1. Introduction
Vascular grafting is a surgical technique or treatment to replace or bypass impaired sections of blood vessels with artificial or natural tubes. The grafts mostly have synthetic materials like polytetrafluoroethylene (PTFE) and polyethylene terephthalate (Dacron), which are found in large quantities and exhibit improved mechanical stability for vascular surgeries [
1,
2]. The grafts are used to restore normal circulation of blood in preventing aneurysm formation and reducing the chances of blockage or rupture of the arteries. They vary in size and shape, which depends on the surgical needs of a particular patient, the site of implantation, and the clinical scenario in question.
Smooth grafts are composed of homogenous structural properties, which reduce friction, hence enhance smooth (laminar) blood flow. This structural advantage is of special value in the large arteries (such as the aorta), where turbulence and flow upheavals are to be avoided [
3]. Corrugated grafts have textured surfaces that are formed by several ridges or folds. This corrugation increases the surface area of the graft, promoting better mechanical fixation and integration of the graft with the surrounding biological structures. Because of these qualities, corrugated grafts are appropriate when placed in the peripheral arteries, where the amount of flexibility with respect to mechanical stability is critical [
4,
5].
Some considerable distinctions between the biological and the mechanical interaction of the smooth and corrugated graft surfaces and blood flow with the vessel walls occur. Smooth-surface grafts normally provide a flowing path to the blood, hence reducing the risks of thromboembolism and hyperplasia of the intimal part. Their simplicity on the surface could, however, be a drawback as they could hinder attachment and proliferation of endothelial cells, which are crucial in sustained graft functionality [
6]. Bacterial adhesion and biofilm formation are significantly influenced by flow dynamics. According to earlier research, biofilm biomass is considerably lower in steady, laminar flows than in disturbed regimes [
7]. Thus, hemodynamic graft geometry optimization may indirectly affect infection susceptibility in addition to being crucial for lowering thrombogenic risk. On the other hand, a topographical advantage is offered by corrugated surfaces that are able to offer a better adhesion of endothelial cells, endothelialization, and a greater biological integration [
8].
A comprehensive review of the literature is available in both instances of synthetic and biological grafts. Further development should be performed in order to highlight the clinical importance, errors in smooth working, and corrugated designs to achieve better results concerning cardiovascular surgeries. A conceptual computational model that depicts the interaction between different materials was created.
2. Method
In this model, fluid–structure interactions, heat transfer, and thermal expansion are considered. The two linked portions, which constitute the vascular graft and diseased artery, are set in a fluid background with complex flow features, which regenerate via viscosity changes, time shear thinning. Structural bending is caused by heating, and then, subsequently, a time-dependent temperature profile is added to the inlet.
2.1. Model Design
To evaluate the biomechanical response of a vascular graft attached to a diseased artery, a coupled fluid–structure interaction (FSI) model was created in COMSOL Multiphysics
®. Hydrodynamic shear conditions have been demonstrated to influence bacterial adhesion and community development [
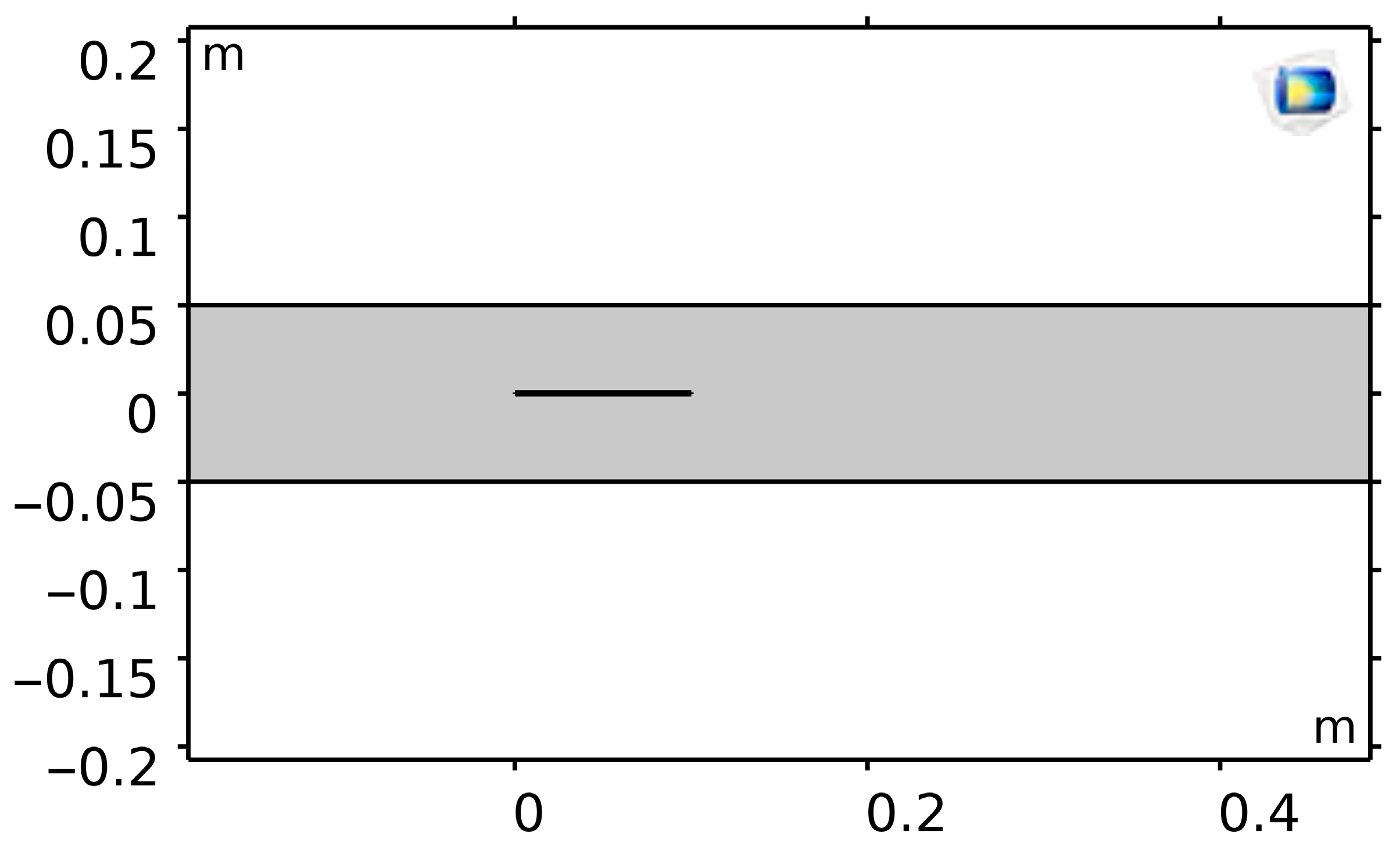
9]. For this reason, wall shear stress (WSS) and flow patterns were carefully evaluated in fluid dynamic simulations. Two bonded strips (10 mm × 0.5 cm each) made up the geometry (
Figure 1), with the lower portion signifying the vascular graft and the upper portion representing the diseased artery. A 70 cm × 10 cm rectangular fluid domain comprised this bi-material system.
In order to simulate vascular fixation, displacements were restricted at the upstream boundary. The junction region was heated with a 50 W evenly distributed heat source. When heated, the graft expanded more than the artery because of differences in their thermal expansion coefficients, which resulted in junctional bending. The graft–artery interaction and deformation patterns could be simulated thanks to this thermal–mechanical coupling.
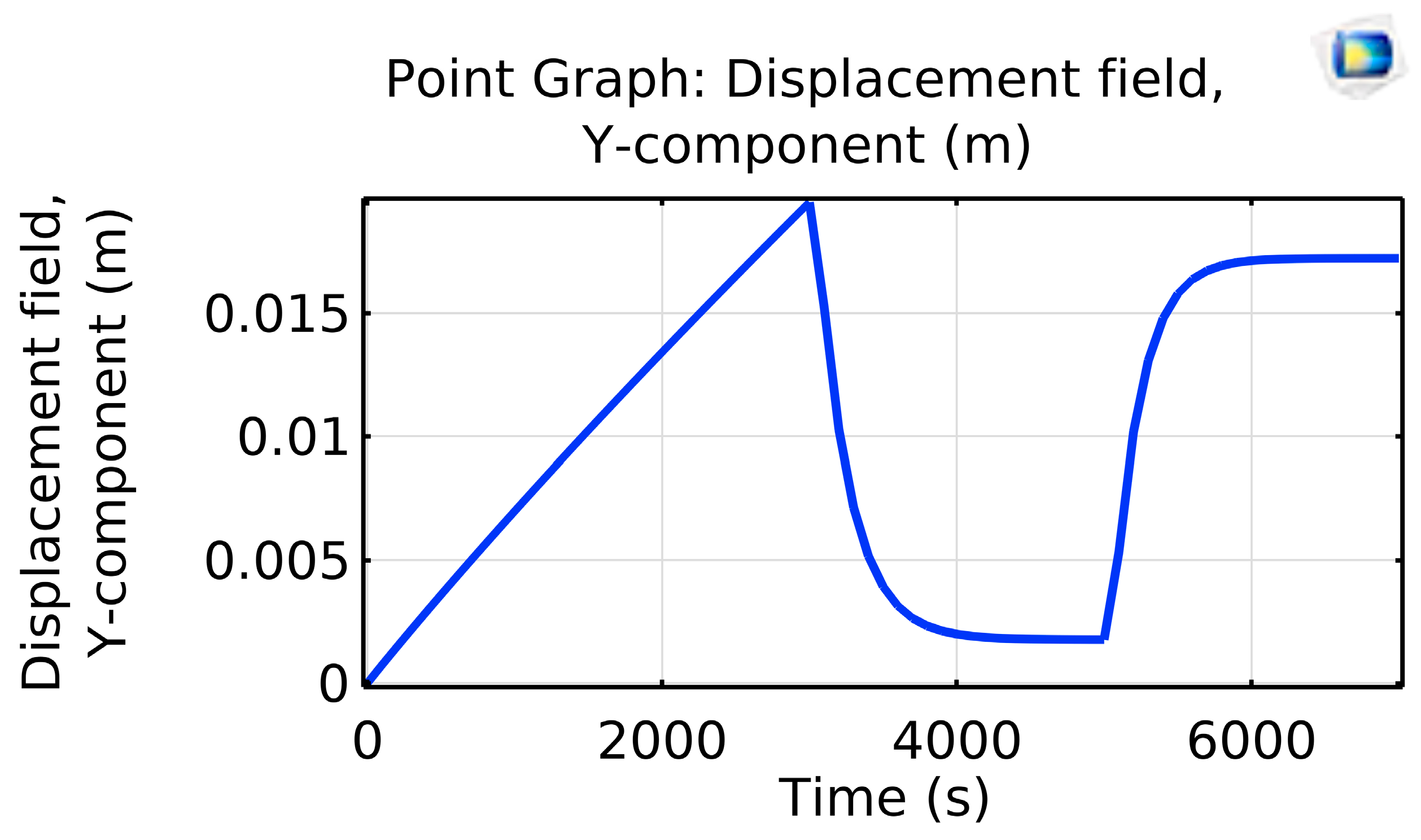
Velocity was set to zero at the inlet and ramped linearly to 0.2 m/s over 3000 s. Likewise, after 5000 s, the inlet temperature ramped up to 593.15 K from its initial setting of 293.15 K (room temperature) (
Figure 2).
2.2. Meshing and Boundary Conditions
At points with high stress gradients, especially at the graft–artery interface, a finite-element mesh was created and refined. Boundary conditions based on biology were enforced:
A pulsatile load is used to model internal arterial pressure.
External restrictions that imitate tissue anchoring.
Time-dependent inlet flow that captures pulsatile hemodynamics is defined.
This made it possible to replicate vascular conditions under coupled FSI in an accurate way.
2.3. Material Properties and Physics Interfaces
Different material properties were assigned to the graft and the arterial wall:
Elastic modulus.
Density.
Poisson’s ratio.
The artery graft composite’s deformation and stress distribution were calculated using the Solid Mechanics interface (study the mechanical behavior in COMSOL using the physiological conditions). Shear-dependent viscosity and thermal expansion were taken into consideration by coupling the physics interfaces of fluid flow and heat transfer.
3. Results and Discussion
Laminar flow through the graft, governed by the Navier–Stokes equations, ensures smooth, orderly blood movement that minimizes arterial shear stress and risk of thromboembolism [
10]. Solid-mechanics analyses using finite-element methods simulate the stresses and strains imposed by pulsatile blood pressure to verify material durability [
11]. Concurrently, heat transfer studies predict how blood-induced temperature changes affect graft thermal expansion and, in turn, flow dynamics and structural integrity [
12]. Multiphysics modeling couples these phenomena—fluid–structure interactions, which reveal how graft deformation alters blood flow [
13]; non-isothermal flow, which integrates thermal and fluid effects [
14]; and thermal expansion analysis assesses dimensional changes under varying temperatures to provide a comprehensive understanding of graft behavior in vivo [
15].
Figure 3 shows that finite-element modeling under 160 mmHg (λ_z = 1.9) predicts peak circumferential stress (~2 × 10
4 Pa) at the inner lumen (red) tapering to the outer wall (blue), pinpointing fatigue-prone regions in the graft design.
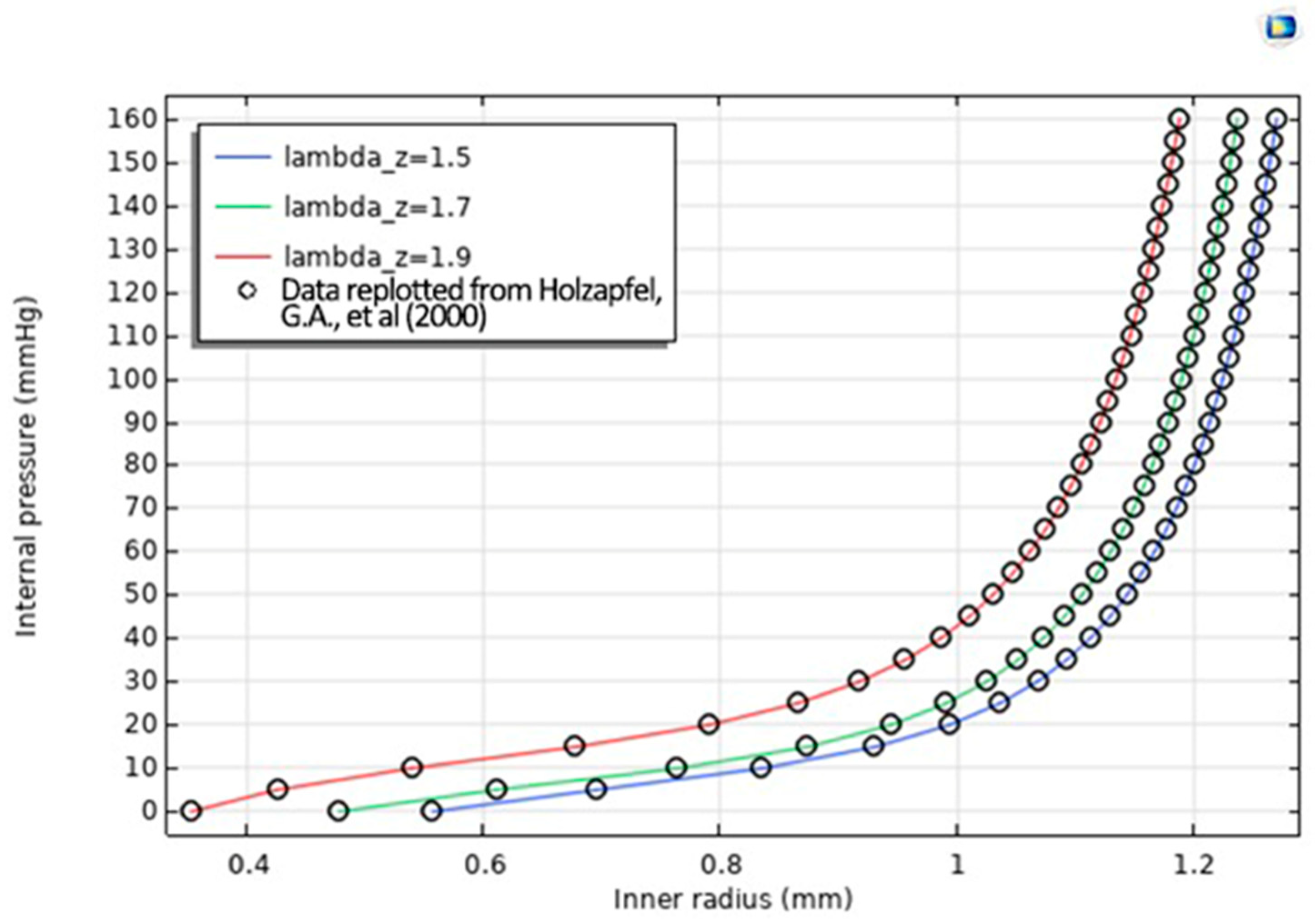
Figure 4 shows the Radius–Pressure Relationship Data replotted from Ref. [
16] in which Simulated inner-radius expansion vs. pressure for λ_z = 1.5, 1.7, 1.9 reproduces the graft’s J-shaped stiffening curve, with λ_z = 1.9 (red) closely matching experimental data (black circles) to validate the bi-material model.
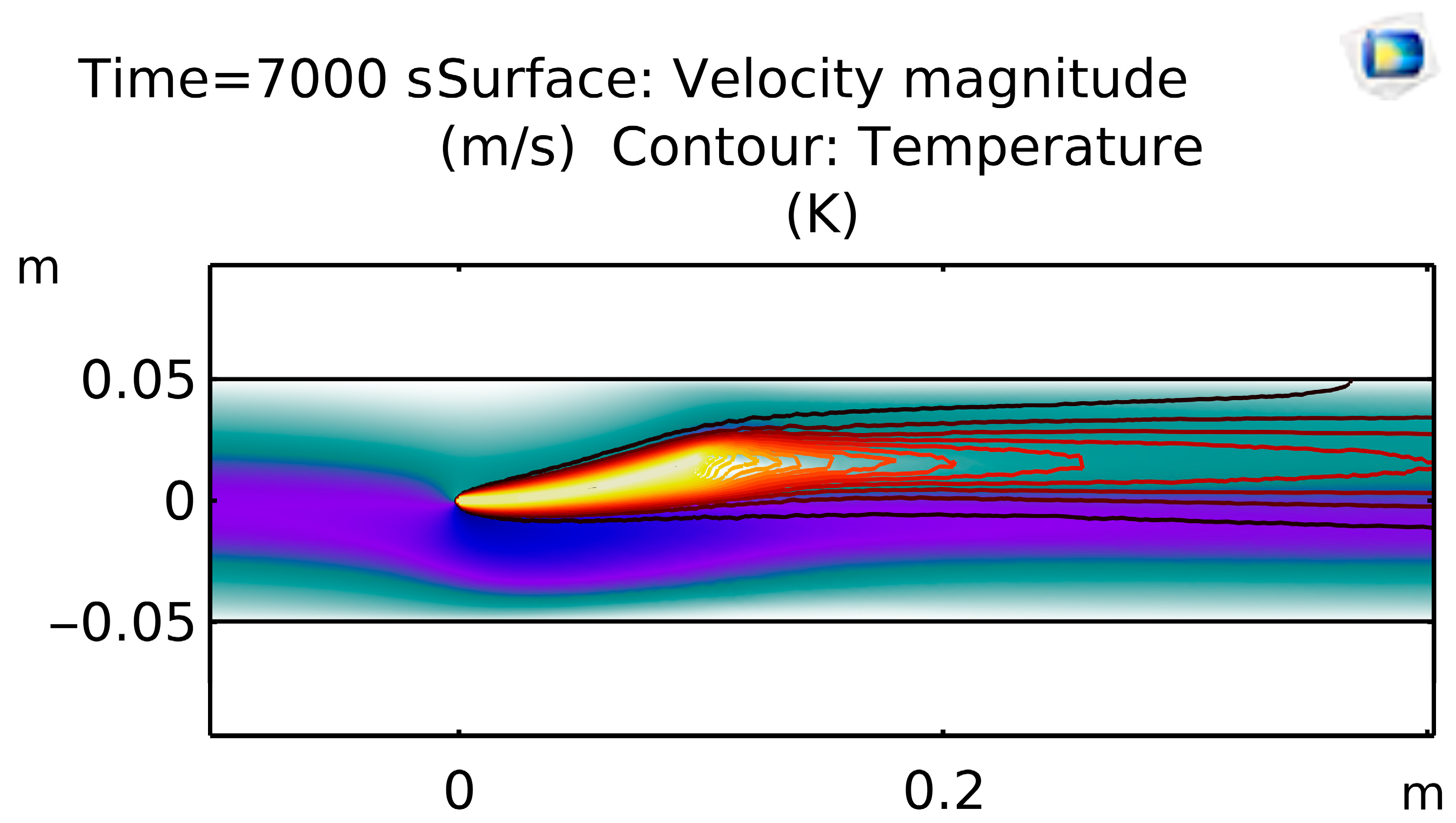
Figure 5 shows the flow velocity in which CFD coupling shows blood accelerating to ~0.4 m/s through the graft constriction at t = 7000 s, creating a localized thermal hotspot (~620 K) in the high-shear jet and a downstream recirculation zone.
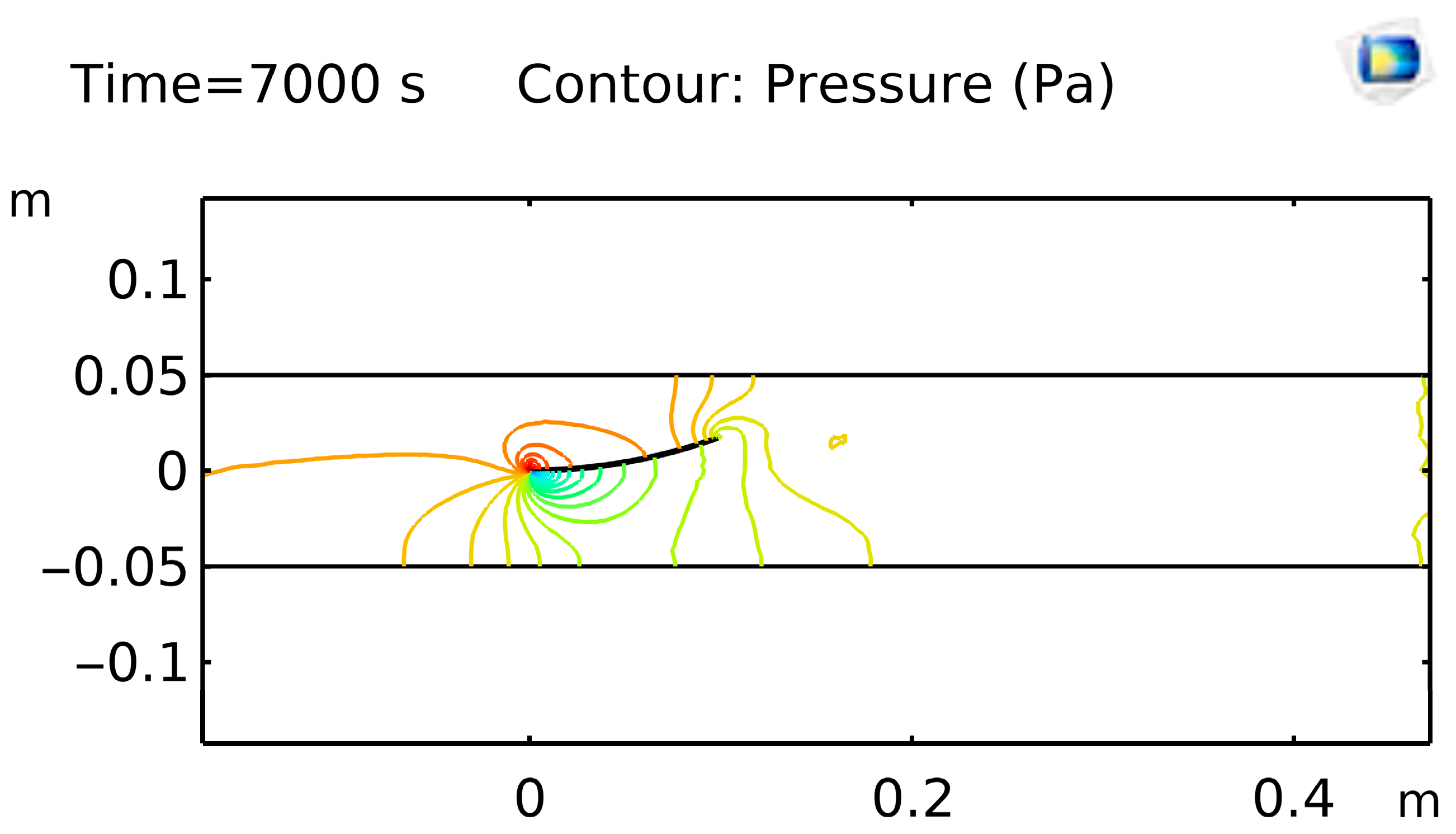
Figure 6 shows pressure mapping at t = 7000 s, revealing a sharp drop across the stenotic segment and a low-pressure recirculation bubble downstream, critical for predicting cavitation risk in graft prototypes.
Figure 7 shows Temperature Distribution with Streamlines—Non-isothermal FSI results overlay temperature (≈620 K peak) and velocity streamlines to highlight convective mixing in the post-stenotic jet and return to baseline (~595 K) regions within the graft.
The simulation solves the coupled FSI problem, providing detailed insights into deformation patterns, stress distribution, and flow dynamics. By comparing the performance of smooth and corrugated grafts, the model reveals critical differences in their mechanical and hemodynamic behavior. The optimization of graft design enhances arterial mechanics and improves clinical outcomes of corrugated grafts. Although direct antimicrobial effects were not examined in this study, the corrugated grafts showed smoother, laminar flow with more uniform WSS distribution and reduced risk of thrombosis. This suggests an indirect protective advantage.
4. Conclusions
Vascular prostheses (grafts) are widely used for hemodialysis blood access, trauma repair, aneurysm repair, and cardiovascular surgeries. General requirements for success in all these applications are similar: the graft must not thrombose and must not trigger hyperplasia. It must not lose mechanical integrity. Vascular grafts used in surgeries today are composed primarily of expanded Teflon (ePTFE) or Dacron fabric. Our work may indirectly reduce infection risks due to the enhanced compliance and flow stability seen in simulations, even though we did not experimentally assess antibacterial activity. These results will be confirmed in subsequent research using surface modification techniques and in vitro testing.
Author Contributions
Conceptualization, H.M., W.A., M.K. (Mishaal Khawar), R.S., N.M., M.K. (Manala Khalid) and K.T.; Methodology, H.M., W.A., M.K. (Mishaal Khawar), R.S., N.M., M.K. (Manala Khalid) and K.T.; Software, H.M., W.A. and M.K. (Mishaal Khawar); Validation, H.M. and W.A.; Formal analysis, H.M., M.K. (Mishaal Khawar), N.M. and K.T.; Investigation, M.K. (Mishaal Khawar), R.S., M.K. (Manala Khalid) and K.T.; Data curation, M.K. (Mishaal Khawar) and R.S.; writing—original draft preparation, M.K. (Mishaal Khawar), W.A., R.S., M.K. (Manala Khalid) and K.T.; writing—review and editing, H.M. and W.A.; Visualization, H.M. and W.A.; Supervision, H.M. and W.A.; Project administration, H.M. All authors have contributed equally to this work. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Zhen, L.; Quiroga, E.; Creason, S.A.; Chen, N.; Sapre, T.R.; Snyder, J.M.; Lindhartsen, S.L.; Fountaine, B.S.; Barbour, M.C.; Faisal, S.; et al. Synthetic vascular graft that heals and regenerates. Biomaterials 2025, 320, 123206. [Google Scholar] [CrossRef] [PubMed]
- Caradu, C.; Jolivet, B.; Pugue, M.; Cazanave, C.; Ducasse, E.; Berard, X. Reconstruction of primary and secondary aortic infections with an antimicrobial graft. J. Vasc. Surg. 2023, 77, 1226–1237. [Google Scholar] [CrossRef] [PubMed]
- Jarad, N.A.; Chami, A.; Weitz, J.I.; Didar, T.F. Advancements in surface modification strategies of vascular grafts to improve biocompatibility and tissue integration. Explor. BioMat-X 2024, 1, 241–265. [Google Scholar] [CrossRef]
- Robinson, A.; Herrera Duran, J.S.; Jiang, D.; Leung, J.; Laude, M.; Nkansah, A.; Guo, L.; Timmins, L.; Cosgriff-Hernandez, E. Design of a corrugated vascular graft with enhanced compliance and kink resistance. J. Biomed. Mater. Res. A 2025, 113, e37975. [Google Scholar] [CrossRef] [PubMed]
- Akbari, S.; Mohebbi-Kalhori, D.; Samimi, A. Effect of corrugated structure on the collapsing of the small-diameter vascular scaffolds. J. Biomater. Appl. 2020, 34, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhou, M.; Li, M.; Li, J.; Zhang, S.; Wang, J. Tailored endothelialization enabled by engineered endothelial cell vesicles accelerates remodeling of small-diameter vascular grafts. Biomater. Transl. 2024, 41, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Tsagkari, E.; Connelly, S.; Liu, Z.; McBride, A.; Sloan, W.T. The role of shear dynamics in biofilm formation. npj Biofilms Microbiomes 2022, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Radke, D.; Jia, W.; Sharma, D.; Fena, K.; Wang, G.; Goldman, J.; Zhao, F. Tissue Engineering at the Blood-Contacting Surface: A Review of Challenges and Strategies in Vascular Graft Development. Adv. Healthc. Mater. 2018, 7, 1701461. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Bawazir, M.; Dhall, A.; Kim, H.-E.; He, L.; Heo, J.; Hwang, G. Implication of surface properties, bacterial motility, and hydrodynamic conditions on bacterial surface sensing and their initial adhesion. Front. Bioeng. Biotechnol. 2021, 9, 643722. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, G.K. An Introduction to Fluid Dynamics; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Zienkiewicz, O.C.; Taylor, R.L. The Finite Element Method for Solid and Structural Mechanics; Butterworth-Heinemann: Oxford, UK, 2005. [Google Scholar]
- Incropera, F.P.; DeWitt, D.P. Fundamentals of Heat and Mass Transfer; Wiley: New York, NY, USA, 2002. [Google Scholar]
- Dowell, E.H. Modeling of Fluid-Structure Interactions; World Scientific: Singapore, 2004. [Google Scholar]
- Kays, W.M.; Crawford, M.E. Convective Heat and Mass Transfer; McGraw-Hill: New York, NY, USA, 1993. [Google Scholar]
- Boley, B.A.; Weiner, J.H. Theory of Thermal Stresses; Dover: New York, NY, USA, 1997. [Google Scholar]
- Holzapfel, G.A.; Gasser, T.C.; Ogden, R.W. A new constitutive framework for arterial wall mechanics and a comparative study of material models. J. Elast. 2000, 61, 1–48. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).