The Role of Plasma Interleukin-6 Levels on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk Scores in Javanese Patients with Chronic Kidney Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Criteria
2.3. Plasma IL-6 Levels
2.4. DNA Isolation and Genotyping of IL-6 Gene SNP
2.5. Data Analysis
3. Results
3.1. Characteristics of the Participants
3.2. Correlation and Path Analysis between Plasma IL-6 Level, ASCVD Risk Score, and Cardiovascular Mortality Risk Score
3.3. Analysis of IL-6 G174C SNP in Javanese CKD Patients
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levey, A.S.; Becker, C.; Inker, L.A. Glomerular filtration rate and albuminuria for detection and staging of acute and chronic kidney disease in adults: A systematic review. JAMA 2015, 313, 837–846. [Google Scholar] [CrossRef]
- Levin, A.; Stevens, P.E.; Bilous, R.W.; Coresh, J.; De Francisco, A.L.M.; De Jong, P.E.; Griffith, K.E.; Hemmelgarn, B.R.; Iseki, K.; Lamb, E.J.; et al. Kidney disease: Improving global outcomes (KDIGO) CKD work group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 1–150. [Google Scholar] [CrossRef]
- Lv, J.-C.; Zhang, L.-X. Prevalence and Disease Burden of Chronic Kidney Disease. In Advances in Experimental Medicine and Biology; Springer: Singapore, 2019; Volume 1165, pp. 3–15. [Google Scholar] [CrossRef]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017 : A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Suriyong, P.; Ruengorn, C.; Shayakul, C.; Anantachoti, P.; Kanjanarat, P. Prevalence of chronic kidney disease stages 3–5 in low- and middle-income countries in Asia: A systematic review and meta-analysis. PLoS ONE 2022, 17, e0264393. [Google Scholar] [CrossRef]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 Diseases and Injuries for 195 countries and territories, 1990-2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Cockwell, P.; Fisher, L.A. The global burden of chronic kidney disease. Lancet 2020, 395, 662–664. [Google Scholar] [CrossRef]
- Rangaswami, J.; Bhalla, V.; Blair, J.E.; Chang, T.I.; Costa, S.; Lentine, K.L.; Lerma, E.V.; Mezue, K.; Molitch, M.; Mullens, W.; et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation 2019, 139, E840–E878. [Google Scholar] [CrossRef]
- Van Der Valk, F.M.; Van Wijk, D.F.; Stroes, E.S.G. Novel anti-inflammatory strategies in atherosclerosis. Curr. Opin. Lipidol. 2012, 23, 532–539. [Google Scholar] [CrossRef]
- Hartman, J.; Frishman, W.H. Inflammation and atherosclerosis: A review of the role of interleukin-6 in the development of atherosclerosis and the potential for targeted drug therapy. Cardiol. Rev. 2014, 22, 147–151. [Google Scholar] [CrossRef]
- Suzuki, S.; Tanaka, K.; Suzuki, N. Ambivalent Aspects of Interleukin-6 in Cerebral Ischemia: Inflammatory versus Neurotrophic Aspects. J. Cereb. Blood Flow Metab. 2009, 29, 464–479. [Google Scholar] [CrossRef]
- Didion, S. Cellular and Oxidative Mechanisms Associated with Interleukin-6 Signaling in the Vasculature. Int. J. Mol. Sci. 2017, 18, 2563. [Google Scholar] [CrossRef]
- Salari, N.; Mansouri, K.; Hosseinian-Far, A.; Ghasemi, H.; Mohammadi, M.; Jalali, R.; Vaisi-Raygani, A. The effect of polymorphisms (174G > C and 572C > G) on the Interleukin-6 gene in coronary artery disease: A systematic review and meta-analysis. Genes Environ. 2021, 43, 1. [Google Scholar] [CrossRef]
- Satti, H.S.; Hussain, S.; Javed, Q. Association of Interleukin-6 Gene Promoter Polymorphism with Coronary Artery Disease in Pakistani Families. Sci. World J. 2013, 2013, 538365. [Google Scholar] [CrossRef]
- Fishman, D.; Faulds, G.; Jeffery, R.; Mohamed-Ali, V.; Yudkin, J.S.; Humphries, S.; Woo, P. The effect of novel polymorphisms in the interleukin-6 (IL-6) gene on IL-6 transcription and plasma IL-6 levels, and an association with systemic-onset juvenile chronic arthritis. J. Clin. Investig. 1998, 102, 1369–1376. [Google Scholar] [CrossRef]
- Zakharyan, R.; Petrek, M.; Arakelyan, A.; Mrazek, F.; Atshemyan, S.; Boyajyan, A. Interleukin-6 promoter polymorphism and plasma levels in patients with schizophrenia. Tissue Antigens 2012, 80, 136–142. [Google Scholar] [CrossRef]
- Gager, G.M.; Biesinger, B.; Hofer, F.; Winter, M.-P.; Hengstenberg, C.; Jilma, B.; Eyileten, C.; Postula, M.; Lang, I.M.; Siller-Matula, J.M. Interleukin-6 level is a powerful predictor of long-term cardiovascular mortality in patients with acute coronary syndrome. Vasc. Pharm. 2020, 135, 106806. [Google Scholar] [CrossRef]
- Volpato, S.; Guralnik, J.M.; Ferrucci, L.; Balfour, J.; Chaves, P.; Fried, L.P.; Harris, T.B. Cardiovascular Disease, Interleukin-6, and Risk of Mortality in Older Women: The Women’s Health and Aging Study. Low. Extrem. 2001, 103, 947–953. [Google Scholar] [CrossRef]
- Spoto, B.; Mattace-Raso, F.; Sijbrands, E.; Leonardis, D.; Testa, A.; Pisano, A.; Pizzini, P.; Cutrupi, S.; Parlongo, R.M.; D’Arrigo, G.; et al. Association of IL-6 and a functional polymorphism in the IL-6 gene with cardiovascular events in patients with CKD. Clin. J. Am. Soc. Nephrol. 2015, 10, 232–240. [Google Scholar] [CrossRef]
- Su, H.; Lei, C.T.; Zhang, C. Interleukin-6 signaling pathway and its role in kidney disease: An update. Front. Immunol. 2017, 8, 405. [Google Scholar] [CrossRef]
- Rauramaa, R.; Väisänen, S.B.; Luong, L.-A.; Schmidt-Trucksäss, A.; Penttilä, I.M.; Bouchard, C.; Töyry, J.; Humphries, S.E. Stromelysin-1 and interleukin-6 gene promoter polymorphisms are determinants of asymptomatic carotid artery atherosclerosis. Arter. Thromb. Vasc. Biol. 2000, 20, 2657–2662. [Google Scholar] [CrossRef]
- Rundek, T.; Elkind, M.S.; Pittman, J.; Boden-Albala, B.; Martin, S.; Humphries, S.E.; Juo, S.-H.H.; Sacco, R.L. Carotid intima-media thickness is associated with allelic variants of stromelysin-1, interleukin-6, and hepatic lipase genes: The Northern Manhattan Prospective Cohort Study. Stroke 2002, 33, 1420–1423. [Google Scholar] [CrossRef]
- McColgan, P.; Thant, K.Z.; Sharma, P. The genetics of sporadic ruptured and unruptured intracranial aneurysms: A genetic meta-analysis of 8 genes and 13 polymorphisms in approximately 20,000 individuals. J. Neurosurg. 2010, 112, 714–721. [Google Scholar] [CrossRef]
- Basso, F.; Lowe, G.D.O.; Rumley, A.; McMahon, A.D.; Humphries, S.E. Interleukin-6 -174G>C polymorphism and risk of coronary heart disease in West of Scotland Coronary Prevention Study (WOSCOPS). Arter. Thromb. Vasc. Biol. 2002, 22, 599–604. [Google Scholar] [CrossRef]
- Aker, S.; Bantis, C.; Reis, P.; Kuhr, N.; Schwandt, C.; Grabensee, B.; Heering, P.; Ivens, K. Influence of interleukin-6 G-174C gene polymorphism on coronary artery disease, cardiovascular complications and mortality in dialysis patients. Nephrol. Dial. Transpl. 2009, 24, 2847–2851. [Google Scholar] [CrossRef]
- Hashad, I.M.; Nosseir, H.; Shaban, G.M.; Abdel Rahman, M.F.; Gad, M.Z. Is there a correlation between -174(G/C) polymorphism of IL-6 gene and the incidence of acute myocardial infarction? J. Genet. Eng. Biotechnol. 2021, 19, 139. [Google Scholar] [CrossRef]
- Kumar, P.; Yadav, A.K.; Kumar, A.; Sagar, R.; Pandit, A.K.; Prasad, K. Association between interleukin-6 (G174C and G572C) promoter gene polymorphisms and risk of ischaemic stroke: A meta-analysis. Ann. Neurosci. 2015, 22, 61–69. [Google Scholar] [CrossRef]
- Gan, G.G.; Subramaniam, R.; Lian, L.H.; Nadarajan, V.S. Ethnic variation in interleukin-6-174 (G/C) polymorphism in the Malaysian population. Balk. J. Med. Genet. 2013, 16, 53–58. [Google Scholar] [CrossRef]
- Sie, M.P.; Sayed-Tabatabaei, F.A.; Oei, H.-H.S.; Uitterlinden, A.G.; Pols, H.A.; Hofman, A.; van Duijn, C.M.; Witteman, J.C. Interleukin 6 -174 G/C promoter polymorphism and risk of coronary heart disease: Results from the Rotterdam study and a meta-analysis. Arter. Thromb. Vasc. Biol. 2006, 26, 212–217. [Google Scholar] [CrossRef]
- Arooj, S.; Khan, M.; Raza, A. Interleukin-6 Gene Promoter Region Polymorphism in Patients with Dilated Cardiomyopathy. Cardiol. Cardiovasc. Med. 2019, 3, 329–339. [Google Scholar] [CrossRef]
- Susilo, H.; Pikir, B.S.; Thaha, M.; Alsagaff, M.Y.; Suryantoro, S.D.; Wungu, C.D.K.; Wafa, I.A.; Pakpahan, C.; Oceandy, D. The Effect of Angiotensin Converting Enzyme (ACE) I/D Polymorphism on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk in Non-Hemodialyzed Chronic Kidney Disease: The Mediating Role of Plasma ACE Level. Genes 2022, 13, 1121. [Google Scholar] [CrossRef]
- Stevens, P.E.; Levin, A. Evaluation and Management of Chronic Kidney Disease: Synopsis of the Kidney Disease: Improving Global Outcomes 2012 Clinical Practice Guideline. Ann. Intern. Med. 2013, 158, 825. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; Jassal, S.K.; Sang, Y.; Ballew, S.H.; E Grams, M.; Surapaneni, A.; Arnlov, J.; Bansal, N.; Bozic, M.; Brenner, H.; et al. Incorporating kidney disease measures into cardiovascular risk prediction: Development and validation in 9 million adults from 72 datasets. EClinicalMedicine 2020, 27, 100552. [Google Scholar] [CrossRef] [PubMed]
- Ghavimi, R.; Sharifi, M.; Mohaghegh, M.A.; Mohammadian, H.; Khadempar, S.; Rezaei, H. Lack of association between rs1800795 (-174 G/C) polymorphism in the promoter region of interleukin-6 gene and susceptibility to type 2 diabetes in Isfahan population. Adv. Biomed. Res. 2016, 5, 18. [Google Scholar] [CrossRef]
- Indonesian Society of Endocrinology. Guidelines on the Management of Dyslipidemia in Indonesia; PB Perkeni: Jakarta, Indonesia, 2019. [Google Scholar]
- Hassan, M.O.; Duarte, R.; Dickens, C.; Dix-Peek, T.; Naidoo, S.; Vachiat, A.; Grinter, S.; Manga, P.; Naicker, S. Interleukin-6 gene polymorhisms and interleukin-6 levels are associated with atherosclerosis in CKD patients. Clin. Nephrol. 2020, 93, S82–S86. [Google Scholar] [CrossRef] [PubMed]
- National Library of Medicine. dbSNP rs1800795 n.d.:2021. Available online: https://www.ncbi.nlm.nih.gov/snp/rs1800795?horizontal_tab=true (accessed on 8 March 2022).
- Herrington, W.; Lacey, B.; Sherliker, P.; Armitage, J.; Lewington, S. Epidemiology of Atherosclerosis and the Potential to Reduce the Global Burden of Atherothrombotic Disease. Circ. Res. 2016, 118, 535–546. [Google Scholar] [CrossRef]
- Wang, Y.; Meng, R.; Liu, G.; Cao, C.; Chen, F.; Jin, K.; Ji, X.; Cao, G. Intracranial atherosclerotic disease. Neurobiol. Dis. 2019, 124, 118–132. [Google Scholar] [CrossRef]
- Weger, M.; Steinbrugger, I.; Haas, A.; März, W.; El-Shabrawi, Y.; Weger, W.; Schmut, O.; Renner, W. Role of the interleukin-6 -174 G>C gene polymorphism in retinal artery occlusion. Stroke 2005, 36, 249–252. [Google Scholar] [CrossRef][Green Version]
- Romano, M.; Sironi, M.; Toniatti, C.; Polentarutti, N.; Fruscella, P.; Ghezzi, P.; Faggioni, R.; Luini, W.; van Hinsbergh, V.; Sozzani, S.; et al. Role of IL-6 and its soluble receptor in induction of chemokines and leukocyte recruitment. Immunity 1997, 6, 315–325. [Google Scholar] [CrossRef]
- Zamani, P.; Schwartz, G.G.; Olsson, A.G.; Rifai, N.; Bao, W.; Libby, P.; Ganz, P.; Kinlay, S.; the Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) Study Investigators. Inflammatory biomarkers, death, and recurrent nonfatal coronary events after an acute coronary syndrome in the MIRACL study. J. Am. Heart Assoc. 2013, 2, e003103. [Google Scholar] [CrossRef]
- Honda, H.; Qureshi, A.R.; Heimbürger, O.; Barany, P.; Wang, K.; Pecoits-Filho, R.; Stenvinkel, P.; Lindholm, B. Serum albumin, C-reactive protein, interleukin 6, and fetuin a as predictors of malnutrition, cardiovascular disease, and mortality in patients with ESRD. Am. J. Kidney Dis. 2006, 47, 139–148. [Google Scholar] [CrossRef]
- Pecoits-Filho, R.; Bárány, P.; Lindholm, B.; Heimbürger, O.; Stenvinkel, P. Interleukin-6 is an independent predictor of mortality in patients starting dialysis treatment. Nephrol. Dial. Transpl. 2002, 17, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Meuwese, C.L.; Snaedal, S.; Halbesma, N.; Stenvinkel, P.; Dekker, F.; Qureshi, A.R.T.; Bárány, P.; Heimburger, O.; Lindholm, B.; Krediet, R.T.; et al. Trimestral variations of C-reactive protein, interleukin-6 and tumour necrosis factor-α are similarly associated with survival in haemodialysis patients. Nephrol. Dial. Transpl. 2011, 26, 1313–1318. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Guo, D.; Perianayagam, M.C.; Tighiouart, H.; Jaber, B.L.; Pereira, B.J.G.; Balakrishnan, V.S. Plasma interleukin-6 predicts cardiovascular mortality in hemodialysis patients. Am. J. Kidney Dis. 2005, 45, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Thaha, M.; Kadariswantiningsih, I.N.; Empitu, M.A. Association of high blood pressure with elevated oxidative stress, inflammatory marker and albuminuria in chronic kidney disease patients. J. Med. 2019, 20, 12–18. [Google Scholar] [CrossRef]
- Cachofeiro, V.; Goicochea, M.; De Vinuesa, S.G.; Oubĩa, P.; Lahera, V.; Lũo, J. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int. 2008, 74, S4–S9. [Google Scholar] [CrossRef]
- Wainstein, M.V.; Mossmann, M.; Araujo, G.N.; Gonçalves, S.C.; Gravina, G.L.; Sangalli, M.; Veadrigo, F.; Matte, R.; Reich, R.; Costa, F.G.; et al. Elevated serum interleukin-6 is predictive of coronary artery disease in intermediate risk overweight patients referred for coronary angiography. Diabetol. Metab. Syndr. 2017, 9, 67. [Google Scholar] [CrossRef]
- Lai, S.; Fishman, E.K.; Lai, H.; Pannu, H.; Detrick, B. Serum IL-6 Levels are Associated with Significant Coronary Stenosis in Cardiovascularly Asymptomatic Inner-City Black Adults in the US. Inflamm. Res. 2009, 58, 15. [Google Scholar] [CrossRef]
- Tuomisto, K.; Jousilahti, P.; Sundvall, J.; Pajunen, P.; Salomaa, V. C-reactive protein, interleukin-6 and tumor necrosis factor alpha as predictors of incident coronary and cardiovascular events and total mortality. A population-based, prospective study. Thromb. Haemost. 2006, 95, 511–518. [Google Scholar] [CrossRef]
- Panichi, V.; Maggiore, U.; Taccola, D.; Migliori, M.; Rizza, G.M.; Consani, C.; Bertini, A.; Sposini, S.; Perez-Garcia, R.; Rindi, P.; et al. Interleukin-6 is a stronger predictor of total and cardiovascular mortality than C-reactive protein in haemodialysis patients. Nephrol. Dial. Transpl. 2004, 19, 1154–1160. [Google Scholar] [CrossRef]
- George, J.; Rapsomaniki, E.; Pujades-Rodriguez, M.; Shah, A.D.; Denaxas, S.; Herrett, E.; Smeeth, L.; Timmis, A.; Hemingway, H. How does cardiovascular disease first present in women and men? Circulation 2015, 132, 1320–1328. [Google Scholar] [CrossRef]
- Leening, M.J.G.; Ferket, B.; Steyerberg, E.W.; Kavousi, M.; Deckers, J.W.; Nieboer, D.; Heeringa, J.; Portegies, M.L.P.; Hofman, A.; Ikram, M.A.; et al. Sex differences in lifetime risk and first manifestation of cardiovascular disease: Prospective population based cohort study. BMJ 2014, 349, g5992. [Google Scholar] [CrossRef] [PubMed]
- Tunstall-Pedoe, H. Myth and paradox of coronary risk and the menopause. Lancet 1998, 351, 1425–1427. [Google Scholar] [CrossRef]
- Howard, N.H.; Mack, W.J. In Perspective: Estrogen Therapy Proves to Safely and Effectively Reduce Total Mortality and Coronary Heart Disease in Recently Postmenopausal Women. Menopause Manag. 2008, 17, 27–32. [Google Scholar]
- Yang, X.-P.; Reckelhoff, J.F. Estrogen, hormonal replacement therapy and cardiovascular disease. Curr. Opin. Nephrol. Hypertens. 2011, 20, 133–138. [Google Scholar] [CrossRef]
- Souza, J.R.M.; Oliveira, R.T.; Blotta, M.H.S.L.; Coelho, O.R. Serum levels of interleukin-6 (Il-6), interleukin-18 (Il-18) and C-reactive protein (CRP) in patients with type-2 diabetes and acute coronary syndrome without ST-segment elevation. Arq. Bras. Cardiol. 2008, 90, 94–99. [Google Scholar] [CrossRef]
- Lee, P.C.; Ho, I.C.; Lee, T.C. Oxidative stress mediates sodium arsenite-induced expression of heme oxygenase-1, monocyte chemoattractant protein-1, and interleukin-6 in vascular smooth muscle cells. Toxicol. Sci. 2005, 85, 541–550. [Google Scholar] [CrossRef]
- Schuett, H.; Luchtefeld, M.; Grothusen, C.; Grote, K.; Schieffer, B. How much is too much? Interleukin-6 and its signalling in atherosclerosis. Thromb. Haemost. 2009, 102, 215–222. [Google Scholar] [CrossRef]
- Nijm, J.; Jonasson, L. Inflammation and cortisol response in coronary artery disease. Ann. Med. 2009, 41, 224–233. [Google Scholar] [CrossRef]
- Vivekanand, J.H.A.; Guillermo, G.; Kunitoshi, I.; Zou, L. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef]
- Hou, H.; Wang, C.; Sun, F.; Zhao, L.; Dun, A.; Sun, Z. Association of interleukin-6 gene polymorphism with coronary artery disease: An updated systematic review and cumulative meta-analysis. Inflamm. Res. 2015, 64, 707–720. [Google Scholar] [CrossRef]
- Song, Y.; Gu, H.D.; He, Y.; Wang, J.W. Role of IL-6 polymorphism on the development of cardiovascular events and coronary artery disease in patients receiving hemodialysis. Genet. Mol. Res. 2015, 14, 2631–2637. [Google Scholar] [CrossRef] [PubMed]
- Chiappelli, M.; Tampieri, C.; Tumini, E.; Porcellini, E.; Caldarera, C.M.; Nanni, S.; Branzi, A.; Lio, D.; Caruso, M.; Hoffmann, E.; et al. Interleukin-6 gene polymorphism is an age-dependent risk factor for myocardial infarction in men. Int. J. Immunogenet. 2005, 32, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Ghazouani, L.; Abboud, N.; Khalifa, S.B.H.; Added, F.; Ben Khalfallah, A.; Nsiri, B.; Mediouni, M.; Mahjoub, T. -174G>C interleukin-6 gene polymorphism in Tunisian patients with coronary artery disease. Ann. Saudi. Med. 2011, 31, 40–44. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tong, Z.; Li, Q.; Zhang, J.; Wei, Y.; Miao, G.; Yang, X. Association between interleukin 6 and interleukin 16 gene polymorphisms and coronary heart disease risk in a Chinese population. J. Int. Med. Res. 2013, 41, 1049–1056. [Google Scholar] [CrossRef]
- Pourhossein, M.; Ghavimi, R.; Alsahebfosoul, F.; Ghaedi, K. Association between rs1800795 (-174 G/C) Polymorphism in the Promoter of IL6 Gene and Risk of Relapsing-Remitting Multiple Sclerosis (RRMS) in Isfahan Population. Open J. Genet. 2014, 4, 407–413. [Google Scholar] [CrossRef][Green Version]
- Meenagh, A.; Williams, F.; A Ross, O.; Patterson, C.; Gorodezky, C.; Hammond, M.; A Leheny, W.; Middleton, D. Frequency of cytokine polymorphisms in populations from western Europe, Africa, Asia, the Middle East and South America. Hum. Immunol. 2002, 63, 1055–1061. [Google Scholar] [CrossRef]
- Pan, M.; Gao, S.P.; Jiang, M.H.; Guo, J.; Zheng, J.G.; Zhu, J.H. Interleukin 6 promoter polymorphisms in normal Han Chinese population: Frequencies and effects on inflammatory markers. J. Investig. Med. 2011, 59, 272–276. [Google Scholar] [CrossRef]
- Muzasti, R.A.; Hariman, H.; Daulay, E.R. The Influence of −174 G/C Interleukin 6 Promoter Gene Polymorphism to Interleukin 6 Concentration in the End Stage Renal Disease Patients with Dialysis: A Single-Center Experience in Indonesia. Open Access Maced. J. Med. Sci. 2020, 8, 563–566. [Google Scholar] [CrossRef]
- Poli, F.; Nocco, A.; Berra, S.; Scalamogna, M.; Taioli, E.; Longhi, E.; Sirchia, G. Allele frequencies of polymorphisms of TNFA, IL-6, IL-10 and IFNG in an Italian Caucasian population. Eur. J. Immunogenet. 2002, 29, 237–240. [Google Scholar] [CrossRef]
- Kurzawski, M.; Pawlik, A.; Czerny, B.; Domański, L.; Rózański, J.; Droździk, M. Frequencies of the common promoter polymorphisms in cytokine genes in a Polish population. Int. J. Immunogenet. 2005, 32, 285–291. [Google Scholar] [CrossRef]
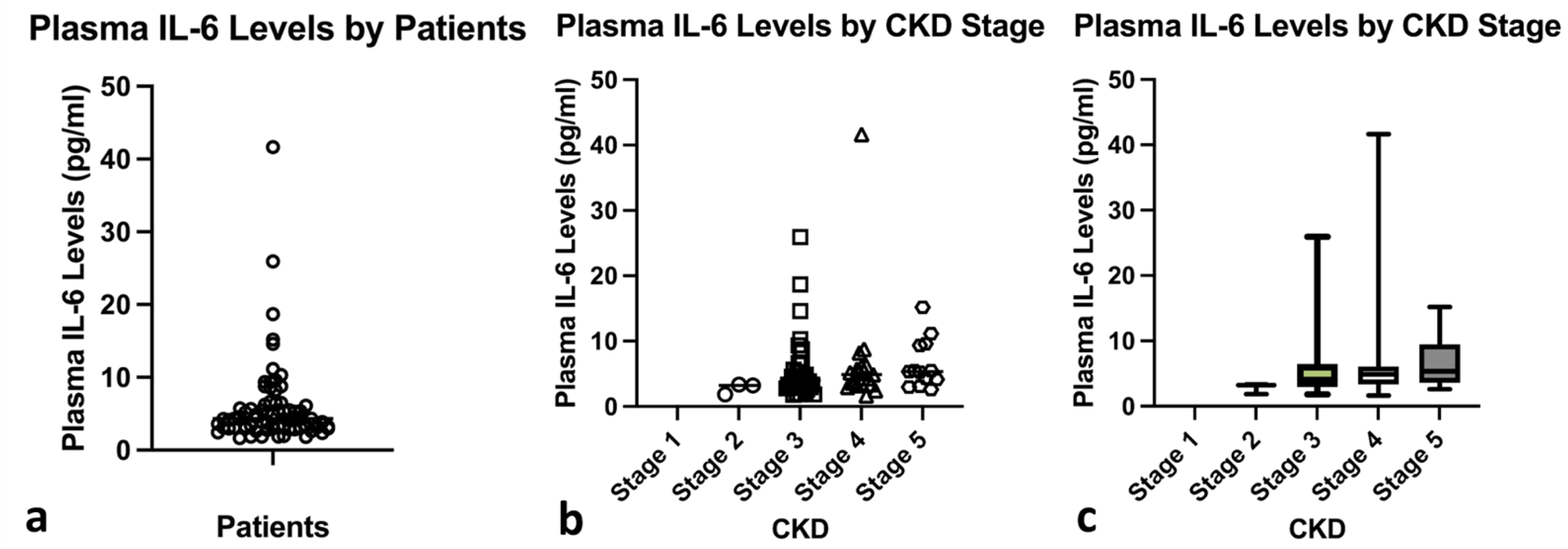
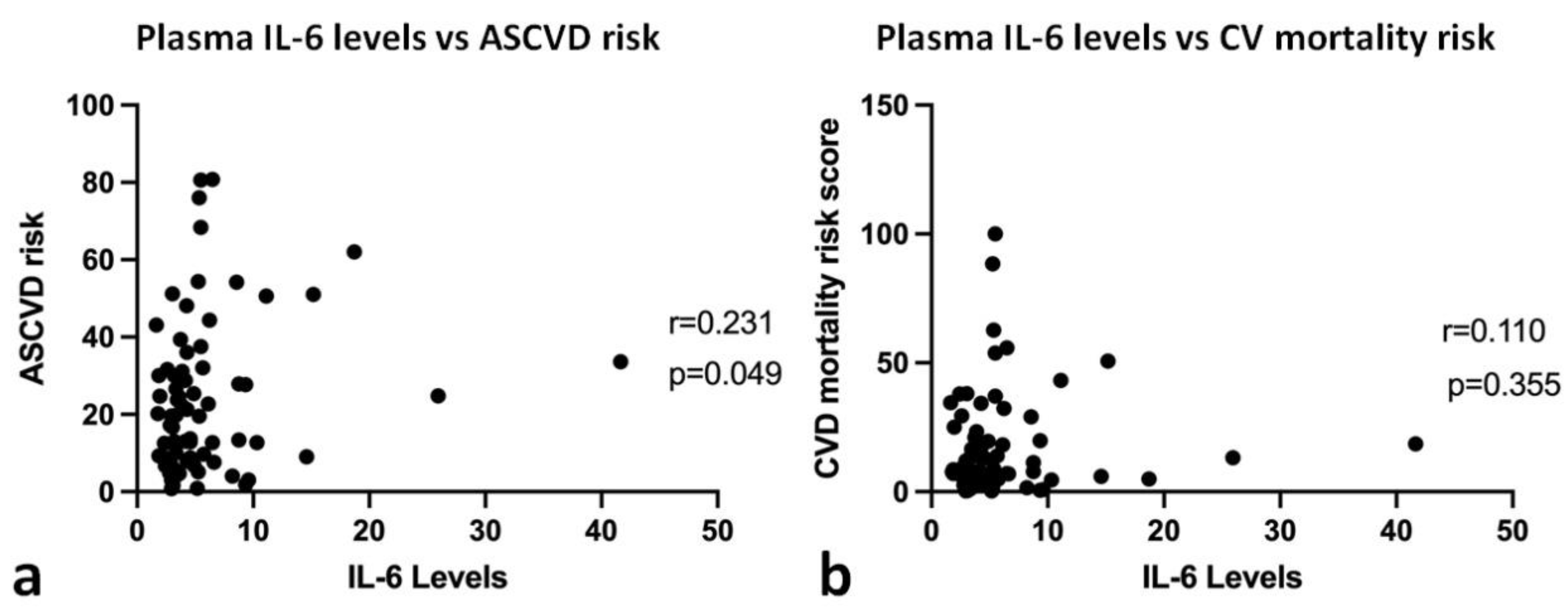
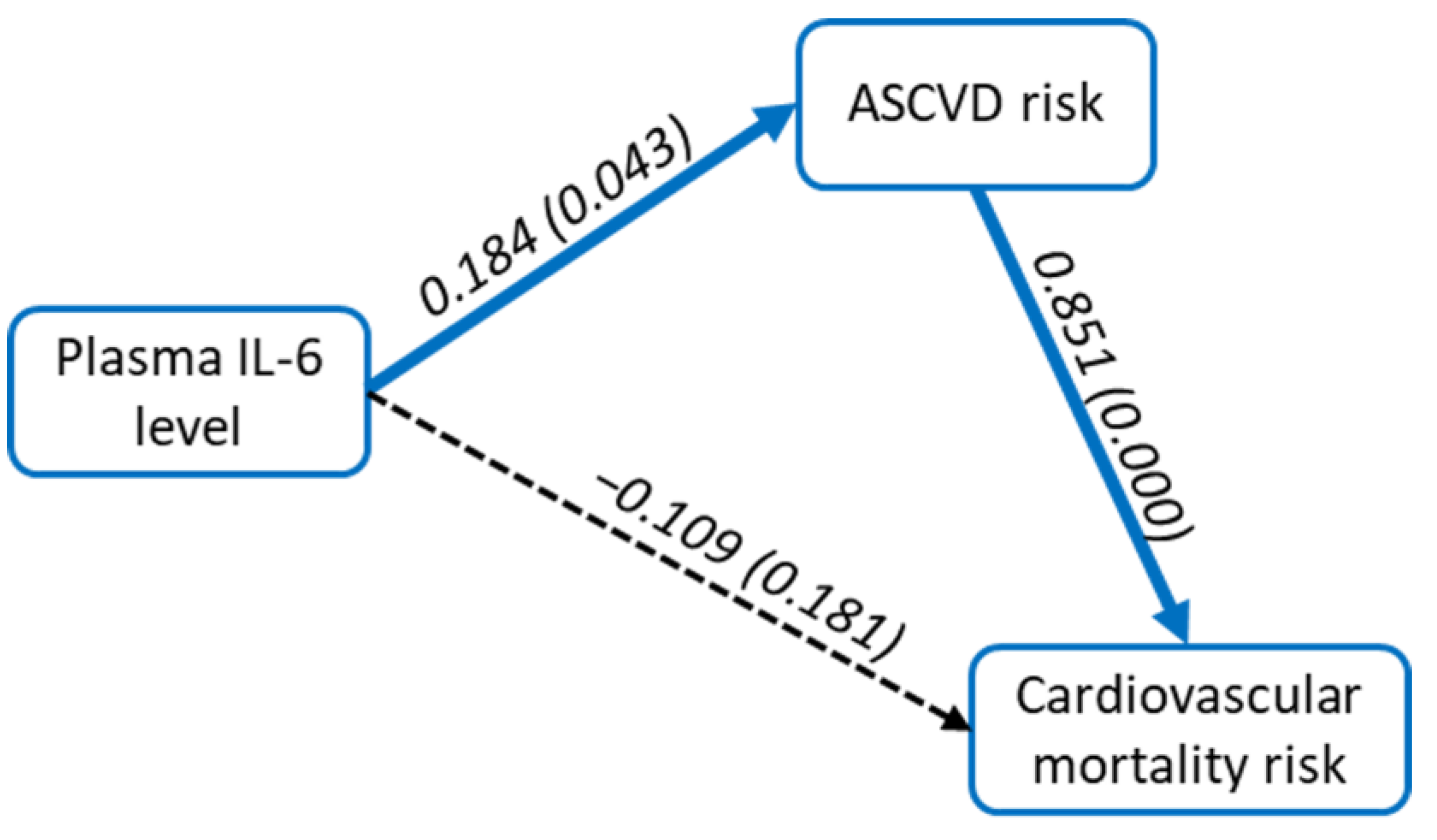

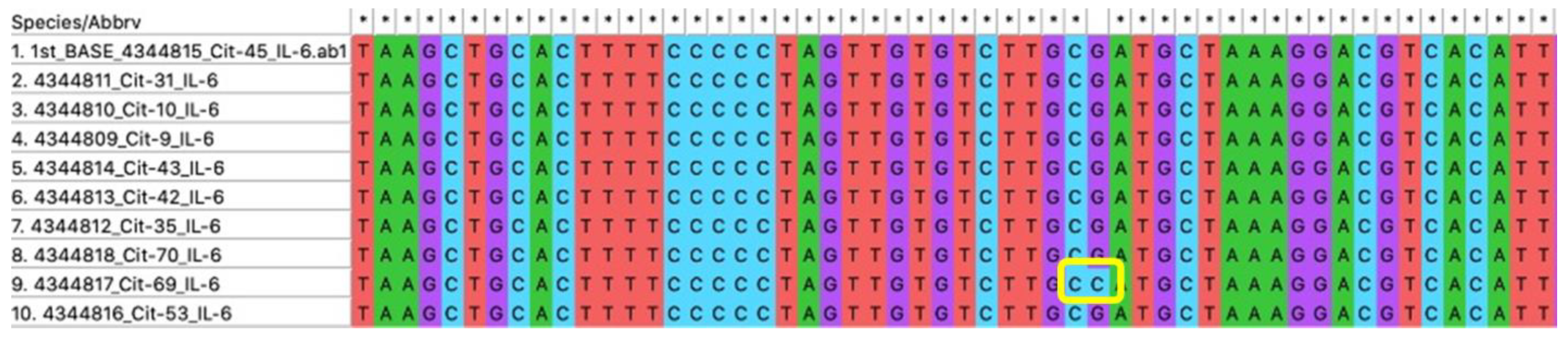
| Variable | Value (n = 73) |
|---|---|
| Gender, male (%) | 38 (52.1) |
| Age (years) | 57.93 ± 7.15 |
| History of type 2 diabetes (%) | 56 (76.7) |
| History of hypertension (%) | 64 (87.7) |
| History of smoking | |
| Non-smoker n (%) | 51 (69.9) |
| Current smoker n (%) | 4 (5.5) |
| Former smoker n (%) | 18 (24.7) |
| Stages of kidney disease | |
| CKD stage 2 n (%) | 3 (4.1) |
| CKD stage 3 n (%) | 38 (52.1) |
| CKD stage 4 n (%) | 20 (27.4) |
| CKD stage 5 n (%) | 12 (16.4) |
| Dyslipidemia n (%) | 58 (79.5%) |
| BMI (Kg/m2) | 26.08 ± 5.16 |
| SBP (mmHg) | 144.27 ± 23.13 |
| DBP (mmHg) | 81.29 ± 11.92 |
| Total cholesterol (mg/dL) | 183.96 ± 52.22 |
| HDL (mg/dL) | 39.75 ± 12.35 |
| Serum creatinine (mg/dL) | 2.64 ± 1.66 |
| e-GFR (mL/min/1.73 m2) | 31.82 ± 14.94 |
| Urine ACR (mg/gram) | 643.33 ± 973.73 |
| Plasma IL-6 (pg/mL) | 5.92 ± 5.83 |
| ASCVD risk score (%) | 23.83 ± 19.82 |
| Cardiovascular mortality risk score (%) | 17.06 ± 19.45 |
| Genotype | n | Frequency (%) | IL-6 Plasma (pg/mL) | ASCVD Risk Score (%) | Cardiovascular Mortality Risk Score (%) | CKD Stage (%) |
|---|---|---|---|---|---|---|
| GG | 71 | 97.26 | 5.48 ± 4.04 | 23.90 ± 19.99 | 17.18 ± 19.74 | Stage 2 (4.2) |
| Stage 3 (52.1) | ||||||
| Stage 4 (26.8) | ||||||
| Stage 5 (16.9) | ||||||
| GC | 1 | 1.37 | 1.874 | 9.30 | 7 | Stage 3 (100) |
| CC | 1 | 1.37 | 41.66 | 33.60 | 18.50 | Stage 4 (100) |
| Total | 73 | 100 | ||||
| Allele | n | Frequency (%) | ||||
| G | 143 | 97.95 | ||||
| C | 3 | 2.05 | ||||
| Total | 146 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Susilo, H.; Thaha, M.; Pikir, B.S.; Alsagaff, M.Y.; Suryantoro, S.D.; Wungu, C.D.K.; Pratama, N.R.; Pakpahan, C.; Oceandy, D. The Role of Plasma Interleukin-6 Levels on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk Scores in Javanese Patients with Chronic Kidney Disease. J. Pers. Med. 2022, 12, 1122. https://doi.org/10.3390/jpm12071122
Susilo H, Thaha M, Pikir BS, Alsagaff MY, Suryantoro SD, Wungu CDK, Pratama NR, Pakpahan C, Oceandy D. The Role of Plasma Interleukin-6 Levels on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk Scores in Javanese Patients with Chronic Kidney Disease. Journal of Personalized Medicine. 2022; 12(7):1122. https://doi.org/10.3390/jpm12071122
Chicago/Turabian StyleSusilo, Hendri, Mochammad Thaha, Budi Susetyo Pikir, Mochamad Yusuf Alsagaff, Satriyo Dwi Suryantoro, Citrawati Dyah Kencono Wungu, Nando Reza Pratama, Cennikon Pakpahan, and Delvac Oceandy. 2022. "The Role of Plasma Interleukin-6 Levels on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk Scores in Javanese Patients with Chronic Kidney Disease" Journal of Personalized Medicine 12, no. 7: 1122. https://doi.org/10.3390/jpm12071122
APA StyleSusilo, H., Thaha, M., Pikir, B. S., Alsagaff, M. Y., Suryantoro, S. D., Wungu, C. D. K., Pratama, N. R., Pakpahan, C., & Oceandy, D. (2022). The Role of Plasma Interleukin-6 Levels on Atherosclerotic Cardiovascular Disease and Cardiovascular Mortality Risk Scores in Javanese Patients with Chronic Kidney Disease. Journal of Personalized Medicine, 12(7), 1122. https://doi.org/10.3390/jpm12071122






