1. Introduction
For decades, polymeric materials have been used in a plethora of application fields. Some of these fields are directly concerned with fire regulations. Indeed, the inherent flammability of polymers is one of the main issues for the use of this kind of material. In order to improve the fire behavior, the polymer industry is working on materials containing flame retardants (FR) to obtain formulations exhibiting low flammability. Various systems, reported in several reviews [
1,
2,
3], have been used to improve the fire reaction of the different types of polymers and containing mainly phosphorus compounds [
4,
5,
6,
7], but also organo-modified layered silicates (OMLS) [
8,
9,
10] and more recently biobased fire retardants [
11,
12,
13]. This wide variety of systems reflects the complexity to find an efficient combination for each kind of polymer. For example, some flame retardants like phosphorus ones are particularly devoted to oxygen and nitrogen containing polymers but they are not relevant for polyolefins.
Poly(lactide) (PLA) is a biobased aliphatic polyester obtained from agricultural plants which is also renewable and biodegradable [
14]. PLA is a thermoplastic well-used in food-packaging technologies [
15] and in biomedical engineering [
16]. The increasing use of this polymer is due to its eco-friendly and biocompatible character. It is one of the most used biopolymers [
17] and is easily processed because of its good melt properties. However, PLA has a low toughness, ductility and a poor thermal stability [
18]. Although PLA is mainly used in packaging, the production of durable goods in PLA is developing, for example in the areas of toys or domestic appliances. Moreover, PLA has an important potential in the field of additive manufacturing and especially for processing by FDM (Fused Deposit Modeling) [
19]. Therefore, flame retarded PLA needs to be developed in order to meet fire safety standards for uses in applications where fire regulations operate.
Polyurethanes are also commonly used in a number of applications often requiring flame retardancy. Segmented ones, consisting of soft and hard segments, are used for various applications such as coatings [
20], seals [
21] or shape memory materials [
22]. As an engineering polymer, polyurethane elastomer (PUE) has been widely used in industrial sectors like aerospace, medicine, automobile manufacturing or shockproof buffer materials due to its high performance in terms of excellent abrasion resistance, chemical resistance and high elastic properties [
23,
24,
25]. PUE can also be injected, molded or extruded. However, the high flammability of polyurethane with melt dripping [
26,
27,
28] is a major limitation of this material. Improving the flame retardancy of PUE is then an important challenge to extend its applications. Recently, the flame retardant properties of PUE have been improved using different kinds of additives [
29,
30,
31,
32,
33].
In this context, intumescent fire retardant (FR) systems based on ammonium polyphosphate (APP) have been developed for many years and have been shown to improve the fire performance of both polymers [
34,
35,
36,
37]. Poly(lactide) and thermoplastic polyurethane have been selected and compared as polymer matrices with the same additives in this study also because only polyurethane contains nitrogen in its chemical composition, which can modify the reactivity towards the different FR systems used.
It was reported that these FR systems could play a role in both condensed and gas phases [
38]. They could reduce the fraction of flammable materials and help in forming a protective char due to a strong dehydrating agent generated by polyphosphoric acid. This char is a residue containing phosphorus which can limit oxygen access to prevent the spread of flammable gases and carbon gasification [
39,
40]. However, a composite with only APP as a flame retardant cannot effectively yield enough fire performance. Hence, it is necessary to combine APP with other flame retardants to achieve synergistic effects on flame retardancy [
41,
42]. In the last 20 years, polymer nanocomposites have attracted much interest. Nanoparticles, such as organomodified layered silicates (OMLS) or carbon nanotubes were used successfully to improve fire reaction properties such as heat release rate, by decreasing the peak heat release rate (pHRR), reinforcing the char residues or reducing the mass loss rate [
43,
44,
45]. Only a few works have reported the use of sepiolite in flame retardant systems, in particular with APP [
46,
47]. This filler belongs to the structural family of 2:1 phyllosilicates with the following formula (Si
12Mg
8O
30)(OH)
4(OH
2)
4·8H
2O [
48,
49]. Sepiolite exhibits a microfibrous morphology with tunnels that grow up in the fiber direction. During the combustion, it has been shown by Lewin et al. [
50] that OMLS could migrate to the surface and consequently, it can be expected that sepiolite could also behave similarly, reinforcing the char layer. Moreover, regarding other kinds of nanoparticles, recent studies have shown that silica could accumulate on the surface during material burning and lead to a significant effect on the reduction of heat release rate of a polymer [
51,
52].
New biobased fire retardants are emerging in order to lead to eco-friendlier composite materials. Lignin is one of the more promising biobased components of fire retardants systems [
53]. It is a byproduct of the paper industry [
54]. Lignin is also a biobased copolymer mainly composed of phenolic groups. The basic units are coumaryl, coniferyl and sinapyl alcohols [
55]. Lignin has a good thermal stability and contributes to flame retardancy in composite materials by promoting the formation of char [
37,
56,
57,
58]. Few papers have been published on the fire behavior of composites and polymer blends containing lignin. Ferry et al. [
56] used unmodified and modified lignin modification in order to improve PBS (polybutylene succinate) fire behavior. Other studies have explored the effect of lignin on the thermal properties of polymers such as poly(ethylene terephtalate) [
59] and for polypropylene [
60]. Lignin has also been studied as an additive in biopolymers, like in polyhydroxybutyrate (PHB) [
61,
62].
This work aims to compare the effectiveness of sepiolite and lignin as potential FR synergistic agents of APP in PLA and PUE. The interest of such compounds was investigated for different mass ratios with APP ranging from 1.5 to 5.67, APP being always the major component. A global loading of 20 wt% of FR components was selected in accordance with the loadings of intumescent FR currently used in research about the fire retardancy of these polymers. The greatest attention was focused on the mechanisms of fire retardancy induced by the presence of both additives in each polymer as well as on their influence on thermal stability. Fire behavior was assessed either at the microscopic scale using combustion microcalorimetry or at the macroscopic scale using cone calorimeter. Moreover, investigations on cone calorimeter residues were carried out to attempt to account for the interactions occurring between the different components during thermal degradation.
2. Materials and Methods
2.1. Polymers and FR Components
The poly(lactide) (PLA) used in this study was supplied by NatureWorks (Minnonka, MN, USA). The polymer is the grade 3251D with a glass transition temperature (Tg) of 55–60 °C, a melt temperature (Tm) of 188–210 °C and a density of 1.24 g/cm3. Polyurethane, supplied from Courbis (Romans-sur-Isère, France), was prepared from polytetramethylene ether glycol (Mn ≈ 2000 g·mol−1), pre-polymer terminated 4,4′-diphenylmethane diisocyanate (pMDI) and 1,4 butanediol (BDO) as chains extender. APP (Exolit AP423) was provided from Clariant (Muttenz, Swiss). Its phosphorus percentage is 31 wt% and the median diameter of particles is 9 µm. Sepiolite (S9 Pangel, Tolsa Company, Madrid, Spain) was purchased from Lavollée S.A. (Levallois-Paris, France). The selected lignin is a commercial low-sulfonated alkaline lignin supplied by Sigma ALDRICH (Darmstadt, Germany). The median diameter of lignin particle is around 50 µm. All chemicals were used without any purification.
2.2. Sample Preparation
The PLA composites were prepared using a twin-screw extruder (1200 mm) Clextral (Firminy, France). The process conditions were the same for all samples. The temperature profile of the barrel was from 80 to 210 °C and the screw rotational speed was set at 250 rpm. The molding process was carried out using an injection molding press (Krauss-Maffei, Munich, Germany) with a closing strength of 50 tons. The blends were injection-molded into plates (100 × 100 × 4 mm3). The temperature profile was 40 °C for the mold and 200 °C for the screw.
For PUE, polyol (68.6 g) and pMDI (106.2 g) were held at room temperature 12 h before vacuum drying at 80 °C for 6 h in a vacuum oven. Then, polyol and pMDI were placed in a plastic beaker and stirred for 5 min, after that 9.2 g of BDO was added. The mixture was then cast in a metallic mold heated at 115 °C to obtain four square sheets of size 100 × 100 × 4 mm3. The mold was maintained at 115 °C for 1 h 30 min, after which the samples were placed at 115 °C for 15 h. To obtain PUE composites, fillers were added with polyol and pMDI.
Table 1 shows all the formulations and the components ratios for the different compounds. A total amount of 20 wt% for the additions was kept constant. It should be noted that a complete substitution of APP in polymers by each additive was not possible or desirable, leading to processing difficulties or very poor thermal stability due to hydrolytic degradation. Hence, investigations on synergistic effects will be only assessed regarding partial substitutions.
2.3. Characterization of Thermal Stability
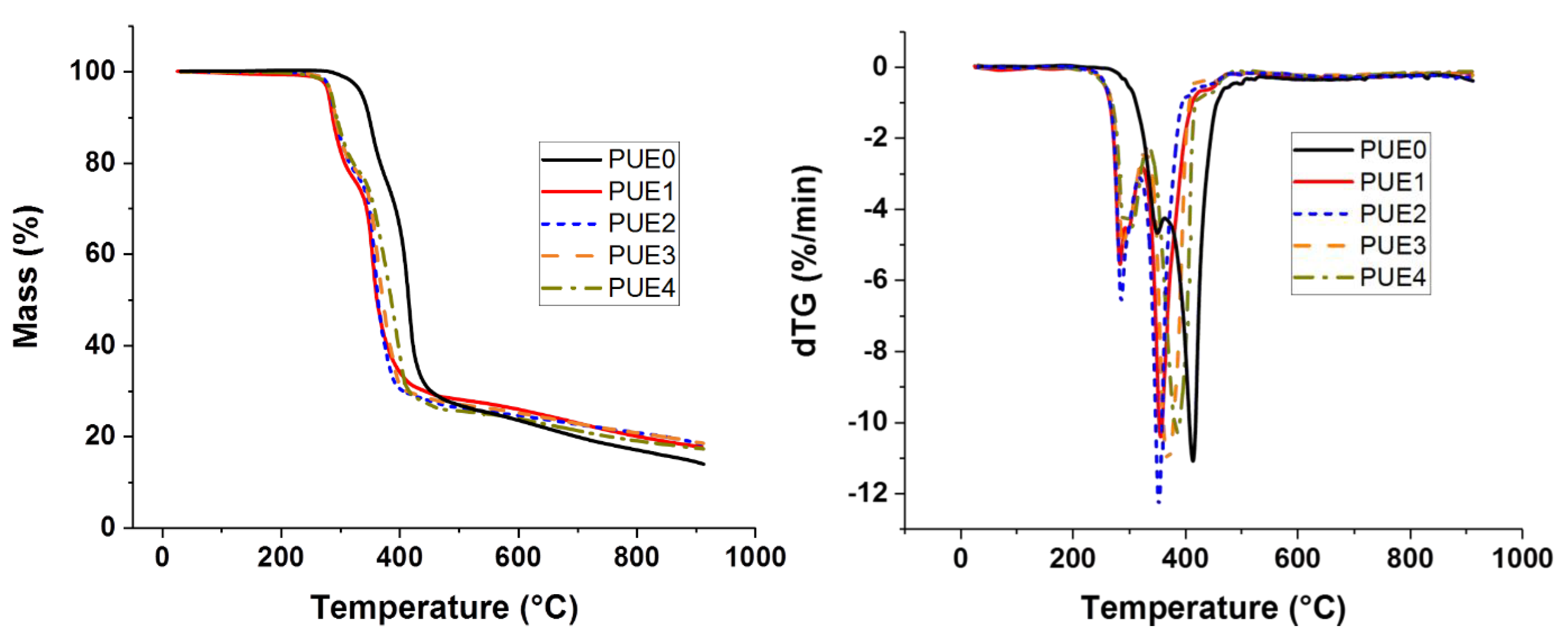
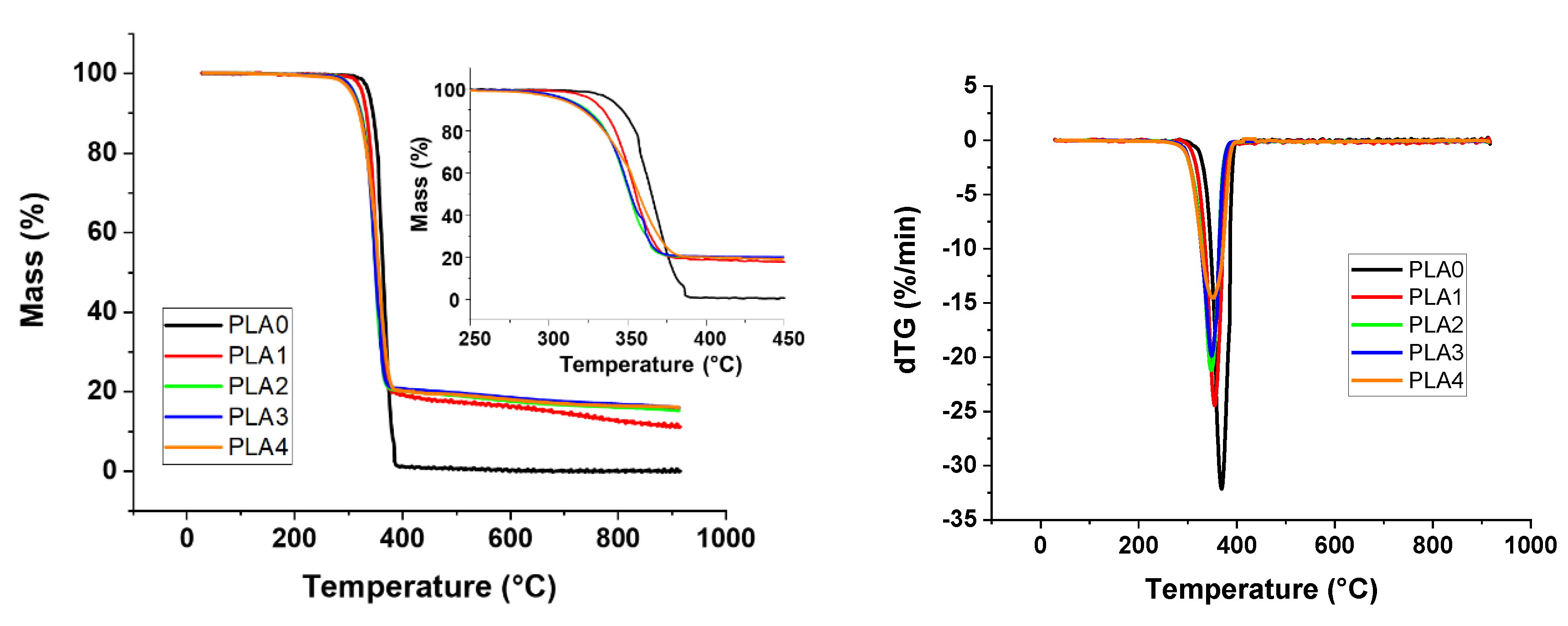
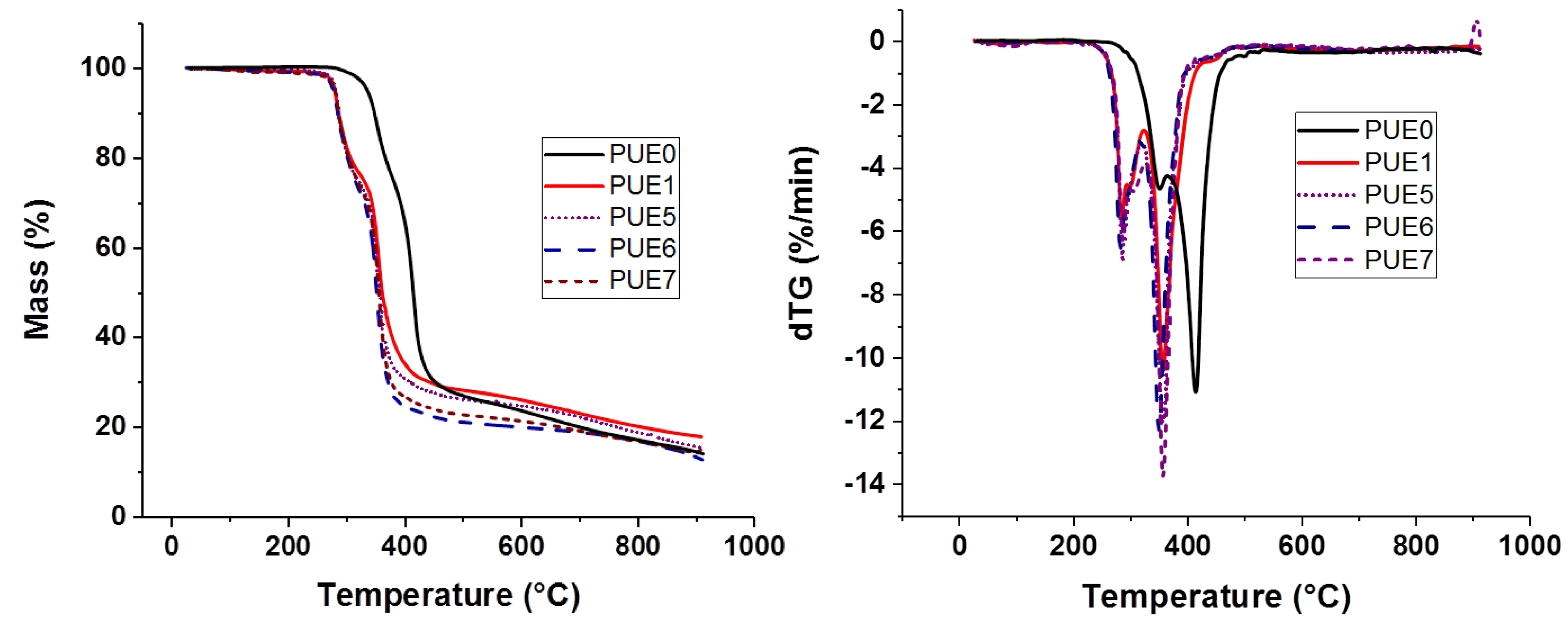
Thermal stability characterizations of samples were performed using a Thermogravimetric Analysis (TGA) apparatus (SETSYS evolution, Setaram, Caluire, France). Experiments were performed in the temperature range from 30 to 900 °C, at heating rate of 10 °C/min under nitrogen atmosphere (40 mL/min). The sample weight was approximately 12 mg. Initial decomposition temperatures (TON-SET) were determined at 5% of weight loss and the maximum degradation rate temperature was measured from the derivates of thermogravimetric curve (dTG) peak maximum. The weight residue left at 900 °C was determined for all the samples.
Thermal degradation of the different nanocomposites was investigated using a Pyrolysis Combustion Flow Calorimeter (PCFC) (Fire Testing Technology, East Grinstead, UK). PCFC was firstly developed by Lyon and Walters [
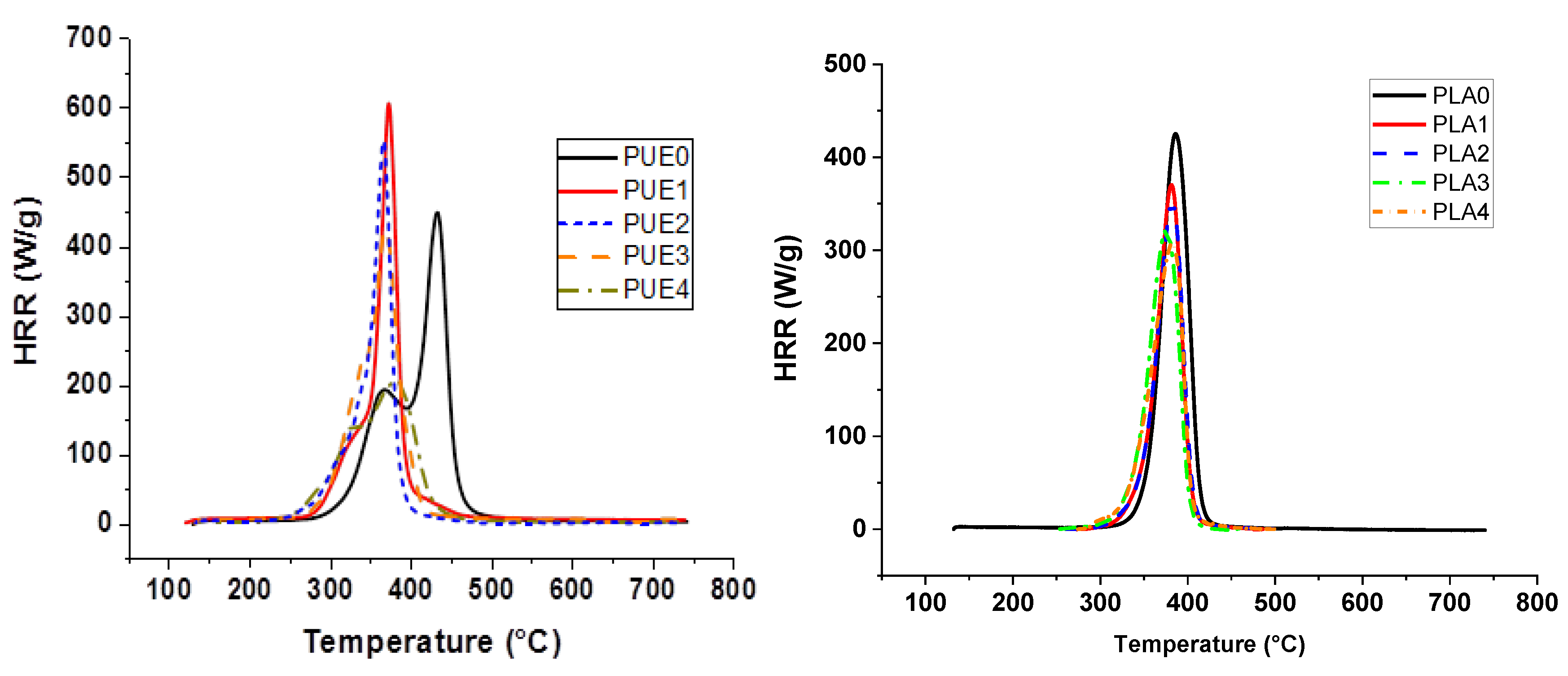
63] to study the thermal degradation of samples at the microscopic scale. Sample weights were around 5 mg. Samples were pyrolyzed at 1 °C/s, then the volatile thermal degradation products were swept from a pyrolysis chamber by an inert gas and combined with excess oxygen in a tubular furnace at flame temperatures, to force complete combustion. Heat release rate (HRR) was measured as a function of temperature. This enables the determination of the peak value of HRR as well as the Total Heat Release (THR, area below the HRR curve). The results were averaged with an error of less than 5%, while the error on temperature of the peak value of HRR and measurement is less than 1%.
2.4. Characterization of Fire Behavior
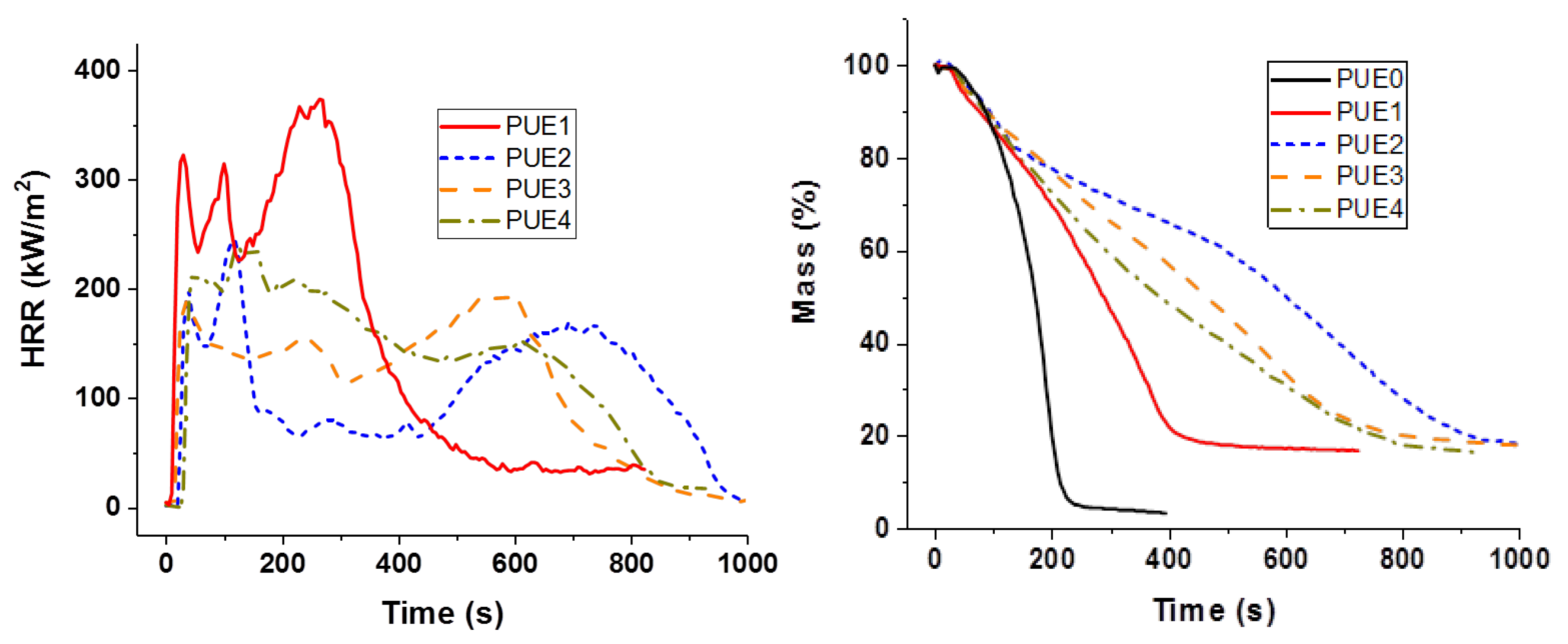
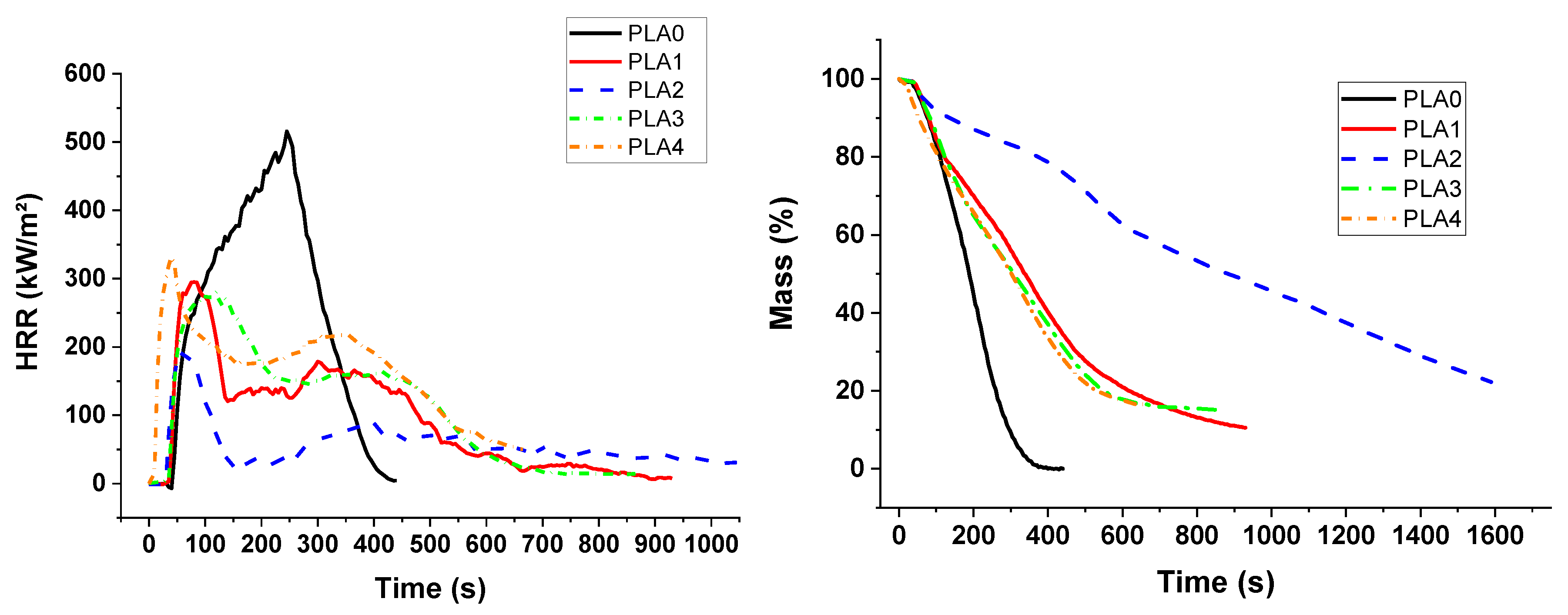
A Fire Testing Technology cone calorimeter was used to evaluate fire reaction properties. The 100 × 100 × 4 mm3 samples were exposed to a radiant heat flux of 50 kW/m2. The distance between the cone and the sample was 25 mm. The air flow was 24 L/s. Heat release rate was measured as a function of time. Then, time to ignition (TTI), Total Heat Released (THR), peak of HRR (pHRR), Maximum Average Rate of Heat Emission maximum value of the average of HRR as function of time (MARHE), and quantity of residue were determined.
For each composition three samples were tested and mean values were reported.
2.5. Characterization of Microstructures and Cone Calorimeter Residues
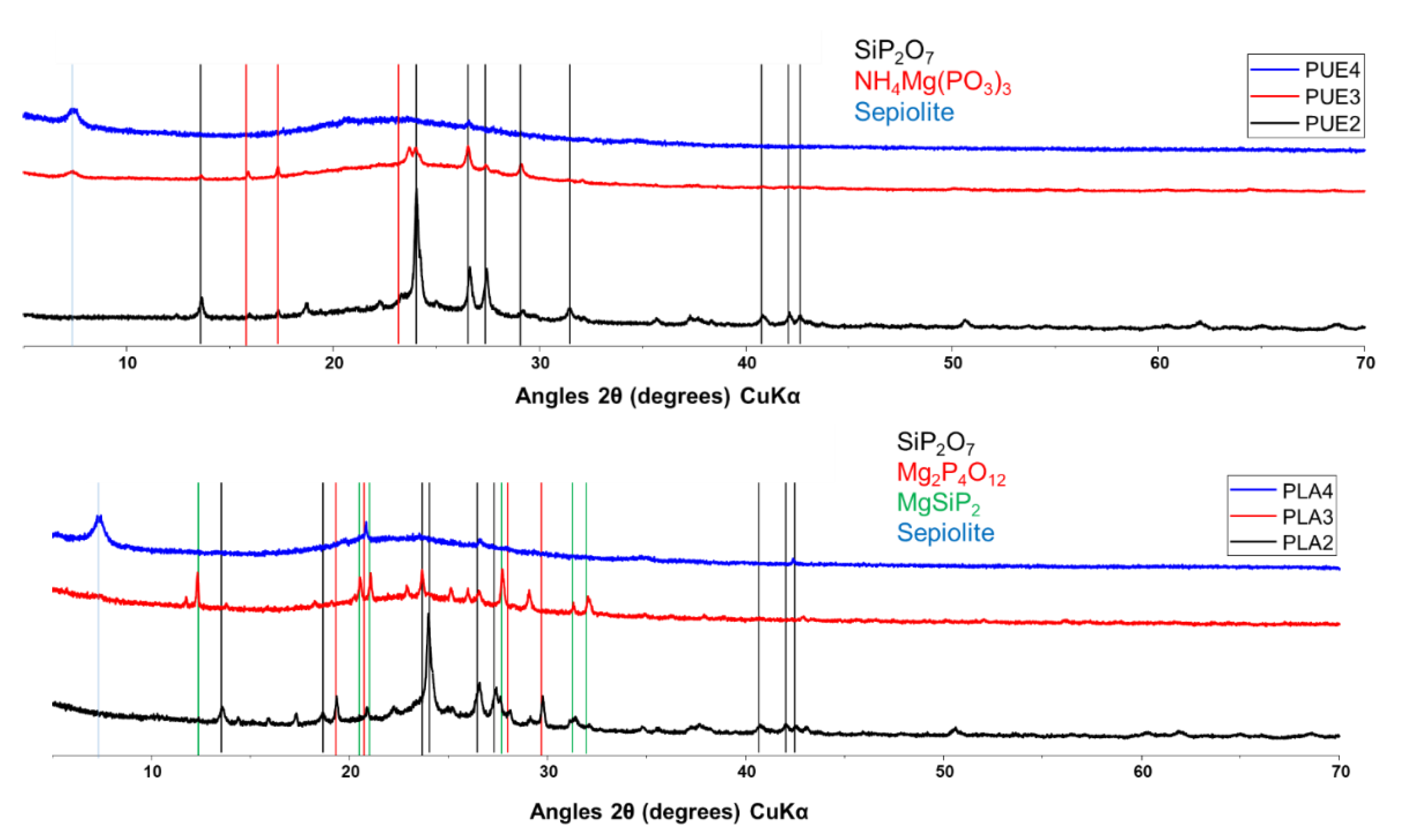
The cone calorimeter residues were analyzed by X-Ray Diffraction (XRD) in order to investigate the degradation mechanisms of the various materials studied. The XRD patterns were obtained using an X-ray diffractometer BRUKER D8 Advance. A scanning angle of 2θ was from 5° to 70° with an X-ray beam (Cu Kα, λ = 1.54 Å).
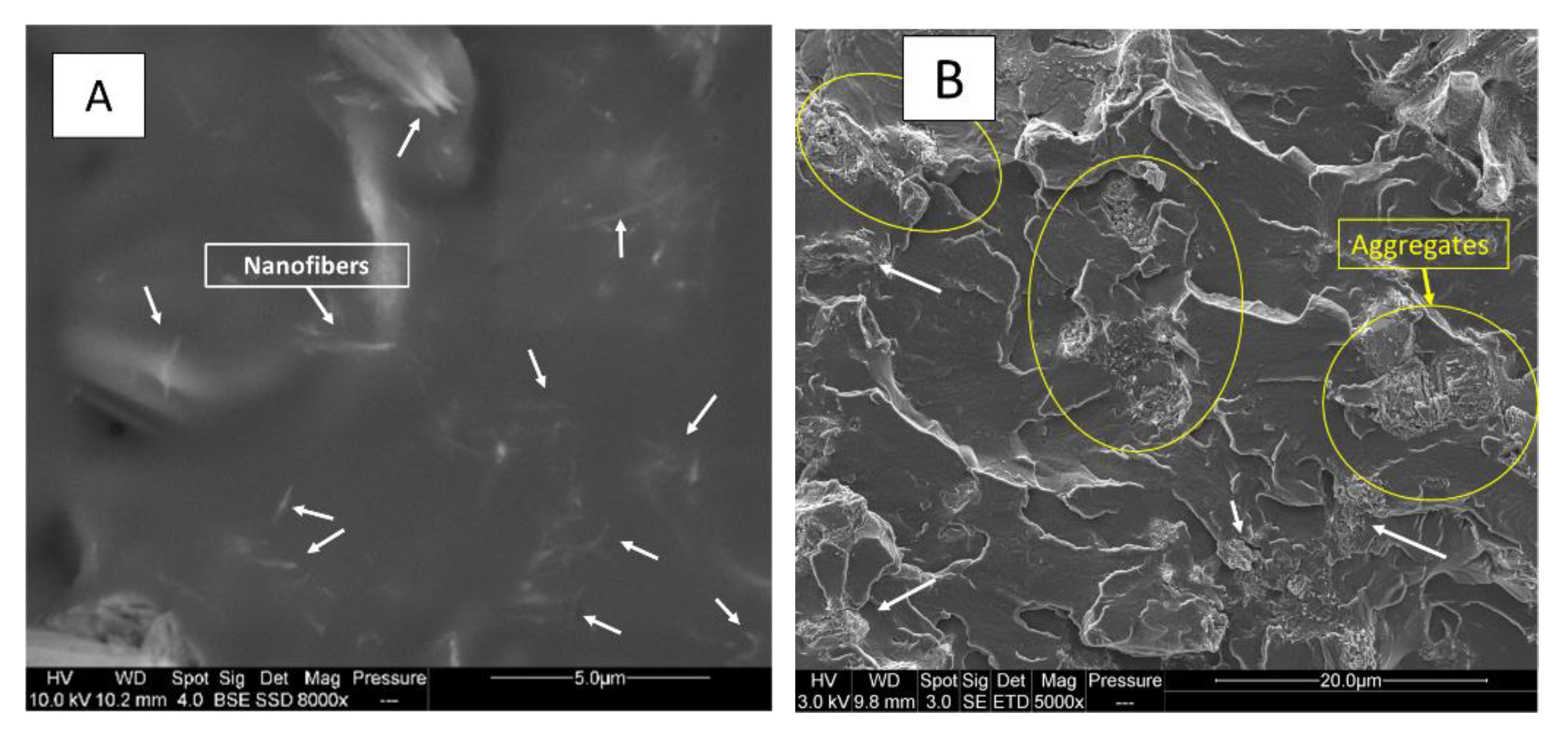
A Scanning Electron Microscopy (SEM) Quanta 200 FEG (FEI Company, Hillsboro, OR, USA) in environmental mode equipped with an X-Max 80N SDD detector was used to observe the samples and the dispersion of fillers inside the polymer matrix. Voltages of 3 kV for PLA, 10 kV for PUE and fillers were respectively used.
2.6. FTIR-ATR Spectroscopy
A FTIR-ATR spectrometer (Vertex 70 FT MIR from Bruker, Billerica, MA, USA) was used to assess the nature of the residue and to determine if the char formed had protected the composite during cone calorimetry testing. The resolution was 4 cm−1 and 32 scans for the background and 32 scans for the spectra acquisition were conducted. The spectral range was from 4000 cm−1 to 400 cm−1 and was analyzed with OPUS software, provided with the spectrometer. The analysis was performed directly on the crystal.
5. Conclusions
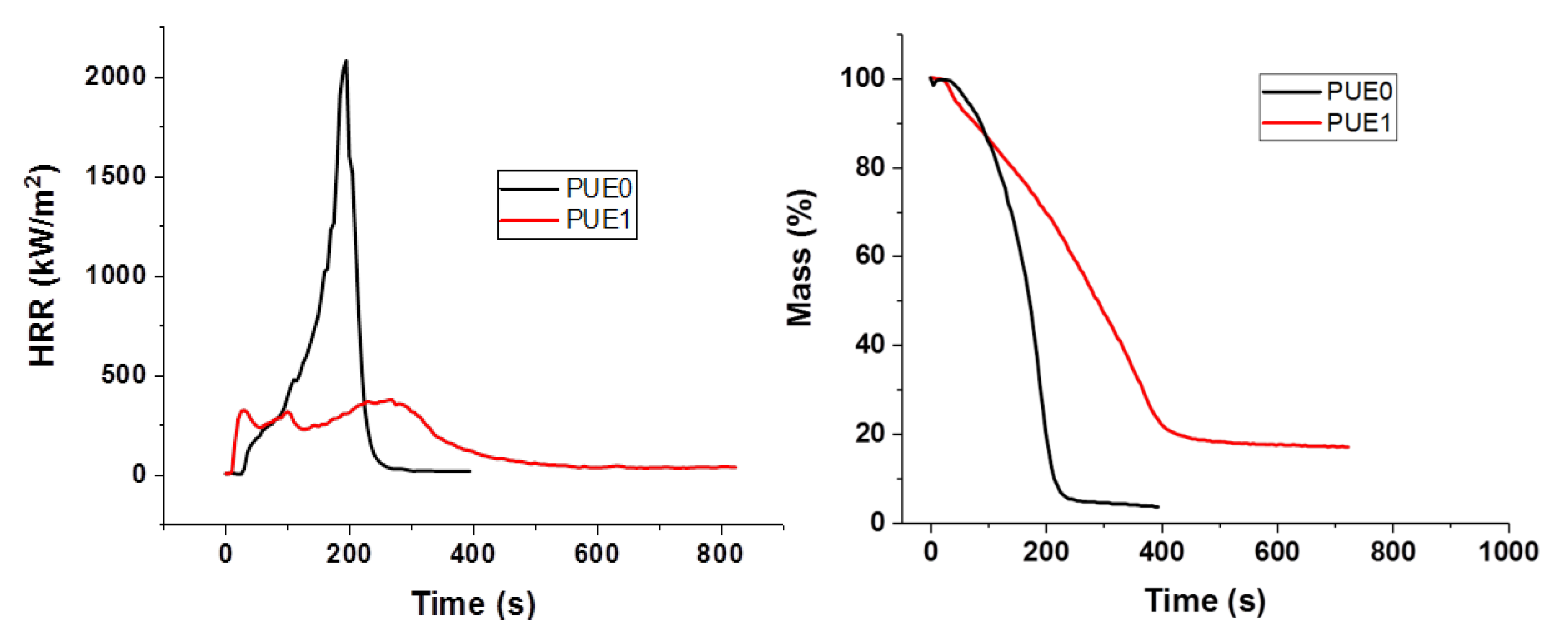
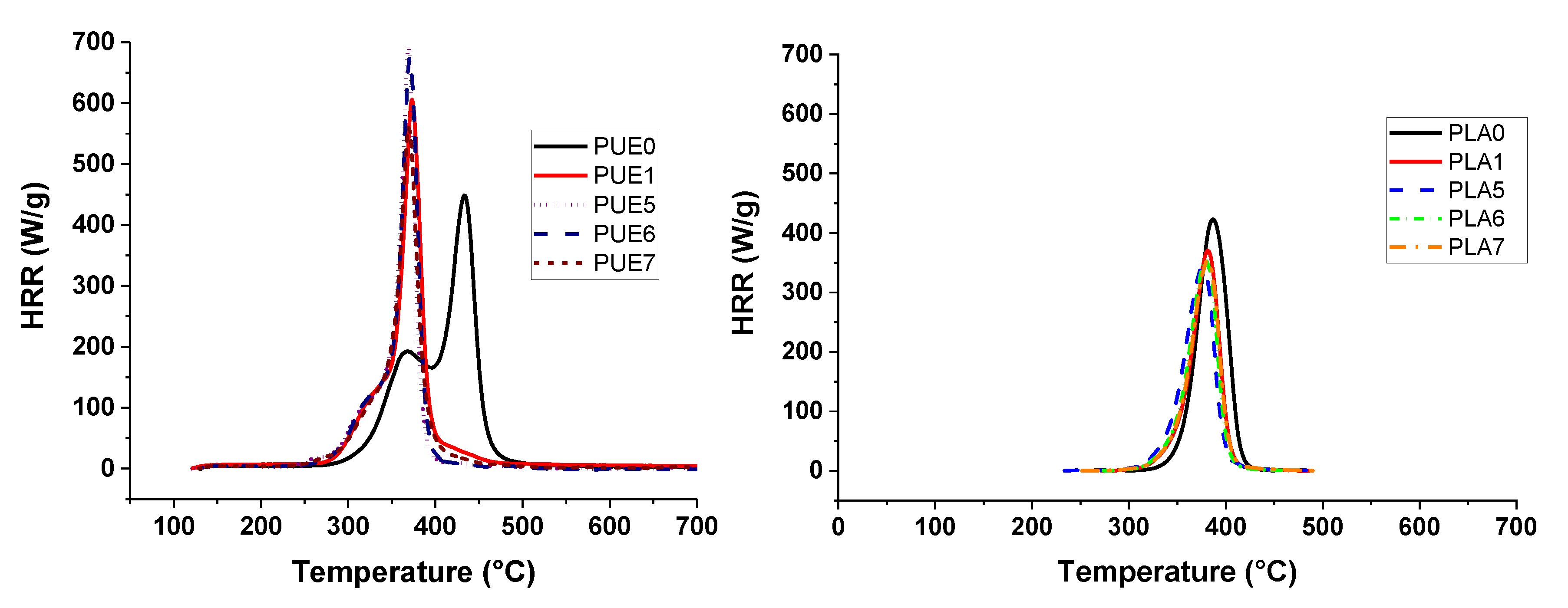
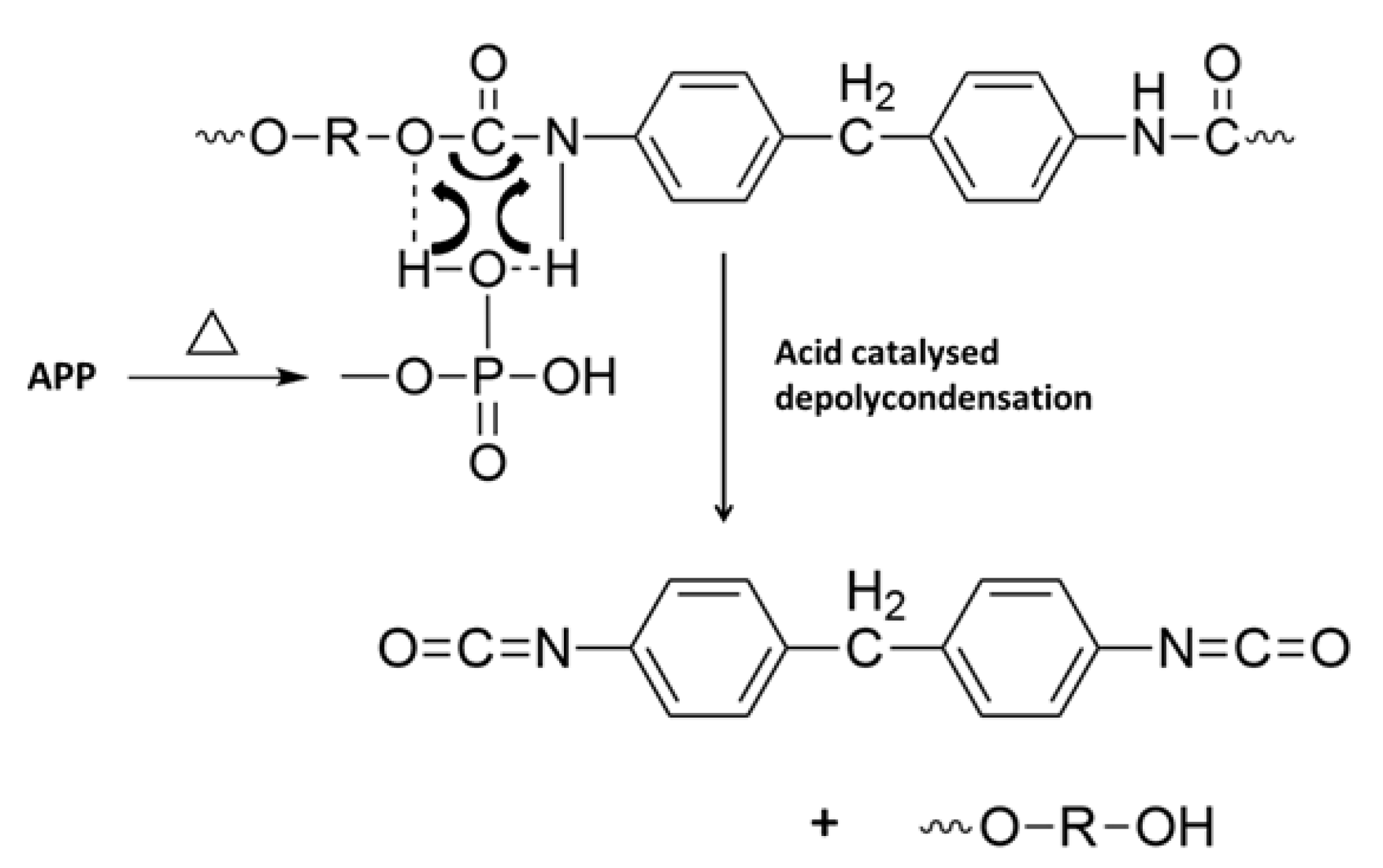
All the flame retardants systems based on APP combined with sepiolite or lignin allowed the fire performance of PUE and PLA to be improved or at least maintained. The use of APP alone led to an improvement of fire parameters in terms of reduced pHRR, THR and MARHE and mass loss rate. However, the action of APP appeared different for both polymers. TGA and PCFC results clearly showed that in the case of PUE, the thermal stability was more strongly impaired than in the case of PLA, in order to promote a charred and intumescent structure. Moreover, for PUE, PCFC showed that the acid catalyzed de-polycondensation caused by APP entails a dramatic release of energy at a lower temperature than was noted for PUE alone.
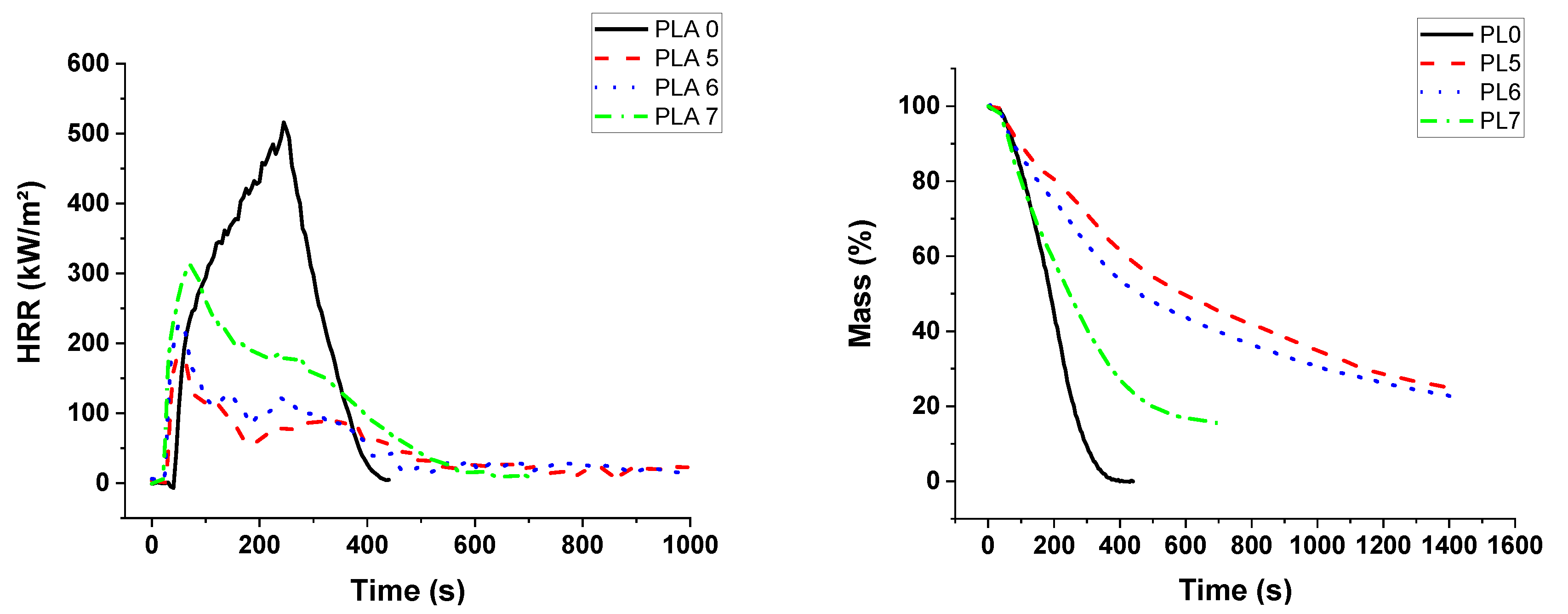
In the cone calorimeter, the reduction in time to ignition for PUE was also due to the detrimental effect of APP on thermal stability in order to promote a charred and intumescent structure, which was not so significant in the case of PLA. Hence, the decrease in pHRR for PUE was more important (82%) than for PLA (43%) for which no significant intumescent structure was found.
Sepiolite was introduced in order to create a barrier effect through a direct reinforcement of the char or through the formation of silicon phosphate and other phosphorus compounds by reaction with APP. A synergistic effect on the fire parameters in the cone calorimeter was noticed for all compositions with PUE. An optimum APP/sepiolite ratio of three was highlighted and it can be concluded that there was no competition between the formation of the intumescent structure from PUE and APP and the formation of silicon phosphate and other phosphorus components from sepiolite and APP. For PLA, the synergistic effect was also highlighted but only for one composition and a higher APP/sepiolite mass ratio (close to six). From TGA curves, it can be noted that sepiolite, for which a barrier effect was expected due to possible restrictions of macromolecular chains, led in fact to a reduction of thermal stability. It can be ascribed to water release and de-hydroxylation of hydroxyl groups leading to PLA hydrolysis. Hence, the interest of sepiolite towards formation of protective structure through silicon phosphate or other phosphorus compounds formation was here limited due to a detrimental effect on thermal stability.
The combination of lignin with APP was expected to impart a stronger charring effect. In the case of PLA, a reduction in pHRR was noticed in PCFC testing without a significant reduction in time for the peak value. From cone calorimetry, synergistic effects on fire performance were observed for an APP/lignin ratio up to three. Conversely, for PUE no synergistic effect was noticed and the amount of residue formed during cone calorimetry tests was slightly reduced. This lack of performance of lignin in PUE can be explained by an increased catalytic effect on de-polycondensation since PCFC tests showed a strong increase in pHRR at the same temperature as PUE with APP alone. Hence, the char promoting effect of lignin was completely offset by its detrimental effect on the thermal stability of PUE, resulting in a strong release of volatile combustibles during the cone calorimeter test.