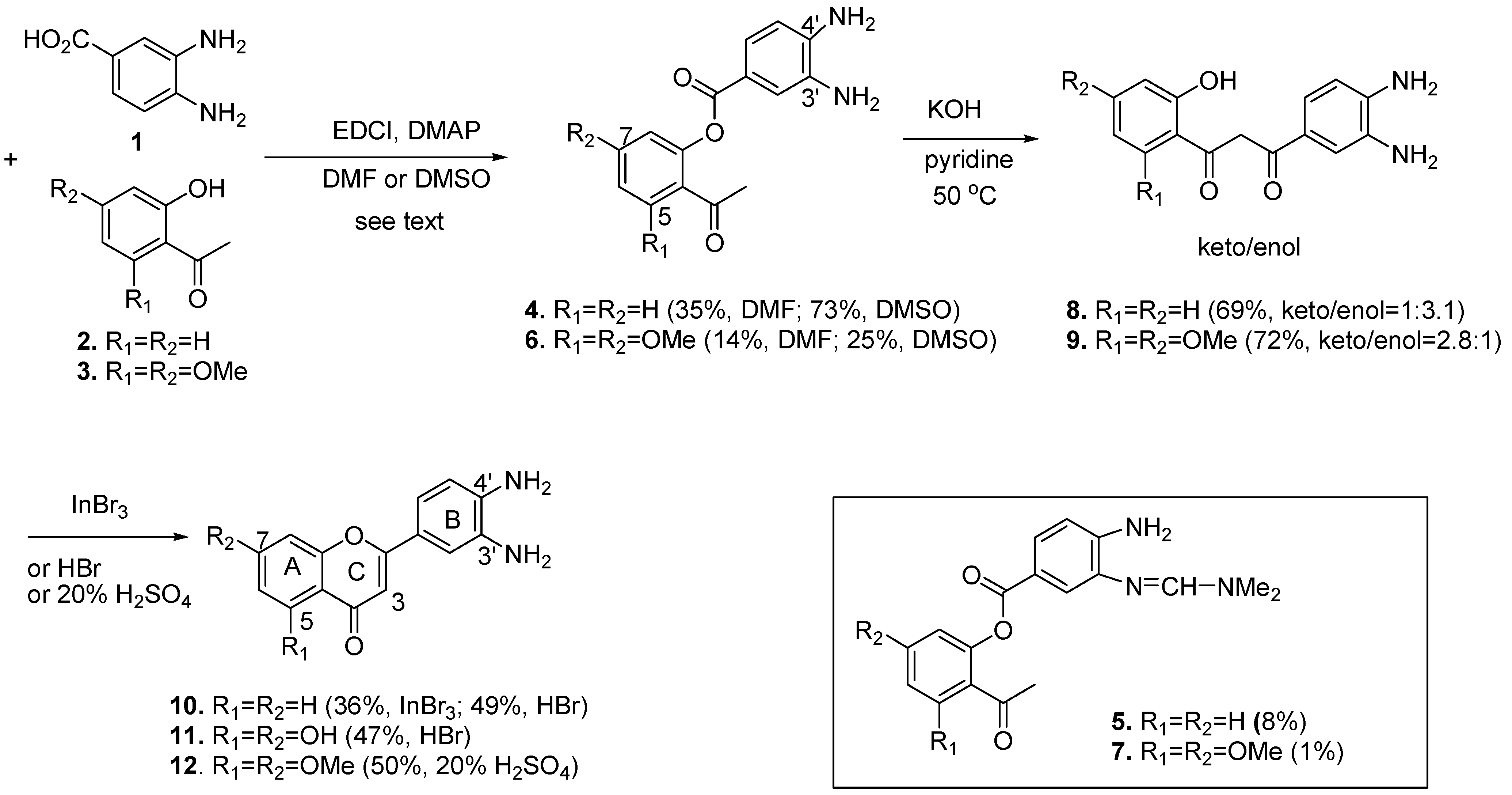
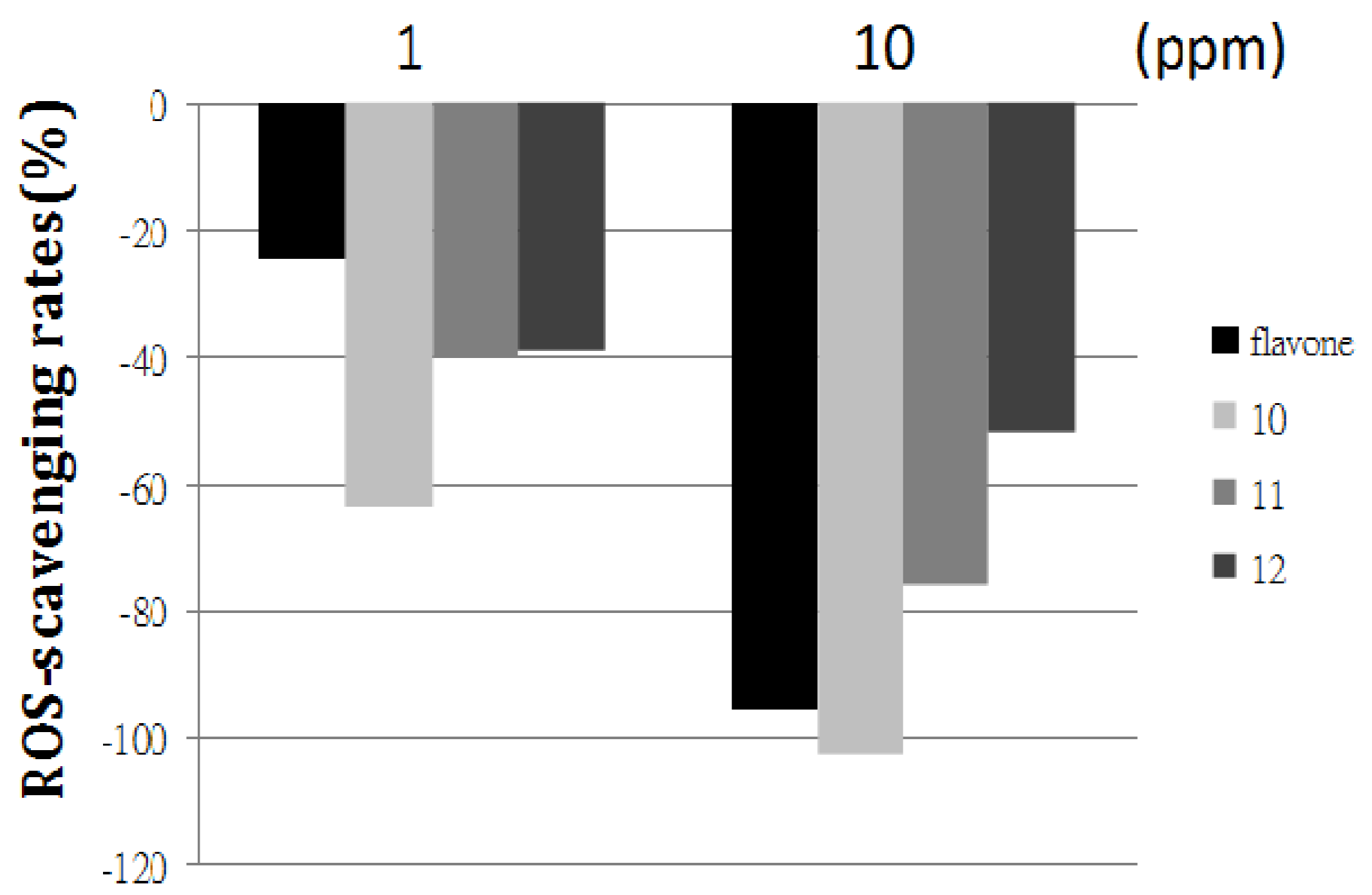
2.2. Comparison of the ROS-Scavenging Ability of Flavone and Aminoflavones 10, 11, and 12
2.3. Flavone and Diaminoflavones Protecting Zebrafish Cells from UV-Induced Apoptosis

3. Experimental
3.1. General
3.2. Synthetic Procedures for the Key Intermediates and Aminoflavones 10, 11, and 12
3.3. Evaluation
3.3.1. Methods for Fish Embryos Maintenance, Chemicals Treatment and Survival Rates Analysis
3.3.2. UVB Exposure, ROS Detection and Data Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Pick, A.; Müller, H.; Mayer, R.; Haenisch, B.; Pajeva, I.K.; Weigt, M.; Bönisch, H.; Müller, C.E.; Wiese, M. Structure-activity relationships of flavonoids as inhibitors of breast cancer resistance protein (BCRP). Bioorg. Med. Chem. 2011, 19, 2090–2102. [Google Scholar] [CrossRef]
- Verma, A.K.; Pratap, R. The biological potential of flavones. Nat. Prod. Rep. 2010, 27, 1571–1593. [Google Scholar] [CrossRef]
- Veitch, N.C.; Grayer, R.J. Flavonoids and their glycosides, including anthocyanins. Nat. Prod. Rep. 2011, 28, 1626–1695. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Lee, E.-R.; Kim, J.-H.; Kang, Y.-J.; Cho, S.-G. The anti-apoptotic and anti-oxidant effect of eriodictyol on UV-induced apoptosis in keratinocytes. Biol. Pharm. Bull. 2007, 30, 32–37. [Google Scholar] [CrossRef]
- Havsteen, B. Flavonoids, a class of natural products of high pharmacological potency. Biochem. Pharmacol. 1983, 32, 1141–1148. [Google Scholar] [CrossRef]
- Formica, J.V.; Regelson, W. Review of the biology of quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Pietta, P.-G. Flavonoids as Antioxidants. J. Nat. Prod 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Oteiza, P.I.; Erlejman, A.G.; Verstraeten, S.V.; Keen, C.L.; Fraga, C.G. Flavonoid-membrane interactions: A protective role of flavonoids at the membrane surface? Clin. Dev. Immunol. 2005, 12, 19–25. [Google Scholar] [CrossRef]
- Ollila, F.; Halling, K.; Vuorela, P.; Vuorela, H.; Slotte, J.P. Characterization of flavonoid-biomembrane interactions. Arch. Biochem. Biophys. 2002, 399, 103–108. [Google Scholar] [CrossRef]
- Han, D.; Williams, E.; Cadenas, E. Mitochondrial respiratory chain-dependent generation of superoxide anion and its release into the intermembrane space. Biochem. J. 2001, 353, 411–416. [Google Scholar] [CrossRef]
- Cos, P.; Ying, L.; Calomme, M.; Hu, J.P.; Cimanga, K.; van Poel, B.; Pieters, L.; Vlietinck, A.J.; Vanden Berghe, D. Structure-activity relationship and classification of flavonoids as inhibitors of xanthine oxidase and superoxide scavengers. J. Nat. Prod. 1998, 61, 71–76. [Google Scholar] [CrossRef]
- Inaba, T.; Tanaka, K.; Takeno, R.; Nagaki, H.; Yoshida, C.; Takano, S. Synthesis and Antiinflammatory Activity of 7-Methanesulfonylamino-6-phenoxychromones. Antiarthritic Effect of the 3-Formylamino Compound (T-614) in Chronic Inflammatory Cisease Models. Chem. Pharm. Bull. 2000, 48, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Yagi, A.; Kabash, A.; Okamura, N.; Haraguchi, H.; Moustafa, S.M.; Khalifa, T.I. Antioxidant, free radical scavenging and anti-inflammatory effects of aloesin derivatives in Aloe vera. Planta Med. 2002, 68, 957–960. [Google Scholar] [CrossRef]
- Dixon, R.A.; Steele, C.L. Flavonoids and isoflavonoids-A gold mine for metabolic engineering. Trends Plant Sci. 1999, 4, 394–400. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.C.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.AM. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar]
- Hernández-Abreu, O.; Castillo-España, P.; León-Rivera, I; Ibarra-Barajas, M.; Villalobos-Molina, R.; González-Christen, J.; Vergara-Galicia, J.; Estrada-Soto, S. Antihypertensive and vasorelaxant effects of tilianin isolated from Agastache mexicana are mediated by NO/cGMP pathway and potassium channel opening. Biochem. Pharmacol. 2009, 78, 54–61. [Google Scholar]
- Torres-Piedra, M.; Ortiz-Andrade, R.; Villalobos-Molina, R.; Singh, N.; Medina-Franco, J.L.; Webster, S.P.; Binnie, M.; Navarrete-Vázquez, G.; Estrada-Soto, S. A comparative study of flavonoid analogues on streptozotocin-nicotinamide induced diabetic rats: Quercetin as a potential antidiabetic agent acting via 11β–Hydroxysteroid dehydrogenase type 1 inhibition. Eur. J. Med. Chem. 2010, 45, 2606–2612. [Google Scholar]
- Kohno, H.; Tanaka, T.; Kawabata, K.; Hirose, Y.; Sugie, S.; Tsuda, H.; Mori, H. Silymarin, a naturally occurring polyphenolic antioxidant flavonoid, inhibits azoxymethane-induced colon carcinogenesis in male F344 rats. Int. J. Cancer 2002, 101, 461–468. [Google Scholar] [CrossRef]
- Cushman, M.; Nagarathnam, D.; Geahlen, R.L. Synthesis and Evaluation of Hydroxylated Flavones and Related Compounds as Potential Inhibitors of the Protein-Tyrosine Kinase p56lck. J. Nat. Prod. 1991, 54, 1345–1352. [Google Scholar] [CrossRef]
- Cushman, M.; Zhu, H.; Geahlen, R.L.; Kraker, A.J. Synthesis and Biochemical Evaluation of a Series of Aminoflavones as Potential Inhibitors of Protein-Tyrosine Kinases p56lck, EGFr, and p60v-src. J. Med. Chem. 1994, 37, 3353–3362. [Google Scholar] [CrossRef]
- Göker, H.; Ayhan, G.; Tuncbilek, M.; Ertan, R.; Leoncini, G.; Garzoglio, R.; Mazzei, M. Synthesis and antiaggregator activity of some new derivatives of 4H-1-benzopyran-4-one. Eur. J. Med. Chem. 1995, 30, 561–567. [Google Scholar]
- Dauzonne, D.; Martinez, L. Synthesis of the 3-aminoflavone-8-acetic acid. Tetrahedron Lett 1995, 36, 1845–1848. [Google Scholar] [CrossRef]
- Akama, T.; Shida, Y.; Sugaya, T.; Ishida, H.; Gomi, K.; Kasai, M. Novel 5-Aminoflavone Derivatives as Specific Antitumor Agents in Breast Cancer. J. Med. Chem. 1996, 39, 3461–3469. [Google Scholar] [CrossRef]
- Dauzonne, D.; Folléas, B.; Martinez, L.; Chabot, G.G. Synthesis and in vitro cytotoxicity of a series of 3-aminoflavones. Eur. J. Med. Chem. 1997, 32, 71–82. [Google Scholar] [CrossRef]
- Tanaka, H.; Stohlmeyer, M.M.; Wandless, T.J.; Taylor, L.P. Synthesis of flavonol derivatives as probes of biological processes. Tetrahedron Lett. 2000, 41, 9735–9739. [Google Scholar] [CrossRef]
- Menichincheri, M.; Ballinari, D.; Bargiotti, A.; Bonomini, L.; Ceccarelli, W.; D’Alessio, R.; Fretta, A.; Moll, J.; Polucci, P.; Soncini, C.; et al. Catecholic Flavonoids Acting as Telomerase Inhibitors. J. Med. Chem. 2004, 47, 6466–6475. [Google Scholar]
- Gao, H.; Kawabata, J. α-Glucosidase inhibition of 6-hydroxyflavones. Part 3: Synthesis and evaluation of 2,3,4-trihydroxybenzoyl-containing flavonoid analogs and 6-aminoflavones as a-glucosidase inhibitors. Bioorg. Med. Chem. 2005, 13, 1661–1671. [Google Scholar] [CrossRef]
- Takechi, A.; Takikawa, H.; Miyake, H.; Sasaki, M. Synthesis of 3-Aminoflavones from 3-Hydroxyflavones via 3-Tosyloxy- or 3-Mesyloxyflavones. Chem. Lett. 2006, 35, 128–129. [Google Scholar] [CrossRef]
- Lecoutey, C.; Fossey, C.; Demuynck, L.; Lefoulon, F.; Fabis, F.; Rault, S. A convenient microwave-assisted 5-amination of flavones. Tetrahedron 2008, 64, 11243–11248. [Google Scholar]
- Lewin, G.; Shridhar, N.B.; Aubert, G.; Thoret, S.; Dubois, J.; Cresteil, T. Synthesis of antiproliferative flavones from calycopterin, major flavonoid of Calycopteris floribunda Lamk. Bioorg. Med. Chem. 2011, 19, 186–196. [Google Scholar] [CrossRef]
- Baker, W. Molecular rearrangement of some o-acyloxyacetophenones and the mechanism of the production of 3-acylchromones. J. Chem. Soc. 1933, 1381–1389. [Google Scholar] [CrossRef]
- Mahal, H.S.; Venkataraman, K. Synthetical experiments in the chromone group. Part XIV. The action of sodamide on 1-acyloxy-2-acetonaphthones. J. Chem. Soc. 1934, 1767–1769. [Google Scholar]
- Barros, A.I.R.N.A.; Silva, A.M.S. Efficient synthesis of nitroflavones by cyclodehydrogenation of 2prime;-hydroxychalcones and by the Baker-Venkataraman method. Monatshefte für Chemie 2006, 137, 1505–1528. [Google Scholar] [CrossRef]
- Lee, Y.R.; Kang, K.Y. Mild and Efficient Method for the Synthesis of Flavones by InCl3-Mediated Cyclization of 1,3-Diketones: Concise Total Synthesis of Lanceolatin B. Lett. Org. Chem. 2007, 4, 440–444. [Google Scholar] [CrossRef]
- Aponte, J.; Castillo, D.; Estevez, Y.; Gonzalez, G.; Arevalo, J.; Hammond, G.B.; Sauvain, M. In vitro and in vivo anti-Leishmania activity of polysubstituted synthetic chalcones. Bioorg. Med. Chem. Lett. 2010, 20, 100–103. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Wen, C.-C.; Lin, C.-Y.; Chou, C.-Y.; Yang, Z.-S.; Wang, Y.-H. UV-Induced fin damage in zebrafish as a system for evaluating the chemopreventive potential of broccoli and cauliflower extracts. Toxicol. Mech. Methods 2011, 21, 63–69. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Wen, C.-C.; Yang, Z.-S.; Cheng, C.-C.; Tsai, J.-N.; Ku, C.-C.; Wu, H.-J.; Chen, Y.-H. Development of a whole-organism model to screen new compounds for sun protection. Mar. Biotechnol. 2009, 11, 419–429. [Google Scholar] [CrossRef]
- Tsai, I.-T.; Yang, Z.-S.; Lin, Z.-Y.; Wen, C.-C.; Cheng, C.-C.; Chen, Y.-H. Flavone is efficient to protect zebrafish fins from UV-induced damages. Drug Chem. Toxicol. 2012, 35, 341–346. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Lee, W.-C.; Liu, C.-F.; Tsai, H.-J. Molecular structure, dynamic expression and promoter analysis of zebrafish (Danio rerio) myf-5 gene. Genesis 2001, 29, 22–35. [Google Scholar] [CrossRef]
- Lee, G.-H.; Chang, M.-Y.; Hsu, C.-H.; Chen, Y.-H. Essential roles of basic helix-loop-helix transcription factors, Capsulin and Musculin, during craniofacial myogenesis of zebrafish. Cell. Mol. Life Sci. 2011, 68, 4065–4078. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Yang, Z.-S.; Wen, C.-C.; Chang, Y.-S.; Wang, B.-C.; Hsiao, C.-A.; Shih, T.-L. Evaluation of the structure-activity relationship of flavonoids as antioxidants and toxicants of zebrafish larvae. Food Chem. 2012, 134, 717–724. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4−12 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shih, T.-L.; Hsiao, C.-A.; Lin, Z.-Y.; Chen, Y.-H. An Alternative Synthesis of 3′,4′-Diaminoflavones to Evaluate Their Antioxidant Ability and Cell Apoptosis of Zebrafish Larvae. Molecules 2012, 17, 8206-8216. https://doi.org/10.3390/molecules17078206
Shih T-L, Hsiao C-A, Lin Z-Y, Chen Y-H. An Alternative Synthesis of 3′,4′-Diaminoflavones to Evaluate Their Antioxidant Ability and Cell Apoptosis of Zebrafish Larvae. Molecules. 2012; 17(7):8206-8216. https://doi.org/10.3390/molecules17078206
Chicago/Turabian StyleShih, Tzenge-Lien, Chih-Ang Hsiao, Zi-Yu Lin, and Yau-Hung Chen. 2012. "An Alternative Synthesis of 3′,4′-Diaminoflavones to Evaluate Their Antioxidant Ability and Cell Apoptosis of Zebrafish Larvae" Molecules 17, no. 7: 8206-8216. https://doi.org/10.3390/molecules17078206
APA StyleShih, T.-L., Hsiao, C.-A., Lin, Z.-Y., & Chen, Y.-H. (2012). An Alternative Synthesis of 3′,4′-Diaminoflavones to Evaluate Their Antioxidant Ability and Cell Apoptosis of Zebrafish Larvae. Molecules, 17(7), 8206-8216. https://doi.org/10.3390/molecules17078206



