Ecology and Epidemiology of Lyme Disease in Western North America
Abstract
Simple Summary
Abstract
1. Introduction
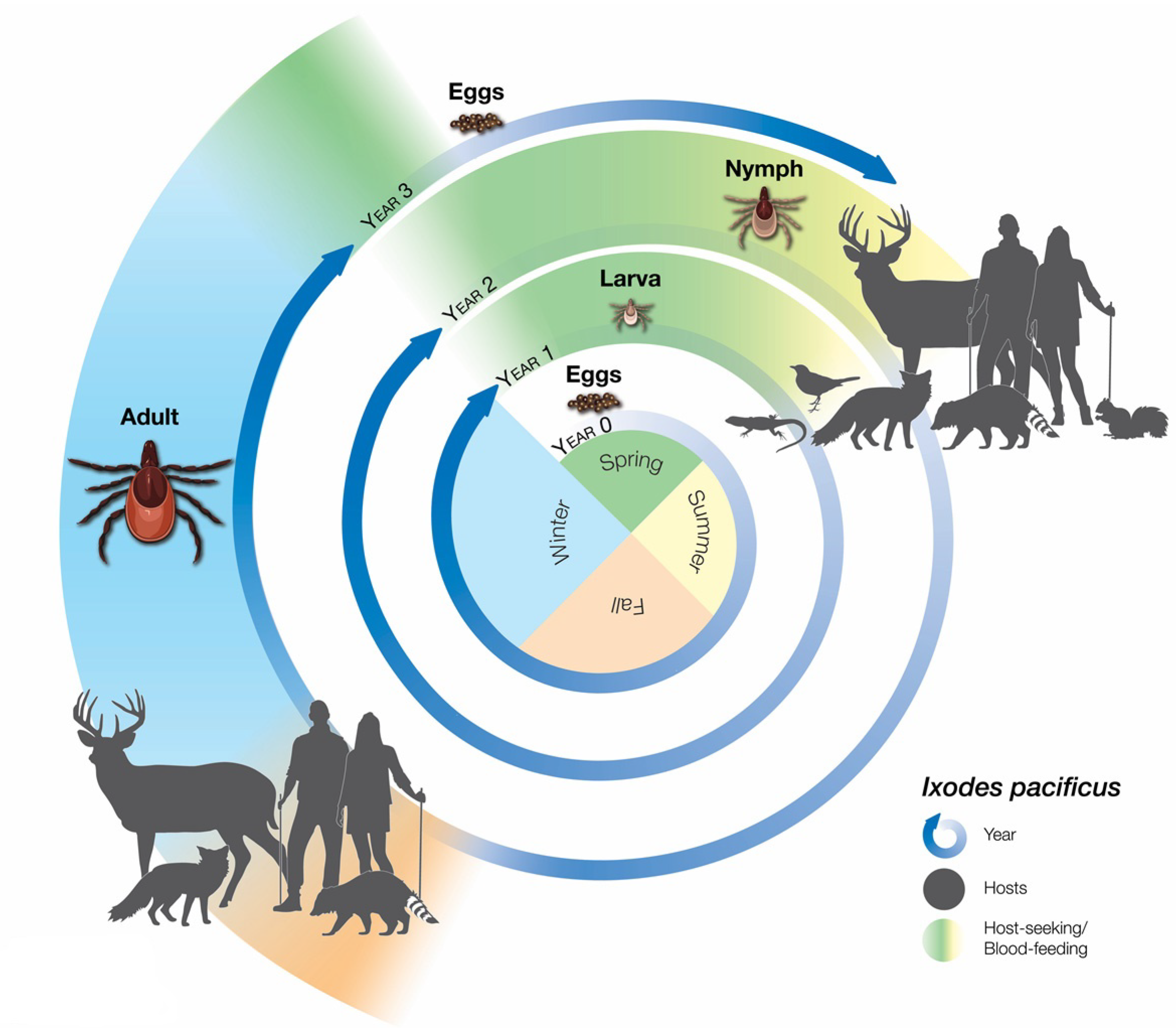
2. Surveillance Efforts to Track Risk of Exposure to Ixodes pacificus
3. Infection Prevalence of Ixodes pacificus with Borrelia burgdorferi
4. Surveillance Efforts Tracking Lyme Disease Cases
5. Mammalian and Reptilian Hosts and Their Roles in Lyme Disease Maintenance and Transmission
6. Avian Hosts and Their Role in Lyme Disease Transmission and Tick Dispersal
7. The Effects of Community-Level Dynamics on Lyme Disease Maintenance and Transmission
8. Additional Tick Vectors of the Lyme Disease Spirochete in Western North America
9. Environmental and Climatic Factors Affecting Ixodes pacificus and Borrelia burgdorferi
10. Modelling the Geographic Distributions of Ixodes pacificus and Borrelia burgdorferi
| Species Distribution Modelling Method | References for Method | Species Dataset Required | Sample Studies |
|---|---|---|---|
| Maximum Entropy (MaxEnt) | Phillips, S.J., R.P. Anderson, and R.E. Schapire (2006). Maximum entropy modeling of species geographic distributions. Ecological Modelling, 190, pp. 231–259. | Presence | [111,112,122,131,133,134,137] |
| Boosted Regression Trees (BRTs) | Elith, J., C. Graham, and the NCEAS species distribution modeling group. (2006). Novel methods improve prediction of species’ distributions from occurrence data. Ecography, 29(2), pp. 129–151. Elith, J. and Leathwick, J.R. (2007). Predicting species’ distributions from museum and herbarium records using multiresponse models fitted with multivariate adaptive regression splines. Diversity and Distributions, 13, pp. 165–175. Leathwick, J.R., Elith, J., and Hastie, T. (2006). Comparative performance of generalized additive models and multivariate adaptive regression splines for statistical modelling of species distributions. Ecological Modelling, 199(2), pp. 188–196. Lehman, A., Overton, J.M., and Leathwick, J.R. (2002). GRASP: generalized regression analysis and spatial prediction. Ecological Modelling, 157, pp. 189–207. | Presence or Presence/Pseudo-absence | [111,112,133,134,137] |
| Multivariate Adaptive Regression Splines (MARS) | Presence/Absence or Pseudo-absence | [111,112,133,134,137] | |
| Generalized Linear Models (GLMs) | Presence/Absence or Pseudo-absence | [111,112,133,134,137] | |
| Genetic Algorithm for Rule-Set Prediction (GARP) | Stockwell, D.R.B., and D.P. Peters. 1999. The GARP modelling system: Problems and solutions to automated spatial prediction. International Journal of Geographical Information Systems, 13, pp. 143–158. | Presence | [109] |
| Random Forest (RF) | Breiman, L. (2001). Random forests. Machine Learning, 45, pp. 5–32. | Presence/Absence or Pseudo-absence | [111,112,133,134,137] |
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rosenberg, R.; Lindsey, N.P.; Fischer, M.; Gregory, C.J.; Hinckley, A.F.; Mead, P.S.; Paz-Bailey, G.; Waterman, S.H.; Drexler, N.A.; Kersh, G.J.; et al. Vital signs: Trends in reported vectorborne disease cases—United States and Territories, 2004–2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 496–501. [Google Scholar] [CrossRef]
- Lindsay, L.R. Present state of common vector-borne diseases in Canada. Can. Commun. Dis. Rep. 2016, 42, 200–201. [Google Scholar] [CrossRef] [PubMed]
- Marques, A.R.; Strle, F.; Wormser, G.P. Comparison of Lyme disease in the United States and Europe. Emerg. Infect. Dis. 2021, 27, 2017–2024. [Google Scholar] [CrossRef] [PubMed]
- Kugeler, K.J.; Schwartz, A.M.; Delorey, M.J.; Mead, P.S.; Hinckley, A.F. Estimating the frequency of Lyme disease diagnoses, United States, 2010–2018. Emerg. Infect. Dis. 2021, 27, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, S.; Koffi, J.; Nelder, M.; Russell, C.; Graham-Derham, S.; Lachance, L.; Adhikari, B.; Badcock, J.; Baidoobonso, S.; Billard, B.; et al. Surveillance for Lyme disease in Canada, 2009–2019. Can. Commun. Dis. Rep. 2022, 48, 219–227. [Google Scholar] [CrossRef]
- Signs and Symptoms of Lyme Disease. Available online: https://www.cdc.gov/lyme/signs_symptoms/index.html (accessed on 25 October 2022).
- Kugeler, K.J.; Griffith, K.S.; Gould, L.H.; Kochanek, K.; Delorey, M.J.; Biggerstaff, B.J.; Mead, P.S. A review of death certificates listing lyme disease as a cause of death in the United States. Clin. Infect. Dis. 2011, 52, 364–367. [Google Scholar] [CrossRef]
- Johnson, R.C.; Schmid, G.P.; Hyde, F.W.; Steigerwalt, A.G.; Brenner, D.J. Borrelia Burgdorferi sp. nov.: Etiologic agent of Lyme disease. Int. J. Syst. Bacteriol. 1984, 34, 496–497. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Beard, C.B. County-scale distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the continental United States. J. Med. Entomol. 2016, 53, 349–386. [Google Scholar] [CrossRef]
- Ogden, N.H.; Lindsay, L.R.; Morshed, M.; Sockett, P.N.; Artsob, H. The emergence of Lyme disease in Canada. Cmaj 2009, 180, 1221–1224. [Google Scholar] [CrossRef]
- Cooley, R.A.; Kohls, G.M. Ixodes californicus Banks, 1904, Ixodes Pacificus n. sp., and Ixodes conepati n. sp. (Acarina: Ixodidae). Pan-Pac. Entomol. 1943, 19, 139–147. [Google Scholar]
- Cooley, R.A.; Kohls, G.M. The genus Ixodes in North America; United States Government Printing Office: Washington, DC, USA, 1945; pp. 21–28. [Google Scholar]
- Lindquist, E.E.; Galloway, T.D.; Artsob, H.; Lindsay, L.R.; Drebot, M.; Wood, H.; Robbins, R.G. A Handbook to the Ticks of Canada (Ixodida: Ixodidae, Argasidae); Biological Survey of Canada: Sackville, NB, Canada, 2016. [Google Scholar]
- Eisen, R.J.; Eisen, L.; Ogden, N.H.; Beard, C.B. Linkages of weather and climate with Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae), enzootic transmission of Borrelia burgdorferi, and Lyme disease in North America. J. Med. Entomol. 2016, 53, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, L.R.; Barker, I.K.; Surgeoner, G.A.; Mcewen, S.A.; Gillespie, T.J.; Addison, E.M. Survival and development of the different life stages of Ixodes scapularis (Acari: Ixodidae) held within four habitats on Long Point, Ontario, Canada. J. Med. Entomol. 1998, 35, 189–199. [Google Scholar] [CrossRef] [PubMed]
- How Ticks Spread Disease. Available online: https://www.cdc.gov/ticks/life_cycle_and_hosts.html#print (accessed on 25 October 2022).
- Schwartz, A.M.; Hinckley, A.F.; Mead, P.S.; Hook, S.A.; Kugeler, K.J. Surveillance for Lyme disease—United States, 2008–2015. MMWR Surveill. Summ. 2017, 66, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, S.; Ogden, N.; Lindsay, L.; Burns, S.; Fleming, S.; Badcock, J.; Hanan, S.; Gaulin, C.; Leblanc, M.; Russell, C.; et al. Surveillance for Lyme disease in Canada: 2009–2015. Can. Commun. Dis. Rep. 2017, 43, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Lyons, L.A.; Brand, M.E.; Gronemeyer, P.; Mateus-Pinilla, N.; Ruiz, M.O.; Stone, C.M.; Tuten, H.C.; Smith, R.L. Comparing contributions of passive and active tick collection methods to determine establishment of ticks of public health concern within Illinois. J. Med. Entomol. 2021, 58, 1849–1864. [Google Scholar] [CrossRef] [PubMed]
- Vector-Borne Disease Section Annual Report 2020. Available online: https://westnile.ca.gov/pdfs/VBDSAnnualReport20.pdf (accessed on 25 October 2022).
- Doggett, J.S.; Kohlhepp, S.; Gresbrink, R.; Metz, P.; Gleaves, C.; Gilbert, D. Lyme disease in Oregon. J. Clin. Microbiol. 2008, 46, 2115–2118. [Google Scholar] [CrossRef] [PubMed]
- Dykstra, E.A.; Oltean, H.N.; Kangiser, D.; Marsden-Haug, N.; Rich, S.M.; Xu, G.; Lee, M.-K.; Morshed, M.G.; Graham, C.B.; Eisen, R.J. Ecology and epidemiology of tickborne pathogens, Washington, USA, 2011–2016. Emerg. Infect. Dis. 2020, 26, 648–832. [Google Scholar] [CrossRef]
- Wilson, C.; Gasmi, S.; Bourgeois, A.-C.; Badcock, J.; Chahil, N.; Kulkarni, M.; Lee, M.-K.; Lindsay, R.; Leighton, P.; Morshed, M.; et al. Surveillance for Ixodes scapularis and Ixodes pacificus ticks and their associated pathogens in Canada, 2019. Can. Commun. Dis. Rep. 2022, 48, 208–218. [Google Scholar] [CrossRef]
- Guillot, C.; Badcock, J.; Clow, K.; Cram, J.; Dergousoff, S.; Dibernardo, A.; Evason, M.; Fraser, E.; Galanis, E.; Gasmi, S.; et al. Sentinel surveillance of Lyme disease risk in Canada, 2019: Results from the first year of the Canadian Lyme Sentinel Network (CaLSeN). Can. Commun. Dis. Rep. 2020, 46, 354–361. [Google Scholar] [CrossRef]
- Lyme Disease in California. Available online: https://storymaps.arcgis.com/stories/f64d0c19a3ab42cf90e8ce38397e96e0 (accessed on 25 October 2022).
- Arthur, D.R.; Snow, K.R. Ixodes pacificus Cooley and Kohls, 1943: Its life-history and occurrence. Parasitology 1968, 58, 893–906. [Google Scholar] [CrossRef]
- Easton, E.R.; Keirans, J.E.; Gresbrink, R.A.; Clifford, C.M. The distribution in Oregon of Ixodes pacificus, Dermacentor andersoni, and Dermacentor occidentalis with a note on Dermacentor variabilis (Acarina: Ixodidae). J. Med. Entomol. 1977, 13, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Arnason, C.S. Biology of the Western Black-Legged Tick, Ixodes pacificus (Cooley and Kohls, 1943): A Potential Vector of Lyme Disease in South Coastal British Columbia. Master’s Thesis, Simon Fraser University, Burnaby, BC, Canada, 1992. [Google Scholar]
- Gregson, J.D. A preliminary report of the lizard-tick relationship on the coast of British Columbia. Proc. Entomol. Soc. Br. Columb. 1934, 31, 17–21. [Google Scholar]
- Eisen, L.; Eisen, R.J.; Lane, R.S. Geographical distribution patterns and habitat suitability models for presence of host-seeking Ixodid ticks in dense woodlands of Mendocino County, California. J. Med. Entomol. 2006, 43, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S.; Steinlein, D.B.; Mun, J. Human behaviors elevating exposure to Ixodes pacificus (Acari: Ixodidae) nymphs and their associated bacterial zoonotic agents in a hardwood forest. J. Med. Entomol. 2004, 41, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Swei, A.; Meentemeyer, R.; Briggs, C.J. Influence of abiotic and environmental factors on the density and infection prevalence of Ixodes pacificus (Acari: Ixodidae) with Borrelia burgdorferi. J. Med. Entomol. 2011, 48, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Pearson, P.; Dykstra, E.; Andrews, E.S.; Rich, S.M. Human-biting Ixodes ticks and pathogen prevalence from California, Oregon, and Washington. Vector Borne Zoonotic Dis. 2019, 19, 106–114. [Google Scholar] [CrossRef]
- Salkeld, D.J.; Porter, W.T.; Loh, S.M.; Nieto, N.C. Time of year and outdoor recreation affect human exposure to ticks in California, United States. Ticks Tick Borne Dis. 2019, 10, 1113–1117. [Google Scholar] [CrossRef]
- Bouchard, C.; Dibernardo, A.; Koffi, J.; Wood, H.; Leighton, P.; Lindsay, L. Increased risk of tick-borne diseases with climate and environmental Changes. Can. Commun. Dis. Rep. 2019, 45, 83–89. [Google Scholar] [CrossRef]
- eTick. Available online: https://www.etick.ca/en (accessed on 25 October 2022).
- iNaturalist. Available online: https://www.inaturalist.org/ (accessed on 25 October 2022).
- Transmission of Lyme Disease. Available online: https://www.cdc.gov/lyme/transmission/index.html (accessed on 25 October 2022).
- Surveillance for Ixodes pacificus and Pathogens Found in This Tick Species in the United States. Available online: https://www.cdc.gov/ticks/resources/TickSurveillance_Ipacificus-P.pdf (accessed on 25 October 2022).
- Hacker, G.M.; Jackson, B.T.; Niemela, M.; Andrews, E.S.; Danforth, M.E.; Pakingan, M.J.; Novak, M.G. A comparison of questing substrates and environmental factors that influence nymphal Ixodes pacificus (Acari: Ixodidae) abundance and seasonality in the Sierra Nevada Foothills of California. J. Med. Entomol. 2021, 58, 1880–1890. [Google Scholar] [CrossRef]
- Clover, J.R.; Lane, R.S. Evidence implicating nymphal Ixodes pacificus (Acari: Ixodidae) in the epidemiology of Lyme disease in California. Am. J. Trop. Med. Hyg. 1995, 53, 237–240. [Google Scholar] [CrossRef]
- Kramer, V.L.; Beesley, C. Temporal and spatial distribution of Ixodes pacificus and Dermacentor occidentalis (Acari: Ixodidae) and prevalence of Borrelia burgdorferi in Contra Costa County, California. J. Med. Entomol. 1993, 30, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S.; Fedorova, N.; Kleinjan, J.E.; Maxwell, M. Eco-epidemiological factors contributing to the low risk of human exposure to Ixodid tick-borne borreliae in Southern California, USA. Ticks Tick Borne Dis. 2013, 4, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S.; Mun, J.; Peribáñez, M.A.; Fedorova, N. Differences in prevalence of Borrelia burgdorferi and Anaplasma spp. infection among host-seeking Dermacentor occidentalis, Ixodes pacificus, and Ornithodoros coriaceus ticks in Northwestern California. Ticks Tick Borne Dis. 2010, 1, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S.; Kucera, T.F.; Barrett, R.H.; Mun, J.; Wu, C.; Smith, V.S. Wild turkey (Meleagris gallopavo) as a host of Ixodid ticks, lice, and Lyme disease spirochetes (Borrelia burgdorferi sensu lato) in California state parks. J. Wildl. Dis. 2006, 42, 759–771. [Google Scholar] [CrossRef]
- Padgett, K.A.; Bonilla, D.L. Novel exposure sites for nymphal Ixodes pacificus within picnic areas. Ticks Tick Borne Dis. 2011, 2, 191–195. [Google Scholar] [CrossRef]
- Lane, R.S.; Loye, J.E. Lyme disease in California: Interrelationship of Ixodes pacificus (Acari: Ixodidae), the western fence lizard (Sceloporus occidentalis), and Borrelia burgdorferi. J. Med. Entomol. 1989, 26, 272–278. [Google Scholar] [CrossRef]
- Dingler, R.J.; Wright, S.A.; Donohue, A.M.; Macedo, P.A.; Foley, J.E. Surveillance for Ixodes pacificus and the tick-borne pathogens Anaplasma phagocytophilum and Borrelia burgdorferi in birds from California’s Inner Coast Range. Ticks Tick Borne Dis. 2014, 5, 436–445. [Google Scholar] [CrossRef]
- Billeter, S.A.; Yoshimizu, M.H.; Hu, R. Species composition and temporal distribution of adult Ixodid ticks and prevalence of Borrelia burgdorferi sensu lato and Rickettsia species in Orange County, California. J. Vector Ecol. 2017, 42, 189–192. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Briggs, C.J. Truncated seasonal activity patterns of the western blacklegged tick (Ixodes pacificus) in Central and Southern California. Ticks Tick Borne Dis. 2016, 7, 234–242. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J.; Lane, R.S. Seasonal activity patterns of Ixodes pacificus nymphs in relation to climatic conditions. Med. Vet. Entomol. 2002, 16, 235–244. [Google Scholar] [CrossRef]
- Lane, R.S.; Manweiler, S.A.; Stubbs, H.A.; Lennette, E.T.; Madigan, J.E.; Lavoie, P.E. Risk factors for Lyme disease in a small rural community in Northern California. Am. J. Epidemiol. 1992, 136, 1358–1368. [Google Scholar] [CrossRef]
- Rose, I.; Yoshimizu, M.H.; Bonilla, D.L.; Fedorova, N.; Lane, R.S.; Padgett, K.A. Phylogeography of Borrelia spirochetes in Ixodes pacificus and Ixodes spinipalpis ticks highlights differential acarological risk of tick-borne disease transmission in Northern versus Southern California. PLoS ONE 2019, 14, e0214726. [Google Scholar] [CrossRef] [PubMed]
- Padgett, K.; Bonilla, D.; Kjemtrup, A.; Vilcins, I.-M.; Yoshimizu, M.H.; Hui, L.; Sola, M.; Quintana, M.; Kramer, V. Large scale spatial risk and comparative prevalence of Borrelia miyamotoi and Borrelia burgdorferi sensu lato in Ixodes pacificus. PLoS ONE 2014, 9, e110853. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, N.; Kleinjan, J.E.; James, D.; Hui, L.T.; Peeters, H.; Lane, R.S. Remarkable diversity of tick or mammalian-associated borreliae in the metropolitan San Francisco Bay Area, California. Ticks Tick Borne Dis. 2014, 5, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Salkeld, D.J.; Lagana, D.M.; Wachara, J.; Porter, W.T.; Nieto, N.C. Examining prevalence and diversity of tick-borne pathogens in questing Ixodes pacificus ticks in California. Appl. Environ. Microbiol. 2021, 87, e00319-21. [Google Scholar] [CrossRef] [PubMed]
- Schwan, T.G.; Schrumpf, M.E.; Karstens, R.H.; Clover, J.R.; Wong, J.; Daugherty, M.; Struthers, M.; Rosa, P.A. Distribution and molecular analysis of Lyme disease spirochetes, Borrelia burgdorferi, isolated from ticks throughout California. J. Clin. Microbiol. 1993, 31, 3096–3108. [Google Scholar] [CrossRef]
- Wright, S.A.; Thompson, M.A.; Miller, M.J.; Knerl, K.M.; Elms, S.L.; Karpowicz, J.C.; Young, J.F.; Kramer, V.L. Ecology of Borrelia burgdorferi in ticks (Acari: Ixodidae), rodents, and birds in the Sierra Nevada Foothills, Placer County, California. J. Med. Entomol. 2000, 37, 909–918. [Google Scholar] [CrossRef]
- Wright, S.A.; Lane, R.S.; Clover, J.R. Infestation of the southern alligator lizard (Squamata: Anguidae) by Ixodes pacificus (Acari: Ixodidae) and its susceptibility to Borrelia burgdorferi. J. Med. Entomol. 1998, 35, 1044–1049. [Google Scholar] [CrossRef]
- Salkeld, D.J.; Lane, R.S. Community ecology and disease risk: Lizards, squirrels, and the Lyme disease spirochete in California, USA. Ecology 2010, 91, 293–298. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Girard, Y.A.; Fedorova, N.; Mun, J.; Slikas, B.; Leonhard, S.; Kitron, U.; Lane, R.S. A spatially-explicit model of acarological risk of exposure to Borrelia burgdorferi-infected Ixodes pacificus nymphs in Northwestern California based on woodland type, temperature, and water vapor. Ticks Tick Borne Dis. 2010, 1, 35–43. [Google Scholar] [CrossRef]
- Feldman, K.A.; Connally, N.P.; Hojgaard, A.; Jones, E.H.; White, J.L.; Hinckley, A.F. Abundance and infection rates of Ixodes scapularis nymphs collected from residential properties in Lyme disease-endemic areas of Connecticut, Maryland, and New York. J. Vector Ecol. 2015, 40, 198–201. [Google Scholar] [CrossRef]
- Hutchinson, M.L.; Strohecker, M.D.; Simmons, T.W.; Kyle, A.D.; Helwig, M.W. Prevalence rates of Borrelia burgdorferi (Spirochaetales: Spirochaetaceae), Anaplasma phagocytophilum (Rickettsiales: Anaplasmataceae), and Babesia microti (Piroplasmida: Babesiidae) in host-seeking Ixodes scapularis (Acari: Ixodidae) from Pennsylvania. J. Med. Entomol. 2015, 52, 693–698. [Google Scholar] [CrossRef]
- Morshed, M.G.; Scott, J.D.; Fernando, K.; Beati, L.; Mazerolle, D.F.; Geddes, G.; Durden, L.A. Migratory songbirds disperse ticks across Canada, and first isolation of the Lyme disease spirochete, Borrelia burgdorferi, from the avian tick, Ixodes auritulus. J. Parasitol. 2005, 91, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Kanji, J.N.; Isaac, A.; Gregson, D.; Mierzejewski, M.; Shpeley, D.; Tomlin, P.; Groeschel, M.; Lindsay, L.R.; Lachance, L.; Kowalewska-Grochowska, K. Epidemiology of ticks submitted from human hosts in Alberta, Canada (2000–2019). Emerg. Microbes Infect. 2022, 11, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.S.; Ramirez, R.A.; Anderson, J.L.; Bernhardt, S.A. Distribution and habitat of Ixodes pacificus (Acari: Ixodidae) and prevalence of Borrelia burgdorferi in Utah. J. Med. Entomol. 2015, 52, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- TCC-3W. Tick Species Submitted in Western Canada, 2022. Available online: http://www.bccdc.ca/Documents/T3WProjectInfographic-FINAL-220615.pdf (accessed on 21 October 2022).
- Surveillance Data. Available online: https://www.cdc.gov/lyme/datasurveillance/surveillance-data.html (accessed on 25 October 2022).
- Lyme Disease Surveillance Report 2019. Available online: https://www.canada.ca/content/dam/phac-aspc/documents/services/publications/diseases-conditions/lyme-disease-surveillance-report-2019/LD-REPORT2019-ENG-Final.pdf (accessed on 25 October 2022).
- Watson, S.C.; Liu, Y.; Lund, R.B.; Gettings, J.R.; Nordone, S.K.; McMahan, C.S.; Yabsley, M.J. A Bayesian spatio-temporal model for forecasting the prevalence of antibodies to Borrelia burgdorferi, causative agent of Lyme disease, in domestic dogs within the contiguous United States. PLoS ONE 2017, 12, e0174428. [Google Scholar] [CrossRef]
- Parasite Prevalence Maps. Available online: https://www.petsandparasites.org/parasite-prevalence-maps#/ (accessed on 25 October 2022).
- Salkeld, D.J.; Castro, M.B.; Bonilla, D.; Kjemtrup, A.; Kramer, V.L.; Lane, R.S.; Padgett, K.A. Seasonal activity patterns of the western black-legged tick, Ixodes pacificus, in relation to onset of human Lyme disease in Northwestern California. Ticks Tick Borne Dis. 2014, 5, 790–796. [Google Scholar] [CrossRef]
- Lane, R.S.; Voie, P.E.L. Lyme borreliosis in California acarological, clinical, and epidemiological Studies. Ann. N. Y. Acad. Sci. 1988, 539, 192–203. [Google Scholar] [CrossRef]
- Manweiler, S.A.; Lane, R.S.; Tempelis, C.H. The western fence lizard Sceloporus occidentalis: Evidence of field exposure to Borrelia burgdorferi in relation to infestation by Ixodes pacificus (Acari: Ixodidae). Am. J. Trop. Med. Hyg. 1992, 47, 328–336. [Google Scholar] [CrossRef]
- Tälleklint-Eisen, L.; Eisen, R.J. Abundance of Ticks (Acari: Ixodidae) Infesting the western fence lizard, Sceloporus occidentalis, in relation to environmental factors. Exp. Appl. Acarol. 1999, 23, 731–740. [Google Scholar] [CrossRef]
- Wright, S.A.; Lemenager, D.A.; Tucker, J.R.; Armijos, M.V.; Yamamoto, S.A. An avian contribution to the presence of Ixodes pacificus (Acari: Ixodidae) and Borrelia burgdorferi on the Sutter Buttes of California. J. Med. Entomol. 2006, 43, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Slowik, T.J.; Lane, R.S. Birds and their ticks in Northwestern California: Minimal contribution to Borrelia burgdorferi enzootiology. J. Parasitol. 2001, 87, 755–761. [Google Scholar] [CrossRef]
- Newman, E.A.; Eisen, L.; Eisen, R.J.; Fedorova, N.; Hasty, J.M.; Vaughn, C.; Lane, R.S. Borrelia burgdorferi sensu lato spirochetes in wild birds in Northwestern California: Associations with ecological factors, bird behavior and tick infestation. PLoS ONE 2015, 10, e0118146. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.A.; Tucker, J.R.; Donohue, A.M.; Castro, M.B.; Kelley, K.L.; Novak, M.G.; Macedo, P.A. Avian hosts of Ixodes pacificus (Acari: Ixodidae) and the detection of Borrelia burgdorferi in larvae feeding on the Oregon junco. J. Med. Entomol. 2011, 48, 852–859. [Google Scholar] [CrossRef]
- Lane, R.S.; Burgdorfer, W. Potential role of native and exotic deer and their associated ticks (Acari: Ixodidae) in the ecology of Lyme disease in California, USA. Zentralbl. Bakteriol. Mikrobiol. Hyg. A. 1986, 263, 55–64. [Google Scholar] [CrossRef]
- Morshed, M.G.; Lee, M.-K.; Man, S.; Fernando, K.; Wong, Q.; Hojgaard, A.; Tang, P.; Mak, S.; Henry, B.; Patrick, D.M. Surveillance for Borrelia burgdorferi in Ixodes ticks and small rodents in British Columbia. Vector Borne Zoonotic Dis. 2015, 15, 701–705. [Google Scholar] [CrossRef]
- Peavey, C.A.; Lane, R.S. Transmission of Borrelia burgdorferi by Ixodes pacificus nymphs and reservoir competence of deer mice (Peromyscus maniculatus) infected by tick-bite. J. Parasitol. 1995, 81, 175–178. [Google Scholar] [CrossRef]
- Lane, R.S.; Loye, J.E. Lyme disease in California: Interrelationship of Ixodid ticks (Acari), rodents, and Borrelia burgdorferi. J. Med. Entomol. 1991, 28, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Eisen, L.; Eisen, R.J.; Lane, R.S. The roles of birds, lizards, and rodents as hosts for the western black-legged tick Ixodes pacificus. J. Vector Ecol. 2004, 29, 295–308. [Google Scholar] [PubMed]
- Eisen, L.; Eisen, R.J.; Mun, J.; Salkeld, D.J.; Lane, R.S. Transmission cycles of Borrelia burgdorferi and B. bissettii in relation to habitat type in Northwestern California. J. Vector Ecol. 2009, 34, 81–91. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Weinstein, S.B.; O’Connor, K.E.; Swei, A. Circulation of tick-borne spirochetes in tick and small mammal communities in Santa Barbara County, California, USA. J. Med. Entomol. 2020, 57, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.N.; Lane, R.S. Reservoir competence of four chaparral-dwelling rodents for Borrelia burgdorferi in California. Am. J. Trop. Med. Hyg. 1996, 54, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Swei, A.; Ostfeld, R.S.; Lane, R.S.; Briggs, C.J. Impact of the experimental removal of lizards on Lyme disease risk. Proc. R. Soc. B Biol. Sci. 2011, 278, 2970–2978. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Hyon, D.W.; McDaniels, A.; O’Connor, K.E.; Swei, A.; Briggs, C.J. Risk of vector tick exposure initially increases, then declines through time in response to wildfire in California. Ecosphere 2018, 9, e02227. [Google Scholar] [CrossRef]
- Salomon, J.; Lawrence, A.; Crews, A.; Sambado, S.; Swei, A. Host infection and community composition predict vector burden. Oecologia 2021, 196, 305–316. [Google Scholar] [CrossRef]
- Brown, R.N.; Lane, R.S. Lyme disease in California: A novel enzootic transmission cycle of Borrelia burgdorferi. Science 1992, 256, 1439–1442. [Google Scholar] [CrossRef]
- Foley, J.E.; Nieto, N.C. The ecology of tick-transmitted infections in the redwood chipmunk (Tamias ochrogenys). Ticks Tick Borne Dis. 2011, 2, 88–93. [Google Scholar] [CrossRef]
- Lane, R.S.; Mun, J.; Eisen, R.J.; Eisen, L. Western gray squirrel (Rodentia: Sciuridae): A primary reservoir host of Borrelia burgdorferi in Californian oak woodlands? J. Med. Entomol. 2005, 42, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Leonhard, S.; Jensen, K.; Salkeld, D.J.; Lane, R.S. Distribution of the Lyme disease spirochete Borrelia burgdorferi in naturally and experimentally infected western gray squirrels (Sciurus griseus). Vector Borne Zoonotic Dis. 2010, 10, 441–447. [Google Scholar] [CrossRef]
- Salkeld, D.J.; Leonhard, S.; Girard, Y.A.; Hahn, N.; Mun, J.; Padgett, K.A.; Lane, R.S. Identifying the reservoir hosts of the Lyme disease spirochete Borrelia burgdorferi in California: The role of the western gray squirrel (Sciurus griseus). Am. J. Trop. Med. Hyg. 2008, 79, 535–540. [Google Scholar] [CrossRef]
- Lane, R.S.; Mun, J.; Eisen, L.; Eisen, R.J. Refractoriness of the western fence lizard (Sceloporus occidentalis) to the Lyme disease group spirochete Borrelia bissettii. J. Parasitol. 2006, 92, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.S. Susceptibility of the western fence lizard (Sceloporus occidentalis) to the Lyme borreliosis spirochete (Borrelia burgdorferi). Am. J. Trop. Med. Hyg. 1990, 42, 75–82. [Google Scholar] [CrossRef]
- Lane, R.S.; Quistad, G.B. Borreliacidal factor in the blood of the western fence lizard (Sceloporus occidentalis). J. Parasitol. 1998, 84, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Durden, L.A.; Anderson, J.F. Infection prevalence of Borrelia Burgdorferi in ticks collected from songbirds in Far-Western Canada. Open J. Anim. Sci. 2015, 5, 232. [Google Scholar] [CrossRef]
- Lilly, M.; Amaya-Mejia, W.; Pavan, L.; Peng, C.; Crews, A.; Tran, N.; Sehgal, R.; Swei, A. Local community composition drives avian Borrelia burgdorferi infection and tick infestation. Vet. Sci. 2022, 9, 55. [Google Scholar] [CrossRef]
- Linders, M.J.; Stinson, D.W. Western Gray Squirrel Recovery Plan; Washington Department of Fish and Wildlife: Olympia, WA, USA, 2007; p. 140. [Google Scholar]
- Nieto, N.C.; Foley, J.E. Evaluation of squirrels (Rodentia: Sciuridae) as ecologically significant hosts for Anaplasma phagocytophilum in California. J. Med. Ent. 2008, 45, 763–769. [Google Scholar] [CrossRef]
- Marsot, M.; Chapuis, J.-L.; Gasqui, P.; Dozières, A.; Masséglia, S.; Pisanu, B.; Ferquel, E.; Vourc’h, G. Introduced siberian chipmunks (Tamias sibiricus barberi) contribute more to Lyme borreliosis risk than native reservoir rodents. PLoS ONE 2013, 8, e55377. [Google Scholar] [CrossRef] [PubMed]
- Slajchert, T.; Kitron, U.D.; Jones, C.J.; Mannelli, A. Role of the eastern chipmunk (Tamias striatus) in the epizootiology of Lyme borreliosis in Northwestern Illinois, USA. J. Wildl. Dis. 1997, 33, 40–46. [Google Scholar] [CrossRef]
- Peavey, C.A.; Lane, R.S.; Damrow, T. Vector competence of Ixodes angustus (Acari: Ixodidae) for Borrelia burgdorferi sensu stricto. Exp. Appl. Acarol. 2000, 24, 77–84. [Google Scholar] [CrossRef]
- Marcum, L. LYME SCI: Lyme-Carrying Ticks in West Differ from Their Eastern Cousins. Lyme Disease. 2022. Available online: https://www.lymedisease.org/ixodes-pacificus-review/# (accessed on 21 October 2022).
- Scott, J.D.; Clark, K.L.; Foley, J.E.; Anderson, J.F.; Bierman, B.C.; Durden, L.A. Extensive distribution of the Lyme disease bacterium, Borrelia burgdorferi sensu lato, in multiple tick species parasitizing avian and mammalian hosts across Canada. Healthcare 2018, 6, 131. [Google Scholar] [CrossRef]
- Brown, R.N.; Peot, M.A.; Lane, R.S. Sylvatic Maintenance of Borrelia burgdorferi (Spirochaetales) in Northern California: Untangling the web of transmission. J. Med. Entomol. 2006, 43, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Eisen, L.; Dolan, M.C.; Piesman, J.; Lane, R.S. Vector competence of Ixodes pacificus and I. spinipalpis (Acari: Ixodidae), and reservoir competence of the dusky-footed woodrat (Neotoma fuscipes) and the deer mouse (Peromyscus maniculatus), for Borrelia bissettii. J. Med. Entomol. 2003, 40, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Burkot, T.R.; Clover, J.R.; Happ, C.M.; DeBess, E.; Maupin, G.O. Isolation of Borrelia burgdorferi from Neotoma fuscipes, Peromyscus maniculatus, Peromyscus boylii, and Ixodes pacificus in Oregon. Am. J. Trop. Med. Hyg. 1999, 60, 453–457. [Google Scholar] [CrossRef]
- Scott, J.D.; Anderson, J.F.; Durden, L.A. Widespread dispersal of Borrelia burgdorferi–infected ticks collected from songbirds across Canada. J. Parasitol. 2012, 98, 49–59. [Google Scholar] [CrossRef]
- MacDonald, A.J.; Hyon, D.W.; Brewington, J.B.; O’Connor, K.E.; Swei, A.; Briggs, C.J. Lyme disease risk in Southern California: Abiotic and environmental drivers of Ixodes pacificus (Acari: Ixodidae) density and infection prevalence with Borrelia burgdorferi. Parasites Vectors 2017, 10, 7. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J.; Chang, C.-C.; Mun, J.; Lane, R.S. Acarologic risk of exposure to Borrelia burgdorferi spirochaetes: Long-term evaluations in North-Western California, with implications for Lyme borreliosis risk-assessment models. Med. Vet. Entomol. 2004, 18, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Padgett, K.A.; Lane, R.S. Life cycle of Ixodes pacificus (Acari: Ixodidae): Timing of developmental processes under field and laboratory conditions. J. Med. Entomol. 2001, 38, 684–693. [Google Scholar] [CrossRef]
- Nieto, N.C.; Holmes, E.A.; Foley, J.E. Survival rates of immature Ixodes pacificus (Acari: Ixodidae) ticks estimated using field-placed enclosures. J. Vector Ecol. 2010, 35, 43–49. [Google Scholar] [CrossRef]
- Foley, J.E.; Queen, E.V.; Sacks, B.; Foley, P. GIS-facilitated spatial epidemiology of tick-borne diseases in coyotes (Canis latrans) in Northern and Coastal California. Comp. Immunol. Microbiol. Infect. Dis. 2005, 28, 197–212. [Google Scholar] [CrossRef]
- Fleshman, A.C.; Graham, C.B.; Maes, S.E.; Foster, E.; Eisen, R.J. Reported county-level distribution of Lyme disease spirochetes, Borrelia burgdorferi sensu stricto and Borrelia mayonii (Spirochaetales: Spirochaetaceae), in host-seeking Ixodes scapularis and Ixodes pacificus ticks (Acari: Ixodidae) in the contiguous United States. J. Med. Entomol. 2021, 58, 1219–1233. [Google Scholar]
- Lane, R.S.; Mun, J.; Peribáñez, M.A.; Stubbs, H.A. Host-seeking behavior of Ixodes pacificus (Acari: Ixodidae) nymphs in relation to environmental parameters in dense-woodland and woodland-grass habitats. J. Vector Ecol. 2007, 32, 342–357. [Google Scholar] [CrossRef] [PubMed]
- Salkeld, D.J.; Nieto, N.C.; Carbajales-Dale, P.; Carbajales-Dale, M.; Cinkovich, S.S.; Lambin, E.F. Disease risk & landscape attributes of tick-borne Borrelia pathogens in the San Francisco Bay Area, California. PLoS ONE 2015, 10, e0134812. [Google Scholar]
- Eisen, R.J.; Clark, R.J.; Monaghan, A.J.; Eisen, L.; Delorey, M.J.; Beard, C.B. Host-seeking phenology of Ixodes pacificus (Acari: Ixodidae) nymphs in Northwestern California in relation to calendar week, woodland type, and weather conditions. J. Med. Entomol. 2016, 54, 125–131. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Lane, R.S. Predicting density of Ixodes pacificus nymphs in dense woodlands in Mendocino County, California, based on geographic information systems and remote sensing versus field-derived data. Am. J. Trop. Med. Hyg. 2006, 74, 632–640. [Google Scholar] [CrossRef]
- Eisen, R.J.; Lane, R.S.; Fritz, C.L.; Eisen, L. Spatial patterns of Lyme disease risk in California based on disease incidence data and modeling of vector-tick exposure. Am. J. Trop. Med. Hyg. 2006, 75, 669–676. [Google Scholar] [CrossRef]
- Mak, S.; Morshed, M.; Henry, B. Ecological niche modeling of Lyme disease in British Columbia, Canada. J. Med. Entomol. 2010, 47, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Peavey, C.A.; Lane, R.S. Density and spatial distribution of Ixodes pacificus (Acari: Ixodidae) in two recreational areas in North Coastal California. Am. J. Trop. Med. Hyg. 2000, 62, 415–422. [Google Scholar] [CrossRef]
- Lane, R.S. Risk of human exposure to vector ticks (Acari: Ixodidae) in a heavily used recreational area in Northern California. Am. J. Trop. Med. Hyg. 1996, 55, 165–173. [Google Scholar] [CrossRef]
- Carella, E.; Orusa, T.; Viani, A.; Meloni, D.; Borgogno-Mondino, E.; Orusa, R. An integrated, tentative remote-sensing approach based on NDVI entropy to model canine distemper virus in wildlife and to prompt science-based management policies. Animals 2022, 12, 1049. [Google Scholar] [CrossRef]
- Orusa, T.; Orusa, R.; Viani, A.; Carella, E.; Borgogno Mondino, E. Geomatics and EO data to support wildlife diseases assessment at landscape level: A pilot experience to map infectious keratoconjunctivitis in chamois and phenological trends in Aosta Valley (NW Italy). Remote Sens. 2020, 12, 3542. [Google Scholar] [CrossRef]
- Suresh, K.P.; Bylaiah, S.; Patil, S.; Kumar, M.; Indrabalan, U.B.; Panduranga, B.A.; Srinivas, P.T.; Shivamallu, C.; Kollur, S.P.; Cull, C.A.; et al. A new methodology to comprehend the effect of El Niño and La Niña oscillation in early warning of anthrax epidemic among livestock. Zoonotic Dis. 2022, 2, 267–290. [Google Scholar] [CrossRef]
- De Marinis, P.; De Petris, S.; Sarvia, F.; Manfron, G.; Momo, E.J.; Orusa, T.; Corvino, G.; Sali, G.; Borgogno, E.M. Supporting pro-poor reforms of agricultural systems in Eastern DRC (Africa) with remotely sensed data: A possible contribution of spatial entropy to interpret land management practices. Land 2021, 10, 1368. [Google Scholar] [CrossRef]
- Orusa, T.; Borgogno Mondino, E. Exploring short-term climate change effects on rangelands and broad-leaved forests by free satellite data in Aosta Valley (Northwest Italy). Climate 2021, 9, 47. [Google Scholar] [CrossRef]
- Guisan, A.; Zimmermann, N.E. Predictive habitat distribution models in ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- MacDonald, A.J.; O’Neill, C.; Yoshimizu, M.H.; Padgett, K.A.; Larsen, A.E. Tracking seasonal activity of the western blacklegged tick across California. J. Appl. Ecol. 2019, 56, 2562–2573. [Google Scholar] [CrossRef]
- MacDonald, A.J.; McComb, S.; O’Neill, C.; Padgett, K.A.; Larsen, A.E. Projected climate and land use change alter western blacklegged tick phenology, seasonal host-seeking suitability and human encounter risk in California. Glob. Chang. Biol. 2020, 26, 5459–5474. [Google Scholar] [CrossRef] [PubMed]
- Eisen, R.J.; Feirer, S.; Padgett, K.A.; Hahn, M.B.; Monaghan, A.J.; Kramer, V.L.; Lane, R.S.; Kelly, M. Modeling climate suitability of the western blacklegged tick in California. J. Med. Entomol. 2018, 55, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.B.; Jarnevich, C.S.; Monaghan, A.J.; Eisen, R.J. Modeling the geographic distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the contiguous United States. J. Med. Entomol. 2016, 53, 1176–1191. [Google Scholar] [CrossRef]
- Porter, W.T.; Barrand, Z.A.; Wachara, J.; DaVall, K.; Mihaljevic, J.R.; Pearson, T.; Salkeld, D.J.; Nieto, N.C. Predicting the current and future distribution of the western black-legged tick, Ixodes pacificus, across the Western US using citizen science collections. PLoS ONE 2021, 16, e0244754. [Google Scholar] [CrossRef]
- Eisen, R.J.; Eisen, L.; Lane, R.S. Remote sensing (normalized difference vegetation index) classification of risk versus minimal risk habitats for human exposure to Ixodes pacificus (Acari: Ixodidae) nymphs in Mendocino County, California. J. Med. Entomol. 2005, 42, 75–81. [Google Scholar] [CrossRef]
- Witmer, F.D.W.; Nawrocki, T.W.; Hahn, M. Modeling geographic uncertainty in current and future habitat for potential populations of Ixodes pacificus (Acari: Ixodidae) in Alaska. J. Med. Entomol. 2022, 59, 976–986. [Google Scholar] [CrossRef] [PubMed]
- BC Centres for Disease Control. Lyme Disease Risk Areas in British Columbia. Available online: http://www.bccdc.ca/resource-gallery/Documents/Statistics%20and%20Research/Statistics%20and%20Reports/Epid/Vector-bourne/Lyme_Disease_Risk_Areas_Map_BC.pdf (accessed on 21 October 2022).
- Hahn, M.B.; Feirer, S.; Monaghan, A.J.; Lane, R.S.; Eisen, R.J.; Padgett, K.A.; Kelly, M. Modeling future climate suitability for the western blacklegged tick, Ixodes pacificus, in California with an emphasis on land access and ownership. Ticks Tick Borne Dis. 2021, 12, 101789. [Google Scholar] [CrossRef] [PubMed]
- Pearson, R.G. Species’ distribution modeling for conservation educators and practitioners. Lessons Conserv. 2010, 3, 54–89. [Google Scholar]
- What is a GCM? Available online: https://www.ipcc-data.org/guidelines/pages/gcm_guide.html (accessed on 25 October 2022).
- IPCC DDC Glossary. Available online: https://www.ipcc-data.org/guidelines/pages/glossary/glossary_r.html (accessed on 25 October 2022).
- TickReport. Available online: https://www.tickreport.com/ (accessed on 25 October 2022).
| Host | Predominant Feeding Ixodes pacificus Life Stage | I. pacificus Infestation Levels | B. burgdorferi s.s. Infection Rates (Host) | B. burgdorferi s.s. Infection Rates (Attaching Ticks) | Studies |
|---|---|---|---|---|---|
| Peromyscus maniculatus | Nymphs | Low | Low | Low | [81,82,83,84,85,86] |
| Neotoma fuscipes | Larva | Moderate | Low | Low | [55,79,84,87,88,89,90,91] |
| Neotamias ochrogenys | Larva | Moderate | N/A | N/A | [92] |
| Sciurus griseus | Larva and nymphs | Moderate | High | High | [60,84,85,93,94,95] |
| Sceloporus occidentalis | Larva and nymphs | High | Low | Low | [47,74,75,76,77,96,97] |
| Elgaria multicarinata | Larva and nymphs | High | Low | Low | [59] |
| Junco hyemalis | Larva and nymphs | High | Low | Low | [48,58,76,78,79] |
| Pipilo maculatus | Larva | High | N/a | Low | [48,58,76,78,79] |
| Catharus guttatus | Larva | High | Low | N/A | [48,58,76,79] |
| Thryomanes bewickii | Larva | High | Low | Low | [58,76,78] |
| Certhia americana | Nymphs | Low | N/A | N/A | [48,78,79] |
| Haemorhous purpureus | Larva and nymphs | Moderate | N/A | N/A | [48,78,79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dizon, C.; Lysyk, T.J.; Couloigner, I.; Cork, S.C. Ecology and Epidemiology of Lyme Disease in Western North America. Zoonotic Dis. 2023, 3, 20-37. https://doi.org/10.3390/zoonoticdis3010004
Dizon C, Lysyk TJ, Couloigner I, Cork SC. Ecology and Epidemiology of Lyme Disease in Western North America. Zoonotic Diseases. 2023; 3(1):20-37. https://doi.org/10.3390/zoonoticdis3010004
Chicago/Turabian StyleDizon, Carl, Tim J. Lysyk, Isabelle Couloigner, and Susan C. Cork. 2023. "Ecology and Epidemiology of Lyme Disease in Western North America" Zoonotic Diseases 3, no. 1: 20-37. https://doi.org/10.3390/zoonoticdis3010004
APA StyleDizon, C., Lysyk, T. J., Couloigner, I., & Cork, S. C. (2023). Ecology and Epidemiology of Lyme Disease in Western North America. Zoonotic Diseases, 3(1), 20-37. https://doi.org/10.3390/zoonoticdis3010004







