Anaerobic Digestion for Producing Renewable Energy—The Evolution of This Technology in a New Uncertain Scenario
Abstract
1. Introduction
2. The Effect of Substrate Composition and Digestion Performance
3. Co-Digestion to Increase Reactor Productivity
| Organic Substrate | Specific Production Potential (M3 CH4/Kg VS) 1 | Reference |
|---|---|---|
| Livestock manure | ||
| Pig manure | 0.30–0.50 | [96,97,98,99,100,101] |
| Poultry manure | 0.03–0.11 | [90,96,100,101] |
| Cattle manure | 0.11–0.54 | [102,103] |
| Organic industrial waste | ||
| Slaughterhouse waste | 0.20–0.80 | [104,105] |
| Brewery waste | 0.3–0.51 | [106,107] |
| Sewage sludge (SS) and co-substrate | ||
| SS | 0.22–0.45 | [35,108,109] |
| SS + grease | 0.4–0.8 | [108,110] |
| SS + glycerol | 0.2–0.4 | [111,112] |
| SS + food wastes | 0.4–0.6 | [86,113] |
| Energy crops | ||
| Corn stover | 0.30–0.40 | [97,114] |
| Sunflower | 0.20–0.40 | [115,116] |
| Rapeseed | 0.25 | [97] |
| Wheat straw (steam explosion pre-treatment) | 0.25–0.35 | [117,118] |
| Rice straw | 0.26 | [119] |
| Grass: Napier grass, Canary grass, King grass | 0.15–0.60 | [120,121,122] |
| Microalgae and cyanobacteria biomass | ||
| Microalgae Chlorella sp. | 0.23–026 | [99,123] |
| Microalgae Nannochloropsis oculata | 0.3–0.35 | [124] |
| Manure + Arthrospira platensis | 0.48 | [125] |
4. Technical and Economic Feasibility of Anaerobic Digestion Plants
| Substrates | Selling Prices | Digester | Costs (EUR) Millions | Reference |
|---|---|---|---|---|
| Dairy cow farm Input: 29,200 t/year Plant treating manure + sheep dung | Electricity: EUR 0.10/kWh Liquid fertilizer: EUR 120/t | 2713 m3 | 0.77 | [146] |
| 2400 beef cattle and glycerine + biomass as co-substrate | Credit claims. Electricity: EUR 0.012/kWh Solid by-product: EUR 28.8/t Liquid by-product: EUR 2.15/t | 2 digesters 3670 m3 each | 2.55 | [147] |
| 8000 t/year Dairy manure, corn stalk, tomato residues | Electricity: EUR 0.13/kWh Heat: EUR 0.0326/kWh Bio-methane: 44/m3 | 1000 m3, wet digestion 250 m3, solid digestion | 0.4–0.5 | [148] |
| Two-phase olive oil mill pomace and pig slurry 7500 t/year and 2450 m3/year of wastewaters + pig slurry 9000 t/year | Electricity: EUR 0.13/kWh Heat: EUR 0.036/kWh Waste management savings: EUR 5/t Compost: EUR 70/t Olive stones: EUR 80/t | Plant size to digest 8750 t/year. | 0.6–1.1 | [149] |
| Small-scale digestion plants Herd size: 50–250 adult caws) | Electricity: EUR 0.158/kWh Heat: EUR 0.295/kWh District heating selling thermal energy price: EUR 0.03/kWh | Based on installed CHP engine power (kWe): 17–55 | 0.29–0.52 | [150] |
5. Integrating Anaerobic Digestion into a Green Energy Producing System
| Substrates | Conversion Processes | Products | Reference |
|---|---|---|---|
| Jerusalem artichoke (Helianthus tuberosus L.) | Extraction | Sugars, succinic acid, Rubisco fraction, proteins | [172] |
| Invasive brown algae (Sargassum muticum) | Drying, extraction, fractionation | Fucoxanthin and hydrolysis liqueur | [184] |
| Food waste | Black soldier fly (BSF) (Hermetia illucen) | High-value insect products: protein, lipids, chitin and frass | [185] |
| Sugar beet | Ethanol fermentation + continuous fermentation with Bacillus coagulans | Ethanol and lactic acid | [186] |
| Grape wine waste | Extraction + hydrolysis + fermentation | Lactic acid, tartaric acid, protein-rich fungal biomass, tannins, polyphenols | [187] |
| Sugarcane distillery (bagasse) and straw | Fermentation + gasification + Fischer–Tropsch synthesis | Ethanol, diesel, jet fuel, gasoline, electricity | [188] |
| Citrus wastes | Pectin extraction anaerobic digestion | Mucic acid production and biogas | [189] |
| Agricultural and livestock wastes | Fermentation + anaerobic digestion | Biogas, electricity, ethanol, butanol, acetate, propionic, lactic acid | [4] |
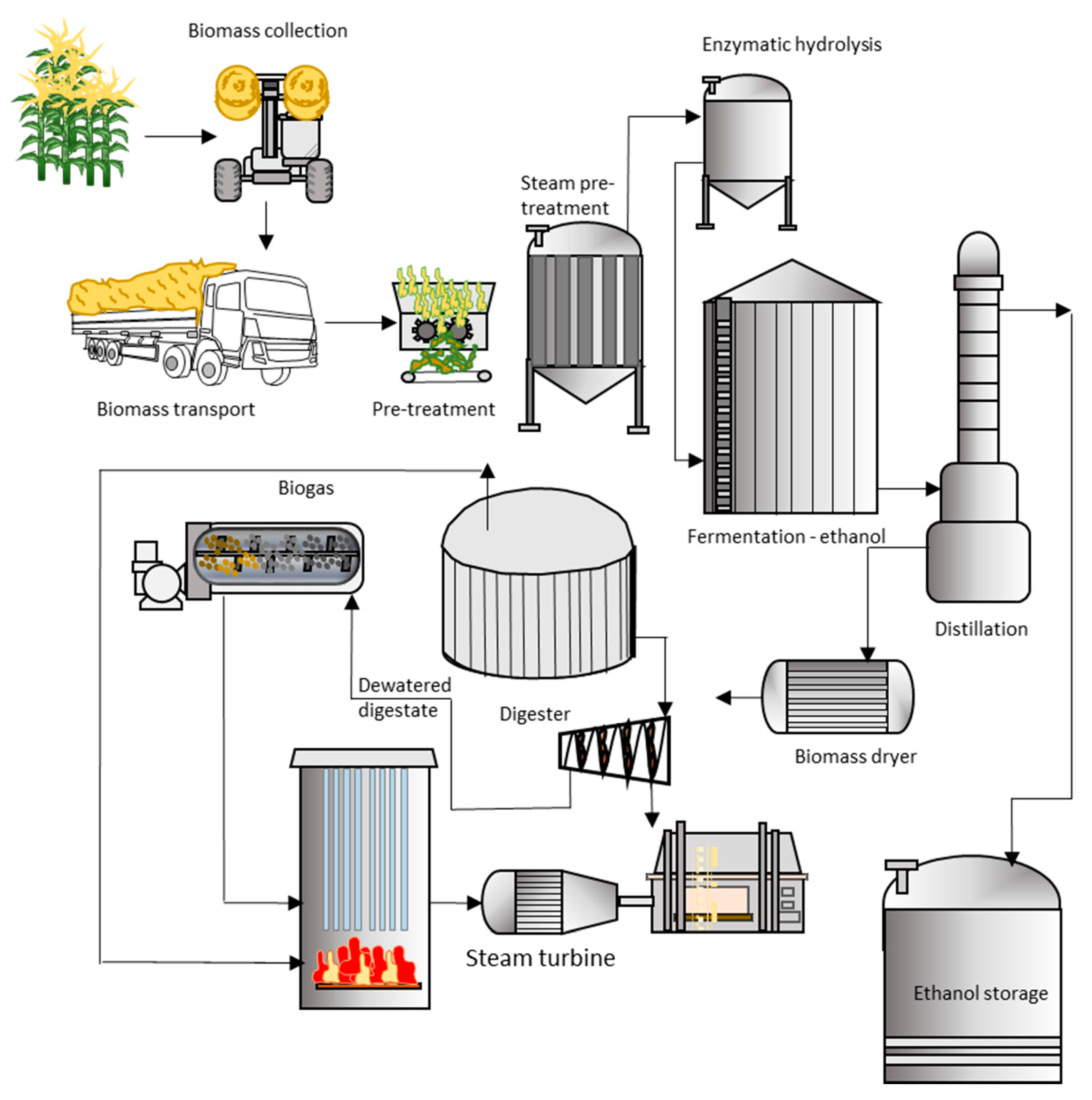
| Switchgrass (Panicum virgatum) | Ethanol fermentation + anaerobic digestion + extraction + thermal process | Ethanol, biogas, electricity, phenol | [190] |
| Sugarcane bagasse (Saccharum officinarum) | Enzymatic hydrolysis, fermentation + anaerobic digestion + combustion | Ethanol, biogas, heat | [191] |
| Meat processing wastes | Hydrolysis − esterification + anaerobic digestion + hydrothermal liquefaction | Biodiesel, biogas, biochar, bio-oil | [192] |
| Wheat straw Animal bedding | Pre-treatment + hydrolysis + fermentation + anaerobic digestion | Ethanol, biogas, energy | [193] |
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Akturk, A.S.; Demirer, G.N. Improved food waste stabilization and valorization by anaerobic digestion through supplementation of conductive materials and trace elements. Sustainability 2020, 12, 5222. [Google Scholar] [CrossRef]
- Pecorini, I.; Peruzzi, E.; Albini, E.; Doni, S.; Macci, C.; Masciandaro, G.; Iannelli, R. Evaluation of MSW compost and digestate mixtures for a circular economy application. Sustainability 2020, 12, 3042. [Google Scholar] [CrossRef]
- Achinas, S.; Euverink, G.J.W. Feasibility study of biogas production from hardly degradable material in co-inoculated bioreactor. Energies 2019, 12, 1040. [Google Scholar] [CrossRef]
- Rekleitis, G.; Haralambous, K.J.; Loizidou, M.; Aravossis, K. Utilization of agricultural and livestock waste in anaerobic digestion (AD): Applying the biorefinery concept in a circular economy. Energies 2020, 13, 4428. [Google Scholar] [CrossRef]
- Chow, W.L.; Chong, S.; Lim, J.W.; Chan, Y.J.; Chong, M.F.; Tiong, T.J.; Chin, J.K.; Pan, G.T. Anaerobic co-digestion of wastewater sludge: A review of potential co-substrates and operating factors for improved methane yield. Processes 2020, 8, 39. [Google Scholar] [CrossRef]
- Nwokolo, N.; Mukumba, P.; Obileke, K.; Enebe, M. Waste to energy: A focus on the impact of substrate type in biogas production. Processes 2020, 8, 1224. [Google Scholar] [CrossRef]
- González, J.; Sánchez, M.E.; Gómez, X. Enhancing anaerobic digestion: The effect of carbon conductive materials. C J. Carbon Res. 2018, 4, 59. [Google Scholar] [CrossRef]
- Muñoz, R.; Meier, L.; Diaz, I.; Jeison, D. A review on the state-of-the-art of physical/chemical and biological technologies for biogas upgrading. Rev. Environ. Sci. Biotechnol. 2015, 14, 727–759. [Google Scholar] [CrossRef]
- Adnan, A.I.; Ong, M.Y.; Nomanbhay, S.; Chew, K.W.; Show, P.L. Technologies for biogas upgrading to biomethane: A review. Bioengineering 2019, 6, 92. [Google Scholar] [CrossRef]
- Janošovský, J.; Marková, E.; Kačmárová, A.; Variny, M. Green dairy plant: Process simulation and economic analysis of biogas use in milk drying. Processes 2020, 8, 1262. [Google Scholar] [CrossRef]
- Kepp, U.; Machenbach, I.; Weisz, N.; Solheim, O.E. Enhanced stabilisation of sewage sludge through thermal hydrolysis-three years of experience with full scale plant. Water Sci. Technol. 2000, 42, 89–96. [Google Scholar] [CrossRef]
- Abu-Orf, M.; Goss, T. Comparing Thermal hydrolysis processes (CAMBI™ and EXELYS™) for solids pretreatmet prior to anaerobic digestion. Digestion 2012, 16, 8–12. [Google Scholar] [CrossRef]
- Available online: https://www.cambi.com/what-we-do/thermal-hydrolysis/how-does-thermal-hydrolysis-work/#:~:text=Thermal%20hydrolysis%20is%20a%20process,to%20preparing%20meals%20using%20steam (accessed on 5 October 2020).
- Available online: https://haarslev.com/industries/environmental/municipal/thermal-hydrolysis-process/ (accessed on 10 October 2020).
- Available online: http://technomaps.veoliawatertechnologies.com/processes/lib/municipal/3472-EN_Brochure_Exelys_0516.pdf (accessed on 12 October 2020).
- Available online: https://sustec.nl/wp-content/uploads/2017/02/16086_TurbotecTHP_LeafletA4_GB_lr.pdf (accessed on 11 October 2020).
- Available online: https://www.dmt-et.com/products/turbotec/ (accessed on 10 November 2020).
- Available online: https://www.aquaenviro.co.uk/wp-content/uploads/2015/06/Lysotherm%C2%AE-Sludge-Hydrolysis-Five-year-experience-with-a-novel-approach-for-operational-savings-Geraats-B.pdf (accessed on 2 November 2020).
- Available online: https://www.hielscher.com/sludge01.htm (accessed on 1 November 2020).
- Eskicioglu, C.; Prorot, A.; Marin, J.; Droste, R.L.; Kennedy, K.J. Synergetic pretreatment of sewage sludge by microwave irradiation in presence of H2O2 for enhanced anaerobic digestion. Water Res. 2008, 42, 4674–4682. [Google Scholar] [CrossRef]
- Pazos, M.; Alcántara, M.T.; Cameselle, C.; Sanromán, M.A. Evaluation of electrokinetic technique for industrial waste decontamination. Sep. Sci. Technol. 2009, 44, 2304–2321. [Google Scholar] [CrossRef]
- Tyagi, V.K.; Lo, S.L. Application of physico-chemical pretreatment methods to enhance the sludge disintegration and subsequent anaerobic digestion: An up to date review. Rev. Environ. Sci. Biotechnol. 2011, 10, 215. [Google Scholar] [CrossRef]
- Tyagi, V.K.; Lo, S.L.; Appels, L.; Dewil, R. Ultrasonic treatment of waste sludge: A review on mechanisms and applications. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1220–1288. [Google Scholar] [CrossRef]
- Neumann, P.; Pesante, S.; Venegas, M.; Vidal, G. Developments in pre-treatment methods to improve anaerobic digestion of sewage sludge. Rev. Environ. Sci. Biotechnol. 2016, 15, 173–211. [Google Scholar] [CrossRef]
- Le, T.M.; Vo, P.T.; Do, T.A.; Tran, L.T.; Truong, H.T.; Xuan Le, T.T.; Chen, Y.H.; Chang, C.C.; Chang, C.Y.; Tran, Q.T.; et al. Effect of assisted ultrasonication and ozone pretreatments on sludge characteristics and yield of biogas production. Processes 2019, 7, 743. [Google Scholar] [CrossRef]
- Martinez-Huitle, C.A.; Ferro, S. Electrochemical oxidation of organic pollutants for the wastewater treatment: Direct and indirect processes. Chem. Soc. Rev. 2006, 35, 1324–1340. [Google Scholar] [CrossRef]
- Feki, E.; Battimelli, A.; Sayadi, S.; Dhouib, A.; Khoufi, S. High-rate anaerobic digestion of waste activated sludge by integration of electro-Fenton process. Molecules 2020, 25, 626. [Google Scholar] [CrossRef]
- Zhen, G.; Lu, X.; Kato, H.; Zhao, Y.; Li, Y.Y. Overview of pretreatment strategies for enhancing sewage sludge disintegration and subsequent anaerobic digestion: Current advances, full-scale application and future perspectives. Renew. Sustain. Energy Rev. 2017, 69, 559–577. [Google Scholar] [CrossRef]
- Xu, Y.; Dai, X. Integrating multi-state and multi-phase treatment for anaerobic sludge digestion to enhance recovery of bio-energy. Sci. Total Environ. 2020, 698, 134196. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, B.; Tian, D.; Jun, H. Bioelectrochemical enhancement of methane production from highly concentrated food waste in a combined anaerobic digester and microbial electrolysis cell. Bioresour. Technol. 2018, 247, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Pickworth, B.; Adams, J.; Panter, K.; Solheim, O.E. Maximising biogas in anaerobic digestion by using engine waste heat for thermal hydrolysis pre-treatment of sludge. Water Sci. Technol. 2006, 54, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Kugel, G. Thermophilic/mesophilic digestion of sewage sludge and organic wastes. J. Environ. Sci. Health A 1996, 31, 2211–2231. [Google Scholar] [CrossRef]
- Kim, H.W.; Han, S.K.; Shin, H.S. The optimisation of food waste addition as a co-substrate in anaerobic digestion of sewage sludge. Waste Manag. Res. 2003, 21, 515–526. [Google Scholar] [CrossRef]
- Gómez, X.; Cuetos, M.J.; Cara, J.; Moran, A.; Garcia, A.I. Anaerobic co-digestion of primary sludge and the fruit and vegetable fraction of the municipal solid wastes: Conditions for mixing and evaluation of the organic loading rate. Renew. Energy 2006, 31, 2017–2024. [Google Scholar] [CrossRef]
- Martínez, E.J.; Sotres, A.; Arenas, C.B.; Blanco, D.; Martínez, O.; Gómez, X. Improving anaerobic digestion of sewage sludge by hydrogen addition: Analysis of microbial populations and process performance. Energies 2019, 12, 1228. [Google Scholar] [CrossRef]
- Moestedt, J.; Westerholm, M.; Isaksson, S.; Schnürer, A. Inoculum source determines acetate and lactate production during anaerobic digestion of sewage sludge and food waste. Bioengineering 2020, 7, 3. [Google Scholar] [CrossRef]
- Mu, L.; Zhang, L.; Zhu, K.; Ma, J.; Ifran, M.; Li, A. Anaerobic co-digestion of sewage sludge, food waste and yard waste: Synergistic enhancement on process stability and biogas production. Sci. Total Environ. 2020, 704, 135429. [Google Scholar] [CrossRef]
- Arenas, C.B.; Meredith, W.; Snape, C.E.; Gómez, X.; González, J.F.; Martinez, E.J. Effect of char addition on anaerobic digestion of animal by-products: Evaluating biogas production and process performance. Environ. Sci. Pollut. Res. 2020, 27, 24387–24399. [Google Scholar] [CrossRef] [PubMed]
- Lü, C.; Shen, Y.; Li, C.; Zhu, N.; Yuan, H. Redox-Active biochar and conductive graphite stimulate methanogenic metabolism in anaerobic digestion of waste-activated sludge: Beyond direct interspecies electron transfer. ACS Sustain. Chem. Eng. 2020, 8, 12626–12636. [Google Scholar] [CrossRef]
- Valero, A.; Valero, A. Thermodynamic rarity and recyclability of raw materials in the energy transition: The need for an in-spiral economy. Entropy 2019, 21, 873. [Google Scholar] [CrossRef]
- Herwig, H. How to teach heat transfer more systematically by involving entropy. Entropy 2018, 20, 791. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lv, W.; Guo, L.; Zhai, M.; Dong, P.; Qi, G. Energy and exergy analysis of rice husk high-temperature pyrolysis. Int. J. Hydrogen Energy 2016, 41, 21121–21130. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Dosta, J.; Romero-Güiza, M.S.; Fonoll, X.; Peces, M.; Astals, S. A critical review on anaerobic co-digestion achievements between 2010 and 2013. Renew. Sustain. Energy Rev. 2014, 36, 412–427. [Google Scholar] [CrossRef]
- Hagos, K.; Zong, J.; Li, D.; Liu, C.; Lu, X. Anaerobic co-digestion process for biogas production: Progress, challenges and perspectives. Renew. Sustain. Energy Rev. 2017, 76, 1485–1496. [Google Scholar] [CrossRef]
- Shah, F.A.; Mahmood, Q.; Rashid, N.; Pervez, A.; Raja, I.A.; Shah, M.M. Co-digestion, pretreatment and digester design for enhanced methanogenesis. Renew. Sustain. Energy Rev. 2015, 42, 627–642. [Google Scholar] [CrossRef]
- González, R.; Rosas, J.G.; Blanco, D.; Smith, R.; Martínez, E.J.; Pastor-Bueis, R.; Gómez, X. Anaerobic digestion of fourth range fruit and vegetable products: Comparison of three different scenarios for its valorisation by life cycle assessment and life cycle costing. Environ. Monit. Assess. 2020, 192, 1–19. [Google Scholar] [CrossRef]
- Naughton, C.C. Will the COVID-19 pandemic change waste generation and composition? The need for more real-time waste management data and systems thinking. Resour. Conserv. Recycl. 2020, 162, 105050. [Google Scholar] [CrossRef]
- Moreno, R.; Martínez, E.J.; Escapa, A.; Martínez, O.; Díez-Antolínez, R.; Gómez, X. Mitigation of volatile fatty acid build-up by the use of soft carbon felt electrodes: Evaluation of anaerobic digestion in acidic conditions. Fermentation 2018, 4, 2. [Google Scholar] [CrossRef]
- Fierro, J.; Martinez, E.J.; Rosas, J.G.; Fernández, R.A.; López, R.; Gomez, X. Co-Digestion of swine manure and crude glycerine: Increasing glycerine ratio results in preferential degradation of labile compounds. Water Air Soil Pollut. 2016, 227, 78. [Google Scholar] [CrossRef]
- Labatut, R.A.; Angenent, L.T.; Scott, N.R. Biochemical methane potential and biodegradability of complex organic substrates. Bioresour. Technol. 2011, 102, 2255–2264. [Google Scholar] [CrossRef] [PubMed]
- Nath, K.; Das, D. Improvement of fermentative hydrogen production: Various approaches. Appl. Microbiol. Biotechnol. 2004, 65, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Nandi, R.; Sengupta, S. Microbial production of hydrogen: An overview. Crit. Rev. Microbiol. 1998, 24, 61–84. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.M.; Trably, E.; Latrille, E.; Carrère, H.; Steyer, J.P. Hydrogen production from agricultural waste by dark fermentation: A review. Int. J. Hydrogen Energy 2010, 35, 10660–10673. [Google Scholar] [CrossRef]
- Moreno, R.; Gómez, X. Dark fermentative H2 production from wastes: Effect of operating conditions. J. Environ. Sci. Eng. A 2012, 1, 936. [Google Scholar]
- Davila-Vazquez, G.; Arriaga, S.; Alatriste-Mondragón, F.; de León-Rodríguez, A.; Rosales-Colunga, L.M.; Razo-Flores, E. Fermentative biohydrogen production: Trends and perspectives. Rev. Environ. Sci. Bio/Technol. 2008, 7, 27–45. [Google Scholar] [CrossRef]
- Peixoto, G.; Pantoja-Filho, J.L.R.; Agnelli, J.A.B.; Barboza, M.; Zaiat, M. Hydrogen and methane production, energy recovery, and organic matter removal from effluents in a two-stage fermentative process. Appl. Biochem. Biotechnol. 2012, 168, 651–671. [Google Scholar] [CrossRef]
- Redondas, V.; Moran, A.; Martínez, J.E.; Fierro, J.; Gómez, X. Effect of methanogenic effluent recycling on continuous H2 production from food waste. Environ. Prog. Sustain. Energy 2015, 34, 227–233. [Google Scholar] [CrossRef]
- Ghimire, A.; Luongo, V.; Frunzo, L.; Lens, P.N.; Pirozzi, F.; Esposito, G. Biohythane production from food waste in a two-stage process: Assessing the energy recovery potential. Environ. Technol. 2020, 1–17. [Google Scholar] [CrossRef]
- Larsen, J.F.; Wallace, J.S. Comparison of emissions and efficiency of a turbocharged lean-burn natural gas and hythane-fueled engine. J. Eng. Gas Turbines Power 1997, 119, 218–222. [Google Scholar] [CrossRef]
- Bolzonella, D.; Mıcoluccı, F.; Battısta, F.; Cavınato, C.; Gottardo, M.; Pıovesan, S.; Pavan, P. Producing biohythane from urban organic wastes. Waste Biomass Valorization 2020, 11, 2367–2374. [Google Scholar] [CrossRef]
- Fernández, C.; Cuetos, M.J.; Martínez, E.J.; Gómez, X. Thermophilic anaerobic digestion of cheese whey: Coupling H2 and CH4 production. Biomass Bioenergy 2015, 81, 55–62. [Google Scholar] [CrossRef]
- Leschine, S.B. Cellulose degradation in anaerobic environments. Ann. Rev. Microbiol. 1995, 49, 399–426. [Google Scholar] [CrossRef]
- Bao, Y.; Dolfing, J.; Wang, B.; Chen, R.; Huang, M.; Li, Z.; Lin, X.; Feng, Y. Bacterial communities involved directly or indirectly in the anaerobic degradation of cellulose. Biol. Fertil. Soils 2019, 55, 201–211. [Google Scholar] [CrossRef]
- Schwarz, W. The cellulosome and cellulose degradation by anaerobic bacteria. Appl. Microbiol. Biotechnol. 2001, 56, 634–649. [Google Scholar] [CrossRef]
- Park, S.; Baker, J.O.; Himmel, M.E.; Parilla, P.A.; Johnson, D.K. Cellulose crystallinity index: Measurement techniques and their impact on interpreting cellulase performance. Biotechnol. Biofuels 2010, 3, 10. [Google Scholar] [CrossRef]
- Yamazawa, A.; Iikura, T.; Morioka, Y.; Shino, A.; Ogata, Y.; Kikuchi, J. Cellulose digestion and metabolism induced biocatalytic transitions in anaerobic microbial ecosystems. Metabolites 2014, 4, 36–52. [Google Scholar] [CrossRef]
- Tambone, F.; Adani, F.; Gigliotti, G.; Volpe, D.; Fabbri, C.; Provenzano, M.R. Organic matter characterization during the anaerobic digestion of different biomasses by means of CPMAS 13C NMR spectroscopy. Biomass Bioenergy 2013, 48, 111–120. [Google Scholar] [CrossRef]
- Gómez, X.; Diaz, M.C.; Cooper, M.; Blanco, D.; Morán, A.; Snape, C.E. Study of biological stabilization processes of cattle and poultry manure by thermogravimetric analysis and 13C NMR. Chemosphere 2007, 68, 1889–1897. [Google Scholar] [CrossRef] [PubMed]
- Gómez, X.; Blanco, D.; Lobato, A.; Calleja, A.; Martínez-Núñez, F.; Martin-Villacorta, J. Digestion of cattle manure under mesophilic and thermophilic conditions: Characterization of organic matter applying thermal analysis and 1H NMR. Biodegradation 2011, 22, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Jiang, Q.; Zhu, R.; Chen, Y.; Sun, T.; Li, M.; Zhai, J.; Shi, D.; Ai, H.; Gu, L.; et al. Enhanced hydrolysis of lignocellulose in corn cob by using food waste pretreatment to improve anaerobic digestion performance. J. Environ. Manag. 2020, 254, 109830. [Google Scholar] [CrossRef] [PubMed]
- Chapleur, O.; Madigou, C.; Civade, R.; Rodolphe, Y.; Mazéas, L.; Bouchez, T. Increasing concentrations of phenol progressively affect anaerobic digestion of cellulose and associated microbial communities. Biodegradation 2016, 27, 15–27. [Google Scholar] [CrossRef]
- Puls, J. Chemistry and biochemistry of hemicelluloses: Relationship between hemicellulose structure and enzymes required for hydrolysis. In Macromolecular Symposia; Hüthig & Wepf Verlag: Basel, Switzerland, 1997; Volume 120, pp. 183–196. [Google Scholar]
- Ghosh, S.; Henry, M.P.; Christopher, R.W. Hemicellulose conversion by anaerobic digestion. Biomass 1985, 6, 257–269. [Google Scholar] [CrossRef]
- Li, W.; Khalid, H.; Zhu, Z.; Zhang, R.; Liu, G.; Chen, C.; Thorin, E. Methane production through anaerobic digestion: Participation and digestion characteristics of cellulose, hemicellulose and lignin. Appl. Energy 2018, 226, 1219–1228. [Google Scholar] [CrossRef]
- Waliszewska, H.; Zborowska, M.; Stachowiak-Wencek, A.; Waliszewska, B.; Czekała, W. Lignin transformation of one-year-old plants during anaerobic digestion (AD). Polymers 2019, 11, 835. [Google Scholar] [CrossRef]
- Kobayashi, T.; Xu, K.Q.; Li, Y.Y.; Inamori, Y. Effect of sludge recirculation on characteristics of hydrogen production in a two-stage hydrogen–methane fermentation process treating food wastes. Int. J. Hydrogen Energy 2012, 37, 5602–5611. [Google Scholar] [CrossRef]
- Qin, Y.; Wu, J.; Xiao, B.; Cong, M.; Hojo, T.; Cheng, J.; Li, Y.Y. Strategy of adjusting recirculation ratio for biohythane production via recirculated temperature-phased anaerobic digestion of food waste. Energy 2019, 179, 1235–1245. [Google Scholar] [CrossRef]
- Qin, Y.; Li, L.; Wu, J.; Xiao, B.; Hojo, T.; Kubota, K.; Cheng, J.; Li, Y.Y. Co-production of biohydrogen and biomethane from food waste and paper waste via recirculated two-phase anaerobic digestion process: Bioenergy yields and metabolic distribution. Bioresour. Technol. 2019, 276, 325–334. [Google Scholar] [CrossRef]
- Xiao, L.P.; Song, G.Y.; Sun, R.C. Effect of Hydrothermal Processing on Hemicellulose Structure. In Hydrothermal Processing in Biorefineries; Ruiz, H., Hedegaard Thomsen, M., Trajano, H., Eds.; Springer: Cham, Switzerland, 2017; pp. 45–94. [Google Scholar] [CrossRef]
- Wagner, A.O.; Lackner, N.; Mutschlechner, M.; Prem, E.M.; Markt, R.; Illmer, P. Biological pretreatment strategies for second-generation lignocellulosic resources to enhance biogas production. Energies 2018, 11, 1797. [Google Scholar] [CrossRef]
- Ahmed, B.; Aboudi, K.; Tyagi, V.K.; Álvarez-Gallego, C.J.; Fernández-Güelfo, L.A.; Romero-García, L.I.; Kazmi, A.A. Improvement of anaerobic digestion of lignocellulosic biomass by hydrothermal pretreatment. Appl. Sci. 2019, 9, 3853. [Google Scholar] [CrossRef]
- Mei, Z.; Liu, X.; Huang, X.; Li, D.; Yan, Z.; Yuan, Y.; Huang, Y. Anaerobic mesophilic codigestion of rice straw and chicken manure: Effects of organic loading rate on process stability and performance. Appl. Biochem. Biotechnol. 2016, 179, 846–862. [Google Scholar] [CrossRef]
- Zhao, C.; Mu, H.; Zhao, Y.; Wang, L.; Zuo, B. Microbial characteristics analysis and kinetic studies on substrate composition to methane after microbial and nutritional regulation of fruit and vegetable wastes anaerobic digestion. Bioresour. Technol. 2018, 249, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Tufaner, F.; Avşar, Y. Effects of co-substrate on biogas production from cattle manure: A review. Int. J. Environ. Sci. Technol. 2016, 13, 2303–2312. [Google Scholar] [CrossRef]
- Li, R.; Chen, S.; Li, X.; Saifullah Lar, J.; He, Y.; Zhu, B. Anaerobic codigestion of kitchen waste with cattle manure for biogas production. Energy Fuels 2009, 23, 2225–2228. [Google Scholar] [CrossRef]
- Ahn, Y.; Lee, W.; Kang, S.; Kim, S.H. Enhancement of sewage sludge digestion by co-digestion with food waste and swine waste. Waste Biomass Valorization 2019, 11, 2421–2430. [Google Scholar] [CrossRef]
- Kang, X.; Liu, Y. Chemically enhanced primary sludge as an anaerobic co-digestion additive for biogas production from food waste. Processes 2019, 7, 709. [Google Scholar] [CrossRef]
- Paulista, L.O.; Boaventura, R.A.; Vilar, V.J.; Pinheiro, A.L.; Martins, R.J. Enhancing methane yield from crude glycerol anaerobic digestion by coupling with ultrasound or A. niger/E. coli biodegradation. Environ. Sci. Pollut. Res. 2020, 27, 1461–1474. [Google Scholar] [CrossRef]
- González, R.; González, J.; Rosas, J.G.; Smith, R.; Gómez, X. Biochar and energy production: Valorizing swine manure through coupling co-digestion and pyrolysis. C J. Carbon Res. 2020, 6, 43. [Google Scholar] [CrossRef]
- Fierro, J.; Martínez, J.E.; Rosas, J.G.; Blanco, D.; Gómez, X. Anaerobic codigestion of poultry manure and sewage sludge under solid-phase configuration. Environ. Prog. Sustain. 2014, 33, 866–872. [Google Scholar] [CrossRef]
- Available online: https://www.mapa.gob.es/es/agricultura/temas/producciones-agricolas/cifras_del_sectorfyh_tcm30-502367.pdf (accessed on 12 November 2020).
- Fonoll, X.; Astals, S.; Dosta, J.; Mata-Alvarez, J. Anaerobic co-digestion of sewage sludge and fruit wastes: Evaluation of the transitory states when the co-substrate is changed. Chem. Eng. J. 2015, 262, 1268–1274. [Google Scholar] [CrossRef]
- Di Maria, F.; Sordi, A.; Cirulli, G.; Micale, C. Amount of energy recoverable from an existing sludge digester with the co-digestion with fruit and vegetable waste at reduced retention time. Appl. Energy 2015, 150, 9–14. [Google Scholar] [CrossRef]
- González, R.; Hernández, J.E.; Gómez, X.; Smith, R.; Arias, J.G.; Martínez, E.J.; Blanco, D. Performance evaluation of a small-scale digester for achieving decentralised management of waste. Waste Manag. 2020, 118, 99–109. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Blanco, D.; González-Arias, J.; García-Cascallana, J.; Gómez, X. Description of a decentralized small scale digester for treating organic wastes. Environments 2020, 7, 78. [Google Scholar] [CrossRef]
- Ahn, H.K.; Smith, M.C.; Kondrad, S.L.; White, J.W. Evaluation of biogas production potential by dry anaerobic digestion of switchgrass–animal manure mixtures. Appl. Biochem. Biotechnol. 2010, 160, 965–975. [Google Scholar] [CrossRef]
- Cuetos, M.J.; Fernández, C.; Gómez, X.; Morán, A. Anaerobic co-digestion of swine manure with energy crop residues. Biotechnol. Bioprocess Eng. 2011, 16, 1044. [Google Scholar] [CrossRef]
- Jurado, E.; Skiadas, I.V.; Gavala, H.N. Enhanced methane productivity from manure fibers by aqueous ammonia soaking pretreatment. Appl. Energy 2013, 109, 104–111. [Google Scholar] [CrossRef]
- Wang, M.; Lee, E.; Zhang, Q.; Ergas, S.J. Anaerobic co-digestion of swine manure and microalgae Chlorella sp.: Experimental studies and energy analysis. BioEnergy Res. 2016, 9, 1204–1215. [Google Scholar] [CrossRef]
- Rubežius, M.; Venslauskas, K.; Navickas, K.; Bleizgys, R. Influence of aerobic pretreatment of poultry manure on the biogas production process. Processes 2020, 8, 1109. [Google Scholar] [CrossRef]
- Wang, F.; Pei, M.; Qiu, L.; Yao, Y.; Zhang, C.; Qiang, H. Performance of anaerobic digestion of chicken manure under gradually elevated organic loading rates. Int. J. Environ. Res. Public Health 2019, 16, 2239. [Google Scholar] [CrossRef] [PubMed]
- Amon, T.; Amon, B.; Kryvoruchko, V.; Zollitsch, W.; Mayer, K.; Gruber, L. Biogas production from maize and dairy cattle manure—Influence of biomass composition on the methane yield. Agric. Ecosyst. Environ. 2007, 118, 173–182. [Google Scholar] [CrossRef]
- Baek, G.; Kim, D.; Kim, J.; Kim, H.; Lee, C. Treatment of cattle manure by anaerobic co-digestion with food waste and pig manure: Methane yield and synergistic effect. Int. J. Environ. Res. Public Health 2020, 17, 4737. [Google Scholar] [CrossRef] [PubMed]
- Ning, Z.; Ji, J.; He, Y.; Huang, Y.; Liu, G.; Chen, C. Effect of lipase hydrolysis on biomethane production from swine slaughterhouse waste in China. Energy Fuels 2016, 30, 7326–7330. [Google Scholar] [CrossRef]
- Cuetos, M.J.; Martinez, E.J.; Moreno, R.; Gonzalez, R.; Otero, M.; Gomez, X. Enhancing anaerobic digestion of poultry blood using activated carbon. J. Adv. Res. 2017, 8, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Kafle, G.K.; Kim, S.H.; Sung, K.I. Ensiling of fish industry waste for biogas production: A lab scale evaluation of biochemical methane potential (BMP) and kinetics. Bioresour. Technol. 2013, 127, 326–336. [Google Scholar] [CrossRef]
- Gunes, B.; Carrié, M.; Benyounis, K.; Stokes, J.; Davis, P.; Connolly, C.; Lawler, J. Optimisation and modelling of anaerobic digestion of whiskey distillery/brewery wastes after combined chemical and mechanical pre-treatment. Processes 2020, 8, 492. [Google Scholar] [CrossRef]
- Davidsson, Å.; Lövstedt, C.; la Cour Jansen, J.; Gruvberger, C.; Aspegren, H. Co-digestion of grease trap sludge and sewage sludge. Waste Manag. 2008, 28, 986–992. [Google Scholar] [CrossRef]
- Martínez, E.J.; Rosas, J.G.; Morán, A.; Gómez, X. Effect of ultrasound pretreatment on sludge digestion and dewatering characteristics: Application of particle size analysis. Water 2015, 7, 6483–6495. [Google Scholar] [CrossRef]
- Noutsopoulos, C.; Mamais, D.; Antoniou, K.; Avramides, C.; Oikonomopoulos, P.; Fountoulakis, I. Anaerobic co-digestion of grease sludge and sewage sludge: The effect of organic loading and grease sludge content. Bioresour. Technol. 2013, 131, 452–459. [Google Scholar] [CrossRef]
- Dos Santos Ferreira, J.; Volschan, I.; Cammarota, M.C. Co-digestion of sewage sludge with crude or pretreated glycerol to increase biogas production. Environ. Sci. Pollut. Res. 2018, 25, 21811–21821. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Abalde, Á.; Guivernau, M.; Prenafeta-Boldú, F.X.; Flotats, X.; Fernández, B. Characterization of microbial community dynamics during the anaerobic co-digestion of thermally pre-treated slaughterhouse wastes with glycerin addition. Bioprocess Biosyst. Eng. 2019, 42, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.; Wang, Y.; Xia, Y.; Chen, G.; Zhang, T. Investigation on the anaerobic co-digestion of food waste with sewage sludge. Appl. Microbiol. Biotechnol. 2017, 101, 7755–7766. [Google Scholar] [CrossRef]
- Joseph, G.; Zhang, B.; Mahzabin Rahman, Q.; Wang, L.; Shahbazi, A. Two-stage thermophilic anaerobic co-digestion of corn stover and cattle manure to enhance biomethane production. J. Environ. Sci. Health A 2019, 54, 452–460. [Google Scholar] [CrossRef]
- Raposo, F.; Borja, R.; Martín, M.A.; Martín, A.; De la Rubia, M.A.; Rincón, B. Influence of inoculum–substrate ratio on the anaerobic digestion of sunflower oil cake in batch mode: Process stability and kinetic evaluation. Chem. Eng. J. 2009, 149, 70–77. [Google Scholar] [CrossRef]
- Zhurka, M.; Spyridonidis, A.; Vasiliadou, I.A.; Stamatelatou, K. Biogas production from sunflower head and stalk residues: Effect of alkaline pretreatment. Molecules 2020, 25, 164. [Google Scholar] [CrossRef] [PubMed]
- Demirbas, A. Biogas potential of manure and straw mixtures. Energy Sources 2006, 28, 71–78. [Google Scholar] [CrossRef]
- Kaldis, F.; Cysneiros, D.; Day, J.; Karatzas, K.A.; Chatzifragkou, A. Anaerobic Digestion of Steam-Exploded Wheat Straw and Co-Digestion Strategies for Enhanced Biogas Production. Appl. Sci. 2020, 10, 8284. [Google Scholar] [CrossRef]
- Mancini, G.; Papirio, S.; Lens, P.N.; Esposito, G. A preliminary study of the effect of bioavailable Fe and Co on the anaerobic digestion of rice straw. Energies 2019, 12, 577. [Google Scholar] [CrossRef]
- Thaemngoen, A.; Saritpongteeraka, K.; Leu, S.Y.; Phuttaro, C.; Sawatdeenarunat, C.; Chaiprapat, S. Anaerobic digestion of napier grass (Pennisetum purpureum) in two-phase dry digestion system versus wet digestion system. BioEnergy Res. 2020, 13, 853–865. [Google Scholar] [CrossRef]
- Kacprzak, A.; Krzystek, L.; Paździor, K.; Ledakowicz, S. Investigation of kinetics of anaerobic digestion of Canary grass. Chem. Pap. 2012, 66, 550–555. [Google Scholar] [CrossRef]
- Pizarro-Loaiza, C.A.; Torres-Lozada, P.; Illa, J.; Palatsi, J.; Bonmatí, A. Effect of harvesting age and size reduction in the performance of anaerobic digestion of Pennisetum grass. Processes 2020, 8, 1414. [Google Scholar] [CrossRef]
- Hidaka, T.; Takabe, Y.; Tsumori, J.; Minamiyama, M. Characterization of microalgae cultivated in continuous operation combined with anaerobic co-digestion of sewage sludge and microalgae. Biomass Bioenergy 2017, 99, 139–146. [Google Scholar] [CrossRef]
- Saleem, M.; Hanif, M.U.; Bahadar, A.; Iqbal, H.; Capareda, S.C.; Waqas, A. The effects of hot water and ultrasonication pretreatment of microalgae (Nannochloropsis oculata) on biogas production in anaerobic co-digestion with cow manure. Processes 2020, 8, 1558. [Google Scholar] [CrossRef]
- Álvarez, X.; Arévalo, O.; Salvador, M.; Mercado, I.; Velázquez-Martí, B. Cyanobacterial biomass produced in the wastewater of the dairy industry and its evaluation in anaerobic co-digestion with cattle manure for enhanced methane production. Processes 2020, 8, 1290. [Google Scholar] [CrossRef]
- Tápparo, D.C.; do Amaral, A.C.; Steinmetz, R.L.R.; Kunz, A. Co-digestion of animal manure and carcasses to increase biogas generation. In Improving Biogas Production; Springer: Cham, Switzerland, 2019; pp. 99–116. [Google Scholar]
- Procházka, J.; Dolejš, P.; Máca, J.; Dohányos, M. Stability and inhibition of anaerobic processes caused by insufficiency or excess of ammonia nitrogen. Appl. Microbiol. Biotechnol. 2012, 93, 439–447. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, K.; Liu, P.; Khan, A.; Xiong, J.; Tian, F.; Li, X. A critical review on the interaction of substrate nutrient balance and microbial community structure and function in anaerobic co-digestion. Bioresour. Technol. 2018, 247, 1119–1127. [Google Scholar] [CrossRef]
- Rasapoor, M.; Young, B.; Brar, R.; Sarmah, A.; Zhuang, W.Q.; Baroutian, S. Recognizing the challenges of anaerobic digestion: Critical steps toward improving biogas generation. Fuel 2020, 261, 116497. [Google Scholar] [CrossRef]
- Alqaralleh, R.M.; Kennedy, K.; Delatolla, R. Improving biogas production from anaerobic co-digestion of thickened waste activated sludge (TWAS) and fat, oil and grease (FOG) using a dual-stage hyper-thermophilic/thermophilic semi-continuous reactor. J. Environ. Manag. 2018, 217, 416–428. [Google Scholar] [CrossRef]
- Marchetti, R.; Vasmara, C.; Bertin, L.; Fiume, F. Conversion of waste cooking oil into biogas: Perspectives and limits. Appl. Microbiol. Biotechnol. 2020, 104, 2833–2856. [Google Scholar] [CrossRef]
- Martínez, E.J.; Fierro, J.; Sánchez, M.E.; Gómez, X. Anaerobic co-digestion of FOG and sewage sludge: Study of the process by Fourier transform infrared spectroscopy. Int. Biodeterior. Biodegrad. 2012, 75, 1–6. [Google Scholar] [CrossRef]
- Kabouris, J.C.; Tezel, U.; Pavlostathis, S.G.; Engelmann, M.; Dulaney, J.A.; Todd, A.C.; Gillette, R.A. Mesophilic and thermophilic anaerobic digestion of municipal sludge and fat, oil, and grease. Water Environ. Res. 2009, 81, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, A.H.; Tao, L. Economic perspectives of biogas production via anaerobic digestion. Bioengineering 2020, 7, 74. [Google Scholar] [CrossRef] [PubMed]
- Moura, I.; Pereira, D. Anaerobic Digestion of MSW: Challenges of a High Cost Technology. In Modeling Innovation Sustainability and Technologies; Springer: Cham, Switzerland, 2018; pp. 295–300. [Google Scholar]
- Bujoczek, G.; Oleszkiewicz, J.; Sparling, R.; Cenkowski, S. High solid anaerobic digestion of chicken manure. J. Agric. Eng. Res. 2000, 76, 51–60. [Google Scholar] [CrossRef]
- Wedwitschka, H.; Gallegos Ibanez, D.; Schäfer, F.; Jenson, E.; Nelles, M. Material characterization and substrate suitability assessment of chicken manure for dry batch anaerobic digestion processes. Bioengineering 2020, 7, 106. [Google Scholar] [CrossRef]
- Massaro, V.; Digiesi, S.; Mossa, G.; Ranieri, L. The sustainability of anaerobic digestion plants: A win–win strategy for public and private bodies. J. Clean Prod. 2015, 104, 445–459. [Google Scholar] [CrossRef]
- Veolia Water. Available online: https://www.veoliawatertechnologies.fi/sites/g/files/dvc3336/files/document/2019/01/3473-EN_Brochure_Exelys_0516-1.pdf (accessed on 10 November 2020).
- Barber, B.; Nilsen, P.J.; Christy, P. Cambi SolidStream®: Thermal Hydrolysis as a pre-treatment for dewatering to further reduce operating costs. Proc. Water Environ. Fed. 2017, 5, 5070–5083. [Google Scholar] [CrossRef]
- Imeni, S.M.; Puy, N.; Ovejero, J.; Busquets, A.M.; Bartroli, J.; Pelaz, L.; Ponsá, S.; Colón, J. Techno-economic assessment of anaerobic co-digestion of cattle manure and wheat straw (raw and pre-treated) at small to medium dairy cattle farms. Waste Biomass Valorization 2020, 11, 4035–4051. [Google Scholar] [CrossRef]
- Rajendran, K.; Murthy, G.S. Techno-economic and life cycle assessments of anaerobic digestion—A review. Biocatal. Agric. Biotechnol. 2019, 20, 101207. [Google Scholar] [CrossRef]
- Piñas, J.A.V.; Venturini, O.J.; Lora, E.E.S.; del Olmo, O.A.; Roalcaba, O.D.C. An economic holistic feasibility assessment of centralized and decentralized biogas plants with mono-digestion and co-digestion systems. Renew. Energy 2019, 139, 40–51. [Google Scholar] [CrossRef]
- Al-Wahaibi, A.; Osman, A.I.; Ala’a, H.; Alqaisi, O.; Baawain, M.; Fawzy, S.; Rooney, D.W. Techno-economic evaluation of biogas production from food waste via anaerobic digestion. Sci. Rep. 2020, 10, 15719. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Decentralized biogas technology of anaerobic digestion and farm ecosystem: Opportunities and challenges. Front. Energy Res. 2014, 2, 10. [Google Scholar] [CrossRef]
- Akbulut, A. Techno-economic analysis of electricity and heat generation from farm-scale biogas plant: Çiçekdağı case study. Energy 2012, 44, 381–390. [Google Scholar] [CrossRef]
- Aui, A.; Li, W.; Wright, M.M. Techno-economic and life cycle analysis of a farm-scale anaerobic digestion plant in Iowa. Waste Manag. 2019, 89, 154–164. [Google Scholar] [CrossRef]
- Li, Y.; Han, Y.; Zhang, Y.; Luo, W.; Li, G. Anaerobic digestion of different agricultural wastes: A techno-economic assessment. Bioresour. Technol. 2020, 315, 123836. [Google Scholar] [CrossRef]
- Orive, M.; Cebrián, M.; Zufía, J. Techno-economic anaerobic co-digestion feasibility study for two-phase olive oil mill pomace and pig slurry. Renew. Energy 2016, 97, 532–540. [Google Scholar] [CrossRef]
- O’Connor, S.; Ehimen, E.; Pillai, S.C.; Lyons, G.; Bartlett, J. Economic and environmental analysis of small-scale anaerobic digestion plants on Irish dairy farms. Energies 2020, 13, 637. [Google Scholar] [CrossRef]
- Oreggioni, G.D.; Gowreesunker, B.L.; Tassou, S.A.; Bianchi, G.; Reilly, M.; Kirby, M.E.; Toop, T.A.; Theodorou, M.K. Potential for energy production from farm wastes using anaerobic digestion in the UK: An economic comparison of different size plants. Energies 2017, 10, 1396. [Google Scholar] [CrossRef]
- Milani, M.; Montorsi, L.; Stefani, M. An integrated approach to energy recovery from biomass and waste: Anaerobic digestion–gasification–water treatment. Waste Manag. Res. 2014, 32, 614–625. [Google Scholar] [CrossRef]
- Monlau, F.; Francavilla, M.; Sambusiti, C.; Antoniou, N.; Solhy, A.; Libutti, A.; Zabaniotou, A.; Barakat, A.; Monteleone, M. Toward a functional integration of anaerobic digestion and pyrolysis for a sustainable resource management. Comparison between solid-digestate and its derived pyrochar as soil amendment. Appl. Energy 2016, 169, 652–662. [Google Scholar] [CrossRef]
- Salman, C.A.; Schwede, S.; Thorin, E.; Yan, J. Predictive modelling and simulation of integrated pyrolysis and anaerobic digestion process. Energy Procedia 2017, 105, 850–857. [Google Scholar] [CrossRef]
- Parmar, K.R.; Ross, A.B. Integration of hydrothermal carbonisation with anaerobic digestion; opportunities for valorisation of digestate. Energies 2019, 12, 1586. [Google Scholar] [CrossRef]
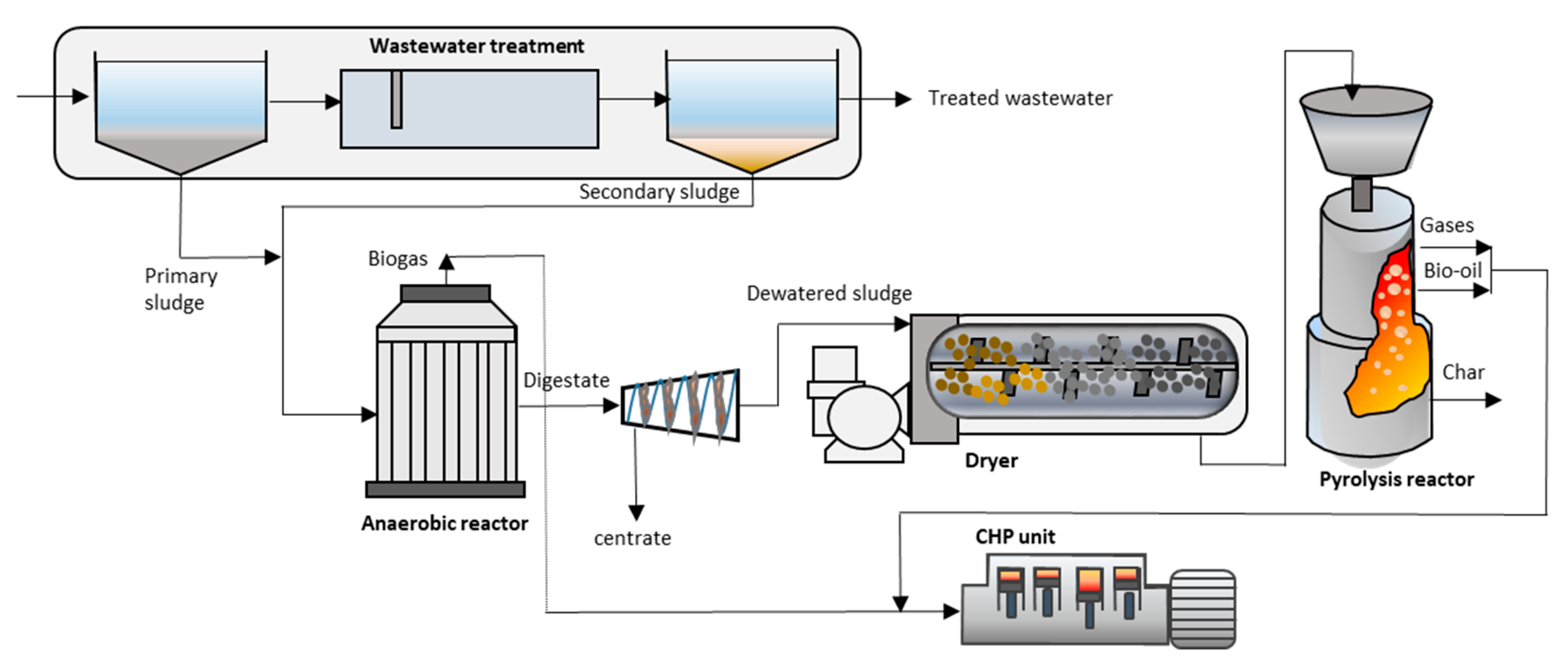
- González-Arias, J.; Gil, M.V.; Fernández, R.Á.; Martínez, E.J.; Fernández, C.; Papaharalabos, G.; Gómez, X. Integrating anaerobic digestion and pyrolysis for treating digestates derived from sewage sludge and fat wastes. Environ. Sci. Pollut. Res. Int. 2020, 27, 32603–32614. [Google Scholar] [CrossRef]
- Gonzalez-Arias, J.; Fernandez, C.; Rosas, J.G.; Bernal, M.P.; Clemente, R.; Sanchez, M.E.; Gomez, X. Integrating anaerobic digestion of pig slurry and thermal valorisation of biomass. Waste Biomass Valorization 2020, 11, 6125–6137. [Google Scholar] [CrossRef]
- Ghysels, S.; Acosta, N.; Estrada, A.; Pala, M.; De Vrieze, J.; Ronsse, F.; Rabaey, K. Integrating anaerobic digestion and slow pyrolysis improves the product portfolio of a cocoa waste biorefinery. Sustain. Energy Fuels 2020, 4, 3712–3725. [Google Scholar] [CrossRef]
- Opatokun, S.A.; Lopez-Sabiron, A.; Ferreira, G.; Strezov, V. Life cycle analysis of energy production from food waste through anaerobic digestion, pyrolysis and integrated energy system. Sustainability 2017, 9, 1804. [Google Scholar] [CrossRef]
- Li, H.; Feng, K. Life cycle assessment of the environmental impacts and energy efficiency of an integration of sludge anaerobic digestion and pyrolysis. J. Clean Prod. 2018, 195, 476–485. [Google Scholar] [CrossRef]
- Alves, J.L.F.; da Silva, J.C.G.; Languer, M.P.; Batistella, L.; Di Domenico, M.; da Silva Filho, V.F.; Muniz-Moreira, R.F.P.; José, H.J. Assessing the bioenergy potential of high-ash anaerobic sewage sludge using pyrolysis kinetics and thermodynamics to design a sustainable integrated biorefinery. Biomass Convers. Biorefin. 2020, 1–12. [Google Scholar] [CrossRef]
- Trippe, F.; Fröhling, M.; Schultmann, F.; Stahl, R.; Henrich, E. Techno-economic analysis of fast pyrolysis as a process step within biomass-to-liquid fuel production. Waste Biomass Valorization 2010, 1, 415–430. [Google Scholar] [CrossRef]
- Campbell, R.M.; Anderson, N.M.; Daugaard, D.E.; Naughton, H.T. Financial viability of biofuel and biochar production from forest biomass in the face of market price volatility and uncertainty. Appl. Energy 2018, 230, 330–343. [Google Scholar] [CrossRef]
- Shahbaz, M.; AlNouss, A.; Parthasarathy, P.; Abdelaal, A.H.; Mackey, H.; McKay, G.; Al-Ansari, T. Investigation of biomass components on the slow pyrolysis products yield using Aspen Plus for techno-economic analysis. Biomass Convers. Biorefin. 2020, 1–13. [Google Scholar] [CrossRef]
- Meyer, P.A.; Snowden-Swan, L.J.; Rappé, K.G.; Jones, S.B.; Westover, T.L.; Cafferty, K.G. Field-to-fuel performance testing of lignocellulosic feedstocks for fast pyrolysis and upgrading: Techno-economic analysis and greenhouse gas life cycle analysis. Energy Fuels 2016, 30, 9427–9439. [Google Scholar] [CrossRef]
- Sharma, H.B.; Vanapalli, K.R.; Cheela, V.S.; Ranjan, V.P.; Jaglan, A.K.; Dubey, B.; Goel, S.; Bhattacharya, J. Challenges, opportunities, and innovations for effective solid waste management during and post COVID-19 pandemic. Resour. Conserv. Recycl. 2020, 162, 105052. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.theguardian.com/environment/gallery/2020/jun/29/coronavirus-and-the-return-of-plastic-in-pictures (accessed on 9 January 2021).
- Vanapalli, K.R.; Sharma, H.B.; Ranjan, V.P.; Samal, B.; Bhattacharya, J.; Dubey, B.K.; Goel, S. Challenges and strategies for effective plastic waste management during and post COVID-19 pandemic. Sci. Total Environ. 2020, 750, 141514. [Google Scholar] [CrossRef] [PubMed]
- Eroğlu, H. Effects of Covid-19 outbreak on environment and renewable energy sector. Environ. Dev. Sustain. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nagaj, R.; Korpysa, J. Impact of COVID-19 on the level of energy poverty in Poland. Energies 2020, 13, 4977. [Google Scholar] [CrossRef]
- Clark, J.H.; Deswarte, F.E. The biorefinery concept—An integrated approach. In Introduction to Chemicals from Biomass; John Wiley & Sons: Chichester, UK, 2008. [Google Scholar]
- Johansson, E.; Prade, T.; Angelidaki, I.; Svensson, S.E.; Newson, W.R.; Gunnarsson, I.B.; Hovmalm, H.P. Economically viable components from Jerusalem artichoke (Helianthus tuberosus L.) in a biorefinery concept. Int. J. Mol. Sci. 2015, 16, 8997–9016. [Google Scholar] [CrossRef]
- Álvarez-Viñas, M.; Flórez-Fernández, N.; Torres, M.D.; Domínguez, H. Successful Approaches for a Red Seaweed Biorefinery. Mar. Drugs 2019, 17, 620. [Google Scholar] [CrossRef]
- Clark, J.H. Green biorefinery technologies based on waste biomass. Green Chem. 2019, 21, 1168–1170. [Google Scholar] [CrossRef]
- Redondo-Gómez, C.; Rodríguez Quesada, M.; Vallejo Astúa, S.; Murillo Zamora, J.P.; Lopretti, M.; Vega-Baudrit, J.R. Biorefinery of biomass of agro-industrial banana waste to obtain high-value biopolymers. Molecules 2020, 25, 3829. [Google Scholar] [CrossRef]
- Demichelis, F.; Laghezza, M.; Chiappero, M.; Fiore, S. Technical, economic and environmental assessment of bioethanol biorefinery from waste biomass. J. Clean. Prod. 2020, 277, 124111. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Conrad, M.; Sun, S.N.; Sanchez, A.; Rocha, G.J.; Romaní, A.; Castro, E.; Torres, A.; Rodríguez-Jasso, R.M.; Andrade, L.P.; et al. Engineering aspects of hydrothermal pretreatment: From batch to continuous operation, scale-up and pilot reactor under biorefinery concept. Bioresour. Technol. 2020, 299, 122685. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Elvira, S.I.; Fdz-Polanco, F. Continuous thermal hydrolysis and anaerobic digestion of sludge. Energy integration study. Water Sci. Technol. 2012, 65, 1839–1846. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.cambi.com/media/1929/barber-et-al-2017-cambi-solidstream-th-as-a-pretreatment-for-dewatering-proc-of-weftec.pdf (accessed on 15 November 2020).
- Ren, T.; Xu, W.; Jia, G.W.; Cai, M. A novel isothermal compression method for energy conservation in fluid power systems. Entropy 2020, 22, 1015. [Google Scholar] [CrossRef]
- Menon, V.; Rao, M. Trends in bioconversion of lignocellulose: Biofuels, platform chemicals & biorefinery concept. Prog. Energy Combust. Sci. 2012, 38, 522–550. [Google Scholar]
- Fernando, S.; Adhikari, S.; Chandrapal, C.; Murali, N. Biorefineries: Current status, challenges, and future direction. Energy Fuels 2006, 20, 1727–1737. [Google Scholar] [CrossRef]
- Alves, C.M.; Valk, M.; de Jong, S.; Bonomi, A.; van der Wielen, L.A.; Mussatto, S.I. Techno-economic assessment of biorefinery technologies for aviation biofuels supply chains in Brazil. Biofuels Bioprod. Biorefin. 2017, 11, 67–91. [Google Scholar] [CrossRef]
- Balboa, E.M.; Moure, A.; Domínguez, H. Valorization of Sargassum muticum biomass according to the biorefinery concept. Mar. Drugs 2015, 13, 3745–3760. [Google Scholar] [CrossRef]
- Ravi, H.K.; Degrou, A.; Costil, J.; Trespeuch, C.; Chemat, F.; Vian, M.A. Larvae mediated valorization of industrial, agriculture and food wastes: Biorefinery concept through bioconversion, processes, procedures, and products. Processes 2020, 8, 857. [Google Scholar] [CrossRef]
- Alves de Oliveira, R.; Schneider, R.; Hoss Lunelli, B.; Vaz Rossell, C.E.; Maciel Filho, R.; Venus, J. A simple biorefinery concept to produce 2G-lactic acid from sugar beet pulp (SBP): A high-value target approach to valorize a waste stream. Molecules 2020, 25, 2113. [Google Scholar] [CrossRef]
- Zacharof, M.P. Grape winery waste as feedstock for bioconversions: Applying the biorefinery concept. Waste Biomass Valorization 2017, 8, 1011–1025. [Google Scholar] [CrossRef]
- Bressanin, J.M.; Klein, B.C.; Chagas, M.F.; Watanabe, M.D.B.; Sampaio, I.L.D.M.; Bonomi, A.; Morais, E.R.D.; Cavalett, O. Techno-economic and environmental assessment of biomass gasification and Fischer–Tropsch synthesis integrated to sugarcane biorefineries. Energies 2020, 13, 4576. [Google Scholar] [CrossRef]
- Ortiz-Sanchez, M.; Solarte-Toro, J.C.; González-Aguirre, J.A.; Peltonen, K.E.; Richard, P.; Alzate, C.A.C. Pre-feasibility analysis of the production of mucic acid from orange peel waste under the biorefinery concept. Biochem. Eng. J. 2020, 161, 107680. [Google Scholar] [CrossRef]
- Cherubini, F.; Jungmeier, G. LCA of a biorefinery concept producing bioethanol, bioenergy, and chemicals from switchgrass. Int. J. LCA 2010, 15, 53–66. [Google Scholar] [CrossRef]
- Rabelo, S.C.; Carrere, H.; Maciel Filho, R.; Costa, A.C. Production of bioethanol, methane and heat from sugarcane bagasse in a biorefinery concept. Bioresour. Technol. 2011, 102, 7887–7895. [Google Scholar] [CrossRef] [PubMed]
- Okoro, O.V.; Sun, Z.; Birch, J. Techno-economic assessment of a scaled-up meat waste biorefinery system: A simulation study. Materials 2019, 12, 1030. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Sebastiá, M.; Gomis-Fons, J.; Galbe, M.; Wallberg, O. Techno-economic evaluation of biorefineries based on low-value feedstocks using the BioSTEAM software: A case study for animal bedding. Processes 2020, 8, 904. [Google Scholar] [CrossRef]
- Zetterholm, J.; Bryngemark, E.; Ahlström, J.; Söderholm, P.; Harvey, S.; Wetterlund, E. Economic evaluation of large-scale biorefinery deployment: A framework integrating dynamic biomass market and techno-economic models. Sustainability 2020, 12, 7126. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sevillano, C.A.; Pesantes, A.A.; Peña Carpio, E.; Martínez, E.J.; Gómez, X. Anaerobic Digestion for Producing Renewable Energy—The Evolution of This Technology in a New Uncertain Scenario. Entropy 2021, 23, 145. https://doi.org/10.3390/e23020145
Sevillano CA, Pesantes AA, Peña Carpio E, Martínez EJ, Gómez X. Anaerobic Digestion for Producing Renewable Energy—The Evolution of This Technology in a New Uncertain Scenario. Entropy. 2021; 23(2):145. https://doi.org/10.3390/e23020145
Chicago/Turabian StyleSevillano, Cristián Arenas, Alby Aguilar Pesantes, Elizabeth Peña Carpio, Elia J. Martínez, and Xiomar Gómez. 2021. "Anaerobic Digestion for Producing Renewable Energy—The Evolution of This Technology in a New Uncertain Scenario" Entropy 23, no. 2: 145. https://doi.org/10.3390/e23020145
APA StyleSevillano, C. A., Pesantes, A. A., Peña Carpio, E., Martínez, E. J., & Gómez, X. (2021). Anaerobic Digestion for Producing Renewable Energy—The Evolution of This Technology in a New Uncertain Scenario. Entropy, 23(2), 145. https://doi.org/10.3390/e23020145







