Complications Following Percutaneous Epidural Neuroplasty: A Narrative Review of Clinical Evidence and the Rationale for Post-Procedural 6 h Inpatient Monitoring Amid Limited Systematic Data
Abstract
1. Introduction
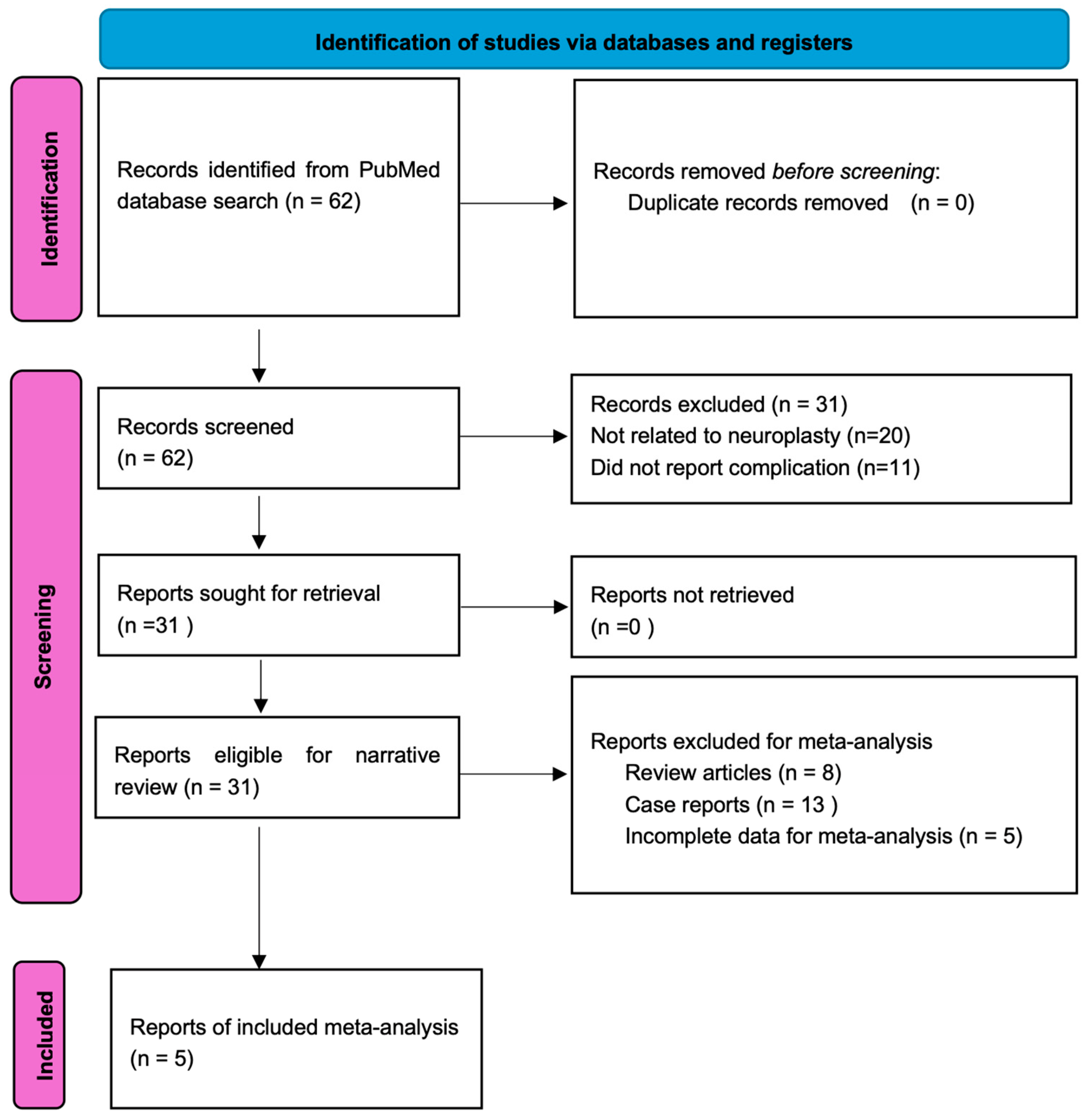
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Data Extraction and Quality Assessment
2.3. Outcomes and Statistical Analysis
3. Results
3.1. Study Selection
3.2. Pathophysiological Basis of Epidural Neuroplasty: Implications for Complication Risk
3.3. Complication Spectrum and Classification
3.4. Complication Incidence and Pooled Analysis
3.5. Complication Type, Severity, and Timing
- Mechanical complications (n ≈ 48):
- Dural puncture: 11;
- Catheter misplacement (vein, paravertebral, intradural): 18;
- Catheter breakage or blockage: 9;
- Problem during withdrawal: 3;
- Intradural cyst formation: 3;
- Procedural failure due to intrathecal passage: 3.
- Neurological complications (n ≈ 47):
- Transient paresthesia: 19;
- Prolonged paresthesia/numbness: 3;
- Urinary/bowel dysfunction: 5;
- Motor weakness: 6;
- Paraplegia or irreversible deficit: 11;
- Sexual dysfunction: 1;
- Headache (post-dural or pneumocephalus-related): 2;
- Persistent neurologic deficit: 5.
- Infectious complications (n ≈ 15):
- Bacterial meningitis or ventriculitis: 3;
- Epidural abscess: 3;
- Local infections at catheter entry site: 7;
- Systemic sepsis or delayed neurologic infection: 2.
- Systemic complications (n ≈ 13):
- Hypotension or vasovagal reaction: 7;
- Respiratory depression: 1;
- Cardiopulmonary arrest or circulatory collapse: 2;
- Pneumocephalus: 3.
- Miscellaneous or rare complications (n ≈ 10):
- Imaging artifact mimicking subarachnoid hemorrhage: 1;
- Persistent hiccup: 1;
- Miscellaneous procedural anomalies (e.g., barotrauma, failed dye spread): 5.
- Mild (n ≈ 52): Headache, local discomfort, and transient symptoms.
- Moderate (n ≈ 38): Urinary retention, temporary motor weakness, and drug-related side effects.
- Severe (n ≈ 43): Epidural hematoma, spinal infection, persistent neurological deficit, and cardiovascular collapse.
- Immediate onset (0–1 h): Most pharmacologic or mechanical events (e.g., intrathecal injection, hypotension).
- Early onset (1–6 h): Epidural hematoma, pneumocephalus, respiratory issues.
- Delayed onset (6–48 h): Infectious complications and neurologic decline.
- Altered spinal anatomy (post-surgical, advanced stenosis or deformity).
- Use of high-risk agents (hypertonic saline, ozone).
- Elderly age or medical comorbidities.
3.6. Clinical Case Highlights and Analysis of High-Risk Complications
4. Discussion
4.1. Medical Rationale for Structured Post-Procedural Inpatient Monitoring
4.2. Recommendations for Clinical Practice and Future Directions
- Patient Selection and Risk Stratification: Comprehensive pre-procedural assessments, including detailed medical history, imaging reviews, and consideration of patient-specific risk factors such as age, anatomical variations, previous spinal surgeries, and comorbid conditions should be strongly recommended. High-risk patients warrant heightened vigilance and possibly extended inpatient monitoring.
- Procedural Best Practices: Adoption of meticulous procedural techniques is crucial to minimize mechanical complications. Practitioners should be rigorously trained in catheter manipulation, medication injection protocols, and the use of imaging modalities such as fluoroscopy or epiduroscopy. Avoidance or cautious use of potentially neurotoxic agents, such as hypertonic saline and ozone, is recommended.
- Structured Inpatient Monitoring: This period should include regular neurological evaluations, monitoring for signs of infection and systemic reactions, and careful assessment of medication-related side effects. Because delayed-onset complications—including infections and neurologic deterioration—may occur up to 14 days after PEN, a follow-up visit is advised following discharge, particularly for high-risk patients.
- Multidisciplinary Collaboration: Implementing a multidisciplinary team approach, involving anesthesiologists, neurologists, rehabilitation specialists, spine surgeons, and nursing staffs, can enhance patient management and early identification of complications. This collaborative framework ensures comprehensive care that addresses both immediate and long-term patient needs.
- Patient Education and Informed Consent: Providing patients with clear information about potential complications, symptoms to watch for post-procedure, and detailed instructions regarding follow-up care can significantly enhance patient compliance and early complication detection post-discharge.
- Future Research Directions: Prospective, multicenter studies and standardized complication-reporting registries are needed to further quantify complication rates, define high-risk patient populations, and refine procedural guidelines. Further studies should also evaluate the cost-effectiveness of structured inpatient monitoring in both high- and low-risk populations as well as the long-term clinical benefits of early complication detection.
- Implementing these recommendations into clinical practice can significantly mitigate the risks associated with PEN, improving patient safety, procedural outcomes, and overall healthcare quality.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Manchikanti, L.; Knezevic, N.N.; Sanapati, M.R.; Boswell, M.V.; Kaye, A.D.; Hirsch, J.A. Effectiveness of Percutaneous Adhesiolysis in Managing Chronic Central Lumbar Spinal Stenosis: A Systematic Review and Meta-Analysis. Pain Physician 2019, 22, E523–E550. [Google Scholar] [CrossRef] [PubMed]
- Dagistan, G.; Ozdemir, I. Assessment of efficacy of percutaneous epidural neuroplasty for lumbar stenosis and failed back surgery syndrome: Effective and safe? Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 11303–11314. [Google Scholar]
- Oh, C.H.; Ji, G.Y.; Shin, D.A.; Cho, P.G.; Yoon, S.H. Clinical Course of Cervical Percutaneous Epidural Neuroplasty in Single-Level Cervical Disc Disease with 12-Month Follow-up. Pain Physician 2017, 20, E941–E949. [Google Scholar] [CrossRef]
- Anderson, S.R.; Racz, G.B.; Heavner, J. Evolution of epidural lysis of adhesions. Pain Physician 2000, 3, 262–270. [Google Scholar] [CrossRef]
- Manchikanti, L.; Staats, P.S.; Singh, V.; Schultz, D.M.; Vilims, B.D.; Jasper, J.F.; Kloth, D.S.; Trescot, A.M.; Hansen, H.C.; Falasca, T.D.; et al. Evidence-based practice guidelines for interventional techniques in the management of chronic spinal pain. Pain Physician 2003, 6, 3–81. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Lee, S.H.; Lee, S.C. Preliminary results of the clinical effectiveness of percutaneous adhesiolysis using a Racz catheter in the management of chronic pain due to cervical central stenosis. Pain Physician 2013, 16, 353–358. [Google Scholar] [CrossRef]
- Choi, E.J.; Yoo, Y.J.; Lee, P.B.; Kim, Y.C.; Lee, S.C.; Moon, J.Y. A Retrospective Study to Evaluate the Effect of Concentration of Hypertonic Saline on Efficacy and Safety of Epidural Adhesiolysis. Anesth. Analg. 2017, 124, 2021–2029. [Google Scholar] [CrossRef]
- Beyaz, S.G.; Altaş, C.; Sayhan, H. Cardiopulmonary Arrest and Pneumoencephaly Developing after Epidural Oxygen-ozone Mixture Therapy. Anesth. Essays Res. 2018, 12, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Toman, H.; Özdemir, U.; Kiraz, H.A.; Lüleci, N. Severe headache following ozone therapy: Pneumocephalus. Agri 2017, 29, 132–136. [Google Scholar] [CrossRef][Green Version]
- Heavner, J.E.; Racz, G.B.; Raj, P. Percutaneous epidural neuroplasty: Prospective evaluation of 0.9% NaCl versus 10% NaCl with or without hyaluronidase. Reg. Anesth. Pain Med. 1999, 24, 202–207. [Google Scholar] [CrossRef]
- Rapčan, R.; Kočan, L.; Mláka, J.; Burianek, M.; Kočanová, H.; Rapčanová, S.; Hess, M.; Hammond, A.; Griger, M.; Venglarčík, M.; et al. A Randomized, Multicenter, Double-Blind, Parallel Pilot Study Assessing the Effect of Mechanical Adhesiolysis vs Adhesiolysis with Corticosteroid and Hyaluronidase Administration into the Epidural Space During Epiduroscopy. Pain Med. 2018, 19, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Erdine, S.; Talu, G.K. Precautions during epidural neuroplasty. Pain Pract. Off. J. World Inst. Pain 2002, 2, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Talu, G.K.; Erdine, S. Complications of epidural neuroplasty: A retrospective evaluation. Neuromodul. J. Int. Neuromodul. Soc. 2003, 6, 237–247. [Google Scholar] [CrossRef]
- Manchikanti, L.; Boswell, M.V.; Singh, V.; Benyamin, R.M.; Fellows, B.; Abdi, S.; Buenaventura, R.M.; Conn, A.; Datta, S.; Derby, R.; et al. Comprehensive evidence-based guidelines for interventional techniques in the management of chronic spinal pain. Pain Physician 2009, 12, 699–802. [Google Scholar] [CrossRef]
- Richardson, J.; McGurgan, P.; Cheema, S.; Prasad, R.; Gupta, S. Spinal endoscopy in chronic low back pain with radiculopathy. A prospect case series. Anaesthesia 2001, 56, 454–460. [Google Scholar] [CrossRef]
- Gazzeri, R.; Tribuzi, S.; Leoni, M.L.G.; Michelli, F.; Occhigrossi, F. Evaluation of Lumbar Adhesiolysis Using a Radiofrequency Catheter During Epiduroscopy in the Treatment of Failed Back Surgery Syndrome (FBSS). Surg. Technol. Int. 2023, 43, 301–306. [Google Scholar] [CrossRef]
- Ege, F. Evaluation of the Efficacy of Caudal Epidural Neuroplasty in Patients with Lumbar Epidural Fibrosis. Cureus 2024, 16, e52606. [Google Scholar] [CrossRef]
- Quan, M.; Hwang, W.H.; Kim, J.H.; Kim, Y.Y. Analysis of pain markers and epidural fibrosis caused by repeated spinal surgery in Sprague-Dawley rats. BMC Musculoskelet. Disord. 2021, 22, 16. [Google Scholar] [CrossRef] [PubMed]
- Manchikanti, L.; Pamapti, V.; Cash, K.A. Protocol for evaluation of the comparative effectiveness of percutaneous adhesiolysis and caudal epidural steroid injections in low back and/or lower extremity pain without post surgery syndrome or spinal stenosis. Pain Physician 2010, 13, E91–E110. [Google Scholar] [CrossRef]
- Ceylan, A.; Aşık, İ.; Özgencil, G.E.; Erken, B. Evaluation of the efficacy of epiduroscopic adhesiolysis in failed back surgery syndrome. Turk. J. Med. Sci. 2019, 49, 249–257. [Google Scholar] [CrossRef]
- Lee, F.; Jamison, D.E.; Hurley, R.W.; Cohen, S.P. Epidural lysis of adhesions. Korean J. Pain 2014, 27, 3–15. [Google Scholar] [CrossRef]
- Sim, J.-H.; Sim, K.-C.; Kim, Y.; Kim, D.-H.; Lee, I.; Shin, J.-W.; Karm, M.-H.; Choi, S.-S. Effectiveness of Epidural Balloon Neuroplasty in Patients With Chronic Spinal Stenosis Accompanied by Redundant Nerve Roots: A Longitudinal Cohort Study. Pain Physician 2022, 25, E841–E850. [Google Scholar]
- Kim, C.H.; Kim, P.; Ju, C.I.; Kim, S.W. Massive Epidural Hematoma Caused by Percutaneous Epidural Neuroplasty: A Case Report. Korean J. Neurotrauma 2023, 19, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.B.; Kim, M.K.; Kim, K.D.; Lim, Y.J. Unintended complication of intracranial subdural hematoma after percutaneous epidural neuroplasty. J. Korean Neurosurg. Soc. 2014, 55, 170–172. [Google Scholar] [CrossRef]
- Oh, C.H.; An, S.D.; Choi, S.H.; Ji, G.Y. Contrast mimicking a subarachnoid hemorrhage after lumbar percutaneous epidural neuroplasty: A case report. J. Med. Case Rep. 2013, 7, 88. [Google Scholar] [CrossRef] [PubMed]
- Noh, S.M.; Kang, H.G. Bacterial meningitis and ventriculitis following cervical neuroplasty. Int. J. Infect. Dis. 2020, 101, 393–394. [Google Scholar] [CrossRef]
- Lee, H.Y.; Wang, H.S.; Kim, S.W.; Ju, C.I. Cerebellar infarction following epidural abscess after epidural neuroplasty. Korean J. Spine 2015, 12, 26–28. [Google Scholar] [CrossRef]
- Lim, Y.S.; Jung, K.T.; Park, C.H.; Wee, S.W.; Sin, S.S.; Kim, J. Acute motor weakness of opposite lower extremity after percutaneous epidural neuroplasty. Korean J. Pain 2015, 28, 144–147. [Google Scholar] [CrossRef]
- Ryu, K.S.; Rathi, N.K.; Kim, G.; Park, C.K. Iatrogenic intradural lumbosacral cyst following epiduroscopy. J. Korean Neurosurg. Soc. 2012, 52, 491–494. [Google Scholar] [CrossRef]
- Lee, C.H.; Son, J.W.; Kim, U. Reverse Takotsubo Cardiomyopathy following Inadvertent Intrathecal Injection during Percutaneous Epidural Neuroplasty. Heart Lung Circ. 2015, 24, e148–e151. [Google Scholar] [CrossRef]
- Kim, T.H.; Shin, J.J.; Lee, W.Y. Surgical treatment of a broken neuroplasty catheter in the epidural space: A case report. J. Med. Case Rep. 2016, 10, 277. [Google Scholar] [CrossRef][Green Version]
- Kang, J.H.; Choi, H.; Kim, J.S.; Lee, M.K.; Park, H.J. A sheared Racz catheter in cervical epidural space for thirty months: A case report. Korean J. Anesthesiol. 2015, 68, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.Y.; Manghnani, P. Acute monoplegia after lysis of epidural adhesions: A case report. Pain Pr. 2008, 8, 404–407. [Google Scholar] [CrossRef]
- Lawton, M.T.; Porter, R.W.; Heiserman, J.E.; Jacobowitz, R.; Sonntag, V.K.; Dickman, C.A. Surgical management of spinal epidural hematoma: Relationship between surgical timing and neurological outcome. J. Neurosurg. 1995, 83, 1–7. [Google Scholar] [CrossRef]
- Engelborghs, S.; Niemantsverdriet, E.; Struyfs, H.; Blennow, K.; Brouns, R.; Comabella, M.; Dujmovic, I.; Van Der Flier, W.; Frölich, L.; Galimberti, D.; et al. Consensus guidelines for lumbar puncture in patients with neurological diseases. Alzheimer’s Dement. 2017, 8, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Apfelbaum, J.L.; Silverstein, J.H.; Chung, F.F.; Connis, R.T.; Fillmore, R.B.; Hunt, S.E.; Nickinovich, D.G.; Schreiner, M.S.; Silverstein, J.H.; Apfelbaum, J.L.; et al. Practice guidelines for postanesthetic care: An updated report by the American Society of Anesthesiologists Task Force on Postanesthetic Care. Anesthesiology 2013, 118, 291–307. [Google Scholar] [PubMed]
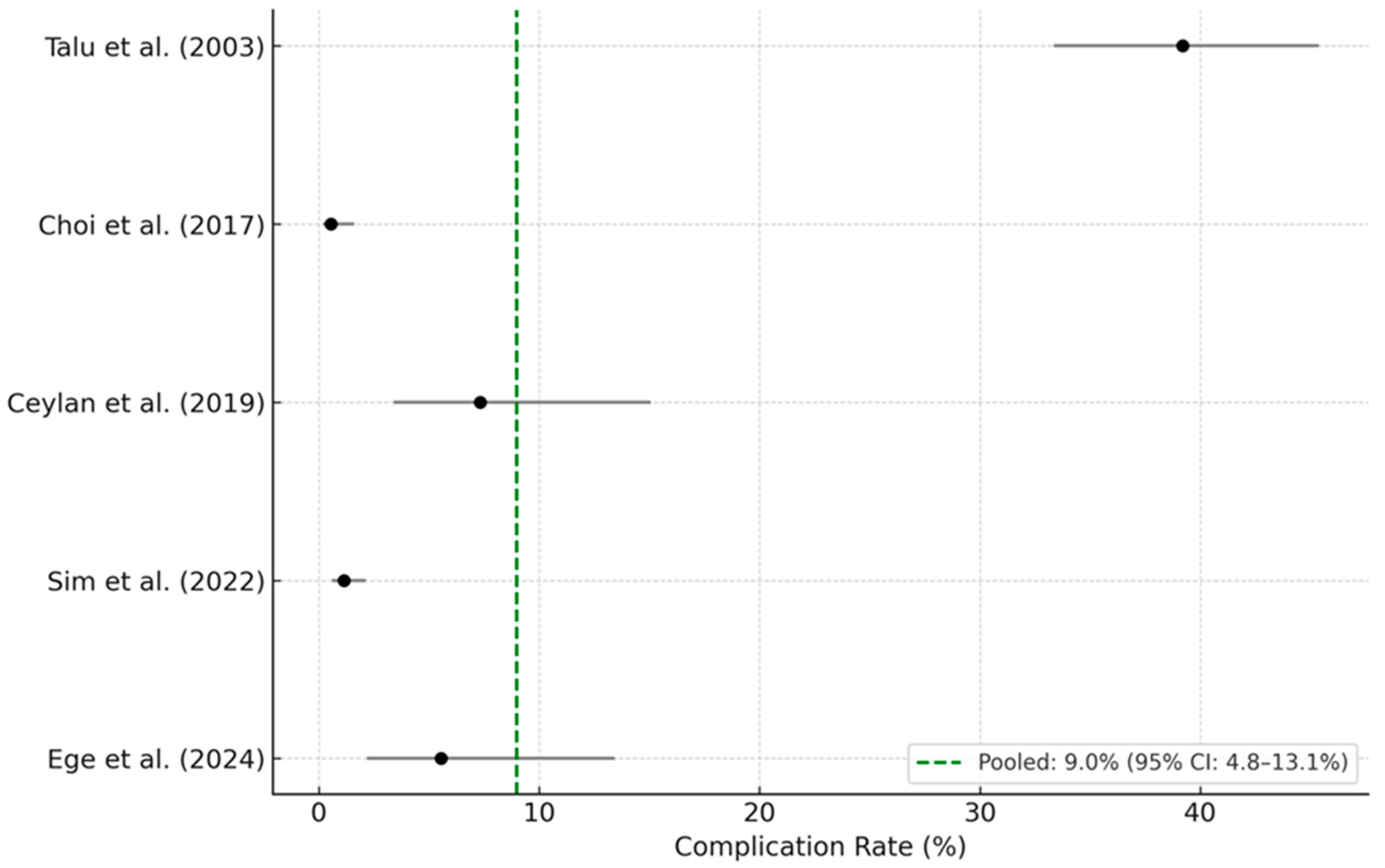
| Study | Year | Country | Population and Indication | Sample Size (N) | Complications (n) | Reported Complications |
|---|---|---|---|---|---|---|
| Sim et al. [22]. | 2022 | South Korea | LSS + RNR undergoing balloon PEN | 793 | 9 | Hypotension, paresthesia, local infection |
| Ceylan et al. [20] | 2019 | Turkey | FBSS treated with epiduroscopy | 82 | 6 | Dural puncture, mild infections, transient symptoms |
| Ege et al. [17] | 2024 | Turkey | Lumbar epidural fibrosis (post-/non-op) | 72 | 4 | Neurologic symptoms, hypotension |
| Talu et al. [13] | 2003 | Turkey | Mixed spinal pain; Racz neuroplasty | 250 | 98 | Dural puncture, catheter issues, infections |
| Choi et al. [7] | 2017 | South Korea | LSS patients with/without sarcopenia treated with balloon PEN | 543 | 3 | Mild hypotension, transient discomfort |
| Authore (Year) | Selection | Comparability | Outcome | Total |
|---|---|---|---|---|
| Sim et al. (2022) [22] | 4 | 2 | 2 | 8 |
| Ceylan et al. (2019) [20] | 4 | 2 | 2 | 8 |
| Ege et al. (2024) [17] | 4 | 2 | 2 | 8 |
| Talu et al. (2003) [13] | 4 | 0 | 2 | 6 |
| Choi et al. (2017) [7] | 4 | 0 | 2 | 6 |
| First Author | Year | Country | Complication Type | Description |
|---|---|---|---|---|
| Kim, CH et al. [23] | 2023 | Korea | Hematoma/Neurologic | Massive epidural hematoma → permanent paralysis |
| Kim, SB et al. [24] | 2014 | Korea | Neurologic | Bilateral subdural hematoma |
| Oh, CH et al. [25] | 2013 | Korea | Imaging artifact | Contrast mimicking SAH on imaging |
| Noh, SM et al. [26] | 2020 | Korea | Infectious | Bacterial meningitis and ventriculitis |
| Lee, HY et al. [27] | 2015 | Korea | Infectious/Neurologic | Epidural abscess → cerebellar infarction |
| Lim, YS et al. [28] | 2015 | Korea | Mechanical | Barotrauma-induced acute motor weakness |
| Ryu, KS et al. [29] | 2012 | Korea | Structural | Iatrogenic intradural cyst formation |
| Lee, CH et al. [30] | 2015 | Korea | Systemic | Reverse Takotsubo cardiomyopathy post-intrathecal injection |
| Kim, TH et al. [31] | 2016 | England | Mechanical | Catheter breakage during procedure |
| Kang JH et al. [32] | 2015 | Korea | Mechanical | Retained Racz catheter fragment |
| Ho, KY et al. [33] | 2008 | USA | Neurologic | Acute monoplegia after adhesiolysis |
| Beyaz, SG et al. [8] | 2018 | India | Systemic | Cardiopulmonary arrest + pneumocephalus post-O2–O3 |
| Torman, H et al. [9] | 2017 | Turkey | Systemic | Severe headache and pneumocephalus |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.H.; Yoon, E.J.; Jo, S.H.; Kim, S.O.; Lee, D.W.; Kim, H.H. Complications Following Percutaneous Epidural Neuroplasty: A Narrative Review of Clinical Evidence and the Rationale for Post-Procedural 6 h Inpatient Monitoring Amid Limited Systematic Data. Medicina 2025, 61, 1397. https://doi.org/10.3390/medicina61081397
Kim JH, Yoon EJ, Jo SH, Kim SO, Lee DW, Kim HH. Complications Following Percutaneous Epidural Neuroplasty: A Narrative Review of Clinical Evidence and the Rationale for Post-Procedural 6 h Inpatient Monitoring Amid Limited Systematic Data. Medicina. 2025; 61(8):1397. https://doi.org/10.3390/medicina61081397
Chicago/Turabian StyleKim, Jae Hun, Eun Jang Yoon, Sung Ho Jo, Sun Ok Kim, Dong Woo Lee, and Hwan Hee Kim. 2025. "Complications Following Percutaneous Epidural Neuroplasty: A Narrative Review of Clinical Evidence and the Rationale for Post-Procedural 6 h Inpatient Monitoring Amid Limited Systematic Data" Medicina 61, no. 8: 1397. https://doi.org/10.3390/medicina61081397
APA StyleKim, J. H., Yoon, E. J., Jo, S. H., Kim, S. O., Lee, D. W., & Kim, H. H. (2025). Complications Following Percutaneous Epidural Neuroplasty: A Narrative Review of Clinical Evidence and the Rationale for Post-Procedural 6 h Inpatient Monitoring Amid Limited Systematic Data. Medicina, 61(8), 1397. https://doi.org/10.3390/medicina61081397






