A Dual-Dynamic Crosslinked Polysaccharide-Based Hydrogel Loaded with Exosomes for Promoting Diabetic Wound Healing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Oxidized Sodium Alginate
2.3. Preparation of 4-Formylphenylboronic Acid Grafted Carboxymethyl Chitosan
2.4. Preparation of Dual-Dynamic Crosslinked Hydrogel Loaded with Exosomes
2.5. Rheological Properties Test
2.6. Injectability and Self-Healing Property Evaluation
2.7. Adhesion Performance Test
2.8. Degradation In Vitro Test
2.9. Measurement of Exosomes Release Profile
2.10. Measurement of Antioxidant Properties
2.11. Hemocompatibility Test
2.12. Cytotoxicity Assay
2.13. Scratch Wound Healing Assays
2.14. Cell Proliferative Behaviors
2.15. Establishment of a Diabetic Skin Wound Model
2.16. Histochemical and Immunofluorescence Staining
2.17. Statistical Analysis
3. Results and Discussion
3.1. Formation and Property of the Dual Dynamic Cross-Linked Hydrogel
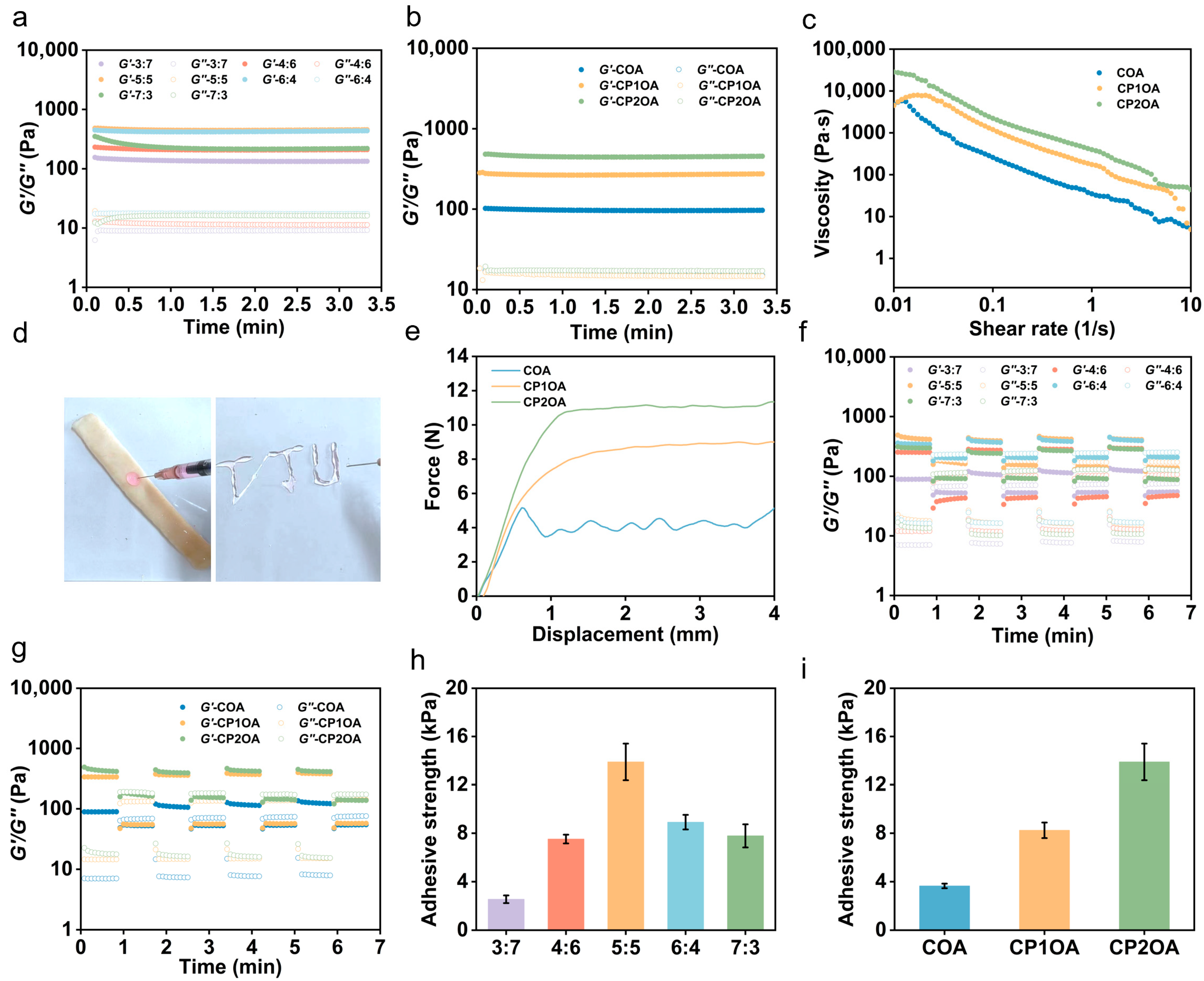
3.2. The Rheological and Adhesive Properties
3.3. ROS Responsiveness and Exos Release Behavior
3.3.1. ROS Responsive Properties
3.3.2. Exos Release Behavior
3.3.3. ROS Scavenging Ability
3.4. In Vitro Biological Properties
3.5. Repair Effects of CPOA@Exos on Diabetic Wound In Vivo
3.6. Modulation of the Diabetic Wound Microenvironment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef]
- Shaw, J.E.; Sicree, R.A.; Zimmet, P.Z. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 2010, 87, 4–14. [Google Scholar] [CrossRef]
- Singh, N.; Armstrong, D.G.; Lipsky, B.A. Preventing foot ulcers in patients with diabetes. JAMA 2005, 293, 217–228. [Google Scholar] [CrossRef]
- Sun, B.K.; Siprashvili, Z.; Khavari, P.A. Advances in skin grafting and treatment of cutaneous wounds. Science 2014, 346, 941–945. [Google Scholar] [CrossRef]
- Zhu, S.; Zhao, B.; Li, M.; Wang, H.; Zhu, J.; Li, Q.; Gao, H.; Feng, Q.; Cao, X. Microenvironment responsive nanocomposite hydrogel with NIR photothermal therapy, vascularization and anti-inflammation for diabetic infected wound healing. Bioact. Mater. 2023, 26, 306–320. [Google Scholar] [CrossRef] [PubMed]
- Hu, N.; Cai, Z.; Jiang, X.; Wang, C.; Tang, T.; Xu, T.; Chen, H.; Li, X.; Du, X.; Cui, W. Hypoxia-pretreated ADSC-derived exosome-embedded hydrogels promote angiogenesis and accelerate diabetic wound healing. Acta Biomater. 2023, 157, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Du, C.; Song, P.; Chen, T.; Rui, S.; Armstrong, D.G.; Deng, W. The Role of Oxidative Stress and Antioxidants in Diabetic Wound Healing. Oxidative Med. Cell. Longev. 2021, 2021, 8852759. [Google Scholar] [CrossRef]
- Lohmann, N.; Schirmer, L.; Atallah, P.; Wandel, E.; Ferrer, R.A.; Werner, C.; Simon, J.C.; Franz, S.; Freudenberg, U. Glycosaminoglycan-based hydrogels capture inflammatory chemokines and rescue defective wound healing in mice. Sci. Transl. Med. 2017, 9, eaai9044. [Google Scholar] [CrossRef]
- Monaghan, M.G.; Borah, R.; Thomsen, C.; Browne, S. Thou shall not heal: Overcoming the non-healing behaviour of diabetic foot ulcers by engineering the inflammatory microenvironment. Adv. Drug Deliv. Rev. 2023, 203, 115120. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, F.; Javaid, S.; Tariq, M.; Mustafa, M. Molecular immunological mechanisms of impaired wound healing in diabetic foot ulcers (DFU), current therapeutic strategies and future directions. Int. Immunopharmacol. 2024, 139, 112713. [Google Scholar] [CrossRef]
- Jin, N.; Wang, Z.; Tang, X.; Jin, N.; Wang, X. Promoting Diabetic Wound Healing through a Hydrogel-Based Cascade Regulation Strategy of Fibroblast-Macrophage. Adv. Healthc. Mater. 2024, 13, 2400526. [Google Scholar] [CrossRef]
- Tian, D.M.; Wan, H.H.; Chen, J.R.; Ye, Y.B.; He, Y.; Liu, Y.; Tang, L.Y.; He, Z.Y.; Liu, K.Z.; Gao, C.J.; et al. In-situ formed elastin-based hydrogels enhance wound healing via promoting innate immune cells recruitment and angiogenesis. Mater. Today Bio. 2022, 15, 100300. [Google Scholar] [CrossRef]
- Villarreal-Leal, R.A.; Healey, G.D.; Corradetti, B. Biomimetic immunomodulation strategies for effective tissue repair and restoration. Adv. Drug Deliv. Rev. 2021, 179, 113913. [Google Scholar] [CrossRef] [PubMed]
- Hesketh, M.; Sahin, K.B.; West, Z.E.; Murray, R.Z. Macrophage phenotypes regulate scar formation and chronic wound healing. Int. J. Mol. Sci. 2017, 18, 1545. [Google Scholar] [CrossRef]
- Boateng, J.; Catanzano, O. Advanced Therapeutic Dressings for Effective Wound Healing—A Review. J. Pharm. Sci. 2015, 104, 3653–3680. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.F.; Bártolo, P.J. Traditional Therapies for Skin Wound Healing. Adv. Wound Care 2016, 5, 208–229. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.D.; Xiao, M.T. Polysaccharide-based hydrogel with photothermal effect for accelerating wound healing. Carbohydr. Polym. 2023, 299, 120228. [Google Scholar] [CrossRef]
- Shao, M.; Shi, Z.; Zhang, X.; Zhai, B.; Sun, J. Synthesis and Properties of Biodegradable Hydrogel Based on Polysaccharide Wound Dressing. Materials 2023, 16, 1358. [Google Scholar] [CrossRef]
- Liang, L.; Zhang, H.; Yao, F.; Li, J. Programmable tissue-adhesive hydrogels with temporal and spatial selectivity. Chem. Soc. Rev. 2025, 54, 12043–12079. [Google Scholar] [CrossRef]
- Wang, X.; Song, R.; Johnson, M.; A, S.; He, Z.; Milne, C.; Wang, X.; Lara-Sáez, I.; Xu, Q.; Wang, W. An Injectable Chitosan-Based Self-Healable Hydrogel System as an Antibacterial Wound Dressing. Materials 2021, 14, 5956. [Google Scholar] [CrossRef]
- Zhao, J.; Zhu, H.; Xu, T.; Xu, S.; Wang, Y.; Hou, Y.; Zhuo, M.; Du, K.; Su, Y.; Han, X.; et al. Self-Healing Oxidized Dextran/Sodium Alginate Hydrogel Dressing with Hemostatic Activity Speeds Up Wound Healing in Burn Injuries. ACS Appl. Mater. Interfaces 2025, 17, 2940–2951. [Google Scholar] [CrossRef]
- Liu, X.; Hu, J.; Hu, Y.; Liu, Y.; Wei, Y.; Huang, D. Multifunctional injectable oxidized sodium alginate/carboxymethyl chitosan hydrogel for rapid hemostasis. Colloids Surf. B Biointerfaces 2025, 245, 114346. [Google Scholar] [CrossRef]
- Xie, M.; Zeng, Y.; Wu, H.; Wang, S.; Zhao, J. Multifunctional carboxymethyl chitosan/oxidized dextran/sodium alginate hydrogels as dressing for hemostasis and closure of infected wounds. Int. J. Biol. Macromol. 2022, 219, 1337–1350. [Google Scholar] [CrossRef]
- Yang, Y.; Zhong, S.; Meng, F.; Cui, X. Multi-Functional hydrogels to promote diabetic wound Healing: A review. Chem. Eng. J. 2024, 497, 154855. [Google Scholar] [CrossRef]
- Gan, Z.; Xiao, Z.; Zhang, Z.; Li, Y.; Liu, C.; Chen, X.; Liu, Y.; Wu, D.; Liu, C.; Shuai, X.; et al. Stiffness-tuned and ROS-sensitive hydrogel incorporating complement C5a receptor antagonist modulates antibacterial activity of macrophages for periodontitis treatment. Bioact. Mater. 2023, 25, 347–359. [Google Scholar] [CrossRef]
- Marco-Dufort, B.; Tibbitt, M.W. Design of moldable hydrogels for biomedical applications using dynamic covalent boronic esters. Mater. Today Chem. 2019, 12, 16–33. [Google Scholar] [CrossRef]
- Miao, Y.; Wei, J.; Chen, X.; Shi, J.; Zhang, L.; Wang, L.; Yang, J.; Ma, L.; Duan, J. Evaluation of living bacterial therapy assisted by pH/reactive oxygen species dual-responsive sodium alginate-based hydrogel for wound infections. Int. J. Biol. Macromol. 2024, 271, 132536. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Li, M.; Mu, L.; Huang, Y.; Liang, Y.; Guo, B. Photothermal/Photodynamic Synergistic Antibacterial Hydrogel Dressing with pH/Glucose Dual Responsive Pirfenidone Release for Diabetic Foot Ulcers. Adv. Funct. Mater. 2025, 35, 2416205. [Google Scholar] [CrossRef]
- Li, J.; Du, C.; Yang, X.; Yao, Y.; Qin, D.; Meng, F.; Yang, S.; Tan, Y.; Chen, X.; Jiang, W.; et al. Instantaneous Self-Healing Chitosan Hydrogels with Enhanced Drug Leakage Resistance for Infected Stretchable Wounds Healing. Small 2025, 21, 2409641. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Liang, B.; Tian, J.; Wu, J. Anti-inflammation biomaterial platforms for chronic wound healing. Biomater. Sci. 2021, 9, 4388–4409. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Xie, Z.; Hao, H.; Tong, C.; Cheng, Y.; Liu, J.; Pang, Y.; Si, Y.; Guo, Y.; Zang, L.; Mu, Y.; et al. Human umbilical cord-derived mesenchymal stem cells elicit macrophages into an anti-inflammatory phenotype to alleviate insulin resistance in type 2 diabetic rats. Stem Cells 2016, 34, 627–639. [Google Scholar] [CrossRef]
- Zhao, H.; Shang, Q.; Pan, Z.; Bai, Y.; Li, Z.; Zhang, H.; Zhang, Q.; Guo, C.; Zhang, L.; Wang, Q. Exosomes From Adipose-Derived Stem Cells Attenuate Adipose Inflammation and Obesity Through Polarizing M2 Macrophages and Beiging in White Adipose Tissue. Diabetes 2017, 67, 235–247. [Google Scholar] [CrossRef]
- Gao, Y.; Yuan, Z.; Yuan, X.; Wan, Z.; Yu, Y.; Zhan, Q.; Zhao, Y.; Han, J.; Huang, J.; Xiong, C.; et al. Bioinspired porous microspheres for sustained hypoxic exosomes release and vascularized bone regeneration. Bioact. Mater. 2022, 14, 377–388. [Google Scholar] [CrossRef]
- Shiekh, P.A.; Singh, A.; Kumar, A. Exosome laden oxygen releasing antioxidant and antibacterial cryogel wound dressing OxOBand alleviate diabetic and infectious wound healing. Biomaterials 2020, 249, 120020. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.-C.; Guo, S.-C.; Li, M.; Ke, Q.-F.; Guo, Y.-P.; Zhang, C.-Q. Chitosan Wound Dressings Incorporating Exosomes Derived from MicroRNA-126-Overexpressing Synovium Mesenchymal Stem Cells Provide Sustained Release of Exosomes and Heal Full-Thickness Skin Defects in a Diabetic Rat Model. Stem Cells Transl. Med. 2017, 6, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Bai, G.; Gu, W.; Niu, C.; Feng, Y.; Wei, Z.; Chen, K.; Guo, X. Boronic ester bonds hydrogel with temperature and pH responsiveness for controlled release fertilizer. Ind. Crops Prod. 2024, 221, 119336. [Google Scholar] [CrossRef]
- Liu, W.; Ge, F.; Du, P.; Yan, X.; Jiang, H. High-performance room-temperature phosphorescence materials based on arylboronic acids activated by a dual hydrogen-bonded network. Chem. Eng. J. 2025, 522, 167561. [Google Scholar] [CrossRef]
- Ma, W.; Dong, W.; Zhao, S.; Du, T.; Wang, Y.; Yao, J.; Liu, Z.; Sun, D.; Zhang, M. An injectable adhesive antibacterial hydrogel wound dressing for infected skin wounds. Biomater. Adv. 2022, 134, 112584. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Jin, M.; Feng, Z.; Zhang, J.; Rong, Y.; Zhang, Y.; Zhang, S.; Yu, Y.; Yang, H.; Wang, T. Injectable decellularzied extracellular matrix hydrogel derived from human umbilical cord: A novel perspective to deal with refractory wound via medical wastes. Mater. Des. 2023, 229, 111877. [Google Scholar] [CrossRef]
- Jing, Y.; Huang, T.; Zhao, B.; Zhao, L.; Zhang, N.; Zhang, K.; Wang, K.; Wang, J.; Hua, J.; Tu, Q. A ROS/glucose stimulated-responsive ADSCs-derived exosomes-release hydrogel system for diabetic wound healing. Chem. Eng. J. 2024, 487, 150561. [Google Scholar] [CrossRef]
- Xie, H.; Wang, Z.; Wang, R.; Chen, Q.; Yu, A.; Lu, A. Self-Healing, Injectable Hydrogel Dressing for Monitoring and Therapy of Diabetic Wound. Adv. Funct. Mater. 2024, 34, 2401209. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lin, D.; Li, Z.; Hao, J.; Xu, X.; Li, X.; Feng, Y.; Lu, X.; Yao, F.; Zhang, H.; Li, J. A Dual-Dynamic Crosslinked Polysaccharide-Based Hydrogel Loaded with Exosomes for Promoting Diabetic Wound Healing. Materials 2026, 19, 445. https://doi.org/10.3390/ma19020445
Lin D, Li Z, Hao J, Xu X, Li X, Feng Y, Lu X, Yao F, Zhang H, Li J. A Dual-Dynamic Crosslinked Polysaccharide-Based Hydrogel Loaded with Exosomes for Promoting Diabetic Wound Healing. Materials. 2026; 19(2):445. https://doi.org/10.3390/ma19020445
Chicago/Turabian StyleLin, Ding, Zhenhao Li, Jianying Hao, Xiaobo Xu, Xiuqiang Li, Yuan Feng, Xiaochen Lu, Fanglian Yao, Hong Zhang, and Junjie Li. 2026. "A Dual-Dynamic Crosslinked Polysaccharide-Based Hydrogel Loaded with Exosomes for Promoting Diabetic Wound Healing" Materials 19, no. 2: 445. https://doi.org/10.3390/ma19020445
APA StyleLin, D., Li, Z., Hao, J., Xu, X., Li, X., Feng, Y., Lu, X., Yao, F., Zhang, H., & Li, J. (2026). A Dual-Dynamic Crosslinked Polysaccharide-Based Hydrogel Loaded with Exosomes for Promoting Diabetic Wound Healing. Materials, 19(2), 445. https://doi.org/10.3390/ma19020445





