Vanadium Compounds as Pro-Inflammatory Agents: Effects on Cyclooxygenases
Abstract
1. Introduction

2. Vanadium Compounds in the Cell
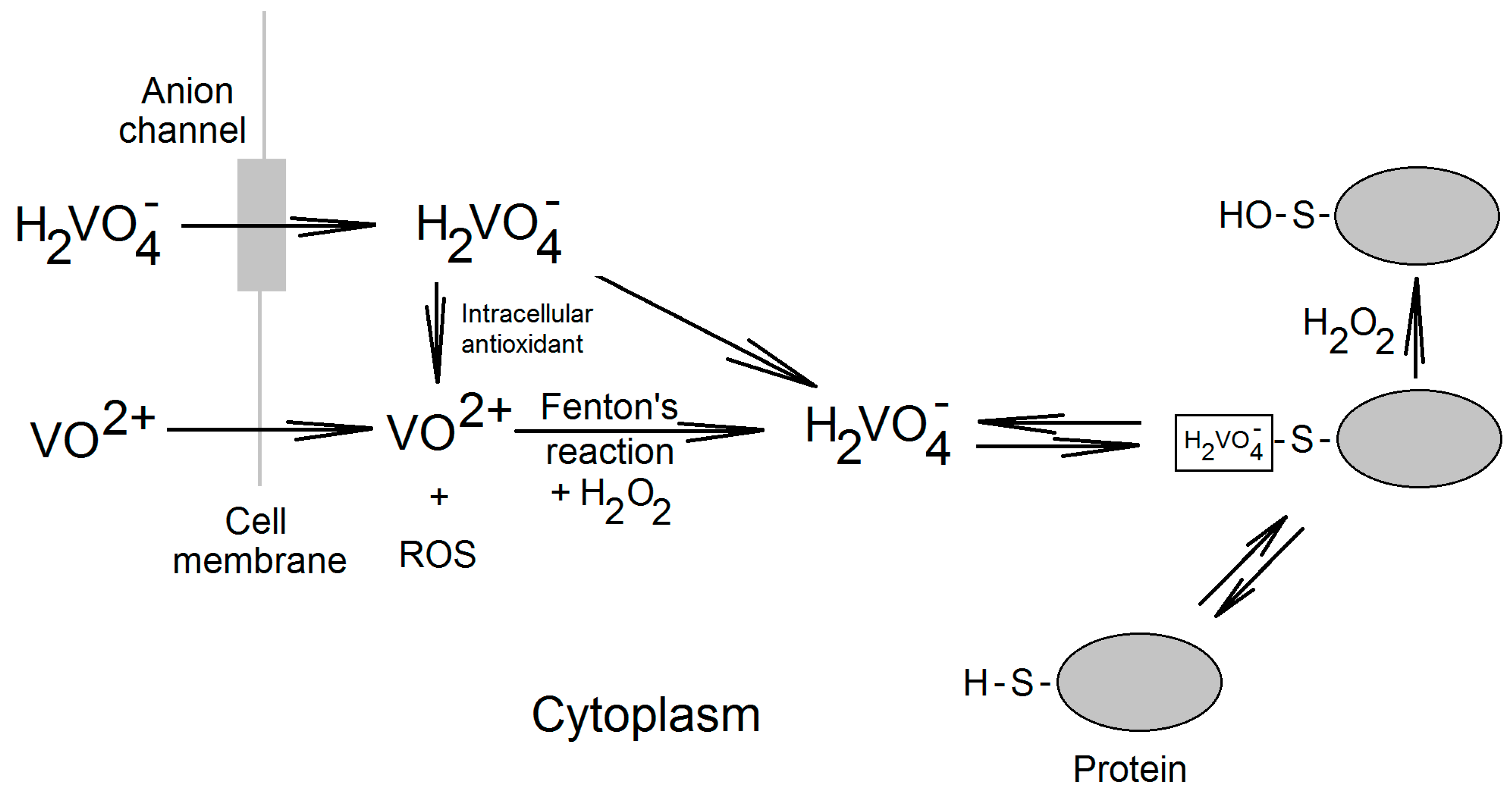
3. Effect of Vanadium Compounds on the Expression of Cyclooxygenases
3.1. Expression of Cyclooxygenases
3.2. Effect on the Expression of Cyclooxygenase-1
3.3. Effect on the Expression of Cyclooxygenase-2
3.3.1. EGFR Signal Transduction Resulting in Increased Expression of Cyclooxygenase-2
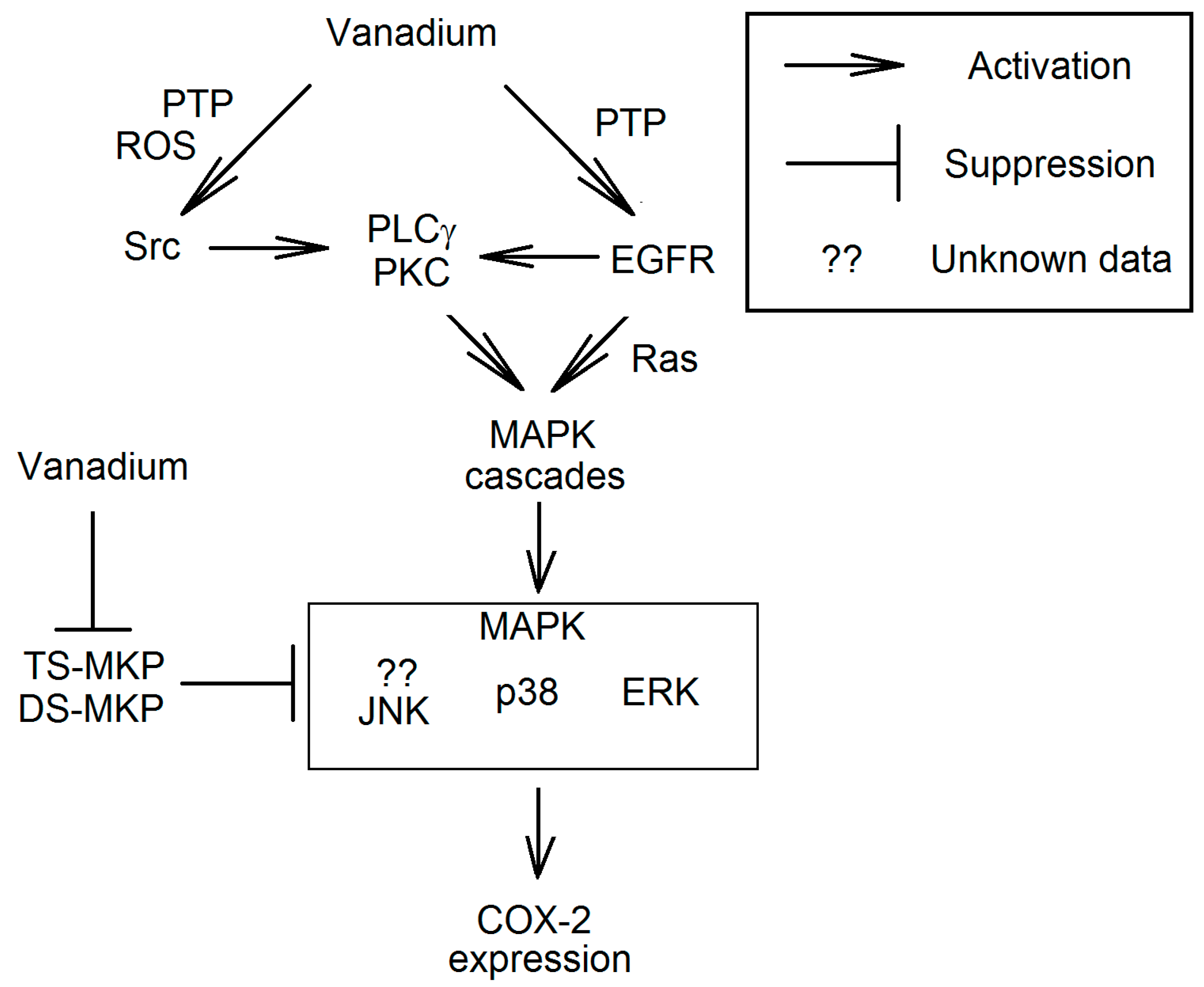
3.3.2. Activation of MAPK Cascades Independent of EGFR
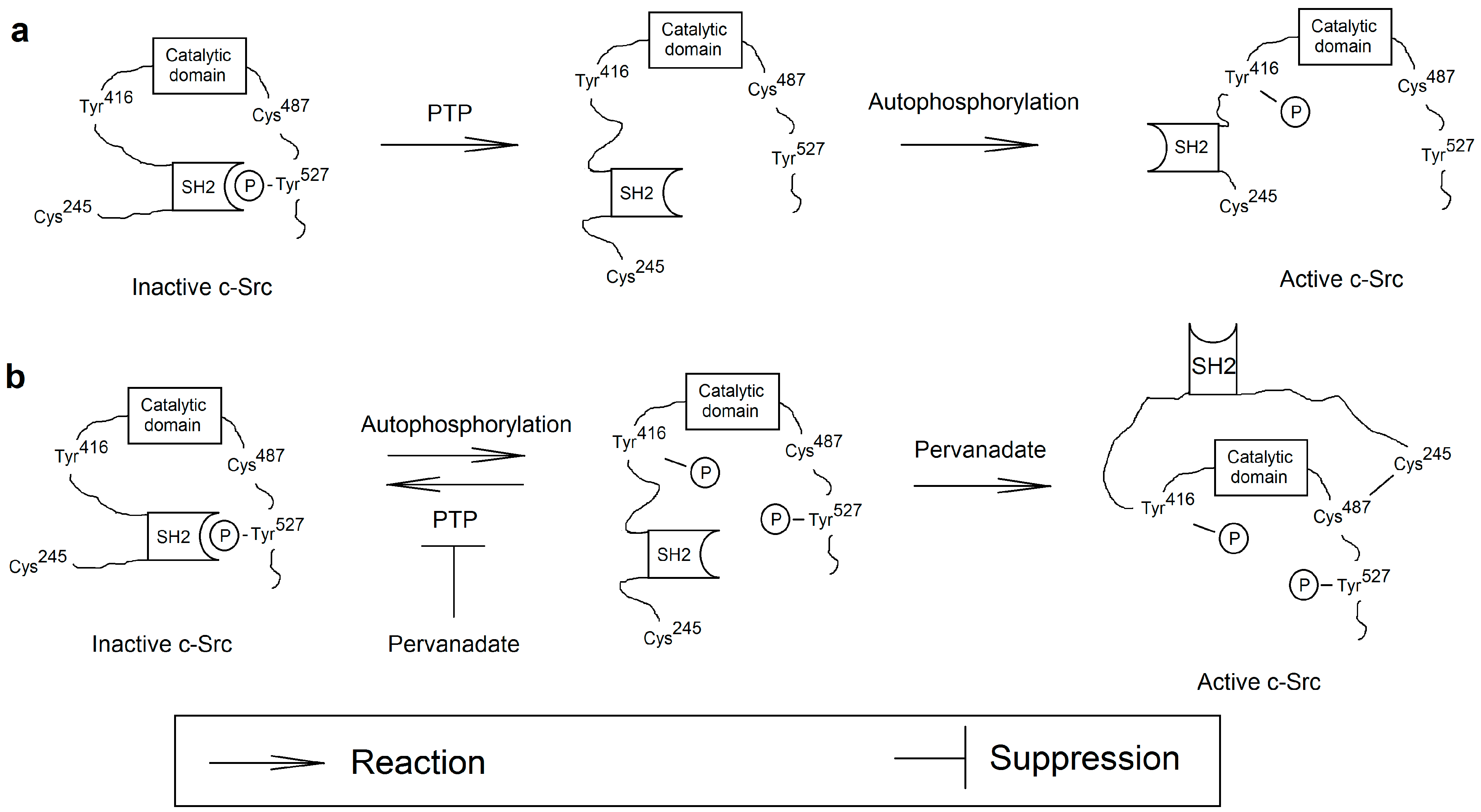
3.3.3. Activation of the Src
3.3.4. The Effect on MAPK Phosphatase
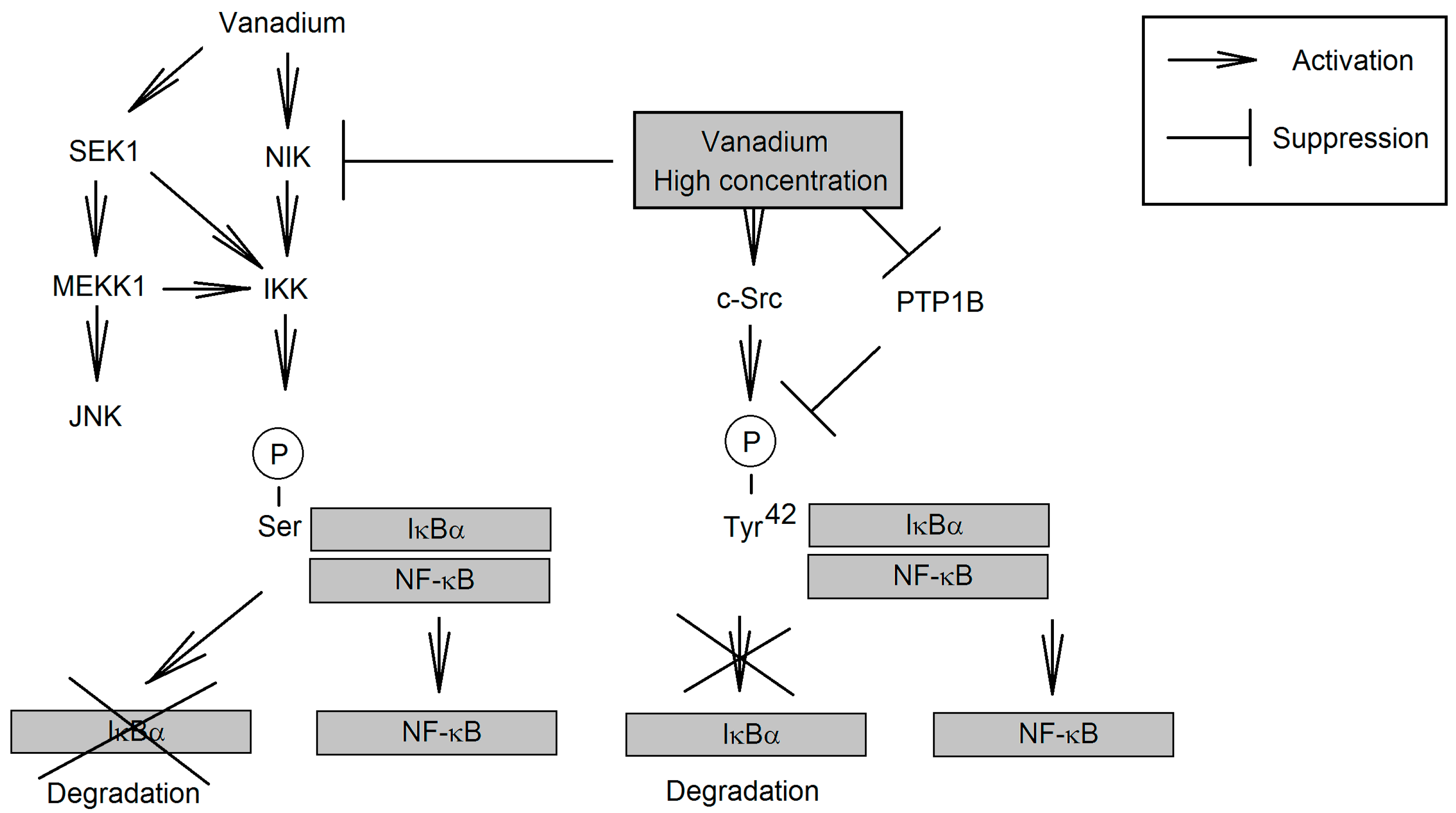
3.3.5. Effect on NF-κB
3.3.6. Effect on the Proteolysis of Cyclooxygenase-2
4. Effect on Cyclooxygenase Activity
5. Effect on the Supply of Substrate for Cyclooxygenases
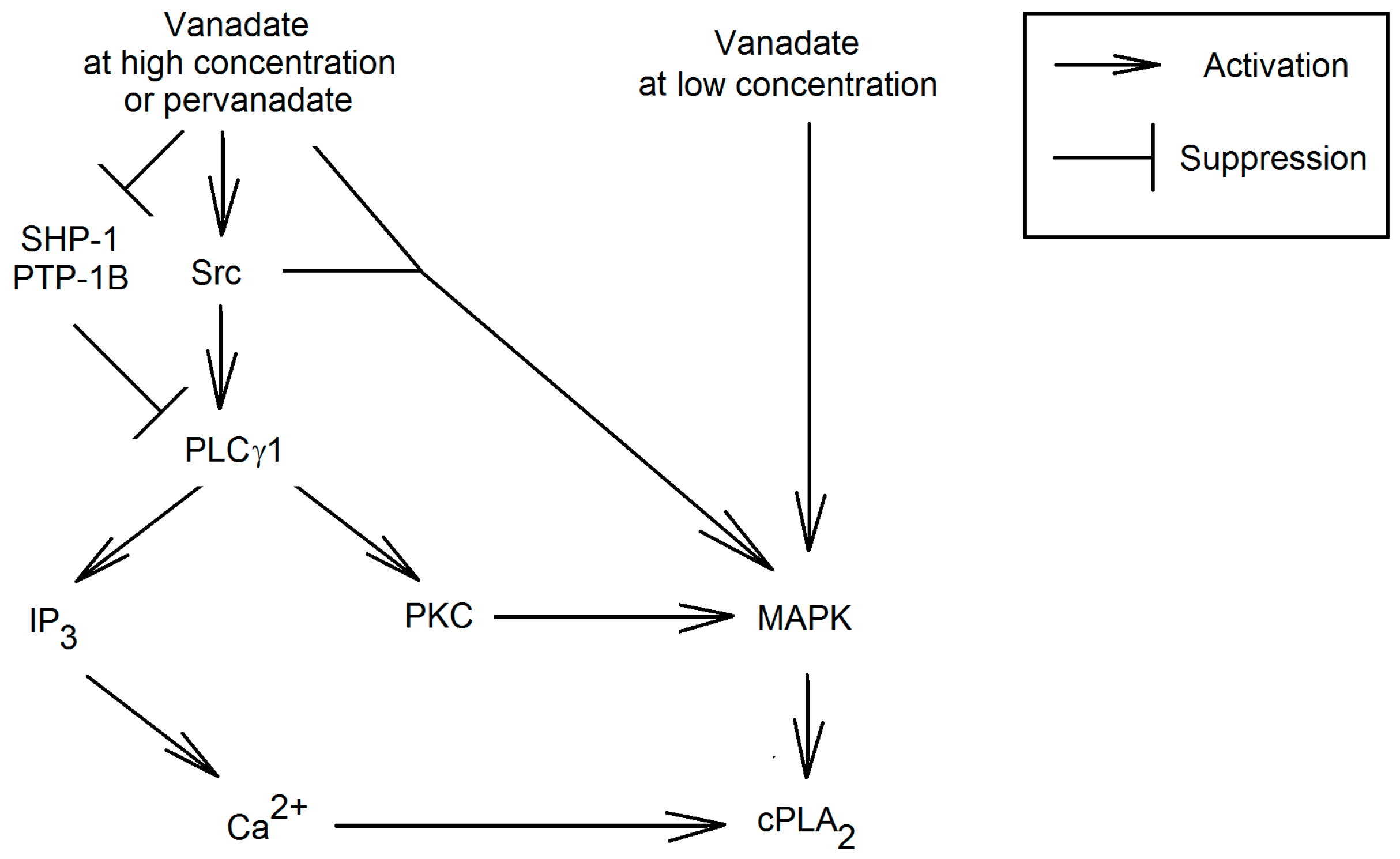
6. Pro-Inflammatory Properties and Therapeutic Use of Vanadium Compounds
Conflicts of Interest
Abbreviations
| AA | arachidonic acid |
| AP-1 | activator protein-1 |
| ASK-1 | apoptosis signal-regulating kinase 1 |
| COX-1 | cyclooxygenase-1 |
| COX-2 | cyclooxygenase-2 |
| COX | cyclooxygenases |
| cPLA2 | cytoplasmic phospholipase A2 |
| DAG | diacylglycerol |
| DS.-MKP | dual specificity MKP |
| EGF | epidermal growth factor |
| EGFR | epidermal growth factor receptor |
| ERK | extracellular signal-regulated kinase |
| HUVEC | human umbilical vein in endothelial cell |
| IKK | IκB kinase |
| IP3 | inositol trisphosphate |
| IκB | inhibitor of NF-κB |
| JNK | c-Jun N-terminal kinase |
| MAPK | mitogen-activated protein kinase |
| MAPKK | mitogen-activated protein kinase kinase |
| MAPKKK | mitogen-activated protein kinase kinase kinase |
| MEKK1 | mitogen-activated protein kinase/ERK kinase kinase 1 |
| MKP | MAPK phosphatase |
| NF-κB | nuclear factor κB |
| NIK | NF-κB-inducing kinase |
| PDGFR | platelet-derived growth factor receptor |
| PGE2 | prostaglandin E2 |
| PKC | protein kinase C |
| PLCγ | phospholipase C-γ |
| PTP | protein tyrosine phosphatases |
| PTP-1B | protein tyrosine phosphatase-1B |
| ROS | reactive oxygen species |
| SEK1 | SAPK/ERK kinase 1 |
| SHP-1 | SH2 domain-containing protein tyrosine phosphatase 1 |
| SHP-2 | SH2 domain-containing protein tyrosine phosphatase 2 |
| Sp1 | specificity protein 1 |
| Src | non-receptor tyrosine kinases of the Src family |
| TS-MKP | tyrosine-specific MKP |
| TxA2 | thromboxane A2 |
References
- Thompson, K.H.; Orvig, C. Vanadium in diabetes: 100 years from Phase 0 to Phase I. J. Inorg. Biochem. 2006, 100, 1925–1935. [Google Scholar] [CrossRef] [PubMed]
- Thompson, K.H.; Lichter, J.; LeBel, C.; Scaife, M.C.; McNeill, J.H.; Orvig, C. Vanadium treatment of type 2 diabetes: A view to the future. J. Inorg. Biochem. 2009, 103, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Kurt, O.; Ozden, T.Y.; Ozsoy, N.; Tunali, S.; Can, A.; Akev, N.; Yanardag, R. Influence of vanadium supplementation on oxidative stress factors in the muscle of STZ-diabetic rats. Biometals 2011, 24, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Missaoui, S.; Ben Rhouma, K.; Yacoubi, M.T.; Sakly, M.; Tebourbi, O. Vanadyl sulfate treatment stimulates proliferation and regeneration of beta cells in pancreatic islets. J. Diabetes Res. 2014, 2014, 540242. [Google Scholar] [CrossRef] [PubMed]
- Pirmoradi, L.; Mohammadi, M.T.; Safaei, A.; Mesbah, F.; Dehghani, G.A. Does the relief of glucose toxicity act as a mediator in proliferative actions of vanadium on pancreatic islet beta cells in streptozocin diabetic rats? Iran. Biomed. J. 2014, 18, 173–180. [Google Scholar] [PubMed]
- Sun, L.; Shi, D.J.; Gao, X.C.; Mi, S.Y.; Yu, Y.; Han, Q. The protective effect of vanadium against diabetic cataracts in diabetic rat model. Biol. Trace Elem. Res. 2014, 158, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Soveid, M.; Dehghani, G.A.; Omrani, G.R. Long-term efficacy and safety of vanadium in the treatment of type 1 diabetes. Arch. Iran. Med. 2013, 16, 408–411. [Google Scholar] [PubMed]
- Evangelou, A.M. Vanadium in cancer treatment. Crit. Rev. Oncol. Hematol. 2002, 42, 249–265. [Google Scholar] [CrossRef]
- Bishayee, A.; Waghray, A.; Patel, M.A.; Chatterjee, M. Vanadium in the detection, prevention and treatment of cancer: the in vivo evidence. Cancer Lett. 2010, 294, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.T.; Liu, Y.J.; Wang, Q.; Yang, X.G.; Wang, K. Reactive-oxygen-species-mediated Cdc25C degradation results in differential antiproliferative activities of vanadate, tungstate, and molybdate in the PC-3 human prostate cancer cell line. J. Biol. Inorg. Chem. 2012, 17, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Suwalsky, M.; Fierro, P.; Villena, F.; Gallardo, M.J.; Jemiola-Rzeminska, M.; Strzalka, K.; Gul-Hinc, S.; Ronowska, A.; Zysk, M.; Szutowicz, A. Effects of sodium metavanadate on in vitro neuroblastoma and red blood cells. Arch. Biochem. Biophys. 2013, 535, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ma, Y.; Xu, Z.; Wang, D.; Zhao, B.; Pan, H.; Wang, J.; Xu, D.; Zhao, X.; Pan, S.; et al. Sodium orthovanadate inhibits growth of human hepatocellular carcinoma cells in vitro and in an orthotopic model in vivo. Cancer Lett. 2014, 351, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Kucera, J.; Byrne, A.R.; Mravcová, A.; Lener, J. Vanadium levels in hair and blood of normal and exposed persons. Sci. Total Environ. 1992, 115, 191–205. [Google Scholar] [CrossRef]
- Nadal, M.; Schuhmacher, M.; Domingo, J.L. Metal pollution of soils and vegetation in an area with petrochemical industry. Sci. Total Environ. 2004, 321, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Pourang, N.; Nikouyan, A.; Dennis, J.H. Trace element concentrations in fish, surficial sediments and water from northern part of the Persian Gulf. Environ. Monit. Assess. 2005, 109, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Moreno, T.; Querol, X.; Alastuey, A.; de la Rosa, J.; Sánchez de la Campa, A.M.; Minguillón, M.; Pandolfi, M.; González-Castanedo, Y.; Monfort, E.; Gibbons, W. Variations in vanadium, nickel and lanthanoid element concentrations in urban air. Sci. Total Environ. 2010, 408, 4569–4579. [Google Scholar] [CrossRef] [PubMed]
- Guzmán-Morales, J.; Morton-Bermea, O.; Hernández-Álvarez, E.; Rodríguez-Salazar, M.T.; García-Arreola, M.E.; Tapia-Cruz, V. Assessment of atmospheric metal pollution in the urban area of Mexico City, using Ficus benjamina as biomonitor. Bull. Environ. Contam. Toxicol. 2011, 86, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Speight, J. Chemical composition. In The Chemistry and Technology of Petroleum, 3rd ed.; CRC Press: New York, NY, USA; Basel, Switzerland, 1999; pp. 215–243. [Google Scholar]
- Bednar, A.J.; Chappell, M.A.; Seiter, J.M.; Stanley, J.K.; Averett, D.E.; Jones, W.T.; Pettway, B.A.; Kennedy, A.J.; Hendrix, S.H.; Steevens, J.A. Geochemical investigations of metals release from submerged coal fly ash using extended elutriate tests. Chemosphere 2010, 81, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.S.; Chang, C.L.; Shen, F.M. Whole blood vanadium in Taiwanese college students. Bull. Environ. Contam. Toxicol. 2004, 73, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Azay, J.; Brès, J.; Krosniak, M.; Teissedre, P.L.; Cabanis, J.C.; Serrano, J.J.; Cros, G. Vanadium pharmacokinetics and oral bioavailability upon single-dose administration of vanadyl sulfate to rats. Fundam. Clin. Pharmacol. 2001, 15, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Holko, P.; Ligeza, J.; Kordowiak, A.M. Sodium orthovanadate affects growth of some human epithelial cancer cells (A549, HTB44, DU145). Folia Biol. 2008, 56, 115–121. [Google Scholar] [CrossRef] [Green Version]
- McNicol, A.; Robertson, C.; Gerrard, J.M. Vanadate activates platelets by enhancing arachidonic acid release. Blood 1993, 81, 2329–2338. [Google Scholar] [PubMed]
- Tsujishita, Y.; Asaoka, Y.; Nishizuka, Y. Regulation of phospholipase A2 in human leukemia cell lines: Its implication for intracellular signaling. Proc. Natl. Acad. Sci. USA 1994, 91, 6274–6278. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.W.; Hsu, Y.W. Cycloheximide-induced cPLA(2) activation is via the MKP-1 down-regulation and ERK activation. Cell Signal. 2000, 12, 457–461. [Google Scholar] [CrossRef]
- Korbecki, J.; Baranowska-Bosiacka, I.; Gutowska, I.; Piotrowska, K.; Chlubek, D. Cyclooxygenase-1 as the main source of proinflammatory factors after sodium orthovanadate treatment. Biol. Trace Elem. Res. 2015, 163, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Greenhough, A.; Smartt, H.J.; Moore, A.E.; Roberts, H.R.; Williams, A.C.; Paraskeva, C.; Kaidi, A. The COX-2/PGE2 pathway: Key roles in the hallmarks of cancer and adaptation to the tumour microenvironment. Carcinogenesis 2009, 30, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Chien, P.S.; Mak, O.T.; Huang, H.J. Induction of COX-2 protein expression by vanadate in A549 human lung carcinoma cell line through EGF receptor and p38 MAPK-mediated pathway. Biochem. Biophys. Res. Commun. 2006, 339, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Boulassel, B.; Sadeg, N.; Roussel, O.; Perrin, M.; Belhadj-Tahar, H. Fatal poisoning by vanadium. Forensic Sci. Int. 2011, 206, e79–e81. [Google Scholar] [CrossRef] [PubMed]
- Crans, D.C.; Smee, J.J.; Gaidamauskas, E.; Yang, L. The chemistry and biochemistry of vanadium and the biological activities exerted by vanadium compounds. Chem. Rev. 2004, 104, 849–902. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, K.; Lu, J.; Crans, D.C. Membrane transport of vanadium compounds and the interaction with the erythrocyte membrane. Coord. Chem. Rev. 2003, 237, 103–111. [Google Scholar] [CrossRef]
- Bruech, M.; Quintanilla, M.E.; Legrum, W.; Koch, J.; Netter, K.J.; Fuhrmann, G.F. Effects of vanadate on intracellular reduction equivalents in mouse liver and the fate of vanadium in plasma, erythrocytes and liver. Toxicology 1984, 31, 283–295. [Google Scholar] [CrossRef]
- Ding, M.; Gannett, P.M.; Rojanasakul, Y.; Liu, K.; Shi, X. One-electron reduction of vanadate by ascorbate and related free radical generation at physiological pH. J. Inorg. Biochem. 1994, 55, 101–112. [Google Scholar] [CrossRef]
- Shi, X.L.; Dalal, N.S. Flavoenzymes reduce vanadium (V) and molecular oxygen and generate hydroxyl radical. Arch. Biochem. Biophys. 1991, 289, 355–361. [Google Scholar] [CrossRef]
- Shi, X.; Dalal, N.S. Hydroxyl radical generation in the NADH/microsomal reduction of vanadate. Free Radic. Res. Commun. 1992, 17, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Dalal, N.S. Vanadate-mediated hydroxyl radical generation from superoxide radical in the presence of NADH: Haber–Weiss vs. Fenton mechanism. Arch. Biochem. Biophys. 1993, 307, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Capella, L.S.; Gefé, M.R.; Silva, E.F.; Affonso-Mitidieri, O.; Lopes, A.G.; Rumjanek, V.M.; Capella, M.A. Mechanisms of vanadate-induced cellular toxicity: Role of cellular glutathione and NADPH. Arch. Biochem. Biophys. 2002, 406, 65–72. [Google Scholar] [CrossRef]
- Crans, D.C.; Zhang, B.; Gaidamauskas, E.; Keramidas, A.D.; Willsky, G.R.; Roberts, C.R. Is vanadate reduced by thiols under biological conditions? Changing the redox potential of V(V)/V(IV) by complexation in aqueous solution. Inorg. Chem. 2010, 49, 4245–4256. [Google Scholar] [CrossRef] [PubMed]
- Huyer, G.; Liu, S.; Kelly, J.; Moffat, J.; Payette, P.; Kennedy, B.; Tsaprailis, G.; Gresser, M.J.; Ramachandran, C. Mechanism of inhibition of protein-tyrosine phosphatases by vanadate and pervanadate. J. Biol. Chem. 1997, 272, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.G.; Zhang, Z.Y. Redox regulation of protein tyrosine phosphatase activity by hydroxyl radical. Biochim. Biophys. Acta 2013, 1834, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Fantus, I.G.; Deragon, G.; Lai, R.; Tang, S. Modulation of insulin action by vanadate: Evidence of a role for phosphotyrosine phosphatase activity to alter cellular signaling. Mol. Cell. Biochem. 1995, 153, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Pugazhenthi, S.; Tanha, F.; Dahl, B.; Khandelwal, R.L. Decrease in protein tyrosine phosphatase activities in vanadate-treated obese Zucker (fa/fa) rat liver. Mol. Cell. Biochem. 1995, 153, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Pugazhenthi, S.; Tanha, F.; Dahl, B.; Khandelwal, R.L. Inhibition of a Src homology 2 domain containing protein tyrosine phosphatase by vanadate in the primary culture of hepatocytes. Arch. Biochem. Biophys. 1996, 335, 273–282. [Google Scholar] [CrossRef]
- Ostman, A.; Frijhoff, J.; Sandin, A.; Böhmer, F.D. Regulation of protein tyrosine phosphatases by reversible oxidation. J. Biochem. 2011, 150, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Tan, Z.; Diltz, C.D.; You, M.; Fischer, E.H. Activation of mitogen-activated protein (MAP) kinase pathway by pervanadate, a potent inhibitor of tyrosine phosphatases. J. Biol. Chem. 1996, 271, 22251–22255. [Google Scholar] [PubMed]
- Lee, K.; Esselman, W.J. Inhibition of PTPs by H2O2 regulates the activation of distinct MAPK pathways. Free Radic. Biol. Med. 2002, 33, 1121–1132. [Google Scholar] [CrossRef]
- Sturla, L.M.; Amorino, G.; Alexander, M.S.; Mikkelsen, R.B.; Valerie, K.; Schmidt-Ullrichr, R.K. Requirement of Tyr-992 and Tyr-1173 in phosphorylation of the epidermal growth factor receptor by ionizing radiation and modulation by SHP2. J. Biol. Chem. 2005, 280, 14597–145604. [Google Scholar] [CrossRef] [PubMed]
- Mbonye, U.R.; Song, I. Posttranscriptional and posttranslational determinants of cyclooxygenase expression. BMB Rep. 2009, 42, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Ueda, N.; Yamashita, R.; Yamamoto, S.; Ishimura, K. Induction of cyclooxygenase-1 in a human megakaryoblastic cell line (CMK) differentiated by phorbol ester. Biochim. Biophys. Acta 1997, 1344, 103–110. [Google Scholar] [CrossRef]
- Okahara, K.; Sun, B.; Kambayashi, J. Upregulation of prostacyclin synthesis-related gene expression by shear stress in vascular endothelial cells. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1922–1926. [Google Scholar] [CrossRef] [PubMed]
- Gibson, L.L.; Hahner, L.; Osborne-Lawrence, S.; German, Z.; Wu, K.K.; Chambliss, K.L.; Shaul, P.W. Molecular basis of estrogen-induced cyclooxygenase type 1 upregulation in endothelial cells. Circ. Res. 2005, 96, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, T.; Tohnai, N. Cyclooxygenase isozymes and their gene structures and expression. Prostaglandins Other Lipid Mediat. 2002, 68–69, 95–114. [Google Scholar] [CrossRef]
- Hwang, D.; Jang, B.C.; Yu, G.; Boudreau, M. Expression of mitogen-inducible cyclooxygenase induced by lipopolysaccharide: Mediation through both mitogen-activated protein kinase and NF-kappaB signaling pathways in macrophages. Biochem. Pharmacol. 1997, 54, 87–96. [Google Scholar] [CrossRef]
- Faour, W.H.; He, Y.; He, Q.W.; de Ladurantaye, M.; Quintero, M.; Mancini, A.; di Battista, J.A. Prostaglandin E(2) regulates the level and stability of cyclooxygenase-2 mRNA through activation of p38 mitogen-activated protein kinase in interleukin-1 beta-treated human synovial fibroblasts. J. Biol. Chem. 2001, 276, 31720–31731. [Google Scholar] [CrossRef] [PubMed]
- Parfenova, H.; Balabanova, L.; Leffler, C.W. Posttranslational regulation of cyclooxygenase by tyrosine phosphorylation in cerebral endothelial cells. Am. J. Physiol. 1998, 274, C72–C81. [Google Scholar] [PubMed]
- Alexanian, A.; Miller, B.; Chesnik, M.; Mirza, S.; Sorokin, A. Post-translational regulation of COX2 activity by FYN in prostate cancer cells. Oncotarget 2014, 5, 4232–4243. [Google Scholar] [PubMed]
- Hirai, K.; Takayama, H.; Tomo, K.; Okuma, M. Protein-tyrosine-kinase-dependent expression of cyclo-oxygenase-1 and -2 mRNAs in human endothelial cells. Biochem. J. 1997, 322, 373–377. [Google Scholar] [PubMed]
- Hirai, K.; Ezumi, Y.; Nishida, E.; Uchiyama, T.; Takayama, H. Comparative study of vanadate- and phorbol ester-induced cyclo-oxygenase-2 expression in human endothelial cells. Thromb. Haemost. 1999, 82, 1545–1552. [Google Scholar] [PubMed]
- DeLong, C.J.; Smith, W.L. An intronic enhancer regulates cyclooxygenase-1 gene expression. Biochem. Biophys. Res. Commun. 2005, 338, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Li, J.J.; Leonard, S.S.; Ye, J.P.; Shi, X.; Colburn, N.H.; Castranova, V.; Vallyathan, V. Vanadate-induced activation of activator protein-1: role of reactive oxygen species. Carcinogenesis 1999, 20, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Chuang, J.Y.; Wang, Y.T.; Yeh, S.H.; Liu, Y.W.; Chang, W.C.; Hung, J.J. Phosphorylation by c-Jun NH2-terminal kinase 1 regulates the stability of transcription factor Sp1 during mitosis. Mol. Biol. Cell 2008, 19, 1139–1151. [Google Scholar] [CrossRef] [PubMed]
- Chu, S. Transcriptional regulation by post-transcriptional modification-role of phosphorylation in Sp1 transcriptional activity. Gene 2012, 508, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Demers, L.M.; Vallyathan, V.; Ding, M.; Lu, Y.; Castranova, V.; Shi, X. Vanadate induction of NF-kappaB involves IkappaB kinase beta and SAPK/ERK kinase 1 in macrophages. J. Biol. Chem. 1999, 274, 20307–20312. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Engelhardt, J.F. Interleukin-1beta induction of NFkappaB is partially regulated by H2O2-mediated activation of NFkappaB-inducing kinase. J. Biol. Chem. 2006, 281, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.J.; Lee, E.K.; Yu, B.P.; Chung, H.Y. Significance of protein tyrosine kinase/protein tyrosine phosphatase balance in the regulation of NF-kappaB signaling in the inflammatory process and aging. Free Radic. Biol. Med. 2009, 47, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Milarski, K.L.; Zhu, G.; Pearl, C.G.; McNamara, D.J.; Dobrusin, E.M.; MacLean, D.; Thieme-Sefler, A.; Zhang, Z.Y.; Sawyer, T.; Decker, S.J.; et al. Sequence specificity in recognition of the epidermal growth factor receptor by protein tyrosine phosphatase 1B. J. Biol. Chem. 1993, 268, 23634–23639. [Google Scholar] [PubMed]
- Agazie, Y.M.; Hayman, M.J. Molecular mechanism for a role of SHP2 in epidermal growth factor receptor signaling. Mol. Cell. Biol. 2003, 23, 7875–7886. [Google Scholar] [CrossRef] [PubMed]
- Keilhack, H.; Tenev, T.; Nyakatura, E.; Godovac-Zimmermann, J.; Nielsen, L.; Seedorf, K.; Böhmer, F.D. Phosphotyrosine 1173 mediates binding of the protein-tyrosine phosphatase SHP-1 to the epidermal growth factor receptor and attenuation of receptor signaling. J. Biol. Chem. 1998, 273, 24839–24846. [Google Scholar] [CrossRef] [PubMed]
- Jorissen, R.N.; Walker, F.; Pouliot, N.; Garrett, T.P.; Ward, C.W.; Burgess, A.W. Epidermal growth factor receptor: Mechanisms of activation and signaling. Exp. Cell Res. 2003, 284, 31–53. [Google Scholar] [CrossRef]
- Wu, W.; Graves, L.M.; Jaspers, I.; Devlin, R.B.; Reed, W.; Samet, J.M. Activation of the EGF receptor signaling pathway in human airway epithelial cells exposed to metals. Am. J. Physiol. 1999, 277, L924–L931. [Google Scholar] [PubMed]
- Wu, W.; Jaspers, I.; Zhang, W.; Graves, L.M.; Samet, J.M. Role of Ras in metal-induced EGF receptor signaling and NF-kappaB activation in human airway epithelial cells. Lung Cell. Mol. Physiol. 2002, 282, L1040–L1048. [Google Scholar] [CrossRef] [PubMed]
- Tao, Q.; Spring, S.C.; Terman, B.I. Comparison of the signaling mechanisms by which VEGF, H2O2, and phosphatase inhibitors activate endothelial cell ERK1/2 MAP-kinase. Microvasc. Res. 2005, 69, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. Src kinase regulation by phosphorylation and dephosphorylation. Biochem. Biophys. Res. Commun. 2005, 331, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ingley, E. Src family kinases: Regulation of their activities, levels and identification of new pathways. Biochim. Biophys. Acta 2008, 1784, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. Src protein-tyrosine kinase structure and regulation. Biochem. Biophys. Res. Commun. 2004, 324, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Chiarugi, P. Src redox regulation: There is more than meets the eye. Mol. Cells 2008, 26, 329–337. [Google Scholar] [PubMed]
- Giannoni, E.; Taddei, M.L.; Chiarugi, P. Src redox regulation: again in the front line. Free Radic. Biol. Med. 2010, 49, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Lluis, J.M.; Buricchi, F.; Chiarugi, P.; Morales, A.; Fernandez-Checa, J.C. Dual role of mitochondrial reactive oxygen species in hypoxia signaling: Activation of nuclear factor-κB via c-SRC and oxidant-dependent cell death. Cancer Res. 2007, 67, 7368–7377. [Google Scholar] [CrossRef] [PubMed]
- Boulven, I.; Robin, P.; Desmyter, C.; Harbon, S.; Leiber, D. Differential involvement of Src family kinases in pervanadate-mediated responses in rat myometrial cells. Cell Signal. 2002, 14, 341–349. [Google Scholar] [CrossRef]
- Fan, C.; Li, Q.; Ross, D.; Engelhardt, J.F. Tyrosine phosphorylation of I kappa B alpha activates NF kappa B through a redox-regulated and c-Src-dependent mechanism following hypoxia/reoxygenation. J. Biol. Chem. 2003, 278, 2072–2080. [Google Scholar] [CrossRef] [PubMed]
- Kmiecik, T.E.; Johnson, P.J.; Shalloway, D. Regulation by the autophosphorylation site in overexpressed pp60c-src. Mol. Cell. Biol. 1988, 8, 4541–4546. [Google Scholar] [PubMed]
- Irtegun, S.; Wood, R.J.; Ormsby, A.R.; Mulhern, T.D.; Hatters, D.M. Tyrosine 416 is phosphorylated in the closed, repressed conformation of c-Src. PLoS ONE 2013, 8, e71035. [Google Scholar] [CrossRef] [PubMed]
- Farooq, A.; Zhou, M.M. Structure and regulation of MAPK phosphatases. Cell Signal. 2004, 16, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Kondoh, K.; Nishida, E. Regulation of MAP kinases by MAP kinase phosphatases. Biochim. Biophys. Acta 2007, 1773, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Turpeinen, T.; Nieminen, R.; Moilanen, E.; Korhonen, R. Mitogen-activated protein kinase phosphatase-1 negatively regulates the expression of interleukin-6, interleukin-8, and cyclooxygenase-2 in A549 human lung epithelial cells. J. Pharmacol. Exp. Ther. 2010, 333, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Misra-Press, A.; Rim, C.S.; Yao, H.; Roberson, M.S.; Stork, P.J. A novel mitogen-activated protein kinase phosphatase. Structure, expression, and regulation. J. Biol. Chem. 1995, 270, 14587–14596. [Google Scholar] [CrossRef] [PubMed]
- Muda, M.; Boschert, U.; Smith, A.; Antonsson, B.; Gillieron, C.; Chabert, C.; Camps, M.; Martinou, I.; Ashworth, A.; Arkinstall, S. Molecular cloning and functional characterization of a novel mitogen-activated protein kinase phosphatase, MKP-4. J. Biol. Chem. 1997, 272, 5141–5151. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Ybanez, M.D.; Ahmadi, S.; Yeh, K.; Kaplowitz, N. Redox regulation of tumor necrosis factor signaling. Antioxid. Redox Signal. 2009, 11, 2245–2263. [Google Scholar] [CrossRef] [PubMed]
- O’Dea, E.; Hoffmann, A. The regulatory logic of the NF-kappaB signaling system. Cold Spring Harb. Perspect. Biol. 2010, 2, a000216. [Google Scholar] [PubMed]
- Barbeau, B.; Bernier, R.; Dumais, N.; Briand, G.; Olivier, M.; Faure, R.; Posner, B.I.; Tremblay, M. Activation of HIV-1 long terminal repeat transcription and virus replication via NF-kappaB-dependent and -independent pathways by potent phosphotyrosine phosphatase inhibitors, the peroxovanadium compounds. J. Biol. Chem. 1997, 272, 12968–12977. [Google Scholar] [CrossRef] [PubMed]
- Lee, F.S.; Peters, R.T.; Dang, L.C.; Maniatis, T. MEKK1 activates both IkappaB kinase alpha and IkappaB kinase beta. Proc. Natl. Acad. Sci. USA 1998, 95, 9319–9324. [Google Scholar] [CrossRef] [PubMed]
- Herscovitch, M.; Comb, W.; Ennis, T.; Coleman, K.; Yong, S.; Armstead, B.; Kalaitzidis, D.; Chandani, S.; Gilmore, T.D. Intermolecular disulfide bond formation in the NEMO dimer requires Cys54 and Cys347. Biochem. Biophys. Res. Commun. 2008, 367, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.J.; Liu, Z.G. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2011, 21, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Siomek, A. NF-κB signaling pathway and free radical impact. Acta Biochim. Pol. 2012, 59, 323–331. [Google Scholar] [PubMed]
- Imbert, V.; Rupec, R.A.; Livolsi, A.; Pahl, H.L.; Traenckner, E.B.; Mueller-Dieckmann, C.; Farahifar, D.; Rossi, B.; Auberger, P.; Baeuerle, P.A.; et al. Tyrosine phosphorylation of I kappa B-alpha activates NF-kappa B without proteolytic degradation of I kappa B-alpha. Cell 1996, 86, 787–798. [Google Scholar] [CrossRef]
- Singh, S.; Darnay, B.G.; Aggarwal, B.B. Site-specific tyrosine phosphorylation of IkappaBalpha negatively regulates its inducible phosphorylation and degradation. J. Biol. Chem. 1996, 271, 31049–31054. [Google Scholar] [CrossRef] [PubMed]
- Béraud, C.; Henzel, W.J.; Baeuerle, P.A. Involvement of regulatory and catalytic subunits of phosphoinositide 3-kinase in NF-kappaB activation. Proc. Natl. Acad. Sci. USA 1999, 96, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, A.; Manna, S.K.; Aggarwal, B.B. Pervanadate-induced nuclear factor-kappaB activation requires tyrosine phosphorylation and degradation of IkappaBalpha. Comparison with tumor necrosis factor-alpha. J. Biol. Chem. 2000, 275, 8549–8555. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Aggarwal, B.B. Protein-tyrosine phosphatase inhibitors block tumor necrosis factor-dependent activation of the nuclear transcription factor NF-kappa B. J. Biol. Chem. 1995, 270, 10631–10639. [Google Scholar] [PubMed]
- Kang, Y.J.; Mbonye, U.R.; DeLong, C.J.; Wada, M.; Smith, W.L. Regulation of intracellular cyclooxygenase levels by gene transcription and protein degradation. Prog. Lipid Res. 2007, 46, 108–125. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Waxman, L.; Goldberg, A.L. Vanadate inhibits the ATP-dependent degradation of proteins in reticulocytes without affecting ubiquitin conjugation. J. Biol. Chem. 1984, 259, 2803–2809. [Google Scholar] [PubMed]
- Kanayama, H.O.; Tamura, T.; Ugai, S.; Kagawa, S.; Tanahashi, N.; Yoshimura, T.; Tanaka, K.; Ichihara, A. Demonstration that a human 26S proteolytic complex consists of a proteasome and multiple associated protein components and hydrolyzes ATP and ubiquitin-ligated proteins by closely linked mechanisms. Eur. J. Biochem. 1992, 206, 567–578. [Google Scholar] [CrossRef] [PubMed]
- Misra, A.; Srivastava, S.; Ankireddy, S.R.; Islam, N.S.; Chandra, T.; Kumar, A.; Barthwal, M.K.; Dikshit, M. Phospholipase C-γ2 via p38 and ERK1/2 MAP kinase mediates diperoxovanadate-asparagine induced human platelet aggregation and sCD40L release. Redox Rep. 2013, 18, 174–185. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murakami, M.; Kudo, I. Phospholipase A2. J. Biochem. 2002, 131, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Goldman, R.; Ferber, E.; Zor, U. Involvement of reactive oxygen species in phospholipase A2 activation: Inhibition of protein tyrosine phosphatases and activation of protein kinases. Adv. Exp. Med. Biol. 1997, 400A, 25–30. [Google Scholar]
- Helgadóttir, A.; Halldórsson, H.; Magnúsdóttir, K.; Kjeld, M.; Thorgeirsson, G. A role for tyrosine phosphorylation in generation of inositol phosphates and prostacyclin production in endothelial cells. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Varecka, L.; Peterajová, E.; Sevcík, J. Vanadate changes Ca2+ influx pathway properties in human red blood cells. Gen. Physiol. Biophys. 1997, 16, 359–372. [Google Scholar] [PubMed]
- Törnquist, K.; Dugué, B.; Ekokoski, E. Protein tyrosine phosphorylation and calcium signaling in thyroid FRTL-5 cells. J. Cell. Physiol. 1998, 175, 211–219. [Google Scholar] [CrossRef]
- Randazzo, P.A.; Olshan, J.S.; Bijivi, A.A.; Jarett, L. The effect of orthovanadate on phosphoinositide metabolism in NIH 3T3 fibroblasts. Arch. Biochem. Biophys. 1992, 292, 258–265. [Google Scholar] [CrossRef]
- Bianchini, L.; Todderud, G.; Grinstein, S. Cytosolic [Ca2+] homeostasis and tyrosine phosphorylation of phospholipase C gamma 2 in HL60 granulocytes. J. Biol. Chem. 1993, 268, 3357–3363. [Google Scholar] [PubMed]
- Ohmori, T.; Yatomi, Y.; Wu, Y.; Osada, M.; Satoh, K.; Ozaki, Y. Wheat germ agglutinin-induced platelet activation via platelet endothelial cell adhesion molecule-1: Involvement of rapid phospholipase C gamma 2 activation by Src family kinases. Biochemistry 2001, 40, 12992–13001. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Ryu, S.H.; Suh, P.G. On/off-regulation of phospholipase C-gamma 1-mediated signal transduction. Adv. Enzym. Regul. 2007, 47, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Machide, M.; Kamitori, K.; Kohsaka, S. Hepatocyte growth factor-induced differential activation of phospholipase cgamma 1 and phosphatidylinositol 3-kinase is regulated by tyrosine phosphatase SHP-1 in astrocytes. J. Biol. Chem. 2000, 275, 31392–31398. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, N.; Shimohama, S.; Hayakawa, T.; Sumida, Y.; Fujimoto, S. Tyrosine phosphorylation and translocation of phospholipase C-gamma 2 in polymorphonuclear leukocytes treated with pervanadate. Biochim. Biophys. Acta 1996, 1314, 167–174. [Google Scholar] [CrossRef]
- Irani, K.; Pham, Y.; Coleman, L.D.; Roos, C.; Cooke, G.E.; Miodovnik, A.; Karim, N.; Wilhide, C.C.; Bray, P.F.; Goldschmidt-Clermont, P.J. Priming of platelet alphaIIbbeta3 by oxidants is associated with tyrosine phosphorylation of beta3. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 1698–706. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shanmugam, N.; Gaw Gonzalo, I.T.; Natarajan, R. Molecular mechanisms of high glucose-induced cyclooxygenase-2 expression in monocytes. Diabetes 2004, 53, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Kordowiak, A.M.; Klein, A.; Goc, A.; Dabroś, W. Comparison of the effect of VOSO4, Na3VO4 and NaVO3 on proliferation, viability and morphology of H35-19 rat hepatoma cell line. Pol. J. Pathol. 2007, 58, 51–57. [Google Scholar] [PubMed]
- Cuesta, S.; Francés, D.; García, G.B. ROS formation and antioxidant status in brain areas of rats exposed to sodium metavanadate. Neurotoxicol. Teratol. 2011, 33, 297–302. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korbecki, J.; Baranowska-Bosiacka, I.; Gutowska, I.; Chlubek, D. Vanadium Compounds as Pro-Inflammatory Agents: Effects on Cyclooxygenases. Int. J. Mol. Sci. 2015, 16, 12648-12668. https://doi.org/10.3390/ijms160612648
Korbecki J, Baranowska-Bosiacka I, Gutowska I, Chlubek D. Vanadium Compounds as Pro-Inflammatory Agents: Effects on Cyclooxygenases. International Journal of Molecular Sciences. 2015; 16(6):12648-12668. https://doi.org/10.3390/ijms160612648
Chicago/Turabian StyleKorbecki, Jan, Irena Baranowska-Bosiacka, Izabela Gutowska, and Dariusz Chlubek. 2015. "Vanadium Compounds as Pro-Inflammatory Agents: Effects on Cyclooxygenases" International Journal of Molecular Sciences 16, no. 6: 12648-12668. https://doi.org/10.3390/ijms160612648
APA StyleKorbecki, J., Baranowska-Bosiacka, I., Gutowska, I., & Chlubek, D. (2015). Vanadium Compounds as Pro-Inflammatory Agents: Effects on Cyclooxygenases. International Journal of Molecular Sciences, 16(6), 12648-12668. https://doi.org/10.3390/ijms160612648






