Na+/H+ Exchangers Involve in Regulating the pH-Sensitive Ion Channels in Mouse Sperm
Abstract
1. Introduction
2. Results
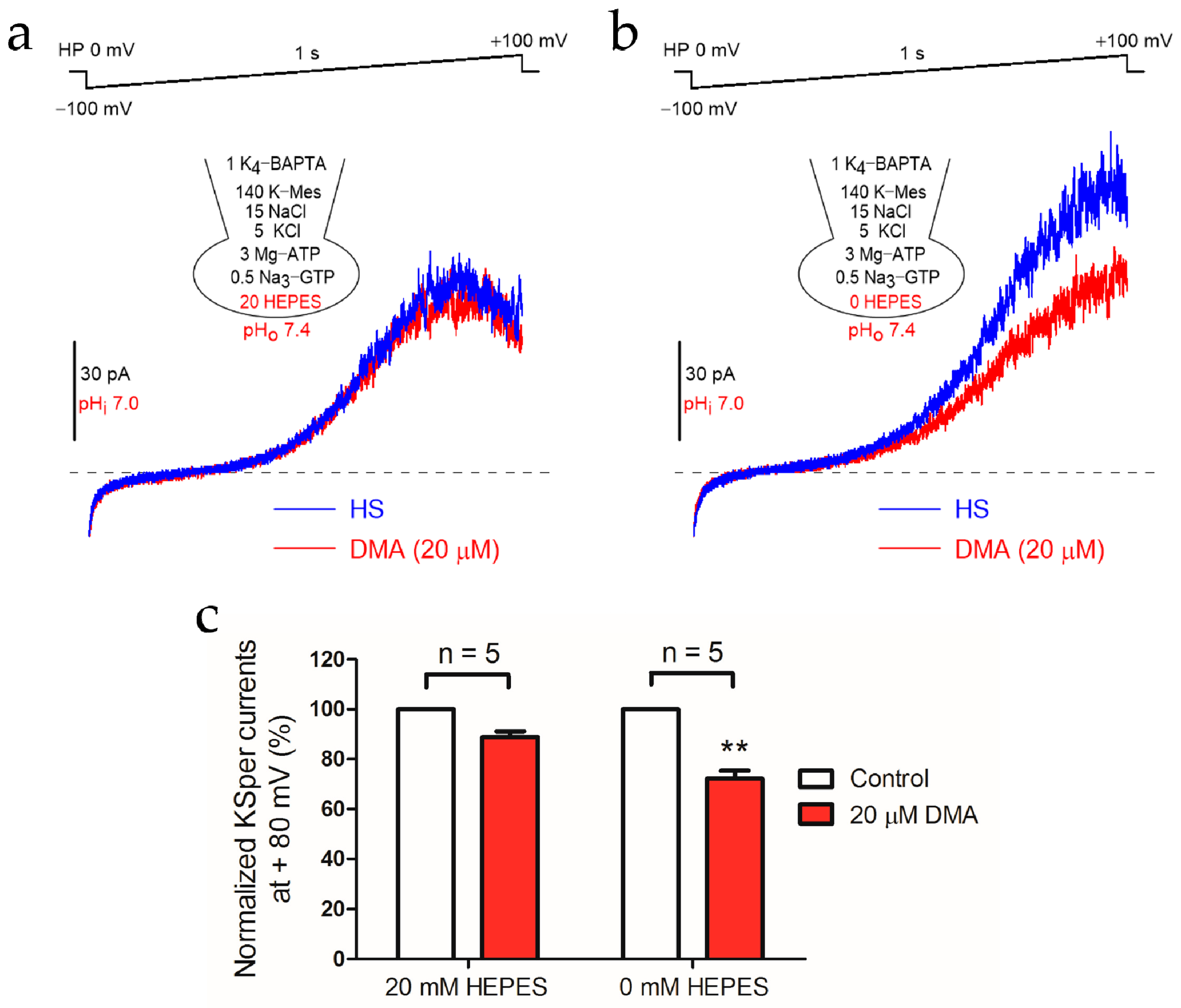
2.1. DMA Impaired the Activation of KSper Channel via Inhibiting NHEs Function
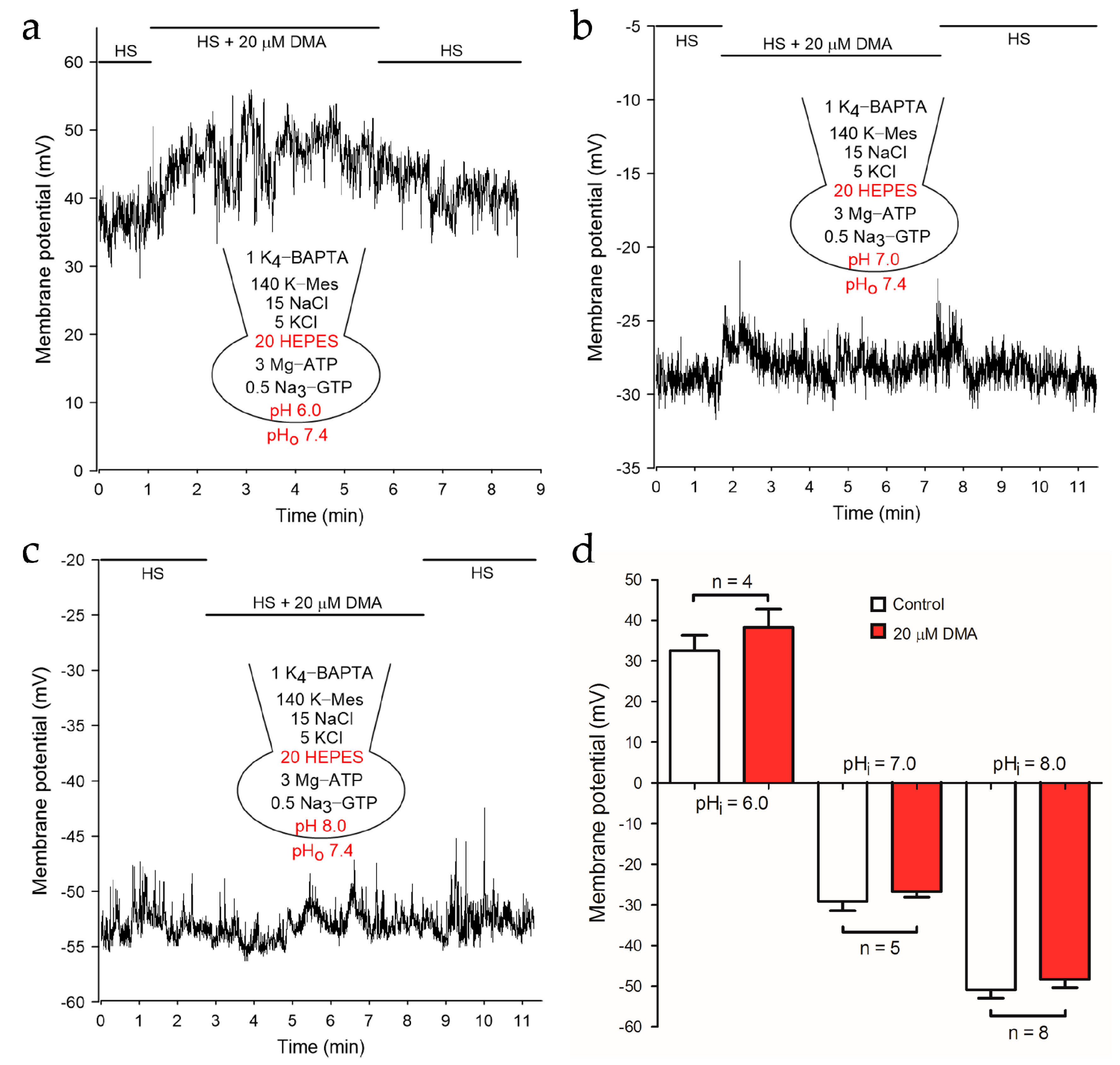
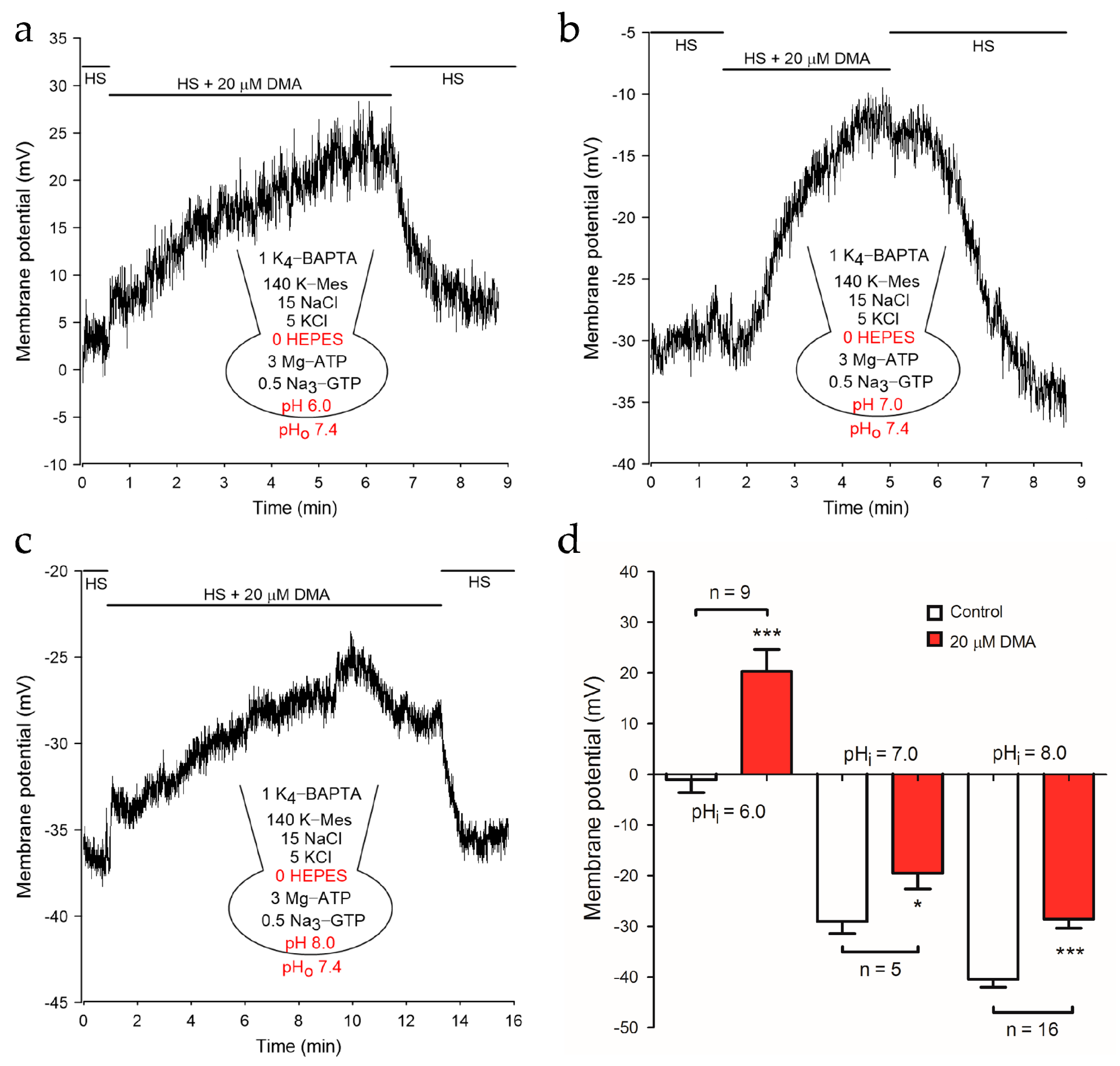
2.2. NHEs Inhibition Resulted in the Depolarization of Membrane Potential
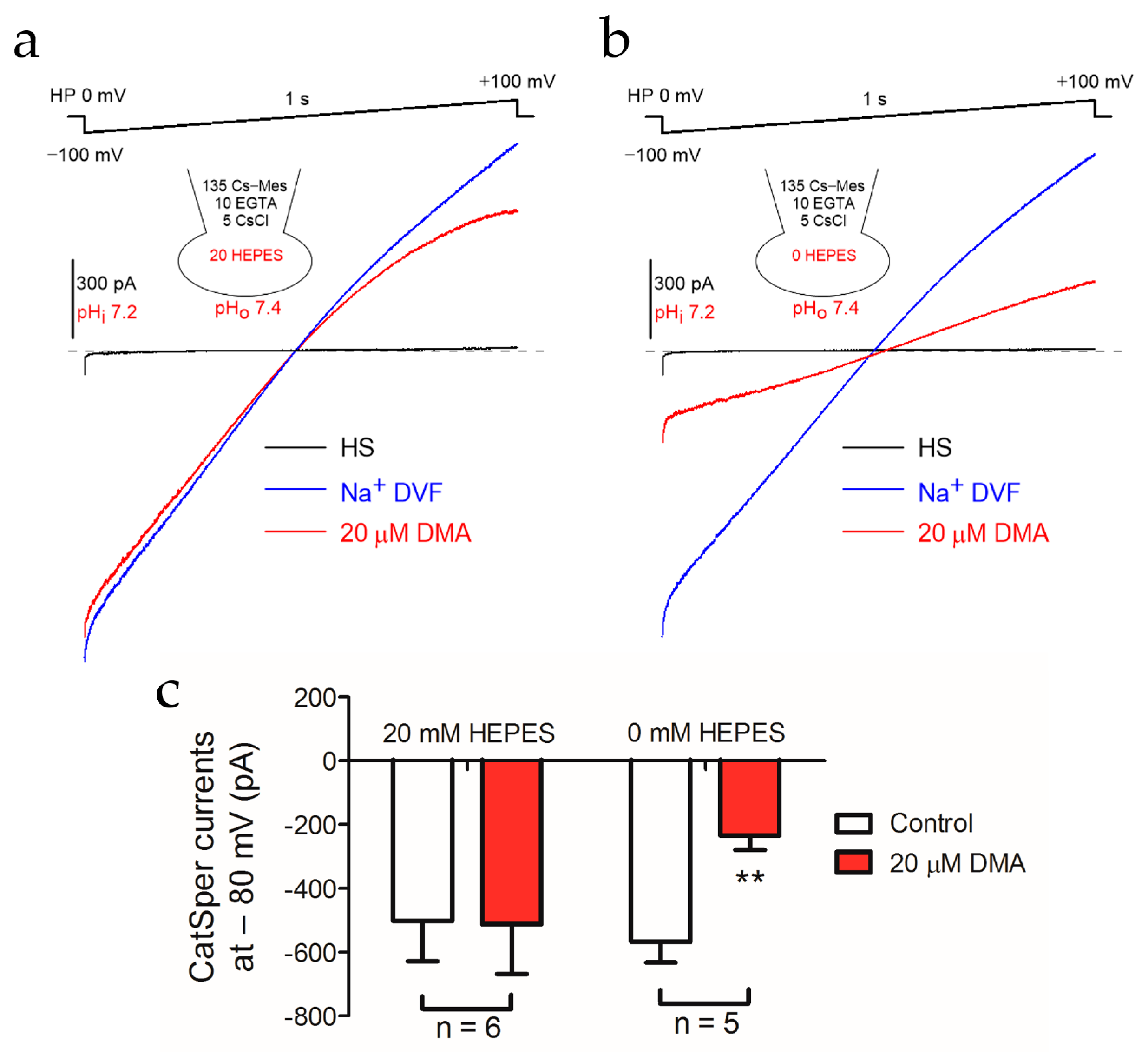
2.3. Alkalization-Activated CatSper Channel and NHEs Were Functional Coupled
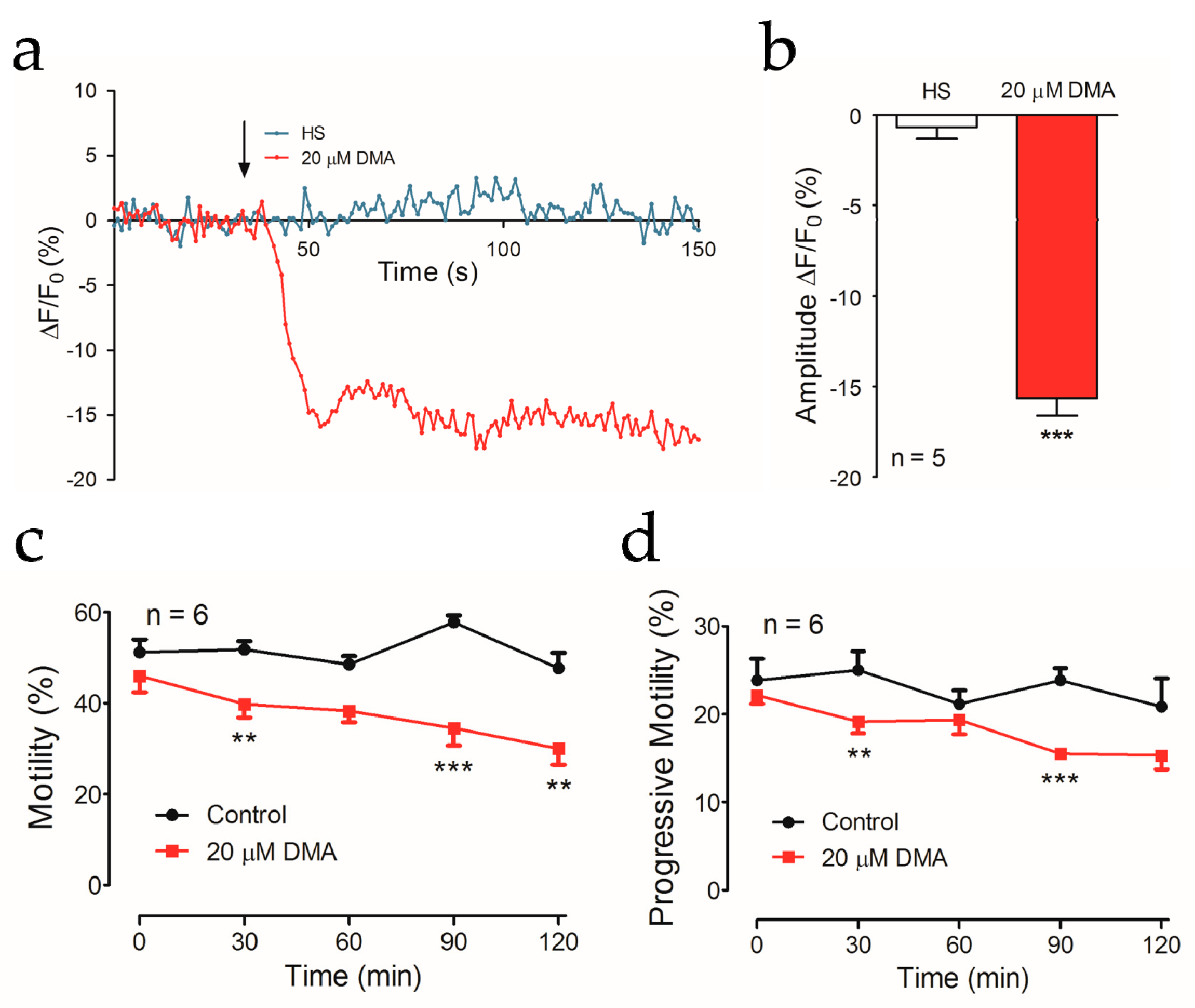
2.4. DMA Treatment Reduced Intracellular Ca2+ Concentration and Impaired Sperm Motility
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Animal Treatment and Sperm Preparation
4.3. Electrophysiology
4.4. Determination of Sperm Intracellular pH and Ca2+
4.5. Assessment of Sperm Motility
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALH | Amplitude of lateral head displacement |
| BAPTA | 1,2-bis(2-aminophenoxy)ethane-N,N,N’,N’-tetraacetic acid |
| BCECF | BCECF-AM |
| BCF | Beat cross frequency |
| BSA | Bovine serum albumin |
| CatSper | Ca2+ channel of sperm |
| DMA | 5-(N, N-dimethyl)-amiloride |
| EFCAB9 | EF-hand calcium-binding domain-containing protein 9 |
| EGTA | Ethylene glycol tetraacetic acid |
| EIPA | 5-(N-ethyl-N-isopropyl)-amiloride |
| HEPES | 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid |
| HP | Holding potential |
| HS | High saline solution |
| Hv1 | Voltage-gated proton channel |
| KSper | Potassium channel of sperm |
| LIN | Linearity |
| Mes | Methanesulfonic acid |
| Na+DVF | Sodium-based divalent free solution |
| NHA | Na+/H+ antiporter |
| NHEs | Na+/H+ exchangers |
| NH4Cl | Ammonium chloride |
| pHi | Intracellular pH |
| SEM | Standard error of the mean |
| sNHE | Sperm-specific NHE |
| VAP | Average path velocity |
| VCL | Curvilinear velocity |
| VSL | Straight-line velocity |
References
- Navarro, B.; Kirichok, Y.; Clapham, D.E. KSper, a pH-sensitive K+ current that controls sperm membrane potential. Proc. Natl. Acad. Sci. USA 2007, 104, 7688–7692. [Google Scholar] [CrossRef]
- Brenker, C.; Zhou, Y.; Müller, A.; Echeverry, F.A.; Trötschel, C.; Poetsch, A.; Xia, X.M.; Bönigk, W.; Lingle, C.J.; Kaupp, U.B.; et al. The Ca2+-activated K+ current of human sperm is mediated by Slo3. Elife 2014, 3, e01438. [Google Scholar] [CrossRef]
- Mannowetz, N.; Naidoo, N.M.; Choo, S.A.; Smith, J.F.; Lishko, P.V. Slo1 is the principal potassium channel of human spermatozoa. Elife 2013, 2, e01009. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Navarro, B.; Perez, G.; Jackson, A.C.; Hsu, S.; Shi, Q.; Tilly, J.L.; Clapham, D.E. A sperm ion channel required for sperm motility and male fertility. Nature 2001, 413, 603–609. [Google Scholar] [CrossRef]
- Kirichok, Y.; Navarro, B.; Clapham, D.E. Whole-cell patch-clamp measurements of spermatozoa reveal an alkaline-activated Ca2+ channel. Nature 2006, 439, 737–740. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Reigada, D.; Mitchell, C.H.; Ren, D. CATSPER channel-mediated Ca2+ entry into mouse sperm triggers a tail-to-head propagation. Biol. Reprod. 2007, 77, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Lishko, P.V.; Botchkina, I.L.; Kirichok, Y. Progesterone activates the principal Ca2+ channel of human sperm. Nature 2011, 471, 387–391. [Google Scholar] [CrossRef]
- Strünker, T.; Goodwin, N.; Brenker, C.; Kashikar, N.D.; Weyand, I.; Seifert, R.; Kaupp, U.B. The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 2011, 471, 382–386. [Google Scholar] [CrossRef]
- Santi, C.M.; Martinez-Lopez, P.; de la Vega-Beltran, J.L.; Butler, A.; Alisio, A.; Darszon, A.; Salkoff, L. The SLO3 sperm-specific potassium channel plays a vital role in male fertility. FEBS Lett. 2010, 584, 1041–1046. [Google Scholar] [CrossRef]
- Zeng, X.H.; Yang, C.; Kim, S.T.; Lingle, C.J.; Xia, X.M. Deletion of the Slo3 gene abolishes alkalization-activated K+ current in mouse spermatozoa. Proc. Natl. Acad. Sci. USA 2011, 108, 5879–5884. [Google Scholar] [CrossRef]
- Quill, T.A.; Sugden, S.A.; Rossi, K.L.; Doolittle, L.K.; Hammer, R.E.; Garbers, D.L. Hyperactivated sperm motility driven by CatSper2 is required for fertilization. Proc. Natl. Acad. Sci. USA 2003, 100, 14869–14874. [Google Scholar] [CrossRef]
- Qi, H.; Moran, M.M.; Navarro, B.; Chong, J.A.; Krapivinsky, G.; Krapivinsky, L.; Kirichok, Y.; Ramsey, I.S.; Quill, T.A.; Clapham, D.E. All four CatSper ion channel proteins are required for male fertility and sperm cell hyperactivated motility. Proc. Natl. Acad. Sci. USA 2007, 104, 1219–1223. [Google Scholar] [CrossRef]
- Chung, J.J.; Navarro, B.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. A novel gene required for male fertility and functional CATSPER channel formation in spermatozoa. Nat. Commun. 2011, 2, 153. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.J.; Miki, K.; Kim, D.; Shim, S.H.; Shi, H.F.; Hwang, J.Y.; Cai, X.; Iseri, Y.; Zhuang, X.; Clapham, D.E. CatSperζ regulates the structural continuity of sperm Ca(2+) signaling domains and is required for normal fertility. Elife 2017, 6, 23082. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.Y.; Mannowetz, N.; Zhang, Y.; Everley, R.A.; Gygi, S.P.; Bewersdorf, J.; Lishko, P.V.; Chung, J.J. Dual Sensing of Physiologic pH and Calcium by EFCAB9 Regulates Sperm Motility. Cell 2019, 177, 1480–1494. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.G.; Publicover, S.J.; Mansell, S.A.; Lishko, P.V.; Williams, H.L.; Ramalingam, M.; Wilson, S.M.; Barratt, C.L.; Sutton, K.A.; Da Silva, S.M. Depolarization of sperm membrane potential is a common feature of men with subfertility and is associated with low fertilization rate at IVF. Hum. Reprod. 2016, 31, 1147–1157. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.F.; Syritsyna, O.; Fellous, M.; Serres, C.; Mannowetz, N.; Kirichok, Y.; Lishko, P.V. Disruption of the principal, progesterone-activated sperm Ca2+ channel in a CatSper2-deficient infertile patient. Proc. Natl. Acad. Sci. USA 2013, 110, 6823–6828. [Google Scholar] [CrossRef]
- Luo, T.; Chen, H.Y.; Zou, Q.X.; Wang, T.; Cheng, Y.M.; Wang, H.F.; Wang, F.; Jin, Z.L.; Chen, Y.; Weng, S.Q.; et al. A novel copy number variation in CATSPER2 causes idiopathic male infertility with normal semen parameters. Hum. Reprod. 2019, 34, 414–423. [Google Scholar] [CrossRef]
- Zeng, X.H.; Yang, C.; Xia, X.M.; Liu, M.; Lingle, C.J. SLO3 auxiliary subunit LRRC52 controls gating of sperm KSPER currents and is critical for normal fertility. Proc. Natl. Acad. Sci. USA 2015, 112, 2599–2604. [Google Scholar] [CrossRef] [PubMed]
- Chávez, J.C.; Ferreira, J.J.; Butler, A.; De La Vega Beltrán, J.L.; Treviño, C.L.; Darszon, A.; Salkoff, L.; Santi, C.M. SLO3 K+ channels control calcium entry through CATSPER channels in sperm. J. Biol. Chem. 2014, 289, 32266–32275. [Google Scholar] [CrossRef]
- Schreiber, M.; Wei, A.; Yuan, A.; Gaut, J.; Saito, M.; Salkoff, L. Slo3, a novel pH-sensitive K+ channel from mammalian spermatocytes. J. Biol. Chem. 1998, 273, 3509–3516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zeng, X.; Lingle, C.J. Slo3 K+ channels: Voltage and pH dependence of macroscopic currents. J. Gen. Physiol. 2006, 128, 317–336. [Google Scholar] [CrossRef] [PubMed]
- Nishigaki, T.; José, O.; González-Cota, A.L.; Romero, F.; Treviño, C.L.; Darszon, A. Intracellular pH in sperm physiology. Biochem. Biophys. Res. Commun. 2014, 450, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Lardy, H.A.; Pounden, W.D.; Phillips, P.H. H ion concentration of various fluids of the genital tract of the cow. Proc. Soc. Exp. Biol. Med. 1940, 44, 517. [Google Scholar] [CrossRef]
- Kleyman, T.R.; Cragoe, E.J., Jr. Amiloride and its analogs as tools in the study of ion transport. J. Membr. Biol. 1988, 105, 1–21. [Google Scholar] [CrossRef]
- Aronson, P.S.; Nee, J.; Suhm, M.A. Modifier role of internal H+ in activating the Na+-H+ exchanger in renal microvillus membrane vesicles. Nature 1982, 299, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Baro Graf, C.; Ritagliati, C.; Torres-Monserrat, V.; Stival, C.; Carizza, C.; Buffone, M.G.; Krapf, D. Membrane Potential Assessment by Fluorimetry as a Predictor Tool of Human Sperm Fertilizing Capacity. Front. Cell Dev. Biol. 2019, 7, 383. [Google Scholar] [CrossRef]
- Molina, L.C.P.; Gunderson, S.; Riley, J.; Lybaert, P.; Borrego-Alvarez, A.; Jungheim, E.S.; Santi, C.M. Membrane Potential Determined by Flow Cytometry Predicts Fertilizing Ability of Human Sperm. Front. Cell Dev. Biol. 2019, 7, 387. [Google Scholar] [CrossRef]
- Hernández-González, E.O.; Sosnik, J.; Edwards, J.; Acevedo, J.J.; Mendoza-Lujambio, I.; López-González, I.; Demarco, I.; Wertheimer, E.; Darszon, A.; Visconti, P.E. Sodium and epithelial sodium channels participate in the regulation of the capacitation-associated hyperpolarization in mouse sperm. J. Biol. Chem. 2006, 281, 5623–5633. [Google Scholar] [CrossRef] [PubMed]
- Publicover, S.; Harper, C.V.; Barratt, C. [Ca2+]i signalling in sperm--making the most of what you’ve got. Nat. Cell Biol. 2007, 9, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.D.; Bernardino, R.L.; Neuhaus-Oliveira, A.; Sousa, M.; Sá, R.; Alves, M.G.; Oliveira, P.F. Physiology of na+/h+ exchangers in the male reproductive tract: Relevance for male fertility. Biol. Reprod. 2014, 91, 11. [Google Scholar] [CrossRef]
- Garcia, M.A.; Meizel, S. Regulation of intracellular pH in capacitated human spermatozoa by a Na+/H+ exchanger. Mol. Reprod. Dev. 1999, 52, 189–195. [Google Scholar] [CrossRef]
- Muzzachi, S.; Guerra, L.; Martino, N.A.; Favia, M.; Punzi, G.; Silvestre, F.; Guaricci, A.C.; Roscino, M.T.; Pierri, C.L.; Dell’Aquila, M.E.; et al. Effect of cariporide on ram sperm pH regulation and motility: Possible role of NHE1. Reproduction 2018, 155, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Woo, A.L.; James, P.F.; Lingrel, J.B. Roles of the Na,K-ATPase alpha4 isoform and the Na+/H+ exchanger in sperm motility. Mol. Reprod. Dev. 2002, 62, 348–356. [Google Scholar] [CrossRef]
- Oberheide, K.; Puchkov, D.; Jentsch, T.J. Loss of the Na(+)/H(+) exchanger NHE8 causes male infertility in mice by disrupting acrosome formation. J. Biol. Chem. 2017, 292, 10845–10854. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; King, S.M.; Quill, T.A.; Doolittle, L.K.; Garbers, D.L. A new sperm-specific Na+/H+ exchanger required for sperm motility and fertility. Nat. Cell Biol. 2003, 5, 1117–1122. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, J.; Bobulescu, I.A.; Quill, T.A.; McLeroy, P.; Moe, O.W.; Garbers, D.L. A sperm-specific Na+/H+ exchanger (sNHE) is critical for expression and in vivo bicarbonate regulation of the soluble adenylyl cyclase (sAC). Proc. Natl. Acad. Sci. USA 2007, 104, 9325–9330. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Huang, J.C.; Zuo, W.L.; Lu, C.L.; Chen, M.; Zhang, X.S.; Li, Y.C.; Cai, H.; Zhou, W.L.; Hu, Z.Y.; et al. A novel testis-specific Na+/H+ exchanger is involved in sperm motility and fertility. Front. Biosci. 2010, 2, 566–581. [Google Scholar] [CrossRef][Green Version]
- Chen, S.R.; Chen, M.; Deng, S.L.; Hao, X.X.; Wang, X.X.; Liu, Y.X. Sodium-hydrogen exchanger NHA1 and NHA2 control sperm motility and male fertility. Cell Death Dis. 2016, 7, e2152. [Google Scholar] [CrossRef] [PubMed]
- Masereel, B.; Pochet, L.; Laeckmann, D. An overview of inhibitors of Na(+)/H(+) exchanger. Eur. J. Med. Chem. 2003, 38, 547–554. [Google Scholar] [CrossRef]
- Xiang, M.; Feng, M.; Muend, S.; Rao, R. A human Na+/H+ antiporter sharing evolutionary origins with bacterial NhaA may be a candidate gene for essential hypertension. Proc. Natl. Acad. Sci. USA 2007, 104, 18677–18681. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Mansell, S.A.; Meyers, S.A.; Lishko, P.V. Flagellar ion channels of sperm: Similarities and differences between species. Cell Calcium. 2015, 58, 105–113. [Google Scholar] [CrossRef]
- Lishko, P.V.; Botchkina, I.L.; Fedorenko, A.; Kirichok, Y. Acid extrusion from human spermatozoa is mediated by flagellar voltage-gated proton channel. Cell 2010, 140, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Yanagimachi, R. Fertility of mammalian spermatozoa: Its development and relativity. Zygote 1994, 2, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Lishko, P.V.; Clapham, D.E.; Navarro, B.; Kirichok, Y. Sperm patch-clamp. Methods Enzym. 2013, 525, 59–83. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, H.; Liu, M.; Zhang, W.; Huang, R.-Z.; Zhao, N.; Chen, C.; Zeng, X.-H. Na+/H+ Exchangers Involve in Regulating the pH-Sensitive Ion Channels in Mouse Sperm. Int. J. Mol. Sci. 2021, 22, 1612. https://doi.org/10.3390/ijms22041612
Kang H, Liu M, Zhang W, Huang R-Z, Zhao N, Chen C, Zeng X-H. Na+/H+ Exchangers Involve in Regulating the pH-Sensitive Ion Channels in Mouse Sperm. International Journal of Molecular Sciences. 2021; 22(4):1612. https://doi.org/10.3390/ijms22041612
Chicago/Turabian StyleKang, Hang, Min Liu, Wei Zhang, Rong-Zu Huang, Na Zhao, Chen Chen, and Xu-Hui Zeng. 2021. "Na+/H+ Exchangers Involve in Regulating the pH-Sensitive Ion Channels in Mouse Sperm" International Journal of Molecular Sciences 22, no. 4: 1612. https://doi.org/10.3390/ijms22041612
APA StyleKang, H., Liu, M., Zhang, W., Huang, R.-Z., Zhao, N., Chen, C., & Zeng, X.-H. (2021). Na+/H+ Exchangers Involve in Regulating the pH-Sensitive Ion Channels in Mouse Sperm. International Journal of Molecular Sciences, 22(4), 1612. https://doi.org/10.3390/ijms22041612




