The Anti-Proliferative Activity of the Hybrid TMS-TMF-4f Compound Against Human Cervical Cancer Involves Apoptosis Mediated by STAT3 Inactivation
Abstract
1. Introduction
2. Results
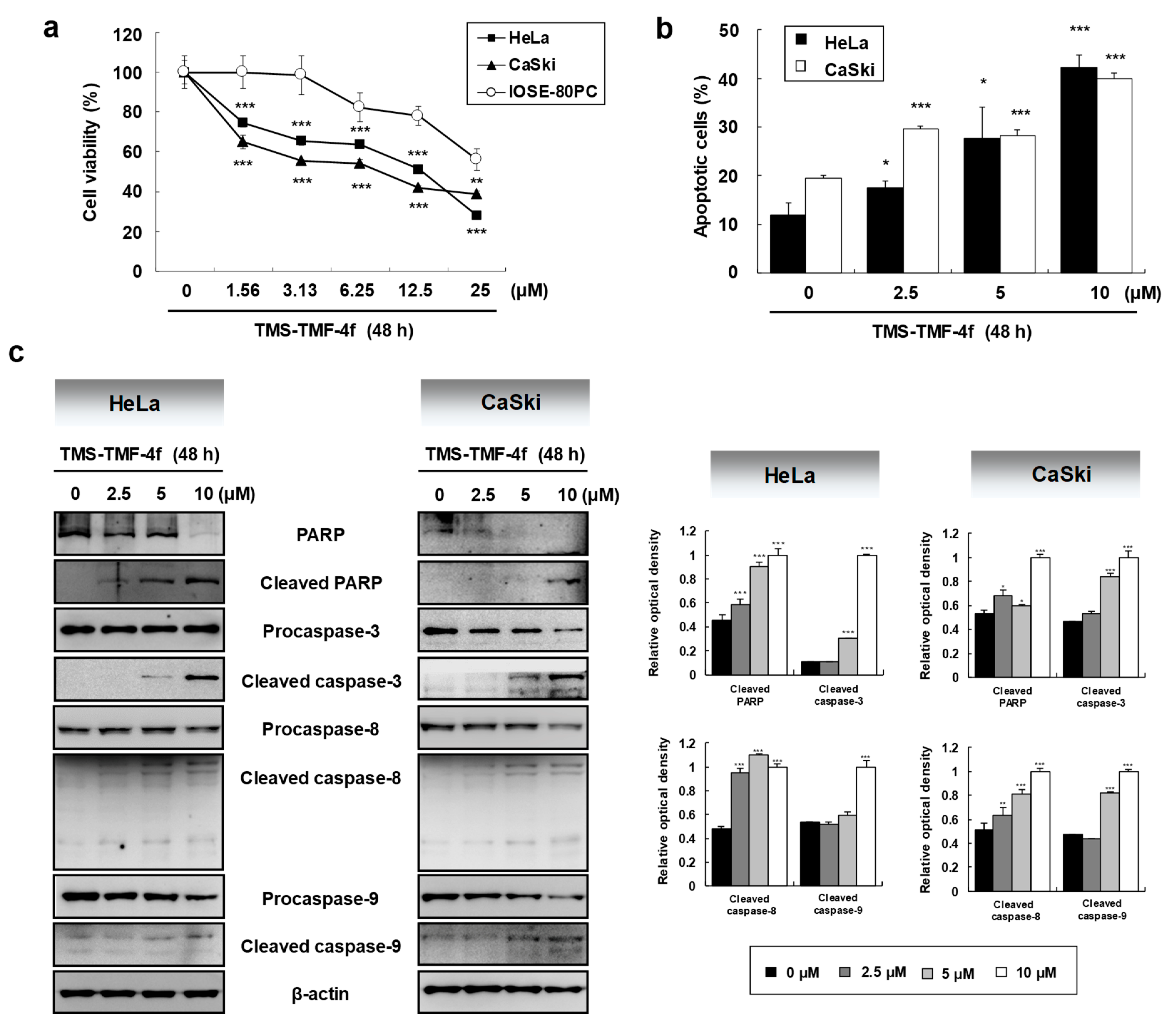
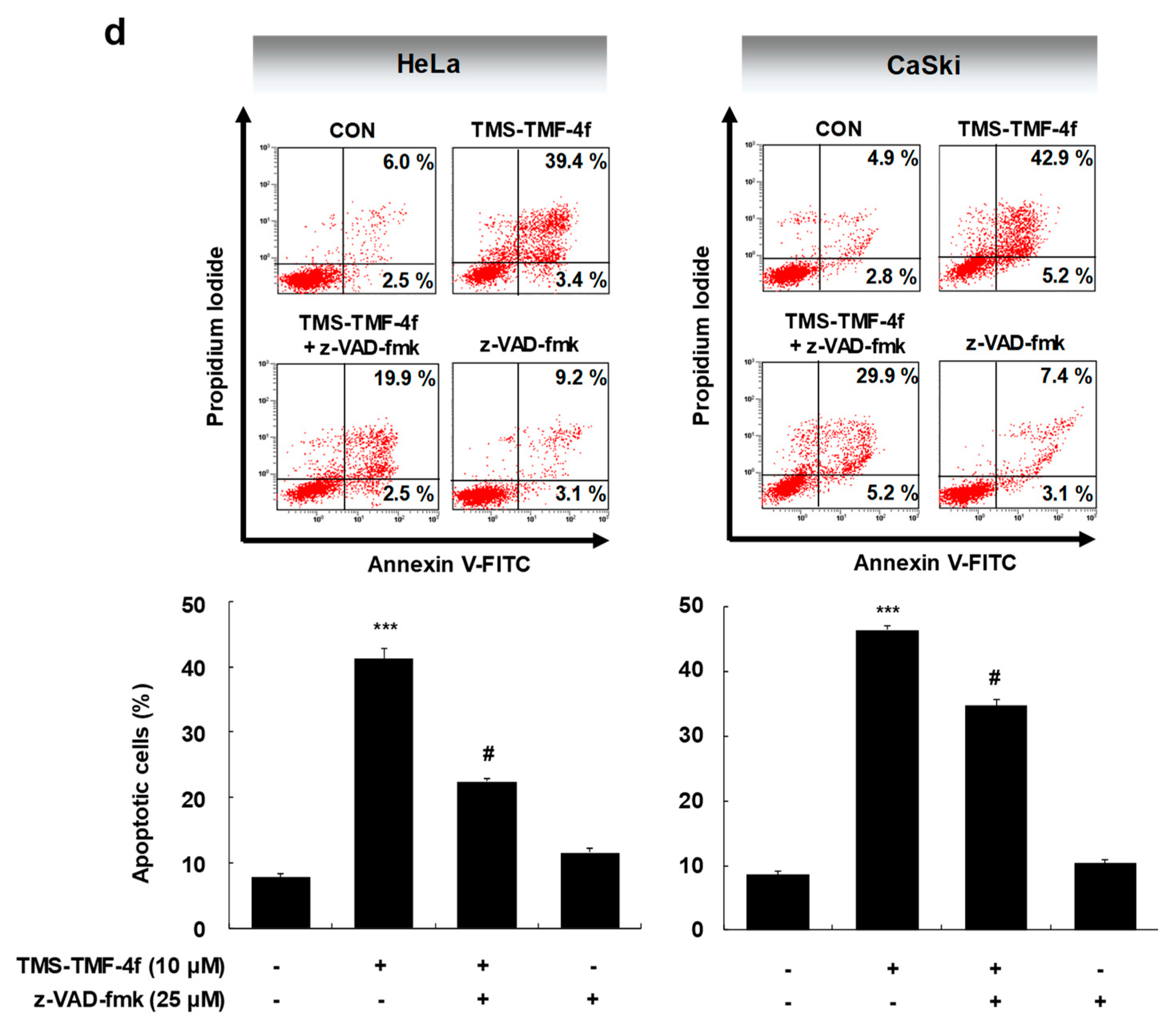
2.1. TMS-TMF-4f Suppresses the Proliferation of HeLa and CaSki Human Cervical Cancer Cells by Inducing Caspase-Dependent Apoptosis
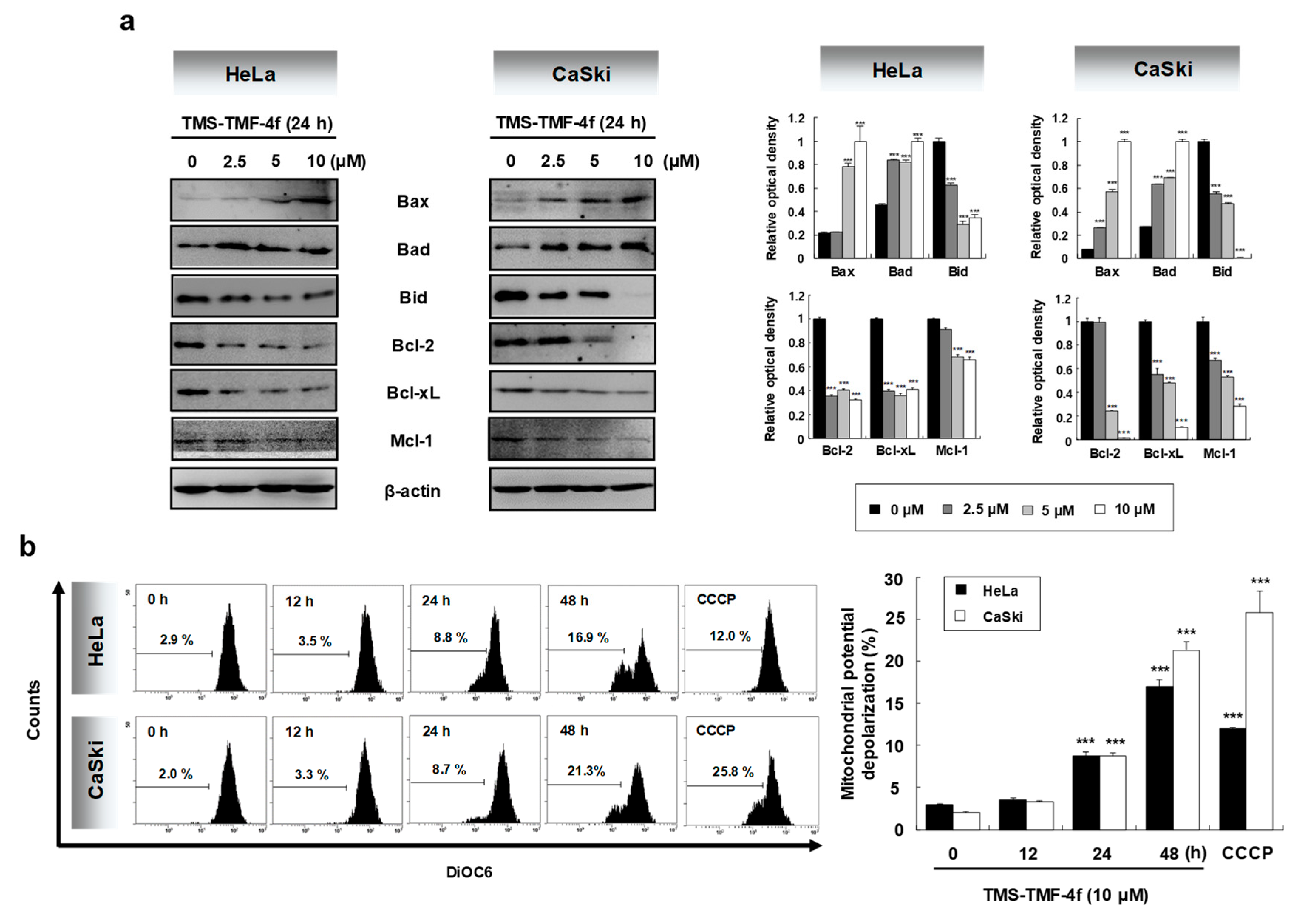
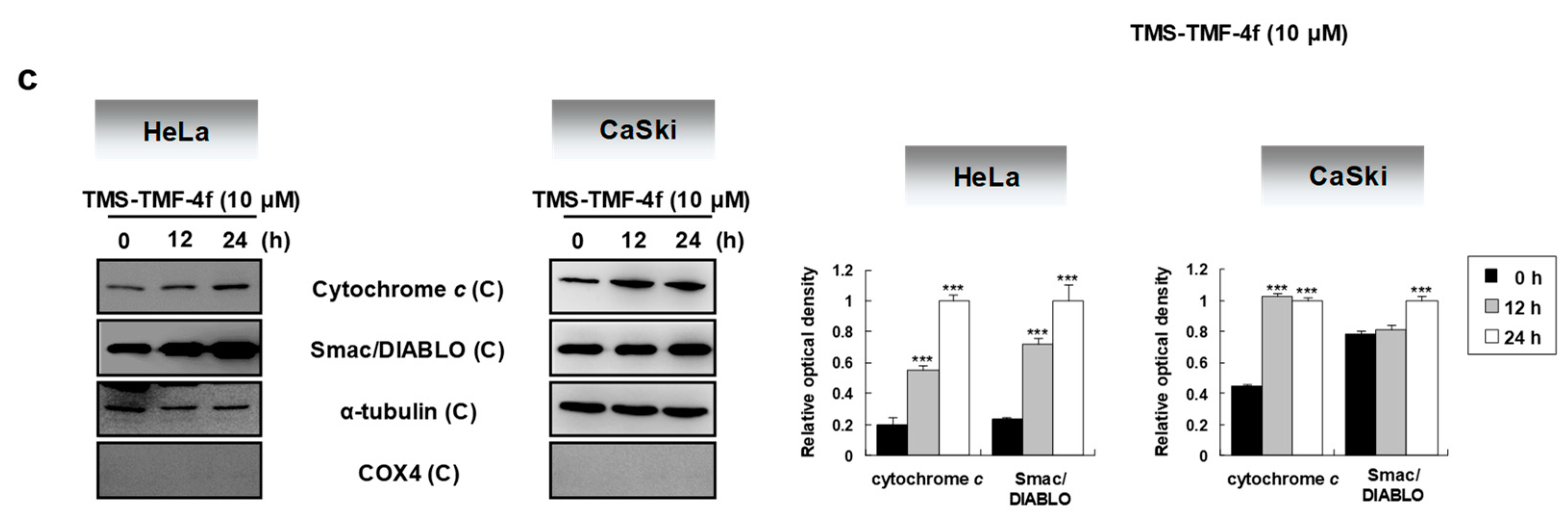
2.2. TMS-TMF-4f Induces Mitochondria-Dependent Apoptosis by Regulating Bcl-2 Family Proteins
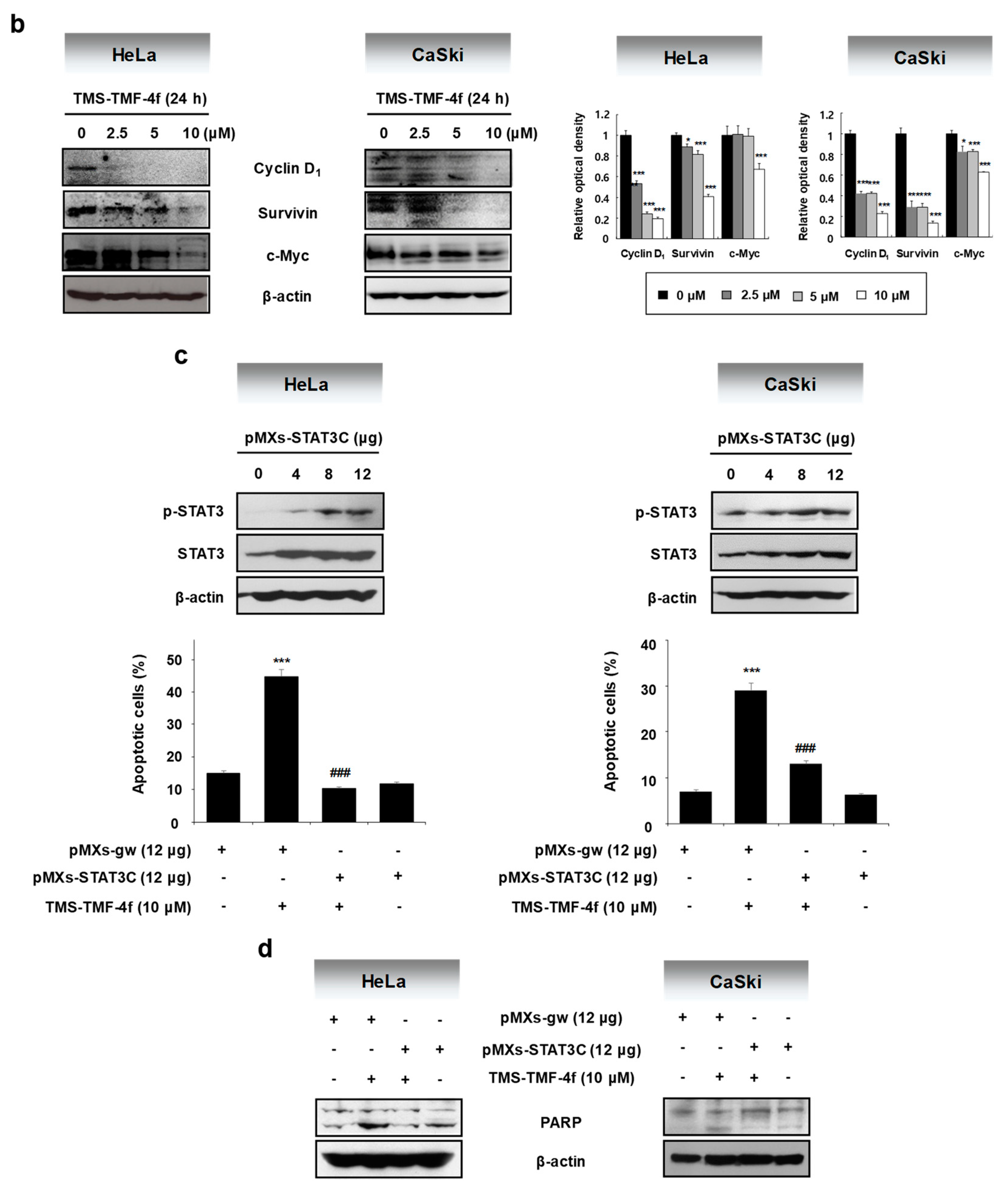
2.3. STAT3 is Involved in TMS-TMF-4f-Induced Apoptosis
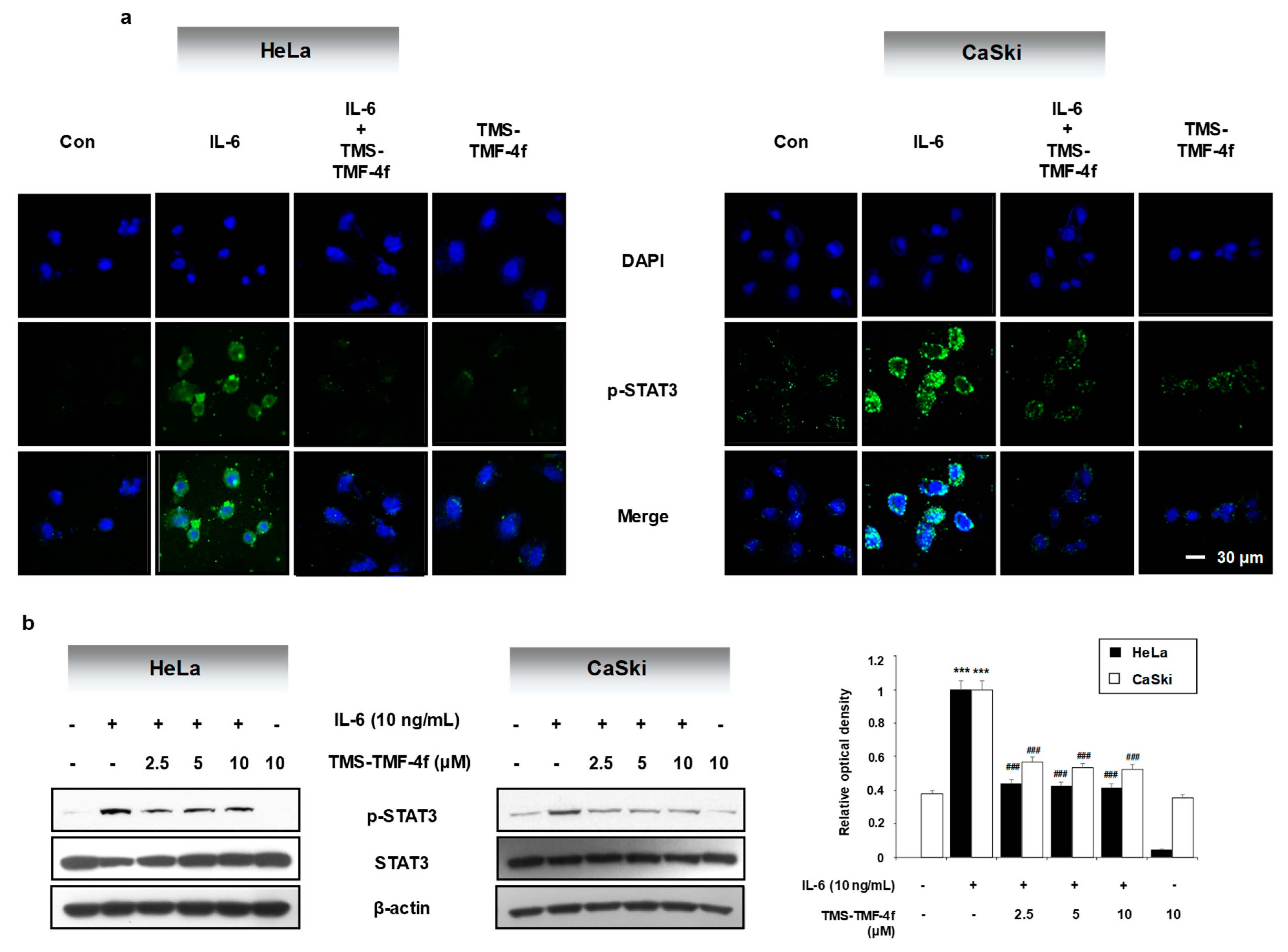
2.4. TMS-TMF-4f Ameliorates IL-6-Induced STAT3 Activation in Human Cervical Cancer Cells
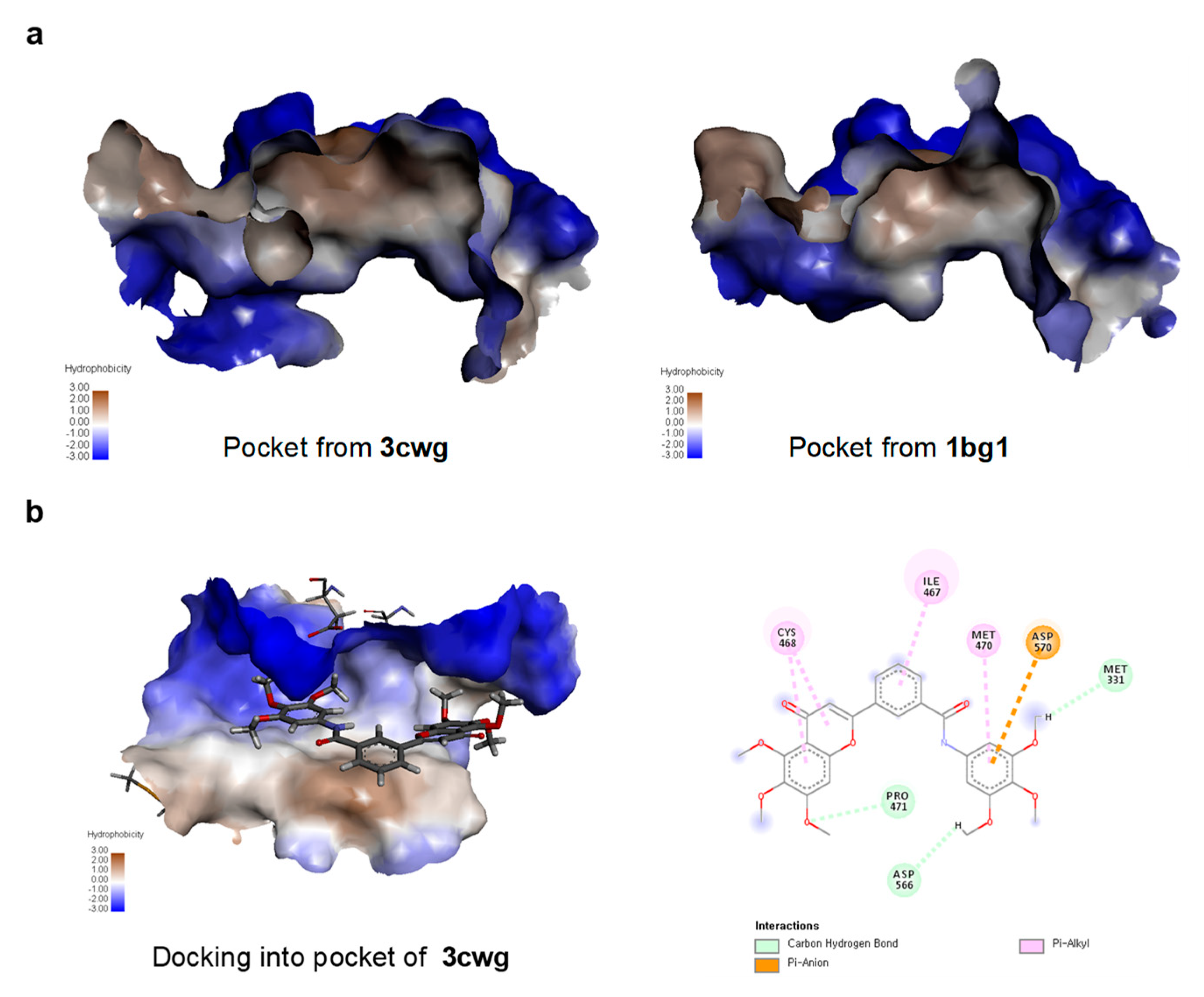
2.5. TMS-TMF-4f Docks into a Pocket Between the DNA-Binding and Ligand-Binding Domains of STAT3
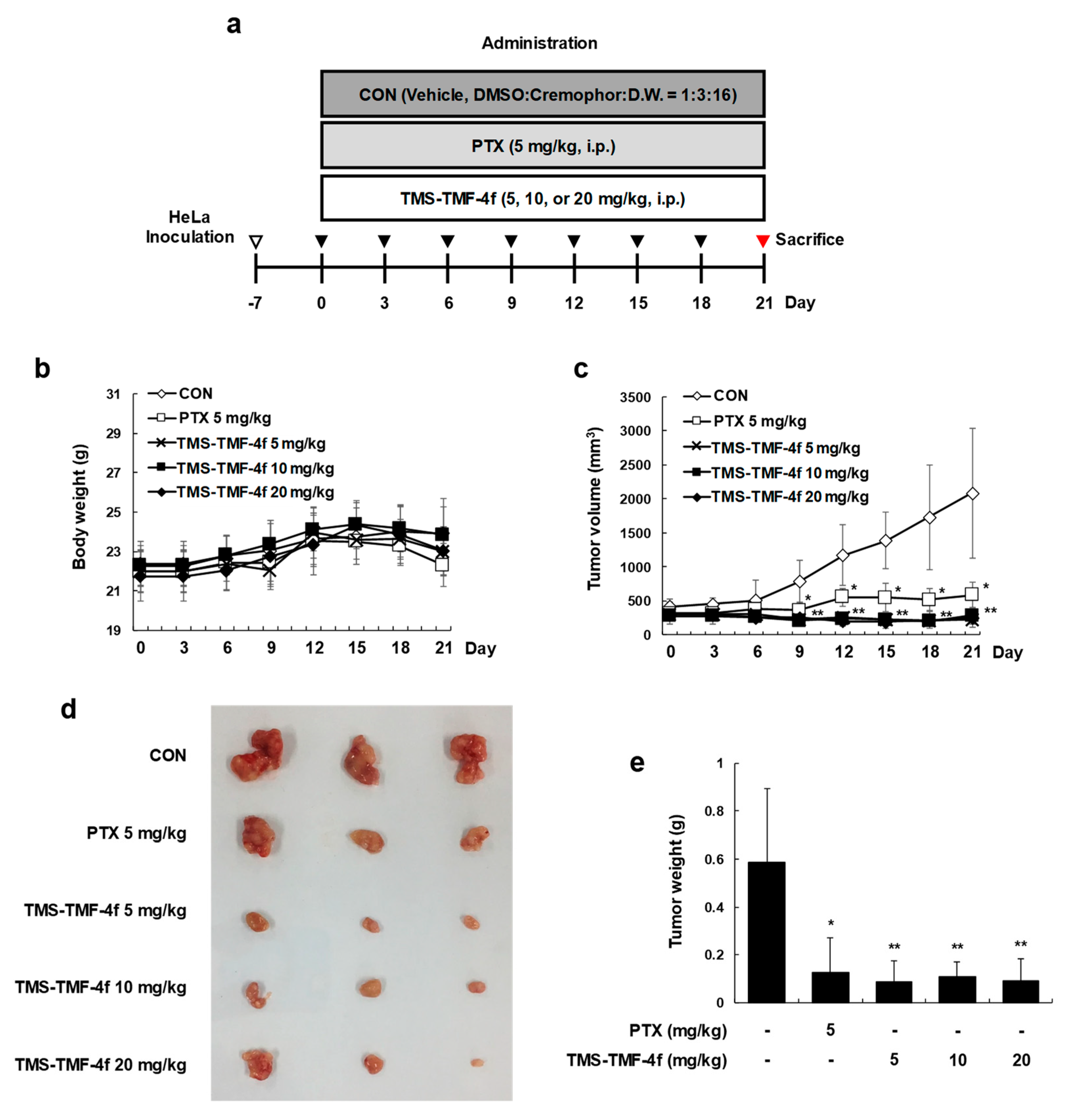
2.6. TMS-TMF-4f Exhibits Significant Antitumor Effects in a Xenograft Cervical Cancer Mouse Model
2.7. TMS-TMF-4f Promotes Apoptosis and Attenuates STAT3 Activation in Cervical Tumor Tissues
3. Discussion
4. Materials and Methods
4.1. Synthesis of Hybrid Analogs
4.2. Cell Culture
4.3. Cell Viability Assay
4.4. Annexin V-FITC and PI double Staining Assay
4.5. Western Blot Analysis
4.6. Measurement of MMP
4.7. Preparation of Cytosolic Fraction
4.8. Immunocytochemistry
4.9. Transfection
4.10. Animals
4.11. In Vivo Tumor Xenograft Studies
4.12. Terminal Deoxynucleotidyl Transferase-Mediated dUTP μM Nick End Labeling (TUNEL) Assay
4.13. Molecular Docking Analysis
4.14. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Small, W., Jr.; Bacon, M.A.; Bajaj, A.; Chuang, L.T.; Fisher, B.J.; Harkenrider, M.M.; Jhingran, A.; Kitchener, H.C.; Mileshkin, L.R.; Viswanathan, A.N.; et al. Cervical cancer: A global health crisis. Cancer 2017, 123, 2404–2412. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tang, D.; Wang, K.; Wang, J.; Zhang, Z.; Chen, Y.; Zhang, X.; Ma, C. HPV genotype prevalence and distribution during 2009–2018 in Xinjiang, China: Baseline surveys prior to mass HPV vaccination. BMC Womens Health 2019, 19, 90. [Google Scholar] [CrossRef] [PubMed]
- Anil, K.; Hemraj, V.; Ram, B.S.; Deepshilkha, G. An anthology of cancer. Innov. Int. J. Med. Pharm. Sci. 2018, 3, 7–14. [Google Scholar]
- Zheng, C.; Qing, S.; Wang, J.; Lu, G.; Li, H.; Lu, X.; Ma, C.; Tang, J.; Yue, X. Diagnosis of cervical squamous cell carcinoma and cervical adenocarcinoma based on Raman spectroscopy and support vector machine. Photodiag. Photodyn. Ther. 2019, 27, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Zhu, W.; Feng, W.; Lee, S.S.; Leung, A.W.; Shen, J.; Gao, L.; Xu, C. A Review of Resveratrol as a Potent Chemoprotective and Synergistic Agent in Cancer Chemotherapy. Front. Pharmacol. 2018, 9, 1534. [Google Scholar] [CrossRef] [PubMed]
- Fathi, N.; Rashidi, G.; Khodadadi, A.; Shahi, S.; Sharifi, S. STAT3 and apoptosis challenges in cancer. Int. J. Biol. Macromol. 2018, 117, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Doherty, J.; Baehrecke, E.H. Life, death and autophagy. Nat. Cell Biol. 2018, 20, 1110–1117. [Google Scholar] [CrossRef]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef]
- Johnson, D.E.; O’Keefe, R.A.; Grandis, J.R. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat. Rev. Clin. Oncol. 2018, 15, 234–248. [Google Scholar] [CrossRef]
- Huynh, J.; Etemadi, N.; Hollande, F.; Ernst, M.; Buchert, M. The JAK/STAT3 axis: A comprehensive drug target for solid malignancies. Semin. Cancer Biol. 2017, 45, 13–22. [Google Scholar] [CrossRef]
- Ray, K.; Ujvari, B.; Ramana, V.; Donald, J. Cross-talk between EGFR and IL-6 drives oncogenic signaling and offers therapeutic opportunities in cancer. Cytokine Growth Factor Rev. 2018, 41, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Song, D.; Li, H.; Yang, Y.; Ma, X.; Deng, S.; Ren, C.; Shu, X. Negative regulators of STAT3 signaling pathway in cancers. Cancer Manag. Res. 2019, 11, 4957–4969. [Google Scholar] [CrossRef] [PubMed]
- Fallowfield, M.E.; Cook, M.G. The value of nucleolar organizer region staining in the differential diagnosis of borderline melanocytic lesions. Histopathology 1989, 14, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Nepali, K.; Sharma, S.; Kumar, D.; Budhiraja, A.; Dhar, K.L. Anticancer hybrids—A patent survey. Recent Pat. Anticancer Drug Discov. 2014, 9, 303–339. [Google Scholar] [CrossRef]
- Hu, H.B.; Liang, H.P.; Li, H.M.; Yuan, R.N.; Sun, J.; Zhang, L.L.; Han, M.H.; Wu, Y. Isolation, modification and cytotoxic evaluation of stilbenoids from Acanthopanax leucorrhizus. Fitoterapia 2018, 124, 167–176. [Google Scholar] [CrossRef]
- Zaki, M.A.; Balachandran, P.; Khan, S.; Wang, M.; Mohammed, R.; Hetta, M.H.; Pasco, D.S.; Muhammad, I. Cytotoxicity and modulation of cancer-related signaling by (Z)- and (E)-3,4,3’,5’-tetramethoxystilbene isolated from Eugenia rigida. J. Nat. Prod. 2013, 76, 679–684. [Google Scholar] [CrossRef]
- Liao, H.L.; Hu, M.K. Synthesis and anticancer activities of 5,6,7-trimethylbaicalein derivatives. Chem. Pharm. Bull. (Tokyo) 2004, 52, 1162–1165. [Google Scholar] [CrossRef][Green Version]
- Rim, H.K.; Yun, C.H.; Shin, J.S.; Cho, Y.W.; Jang, D.S.; Ryu, J.H.; Park, H.; Lee, K.T. 5,6,7-trimethoxyflavone suppresses pro-inflammatory mediators in lipopolysaccharide-induced RAW 264.7 macrophages and protects mice from lethal endotoxin shock. Food Chem. Toxicol. 2013, 62, 847–855. [Google Scholar] [CrossRef]
- Hassan, A.H.E.; Choi, E.; Yoon, Y.M.; Lee, K.W.; Yoo, S.Y.; Cho, M.C.; Yang, J.S.; Kim, H.I.; Hong, J.Y.; Shin, J.S.; et al. Natural products hybrids: 3,5,4’-Trimethoxystilbene-5,6,7-trimethoxyflavone chimeric analogs as potential cytotoxic agents against diverse human cancer cells. Eur. J. Med. Chem. 2019, 161, 559–580. [Google Scholar] [CrossRef]
- Tome-Carneiro, J.; Larrosa, M.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.A.; Garcia-Conesa, M.T.; Espin, J.C. Resveratrol and clinical trials: The crossroad from in vitro studies to human evidence. Curr. Pharm. Des. 2013, 19, 6064–6093. [Google Scholar] [CrossRef]
- Signorelli, P.; Ghidoni, R. Resveratrol as an anticancer nutrient: Molecular basis, open questions and promises. J. Nutr. Biochem. 2005, 16, 449–466. [Google Scholar] [CrossRef] [PubMed]
- Won, J.H.; Chung, K.S.; Park, E.Y.; Lee, J.H.; Choi, J.H.; Tapondjou, L.A.; Park, H.J.; Nomura, M.; Hassan, A.H.E.; Lee, K.T. 23-Hydroxyursolic Acid Isolated from the Stem Bark of Cussonia bancoensis Induces Apoptosis through Fas/Caspase-8-Dependent Pathway in HL-60 Human Promyelocytic Leukemia Cells. Molecules 2018, 23, 3306. [Google Scholar] [CrossRef] [PubMed]
- Ilmarinen, P.; Moilanen, E.; Kankaanranta, H. Mitochondria in the center of human eosinophil apoptosis and survival. Int. J. Mol. Sci. 2014, 15, 3952–3969. [Google Scholar] [CrossRef] [PubMed]
- Webster, K.A. Mitochondrial membrane permeabilization and cell death during myocardial infarction: Roles of calcium and reactive oxygen species. Future Cardiol. 2012, 8, 863–884. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.J.; Tait, S.W.G. Targeting BCL-2 regulated apoptosis in cancer. Open Biol. 2018, 8, 180002. [Google Scholar] [CrossRef]
- Kim, H.; Kim, D.; Choi, S.A.; Kim, C.R.; Oh, S.K.; Pyo, K.E.; Kim, J.; Lee, S.H.; Yoon, J.B.; Zhang, Y.; et al. KDM3A histone demethylase functions as an essential factor for activation of JAK2-STAT3 signaling pathway. Proc. Natl. Acad. Sci. USA 2018, 115, 11766–11771. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Ray, R.M.; Johnson, L.R. STAT3-mediated transcription of Bcl-2, Mcl-1 and c-IAP2 prevents apoptosis in polyamine-depleted cells. Biochem. J. 2005, 392, 335–344. [Google Scholar] [CrossRef]
- Oh, S.B.; Hwang, C.J.; Song, S.Y.; Jung, Y.Y.; Yun, H.M.; Sok, C.H.; Sung, H.C.; Yi, J.M.; Park, D.H.; Ham, Y.W.; et al. Anti-cancer effect of tectochrysin in NSCLC cells through overexpression of death receptor and inactivation of STAT3. Cancer Lett. 2014, 353, 95–103. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Hassan, M.; Watari, H.; AbuAlmaaty, A.; Ohba, Y.; Sakuragi, N. Apoptosis and molecular targeting therapy in cancer. BioMed Res. Int. 2014, 2014, 150845. [Google Scholar] [CrossRef]
- Jan, R.; Chaudhry, G.E. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019, 9, 205–218. [Google Scholar] [CrossRef]
- Khan, K.H.; Blanco-Codesido, M.; Molife, L.R. Cancer therapeutics: Targeting the apoptotic pathway. Crit. Rev. Oncol. Hematol. 2014, 90, 200–219. [Google Scholar] [CrossRef]
- Igney, F.H.; Krammer, P.H. Death and anti-death: Tumour resistance to apoptosis. Nat. Rev. Cancer 2002, 2, 277–288. [Google Scholar] [CrossRef]
- Locksley, R.M.; Killeen, N.; Lenardo, M.J. The TNF and TNF receptor superfamilies: Integrating mammalian biology. Cell 2001, 104, 487–501. [Google Scholar] [CrossRef]
- Fulda, S. Targeting extrinsic apoptosis in cancer: Challenges and opportunities. Semin. Cell Dev. Biol. 2015, 39, 20–25. [Google Scholar] [CrossRef]
- Chong, S.J.; Low, I.C.; Pervaiz, S. Mitochondrial ROS and involvement of Bcl-2 as a mitochondrial ROS regulator. Mitochondrion 2014, 19 Pt A, 39–48. [Google Scholar] [CrossRef]
- Martinez-Fabregas, J.; Diaz-Moreno, I.; Gonzalez-Arzola, K.; Janocha, S.; Navarro, J.A.; Hervas, M.; Bernhardt, R.; Velazquez-Campoy, A.; Diaz-Quintana, A.; De la Rosa, M.A. Structural and functional analysis of novel human cytochrome C targets in apoptosis. Mol. Cell. Proteom. 2014, 13, 1439–1456. [Google Scholar] [CrossRef]
- Chinnaiyan, A.M. The apoptosome: Heart and soul of the cell death machine. Neoplasia 1999, 1, 5–15. [Google Scholar] [CrossRef]
- Aldawsari, F.S.; Velazquez-Martinez, C.A. 3,4’,5-trans-Trimethoxystilbene; a natural analogue of resveratrol with enhanced anticancer potency. Investig. New Drugs 2015, 33, 775–786. [Google Scholar] [CrossRef]
- Wei, L.H.; Kuo, M.L.; Chen, C.A.; Chou, C.H.; Lai, K.B.; Lee, C.N.; Hsieh, C.Y. Interleukin-6 promotes cervical tumor growth by VEGF-dependent angiogenesis via a STAT3 pathway. Oncogene 2003, 22, 1517–1527. [Google Scholar] [CrossRef]
- Shukla, S.; Shishodia, G.; Mahata, S.; Hedau, S.; Pandey, A.; Bhambhani, S.; Batra, S.; Basir, S.F.; Das, B.C.; Bharti, A.C. Aberrant expression and constitutive activation of STAT3 in cervical carcinogenesis: Implications in high-risk human papillomavirus infection. Mol. Cancer 2010, 9, 282. [Google Scholar] [CrossRef] [PubMed]
- Spitzner, M.; Ebner, R.; Wolff, H.A.; Ghadimi, B.M.; Wienands, J.; Grade, M. STAT3: A Novel Molecular Mediator of Resistance to Chemoradiotherapy. Cancers (Basel) 2014, 6, 1986–2011. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, P.K.; Li, C.; Lin, J. Inhibition of STAT3 signaling blocks the anti-apoptotic activity of IL-6 in human liver cancer cells. J. Biol. Chem. 2010, 285, 27429–27439. [Google Scholar] [CrossRef] [PubMed]
- Miao, J.W.; Liu, L.J.; Huang, J. Interleukin-6-induced epithelial-mesenchymal transition through signal transducer and activator of transcription 3 in human cervical carcinoma. Int. J. Oncol. 2014, 45, 165–176. [Google Scholar] [CrossRef]
- Dethlefsen, L.A.; Prewitt, J.M.; Mendelsohn, M.L. Analysis of tumor growth curves. J. Natl. Cancer Inst. 1968, 40, 389–405. [Google Scholar] [CrossRef]
- Grosdidier, A.; Zoete, V.; Michielin, O. SwissDock, a protein-small molecule docking web service based on EADock DSS. Nucleic Acids Res. 2011, 39, W270–W277. [Google Scholar] [CrossRef]
- Hassan, A.H.E.; Park, H.R.; Yoon, Y.M.; Kim, H.I.; Yoo, S.Y.; Lee, K.W.; Lee, Y.S. Antiproliferative 3-deoxysphingomyelin analogs: Design, synthesis, biological evaluation and molecular docking of pyrrolidine-based 3-deoxysphingomyelin analogs as anticancer agents. Bioorg. Chem. 2019, 84, 444–455. [Google Scholar] [CrossRef]
- Farag, A.K.; Hassan, A.H.E.; Jeong, H.; Kwon, Y.; Choi, J.G.; Oh, M.S.; Park, K.D.; Kim, Y.K.; Roh, E.J. First-in-class DAPK1/CSF1R dual inhibitors: Discovery of 3,5-dimethoxy-N-(4-(4-methoxyphenoxy)-2-((6-morpholinopyridin-3-yl)amino)pyrimidi n-5-yl)benzamide as a potential anti-tauopathies agent. Eur. J. Med. Chem. 2019, 162, 161–175. [Google Scholar] [CrossRef]


| Compound (TMS-TMF) | a IC50 (μM) | ||||
|---|---|---|---|---|---|
| HCT116 | A549 | AsPC-1 | HeLa | ||
| 4a | 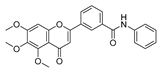 | 38.27 ± 1.60 | 46.61 ± 3.12 | 39.11 ± 2.23 | 35.55 ± 2.54 |
| 4b | 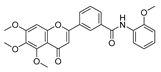 | 27.60 ± 7.55 | 27.86 ± 6.47 | 25.57 ± 6.48 | 14.21 ± 4.63 |
| 4c | 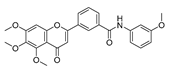 | 36.27 ± 4.76 | 24.41 ± 2.49 | 31.41 ± 1.45 | 34.99 ± 1.70 |
| 4d |  | 33.57 ± 5.38 | 41.85 ± 3.69 | >50 | >50 |
| 4e | 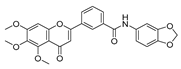 | 30.54 ± 5.21 | >50 | >50 | >50 |
| 4f | 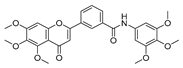 | 17.33 ± 2.76 | 16.79 ± 3.85 | 14.68 ± 1.65 | 12.07 ± 1.84 |
| Etoposide | 21.45 ± 3.82 | 24.25 ± 2.99 | 29.77 ± 3.08 | 32.70 ± 2.56 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, J.Y.; Chung, K.-S.; Shin, J.-S.; Lee, J.-H.; Gil, H.-S.; Lee, H.-H.; Choi, E.; Choi, J.-H.; Hassan, A.H.E.; Lee, Y.S.; et al. The Anti-Proliferative Activity of the Hybrid TMS-TMF-4f Compound Against Human Cervical Cancer Involves Apoptosis Mediated by STAT3 Inactivation. Cancers 2019, 11, 1927. https://doi.org/10.3390/cancers11121927
Hong JY, Chung K-S, Shin J-S, Lee J-H, Gil H-S, Lee H-H, Choi E, Choi J-H, Hassan AHE, Lee YS, et al. The Anti-Proliferative Activity of the Hybrid TMS-TMF-4f Compound Against Human Cervical Cancer Involves Apoptosis Mediated by STAT3 Inactivation. Cancers. 2019; 11(12):1927. https://doi.org/10.3390/cancers11121927
Chicago/Turabian StyleHong, Joo Young, Kyung-Sook Chung, Ji-Sun Shin, Jeong-Hun Lee, Hyo-Sun Gil, Hwi-Ho Lee, Eunwoo Choi, Jung-Hye Choi, Ahmed H.E. Hassan, Yong Sup Lee, and et al. 2019. "The Anti-Proliferative Activity of the Hybrid TMS-TMF-4f Compound Against Human Cervical Cancer Involves Apoptosis Mediated by STAT3 Inactivation" Cancers 11, no. 12: 1927. https://doi.org/10.3390/cancers11121927
APA StyleHong, J. Y., Chung, K.-S., Shin, J.-S., Lee, J.-H., Gil, H.-S., Lee, H.-H., Choi, E., Choi, J.-H., Hassan, A. H. E., Lee, Y. S., & Lee, K.-T. (2019). The Anti-Proliferative Activity of the Hybrid TMS-TMF-4f Compound Against Human Cervical Cancer Involves Apoptosis Mediated by STAT3 Inactivation. Cancers, 11(12), 1927. https://doi.org/10.3390/cancers11121927






