HbA1c and Glucose Management Indicator Discordance Associated with Obesity and Type 2 Diabetes in Intermittent Scanning Glucose Monitoring System
Abstract
:1. Introduction
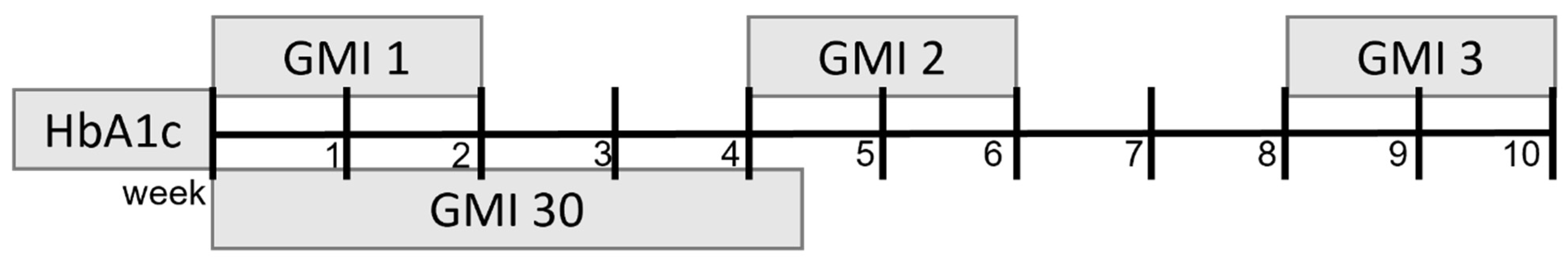
2. Materials and Methods
Statistical Analysis
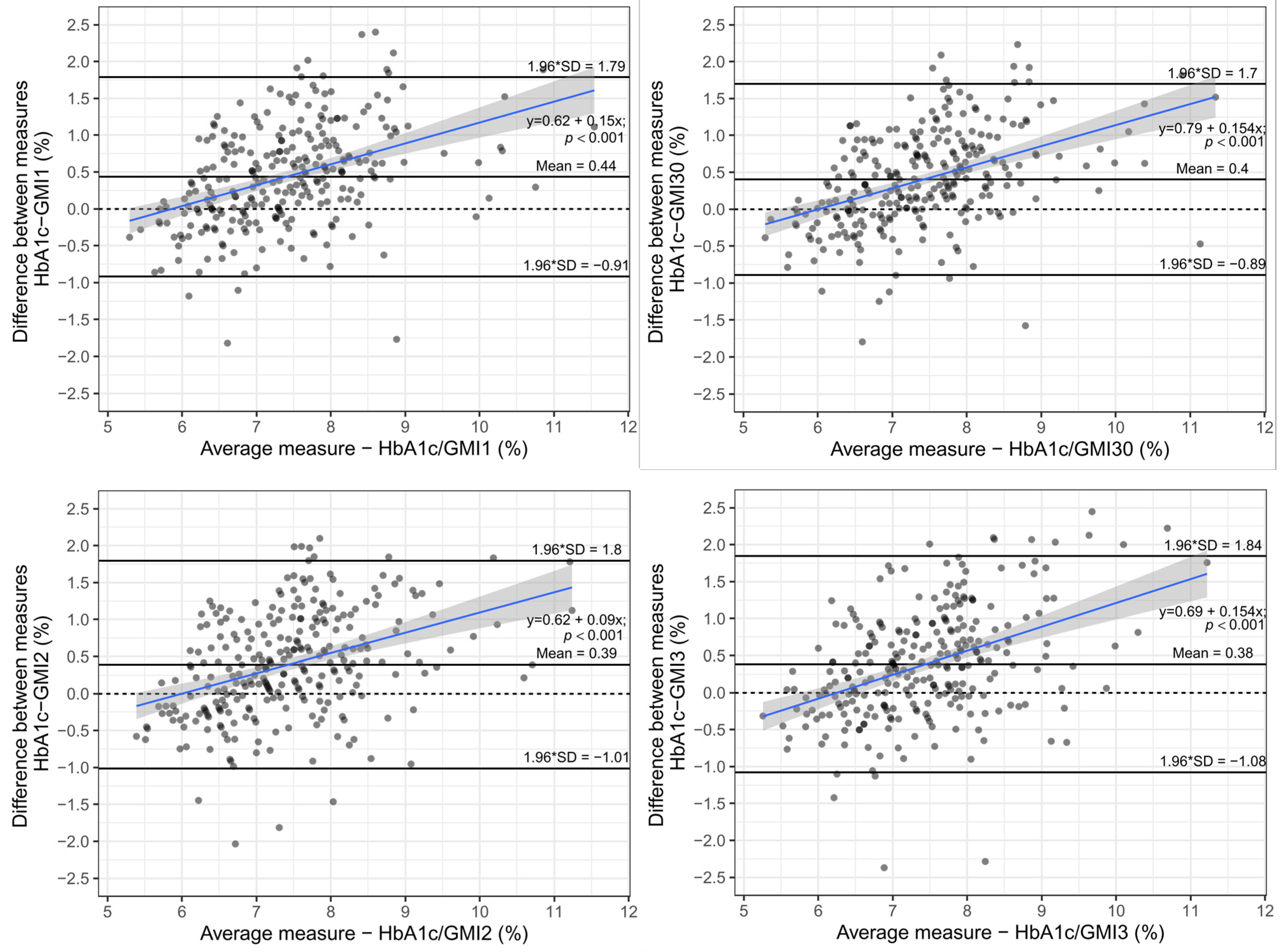
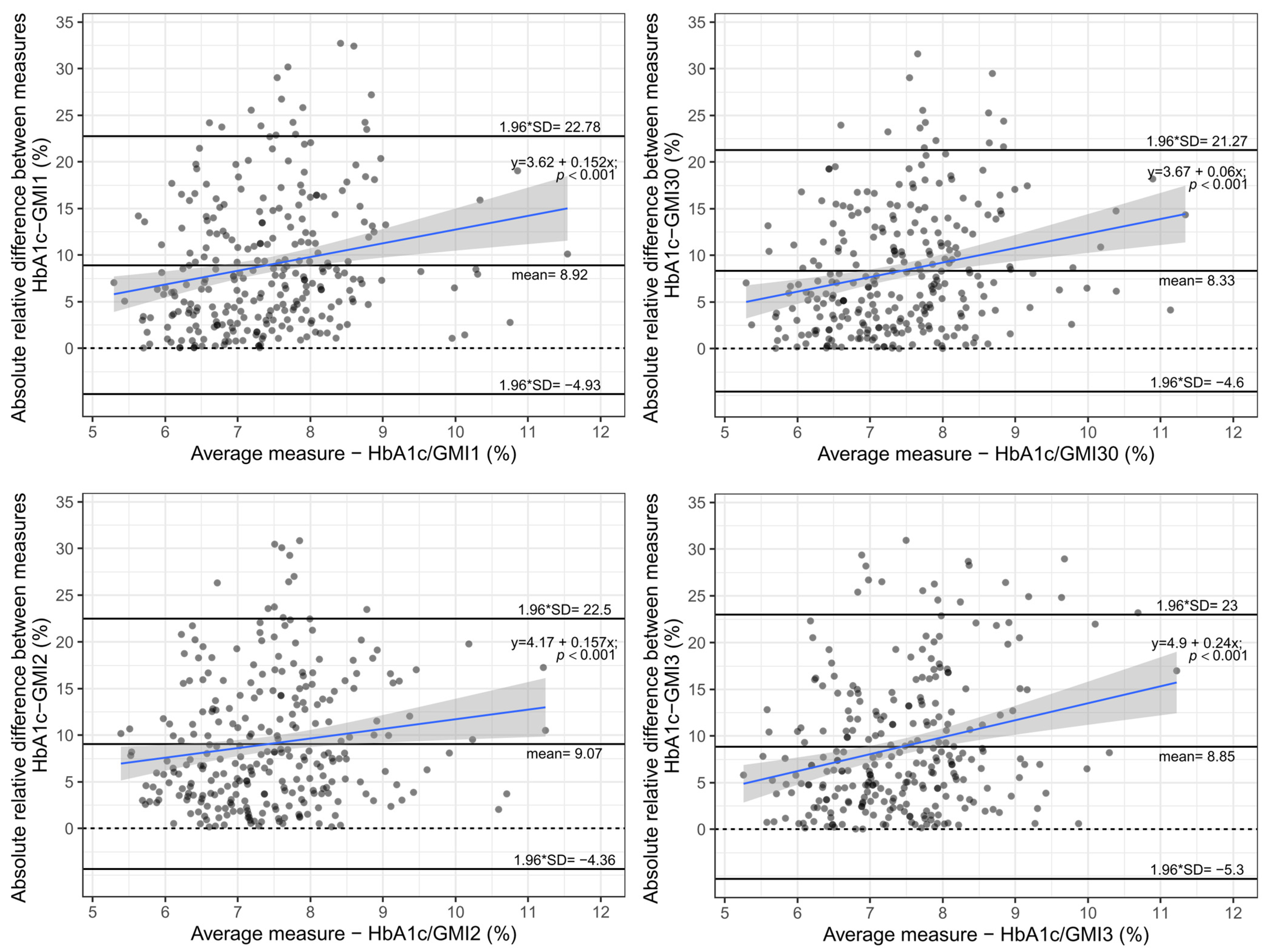
3. Results
3.1. Quality of Glycaemic Control
3.2. Analysis of Differences
3.3. Regression Analysis
4. Discussion
Limitations/Strengths
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tyndall, V.; Stimson, R.H.; Zammitt, N.N.; Ritchie, S.A.; McKnight, J.A.; Dover, A.R.; Gibb, F.W. Marked Improvement in HbA1c Following Commencement of Flash Glucose Monitoring in People with Type 1 Diabetes. Diabetologia 2019, 62, 1349–1356. [Google Scholar] [CrossRef] [Green Version]
- Raj, R.; Mishra, R.; Jha, N.; Joshi, V.; Correa, R.; Kern, P.A. Time in Range, as Measured by Continuous Glucose Monitor, as a Predictor of Microvascular Complications in Type 2 Diabetes: A Systematic Review. BMJ Open Diabetes Res. Care 2022, 10, e002573. [Google Scholar] [CrossRef] [PubMed]
- Bergenstal, R.M.; Beck, R.W.; Close, K.L.; Grunberger, G.; Sacks, D.B.; Kowalski, A.; Brown, A.S.; Heinemann, L.; Aleppo, G.; Ryan, D.B.; et al. Glucose Management Indicator (GMI): A New Term for Estimating A1C From Continuous Glucose Monitoring. Diabetes Care 2018, 41, 2275–2280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riddlesworth, T.D.; Beck, R.W.; Gal, R.L.; Connor, C.G.; Bergenstal, R.M.; Lee, S.; Willi, S.M. Optimal Sampling Duration for Continuous Glucose Monitoring to Determine Long-Term Glycemic Control. Diabetes Technol. Ther. 2018, 20, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Aberer, F.; Hochfellner, D.A.; Mader, J.K. Application of Telemedicine in Diabetes Care: The Time Is Now. Diabetes Ther. 2021, 12, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Perlman, J.E.; Gooley, T.A.; McNulty, B.; Meyers, J.; Hirsch, I.B. HbA1c and Glucose Management Indicator Discordance: A Real-World Analysis. Diabetes Technol. Ther. 2021, 23, 253–258. [Google Scholar] [CrossRef]
- Yamada, M.; Okada, S.; Oda, H.; Nakajima, Y.; Bastie, C.C.; Kasai, Y.; Osaki, A.; Shimoda, Y.; Shibusawa, R.; Uehara, R.; et al. Evaluation of the Relationship between Glycated Hemoglobin A1c and Mean Glucose Levels Derived from the Professional Continuous Flash Glucose Monitoring System. Endocr. J. 2020, 67, 531–536. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Peralta, F.; Choudhary, P.; Cosson, E.; Irace, C.; Rami-Merhar, B.; Seibold, A. Understanding the Clinical Implications of Differences between Glucose Management Indicator and Glycated Haemoglobin. Diabetes Obes. Metab. 2022, 24, 599–608. [Google Scholar] [CrossRef]
- Beck, R.W.; Connor, C.G.; Mullen, D.M.; Wesley, D.M.; Bergenstal, R.M. The Fallacy of Average: How Using HbA1c Alone to Assess Glycemic Control Can Be Misleading. Diabetes Care 2017, 40, 994–999. [Google Scholar] [CrossRef] [Green Version]
- Hansen, K.W.; Bibby, B.M. Glycemic Metrics Derived From Intermittently Scanned Continuous Glucose Monitoring. J. Diabetes Sci. Technol. 2020, 16, 113–119. [Google Scholar] [CrossRef]
- Cohen, R.M.; Franco, R.S.; Khera, P.K.; Smith, E.P.; Lindsell, C.J.; Ciraolo, P.J.; Palascak, M.B.; Joiner, C.H. Red Cell Life Span Heterogeneity in Hematologically Normal People Is Sufficient to Alter HbA1c. Blood 2008, 112, 4284–4291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grimsmann, J.M.; Sengbusch, S.; von Freff, M.; Ermer, U.; Placzek, K.; Danne, T.; Hammer, E.; Holl, R.W. Glucose Management Indicator Based on Sensor Data and Laboratory HbA1c in People With Type 1 Diabetes From the DPV Database: Differences by Sensor Type. Diabetes Care 2020, 43, e111–e112. [Google Scholar] [CrossRef] [PubMed]
- Ólafsdóttir, A.F.; Attvall, S.; Sandgren, U.; Dahlqvist, S.; Pivodic, A.; Skrtic, S.; Theodorsson, E.; Lind, M. A Clinical Trial of the Accuracy and Treatment Experience of the Flash Glucose Monitor FreeStyle Libre in Adults with Type 1 Diabetes. Diabetes Technol. Ther. 2017, 19, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Freckmann, G.; Link, M.; Pleus, S.; Westhoff, A.; Kamecke, U.; Haug, C. Measurement Performance of Two Continuous Tissue Glucose Monitoring Systems Intended for Replacement of Blood Glucose Monitoring. Diabetes Technol. Ther. 2018, 20, 541–549. [Google Scholar] [CrossRef]
- Petrofsky, J.S.; McLellan, K.; Prowse, M.; Bains, G.; Berk, L.; Lee, S. The Effect of Body Fat, Aging, and Diabetes on Vertical and Shear Pressure in and under a Waist Belt and Its Effect on Skin Blood Flow. Diabetes Technol. Ther. 2010, 12, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Mensh, B.D.; Wisniewski, N.A.; Neil, B.M.; Burnett, D.R. Susceptibility of Interstitial Continuous Glucose Monitor Performance to Sleeping Position. J. Diabetes Sci. Technol. 2013, 7, 863–870. [Google Scholar] [CrossRef] [Green Version]
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223. [Google Scholar] [CrossRef] [Green Version]
- Didyuk, O.; Econom, N.; Guardia, A.; Livingston, K.; Klueh, U. Continuous Glucose Monitoring Devices: Past, Present, and Future Focus on the History and Evolution of Technological Innovation. J. Diabetes Sci. Technol. 2020, 15, 676–683. [Google Scholar] [CrossRef]
- Xu, Y.; Grimsmann, J.M.; Karges, B.; Hofer, S.; Danne, T.; Holl, R.W.; Ajjan, R.A.; Dunn, T.C. Personal Glycation Factors and Calculated Hemoglobin A1c for Diabetes Management: Real-World Data from the Diabetes Prospective Follow-up (DPV) Registry. Diabetes Technol. Ther. 2021, 23, 452–459. [Google Scholar] [CrossRef]
- Wright, L.A.-C.; Hirsch, I.B. Metrics Beyond Hemoglobin A1C in Diabetes Management: Time in Range, Hypoglycemia, and Other Parameters. Diabetes Technol. Ther. 2017, 19, S-16–S-26. [Google Scholar] [CrossRef] [Green Version]
- Pleus, S.; Kamecke, U.; Waldenmaier, D.; Link, M.; Zschornack, E.; Jendrike, N.; Haug, C.; Freckmann, G. Time in Specific Glucose Ranges, Glucose Management Indicator, and Glycemic Variability: Impact of Continuous Glucose Monitoring (CGM) System Model and Sensor on CGM Metrics. J. Diabetes Sci. Technol. 2020, 15, 1104–1110. [Google Scholar] [CrossRef] [PubMed]
- Battelino, T.; Danne, T.; Bergenstal, R.M.; Amiel, S.A.; Beck, R.; Biester, T.; Bosi, E.; Buckingham, B.A.; Cefalu, W.T.; Close, K.L.; et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations From the International Consensus on Time in Range. Diabetes Care 2019, 42, 1593–1603. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Variable | Mean ± Standard Deviations |
|---|---|
| N = 278 | |
| Sex (f/m) | 47.5%/52.5% |
| Age (y) | 54.8 ± 15.75 |
| BMI (kg/m2) | 27.94 ± 6.14 |
| RR sys (mmHg) | 139.05 ± 20.63 |
| RR dia (mmHg) | 83.13 ± 12.24 |
| Type diabetes | |
| Type 1 | 155 (55.76%) |
| Type 2 | 91 (32.7%) |
| other types of diabetes | 32 (11.5%) |
| Therapy for diabetes | |
| no insulin | 4 (1.44%) |
| basal only | 13 (4.68%) |
| basal + prandial | 230 (82.73%) |
| Insulin pump | 26 (9.35%) |
| Only SA insulin | 1 (0.36%) |
| Mix-Insulin | 4 (1.44%) |
| Metformin | 71 (25.54%) |
| HbA1c (mmol/mol) | 59.87 ± 13.01 |
| HbA1c (%) | 7.63 ± 1.19 |
| HbA1c <7% vs. >7% | 82 (29.5%) vs. 196 (70.5%) |
| duration of diabetes (n194; y) | 19.68 ± 14.77 (span: 0.3–66 y) |
| Mean ± SD | |
|---|---|
| GMI1 (-d14-d0) | 7.19 ± 0.92 |
| Mean glucose (-d14-d0) | 162.23 ± 38.39 |
| Time in range (%) | 60.71 ± 21.53 |
| TIR >70% vs. <70% of time | 99 (35.6%) vs. 179 (64.4%) |
| Time above range (%) | 34.66 ± 22.51 |
| Time below range (%) | 4.59 ± 5.14 |
| Time below range >4% of time vs. <4% of time | 127 (45.7%) vs. 151 (54.3%) |
| CV (%) | 35 ± 9 |
| GMI30 (-30d-d0) | 7.22 ± 0.91 |
| Mean glucose 30 (-30d-d0) | 163.63 ± 38.21 |
| GMI2 (-6w/-4w) | 7.24 ± 0.93 |
| Mean glucose2 (-6w/-4w) | 164.13 ± 38.83 |
| GMI3 (-10w/-8w) | 7.25 ± 0.89 |
| Mean glucose 3 (-10w/-8w) | 164.51 ± 37.22 |
| R | p-Value | |
|---|---|---|
| Correlation GMI1/HbA1c | 0.82 | <0.001 |
| Correlation GMI30/HbA1c | 0.84 | <0.001 |
| Correlation GMI2/HbA1c | 0.80 | <0.001 |
| Correlation GMI3/HbA1c | 0.78 | <0.001 |
| Estimate | 95% CI (Upper/Lower Limit) | R-Squared | p-Value | |
|---|---|---|---|---|
| Model statistic | 0.1344 | 9.22 × 10−5 | ||
| Intercept | 2.415 | 8.38/13.21 | ||
| Age | 0.004 | −5.2 × 10−5/0.01 | 0.053 | |
| BMI | 0.012 | 0.001/0.022 | 0.029 | |
| Sex | 0.03 | −0.086/0.146 | 0.61 | |
| Type 2 diabetes as factor | 0.16 | 8.286 × 10−4/0.322 | 0.0488 | |
| Type 3 diabetes as factor | −0.02 | −0.222/0.172 | 0.817 | |
| CV | 0.875 | −0.107/1.856 | 0.080 | |
| TiR | −0.028 | −0.136/0.08 | 0.610 | |
| TaR | −0.028 | −0.136/0.079 | 0.605 | |
| TbR | −0.027 | −0.136/0.082 | 0.626 | |
| Absolute difference GMI1/GMI2 | 0.037 | −0.16/0.234 | 0.71 | |
| Absolute difference GMI1/GMI3 | 0.218 | 0.059/0.378 | 0.0074 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fellinger, P.; Rodewald, K.; Ferch, M.; Itariu, B.; Kautzky-Willer, A.; Winhofer, Y. HbA1c and Glucose Management Indicator Discordance Associated with Obesity and Type 2 Diabetes in Intermittent Scanning Glucose Monitoring System. Biosensors 2022, 12, 288. https://doi.org/10.3390/bios12050288
Fellinger P, Rodewald K, Ferch M, Itariu B, Kautzky-Willer A, Winhofer Y. HbA1c and Glucose Management Indicator Discordance Associated with Obesity and Type 2 Diabetes in Intermittent Scanning Glucose Monitoring System. Biosensors. 2022; 12(5):288. https://doi.org/10.3390/bios12050288
Chicago/Turabian StyleFellinger, Paul, Karin Rodewald, Moritz Ferch, Bianca Itariu, Alexandra Kautzky-Willer, and Yvonne Winhofer. 2022. "HbA1c and Glucose Management Indicator Discordance Associated with Obesity and Type 2 Diabetes in Intermittent Scanning Glucose Monitoring System" Biosensors 12, no. 5: 288. https://doi.org/10.3390/bios12050288
APA StyleFellinger, P., Rodewald, K., Ferch, M., Itariu, B., Kautzky-Willer, A., & Winhofer, Y. (2022). HbA1c and Glucose Management Indicator Discordance Associated with Obesity and Type 2 Diabetes in Intermittent Scanning Glucose Monitoring System. Biosensors, 12(5), 288. https://doi.org/10.3390/bios12050288






