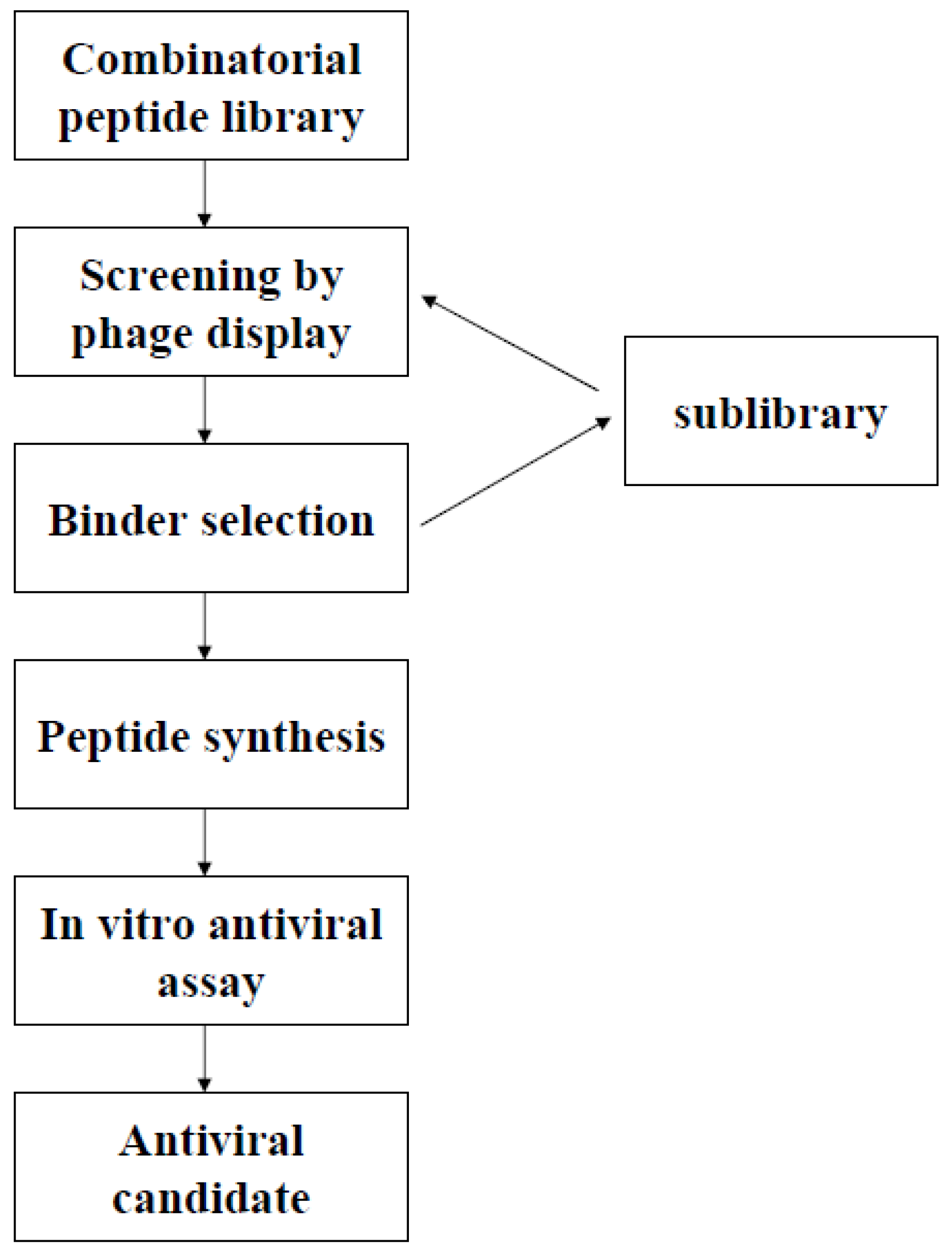
Phage Display of Combinatorial Peptide Libraries: Application to Antiviral Research
Abstract
:1. Introduction
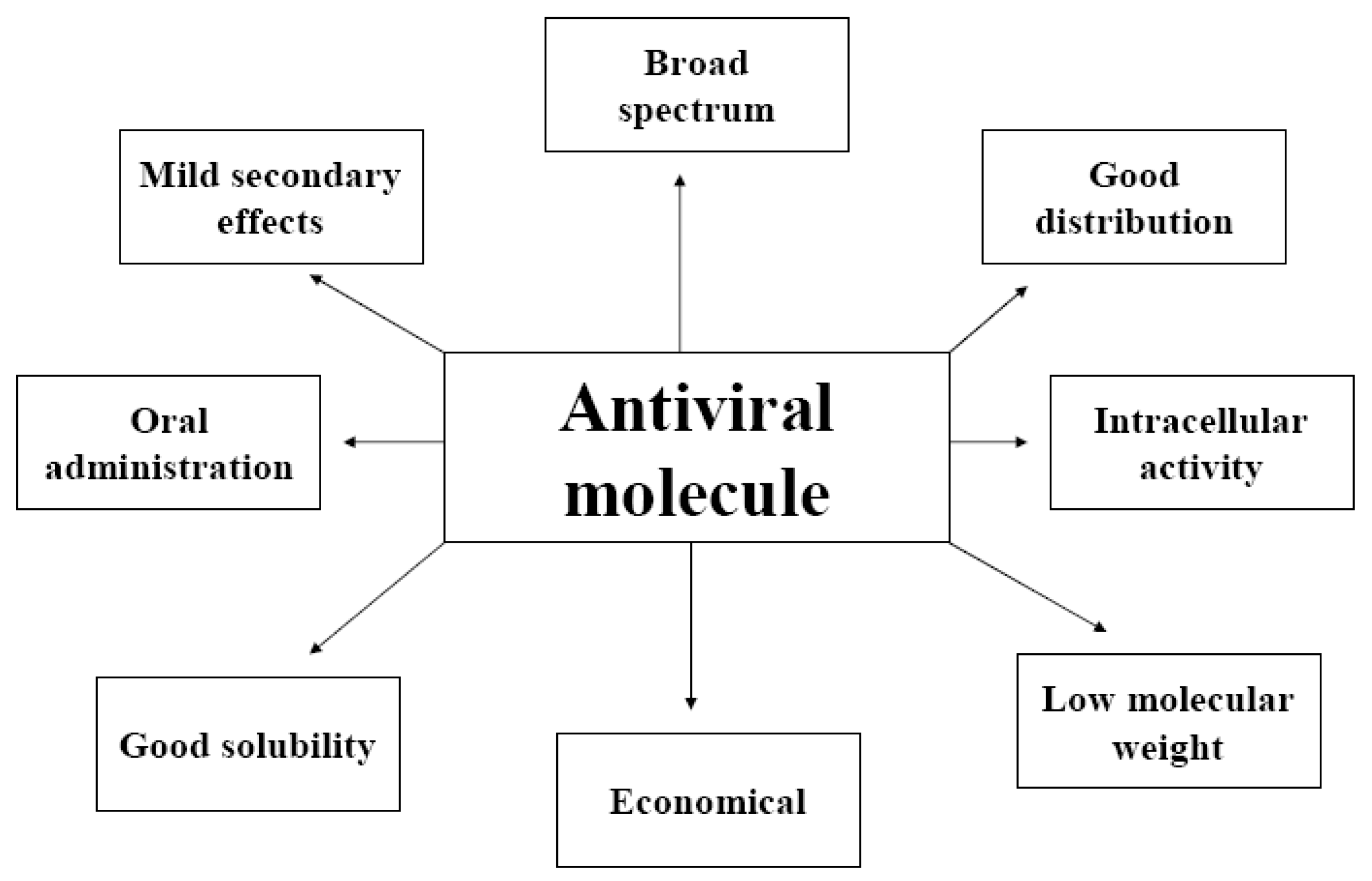
- A cognitive approach based on knowledge about the structure/function of the viral components and the interactions they establish within the complex and, possibly, with cellular partners.
- A random approach involving the high-throughput screening of highly diverse combinatorial molecule libraries generated by natural or organic chemistry (nucleic acids, peptides, proteins). This approach is particularly useful for the rapid isolation of candidate antiviral molecules [9] in cases in which little is known about the target protein, ruling out the use of more rational techniques [7].
3. The Choice of the Target Protein: A Critical Decision
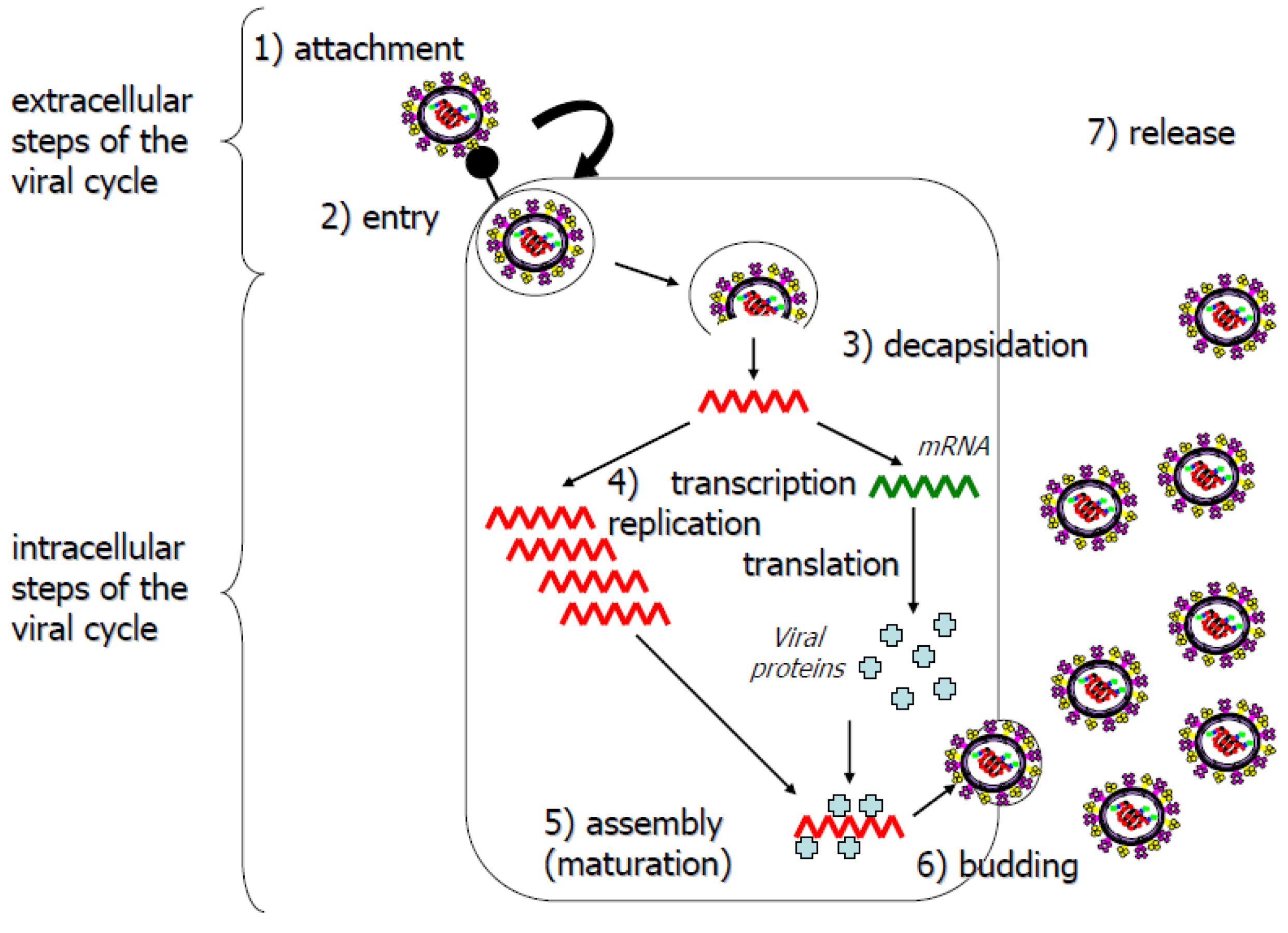
- (1). Targeting of the viral proteins responsible for virus entry into cells (extracellular steps)
- (2). Targeting of the various elements of the viral replication complex and the cellular partners of viral proteins within infected cells (intracellular steps).
| Viral replication step | Virus | Target | Library | Year | Ref. |
|---|---|---|---|---|---|
| Extracellular steps | IBV | whole virus | 12-mer | 2006 | [48] |
| CMV | whole virus | 9-mer | 1999 | [49] | |
| Human rotavirus | whole virus | 15-mer | 2007 | [50] | |
| WSSV | whole virus | 10-mer | 2003 | [51] | |
| NDV | whole virus | Cyclic 7-mer | 2002 | [52,53] | |
| NDV | whole virus | 7-mer | 2005 | [54] | |
| GCHV | whole virus | 9-mer | 2000 | [55] | |
| ANDV | whole virus | 9-mer | 2009 | [34] | |
| AIV (H9N2) | whole virus | 7-mer | 2009 | [35,56] | |
| West-Nile | protein E | Mouse brain cDNA | 2007 | [57] | |
| HCV | protein E2 | 7-mer | 2010 | [59] | |
| HBV | protein domain PreS | 12-mer | 2007 | [58] | |
| HIV-1 | protein gp41 | 10-mer | 1999 | [37] | |
| HIV-1 | protein gp41 | 8-mer / 7-mer | 2007 | [8] | |
| HIV-1 | protein gp41 | 8-mer / 7-mer | 2010 | [13] | |
| Influenza A | protein HA | 15-mer | 2010 | [14] | |
| SNV, HTNV, PHV | integrin alpha/beta | cyclic 9-mer | 2005 | [73] | |
| Intracellular steps | HBV | protein HBcAg | 6-mer | 1995 | [60] |
| HBV | protein HBcAg | C-7-mer-C | 2003 | [20] | |
| HBV | protein HBsAg | C-7-mer-C | 2005 | [62] | |
| HIV | integrase | 7-mer | 2004 | [65] | |
| HIV | protein GAG | 12-mer | 2005 | [66] | |
| HIV | protein Tat | fd pVIII fragments | 2005 | [69] | |
| HIV | protein LcK | 12-mer | 2005 | [71] | |
| HCV | polymerase NS5B | C-7-mer-C | 2003 | [63] | |
| HCV | polymerase NS5B | 7-mer/12-mer/C-9-mer-C | 2008 | [64] | |
| HPV16 | protein E2 | 7-mer / 12-mer | 2003 | [36] | |
| VSV | IFN receptor | 7-mer | 2008 | [75] | |
| HCV | interleukin 10 | 15-mer | 2011 | [76] |
3.3. Targeting cellular proteins
4. Conclusions
Acknowledgments
References and Notes
- Smith, G.P. Filamentous fusion phage: Novel expression vectors that display cloned antigens on the virion surface. Science 1985, 228, 1315–1317. [Google Scholar]
- Souriau, C.; Hua, T.; Lefranc, M.; Weill, M. Présentation à la surface de phages filamenteux: Les multiples applications du phage display. Médecine/Sciences 1998, 14, 300–309. [Google Scholar]
- Decaffmeyer, M.; Thomas, A.; Brasseur, R. Les médicaments peptidiques: Mythe ou réalité ? Biotechnol. Agron. Soc. 2008, 12, 2057. [Google Scholar]
- Brissette, R.; Goldstein, N.I. The use of phage display peptide libraries for basic and translational research. Methods Mol. Biol. 2007, 383, 203–213. [Google Scholar]
- Loregian, A.; Palu, G. Disruption of the interactions between the subunits of herpesvirus DNA polymerases as a novel antiviral strategy. Clin. Microbiol. Infect. 2005, 11, 437–446. [Google Scholar] [CrossRef]
- Loregian, A.; Palu, G. Disruption of protein-protein interactions: Towards new targets for chemotherapy. J. Cell Physiol. 2005, 204, 750–762. [Google Scholar] [CrossRef]
- Vicent, M.J.; Perez-Paya, E.; Orzaez, M. Discovery of inhibitors of protein-protein interactions from combinatorial libraries. Curr. Top. Med. Chem. 2007, 7, 83–95. [Google Scholar]
- Welch, B.D.; VanDemark, A.P.; Heroux, A.; Hill, C.P.; Kay, M.S. Potent D-peptide inhibitors of HIV-1 entry. Proc. Natl. Acad. Sci. USA 2007, 104, 16828–16833. [Google Scholar]
- Vidal, M.; Endoh, H. Prospects for drug screening using the reverse two-hybrid system. Trends Biotechnol. 1999, 17, 374–381. [Google Scholar] [CrossRef]
- Xie, D.; Yao, C.; Wang, L.; Min, W.; Xu, J.; Xiao, J.; Huang, M.; Chen, B.; Liu, B.; Li, X.; Jiang, H. An albumin-conjugated peptide exhibits potent anti-HIV activity and long in vivo half-life. Antimicrob. Agents Chemother. 2010, 54, 191–196. [Google Scholar] [CrossRef]
- Edwards, C.M.; Cohen, M.A.; Bloom, S.R. Peptides as drugs. QJM 1999, 92, 1–4. [Google Scholar] [CrossRef]
- Ladner, R.C.; Sato, A.K.; Gorzelany, J.; de Souza, M. Phage display-derived peptides as therapeutic alternatives to antibodies. Drug Discov. Today 2004, 9, 525–529. [Google Scholar] [CrossRef]
- Welch, B.D.; Francis, J.N.; Redman, J.S.; Paul, S.; Weinstock, M.T.; Reeves, J.D.; Lie, Y.S.; Whitby, F.G.; Eckert, D.M.; Hill, C.P.; Root, M.J.; Kay, M.S. Design of a potent D-peptide HIV-1 entry inhibitor with a strong barrier to resistance. J. Virol. 2010, 84, 11235–11244. [Google Scholar]
- Matsubara, T.; Onishi, A.; Saito, T.; Shimada, A.; Inoue, H.; Taki, T.; Nagata, K.; Okahata, Y.; Sato, T. Sialic acid-mimic peptides as hemagglutinin inhibitors for anti-influenza therapy. J. Med. Chem. 2010, 53, 4441–4449. [Google Scholar]
- Smith, G.P.; Petrenko, V.A. Phage display. Chem. Rev. 1997, 97, 391–410. [Google Scholar]
- Newton, J.R.; Deutscher, S.L. In vivo bacteriophage display for the discovery of novel peptide-based tumor-targeting agents. Methods Mol. Biol. 2009, 504, 275–290. [Google Scholar] [CrossRef]
- Devlin, J.J.; Panganiban, L.C.; Devlin, P.E. Random peptide libraries: A source of specific protein binding molecules. Science 1990, 249, 404–406. [Google Scholar]
- Hall, P.R.; Hjelle, B.; Brown, D.C.; Ye, C.; Bondu-Hawkins, V.; Kilpatrick, K.A.; Larson, R.S. Multivalent presentation of antihantavirus peptides on nanoparticles enhances infection blockade. Antimicrob. Agents Chemother. 2008, 52, 2079–2088. [Google Scholar] [CrossRef]
- Lowman, H.B. Bacteriophage display and discovery of peptide leads for drug development. Annu Rev. Biophys. Biomol. Struct. 1997, 26, 401–424. [Google Scholar] [CrossRef]
- Ho, K.L.; Yusoff, K.; Seow, H.F.; Tan, W.S. Selection of high affinity ligands to hepatitis B core antigen from a phage-displayed cyclic peptide library. J. Med. Virol. 2003, 69, 27–32. [Google Scholar] [CrossRef]
- Ladner, R.C. Constrained peptides as binding entities. Trends Biotechnol. 1995, 13, 426–430. [Google Scholar] [CrossRef]
- Privalov, P.L.; Gill, S.J. Stability of protein structure and hydrophobic interaction. Adv. Protein Chem. 1988, 39, 191–234. [Google Scholar] [CrossRef]
- Sergeeva, A.; Kolonin, M.G.; Molldrem, J.J.; Pasqualini, R.; Arap, W. Display technologies: Application for the discovery of drug and gene delivery agents. Adv. Drug Deliv. Rev. 2006, 58, 1622–1654. [Google Scholar] [CrossRef]
- Pande, J.; Szewczyk, M.M.; Grover, A.K. Phage display: Concept, innovations, applications and future. Biotechnol. Adv. 2010, 28, 849–858. [Google Scholar] [CrossRef]
- Falciani, C.; Lozzi, L.; Pini, A.; Bracci, L. Bioactive peptides from libraries. Chem. Biol. 2005, 12, 417–426. [Google Scholar] [CrossRef]
- Scott, J.K.; Smith, G.P. Searching for peptide ligands with an epitope library. Science 1990, 249, 386–390. [Google Scholar]
- Krumpe, L.R.; Atkinson, A.J.; Smythers, G.W.; Kandel, A.; Schumacher, K.M.; McMahon, J.B.; Makowski, L.; Mori, T. T7 lytic phage-displayed peptide libraries exhibit less sequence bias than M13 filamentous phage-displayed peptide libraries. Proteomics 2006, 6, 4210–4222. [Google Scholar]
- Castagnoli, L.; Zucconi, A.; Quondam, M.; Rossi, M.; Vaccaro, P.; Panni, S.; Paoluzi, S.; Santonico, E.; Dente, L.; Cesareni, G. Alternative bacteriophage display systems. Comb. Chem. High Throughput Screen. 2001, 4, 121–133. [Google Scholar]
- Burritt, J.B.; Bond, C.W.; Doss, K.W.; Jesaitis, A.J. Filamentous phage display of oligopeptide libraries. Anal. Biochem. 1996, 238, 1–13. [Google Scholar] [CrossRef]
- Levin, A.M.; Weiss, G.A. Optimizing the affinity and specificity of proteins with molecular display. Mol. Biosyst. 2006, 2, 49–57. [Google Scholar] [CrossRef]
- Sidhu, S.S. Engineering M13 for phage display. Biomol. Eng. 2001, 18, 57–63. [Google Scholar] [CrossRef]
- O'Connell, D.; Becerril, B.; Roy-Burman, A.; Daws, M.; Marks, J.D. Phage versus phagemid libraries for generation of human monoclonal antibodies. J. Mol. Biol. 2002, 321, 49–56. [Google Scholar] [CrossRef]
- Jestin, J.L. Functional cloning by phage display. Biochimie 2008, 90, 1273–1278. [Google Scholar] [CrossRef]
- Hall, P.R.; Hjelle, B.; Njus, H.; Ye, C.; Bondu-Hawkins, V.; Brown, D.C.; Kilpatrick, K.A.; Larson, R.S. Phage display selection of cyclic peptides that inhibit Andes virus infection. J. Virol. 2009, 83, 8965–8969. [Google Scholar]
- Rajik, M.; Jahanshiri, F.; Omar, A.R.; Ideris, A.; Hassan, S.S.; Yusoff, K. Identification and characterisation of a novel anti-viral peptide against avian influenza virus H9N2. Virol. J. 2009, 6, 74. [Google Scholar] [CrossRef]
- Fujii, T.; Austin, D.; Guo, D.; Srimatkandada, S.; Wang, T.; Kubushiro, K.; Masumoto, N.; Tsukazaki, K.; Nozawa, S.; Deisseroth, A.B. Peptides inhibitory for the transcriptional regulatory function of human papillomavirus E2. Clin. Cancer Res. 2003, 9, 5423–5428. [Google Scholar]
- Eckert, D.M.; Malashkevich, V.N.; Hong, L.H.; Carr, P.A.; Kim, P.S. Inhibiting HIV-1 entry: Discovery of D-peptide inhibitors that target the gp41 coiled-coil pocket. Cell 1999, 99, 103–115. [Google Scholar] [CrossRef]
- Heyd, B.; Pecorari, F.; Collinet, B.; Adjadj, E.; Desmadril, M.; Minard, P. In vitro evolution of the binding specificity of neocarzinostatin, an enediyne-binding chromoprotein. Biochemistry 2003, 42, 5674–5683. [Google Scholar] [CrossRef]
- Knobel, D.L.; Cleaveland, S.; Coleman, P.G.; Fevre, E.M.; Meltzer, M.I.; Miranda, M.E.; Shaw, A.; Zinsstag, J.; Meslin, F.X. Re-evaluating the burden of rabies in Africa and Asia. Bull World Health Organ. 2005, 83, 360–368. [Google Scholar]
- Massé, N.; Selisko, B.; Malet, H.; Peyrane, F.; Debarnot, C.; Decroly, E.; Benarroch, D.; Egloff, M.; Guillemot, J.; Alvarez, K.; Canard, B. Le virus de la dengue: Cibles virales et antiviraux. Virologie 2007, 11, 121–133. [Google Scholar]
- Rodi, D.J.; Soares, A.S.; Makowski, L. Quantitative assessment of peptide sequence diversity in M13 combinatorial peptide phage display libraries. J. Mol. Biol. 2002, 322, 1039–1052. [Google Scholar]
- Malone, J.; Sullivan, M.A. Analysis of antibody selection by phage display utilizing anti-phenobarbital antibodies. J. Mol. Recognit. 1996, 9, 738–745. [Google Scholar] [CrossRef]
- Schier, R.; Marks, J.D. Efficient in vitro affinity maturation of phage antibodies using BIAcore guided selections. Hum. Antibodies Hybridomas 1996, 7, 97–105. [Google Scholar]
- Adey, N.B.; Mataragnon, A.H.; Rider, J.E.; Carter, J.M.; Kay, B.K. Characterization of phage that bind plastic from phage-displayed random peptide libraries. Gene 1995, 156, 27–31. [Google Scholar] [CrossRef]
- Leyssen, P.; De Clercq, E.; Neyts, J. Molecular strategies to inhibit the replication of RNA viruses. Antiviral Res. 2008, 78, 9–25. [Google Scholar] [CrossRef]
- Aman, M.J.; Kinch, M.S.; Warfield, K.; Warren, T.; Yunus, A.; Enterlein, S.; Stavale, E.; Wang, P.; Chang, S.; Tang, Q.; Porter, K.; Goldblatt, M.; Bavari, S. Development of a broad-spectrum antiviral with activity against Ebola virus. Antiviral Res. 2009, 83, 245–251. [Google Scholar]
- Bray, M. Highly pathogenic RNA viral infections: Challenges for antiviral research. Antiviral Res. 2008, 78, 1–8. [Google Scholar] [CrossRef]
- Peng, B.; Chen, H.; Tan, Y.; Jin, M.; Guo, A. Identification of one peptide which inhibited infectivity of avian infectious bronchitis virus in vitro. Sci. Chin. C Life Sci. 2006, 49, 158–163. [Google Scholar] [CrossRef]
- Gough, K.C.; Cockburn, W.; Whitelam, G.C. Selection of phage-display peptides that bind to cucumber mosaic virus coat protein. J. Virol. Methods 1999, 79, 169–180. [Google Scholar] [CrossRef]
- Yao, N.; Yao, L.G.; Zhang, X.M.; Guo, T.L.; Kan, Y.C. Screening for peptides of anti-rotavirus by phage-displayed technique. Sheng Wu Gong Cheng Xue Bao 2007, 23, 403–408. [Google Scholar]
- Yi, G.; Qian, J.; Wang, Z.; Qi, Y. A phage-displayed peptide can inhibit infection by white spot syndrome virus of shrimp. J. Gen. Virol. 2003, 84, 2545–2553. [Google Scholar] [CrossRef]
- Ramanujam, P.; Tan, W.S.; Nathan, S.; Yusoff, K. Novel peptides that inhibit the propagation of Newcastle disease virus. Arch. Virol. 2002, 147, 981–993. [Google Scholar] [CrossRef]
- Chia, S.L.; Tan, W.S.; Shaari, K.; Abdul Rahman, N.; Yusoff, K.; Satyanarayanajois, S.D. Structural analysis of peptides that interact with Newcastle disease virus. Peptides 2006, 27, 1217–1225. [Google Scholar] [CrossRef]
- Ozawa, M.; Ohashi, K.; Onuma, M. Identification and characterization of peptides binding to newcastle disease virus by phage display. J. Vet. Med. Sci. 2005, 67, 1237–1241. [Google Scholar]
- Wang, B.; Ke, L.H.; Jiang, H.; Li, C.Z.; Tien, P. Selection of a specific peptide from a nona-peptide library for in vitro inhibition of grass carp hemorrhage virus replication. Virus Res. 2000, 67, 119–125. [Google Scholar] [CrossRef]
- Rajik, M.; Omar, A.R.; Ideris, A.; Hassan, S.S.; Yusoff, K. A novel peptide inhibits the influenza virus replication by preventing the viral attachment to the host cells. Int. J. Biol. Sci. 2009, 5, 543–548. [Google Scholar]
- Bai, F.; Town, T.; Pradhan, D.; Cox, J.; Ashish; Ledizet, M.; Anderson, J.F.; Flavell, R.A.; Krueger, J.K.; Koski, R.A.; Fikrig, E. Antiviral peptides targeting the west nile virus envelope protein. J. Virol. 2007, 81, 2047–2055. [Google Scholar]
- Deng, Q.; Zhai, J.W.; Michel, M.L.; Zhang, J.; Qin, J.; Kong, Y.Y.; Zhang, X.X.; Budkowska, A.; Tiollais, P.; Wang, Y.; Xie, Y.H. Identification and characterization of peptides that interact with hepatitis B virus via the putative receptor binding site. J. Virol. 2007, 81, 4244–4254. [Google Scholar]
- Hong, H.W.; Lee, S.W.; Myung, H. Selection of peptides binding to HCV e2 and inhibiting viral infectivity. J. Microbiol. Biotechnol. 2010, 20, 1769–1771. [Google Scholar]
- Dyson, M.R.; Murray, K. Selection of peptide inhibitors of interactions involved in complex protein assemblies: Association of the core and surface antigens of hepatitis B virus. Proc. Natl. Acad. Sci. USA 1995, 92, 2194–2198. [Google Scholar] [CrossRef]
- Bottcher, B.; Tsuji, N.; Takahashi, H.; Dyson, M.R.; Zhao, S.; Crowther, R.A.; Murray, K. Peptides that block hepatitis B virus assembly: Analysis by cryomicroscopy, mutagenesis and transfection. EMBO J. 1998, 17, 6839–6845. [Google Scholar] [CrossRef]
- Tan, W.S.; Tan, G.H.; Yusoff, K.; Seow, H.F. A phage-displayed cyclic peptide that interacts tightly with the immunodominant region of hepatitis B surface antigen. J. Clin. Virol. 2005, 34, 35–41. [Google Scholar] [CrossRef]
- Amin, A.; Zaccardi, J.; Mullen, S.; Olland, S.; Orlowski, M.; Feld, B.; Labonte, P.; Mak, P. Identification of constrained peptides that bind to and preferentially inhibit the activity of the hepatitis C viral RNA-dependent RNA polymerase. Virology 2003, 313, 158–169. [Google Scholar] [CrossRef]
- Kim, M.S.; Park, C.; Lee, J.H.; Myung, H. Selection and target-site mapping of peptides inhibiting HCV NS5B polymerase using phage display. J. Microbiol. Biotechnol. 2008, 18, 328–333. [Google Scholar]
- Desjobert, C.; de Soultrait, V.R.; Faure, A.; Parissi, V.; Litvak, S.; Tarrago-Litvak, L.; Fournier, M. Identification by phage display selection of a short peptide able to inhibit only the strand transfer reaction catalyzed by human immunodeficiency virus type 1 integrase. Biochemistry 2004, 43, 13097–13105. [Google Scholar]
- Sticht, J.; Humbert, M.; Findlow, S.; Bodem, J.; Muller, B.; Dietrich, U.; Werner, J.; Krausslich, H.G. A peptide inhibitor of HIV-1 assembly in vitro. Nat. Struct. Mol. Biol. 2005, 12, 671–677. [Google Scholar]
- Dietz, J.; Koch, J.; Kaur, A.; Raja, C.; Stein, S.; Grez, M.; Pustowka, A.; Mensch, S.; Ferner, J.; Moller, L.; Bannert, N.; Tampe, R.; Divita, G.; Mely, Y.; Schwalbe, H.; Dietrich, U. Inhibition of HIV-1 by a peptide ligand of the genomic RNA packaging signal Psi. Chem. Med. Chem. 2008, 3, 749–755. [Google Scholar]
- Enshell-Seijffers, D.; Smelyanski, L.; Gershoni, J.M. The rational design of a 'type 88' genetically stable peptide display vector in the filamentous bacteriophage fd. Nucl. Acids Res. 2001, 29, E50. [Google Scholar] [CrossRef]
- Krichevsky, A.; Rusnati, M.; Bugatti, A.; Waigmann, E.; Shohat, S.; Loyter, A. The fd phage and a peptide derived from its p8 coat protein interact with the HIV-1 Tat-NLS and inhibit its biological functions. Antiviral Res. 2005, 66, 67–78. [Google Scholar] [CrossRef]
- Moran, G.J.; Talan, D.A.; Abrahamian, F.M. Biological terrorism. Infect Dis. Clin. North Am. 2008, 22, 145–187. [Google Scholar] [CrossRef]
- Tran, T.; Hoffmann, S.; Wiesehan, K.; Jonas, E.; Luge, C.; Aladag, A.; Willbold, D. Insights into human Lck SH3 domain binding specificity: Different binding modes of artificial and native ligands. Biochemistry 2005, 44, 15042–15052. [Google Scholar] [CrossRef]
- Stangler, T.; Tran, T.; Hoffmann, S.; Schmidt, H.; Jonas, E.; Willbold, D. Competitive displacement of full-length HIV-1 Nef from the Hck SH3 domain by a high-affinity artificial peptide. Biol. Chem. 2007, 388, 611–615. [Google Scholar]
- Larson, R.S.; Brown, D.C.; Ye, C.; Hjelle, B. Peptide antagonists that inhibit Sin Nombre virus and hantaan virus entry through the beta3-integrin receptor. J. Virol. 2005, 79, 7319–7326. [Google Scholar] [CrossRef]
- Tzianabos, A.O. Polysaccharide immunomodulators as therapeutic agents: Structural aspects and biologic function. Clin. Microbiol. Rev. 2000, 13, 523–533. [Google Scholar] [CrossRef]
- Zhang, Q.; Bai, G.; Chen, J.Q.; Tian, W.; Cao, Y.; Pan, P.W.; Wang, C. Identification of antiviral mimetic peptides with interferon alpha-2b-like activity from a random peptide library using a novel functional biopanning method. Acta Pharmacol. Sin. 2008, 29, 634–640. [Google Scholar] [CrossRef]
- Diaz-Valdes, N.; Manterola, L.; Belsue, V.; Riezu-Boj, J.I.; Larrea, E.; Echeverria, I.; Llopiz, D.; Lopez-Sagaseta, J.; Lerat, H.; Pawlotsky, J.M.; Prieto, J.; Lasarte, J.J.; Borras-Cuesta, F.; Sarobe, P. Improved dendritic cell-based immunization against hepatitis C virus using peptide inhibitors of interleukin 10. Hepatology 2011, 53, 23–31. [Google Scholar]
- Padwad, Y.S.; Mishra, K.P.; Jain, M.; Chanda, S.; Ganju, L. Dengue virus infection activates cellular chaperone Hsp70 in THP-1 cells: Downregulation of Hsp70 by siRNA revealed decreased viral replication. Viral. Immunol. 2010, 23, 557–565. [Google Scholar] [CrossRef]
- Wunner, W.H.; Pallatroni, C.; Curtis, P.J. Selection of genetic inhibitors of rabies virus. Arch. Virol. 2004, 149, 1653–1662. [Google Scholar]
- Pelet, T.; Miazza, V.; Mottet, G.; Roux, L. High throughput screening assay for negative single stranded RNA virus polymerase inhibitors. J. Virol. Methods 2005, 128, 29–36. [Google Scholar]
- Strobel, H.; Ladant, D.; Jestin, J.L. In vitro selection for enzymatic activity: A model study using adenylate cyclase. J. Mol. Biol. 2003, 332, 1–7. [Google Scholar]
- Soumillion, P.; Jespers, L.; Bouchet, M.; Marchand-Brynaert, J.; Winter, G.; Fastrez, J. Selection of beta-lactamase on filamentous bacteriophage by catalytic activity. J. Mol. Biol. 1994, 237, 415–422. [Google Scholar] [CrossRef]
- Pedersen, H.; Holder, S.; Sutherlin, D.P.; Schwitter, U.; King, D.S.; Schultz, P.G. A method for directed evolution and functional cloning of enzymes. Proc. Natl. Acad. Sci. USA 1998, 95, 10523–10528. [Google Scholar]
- Yamamoto, K.; Taniai, M.; Torigoe, K.; Yamamoto, S.; Arai, N.; Suemoto, Y.; Yoshida, K.; Okura, T.; Mori, T.; Fujioka, N.; Tanimoto, T.; Miyata, M.; Ariyasu, H.; Ushio, C.; Fujii, M.; Ariyasu, T.; Ikeda, M.; Ohta, T.; Kurimoto, M.; Fukuda, S. Creation of interferon-alpha8 mutants with amino acid substitutions against interferon-alpha receptor-2 binding sites using phage display system and evaluation of their biologic properties. J. Interferon Cytokine Res. 2009, 29, 161–170. [Google Scholar]
- Kalie, E.; Jaitin, D.A.; Abramovich, R.; Schreiber, G. An interferon alpha2 mutant optimized by phage display for IFNAR1 binding confers specifically enhanced antitumor activities. J. Biol. Chem. 2007, 282, 11602–11611. [Google Scholar] [CrossRef]
- Wu, H.; Pfarr, D.S.; Tang, Y.; An, L.L.; Patel, N.K.; Watkins, J.D.; Huse, W.D.; Kiener, P.A.; Young, J.F. Ultra-potent antibodies against respiratory syncytial virus: Effects of binding kinetics and binding valence on viral neutralization. J. Mol. Biol. 2005, 350, 126–144. [Google Scholar]
- Sample Availability: Not available.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Castel, G.; Chtéoui, M.; Heyd, B.; Tordo, N. Phage Display of Combinatorial Peptide Libraries: Application to Antiviral Research. Molecules 2011, 16, 3499-3518. https://doi.org/10.3390/molecules16053499
Castel G, Chtéoui M, Heyd B, Tordo N. Phage Display of Combinatorial Peptide Libraries: Application to Antiviral Research. Molecules. 2011; 16(5):3499-3518. https://doi.org/10.3390/molecules16053499
Chicago/Turabian StyleCastel, Guillaume, Mohamed Chtéoui, Bernadette Heyd, and Noël Tordo. 2011. "Phage Display of Combinatorial Peptide Libraries: Application to Antiviral Research" Molecules 16, no. 5: 3499-3518. https://doi.org/10.3390/molecules16053499
APA StyleCastel, G., Chtéoui, M., Heyd, B., & Tordo, N. (2011). Phage Display of Combinatorial Peptide Libraries: Application to Antiviral Research. Molecules, 16(5), 3499-3518. https://doi.org/10.3390/molecules16053499