Aperiodicity in Low Dimensions
Abstract
1. Outline and Scope of the Review
2. Quasicrystals in Three Dimension
2.1. Basic Facts About Structure of 3D Quasicrystals
- Icosahedral quasicrystals, which display pentagonal and icosahedral symmetries, like Aluminum–Manganese alloys.
- Decagonal quasicrystals with 10-fold symmetry in one direction and periodicity along another, like Aluminum–Nickel–Cobalt intermetallics.
- Dodecagonal quasicrystals, which display 12-fold symmetry, are typically found in transition metal alloys. Example: Aluminum–Cobalt–Nickel systems.
- Octagonal and other quasicrystals are rare quasicrystals that exhibit 8-fold or other non-crystallographic symmetries.
2.2. Fundamental Properties of 3D Quasicrystals
3. Quasicrystals in Low Dimensions
3.1. Departure from Periodicity of Free-Standing, Highly Symmetrical 2D Lattices
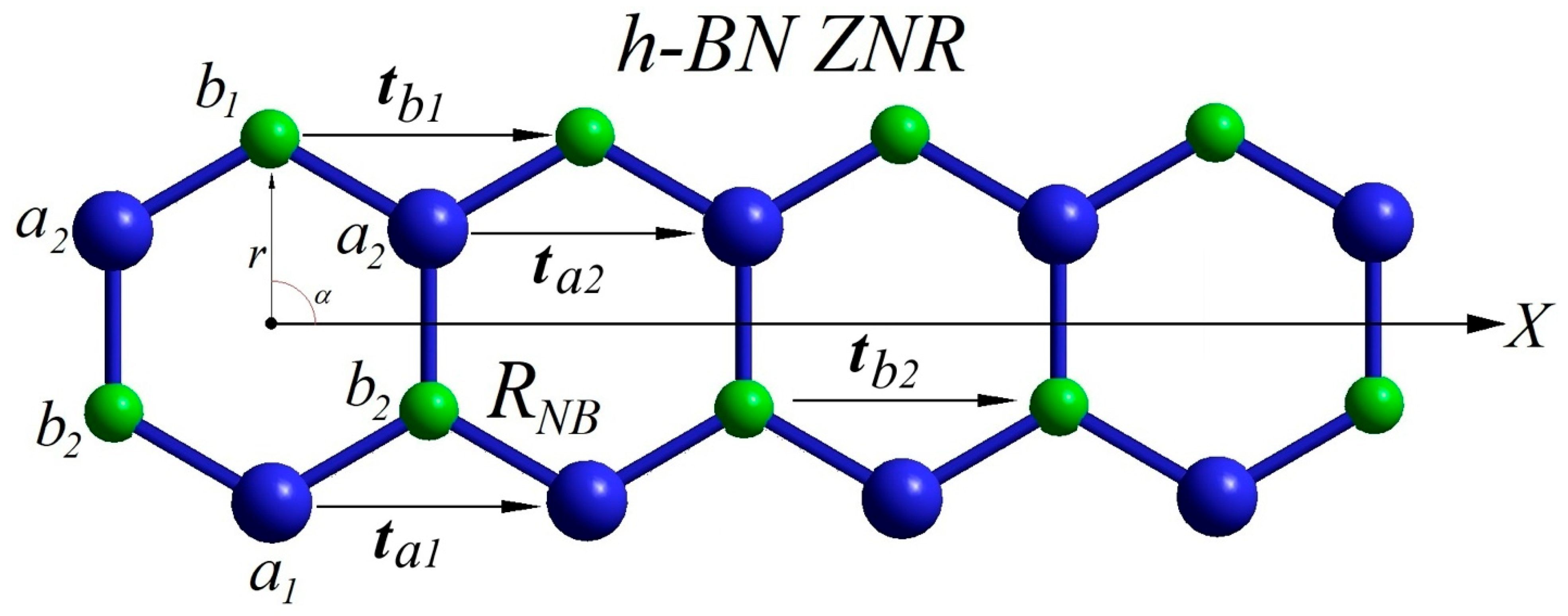
3.2. Topological Instability of Low-Dimensional Lattices with Multiple Nonequivalent Sublattices: Topology Conservation, Theorem, Theorema Egregium, and Euler–Gauss–Bonnet Theorem
“To conserve planar topology of one-unit-cell-thick planar crystals with negligible stabilizing force constant in the perpendicular direction, and to avoid uncompensated mechanical stress perpendicular to the regular lattice plane, the free-standing constituting fragments (unit cells) must perfectly fit the low-dimensional space. Due to the leading contribution of the stretching force constants to total energy, any small regular structural mismatch should accumulate and lead to motion of the crystalline lattice in the perpendicular direction to the plane to compensate the accumulated mechanical stress.”
3.3. Two-Dimensional Quasicrystals Based on Penrose Tilings
3.4. Aperiodicity in 2D Incommensurate Lattices
3.5. Zero-Dimensional Finite-Sized Aperiodic Crystalline Solids Based on Closed-Shell Multiply Twinned sp3 Carbon and Silicon Clusters
4. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Levine, D.; Steinhardt, P.J. Quasicrystals: A new class of ordered structures. Phys. Rev. Lett. 1984, 53, 2477–2480. [Google Scholar] [CrossRef]
- Levine, D.; Steinhardt, P.J. Quasicrystals. I. Definition and structure. Phys. Rev. B 1986, 34, 596. [Google Scholar] [CrossRef] [PubMed]
- Elser, V.; Henley, C.L. Crystal and quasicrystal structures in Al–Mn–Si alloys. Phys. Rev. Lett. 1985, 55, 2883–2886. [Google Scholar] [CrossRef]
- Noya, E.G.; Wong, C.K.; Llombart, P.; Doye, J.P.K. How to design an icosahedral quasicrystal through directional bonding. Nature 2021, 596, 367–371. [Google Scholar] [CrossRef]
- Penrose, R. The role of aesthetics in pure and applied mathematical research. Bull. Inst. Math. Appl. 1974, 10, 266–271. [Google Scholar]
- Penrose, R. Pentaplexity: A class of nonperiodic tilings of the plane. Math. Intell. 1979, 2, 32–37. [Google Scholar] [CrossRef]
- Dubois, J.-M. Properties and applications of quasicrystals and complex metallic alloys. Chem. Soc. Rev. 2012, 41, 6760–6777. [Google Scholar] [CrossRef] [PubMed]
- Scherrer, W. Die Einlagerung eines regulären Vielecks in ein Gitter. Elem. Der Math. 1946, 1, 97–98. [Google Scholar]
- Bamberg, J.; Cairns, G.; Kilminster, D. The crystallographic restriction, permutations, and Goldbach’s conjecture. Am. Math. Mon. 2003, 110, 202–209. [Google Scholar] [CrossRef]
- Bončina, T.; Zupanič, F. Metallography of quasicrystals in Al-alloys. Materials 2025, 18, 4575. [Google Scholar] [CrossRef]
- Steurer, W. The structure of quasicrystals. Mater. Sci. Forum. 1994, 150–151, 15–34. [Google Scholar] [CrossRef]
- Louzguine-Luzgin, D.V.; Inoue, A. Formation and properties of quasicrystals. Annu. Rev. Mater. Res. 2008, 38, 403–423. [Google Scholar] [CrossRef]
- Landau, L.D.; Lifshitz, E.M. Course of Theoretical Physics, Vol. 5: Statistical Physics, Part 1, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2005; Volume 137, pp. 432–436. [Google Scholar]
- Mermin, N.D. Crystalline order in two dimensions. Phys. Rev. 1968, 176, 250–254. [Google Scholar] [CrossRef]
- Nelson, D.R.; Piran, T.; Weinberg, S. (Eds.) Statistical Mechanics of Membranes and Surfaces; World Scientific: Singapore, 2004. [Google Scholar]
- Nelson, D.R.; Peliti, L. Fluctuations in membranes with crystalline and hexatic order. J. Physique 1987, 48, 1085–1092. [Google Scholar] [CrossRef]
- Le Doussal, P.; Radzihovsky, L. Self-consistent theory of polymerized membranes. Phys. Rev. Lett. 1992, 69, 1209–1212. [Google Scholar] [CrossRef]
- Mermin, N.D.; Wagner, H. Absence of ferromagnetism or antiferromagnetism in one- or two-dimensional isotropic Heisenberg models. Phys. Rev. Lett. 1966, 17, 1133–1136. [Google Scholar] [CrossRef]
- Hohenberg, P.C. Existence of long-range order in one and two dimensions. Phys. Rev. 1967, 158, 383. [Google Scholar] [CrossRef]
- Zakharchenko, K.V.; Katsnelson, M.I.; Fasolino, A. Atomistic simulations of structural and thermodynamic properties of bilayer graphene. Phys. Rev. Lett. 2009, 102, 046808. [Google Scholar] [CrossRef]
- Los, J.H.; Zakharchenko, K.V.; Katsnelson, M.I. Scaling properties of flexible membranes from atomistic simulations: Application to graphene. Phys. Rev. B 2009, 80, 121405. [Google Scholar] [CrossRef]
- Fasolino, A.; Los, J.H.; Katsnelson, M.I. Intrinsic ripples in graphene. Nat. Mater. 2007, 6, 858–861. [Google Scholar] [CrossRef]
- Meyer, J.C.; Geim, A.K.; Katsnelson, M.I.; Novoselov, K.S.; Booth, T.J.; Roth, S. The structure of suspended graphene sheets. Nature 2007, 446, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Girit, C.; Meyer, J.C.; Erni, R.; Roseli, M.D.; Kisielowski, C.; Yang, L.; Park, C.H.; Crommie, M.F.; Cohen, M.L.; Loie, S.G.; et al. Graphene at the Edge: Stability and Dynamics. Science 2009, 323, 1705–1708. [Google Scholar] [CrossRef]
- Bao, W.; Miao, F.; Chen, Z.; Zhang, H.; Jang, W.; Dames, C.; Lau, C.N. Controlled ripple texturing of suspended graphene and ultrathin graphite membranes. Nat. Nanotechnol. 2009, 4, 562–566. [Google Scholar] [CrossRef]
- Tapasztó, L.; Dumitrică, T.; Kim, S.J.; Nemes-Incze, P.; Hwang, C.; Biró, L.P. Breakdown of continuum mechanics for nanometre-wavelength rippling of graphene. Nat. Phys. 2012, 8, 739–742. [Google Scholar] [CrossRef]
- Larach, S.; Shrader, R.E. Electroluminescence from boron nitride. Phys. Rev. 1956, 102, 582. [Google Scholar] [CrossRef]
- Elias, D.C.; Nair, R.R.; Mohiuddin, T.M.G.; Morozov, S.V.; Blake, P.; Halsall, M.P.; Ferrari, A.C.; Boukhalv, D.W.; Katsnelson, M.I.; Geim, A.K.; et al. Novoselov, Control of graphene’s properties by reversible hydrogenation: Evidence for graphane. Science 2009, 323, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Ketabi, N.; Tolhurst, T.M.; Leedahl, B.; Liu, H.; Li, Y.; Moewes, A. How functional groups change the electronic structure of graphdiyne: Theory and experiment. Carbon 2017, 123, 1–6. [Google Scholar] [CrossRef]
- Jia, Z.; Li, Y.; Zuo, Z.; Liu, H.; Huang, C.; Li, Y. Synthesis and properties of 2D carbon—Graphdiyne. Acc. Chem. Res. 2017, 50, 2470–2478. [Google Scholar] [CrossRef]
- Desyatkin, V.G.; Martin, W.B.; Aliev, A.E.; Chapman, N.E.; Fonseca, A.F.; Galvão, D.S.; Miller, E.R.; Stone, K.H.; Wang, Z.; Zakhidov, D.; et al. Scalable synthesis and characterization of multilayer γ-graphyne, new carbon crystals with a small direct band gap. J. Am. Chem. Soc. 2022, 144, 12646–12655. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Cho, S.M.; Lin, S.; Chen, Z.; Choi, W.; Kim, Y.-M.; Yun, E.; Baek, E.H.; Ryu, D.H.; Lee, H. Constructing two-dimensional holey graphyne with unusual annulative π-extension. Matter 2022, 5, 2306–2318. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Wang, Q.; Jin, Y.; Huang, D.; Cui, Q.; Zou, G. Nitrogen-rich carbon nitride hollow vessels: Synthesis, characterization, and their properties. J. Phys. Chem. B 2010, 114, 9429–9434. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Wang, X.Q.; Luo, H.M.; Dai, S. Fluidic Carbon Precursors for Formation of Functional Carbon under Ambient Pressure Based on Ionic Liquids. Adv. Mater. 2010, 22, 1004. [Google Scholar] [CrossRef] [PubMed]
- Chernozatonskii, L.A.; Sorokin, P.B.; Kvashnin, A.G.; Kvashnin, D.G. Diamond-like C2H nanolayer, diamane: Simulation of the structure and properties. JETP Lett. 2009, 90, 134–138. [Google Scholar] [CrossRef]
- Bakharev, P.V.; Huang, M.; Saxena, M.; Lee, S.W.; Joo, S.H.; Park, S.O.; Dong, J.; Camacho-Mojica, D.C.; Jin, S.; Kwon, Y.; et al. Chemically induced transformation of CVD-grown bilayer graphene into fluorinated single-layer diamond. Nat. Nanotechnol. 2020, 15, 59–66. [Google Scholar] [CrossRef]
- O’Hare, A.; Kusmartsev, F.V.; Kugel, K.I.; Stable, A. “Flat” Form of Two-Dimensional Crystals: Could Graphene, Silicene, Germanene Be Minigap Semiconductors? Nano Lett. 2012, 12, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Avramov, P. Topology conservation theorem, quantum instability and violation of subperiodic symmetry of complex low-dimensional lattices. Acta Crystallogr. 2023, A79, C325. [Google Scholar] [CrossRef]
- Avramov, P.V.; Kuklin, A.V. Topological and Quantum Stability of Low-Dimensional Crystalline Lattices with Multiple Nonequivalent Sublattices. New J. Phys. 2022, 24, 103015. [Google Scholar] [CrossRef]
- Taskaev, S.; Skokov, K.; Khovaylo, V.; Donner, W.; Faske, T.; Dudorov, A.; Gorkavyi, N.; Muratov, D.S.; Savosteenko, G.; Dyakonov, A.; et al. Exotic carbon microcrystals in meteoritic dust of the Chelyabinsk superbolide: Experimental investigations and theoretical scenarios of their formation. Eur. Phys. J. Plus 2022, 137, 562. [Google Scholar] [CrossRef]
- Kuklin, A.; Ågren, H.; Avramov, P.V. Structural stability of single-layer PdSe2 with pentagonal puckered morphology and its nanotubes. Phys. Chem. Chem. Phys. 2020, 22, 8289–8295. [Google Scholar] [CrossRef]
- Avramov, P.; Demin, V.; Luo, M.; Choi, C.H.; Sorokin, P.B.; Yakobson, B.; Chernozatonskii, L. Translation Symmetry Breakdown in Low-Dimensional Lattices of Pentagonal Rings. J. Phys. Chem. Lett. 2015, 6, 4525–4531. [Google Scholar] [CrossRef]
- Avramov, P.V.; Fedorov, D.G.; Sorokin, P.B.; Sakai, S.; Entani, S.; Ohtomo, M.; Matsumoto, Y.; Naramoto, H. Intrinsic Edge Asymmetry in Narrow Zigzag Hexagonal Heteroatomic Nanoribbons Causes their Subtle Uniform Curvature. J. Phys. Chem. Lett. 2012, 3, 2003–2008. [Google Scholar] [CrossRef]
- Gauss, G.F. Disquisitiones Generales Circa Superficies Curvas; Typis Dieterichianis: Göttingen, Germany, 1827. [Google Scholar]
- do Carmo, M.P. Differential Geometry of Curves and Surfaces; Prentice-Hall: Hoboken, NJ, USA, 1976; Volume 3. [Google Scholar]
- Weisstein, E.W. Gauss’s Theorema Egregium, MathWorld—A Wolfram Web Resource. Available online: https://mathworld.wolfram.com/TheoremaEgregium.html (accessed on 10 December 2025).
- Neto, A.H.C.; Guinea, F.; Peres, N.M.R.; Novoselov, K.S.; Geim, A.K. The electronic properties of graphene. Rev. Mod. Phys. 2009, 81, 109–162. [Google Scholar] [CrossRef]
- Lee, J.M. Introduction to Riemannian Manifolds, Graduate Texts in Mathematics; Springer: Cham, Switzerland, 2018; Volume 176. [Google Scholar] [CrossRef]
- Cheeger, J.; Müller, W.; Schrader, R. On the curvature of piecewise flat spaces. Commun. Math. Phys. 1984, 92, 405–454. [Google Scholar] [CrossRef]
- Kroto, H.; Heath, J.R.; O’Brien, S.C.; Curl, R.F. Smalley C60: Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar] [CrossRef]
- Prinz, V.Y.; Seleznev, V.A.; Gutakovsky, A.K.; Chehovskiy, A.V.; Preobrazhenskii, V.V.; Putyato, M.A.; Gavrilova, T.A. Free-standing and overgrown InGaAs=GaAs nanotubes, nanohelices and their arrays. Physica E 2000, 6, 828–831. [Google Scholar] [CrossRef]
- Kotakoski, J.; Krasheninnikov, A.V.; Kaiser, U.; Meyer, J.C. From Point Defects in Graphene to Two-Dimensional Amorphous Carbon. Phys. Rev. Lett. 2011, 106, 105505. [Google Scholar] [CrossRef]
- Kotakoski, J.; Meyer, J.C.; Kurasch, S.; Santos-Cottin, D.; Kaiser, U.; Krasheninnikov, A.V. Stone-Wales-type transformations in carbon nanostructures driven by electron irradiation. Phys. Rev. B 2011, 83, 245420. [Google Scholar] [CrossRef]
- Kuzubov, A.A.; Avramov, P.V.; Tomilin, F.N.; Ovchinnikov, S.G. Theoretical investigation of toroidal forms of carbon and their endohedral derivatives with Li ions. Phys. Solid State 2001, 43, 1982–1988. [Google Scholar] [CrossRef]
- Grünbaum, B.; Shephard, G.C. Tilings and Patterns; W. H. Freeman and Company: New York, NY, USA, 1987. [Google Scholar]
- Yadav, T.P.; Woellner, C.F.; Sinha, S.K.; Sharifi, T.; Apte, A.; Mukhopadhyay, N.K.; Srivastava, O.N.; Vajtai, R.; Galvao, D.S.; Tiwary, C.S.; et al. Liquid exfoliation of icosahedral quasicrystals. Adv. Funct. Mater. 2018, 28, 1801181. [Google Scholar] [CrossRef]
- Yadav, T.P.; Woellner, C.F.; Sharifi, T.; Sinha, S.K.; Qu, L.-L.; Apte, A.; Mukhopadhyay, N.K.; Srivastava, O.N.; Vajtai, R.; Galvão, D.S.; et al. Extraction of Two-Dimensional Aluminum Alloys from Decagonal Quasicrystals. ACS Nano 2020, 14, 7435–7443. [Google Scholar] [CrossRef]
- Ngoc, B.P.; Geantet, C.; Aouine, M.; Bergeret, G.; Marlin, S.R.S. Quasicrystal derived catalyst for steam reforming of methanol. Int. J. Hydrogen Energy 2008, 33, 1000–1007. [Google Scholar] [CrossRef]
- Kameoka, S.; Tanabe, T.; Tsai, A.P. Al-Cu-Fe quasicrystals for steam reforming of methanol: A new form of copper catalysts. Catal. Today 2004, 93, 23–26. [Google Scholar] [CrossRef]
- Tanabe, T.; Kameoka, S.; Tsai, A.P. Microstructure of leached Al-Cu-Fe quasicrystal with high catalytic performance for steam reforming of methanol. Appl. Catal. A Gen. 2010, 384, 241–251. [Google Scholar] [CrossRef]
- Tanabe, T.; Kameoka, S.; Tsai, A.P. Evolution of microstructure induced by calcination in leached Al-Cu-Fe quasicrystal and its effects on catalytic activity. J. Mater. Sci. 2011, 46, 2242–2250. [Google Scholar] [CrossRef]
- Pandey, S.K.; Bhatnagar, A.; Mishra, S.S.; Yadav, T.P.; Shaz, M.A.; Srivastava, O.N. Curious Catalytic Characteristics of Al–Cu– Fe Quasicrystal for De/Rehydrogenation of MgH2. J. Phys. Chem. C 2017, 121, 24936–24944. [Google Scholar] [CrossRef]
- Yamamoto, A. Software package for structure analysis of quasicrystals. Sci. Technol. Adv. Mater. 2008, 9, 013001. [Google Scholar] [CrossRef] [PubMed]
- Kittel, C. Introduction to Solid State Physics, 8th ed.; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Zallen, R. The Physics of Amorphous Solids; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- van Smaalen, S. Incommensurate Crystallography, IUCr Monographs; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Grimm, U.; Scheffer, M. Incommensurate Crystals and Quasicrystals. In Encyclopedia of Physical Science and Technology; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2001; Volume 7. [Google Scholar]
- Suck, J.-B. Lattice Dynamics: Aperiodic Crystals. In Encyclopedia of Condensed Matter Physics; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2005; Volume 3. [Google Scholar]
- Stampfli, P. A dodecagonal quasiperiodic lattice in two dimensions. Helv. Phys. Acta 1986, 59, 1260. [Google Scholar]
- Koren, E.; Duerig, U. Superlubricity in quasicrystalline twisted bilayer graphene. Phys. Rev. B 2016, 93, 201404. [Google Scholar] [CrossRef]
- Ahn, S.J.; Moon, P.; Kim, T.-H.; Kim, H.-W.; Shin, H.-C.; Kim, E.H.; Cha, H.W.; Kahng, S.-J.; Kim, P.; Koshino, M.; et al. Dirac electrons in a dodecagonal graphene quasicrystal. Science 2018, 361, 782–786. [Google Scholar] [CrossRef]
- Santos, J.M.B.L.D.; Peres, N.M.R.; Neto, A.H.C. Graphene bilayer with a twist: Electronic structure. Phys. Rev. Lett. 2007, 99, 256802. [Google Scholar] [CrossRef]
- Kraus, Y.E.; Ringel, Z.; Zilberberg, O. Four-dimensional quantum Hall effect in a two-dimensional quasicrystal. Phys. Rev. Lett. 2013, 111, 226401. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Sun, J.; Du, X.; Tian, F.; Jiang, L. The Formation of Multiply Twinning Structure and Photoluminescence of Well-Dispersed Nanodiamonds Produced by Pulsed-Laser Irradiation. Diam. Relat. Mater. 2008, 17, 142–146. [Google Scholar] [CrossRef]
- Wentorf, R.H. The Art and Science of Growing Crystals; Gilman, J., Ed.; Wiley: New York, NY, USA, 1963; p. 493. [Google Scholar]
- Casanova, R.; Simon, B.; Turco, G. A Repeated Twin in Natural Diamond From Tortiya, Ivory Coast. Am. Mineral. 1972, 57, 1871–1873. [Google Scholar]
- Matsumoto, S.; Matsui, Y. Electron Microscopic Observation of Diamond Particles Grown from the Vapor Phase. J. Mater. Sci. 1983, 18, 1785–1793. [Google Scholar] [CrossRef]
- Palache, C. Multiple Twins of Diamond and Sphalerite. Am. Mineral. 1932, 17, 360–361. [Google Scholar]
- Carrington, W.A.; Hanssen, L.M.; Snail, K.A.; Oakes, D.B.; Butler, J.E. Diamond growth in O2 + C2H4 and O2 + C2H2 flames. Metall. Trans. A 1989, 20, 1282–1284. [Google Scholar] [CrossRef]
- Ohsumi, K.; Takase, T.; Hagiya, K.; Shimizugawa, Y.; Miyamoto, M.; Mitsuda, Y.; Ohmasa, M. Characterization of 5-μm-sized icosahedral chemical vapor deposited diamond by synchrotron x-ray diffraction with the Laue method. Rev. Sci. Instrum. 1992, 63, 1181–1184. [Google Scholar] [CrossRef]
- Miki-Yoshida, M.; Rendón, L.; Tehuacanero, S.; José-Yacamán, M. Icosahedral, decahedral and single faulted particles obtained from carbon soot. Surf. Sci. 1993, 284, L444. [Google Scholar] [CrossRef]
- Sunkara, M.K.; Angus, J.C.; Hayman, C.C.; Buck, F.A. Nucleation of diamond crystals. Carbon 1990, 28, 745. [Google Scholar] [CrossRef]
- Mani, R.C.; Sunkara, M.K. Kinetic faceting of multiply twinned diamond crystals during vapor phase synthesis. Diam. Relat. Mater. 2003, 12, 324. [Google Scholar] [CrossRef]
- Iakoubovskii, K.; Adriaenssens, G.J. Photoluminescence in CVD Diamond Films. Phys. Status Solidi (a) 1999, 172, 123–129. [Google Scholar] [CrossRef]
- Butler, J.E.; Windischmann, H. Developments in CVD-Diamond Synthesis During the Past Decade. MRS Bull. 1998, 23, 22–27. [Google Scholar] [CrossRef]
- Narayan, J.; Srivatsa, A.R.; Peters, M.; Yokota, S.; Ravi, K.V. On epitaxial growth of diamond films on (100) silicon substrates. Appl. Phys. Lett. 1988, 53, 1823. [Google Scholar] [CrossRef]
- Narayan, J.; Srivatsa, A.R.; Ravi, K.V. Mechanism of formation of <110> oriented fivefold microcrystallites in diamond films. Appl. Phys. Lett. 1989, 54, 1659. [Google Scholar] [CrossRef]
- Angus, J.C.; Buck, F.A.; Sunkara, M.K.; Groth, T.F.; Hayman, C.C.; Gat, R. Diamond growth at low pressures. MRS Bull. 1989, 14, 38. [Google Scholar] [CrossRef]
- Zeger, L.; Kaxiras, E. New Model for Icosahedral Carbon Clusters and the Structure of Collapsed Fullerite. Phys. Rev. Lett. 1993, 70, 2920. [Google Scholar] [CrossRef] [PubMed]
- Breza, J.J.; Kadlečíková, M.; Vojs, M.; Michalka, M.; Veselý, M.; Daniš, T. Diamond icosahedron on a TiN-coated steel substrate. Microelectron. J. 2004, 35, 709–712. [Google Scholar] [CrossRef]
- Lee, S.; Glotzer, S.C. Entropically engineered formation of fivefold and icosahedral twinned clusters of colloidal shapes. Nat. Commun. 2022, 13, 7362. [Google Scholar] [CrossRef]
- Song, C.W.; Lee, Y.H.; Choi, S.; Hwang, N.-M.; Kim, K.H. Formation of pentagonal dimples in icosahedral diamond crystals grown by hot filament chemical vapor deposition: Approach by non-classical crystallization. Coatings 2019, 9, 269. [Google Scholar] [CrossRef]
- Pipkin, N.J.; Davies, D.J. The crystal morphology of cyclic twins in synthetic diamond grits. Philos. Mag. A 1979, 40, 435–443. [Google Scholar] [CrossRef]
- Sunkara, M.K. Monte Carlo Simulation of Diamond Nucleation and Growth. Ph.D. Thesis, Case Western Reserve University, Cleveland, OH, USA, 1993. [Google Scholar]
- Angus, J.C.; Argoitia, A.; Gat, R.; Li, Z.; Sunkara, M.; Wang, L.; Wang, Y. Chemical vapour deposition of diamond. Philos. Trans. R. Soc. Lond. A 1993, 342, 195–208. [Google Scholar] [CrossRef]
- Wei, Q.-P.; Ma, L.; Ye, J.; Yu, Z.-M. Growth mechanism of icosahedral and other five-fold symmetric diamond crystals. Trans. Nonferrous Met. Soc. China 2015, 25, 1587–1598. [Google Scholar] [CrossRef]
- Lemmlein, G.G.; Kliya, M.O.; Chernov, A.A. Morphological Investigation of Artificial Diamond Crystals. Sov. Phys. Crystallogr. 1964, 9, 181–189. [Google Scholar]
- Nesse, W.D. Introduction to Mineralogy; Oxford University Press: New York, NY, USA, 2000; pp. 87–91. [Google Scholar]
- Klein, C.; Hurlbut, C.S., Jr. Manual of Mineralogy, 21st ed.; Wiley: New York, NY, USA, 1993; pp. 102–106. [Google Scholar]
- Kobashi, K. Diamond Films: Chemical Vapor Deposition for Oriented and Heteroepitaxial Growth; Elsevier: Amsterdam, The Netherlands, 2005; p. 9. [Google Scholar]
- Frondel, C.; Marvin, U.B. Lonsdaleite, a Hexagonal Polymorph of Diamond. Nature 1967, 214, 587–589. [Google Scholar] [CrossRef]
- Melchakova, I.A.; Oyeniyi, G.T.; Engelgardt, D.R.; Polyutov, S.P.; Avramov, P.V. Pentagonal Symmetry in Aperiodic 0D Multiply-Twinned Nano- and Mezodiamonds. J. Phys. Chem. A 2024, 128, 8591–8614. [Google Scholar] [CrossRef] [PubMed]
- Danilenko, V.V. On the History of the Discovery of Nanodiamond Synthesis. Phys. Solid State 2004, 46, 595–599. [Google Scholar] [CrossRef]
- Chernozatonskii, L.A.; Sorokin, P.B.; Kuzubov, A.A.; Sorokin, B.P.; Kvashnin, A.G.; Kvashnin, D.G.; Avramov, P.V.; Yakobson, B.I. Influence of Size Effect on the Electronic and Elastic Properties of Diamond Films with Nanometer Thickness. J. Phys. Chem. C 2011, 115, 132–136. [Google Scholar] [CrossRef]
- Kvashnin, A.G.; Avramov, P.V.; Kvashnin, D.G.; Chernozatonskii, L.A.; Sorokin, P.B. The Features of Electronic, Mechanical and Electromechanical Properties of Fluorinated Diamond Films of Nanometer Thickness. J. Phys. Chem. C 2017, 121, 28484–28489. [Google Scholar] [CrossRef]
- Dorignac, D.; Delclos, S.; Phillipp, F. Atomic structure of a complex defect configuration in synthetic diamond: A fivefold twin center connected to two high-order grain boundaries. Philos. Mag. B 2001, 81, 1879–1891. [Google Scholar] [CrossRef]
- Vaughan, D.J.; Pattrick, R.A.D. Mineralogy: An Introduction to the Study of Minerals and Crystals; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Cullity, B.D.; Stock, S.R. Elements of X-Ray Diffraction, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2001. [Google Scholar]
- Iijima, S. Fine Particles of Silicon. I. Crystal Growth of Spherical Particles of Si. Jpn. J. Appl. Phys. 1987, 26, 357. [Google Scholar] [CrossRef]
- Iijima, S. Fine Particles of Silicon. II. Decahedral Multiply-Twinned Particles. Jpn. J. Appl. Phys. 1987, 26, 365. [Google Scholar] [CrossRef]
- Saito, Y. Crystal structure and habit of silicon and germanium particles grown in argon gas. J. Cryst. Growth 1979, 47, 61–72. [Google Scholar] [CrossRef]
- Yamazoe, S.; Takano, S.; Kurashige, W.; Yokoyama, T.; Nitta, K.; Negishi, Y.; Tsukuda, T. Hierarchy of bond stiffnesses within icosahedral-based gold clusters protected by thiolates. Nat. Commun. 2016, 7, 10414. [Google Scholar] [CrossRef]
- Ino, S. Epitaxial Growth of Metals on Rocksalt Faces Cleaved in Vacuum. II. Orientation and Structure of Gold Particles Formed in Ultrahigh Vacuum. J. Phys. Soc. Jpn. 1966, 21, 346–362. [Google Scholar] [CrossRef]
- Ogawa, S.; Ino, S. Formation of Multiply-Twinned Particles in the Nucleation Stage of Film Growth. J. Vac. Sci. Technol. 1969, 6, 527–534. [Google Scholar] [CrossRef]
- Komoda, T. Study on the Structure of Evaporated Gold Particles by Means of a High Resolution Electron Microscope. Jpn. J. Appl. Phys. 1968, 7, 27–30. [Google Scholar] [CrossRef]
- Jang, F.J.-H.; Lee, E.; Park, J.; Kim, G.; Hong, S.; Kwon, Y.-U. Rational syntheses of core-shell Fex@Pt nanoparticles for the study of electrocatalytic oxygen reduction reaction. Sci. Rep. 2013, 3, 2872. [Google Scholar] [CrossRef]
- Bühler, J.; Prior, Y. Study of morphological behavior of single diamond crystals. J. Cryst. Growth 2000, 209, 779–788. [Google Scholar] [CrossRef]
- Williams, B.E.; Kong, H.S.; Glass, J.T. Electron microscopy of vapor phase deposited diamond. J. Mater. Res. 1990, 5, 801. [Google Scholar] [CrossRef]
- Avramov, P.V.; Fedorov, D.G.; Sorokin, P.B.; Chernozatonskii, L.A. Gordon, Atomic and Electronic Structure of New Hollow-Based Symmetric Families of Silicon Nanoclusters. J. Phys. Chem. C 2007, 111, 18824. [Google Scholar] [CrossRef]
- Mackay, A.L. A dense non-crystallographic packing of equal spheres. Acta Crystallogr. 1962, 15, 916–918. [Google Scholar] [CrossRef]
- Bagley, B.G. Five-Fold Pseudosymmetry. Nature 1970, 225, 1040. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Hyde, B.G. Crystal Structures I: Patterns and Symmetry; Mineralogical Society of America: Washington, DC, USA, 1996. [Google Scholar]
- Zeger, L.; Kaxiras, E. Compact Carbon Clusters with Tetrahedral Bonding and Icosahedral Symmetry. Comp. Mater. Sci. 1993, 1, 403. [Google Scholar] [CrossRef]
- Zhao, Y.; Kim, Y.-H.; Du, M.-H.; Zhang, S.B. First-Principles Prediction of Icosahedral Quantum Dots for Tetravalent Semiconductors. Phys. Rev. Lett. 2004, 93, 015502. [Google Scholar] [CrossRef]
- Savosteenko, G.; Taskaev, S.; Avramov, P. Structure and Raman spectra of exotic carbon microcrystals from meteoritic dust of Chelyabinsk superbolide. Nanomaterials 2023, 13, 73. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M. The Isoperimetric Problem for Polyhedra. Tohoku Math. J. 1935, 40, 226. [Google Scholar]
- Goldberg, M. A Class of Multi-Symmetric Polyhedra. Tohoku Math. J. 1937, 43, 104. [Google Scholar]
- Fowler, P.W.; Rogers, K.M. Spiral codes and Goldberg representations of icosahedral fullerenes and octahedral analogues. J. Chem. Inf. Comput. Sci. 2001, 41, 108. [Google Scholar] [CrossRef]
- King, R.B.; Diudea, M.V. The Chirality of Icosahedral Fullerenes: A Comparison of the Tripling (leapfrog), Quadrupling (chamfering), and Septupling (capra) Transformations. J. Math. Chem. 2006, 39, 597. [Google Scholar] [CrossRef]
- Kramers, H.A. Théorie générale de la rotation paramagnétique dans les cristaux. Proc. Acad. Sci. Amst. 1930, 33, 959–972. [Google Scholar]
- Wigner, E.P. Group Theory and Its Application to the Quantum Mechanics of Atomic Spectra; Academic Press: New York, NY, USA, 1959. [Google Scholar] [CrossRef]
- Hasan, M.Z.; Kane, C.L. Colloquium: Topological insulators. Rev. Mod. Phys. 2010, 82, 3045–3067. [Google Scholar] [CrossRef]
- Dresselhaus, M.S.; Dresselhaus, G.; Jorio, A. Group Theory: Application to the Physics of Condensed Matter; Springer: Berlin, Germany, 2008. [Google Scholar] [CrossRef]
- Bradley, C.J.; Cracknell, A.P. The Mathematical Theory of Symmetry in Solids: Representation Theory for Point Groups and Space Groups; Clarendon Press: Oxford, UK, 2009. [Google Scholar]
- Steinhardt, P.J.; Ostlund, S. (Eds.) The Physics of Quasicrystals; World Scientific: Singapore, 1987. [Google Scholar]
- Fu, L.; Kane, C.K. Topological insulators with inversion symmetry. Phys. Rev. B 2007, 76, 045302. [Google Scholar] [CrossRef]
- Ishii, F.; Mizuta, Y.; Sawahata, H.; Yamaguchi, N. First-principles study of oxide skyrmion crystal Chern insulator. APS March Meet. Abstr. 2019, 2019, H41-006. [Google Scholar]
- Hubert, H.; Devouard, B.; Garvie, L.A.J.; O’Keeffe, M.; Buseck, P.R.; Petuskey, W.T.; McMillan, P.F. Icosahedral packing of B12 icosahedra in boron suboxide (B6O). Nature 1998, 391, 376–378. [Google Scholar] [CrossRef]
- Naslain, R. Crystal Chemistry of Boron and of Some Boron-Rich Phases; Preparation of Boron Modifications. In Boron and Refractory Boride; Matkovitch, V.I., Ed.; Springer: Berlin/Heidelberg, Germany, 1977; pp. 139–202. [Google Scholar]
- Ignatiev, N.K. On a practical method for finding dense packings of n-dimensional spheres. Sib. Mat. Zhurnal 1964, 5, 815–819. [Google Scholar]
- Doraiswamy, N.; Marks, L.D. Preferred structures in small particles. Philos. Mag. B 1995, 71, 291–310. [Google Scholar] [CrossRef]
- Gammons, C.H. Hydrothermal synthesis of gold grains with apparent five-fold symmetry. Can. Mineral. 1996, 34, 1–8. [Google Scholar]
- Uyeda, R. Morphology of Crystals; Sunagawa, I., Ed.; Terra Scientific Publishing Company: Tokyo, Japan, 1987. [Google Scholar]
- Ohashi, W.; Spaepen, F. Stable Ga–Mg–Zn quasiperiodic crystals with pentagonal dodecahedral solidification morphology. Nature 1987, 330, 555–556. [Google Scholar] [CrossRef]
- Dubost, B.; Lang, J.M.; Tanaka, M.; Sainfort, P.; Audier, M. Large AlCuLi single quasicrystals with triacontahedral solidification morphology. Nature 1986, 324, 48–50. [Google Scholar] [CrossRef]
- Lundström, T. Structure and Bulk Modulus of High-Strength Boron Compounds. J. Solid State Chem. 1997, 133, 88–92. [Google Scholar] [CrossRef]
- Vast, N.; Baroni, S.; Zerah, G.; Besson, J.M.; Polian, A.; Grimsditch, M.; Chervin, J.C. Lattice Dynamics of Icosahedral α-Boron under Pressure. Phys. Rev. Lett. 1997, 78, 693–696. [Google Scholar] [CrossRef]
- Kobayashi, M.; Higashi, I.; Brodhag, C.; Thévenot, F. Structure of B6O boron-suboxide by Rietveld refinement. J. Mater. Sci. 1993, 28, 2129–2134. [Google Scholar] [CrossRef]
- Lundström, T.; Andreev, Y.G. Superhard boron-rich borides and studies of the B–C–N system. Mater. Sci. Eng. A 1996, 209, 16–22. [Google Scholar] [CrossRef]
- Bolmgren, H.; Lundström, T.; Okada, S. Boron-Rich Solids; Emin, D., Aselage, T., Beckel, C.L., Howard, I.A., Wood, C., Eds.; AIP Conf. Proc. No. 231; American Institute of Physics: New York, NY, USA, 1991; pp. 197–200. [Google Scholar]
- Donohue, J. The Structures of the Elements; Wiley: New York, NY, USA, 1974. [Google Scholar] [CrossRef]
- Hoard, J.L.; Sullenger, D.B.; Kennard, C.H.L.; Hughes, R.E. The structure analysis of β-rhombohedral boron. J. Solid State Chem. 1970, 1, 268–277. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Avramov, P.V.; Tian, H.; Li, L. Aperiodicity in Low Dimensions. Materials 2026, 19, 446. https://doi.org/10.3390/ma19030446
Avramov PV, Tian H, Li L. Aperiodicity in Low Dimensions. Materials. 2026; 19(3):446. https://doi.org/10.3390/ma19030446
Chicago/Turabian StyleAvramov, Pavel V., Hao Tian, and Li Li. 2026. "Aperiodicity in Low Dimensions" Materials 19, no. 3: 446. https://doi.org/10.3390/ma19030446
APA StyleAvramov, P. V., Tian, H., & Li, L. (2026). Aperiodicity in Low Dimensions. Materials, 19(3), 446. https://doi.org/10.3390/ma19030446








