Application of Activated Carbon/Alginate Composite Beads for the Removal of 2-Methylisoborneol from Aqueous Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of the Adsorbent
2.2.2. Characterization of the Adsorbent
2.2.3. Adsorption Experiments
2.2.4. Adsorption Kinetic Models
- Pseudo-first order kinetic model
- Pseudo-second order kinetic model
2.2.5. Adsorption Isotherm Models
- Langmuir isotherm model
- Freundlich isotherm model
- Temkin model
2.2.6. Regeneration Experiments
2.2.7. Analysis of 2-MIB
3. Results and Discussion
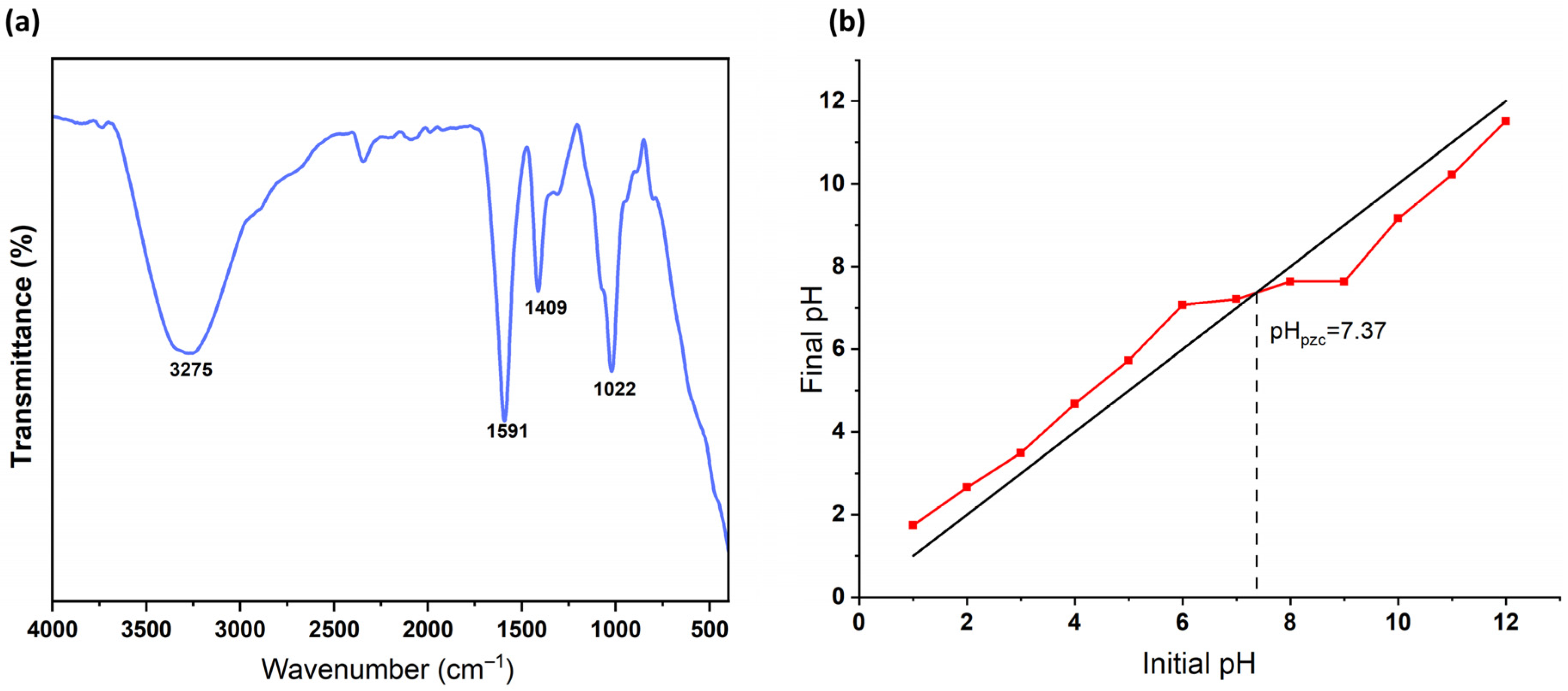
3.1. Characterization of the Adsorbent
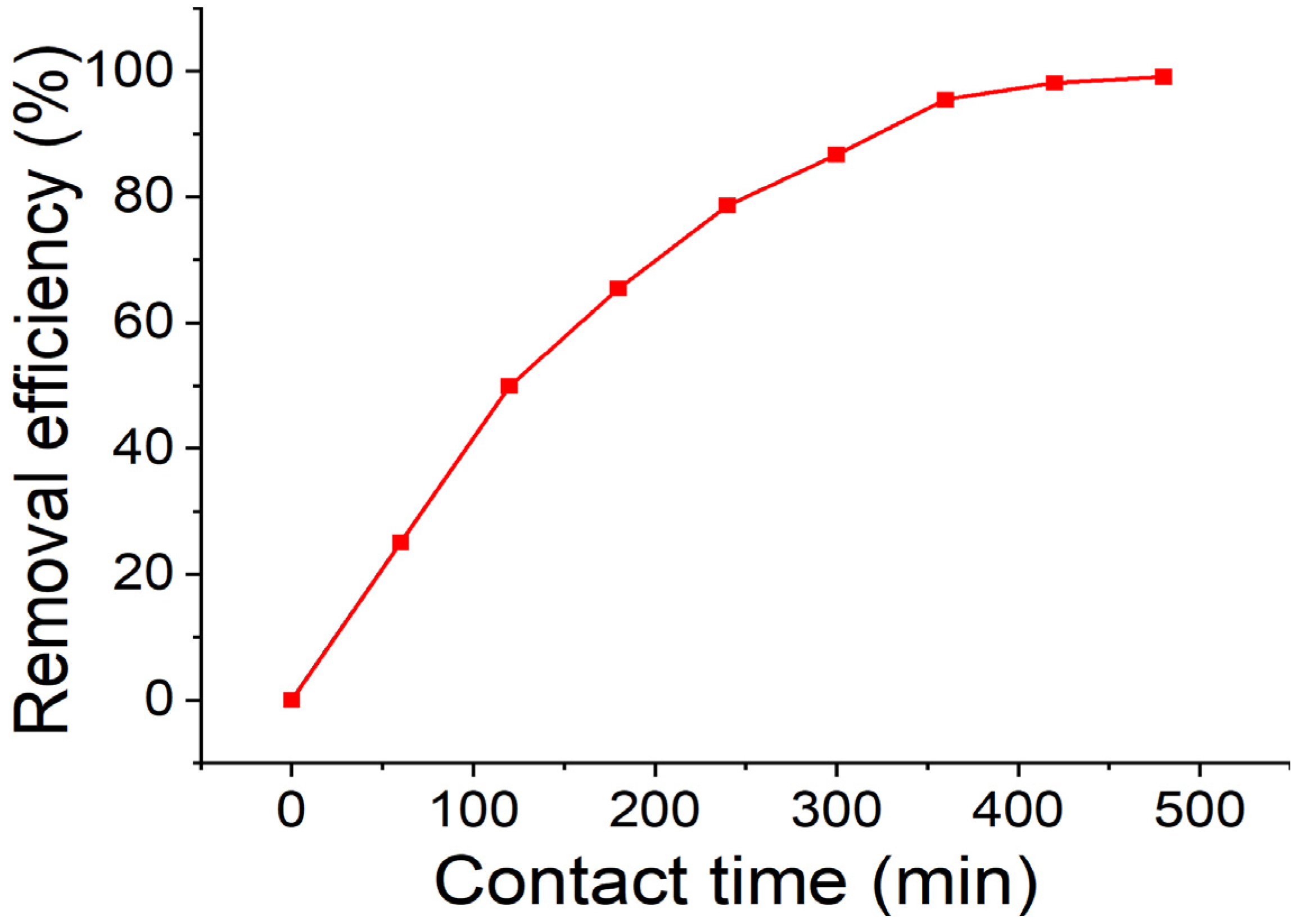
3.2. Effect of Contact Time
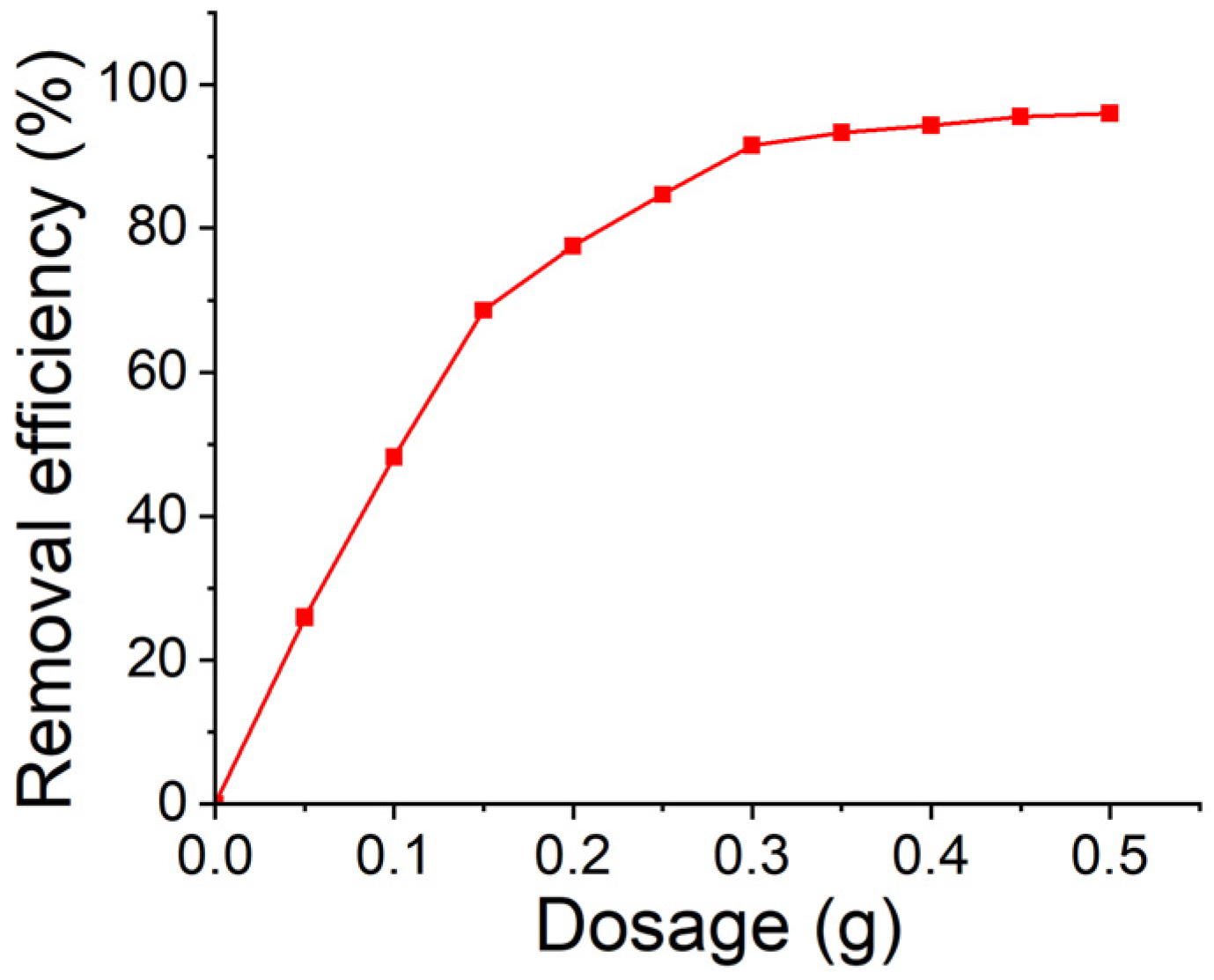
3.3. Effect of Adsorbent Dosage
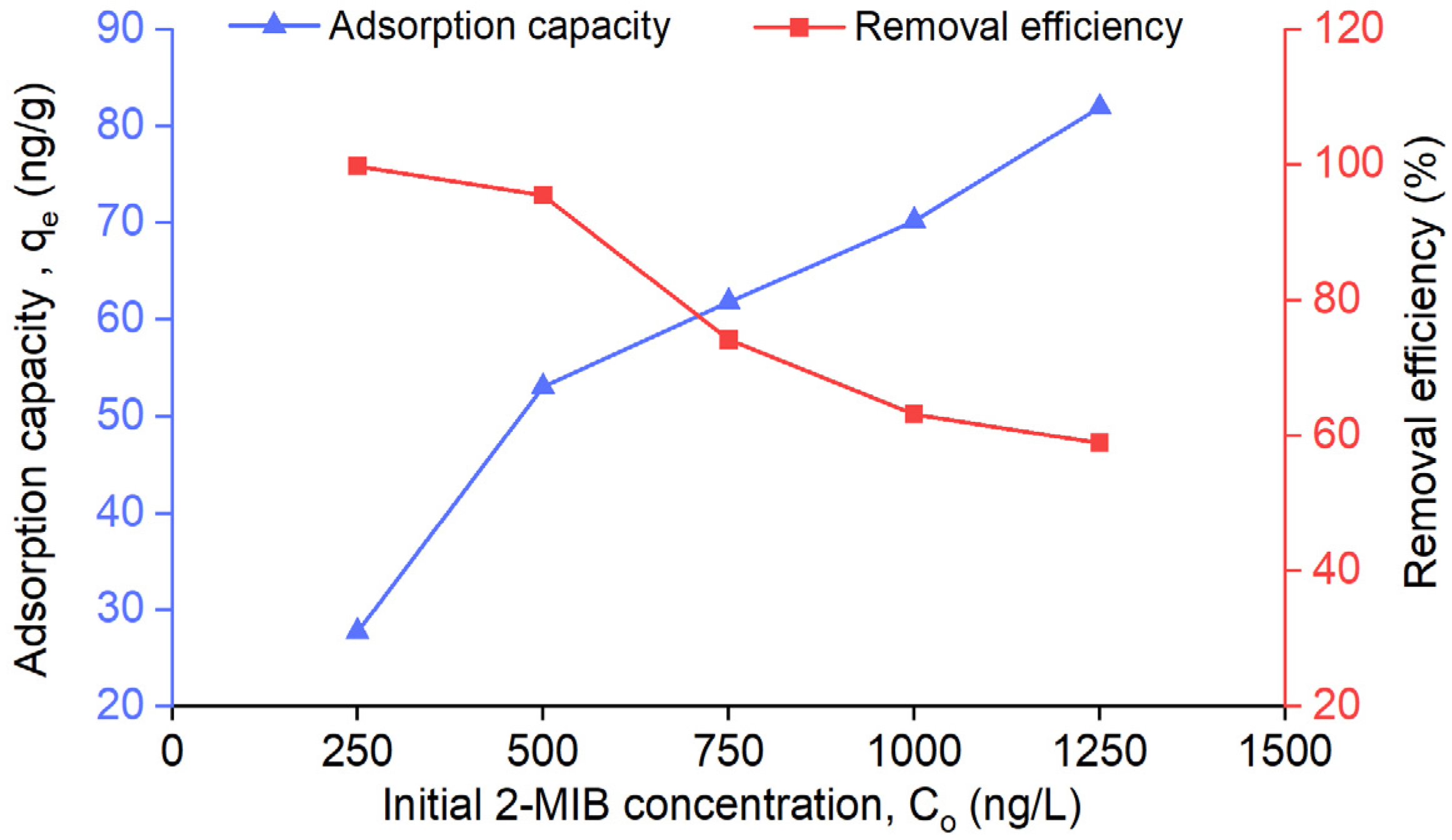
3.4. Effect of Initial Concentration of 2-MIB
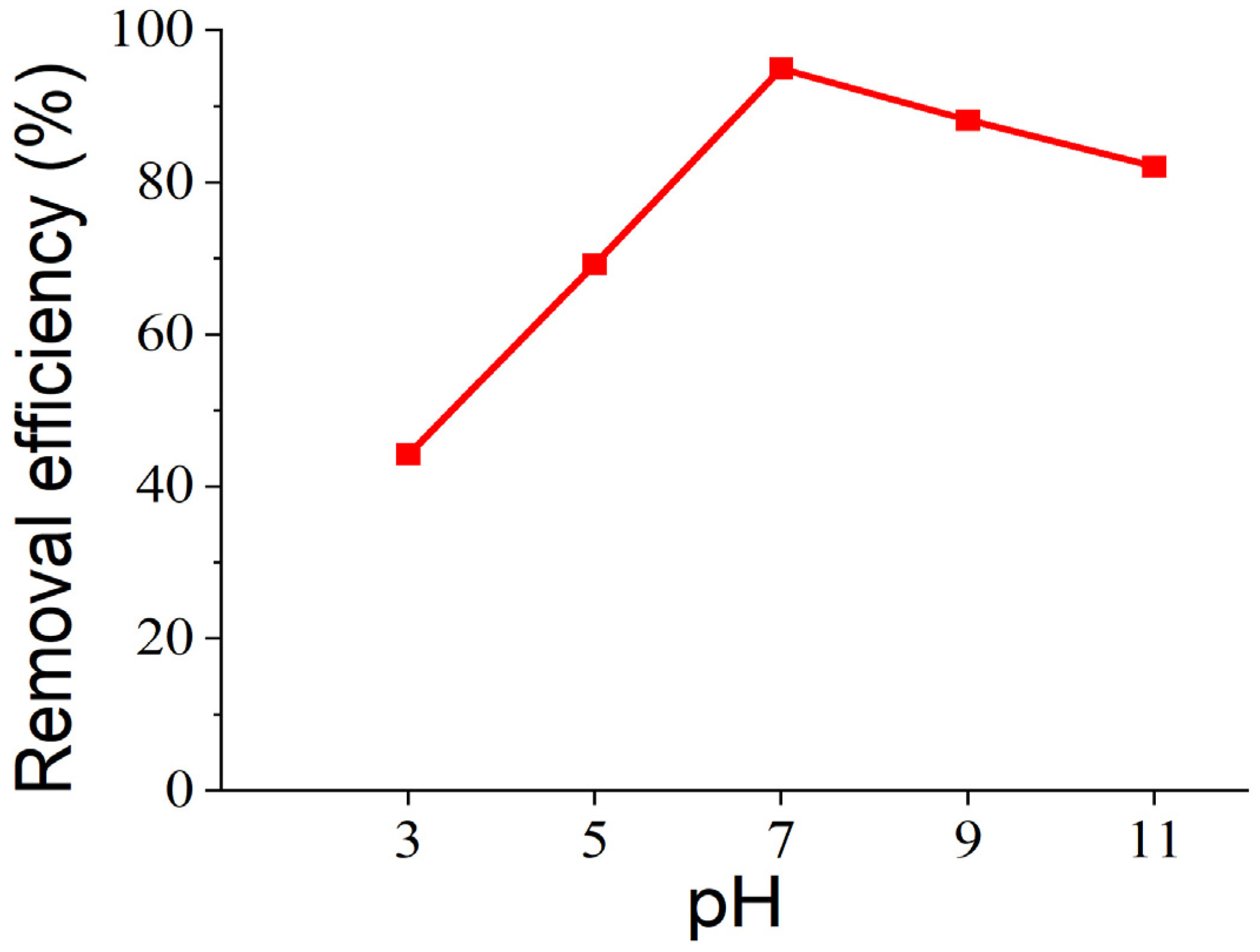
3.5. Effect of Initial pH
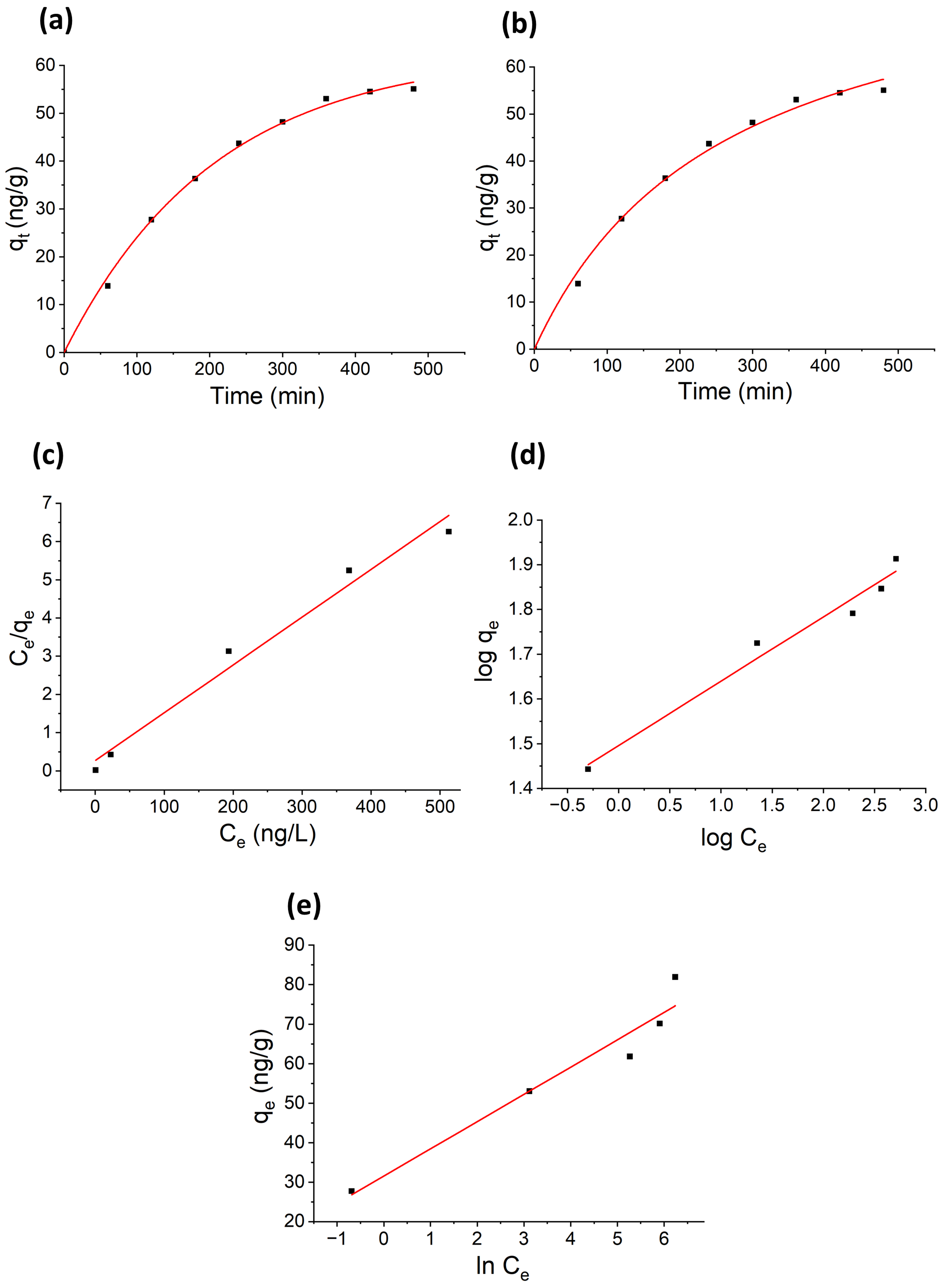
3.6. Adsorption Kinetic Study
3.7. Adsorption Isotherm Study
3.8. Regeneration Studies
4. Limitations and Future Work
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2-MIB | 2-methylisoborneol |
| AC/alginate | Activated carbon/alginate |
| SEM | Scanning electron microscopy |
| FTIR | Fourier transform infrared spectroscopy |
| PAC | Powdered activated carbon |
| GAC | Granular activated carbon |
| GCMS | Gas chromatography/mass spectrometry |
| SPME | Solid phase microextraction |
| PFO | Pseudo first order |
| PSO | Pseudo second order |
References
- Chapra, S.C.; Boehlert, B.; Fant, C.; Bierman, V.J.; Henderson, J.; Mills, D.; Mas, D.M.L.; Rennels, L.; Jantarasami, L.; Martinich, J.; et al. Climate Change Impacts on Harmful Algal Blooms in U.S. Freshwaters: A Screening-Level Assessment. Environ. Sci. Technol. 2017, 51, 8933–8943. [Google Scholar] [CrossRef]
- Paerl, H.W.; Paul, V.J. Climate Change: Links to Global Expansion of Harmful Cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-S.; Yuan, D.-X.; Weng, T.-P. Pilot Study of Drinking Water Treatment with GAC, O3/BAC and Membrane Processes in Kinmen Island, Taiwan. Desalination 2010, 263, 271–278. [Google Scholar] [CrossRef]
- Sugiura, N.; Iwami, N.; Inamori, Y.; Nishimura, O.; Sudo, R. Significance of Attached Cyanobacteria Relevant to the Occurrence of Musty Odor in Lake Kasumigaura. Water Res. 1998, 32, 3549–3554. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Elbestawy, A.R.; Ellakany, H.F.; Abaza, S.S.; Geneedy, A.M.; Salem, H.M.; Taha, A.E.; Swelum, A.A.; Omer, F.A. Undesirable Odour Substances (Geosmin and 2-Methylisoborneol) in Water Environment: Sources, Impacts and Removal Strategies. Mar. Pollut. Bull. 2022, 178, 113579. [Google Scholar] [CrossRef]
- Gerber, N.N. Volatile Substances from Actinomycetes: Their Role in the Odor Pollution of Water. CRC Crit. Rev. Microbiol. 1979, 7, 191–214. [Google Scholar] [CrossRef]
- Persson, P.-E. Odorous Algal Cultures in Culture Collections. Water Sci. Technol. 1988, 20, 211–213. [Google Scholar] [CrossRef]
- Young, W.; Horth, H.; Crane, R.; Ogden, T.; Arnott, M. Taste and Odour Threshold Concentrations of Potential Potable Water Contaminants. Water Res. 1996, 30, 331–340. [Google Scholar] [CrossRef]
- Bruce, D.; Westerhoff, P.; Brawley-Chesworth, A. Removal of 2-Methylisoborneol and Geosmin in Surface Water Treatment Plants in Arizona. J. Water Supply Res. Technol.—AQUA 2002, 51, 183–198. [Google Scholar] [CrossRef]
- Hafuka, A.; Nagasato, T.; Yamamura, H. Application of Graphene Oxide for Adsorption Removal of Geosmin and 2-Methylisoborneol in the Presence of Natural Organic Matter. Int. J. Environ. Res. Public Health 2019, 16, 1907. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhou, L.; Gao, J.; Zhu, J.; Zhou, J. Process Optimization and Mechanism Revealing of KMnO4 Pre-Oxidation Coupled Powdered Activated Carbon Adsorption for 2-MIB Removal. J. Water Process Eng. 2023, 53, 103705. [Google Scholar] [CrossRef]
- Zamyadi, A.; Henderson, R.; Stuetz, R.; Hofmann, R.; Ho, L.; Newcombe, G. Fate of Geosmin and 2-Methylisoborneol in Full-Scale Water Treatment Plants. Water Res. 2015, 83, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Ho, L.; Hoefel, D.; Bock, F.; Saint, C.P.; Newcombe, G. Biodegradation Rates of 2-Methylisoborneol (MIB) and Geosmin through Sand Filters and in Bioreactors. Chemosphere 2007, 66, 2210–2218. [Google Scholar] [CrossRef]
- Cook, D.; Newcombe, G.; Sztajnbok, P. The Application of Powdered Activated Carbon for MIB and Geosmin Removal: Predicting PAC Doses in Four Raw Waters. Water Res. 2001, 35, 1325–1333. [Google Scholar] [CrossRef]
- Graham, M.; Summers, R.; Simpson, M.; MacLeod, B. Modeling Equilibrium Adsorption of 2-Methylisoborneol and Geosmin in Natural Waters. Water Res. 2000, 34, 2291–2300. [Google Scholar] [CrossRef]
- Yuan, J.; Mortazavian, S.; Crowe, G.; Flick, R.; Passeport, E.; Hofmann, R. Evaluating the Relative Adsorption and Biodegradation of 2-Methylisoborneol and Geosmin across Granular Activated Carbon Filter-Adsorbers. Water Res. 2022, 215, 118239. [Google Scholar] [CrossRef]
- Tosa, K.; Nakamura, G.; Miyabayashi, K.; Ishisaki, H.; Takahashi, Y. Adsorption of Geosmin and 2-MIB to Porous Coordination Polymer MIL-53 (Al). J. Water Environ. Technol. 2022, 20, 212–218. [Google Scholar] [CrossRef]
- Ellis, J.; Korth, W. Removal of Geosmin and Methylisoborneol from Drinking Water by Adsorption on Ultrastable Zeolite-Y. Water Res. 1993, 27, 535–539. [Google Scholar] [CrossRef]
- Asghar, A.; Khan, Z.; Maqbool, N.; Qazi, I.A.; Awan, M.A. Comparison of Adsorption Capability of Activated Carbon and Metal Doped TiO2 for Geosmin and 2-MIB Removal from Water. J. Nanomater. 2015, 2015, 479103. [Google Scholar] [CrossRef]
- Xue, Q.; Chen, R.; Sakharkar, M.K.; Utsumi, M.; Li, M.; Shimizu, K.; Zhang, Z.; Sugiura, N. Development of a Ceramic Adsorbent for the Removal of 2-Methylisoborneol from Aqueous Solution. Desalination 2011, 281, 293–297. [Google Scholar] [CrossRef]
- Liu, L.; Li, L.; Zuo, Y.; Huang, Y.; Song, L. Adsorption of 2-Methylisoborneol and Geosmin by a Low-Cost Hybrid Adsorbent Synthesized from Fly Ash and Bentonite. J. Water Supply Res. Technol.—AQUA 2011, 60, 478–485. [Google Scholar] [CrossRef]
- Matsui, Y.; Nakao, S.; Sakamoto, A.; Taniguchi, T.; Pan, L.; Matsushita, T.; Shirasaki, N. Adsorption Capacities of Activated Carbons for Geosmin and 2-Methylisoborneol Vary with Activated Carbon Particle Size: Effects of Adsorbent and Adsorbate Characteristics. Water Res. 2015, 85, 95–102. [Google Scholar] [CrossRef]
- Zoschke, K.; Engel, C.; Börnick, H.; Worch, E. Adsorption of Geosmin and 2-Methylisoborneol onto Powdered Activated Carbon at Non-Equilibrium Conditions: Influence of NOM and Process Modelling. Water Res. 2011, 45, 4544–4550. [Google Scholar] [CrossRef] [PubMed]
- Chae, A.N.; Shin, J.W.; Cho, K.W.; Lee, B.C.; Song, K.G. Removal of Geosmin and 2-Methylisoborneol in Drinking Water by Powdered Activated Carbon. KSCE J. Civ. Environ. Eng. Res. 2017, 37, 475–483. [Google Scholar] [CrossRef][Green Version]
- Park, S.-M.; Heo, T.-Y.; Park, N.-B.; Na, K.-J.; Jun, H.-B.; Jung, J.-Y. Application of Air Stripping to Powdered Activated Carbon Adsorption of Geosmin and 2-Methylisoborneol. J. Water Supply Res. Technol.—AQUA 2010, 59, 492–500. [Google Scholar] [CrossRef]
- Bong, T.; Kang, J.-K.; Yargeau, V.; Nam, H.-L.; Lee, S.-H.; Choi, J.-W.; Kim, S.-B.; Park, J.-A. Geosmin and 2-Methylisoborneol Adsorption Using Different Carbon Materials: Isotherm, Kinetic, Multiple Linear Regression, and Deep Neural Network Modeling Using a Real Drinking Water Source. J. Clean. Prod. 2021, 314, 127967. [Google Scholar] [CrossRef]
- Watanabe, T.; Amano, Y.; Machida, M. The Adsorption Mechanism and Rapid Screening of Activated Carbon for 2-Methylisoborneol Adsorption. TANSO 2013, 2013, 124–134. [Google Scholar] [CrossRef][Green Version]
- Yuan, J.; Huang, Y.; Nie, Z.; Hofmann, R. The Effect of Water Temperature on the Removal of 2-Methylisoborneol and Geosmin by Preloaded Granular Activated Carbon. Water Res. 2020, 183, 116065. [Google Scholar] [CrossRef]
- Pan, L.; Matsui, Y.; Matsushita, T.; Shirasaki, N. Superiority of Wet-Milled over Dry-Milled Superfine Powdered Activated Carbon for Adsorptive 2-Methylisoborneol Removal. Water Res. 2016, 102, 516–523. [Google Scholar] [CrossRef]
- Xin, X.; Wang, M.; Ge, X.; Zhao, Q.; Sun, S.; Jia, R. Highly Efficient Removal of Geosmin and 2-Methylisoborneol by Carboxylated Multi-Walled Carbon Nanotubes. Monatshefte Chem.-Chem. Mon. 2014, 145, 747–754. [Google Scholar] [CrossRef]
- Yuan, J.; Hofmann, R. Adsorption and Biodegradation of 2-Methylisoborneol and Geosmin in Drinking Water Granular Activated Carbon Filters: A Review and Meta-Analysis. J. Hazard. Mater. 2022, 440, 129838. [Google Scholar] [CrossRef]
- Newcombe, G.; Morrison, J.; Hepplewhite, C.; Knappe, D. Simultaneous Adsorption of MIB and NOM onto Activated Carbon: II. Competitive Effects. Carbon 2002, 40, 2147–2156. [Google Scholar] [CrossRef]
- Matsui, Y.; Nakao, S.; Taniguchi, T.; Matsushita, T. Geosmin and 2-Methylisoborneol Removal Using Superfine Powdered Activated Carbon: Shell Adsorption and Branched-Pore Kinetic Model Analysis and Optimal Particle Size. Water Res. 2013, 47, 2873–2880. [Google Scholar] [CrossRef]
- Wang, F.; Li, X.; Liu, T.; Li, X.; Cui, Y.; Xu, L.; Huo, S.; Zou, B.; Qian, J.; Ma, A. Removal of Taste and Odor Compounds from Water: Methods, Mechanism and Prospects. Catalysts 2023, 13, 1356. [Google Scholar] [CrossRef]
- Kim, K.T.; Park, Y.-G. Geosmin and 2-MIB Removal by Full-Scale Drinking Water Treatment Processes in the Republic of Korea. Water 2021, 13, 628. [Google Scholar] [CrossRef]
- Andreadakis, A.D.; Mamais, D.; Gavalakis, E.A.; Noutsopoulos, C.; Kouris, N.; Nikitopoulos, G. Removal of Taste and Odour from Potable Water by Ozone and Powdered Activated Carbon (PAC). Int. J. Environ. Waste Manag. 2010, 5, 392–409. [Google Scholar] [CrossRef]
- Pawar, R.R.; Ingole, P.G.; Lee, S.-M. Use of Activated Bentonite-Alginate Composite Beads for Efficient Removal of Toxic Cu2+ and Pb2+ Ions from Aquatic Environment. Int. J. Biol. Macromol. 2020, 164, 3145–3154. [Google Scholar] [CrossRef] [PubMed]
- Benhouria, A.; Islam, M.A.; Zaghouane-Boudiaf, H.; Boutahala, M.; Hameed, B. Calcium Alginate–Bentonite–Activated Carbon Composite Beads as Highly Effective Adsorbent for Methylene Blue. Chem. Eng. J. 2015, 270, 621–630. [Google Scholar] [CrossRef]
- Steinbüchel, A.; Rhee, S.K. (Eds.) Polysaccharides and Polyamides in the Food Industry: Properties, Production, and Patents; WILEY-VCH: Weinheim, Germany, 2005; pp. 1–30. ISBN 978-3-527-31345-7. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and Biomedical Applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Chitosan-Based Biosorbents: Modification and Application for Biosorption of Heavy Metals and Radionuclides. Bioresour. Technol. 2014, 160, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Sutirman, Z.A.; Sanagi, M.M.; Aini, W.I.W. Alginate-Based Adsorbents for Removal of Metal Ions and Radionuclides from Aqueous Solutions: A Review. Int. J. Biol. Macromol. 2021, 174, 216–228. [Google Scholar] [CrossRef]
- Torres, M.R.; Sousa, A.P.; Silva Filho, E.A.; Melo, D.F.; Feitosa, J.P.; de Paula, R.C.; Lima, M.G. Extraction and Physicochemical Characterization of Sargassum Vulgare Alginate from Brazil. Carbohydr. Res. 2007, 342, 2067–2074. [Google Scholar] [CrossRef]
- Faidi, A.; Lassoued, M.A.; Becheikh, M.E.H.; Touati, M.; Stumbé, J.-F.; Farhat, F. Application of Sodium Alginate Extracted from a Tunisian Brown Algae Padina Pavonica for Essential Oil Encapsulation: Microspheres Preparation, Characterization and in Vitro Release Study. Int. J. Biol. Macromol. 2019, 136, 386–394. [Google Scholar] [CrossRef]
- Hassan, A.; Abdel-Mohsen, A.; Fouda, M.M. Comparative Study of Calcium Alginate, Activated Carbon, and Their Composite Beads on Methylene Blue Adsorption. Carbohydr. Polym. 2014, 102, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Bée, A.; Talbot, D.; Abramson, S.; Dupuis, V. Magnetic Alginate Beads for Pb (II) Ions Removal from Wastewater. J. Colloid Interface Sci. 2011, 362, 486–492. [Google Scholar] [CrossRef]
- Fiol, N.; Poch, J.; Villaescusa, I. Chromium (VI) Uptake by Grape Stalks Wastes Encapsulated in Calcium Alginate Beads: Equilibrium and Kinetics Studies. Chem. Speciat. Bioavailab. 2004, 16, 25–33. [Google Scholar] [CrossRef]
- Vincent, T.; Parodi, A.; Guibal, E. Pt Recovery Using Cyphos IL-101 Immobilized in Biopolymer Capsules. Sep. Purif. Technol. 2008, 62, 470–479. [Google Scholar] [CrossRef]
- Hassan, A.; Abdel-Mohsen, A.; Elhadidy, H. Adsorption of Arsenic by Activated Carbon, Calcium Alginate and Their Composite Beads. Int. J. Biol. Macromol. 2014, 68, 125–130. [Google Scholar] [CrossRef]
- Davranche, M.; Lacour, S.; Bordas, F.; Bollinger, J.-C. An Easy Determination of the Surface Chemical Properties of Simple and Natural Solids. J. Chem. Educ. 2003, 80, 76. [Google Scholar] [CrossRef]
- Senavirathna, M.D.H.J.; Jayasekara, M.A.D.D. Temporal Variation of 2-MIB and Geosmin Production by Pseudanabaena Galeata and Phormidium Ambiguum Exposed to High-intensity Light. Water Environ. Res. 2023, 95, e10834. [Google Scholar] [CrossRef]
- Ahmad, A.; Loh, M.; Aziz, J. Preparation and Characterization of Activated Carbon from Oil Palm Wood and Its Evaluation on Methylene Blue Adsorption. Dye. Pigment. 2007, 75, 263–272. [Google Scholar] [CrossRef]
- Saarai, A.; Kasparkova, V.; Sedlacek, T.; Sáha, P. On the Development and Characterisation of Crosslinked Sodium Alginate/Gelatine Hydrogels. J. Mech. Behav. Biomed. Mater. 2013, 18, 152–166. [Google Scholar] [CrossRef]
- Larosa, C.; Salerno, M.; de Lima, J.S.; Meri, R.M.; da Silva, M.F.; de Carvalho, L.B.; Converti, A. Characterisation of Bare and Tannase-Loaded Calcium Alginate Beads by Microscopic, Thermogravimetric, FTIR and XRD Analyses. Int. J. Biol. Macromol. 2018, 115, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Mustapha, S.; Tijani, J.O.; Ndamitso, M.; Abdulkareem, A.S.; Shuaib, D.T.; Mohammed, A.K. A Critical Review on Geosmin and 2-Methylisoborneol in Water: Sources, Effects, Detection, and Removal Techniques. Env. Monit Assess 2021, 193, 204. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Yu, J.; Wang, Q.; Wang, C.; Du, Y.; Liu, Z.; Zhang, L.; Liu, Z.; Jing, C.; Tang, J. Enhancing the Adsorption Performance of 2-Methylisoborneol by Activated Carbon by Promoting Hydrophobic Effects. ACS EST Water 2022, 2, 1789–1798. [Google Scholar] [CrossRef]
- Thiel, P.; Cullum, P. Evaluating the Performance of Different Powdered Activated Carbons (PAC) for Taste and Odour Reduction. In Proceeding of the 32nd Annual Water Industry Operations Workshop, Rockhampton, QLD, Australia, 17–19 April 2007. [Google Scholar]
- Newcombe, G.; Drikas, M.; Hayes, R. Influence of Characterised Natural Organic Material on Activated Carbon Adsorption: II. Effect on Pore Volume Distribution and Adsorption of 2-Methylisoborneol. Water Res. 1997, 31, 1065–1073. [Google Scholar] [CrossRef]
- Yu, J.; Yang, M.; Lin, T.-F.; Guo, Z.; Zhang, Y.; Gu, J.; Zhang, S. Effects of Surface Characteristics of Activated Carbon on the Adsorption of 2-Methylisobornel (MIB) and Geosmin from Natural Water. Sep. Purif. Technol. 2007, 56, 363–370. [Google Scholar] [CrossRef]


| Kinetic Studies | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pseudo First-Order | Pseudo Second Order | |||||||||
| qeq calc. (ng g−1) | k1 × 10−3 (min−1) | R2 | AIC | BIC | qeq calc. (ng g−1) | k2 × 10−5 (g ng−1 min−1) | R2 | AIC | BIC | |
| 62.64 | 4.84 | 0.99 | 9.73 | 5.52 | 88.79 | 4.29 | 0.99 | 16.81 | 12.60 | |
| Isotherm Studies | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Langmuir Model | Freundlich Model | Temkin Model | ||||||||
| qmax (ng g−1) | KL (L ng−1) | R2 | Kf | n | R2 | KT (L ng−1) | n | R2 | ||
| 79.93 | 0.046 | 0.97 | 31.31 | 6.95 | 0.96 | 95.88 | 6.9 | 0.92 | ||
| Adsorbent | Initial 2-MIB Concentration | Dose | Contact Time | Removal Efficiency or qm | Ref. |
|---|---|---|---|---|---|
| Ceramic adsorbent | 200 ng/L | 200 ng/L | 600 min | >80% | [20] |
| Fly ash-bentonite adsorbent | 42–234 ng/L | 15 mg/L | 60 min | 59.9% | [21] |
| PAC (coconut shell) | 100 ng/L | 32.14 mg/L | 20 min | >80% | [26] |
| PAC (coconut shell) | 100 ng/L | 25 mg/L | 3–5 days | 85–95% | [14] |
| PAC (coconut shell) PAC (wood based) PAC (coal based) | N/A | 15 mg/L | 30 min | 48% | [57] |
| 62% | |||||
| 79% | |||||
| PAC (coal based) | 327 ng/L | 13 mg/L | 5 days | 95–99% | [58] |
| 880 ng/L | 96–99% | ||||
| PAC (coal based) PAC (wood based) | 100 ng/L | 2–30 mg/L | 3 days | 10–20 ng/mg 18 ng/mg | [59] |
| AC/alginate composite | 500 ng/L | 0.45 g | 360 min | 79.94 ng/g | Current study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balasooriya, I.L.; Senavirathna, M.D.H.J.; Wang, W. Application of Activated Carbon/Alginate Composite Beads for the Removal of 2-Methylisoborneol from Aqueous Solution. AppliedChem 2025, 5, 32. https://doi.org/10.3390/appliedchem5040032
Balasooriya IL, Senavirathna MDHJ, Wang W. Application of Activated Carbon/Alginate Composite Beads for the Removal of 2-Methylisoborneol from Aqueous Solution. AppliedChem. 2025; 5(4):32. https://doi.org/10.3390/appliedchem5040032
Chicago/Turabian StyleBalasooriya, Iresha Lakmali, Mudalige Don Hiranya Jayasanka Senavirathna, and Weiqian Wang. 2025. "Application of Activated Carbon/Alginate Composite Beads for the Removal of 2-Methylisoborneol from Aqueous Solution" AppliedChem 5, no. 4: 32. https://doi.org/10.3390/appliedchem5040032
APA StyleBalasooriya, I. L., Senavirathna, M. D. H. J., & Wang, W. (2025). Application of Activated Carbon/Alginate Composite Beads for the Removal of 2-Methylisoborneol from Aqueous Solution. AppliedChem, 5(4), 32. https://doi.org/10.3390/appliedchem5040032






